Abstract
Background
Poststroke depression (PSD) is related to adverse functional and cognitive prognosis in stroke patients. The participation of kynurenine pathway metabolites in depression has been previously proposed; however, there are few studies on its role in PSD and disability in stroke.
Objective
To investigate if there is a correlation between serum kynurenines levels with poststroke anxiety and depression symptoms and disability scales.
Methods
A cross-sectional case-control study was conducted in patients with first stroke, of >1 month and <1 year of evolution, with no history of previous psychiatric or neurological disorders; the Hospital Anxiety and Depression Scale (HADS), Montreal Cognitive Assessment (MoCA), functional evaluations (Barthel index, Functional Independence Measure [FIM]) were applied and serum kynurenines (Kyns) were determined.
Results
Sixty patients were included; significant depressive symptoms were found in 63% of the cases; a significant and positive correlation was obtained between levels of 3-hydroxykynurenine (3-HK) with HADS-T (r = 0.30, P = .025) and HADS-D (r = 0.28, P = .039). Depressed patients showed significantly higher levels of 3HK (P = .048) and KYNA (P = .0271) than nondepressed patients; the 3HK levels were inversely correlated with functional scales: Barthel index (r = −0.31, P = .02), FIM (r = −0.40, P = .01); in addition, serum 3HK levels were significantly higher in patients with poor sleep quality (P = .0190).
Conclusions
Serum Kyns show correlation with the presence and severity of depressive symptoms and with the disability and sleep quality. Kyns may be a potential marker of depression risk and disability in stroke in future.
Introduction
Stroke not only continues to be one of the main causes of mortality and morbidity worldwide but also represents the first cause of global disability in the adult population.1,2 Poststroke depression (PSD) is a complication observed in 30% to 50% of patients and its importance lies in the fact that its presence is related to an increased risk of morbidity and mortality, as well as poor functional and cognitive prognosis.3-5 Several risk factors involved in the development of PSD have been reported: previous depression, higher level of disability, severity of stroke, female sex, age >70 years, low socioeconomic level, and so on.6,7
The pathophysiology of PSD is complex and has been proposed to involve both biological factors or factors related to brain damage (site of injury, extension of the lesion, neurochemical alterations, persistent inflammation, etc.) and environmental factors (level of disability, socioeconomic status, etc).
8
In this sense, the participation of the metabolites of tryptophan and especially those belonging to the kynurenine pathway (KP) in the origin of the depression has recently been proposed.9,10 KP is the main route of catabolism of tryptophan in the central nervous system (CNS) and owes its importance to the fact that some of its metabolites have shown neuroactive properties. For example, quinolinic acid (QUIN) has shown to activate N-methyl-
Methods
The present study was carried out in strict adherence to the Declaration of Helsinki (1964) and to the current national sanitary regulations in the field of human research and was also evaluated and authorized by the research and bioethics committee of the Instituto Nacional de Rehabilitación LGII. All patients authorized their participation by signing an informed consent.
Selection of Patients
Adult patients (>18 years) was recruited of any sex that met the following inclusion criteria: (1) first event of ischemic or hemorrhagic stroke corroborated by neuroimaging study, (2) no history of previous neurological or psychiatric diseases, (3) evolution time of the stroke of >1 month, but <1 year, (4) that could reliably answer the evaluations of the mood and cognition; patients with severe aphasia were excluded or patients with acute complications that prevent their objective assessment (eg, delirium, electrolyte imbalance, etc), and (5) no current consumption of antidepressants, drug abuse, immunosuppressants, immunomodulators, or other drugs that may affect their cognitive performance or their mood status.
Clinical, Functional, Cognitive, and Mood Assessments
In a single interview the following evaluations were carried out, a questionnaire of demographic and clinical variables: age, sex, occupation, marital status, socioeconomic status, stroke evolution time, type of neurological sequelae and site of neuroanatomical lesion, and so on. Functional evaluations: Barthel index and Functional Independence Measure (FIM). For cognitive evaluation the Montreal Cognitive Assessment (MoCA) was applied; to assess the sleep quality, the Pittsburgh Sleep Quality Index (PSQI) was applied; and for the evaluation of symptoms of anxiety and depression, the Hospital Anxiety and Depression Scale (HADS) was used; which is the most widely used and supported screening instrument for the evaluation of anxiety and depression symptoms in patients with stroke, 5 this instrument can be scored as a total score (HADS-T) or for each of its subcomponents: anxiety (HADS-A) or depression (HADS-D).
Measurement of Serum Kynurenines
At the end of the clinical evaluations, a sample of peripheral venous blood was taken from the antecubital vein of the nondominant arm, and 10 cm3 of blood was obtained in a tube without anticoagulant, after allowing the sample to coagulate is proceeded to the centrifugation process at 2500 rpm for 20 minutes to then separate the serum and store it at −70 °C until further processing. All blood samples were taken between 9:00 and 13:00 hours in the morning.
Tryptophan (Trp), L-kynurenine (L-Kyn), and KYNA levels were measured by an HPLC (high-performance liquid chromatography) method with fluorescence detection. 18 Briefly, 20 μL of serum supernatant sample were injected onto an Eclipse XDB-C18 reverse phase column (5-μm, 4.6 × 150 mm), and isocratically eluted with a mobile phase consisting of 50 mM sodium acetate, 250 mM of zinc acetate, and 3% of acetonitrile, pH adjusted to 6.2 with glacial acetic acid, at a flow rate of 1 mL/min; L-Kyn was eluted with the same mobile phase but without acetonitrile. KYNA was detected at excitation wavelength: 344 nm, emission wavelength: 398 nm, and L-Kyn at excitation wavelength: 368 nm, emission wavelength: 480 nm. The retention time for KYNA was ~7 minutes and for L-Kyn it was ~8 minutes. Trp levels were determined using a ZORBAX Eclipse AAA column (3.5-μm, 4.6 × 150 mm), and isocratically eluted with a mobile phase containing 100 mM of zinc acetate and 3% of acetonitrile (pH adjusted to 4.2) at a flow rate of 1 mL/min. Trp was detected at excitation wavelength: 254 nm, emission wavelength: 404 nm. The retention time of Trp was ~5 minutes.
The 3HK levels were determined using an electrochemical method. 18 Briefly, 40 μL of sample were injected onto a Adsorbosphere Catecholamine C18 reverse phase column (3 μm, 4.6 mm × 100 mm) and eluted at a constant flow rate of 0.6 mL/min with a mobile phase containing 9% of triethylamine, 0.59% phosphoric acid, 0.27 mM ethylenediaminetetraacetic acid (EDTA), and 8.9 mM heptane sulfonic acid. The retention time was ~11 minutes.
Statistic Analysis
In first place, descriptive statistics were used to summarize the characteristics of the sample, then a normality test of the different quantitative variables was performed using the Shapiro-Wilk test, later Spearman or Pearson correlation tests were applied between quantitative variables (functional scales and mood assessment vs serum kynurenines), to finally search significant differences between groups of depressed and nondepressed patients using the Mann-Whitney test. For statistical analysis, the GraphPad Prism version 6.0 software was used. To establish significant differences and correlations, a P value less than or equal to .05 was considered significant.
Results
A total of 90 patients were recruited; however, 30 patients were eliminated from the study because they did not meet all the inclusion criteria or the blood sample that was obtained was insufficient to perform its analysis correctly. The final sample of patients included 60 patients: 27 women and 33 men, the mean age was 57.3 years (SD ±14 years), the mean evolution time since stroke was 5.2 months (SD ±3.5 months), on the type of stroke: 82% (n = 49) were ischemic and 18% (n = 11) were hemorrhagic, on the side of brain lesion: 55% had right and 45% left injuries; the average number of comorbidities was 2.6 for each patient with a range of 1 to 6 comorbidities, the most frequent being hypertension (66%), dyslipidemia (57%), and diabetes mellitus (42%); body mass index mean was 26.28 kg/m2 (SD ±3.6 kg/m2); the average years of education was 10.2 years (SD ±5.3 years) and the mean of socioeconomic level was 2.3 (SD ±1.0) range from 0 to 6 (considering that 6 is the highest socioeconomic level and 0 the lowest).
Using the suggested cutoff points reported in the literature for patients with stroke of the HADS-A and HADS-D score (>6 points), 19 it was observed that 63% (n = 38) of the patients showed significant depressive symptoms, while 53% (n = 32) of the patients showed significant anxiety symptoms, and 45% (n = 27) had significant symptoms of both; no difference was observed in the presentation of significant depressive or anxious symptoms in relation to sex. Considering the cutoff point of the PSQI scale (>5 points), 50% (n = 30) of the patients showed poor sleep quality. Regarding the cognitive evaluation, 68% (n = 41) of the patients showed some degree of cognitive impairment (MoCA <24 points, as has been suggested in the Mexican population). 20 When performing a comparative analysis of basal features between depressed versus nondepressed patients, the only differences observed were in years of education and PSQI score (Table 1).
Main Features and Results of Evaluations of Total Sample and Comparison Between Depressed and Nondepressed Patients.
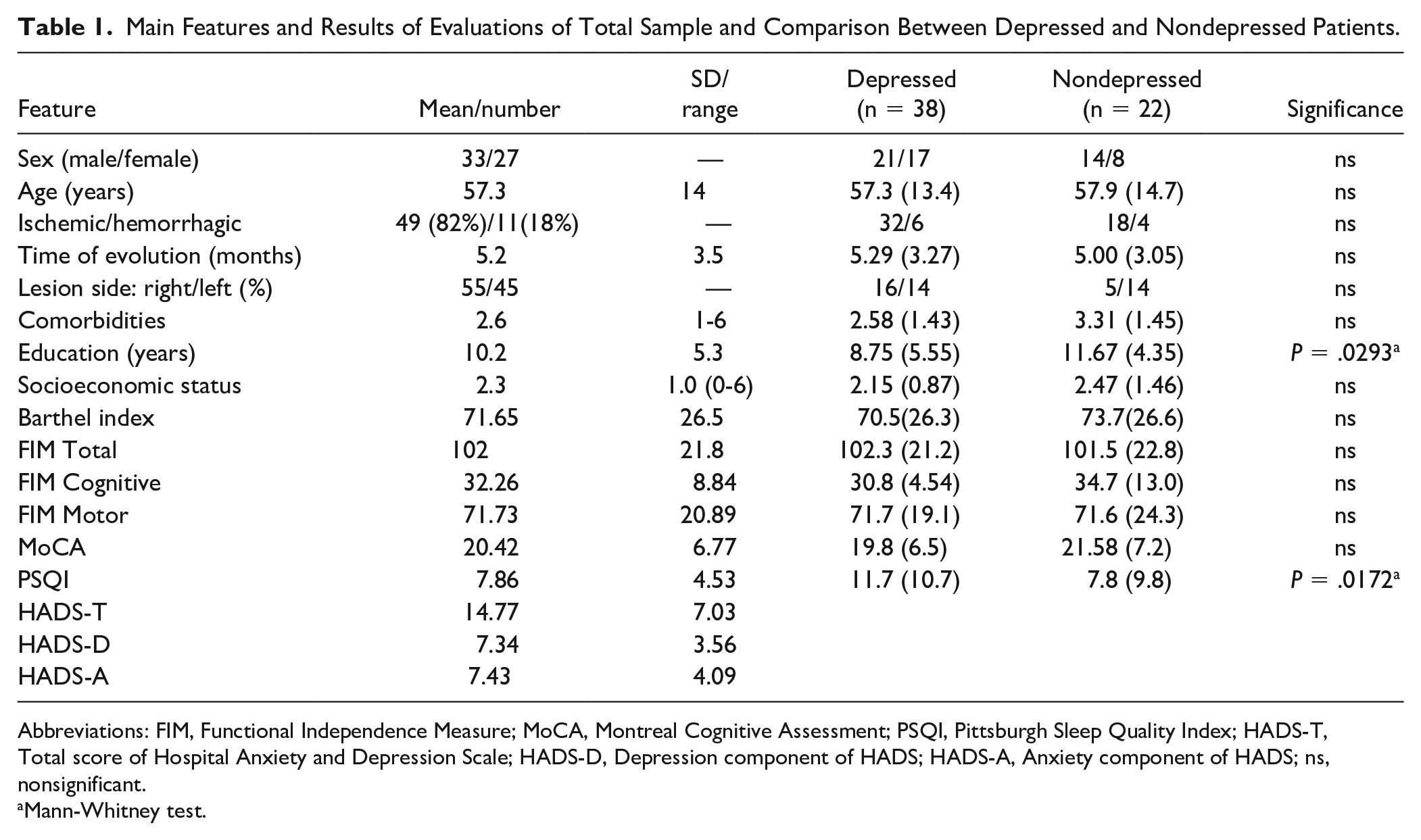
Abbreviations: FIM, Functional Independence Measure; MoCA, Montreal Cognitive Assessment; PSQI, Pittsburgh Sleep Quality Index; HADS-T, Total score of Hospital Anxiety and Depression Scale; HADS-D, Depression component of HADS; HADS-A, Anxiety component of HADS; ns, nonsignificant.
Mann-Whitney test.
When the correlations between the scores of the HADS with the serum levels of the different kynurenines (L-Kyn, KYNA, QUIN, and KYNA) and tryptophan was made, only a significant positive correlation was found between the 3HK levels with the HADS-T (r = 0.30; P = .025) and the HADS- D (r = 0.28, P = .039). Performing a correlation analysis between the kynurenine levels and the different functional evaluations, also a significant and inverse correlation was observed between the serum levels of 3HK and the Barthel index (r = −0.2795, P = .0042) and the total FIM (r = −0.4721, P = .0004). When performing a correlation or association analysis between the subscales of the HADS with the demographic variables and the functional, cognitive, or sleep scales (age, sex, socioeconomic status, Barthel index, FIM, MoCA, PSQI) only a significant correlation of the PSQI was obtained with the 3 subscales of the HADS (Pearson test): HADS-T (r = 0.5789, P < .0001); HADS-D (r = 0.4378, P = .0023); HADS-A (r = 0.6030, P < .0001) (Figure 1).
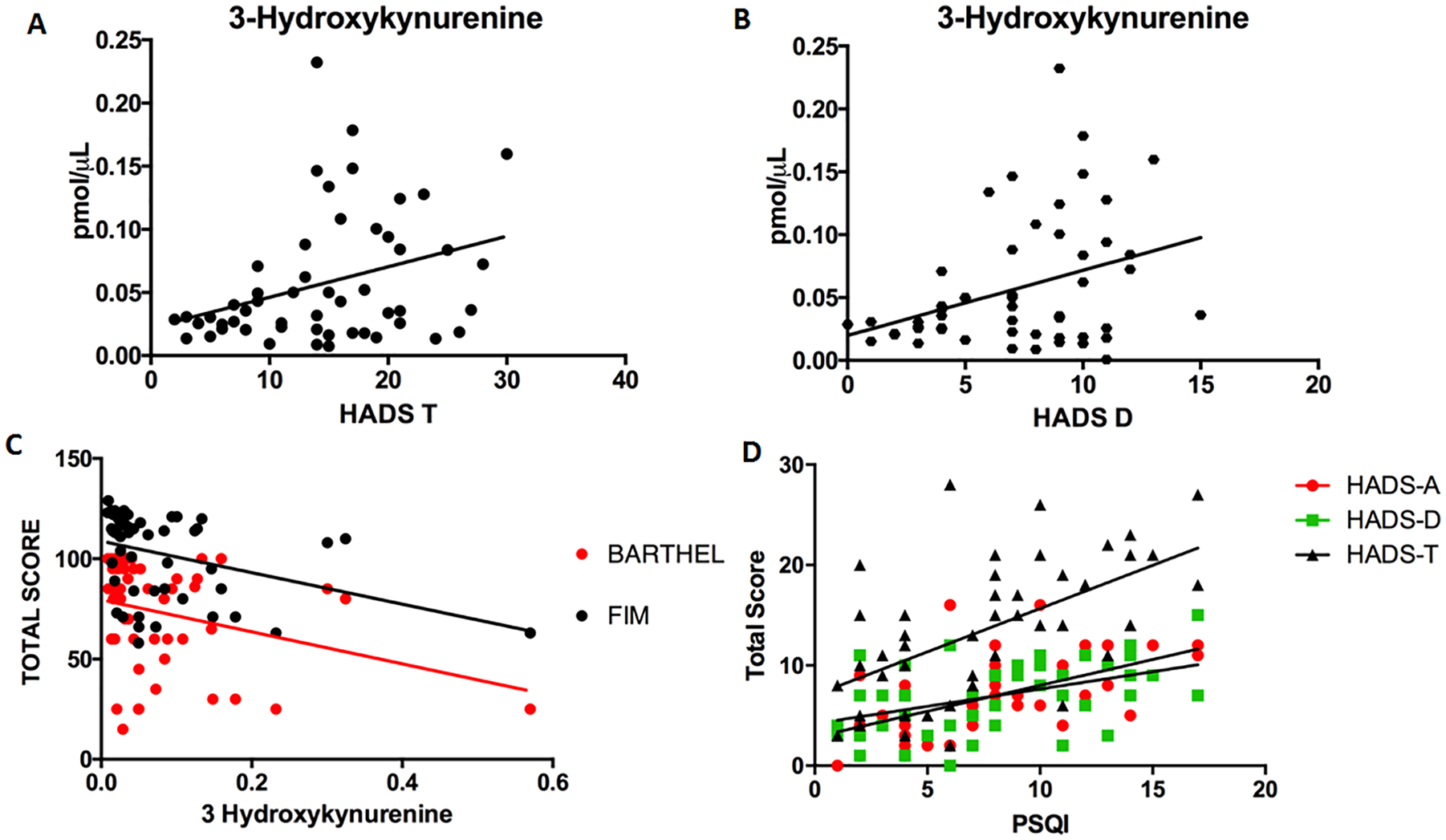
Correlation plots between the 3HK serum levels and HADS scale in the entire patient sample. A significant but slight positive correlation (Spearman test) was observed with the HADS-T (A) (r = 0.30, P = .025) and HADS-D (B) (r = 0.28, P = .039). (C) Correlation plot between 3HK serum levels and functional assessments. A significant but slight inverse correlation was found between the 3HK serum levels with the Barthel index (Spearman test, r = −0.2795, P = .0421) and with the FIM scale (Spearman test, r = −0.4721, P = .0004). (D) Correlation plot between PSQI and HADS subscales. Significant correlation of the PSQI was obtained with the 3 subscales of the HADS (Pearson test): HADS-T (r = 0.5789, P < .0001); HADS-D (r = 0.4378, P = .0023); HADS-A (r = 0.6030, P < .0001). 3HK, 3-hydroxykynurenine; HADS-T, Total score of Hospital Anxiety and Depression Scale; HADS-D, Depression component of HADS; HADS-A, Anxiety component of HADS; FIM, Functional Independence Measure; PSQI, Pittsburgh Sleep Quality Index.
When dividing the sample in patients with or without significant depressive symptoms, it was also observed that depressed patients showed significantly higher levels of 3HK (Mann-Whitney test P = .048) and KYNA (Mann-Whitney test P = .0271) than nondepressed patients. Finally, when dividing the sample in patients with good or poor sleep quality (PSQI >6 points), a significant increase in serum levels of 3HK was also observed in poor sleep quality group (unpaired T test, P = .0190) (Figure 2).
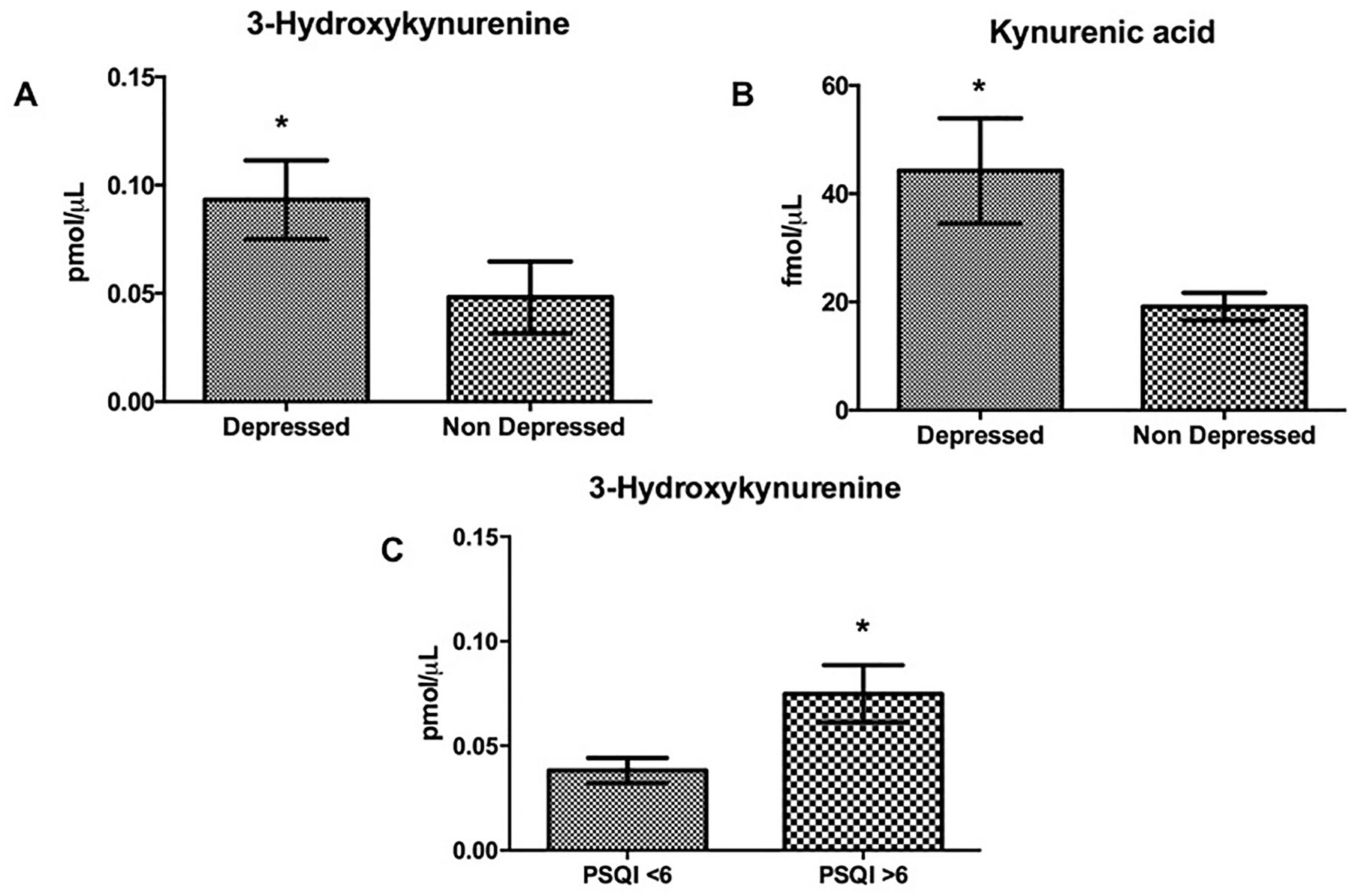
Significant differences in 3HK (A) and KYNA (B) serum levels was observed between depressed and nondepressed patients (Mann-Whitney test, P = .0497; P = .0271), with a significantly higher level of 3HK and KYNA in subjects with depression. (C) A significant difference was observed in 3HK serum levels between good and poor sleep quality patients, with significantly higher levels of 3HK in the group of patients with poor sleep quality (unpaired T test, P = .0190). The bars represent mean ± SEM. 3HK, 3-hydroxykynurenine; KYNA, kynurenic acid.
Discussion
Depression is a complication that frequently occurs in patients with stroke, global epidemiological studies report a frequency of 30% to 50% of cases. 7 Several studies suggest that the presence of depression is associated with a functional, cognitive, and vital adverse prognosis.3,21 For this reason, it is particularly important to know their risk factors, as well as to carry out an early diagnosis and treatment to try to reduce the associated disability and the costs of care.
Etiology of the PSD is considered multifactorial, a combination of biological and environmental factors; in this sense one of the most supported biological hypotheses currently is related with the participation of inflammatory mediators. 22 In this sense it has been shown that the proinflammatory environment increases the activation of the KP in the CNS.23,24 However, the neurobiological relevance of this route lies mainly in that there is evidence that some of its intermediate metabolites have redox and neuroactive properties, modulating glutamatergic, cholinergic, and dopaminergic neurotransmission. 25 Previous meta analytic studies have shown consistent changes in serum levels of KP metabolites: a reduction of serum tryptophan, KYNA, and L-Kyn and an increase of QUIN and 3HK in patients with major depression.9,10,26,27 However, the relationship of serum kynurenines with anxious-depressive symptoms in patients with stroke has been comparatively little studied.14-16 This study is relevant because it is the first report that shows a positive association of serum kynurenines levels with anxious-depressive symptoms and disability in patients with stroke.
In our study, the clinical and demographic characteristics (age, education, evolution time, type of stroke, laterality of the lesion, comorbidities, etc) of our sample were very similar to those reported by a previous study conducted in patients with stroke in Mexican population. 28 However, these characteristics differ in few aspects with regard to some international studies since our population had a lower average age and a lower socioeconomic and educational level than in international studies.29,30 In our study, we observed a frequency of significant depressive and anxious symptoms of 63% and 53%, respectively, these numbers are similar to those reported in the previous study by Arauz et al 28 (56% vs 63%); however, they are superior to those reported in the international literature (25.4% and 31%).29,30 This divergence may be due to multiple factors, for example, that different instruments were used for the diagnosis of depression, such as the DSM-IV (Diagnostic and Statistical Manual of Mental Disorders, fourth edition) or ICD-10 (International Classification of Diseases, 10th revision) in international studies, while in our study we used screening scales such as the HADS or the Beck Depression Inventory, 28 which are more sensitive but they can overestimate the frequency depression. Despite this, HADS continues to be the most widely used and supported mood assessment instrument in patients with stroke. 5 Additionally, patients were evaluated at different times of stroke, which may affect the frequency of presentation of the PSD. 31 When performing the analysis of correlation and association of the different subscales of HADS with the demographic, clinical, and other scales, only a significant positive correlation of all the subscales of the HADS was observed with the sleep quality (PSQI); this is consistent with reported in the international literature in relation to association between major depression and sleep. 32 However, poor sleep quality is not a factor that has been previously reported consistently in PSD, possibly because sleep quality is not an assessment that is routinely performed in patients with stroke. 33
In the present study, we found a positive significant correlation between the serum levels of 3HK with the HADS-T (r = 0.30) and HADS-D (r = 0.28), that is to say, at higher serum levels of 3HK also increase the depressive symptoms. These results differ from that obtained in previous studies, for example, the research conducted by Bensimon et al, 15 in which 86 patients with stroke were studied but there were important differences in the selection criteria of the patients with regard to our study: In their sample, patients with hemorrhagic stroke and cognitive impairment were excluded; on the other hand, patients with a higher average age (71.7 years), with antidepressants and with previous stroke were included; all these are factors that can influence serum Kyns levels, resulting in several confounding factors. In this study, an association was found between the rates of proinflammatory cytokines with depressive symptoms and fatigue, but there was no association with Kyns levels; however, in this study only Trp and L-Kyn were determined and no made determinations of other Kyns; so it is possible that due to the activation of the KP much of the L-Kyn is metabolizing to other intermediaries such as 3HK and KYNA and that explains the fact that no association has been observed with the levels of L-Kyn. Kyn or the Trp/L-Kyn index in that study. 12
On the other hand, the study conducted by Ormstad et al, 16 included 54 patients with the first stroke and measured the serum Kyns (L-Kyn, KYNA, QUIN), as well as Trp, serotonin, and other competitive amino acids at acute stroke (<72 hours) and correlated it with fatigue and depressive symptoms at 18 months. In this study, only an association of high KYNA levels with the severity of fatigue was found at 18 months of evolution, but no association of Kyns with depressive symptoms was observed. The discrepancy with our results may be due mainly to the fact that these patients were studied in acute stage, where the proinflammatory environment may be especially active due to stroke itself but may also be increased by the acute complications that may occur in patients in acute stage (eg, infection). In this sense it is possible that the Kyns levels in the subacute or chronic stage of the stroke are more stable (also due to the clinical stability of the patients) and perhaps for this reason have a greater association and influence on mood symptoms such as we observe in our results. 34 Additionally, there is evidence that in the acute stage of stroke, Kyns are associated with the severity of stroke and the extension of the brain damage, so perhaps in the acute stage, Kyns may be more a marker of acute brain damage and inflammation. 17 It is possible that in those patients in whom there is a persistence of the proinflammatory state for various factors this produces a greater activation of the KP and increase in the Kyns, which could generate various clinical consequences such as alterations in mood, fatigue or even cognitive impairment. 35
Consistently, in our study when the sample was divided into patients with and without depression, significantly higher serum levels were also observed in 3HK and KYNA in the group of depressed patients, which also supports the hypothesis that KP stimulation increases Kyns levels, which may be related to the development of PSD. The mechanisms by which Kyns can participate in the origin of the symptoms of PSD are not yet known with precision but there are several possibilities: KYNA is an endogenous antagonist of NMDA receptors, so it is demonstrated that increases in KYNA at the brain, they can impairs plasticity processes related to learning and memory, which may be related to the negative cognitive symptoms present in patients with depression.36,37 Interestingly, recent experimental studies show that KYNA can also show antidepressant-like effects in rats, so it is possible that the increase observed in our study may represent an endogenous compensation mechanism, however, these initial results should be replicated in the future. 38 However, it is prudent to point out that in our study the Kyns determinations were performed in the serum and it is demonstrated that KYNA crosses poorly the blood-brain barrier. 12 Therefore, the physiological significance of serum KYNA elevation may be different, for example, it may be related to an immunosuppressive effect, a phenomenon that has previously been demonstrated in major depression.39,40 It is also important to mention that, as has been previously proposed, the elevation of KYNA and other kynurenines in acute stroke may represent a mechanism of secondary tissue damage, 41 while the elevation of other kynurenines and the reduction of KYNA may be more related to the origin of the depressive symptoms as demonstrated in major depression. 26 However, a recent study shows that there is a close correlation between serum levels of Kyns and their levels in the cerebrospinal fluid, which can validate the use of serum Kyns as a good biomarker. 42
On the other hand, 3HK and its metabolite 3-hydroxyanthranillic acid, has shown an ambivalent role, behaving on the one hand as a toxin that induces damage by oxidative stress, and on the other exhibiting protective properties as a scavenger of free radicals43-45; whereby 3HK can represent, on the one hand, a tissue damage marker in stroke, which is supported by the fact that we also observe a significant negative correlation between serum levels of 3HK and disability in functional scales (Barthel index and FIM), which had not been previously reported. However, on the other hand, there is recent preclinical evidence that has shown that 3HK also have the ability to decrease the excitatory postsynaptic potentials at hippocampus, so perhaps through these mechanisms 3HK can also have a negative effect on the learning and memory processes in patients with PSD.46,47
Interestingly, when dividing patients with poor or good sleep quality in our study, we also found a significant increase in serum levels of 3HK in patients with poor sleep quality, which may be in accordance with previous experimental studies in where increase of Kyns brain levels in rats produces a reduction in the duration of REM (rapid eye movement) sleep, alterations in sleep architecture, and cognitive deficits. 48 However, the role of Kyns in the physiology of sleep and its disorders has not yet been adequately studied.
Finally, we consider relevant to point out some limitations of the present study. In first place, the small size of the sample, which although is comparable to previous publications, it will be important to replicate the present results in a larger sample of patients so that the results can be more generalizable to all patients with sequelae of stroke. Another limitation is the lack of control of fasting at the time of blood sampling, which can affect serum levels of Kyns depending on diet of subjects and finally the large number of comorbidities that can also influence the levels of Kyns; however, in this sense it is important to note that the number of comorbidities did not show an association in Kyns levels in the present study.
Conclusions
In conclusion, the levels of 3HK and KYNA correlated significantly with the severity of depressive symptoms in patients with stroke and also 3HK levels were significantly related to the degree of disability and the quality of sleep. Therefore, serum Kyns levels may represent a potential good marker of risk of PSD and disability in patients with stroke in the future.
Footnotes
Author Contributions
PCM and VPC contributed to the study’s conception, acquisition of the data, analysis and interpretation of the data, and drafting of the manuscript. BEC, PTG, JAMC, MRB, and JQF contributed to recruitment of patients, the acquisition of the data and the analysis and interpretation of the clinical data. FRC, GGC, ISV, KBM, and KFP contributed to the acquisition of the data (scales assessment). LSC, DRO, and LRC contributed to the analysis of the serum biomarkers.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Consejo Nacional de Ciencia y Tecnología (CONACYT) Grant 262010. However, funding source had no influence on decision to submit this manuscript for publication. We declare that there has been no other significant financial support for this work that could have influenced its outcome.
