Abstract
Background
Studies examining the effects of therapeutic interventions after stroke often focus on changes in loss of body function/structure (impairment). However, improvements in activities limitations and participation restriction are often higher patient priorities, and the relationship that these measures have with loss of body function/structure is unclear.
Objective
This study measured gains across WHO International Classification of Function (ICF) dimensions and examined their interrelationships.
Methods
Subjects were recruited 11 to 26 weeks after hemiparetic stroke. Over a 3-week period, subjects received 12 sessions of intensive robot-based therapy targeting the distal arm. Each subject was assessed at baseline and at 1 month after end of therapy.
Results
At baseline, subjects (n = 40) were 134.7 ± 32.4 (mean ± SD) days poststroke and had moderate-severe arm motor deficits (arm motor Fugl-Meyer score of 35.6 ± 14.4) that were stable. Subjects averaged 2579 thumb movements and 1298 wrist movements per treatment session. After robot therapy, there was significant improvement in measures of body function/structure (Fugl-Meyer score) and activity limitations (Action Research Arm Test, Barthel Index, and Stroke Impact Scale–Hand), but not participation restriction (Stroke Specific Quality of Life Scale). Furthermore, while the degree of improvement in loss of body function/structure was correlated with improvement in activity limitations, neither improvement in loss of body function/structure nor improvement in activity limitations was correlated with change in participation restriction.
Conclusions
After a 3-week course of robotic therapy, there was improvement in body function/structure and activity limitations but no reduction in participation restriction.
Introduction
Stroke remains a leading cause of long-term disability worldwide. 1 Deficits in function of the upper extremity represent a significant contributor to decreased function and quality of life poststroke. 2 Interventions such as constraint-induced movement therapy 3 and robot-assisted therapy4,5 have emerged as promising approaches to promote return of function beyond that which is regained during spontaneous recovery and from conventional rehabilitation. However, the degree to which specific interventions affect disability and improve function after stroke is still poorly understood.6,7
Numerous outcome measures are available that assess the effect of stroke across all dimensions of the World Health Organization (WHO) International Classification of Function, Disability, and Health (ICF),8,9 complicating selection of appropriate measurement tools when assessing novel rehabilitation-related interventions.10,11 Assessments of stroke-related
In general, a limited relationship has been found across WHO ICF dimensions such as loss of function/structure and activity,12,15,16 and data are conflicting regarding the degree of correlation that exists. For example, a meta-analysis of electromechanical and robot-assisted arm training described high-quality evidence that such interventions improve outcomes in both function/structure and activity, but there was limited evaluation of effects on participation. 17 The Fugl-Meyer (FM) motor scale,18,19 a commonly employed measure of function/structure, was shown to demonstrate robust correlation with the Action Research Arm Test (ARAT), a measure of activity, when performed by expert raters. 19 On the other hand, studies of constraint-induced movement therapy report discrepancies between treatment-related changes in function/structure when compared with activity. 20 Another report found that a majority of patients demonstrating no measurable upper extremity motor impairment in an assessment of function/structure continued to report deficits as measured by assessments of activity and participation. 21 This inconsistent relationship may be due to numerous factors having greater influence on outcome as one moves from measurements of function/structure to activity or participation. 16 For example, multiple studies have shown improvements in patients’ functional independence measure (FIM) scores (FIM is a measure of activity) over the course of inpatient rehabilitation despite minimal change in FM total score 22 or National Institutes of Health Stroke Scale (NIHSS) 23 (both assessments of function/structure), potentially related to use of assistive devices or learning compensatory skills.
In the setting of a clinical trial on intensive robot-assisted therapy, the current study had two primary aims. First, the study aimed to provide a detailed assessment of the effect of robot-assisted therapy across all ICF dimensions. Second, the study also aimed to assess the degree to which therapy-related improvements in function/structure were related to improvements in activity and participation. We hypothesized that robot-assisted therapy would result in clinically and statistically significant gains in function/structure with smaller gains in activity and participation.
Methods
Subject Enrollment
Forty-one individuals 11 to 26 weeks after stroke onset gave informed consent to participate in a longitudinal study of standardized intensive robot-assisted therapy targeting the distal arm (clinicaltrials.gov, ID NCT01244243). In one individual, baseline imaging revealed an incidental finding that met exclusion criteria. Therefore, results represent the remaining 40 eligible subjects. Neuroimaging data from a subset of these subjects have previously been reported. 24 Study procedures were approved by the University of California, Irvine Institutional Review Board.
Inclusion and exclusion criteria were designed to select for individuals with a wide range of motor deficits in whom spontaneous arm motor recovery had reached a plateau. All subjects met the following inclusion criteria: (1) age ≥18 years; (2) hemorrhagic or ischemic stroke with onset 11 to 26 weeks prior to enrollment; (3) residual arm motor deficits, defined as ARAT score <52 or 9-hole peg test score >25% longer in the affected compared with unaffected hand; and (4) preservation of voluntary movement in the distal upper extremity, as demonstrated by ≥5° of active range of motion in the wrist or index finger metacarpophalangeal joint of the affected side. Individuals were excluded from the study if they had a contraindication to MRI, severe cognitive impairment (Mini Mental Status Examination [MMSE] score <27), a comorbid diagnosis impacting the function of the affected upper extremity, or unstable arm motor status. Stable arm motor status was defined as change in FM score ≤2 points across 2 successive baseline assessments that spanned ≥1 week prior to initiation of robot therapy. This was a single treatment-arm study, with no comparison or placebo group.
Robot Therapy
Robot-assisted therapy consisted of 12 treatment sessions across 3 weeks. Sessions occurred 4 days per week for 2 hours per day, totaling 24 total hours of delivered therapy. All subjects completed at least 11 of 12 prescribed robot therapy sessions. This robot has been previously described 5 (Figure 1). In brief, the robotic device has 3 degrees of freedom, is pneumatically actuated, and is back-driveable. The 3 degrees are rotational movement of the wrist, thumb, and fingers in the plane of gravity, with the fingers moving as a single unit about the metacarpophalangeal joint. Specifically, the device assists in power grip and release movements in a pattern that combines wrist extension with hand grasp, then wrist flexion with hand release. Robot-assisted joint movement is achieved by a lever design, in which a pneumatic cylinder is mounted at the opposite end of a lever from the limb interface, with a revolute joint in between. Back-driveability of the robot permits the subject to freely drive movements when active assistance is not engaged.
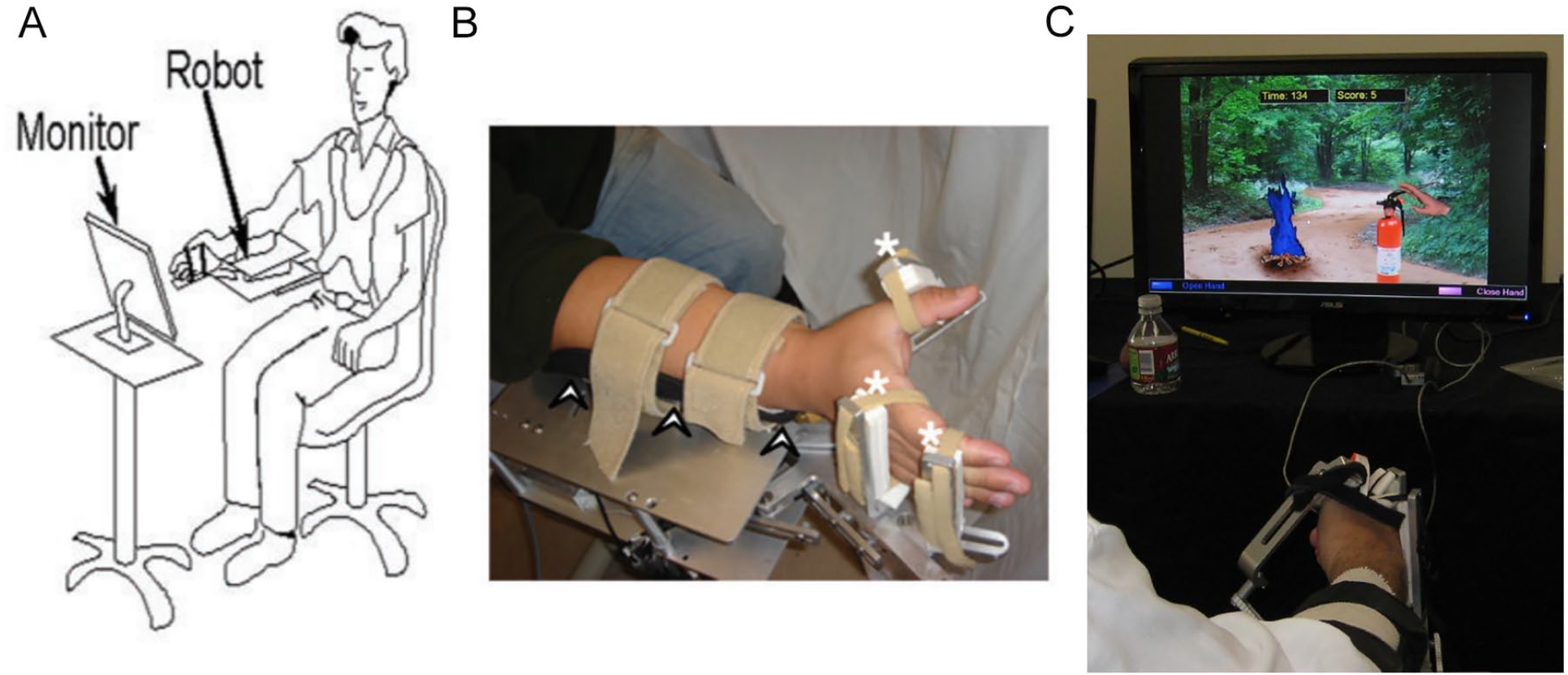
(A) Diagram of subject’s posture, relationship to robot, and relationship to computer monitor during therapy. (B) Demonstration of subject’s hand and arm interfacing with the robot. Arrowheads denote ulnar forearm rest. White asterisks indicate soft Velcro straps connecting the subject’s hand to the robot. (C) Example of virtual reality game, in which hand grasp turned on fire extinguisher while grasp release turned off fire extinguisher during Smokey the Bear game. Figure partially reprinted with permission from previously published material. 5
At the start of each session, a trained therapist (licensed occupational therapist or physical therapist) assisted in robot adjustments to accommodate differences in hand sizes across subjects and to ensure maximal biomechanical advantages in robot-assisted movements. The therapist also determined the comfortable passive and active range of motion at each of the three degrees, for each subject, and adjusted the robot’s hard stops accordingly. Movement repetitions were calculated from each subject’s pretreatment range of motion, such that a movement cycle was counted when measured active movement exceeded a preset threshold of the baseline active range of motion at a specified joint.
Each robot-assisted therapy session began with robot-assisted passive stretching, then grasp-release exercises. A wide range of real objects was placed in the hand, and the subject was asked to describe features such as texture or temperature. Next, and occupying most of the session were repeated grasp-release movements of the affected hand coupled to video games (eg, squeezed mustard onto a hot dog consumed by Homer Simpson, or hand closure in the robot squeezed a fire extinguisher to douse a forest fire, with approval of Smokey the Bear, Figure 1C). These games emphasized control of hand movement range, speed, and timing. For each movement, the subject-initiated hand grasp, and if the subject did not complete the full movement, the pneumatic cylinder was activated to assist in completing hand grasp. In most games, movement was initiated in response to a simple computer-generated cue; in others, movement onset time was self-initiated, in an attempt to activate supplementary motor area 25 ; and in others, choice of movement was guided by cues that corresponded to regularly changing rules displayed on the screen, in an attempt to activate dorsal premotor cortex. 26 The therapist was at the patient’s side at all times to monitor and adjust robot-subject interface, clarify gameplay, and address any questions that arose. In total, the robot recorded an average of 11 278 finger movements, 28 970 thumb movements, and 15 759 wrist movements across the 3 weeks of therapy; average movement repetitions per day were 954 for finger movements, 2579 for thumb movements, and 1298 for wrist movements. For some games, hand grasp required movement of the fingers and thumb only, while other games required movement of the wrist and thumb. As a result, the number of movement repetitions was greatest for the thumb.
Study Design
Clinical assessments were performed at 3 time points: (1) ≥2 weeks prior to therapy (first baseline visit), (2) ≥1 week prior to therapy and at least 7 days after first baseline visit (second baseline visit), and (3) 1 month following completion of therapy. Performance of 2 baseline assessments prior to initiation of therapy was done in order to ensure subjects had reached a stable plateau in motor recovery of the affected distal upper extremity.
Clinical Outcome Measures
All clinical assessments were performed by 1 of 2 physical therapists blinded to the subjects’ performance during therapy. The primary outcome measures were the Upper Extremity FM Assessment 18 and the ARAT, 27 which have good reliability, reproducibility, and responsiveness to therapy. 19
Assessments were categorized per the ICF dimensions of stroke recovery8,28 as defined by the RehabMeasures Database at the Shirley Ryan AbilityLab (https://www.sralab.org/rehabilitation-measures). Measures of
Statistical Analysis
For FM, ARAT, and Box and Blocks assessments, evaluation of robot therapy–related improvement was calculated with regards to the mean of the 2 baseline assessments. For all other measures, therapy-related improvement was calculated with respect to scores at the first baseline visit.
Paired testing was used to determine the statistical significance of therapy-related improvement. Measures that were normally distributed or could be transformed to a normal distribution were evaluated using the paired
Results
Subject Characteristics
Demographics for the 40 subjects are summarized in Table 1. Subjects were mainly right-hand dominant. Across the 40 subjects, the index infarct was in the left hemisphere in 21 and in the right hemisphere in 19. Ten individuals had more than 1 infarct. There was significant variability in size and location of infarcts (33.1 ± 50.0 cm3, mean ± SD). Mean time between the 2 baseline assessments was 15.6 ± 6.0 days (range 7-30 days). FM, ARAT, and Box and Blocks Test scores did not change across the 2 baseline assessments (
Subject demographics.

Clinical Assessments at Baseline and Posttreatment.
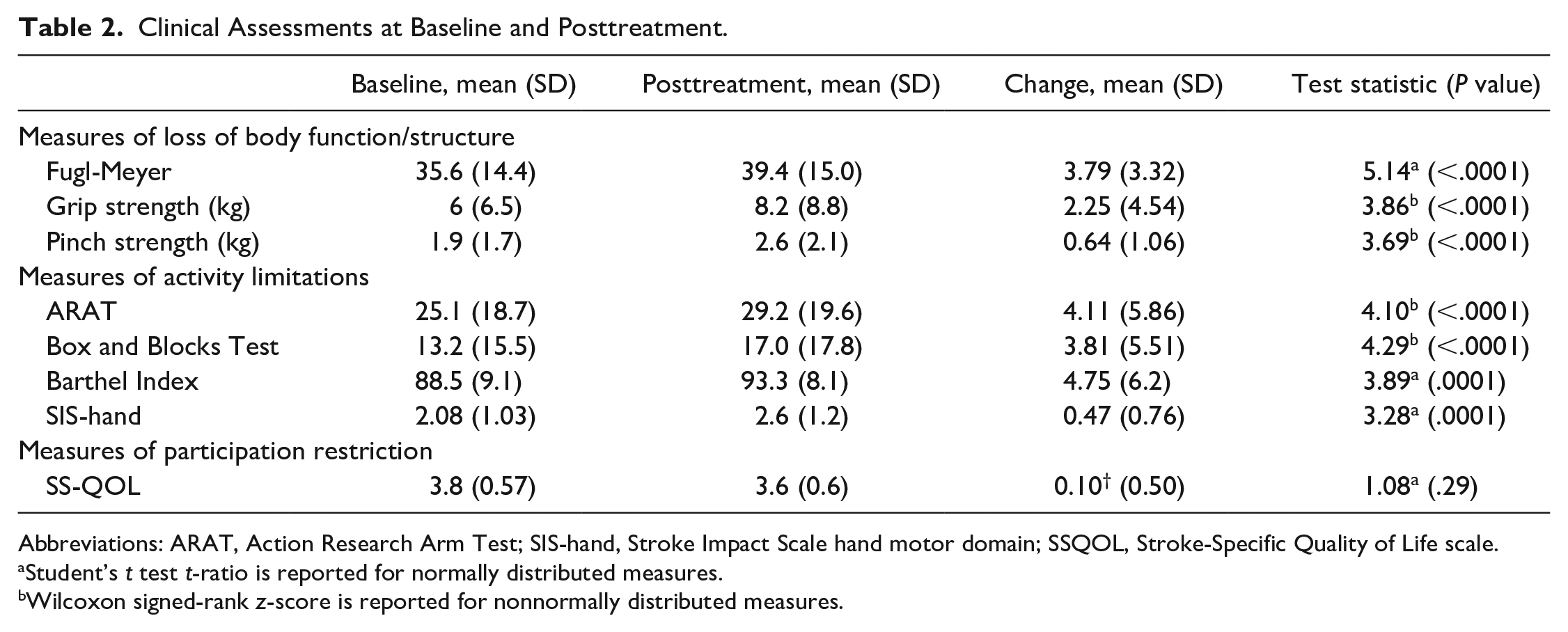
Abbreviations: ARAT, Action Research Arm Test; SIS-hand, Stroke Impact Scale hand motor domain; SSQOL, Stroke-Specific Quality of Life scale.
Student’s
Wilcoxon signed-rank
Outcomes After Robot Therapy
Subjects demonstrated statistically significant improvement in all measures of function/structure and activity after robot therapy, from baseline to the posttherapy assessment (Table 2). The group did not show statistically significant change in SS-QOL, the only measure of participation. Across all measures, the largest effect sizes were noted for improvement in FM (
Correlation Across ICF Dimensions
Correlations for improvements in measures both within and across ICF dimensions are detailed in Table 3. Briefly, modest correlations were demonstrated between measures within each ICF dimension. However, the strongest correlations were found between measures of function/structure and activity. For example, improvements in FM were robustly correlated with improvements in ARAT (Figure 2A,
Correlations Between Clinical Measures.
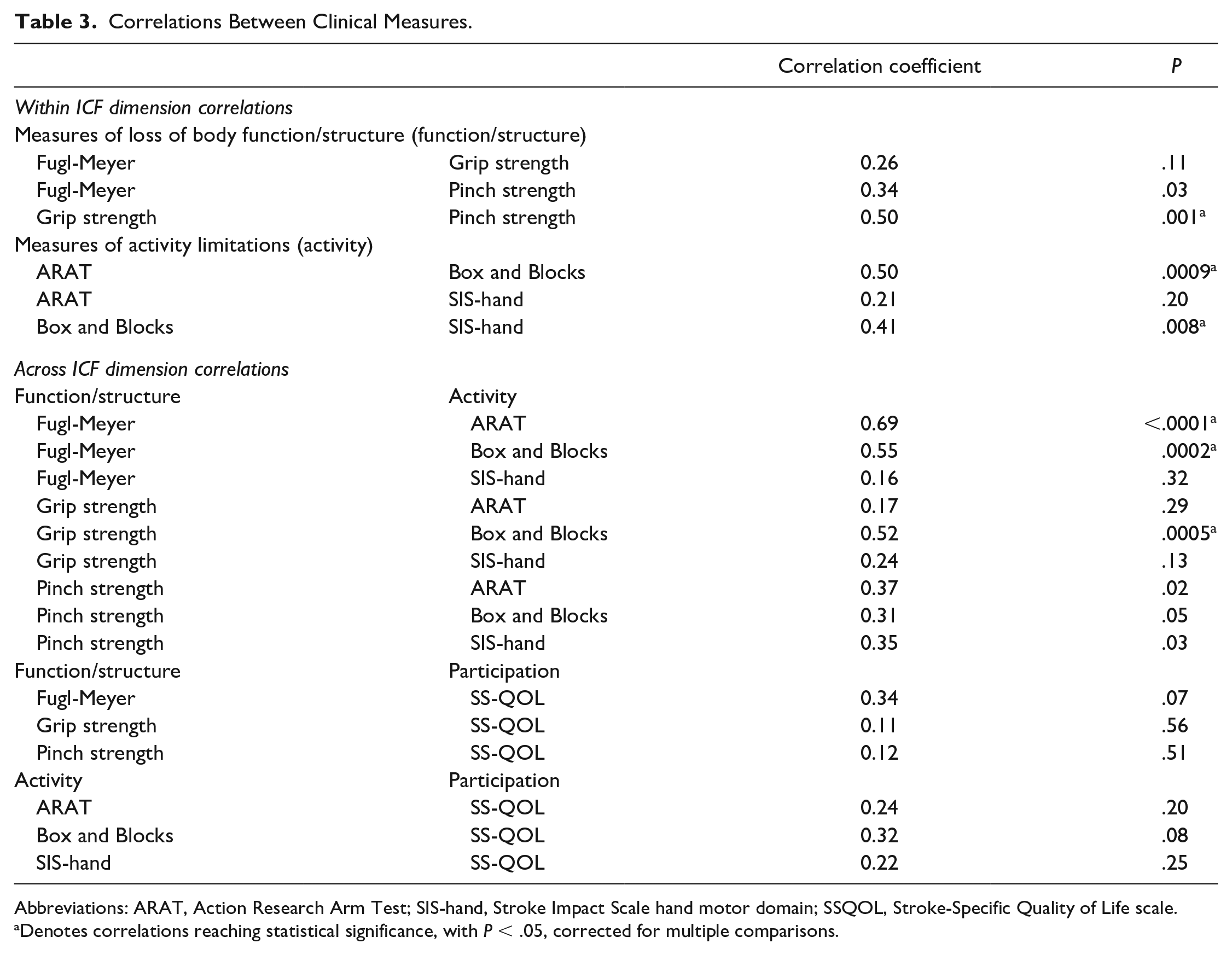
Abbreviations: ARAT, Action Research Arm Test; SIS-hand, Stroke Impact Scale hand motor domain; SSQOL, Stroke-Specific Quality of Life scale.
Denotes correlations reaching statistical significance, with
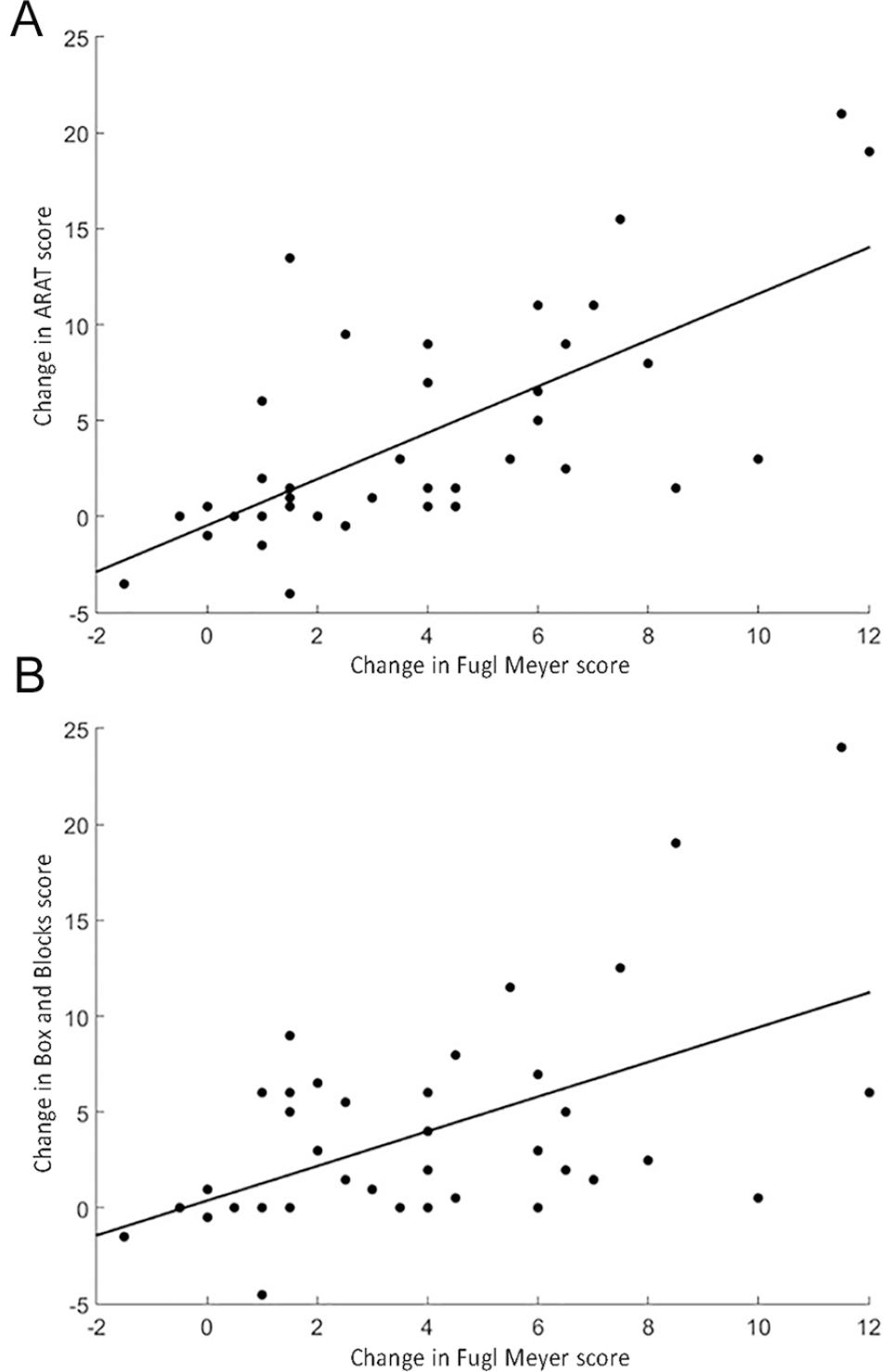
Scatterplot demonstrating the relationship between change in Upper Extremity Fugl-Meyer Assessment score and change in (A) Action Research Arm Test (
Changes in SS-QOL were not correlated with improvements in any measure of function/structure or activity, including FM, ARAT, and SIS-hand.
Discussion
Stroke-related deficits in upper extremity function remain a significant contributor to disability worldwide, with persistent deficits directly linked to activity limitations, participation restriction, poorer quality of life, and decreased subjective well-being.1,21,33 Novel interventions, including robot-assisted therapy, have been developed to reduce stroke-related disability. However, available data provide a limited understanding of the degree to which such interventions produce changes across ICF dimensions. Furthermore, there have been limited studies that directly examine the degree to which treatment-related gains in one ICF dimension are associated with gains across other ICF dimensions.
The current study found that subjects who completed a course of robot-based therapy demonstrated statistically significant improvements in stroke-related loss of body function/structure and activity limitations, but not in participation restriction. Specifically, robot-assisted therapy was associated with statistically significant gains across all measures of function/structure and activity, including both primary endpoints: FM (a measure of function/structure) and ARAT (a measure of activity). Estimates of the minimal clinically important difference (MCID) for the FM and ARAT have been estimated to be 10% of the scale maximum,18,34 or 6.6 and 5.7 points, respectively. Using this estimate, 17.5% of the study patients achieved MCID on the FM, and 32.5% on the ARAT, after robot-assisted therapy. Lo et al 4 have suggested that for some subjects with chronic stroke, gains below MCID might be clinically meaningful. SIS-hand, which is a patient-reported measure of activity, also showed a statistically significant improvement after therapy, though effect size was notably smaller. Improvements in measures of function/structure were robustly correlated with measures of activity, consistent with prior studies that found measures of function/structure to be significantly correlated with measures of activity, including Barthel Index, FIM, and SIS-16.35,36 Together, the current results provide an in-depth examination of the degree to which clinical improvements after robot-based therapy extend across ICF dimensions.
Current findings are similar to previously published large-scale clinical trials in which an experimental intervention produced significant gains in measures of function/structure and activity, but not in measures of participation.3,37,38 This difficulty to generate changes in participation likely reflects both the multitude of factors that contribute to participation in society and the complex interaction of such factors. 1 Previous studies have identified demographic variables, socioeconomic status, injury characteristics, medical and psychiatric comorbidities, social support, and functional status as contributors to deficits in poststroke participation.2,39 Given that participation can be influenced by so many variables, including those related to both the function/structure and activity dimensions, 40 it may be that detection of meaningful improvements in participation requires a longer time period in order to develop and thus an extended follow-up period, consistent with the observation from longitudinal studies that quality of life after stroke can take months to years to manifest. 41 In line with this, Wolf et al 42 found initial changes in SIS functional domains were only followed by improvements in SIS participation domains after a period of 12 to 24 months beyond when the intervention was completed.
The high number and density of movement repetitions provided by the robot represent several strengths of the current study. First, standardization of treatment by the robot minimizes variability in conventional rehabilitation interventions, which could confound the relationship between improvements after robot-assisted therapy across ICF dimensions. 43 Second, robot-based interventions often deliver a higher intensity of movement repetitions compared with conventional interventions.43,44 In one study, conventional therapist-administered interventions provided an average of 32 repetitions/session, 45 which is too few movements to induce neural plasticity 46 and an impediment to improving behavior, particularly in higher order ICF dimensions, including participation. 47 In contrast, patients in the current study averaged 2579 thumb movements and 1298 wrist movements per treatment session, exceeding the 600 to 700 repetitions per day 48 in rats and the 924 49 movements per day in primates considered necessary to realize functional benefits with stroke rehabilitation. Third, each hand movement in the robot was coupled to video games. Games promote patient involvement in health care50,51 and motivate patients to engage in enjoyable play behavior that involves therapeutically relevant movements.52,53 Use of games alters cognitive context 54 and, compared with rote movement repetition, increases activity in cognitive networks in patients with stroke. 55
After robot-assisted therapy, improvements in function/structure and activity were not associated with improvements in participation. Recently published results from 2 large-scale clinical trials also reported a dissociation between treatment-related motor gains and change in measures of participation.16,38 Lang et al 38 found that while the majority of subjects reported overall perception of meaningful change with treatment, as a group they did not demonstrate statistically significant improvement in measures of participation. A cross-sectional study 31 comparing SIS with SS-QOL also reported significant dissociation, with SIS subscores being more responsive to treatment as compared to SS-QOL subscores. On the other hand, Roth and Lovell 56 reported a correlation between FIM scores >80 at 1 year poststroke and increased community/home participation as defined by the Frenchay Activities Index. Together, these results highlight an ongoing need for robust, responsive, and specific measures of participation, particularly when evaluating the effect of novel interventions for reducing stroke disability. Strikingly, in a review of the 116 instruments on www.rehabmeasures.org 28 designated for stroke, only 15 instruments were strictly participation measures. The overwhelming majority of those instruments assess only 3 or 4 items to characterize overall quality of life, and thus provide coarse gradation across patients. Under-scoring the limited attention to participation in studies of stroke rehabilitation therapies, a report by Salter et al 57 showed only 25% of randomized controlled trials of stroke rehabilitation in the past 4 decades included an assessment of participation. 57 These prior studies indicate the ongoing interest and the paucity of knowledge regarding the translation between improvements in function/structure and participation, changes that are more clinically meaningful to patients.
Several limitations are associated with the current study. First, subjects were studied 11 to 26 weeks poststroke. As such, it is unknown the degree to which the current findings can be extended to patients who initiate therapy at an earlier or later time poststroke. In addition, while the literature reports 95% of patients with upper extremity deficits reach recovery plateau by 11 weeks poststroke, 58 the improvements demonstrated after robot-assisted therapy (Table 2) may not be entirely attributable to the robotic intervention and instead may be confounded by a degree of natural recovery. Any contributions of natural recovery to the current results are likely attenuated in this study, however, as serial baseline assessments indicated that subjects had reached stable arm motor status at the time of enrollment. The primary endpoint of the study was at 1 month posttherapy, which was an additional study limitation. It may be that any long-term changes in participation and their relationship over time 59 with structure/function and activities, were not captured as a result of this 1-month focus. Finally, reflecting the paucity of meaningful participation measures reported in the stroke literature, the SS-QOL was the only participation measure used in the current study. Although other measures of participation are available, the SS-QOL has been validated in stroke patients in multiple studies60,61 and includes measures of quality of life as they relate to upper extremity function, making it especially suited for the current study.
Though the generalizability of these results to robot-assisted therapy beyond the upper extremity is yet unknown, studies showing correlations between function/structure of the lower extremity 62 and activity using the Barthel Index,35,62 SIS-16, 36 and FIM22,36 suggest that further studies in other key components of movement are warranted and may show similar relationships.
Conclusions
Novel interventions to reduce stroke disability are in development. 63 The current results demonstrate robot-assisted therapy, specifically, supports improvements in stroke-related loss of body function/structure, as well as activity limitations. The results also underscore concerns regarding the incomplete relationships between therapy-related improvement in function/structure and activity (often the primary endpoints in clinical trials), and improvements in participation, which are often prioritized by patients. 1 Ultimately, this study highlights the need 64 for further development of instruments that provide accurate, specific, reliable, reproducible, and granulated assessments of participation restriction after stroke.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Cramer serves as a consultant for Abbvie, Constant Therapeutics, MicroTransponder, Neurolutions, SanBio, Fujifilm Toyama Chemical Co, and TRCare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Author has received funding from National Institute of Neurological Disorders and Stroke, R01 NS059909 (Grant id) and National Center for Advancing Translational Sciences, K24 HD074722 (Grant id).
