Abstract
Background
Sleep disorders have a prevalence of 30% to 70% in post-stroke individuals. The presence of sleep disorders and poor sleep quality after stroke can affect important functions and lead to worse outcomes. However, most studies are restricted to the acute post-stroke stage only.
Objective
To investigate the frequency of self-reported sleep alterations in a sample of chronic stroke individuals and to identify which self-reported sleep alterations were associated with disability.
Methods
Prospective exploratory study. Self-reported sleep alterations were measured by the Pittsburgh Sleep Quality Index, Insomnia Severity Index, Epworth Sleepiness Scale, and STOP-Bang Questionnaire. The dependent variable was measured 3 years after the first contact by the Modified Rankin Scale (mRS). Step-wise multiple linear regression analysis was employed to identify which sleep alterations were associated with disability.
Results
Sixty-five individuals with stroke participated. About 67.7% of participants had poor sleep quality, 52.4% reported insomnia symptoms, 33.9% reported excessive daytime sleepiness, and 80.0% were classified as intermediate or high risk for obstructive sleep apnea. Only risk for obstructive sleep apnea was significantly associated with disability and explained 5% of the variance in the mRS scores.
Conclusion
Self-reported sleep alterations had a considerable frequency in a sample of chronic stroke individuals. The risk of obstructive sleep apnea was associated with disability in the chronic stage of stroke. Sleep alterations must be considered and evaluated in the rehabilitation process even after a long period since the stroke onset.
Introduction
Stroke is one of the leading causes of death and disability in the world. 1 Post-stroke individuals can present different clinical manifestations, such as motor, cognitive, and emotional alterations. In addition, these individuals may present important changes in sleep, including a reduction of total sleep time, increased sleep latency and alterations in breathing patterns during sleep. 2
The prevalence of sleep disorders in this population ranges from 30% to 70%, and the main sleep disorders described in stroke survivors are insomnia and obstructive sleep apnea.3,4 Insomnia is defined as complaints about sleep quality and quantity, associated with difficulties initiating sleep or maintaining sleep for at least 3 nights per week over 3 months or more, with impacts on functionality. Insomnia after a stroke can be explained by different mechanisms, including damage to central nervous system structures or the use of different medications. In addition, it is associated with lower quality of life. 5
Obstructive sleep apnea is the most prevalent sleep disorder in post-stroke individuals with a reported prevalence ranging from 50% to 70%. 4 It is characterized by repeated episodes of partial or total airflow obstruction during sleep due to the collapse of the upper airways, which results in reduced oxygen saturation. Studies have shown that obstructive sleep apnea is associated with metabolic disorders, cardiovascular diseases, and all-cause mortality. 6 Furthermore, obstructive sleep apnea is an independent risk factor for stroke, and studies suggest a relationship between obstructive sleep apnea and the occurrence of ischemic stroke during sleep, with the first signs of stroke first noted upon awakening from sleep (wake-up stroke).7,8
The main obstructive sleep apnea symptoms include snoring, sleep fragmentation, witnessed apneas, morning headaches, decreased concentration, memory loss, and excessive daytime sleepiness. Excessive daytime sleepiness is a common symptom in stroke individuals and can be explained by insufficient sleep, sleep fragmentation, or the presence of sleep disorders. 9 In the acute stage of stroke, excessive daytime sleepiness has an important role in outcomes with patients with this symptom showing 10 times more chances to go to a nursing home and have worse disability. 10
Different studies show that sleep can influence in post-stroke rehabilitation and has an important role in motor learning and cognitive performance.3,11,12 In stroke survivors, worse sleep quality is associated with a worse level of independence and minor improvement in rehabilitation in the acute stage. 11 Sleep disturbances are associated with balance and gait after 4 weeks of rehabilitation, with individuals with sleep disturbances showing a worse balance and gait function. 12 In another study, obstructive sleep apnea was associated with worse cognitive status, neurological status, independence, and longer periods of hospitalization. 13
However, most of the studies addressed only the acute stage of stroke and it is known that sleep problems are still present in the chronic stage and can influence rehabilitation, quality of life, mortality, and stroke recurrence.2,14,15 Long-term monitoring of post-stroke sleep alterations and their impact on disability is extremely important, especially considering that sleep alterations can persist for years after the stroke onset. 14 Most published studies that investigated the relationship between sleep alterations and disability have cross-sectional or retrospective designs, which do not allow the establishment of temporal relationships between variables or are subject to limited control in the measurement of outcome variables. 3
Therefore, it is essential to investigate the relationship between sleep alterations and disability after a long period since the stroke onset, especially considering that sleep alterations can have long-term negative impacts on the disability of these individuals. A better understanding of the relationship between sleep alterations and disability can improve the process of evaluation and the planning of the interventions, which can result in better neurological recovery and quality of life for this population. The objectives of the present study were to describe the frequency of self-reported sleep alterations in a sample of chronic stroke individuals and to identify which self-reported sleep alterations were associated with disability.
Materials and Methods
Design
A prospective exploratory study that adhered to the Strengthening the Reporting of Observational Studies in Epidemiology guidelines was carried out. Participants were recruited from the local community of a metropolitan city in Brazil, by means of advertisements and contact lists from previous research projects, from February to December 2019 and were followed up over 3 years since the first contact. Those participants, who were willing to participate, were physically screened and informed about the specific objectives of the present study.
Participants
Individuals with stroke were included if they were at the chronic stage (at least 6 months since the onset of the stroke), were above 20 years of age, and had no cognitive deficits, which were screened by education-adjusting scores on the Mini-Mental Examination (13 for individuals with illiteracy, 18 for those with elementary and middle education schooling, and 26 for those with basic education). 16 Those, who had comprehension aphasia and any other disabling neurological condition, not related to stroke that could influence measurement, were excluded.
The participants were informed about the objective of the study and were invited to provide written consent, based upon previous approval from the Institutional Ethical Review Board (#02465118.9.0000.5149). If an illiterate individual was interested in participating in the study, the consent form would be read in simple language with pauses to ensure understanding. At the end, the researcher must clearly ask if the individual wants to participate in the study. The answer must be yes or no. The entire process must be recorded in video and sent to the ethics committee.
Then, sociodemographic and clinical data, for example, age, time since the stroke on set, type of stroke, self-reported sleep alterations, and use of sleep medication, were collected for characterization purposes, followed by the collection of the independent variables in a research laboratory setting. All data were collected by trained research personnel. Three years after the first contact, interviews were then conducted by telephone by trained interviewers, who questioned the participants about their current disability. The timeline of the study is presented in Figure 1.
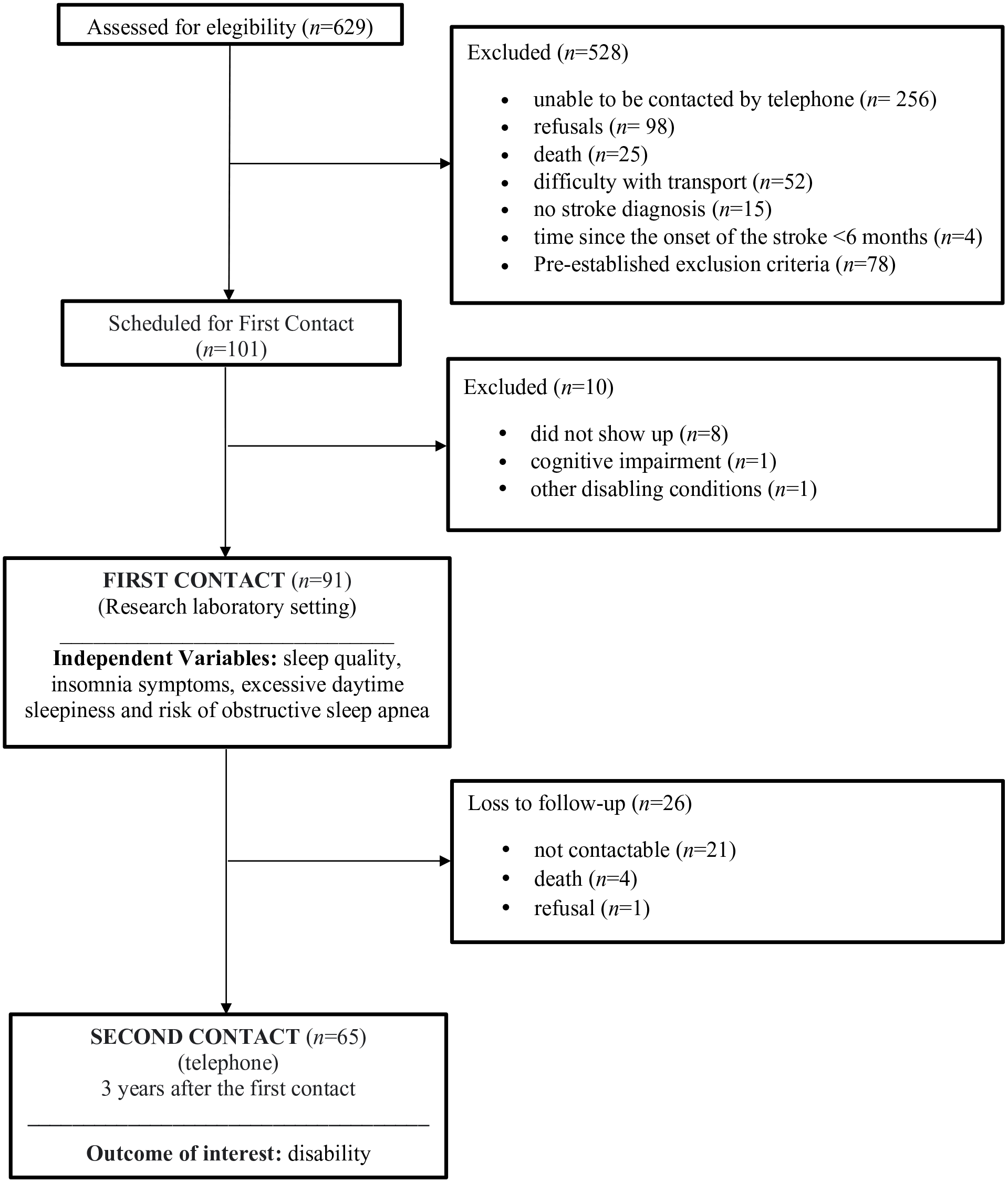
Study design and flow of participants through the study.
Outcomes
Dependent Variable: Disability
Disability was measured 3 years after the first contact by the Brazilian Version of the Modified Rankin Scale (mRS), 17 through a telephone interview. This scale is used to measure disability after stroke with scores ranging from 0 (no disability/dependence) to 6 (death) points.
The mRS is the most used scale to asses disability after stroke and can be applied by telephone with adequate validity and reliability. 17 The validity of the mRS has been demonstrated by comparisons with the American Heart Association’s Stroke Outcomes Classification, the Barthel Index, and the motor component of the Functional Independence Measure.18,19 Moreover, strong test-re-test reliability of the mRS was reported and inter-rater reliability of the mRS ranged from moderate to high. 20
Independent Variables: Self-Reported Sleep Alterations
The 4 independent variables were sleep quality, insomnia symptoms, excessive daytime sleepiness, and risk of obstructive sleep apnea.
Sleep quality was measured by the Brazilian version of the Pittsburgh Sleep Quality Index (PSQI). 21 The PSQI is a questionnaire that subjectively measures sleep quality, sleep duration, sleep latency, daytime dysfunction, use of sleep medication, and sleep disturbances. The total score ranges from 0 to 21 and higher scores indicate worse sleep quality. It is suggested a cut-off score of 5, with patients with a PSQI score higher than 5 being classified as poor sleepers, and those with PSQI scores lower or equal to 5 being classified as good sleepers. 22 The questionnaire is a valid and reliable tool to measure sleep quality in clinical and non-clinical samples. The PSQI has a sensitivity of 89% and a specificity of 86% in detecting good and poor sleepers. 22
Insomnia symptoms were measured by the Brazilian version of the Insomnia Severity Index (ISI). The ISI is a 7-item questionnaire that measures the patient’s perception of the severity and impact of insomnia. 23 Each item can be assigned a score from 0 to 4, resulting in a total score ranging from 0 to 28. Higher scores indicate more severe insomnia symptoms. The total score can be classified as no insomnia symptoms (0-7), mild insomnia symptoms (8-14), moderate insomnia symptoms (15-21), and severe insomnia symptoms (22-28). 24 The Brazilian Version of the ISI is a valid and reliable tool to measure insomnia symptoms in adults. A cutoff ISI score of 8 shows a sensitivity of 95.8% and a specificity of 78.3% to detect insomnia cases. 24
Excessive daytime sleepiness was measured by the Brazilian version of Epworth Sleepiness Scale (ESS). 25 The ESS is a tool that measures self-reported daytime sleepiness in 8 different situations and ranges from 0 to 24. Higher scores indicate a greater probability of falling asleep. 25 A score higher than 10 implies the presence of excessive daytime sleepiness. The scale has good construct validity for use in stroke and adequate test-retest reliability. The EES has a sensitivity of 36% and a specificity of 62% in detecting sleep disorders of breathing in individuals post-stroke. 26
The risk of obstructive sleep apnea was measured by the Brazilian version of the STOP-Bang Questionnaire. The questionnaire consists of 8 questions that measure the presence of snoring, tiredness, observed apnea during sleep, hypertension, body mass index over 35 kg/m2, age older than 50 years, and neck circumference above 43 cm for males and 41 cm for females. Scores range from 0 to 8 points with 1 point assigned to each positive answer. The scoring criteria are: low risk, for those who answered yes to 0 to 2 questions, intermediated risk, for those who answered yes to 3 to 4 questions, and high risk for those who answered yes to 5 to 8 questions. Subjects were also classified as high risk if they answered yes to 2 of the 4 questions addressing snoring, tiredness, observed apnea during sleep, or hypertension combined with one of the following characteristics: male gender, body mass index over 35 kg/m2, or neck circumference above 43 cm for male and 41 cm for female. The questionnaire is a valid and reliable tool for screening for obstructive sleep apnea in adults, when compared to polysomnography results, and has high test-retest reliability (k = 0.83-0.91). 27 STOP-Bang Questionnaire has a sensitivity of 87% and a specificity of 56% in detecting obstructive sleep apnea in individuals post-stroke. 28
Data Analyses
The sample size of at least 60 participants was estimated to include at least 15 participants per independent variable in the regression model. 29 Descriptive statistics, tests for normality (Kolmogorov–Smirnov), and equality of variances (Levene) were calculated for all outcomes. Spearmans’s correlation coefficients were calculated as the initial step, to determine which variables would be included in the regression model. The level of significance was set at P < .10, to ensure that independent variables were not excluded at this stage. Variables that were significantly correlated with the disability, were, then, inserted into the regression model.
Step-wise multiple linear regression analysis was employed to identify which of the independent variables that showed to be significantly correlated with the mRS scores would explain disability, 3 years after the stroke. Prior to performing regression, the data were evaluated, to determine if they fulfilled all requirements for regression analysis, such as linearity, homoscedasticity, independence, and normality of the residuals, as well as multicollinearity and outliers. Variable entry for the regression was set at .05, and removal was set at .10. Plots of the residuals against the predicted values and histograms of the residuals were also examined, to determine any violation of the assumptions for the regression analyses, mainly the normality of the residuals.
All analyses were carried out using the Statistical Package for the Social Sciences for Windows software (version 21.0), at a significance level of 5%.
Results
Flow of Participants Through the Study
From a list of 629 potential participants, 528 were excluded for the following reasons: unable to be contacted by telephone (n = 256), refusals (n = 98), death (n = 25), difficulty with transport (n = 52), no stroke diagnosis (n = 15), time since the onset of the stroke less than 6 months (n = 4), and pre-established exclusion criteria (n = 78). One hundred and one volunteers were scheduled for the measurement sessions. Of these, 8 did not show up, 1 had cognitive impairment, and 1 had other disabling conditions. After 3 years, 21 participants were not contactable, 1 refused to participate, and 4 had died, leaving 65 participants (Figure 1).
Participants’ Characteristics
A total of 65 individuals participated in the present study with a mean age of 59.8 (standard deviation [SD] = 12.9) years. The mean time since the stroke onset was 62.2 (SD = 59.9) months and 53 (81.5%) had an ischemic stroke. Regarding the self-reported sleep alterations, 44 (67.7%) participants had poor sleep quality, 34 (52.4%) reported insomnia symptoms, and 22 (33.9%) reported excessive daytime sleepiness.
According to the risk classification for obstructive sleep apnea 13 (20.0%) subjects were classified as low risk, 24 (36.9%) were classified as intermediate risk, and 28 (43.1%) as high risk. However, only 9 (13.8%) individuals were referred for polysomnography after the stroke and 3 (4.6%) reported using continuous positive airway pressure (CPAP). About 13 (20.0%) participants, who were diagnosed with ischemic stroke, reported that the first signs of the stroke were noted upon awakening from sleep (wake-up stroke). After 3 years, 17 (26.2%) participants were diagnosed with a new comorbidity and the mean mRS was 1.5 (0.9). Their main characteristics are summarized in Table 1.
Clinical and Demographic Characteristics of the Participants.
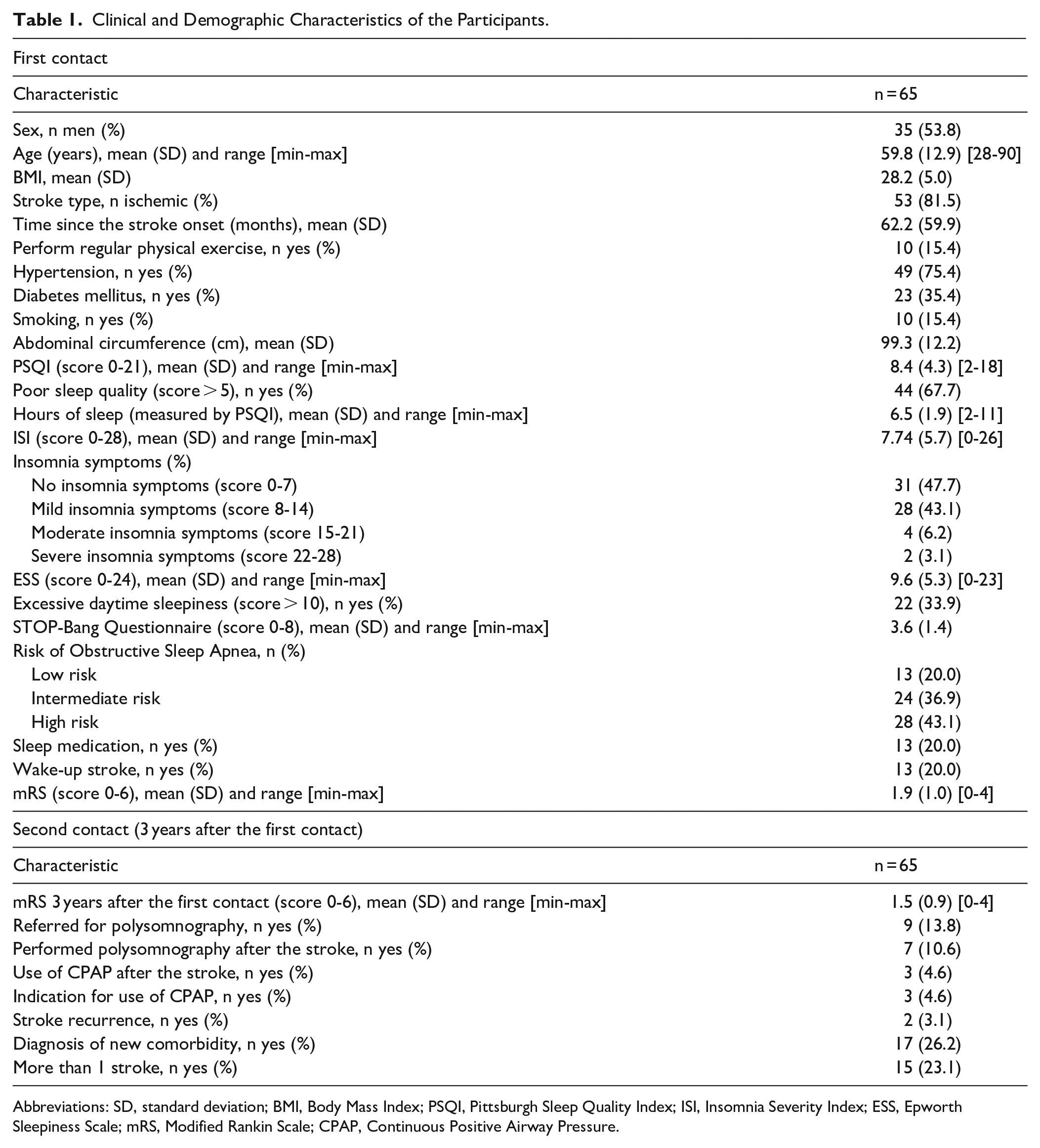
Abbreviations: SD, standard deviation; BMI, Body Mass Index; PSQI, Pittsburgh Sleep Quality Index; ISI, Insomnia Severity Index; ESS, Epworth Sleepiness Scale; mRS, Modified Rankin Scale; CPAP, Continuous Positive Airway Pressure.
Regression Analysis
Significant and positive correlations were found between the mRS scores and sleep quality (rs = .221, P = .092) and risk of obstructive sleep apnea (rs = .225, P = .071), that is, PSQI scores and STOP-Bang Questionnaire scores. However, no significant correlations were found between the mRS scores and insomnia symptoms (rs = .121, P = .336), and excessive daytime sleepiness (rs = .038, P = .761), that is, ISI scores and ESS scores (Table 2). Therefore, sleep quality and risk of obstructive sleep apnea were, then, inserted into the regression model.
Correlation Coefficients and Statistical Significance (P Values) Between Disability and All Measured Independent Variables (n = 65).
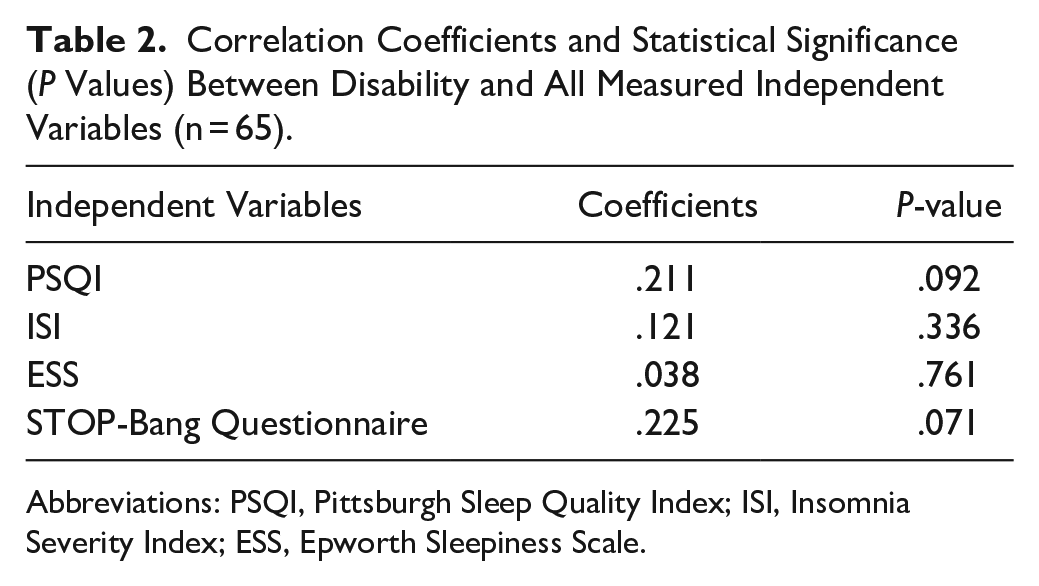
Abbreviations: PSQI, Pittsburgh Sleep Quality Index; ISI, Insomnia Severity Index; ESS, Epworth Sleepiness Scale.
The regression analysis revealed that only the STOP-Bang Questionnaire was retained in the model and explained 5% of the variance in the mRS scores (Table 3). The STOP-Bang Questionnaire scores were positively correlated with the mRS. This indicated that individuals, who had a higher risk of obstructive sleep apnea, were more likely to report worse disability.
Results of the Linear Regression Analysis Regarding the Independent Variables of Disability After Stroke (n = 65).

Abbreviations: B, regression coefficient, followed by the respective standard error; F, critical value; P, P-value; CI, confidence interval; β, standardized regression coefficient; R2, coefficient of determination; SEE, standard error of estimate.
Discussion
The present study aimed at identifying the frequency of self-reported sleep alterations in individuals with chronic stroke, and whether those self-reported sleep alterations were associated with disability in the chronic stage of stroke, 3 years after the first assessment. The participants showed moderate to high frequencies of poor sleep quality, insomnia symptoms, excessive daytime sleepiness, and risk for obstructive sleep apnea. Only the STOP-Bang Questionnaire was retained in the regression model, suggesting that the risk of obstructive sleep apnea was associated with disability.
These results are similar to other studies that investigated the frequency of self-reported sleep alterations after stroke.15,30-32. Ho et al 30 reported a poor sleep quality frequency of 64%, while Oliveira et al 31 have found a frequency of 71% in individuals with chronic stroke. Concerning the presence of insomnia symptoms, Polomaki et al 32 corroborate the present study results. They reported that 49% of the post-stroke individuals showed insomnia complaints, 18 months after stroke. However, Glozier et al 15 have found a lower frequency with 30% to 37% of the participants reporting insomnia symptoms, 6 and 12 months after the stroke. These distinct results may be due to differences in the instruments used to measure insomnia symptoms (Karolinska Sleep Questionnaire vs Insomnia Severity Index) and in the characteristics of the participants. The maximum age of the participants of the Glozier et al 15 study was 65 years, in contrast to 90 years in the present study.
Studies have investigated the frequency of excessive daytime sleepiness after stroke.33,34 Martynowicz et al, 33 using the same classification criteria as the present study, reported 33% of excessive daytime sleepiness 3 months after stroke, which is quite similar to the present study. Mills et al, 34 have also found similar results with 41% of post-stroke individuals reporting excessive daytime sleepiness and 54% of the participants reporting sleeping during the day with a mean time of 1 hour. Regarding the risk of obstructive sleep apnea, Sarfo et al 35 reported a frequency of 87% of intermediate or high risk for obstructive sleep apnea in individuals with chronic stroke. Gottlieb et al, 2 have performed polysomnography in a small sample of 28 chronic stroke individuals and have found that 57% of the participants had undiagnosed moderate or severe obstructive sleep apnea. Thus, these results show that sleep alterations are present in the chronic stage of stroke in moderate to high frequencies.
Despite the moderate to high frequencies of self-reported sleep alterations observed through the application of questionnaires, only 9 (13.8%) individuals reported being referred to polysomnography. Similar results have been found by Brown et al 36 in a sample of post-stroke patients in the United States, with only 6% of the participants being referred to obstructive sleep apnea diagnostic test after stroke and only 5% of the patients reported being directly questioned about snoring, one of the main obstructive sleep apnea symptoms. These findings may be explained by the high cost and low availability of polysomnography in most of clinical settings, but also by the lack of knowledge of most of healthcare professionals about the relationship between stroke and sleep disorders. Strategies to overcome these barriers should be addressed. Also, it is important to consider that obstructive sleep apnea can contribute to the development of other conditions, such as diabetes, dyslipidemia, arterial hypertension, and arrhythmia, all important risk factors for the occurrence of stroke. 6 Indeed, the present study showed that 26.2% of the participants were diagnosed with a new comorbidity in a period of 3 years and 20.0% reported wake-up stroke.
The frequency of wake-up stroke and its association with obstructive sleep apnea have been investigated.7,8 Similar to the present study, Barreto et al 37 and Schutz et al 38 reported wake-up stroke rates of 29% and 30%, respectively. A recent meta-analysis that included 13 studies with stroke individuals has concluded that wake-up stroke individuals have worse sleep disorders of breathing than non-wake-up stroke ones, which might indicate that obstructive sleep apnea plays an important role in the occurrence of the wake-up stroke. 8 Moreover, some studies suggested that individuals diagnosed with wake-up stroke may have worse functional outcomes. 7 Thus, individuals diagnosed with wake-up stroke need particularly to be questioned about sleep alterations and to be closely monitored since the early hours after the stroke.
The present study showed that only the risk of obstructive sleep apnea was significantly associated with disability and explained 5% of the variance in the mRS scores. To strengthen the argument that the risk of obstructive sleep apnea, measured by the STOP-Bang Questionnaire, could contribute to future disability, the correlation coefficient between the mRS baseline values and the STOP-Bang Questionnaire scores was calculated. The lack of significant correlation strengthens the contribution of the risk of sleep apnea to future disability.
According to our knowledge, this is one of the first longitudinal studies that have investigated the relationship between self-reported sleep alterations and disability in individuals with chronic stroke. Other studies have investigated the relationship between sleep alterations and disability in different stages of stroke recovery.4,37,39-41 Camilo et al 4 have found similar results to those of the present study with severe sleep apnea in the acute stage being an independent factor associated with disability after 6 months since the ischemic stroke onset. Menon et al 39 and Barreto et al, 37 have also found similar results with individuals diagnosed with obstructive sleep apnea by polysomnography in the acute stage of stroke, showing worse mRS scores after 3 months and after 12 months respectively.
Other studies have not found an association between obstructive sleep apnea and disability after stroke.40,41 Ahn et al 40 found a difference in disability at hospital discharge post-stroke between individuals with and without obstructive sleep apnea. However, the study found no difference in recovery between hospital discharge and 3 months after stroke in individuals with and without obstructive sleep apnea. Bassetti et al, 41 found similar disability measured by mRS 6 months after stroke for individuals with and without obstructive sleep apnea. The differences between the results may be due to the differences in measures and criteria used to classify obstructive sleep apnea. Moreover, most of the studies have carried out the investigation of obstructive sleep apnea in individuals who were hospitalized, a few hours after the stroke onset. It is known that during this period the neurological condition is often unstable and the patient may be exposed to several aspects that can interfere with sleep, such as noise, medication, and routine interventions by the medical team.
In the present study sleep quality, insomnia symptoms, and excessive daytime sleepiness were not associated with disability. These findings are supported by other studies. Tang et al, 42 did not find a difference in daily living activities between groups with or without insomnia symptoms, 3 months after stroke, while Zhang et al 43 did not identify poor sleep quality and excessive daytime sleepiness as risk factors for poor neurological function in the chronic stage of stroke. However, contrary to the present results, Zhang et al 43 did not identify a high risk for obstructive sleep apnea as a risk factor for poor neurological function, 4 years after stroke. These conflicting results may be explained by the fact that the risk of obstructive sleep apnea was measured by the Berlim Questionnaire, which has a lower sensitivity for sleep-disordered breathing in post-stroke individuals, in comparison to the STOP-Bang Questionnaire. 26 Furthermore, Fulk et al, 3 in a systematic review found mixed results with approximately 50% of the studies showing significant differences in disability between stroke individuals with and without sleep disorders and the other 50% showing no difference. It is important to consider a few points: most of the studies included in the systematic review investigated the acute stage of stroke, the chronic stage was defined as at least 3 months after the stroke onset, and in most of the studies, the sleep assessment was performed in the acute stage of stroke, and many studies had no follow-up.
The main strength of the present study is the longitudinal nature that allows the establishment of a temporal relationship between self-reported sleep alterations and disability. Furthermore, the study only included individuals in the chronic stage of stroke, when neurological and environmental conditions are more stable and may have less influence on the results. It is also necessary to acknowledge the study limitations since sleep alterations were measured only by questionnaires, instead of polysomnography, which is the gold standard. However, polysomnography is a high-cost and technically complex exam and may not be available in most clinical settings. Another alternative could be actigraphy, a non-invasive method for evaluating sleep-wake cycles, based on continuous monitoring of body movements and the identification of periods of activity and rest. Its advantage is to provide information for extended periods in the user’s natural environment. 44 Although actigraphy is a good method for assessing sleep, the equipment is not available in most clinical settings and has a relatively high cost.
The use of validated questionnaires might be a good alternative for clinical practice, especially in areas with limited resources, being fast, low-cost, and with no need for equipment. In addition, another limitation of the study was the long follow-up period of 3 years without multiple assessments, which may have contributed to the loss of follow-up of part of the participants. It is important to consider that the first part of the study was completed a few months before the coronavirus disease 2019 pandemic, being necessary adjustments for the feasibility of the second part of the study.
It is also important to mention the use of a self-report instrument to assess disability, in which self-report can be influenced by different factors such as comorbidities and socioeconomic factors.45,46 However, the mRS is the most used scale to asses disability after stroke and is a valid and reliable instrument. In the future, longitudinal studies with multiple follow-ups and larger samples, using subjective and objective measures of sleep alterations should be conducted, in order to better understand the relationship between sleep alterations and disability in this population.
Conclusions
The findings of the present study showed that self-reported sleep alterations had a moderate to high frequency in a sample of individuals with chronic stroke. However, few of these individuals were referred for polysomnography. The risk of obstructive sleep apnea was associated with disability in the chronic stage of stroke. Therefore, sleep alterations, especially obstructive sleep apnea, should be considered and evaluated in the rehabilitation process, even after a long period since the stroke onset.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Brazilian Government Funding Agencies (CAPES- code 001, CNPq, FAPEMIG, and PRPq/UFMG).
