Abstract
Background. Sleep changes are frequently reported following traumatic brain injury (TBI) and have an impact on rehabilitation and quality of life following injury. Potential causes include injury to brain regions associated with sleep regulation, as well as secondary factors, including depression, anxiety, and pain. Understanding the nature and causes of sleep changes following TBI represents a vital step in developing effective treatments. Objective. The study aimed to investigate subjective sleep changes in a community-based sample of individuals with TBI in comparison with noninjured age- and sex-matched controls and to explore the impact of secondary factors (pain, anxiety, depression, employment) on these self-reported sleep changes. Methods: A total of 153 participants with mild to severe TBI and 128 noninjured controls completed self-report measures relating to their sleep quality, daytime sleepiness, mood, fatigue, and pain and completed a sleep diary each day for 7 days. Results. Compared with the noninjured controls, participants with TBI reported significantly poorer sleep quality and higher levels of daytime sleepiness; sleep diaries revealed longer sleep onset latency, poorer sleep efficiency, longer sleep duration, and more frequent daytime napping in the TBI group, as well as earlier bedtimes and greater total sleep duration. Anxiety, depression, and pain were associated with poorer sleep quality. Greater injury severity was also associated with a need for longer sleep time. Conclusion. These findings highlight the importance of assessing and addressing pain, anxiety, and depression as part of the process of treating TBI-related sleep disturbances.
Keywords
Introduction
In recent years, there has been increasing awareness of changes in sleep following traumatic brain injury (TBI). 1 Although the frequency of reported changes varies widely, between 30% and 80%, it is significantly higher than in the general population. 2 These changes have been documented both in the acute recovery stages during inpatient rehabilitation3,4 and in the longer term, up to many years after injury.5-8 They have been reported as often, if not more frequently, by individuals with mild TBI as by those with moderate to severe injuries.5,6,9,10 Occurring in the context of brain injury, sleep disturbance may have an impact on rehabilitation and daily activities, 11 as well as on well-being and quality of life. 12
Obtaining a clear understanding of the nature and causes of sleep changes represents a vital step in the development of effective treatments. There has been considerable variability in sleep complaints documented, including the presence of nightmares, delayed sleep onset, early awakening, poor sleep efficiency, excessive daytime sleepiness, in addition to disorders of sleep (ie, sleep apnea, insomnia, hypersomnia, periodic limb movements, narcolepsy).2,13-16 The etiology of sleep disturbances following TBI is presently unclear. They may be attributed to injury to brain regions, pathways, and neurotransmitter systems associated with sleep regulation, including the suprachiasmatic nucleus, hypothalamus, midbrain, and ascending reticular activating system.13,17-20 However, secondary symptoms, including depression, anxiety,4,5,14,21 fatigue, and pain6,22,23 have also been associated with sleep complaints in people with TBI. The direction of the association between sleep, anxiety, and depression remains unclear.5,24 Fatigue may increase frequency of daytime napping, which in turn may affect nighttime sleep quality. 6 Sleep disturbance may also exacerbate pain. 23 Further understanding the impact of secondary symptoms on self-reported sleep and sleep quality has implications for effective and tailored treatment of these complaints.
Studies to date have used variable measures to document sleep changes, some of which have not been validated for use in the TBI population. Studying patients in inpatient settings is problematic, given the potential impact of environmental factors and imposed routines on sleep in these settings. Other studies have recruited patients from the community at long periods after injury, 6 thus potentially biasing findings toward those who have greater psychopathology. Very few studies have employed demographically matched control groups.
We have previously reported results from a preliminary study by Parcell et al 5 and Ponsford et al 16 involving 63 patients with mild to severe TBI who were on average 230 days postinjury. We documented high rates of sleep changes and a decline in sleep quality relative to preinjury, as well as increased nighttime awakenings in sleep diary reports. Higher levels of anxiety and depression were present in those with milder injuries. Both these factors were associated with greater self-reported sleep change postinjury, poorer sleep quality (measured using a general sleep questionnaire), and increased nighttime awakenings shown in sleep diary reports. The impact of pain was not examined in this study.
The present study extends these findings to a larger sample and includes some additional measures. Specifically, sleep quality was assessed using the Pittsburgh Sleep Quality Index (PSQI), a measure validated for use in the TBI population since commencement of the previous study. Pain was assessed using the Brief Pain Inventory (BPI). The aim of the study was to further investigate subjective sleep changes in an expanded, community-based sample of individuals with TBI in comparison with noninjured age- and sex-matched controls and to explore the impact of secondary factors, including pain, anxiety, and depression on these self-reported sleep changes. It was hypothesized that individuals with TBI would report more sleep changes than controls, poorer sleep quality, increased sleep onset latency, more nighttime awakenings, and greater daytime sleepiness and that these changes would be associated with anxiety, depression, and pain.
Methods
Participants
Study participants were individuals with mild to severe TBI, consecutively recruited from the community following inpatient rehabilitation at Epworth Hospital, Melbourne; and age- and sex-matched noninjured controls recruited from the general population. Participants were required to be aged 16 to 65 years; have sufficient English language ability to complete the questionnaires; no history of transmeridian travel across >1 time zone in the preceding 3 months and no shift work in the previous 12 months; no preinjury sleep disorder, as indicated by significant other’s responses on a screening checklist for classic symptoms of periodic limb movement disorder, sleep apnea, or insomnia; no current use of benzodiazepines or other sleeping medications; and no history of previous head injury, neurologic disorder, or major psychiatric disorder. Noninjured control participants met the same criteria and had never sustained a head injury.
Procedure
Individuals with TBI were enrolled into the study at least 2 weeks postdischarge from hospital. They were asked to complete a range of self-report measures and questionnaires relating to their sleep and to prospectively record their sleep pattern daily for 7 days in a sleep diary. Participants were instructed to complete the diary on awakening each morning and again at the end of each day to record napping behavior. The noninjured control participants completed the same measures. In addition to these measures, all participants who were newly recruited into the study also completed the PSQI and BPI. These questionnaires were added to the protocol of the previous reported study 5 to provide a subjective global measure of sleep quality using a measure validated for use in the TBI population 25 and to allow for investigation of the impact of pain on sleep changes. The protocol was approved by the Human Research and Ethics Committees of Monash University and Epworth Hospital.
Measures
Demographic and Medical History
Participant demographics were collected from interviews. Following consent, injury characteristics (time since injury, duration of posttraumatic amnesia [PTA], and Glasgow Coma score [GCS]) were obtained from medical files. Duration of PTA was measured prospectively using the Westmead Posttraumatic Amnesia Scale. 26
Sleep–Wake Diary
A 7-day sleep–wake diary was used to assess subjective sleep timing and continuity. Information obtained for each day included estimates of time of going to bed (bedtime), time of sleep onset, number, and duration of nighttime awakenings, time of morning awakening (wake time), number and duration of daytime naps, and caffeine and alcohol consumption. Nocturnal sleep duration (minutes) and sleep efficiency (percentage and time in bed spent asleep) were calculated from provided data.
Epworth Sleepiness Scale
The Epworth Sleepiness Scale (ESS) 27 is widely used in research and clinical practice to identify people with excessive daytime sleepiness. The ESS was used as a subjective measure of daytime sleepiness and required participants to rate their likelihood of dozing or falling asleep in specified sedentary situations. A total score of ≥10 (range, 0-24) is considered an indicator of excessive daytime sleepiness. 27 It has been used in a number of previous studies documenting sleep changes following TBI5,14,28-30
Pittsburgh Sleep Quality Index
The PSQI was added to the measures in the previous study 5 to provide an additional measure of self-reported sleep quality previously shown to be sensitive to sleep disturbance in the TBI population. 31 It has been recommended for routine assessment of sleep in patients with TBI. 2 The PSQI comprises 19 self-rated questions producing a global score ranging from 0 to 21. Higher scores represent poorer sleep quality with a global score of >5 indicative of poor sleep quality. 31 In an outpatient TBI sample, a cutoff score of >8 has demonstrated high sensitivity (93%) and specificity (100%) to insomnia. 25
General Sleep Questionnaire
Participants used a tick-box format to comment on whether their sleep had changed and in what way since the head injury (TBI group) or in the past 3 months (noninjured control group). They were asked to identify factors that restricted their sleep. In addition, 2 sleep quality rating scales, derived from the General Sleep Habits Questionnaire 32 used by Parsons and ver Beek 33 were also administered. The first was a 6-point scale assessing “How do you feel on waking in the morning?” Responses ranged from “refreshed” to “need more sleep.” The second scale was a 4-point scale asking “How rested do you feel in the morning?” Reponses ranged from “very rested” to “not rested at all.” Previous 5 and current analysis shows that TBI participant responses to both scales correlate significantly (n = 144, r = 0.79, P < .0001). Information from both scales was summed to give a sleep quality score between 2 and 10, with higher scores reflecting less satisfaction with sleep.
Hospital Anxiety and Depression Scale
The Hospital Anxiety and Depression Scale (HADS) 34 is a brief, self-report measure of symptoms of anxiety and depression. It includes relatively few symptoms which may overlap with TBI symptoms and none relating to sleep disturbance. It comprises 14 items rated on a 5-point scale to indicate severity. Scores range from 0 to 21, with severity of symptoms rated as normal (0-7), mild (8-10), moderate (11-14), or severe (15-21). The HADS is considered a reliable measure of anxiety and depression in the TBI population.35-37
Brief Pain Inventory
The BPI was added because of the documented impact of pain on sleep following TBI. 6 The BPI is a short, self-administered questionnaire asking individuals to rate their current pain intensity and pain in the past 24 hours at its worst, least, and on average by using a scale of 0 (“no pain”) to 10 (“pain as bad as you can imagine”). 38 Previous research has indicated that assessment of “worst pain” intensity correlates with severity of functional interference in radiology patients. 39 The BPI has been used in prior research within the trauma population. 40
Data Analysis
Analyses were performed using SPSS Version 20 (IBM SPSS Inc, Chicago, Illinois). Between-group comparisons were conducted using parametric independent t tests and χ2 tests for categorical variables. Where indicated, between-group comparisons were also conducted using Mann–Whitney nonparametric tests because of nonnormality of the distribution. Associations between selected variables were examined using Pearson r and Spearman ρ, as specified. Linear regression was conducted to examine the association of various factors with sleep quality.
Results
Participant Characteristics
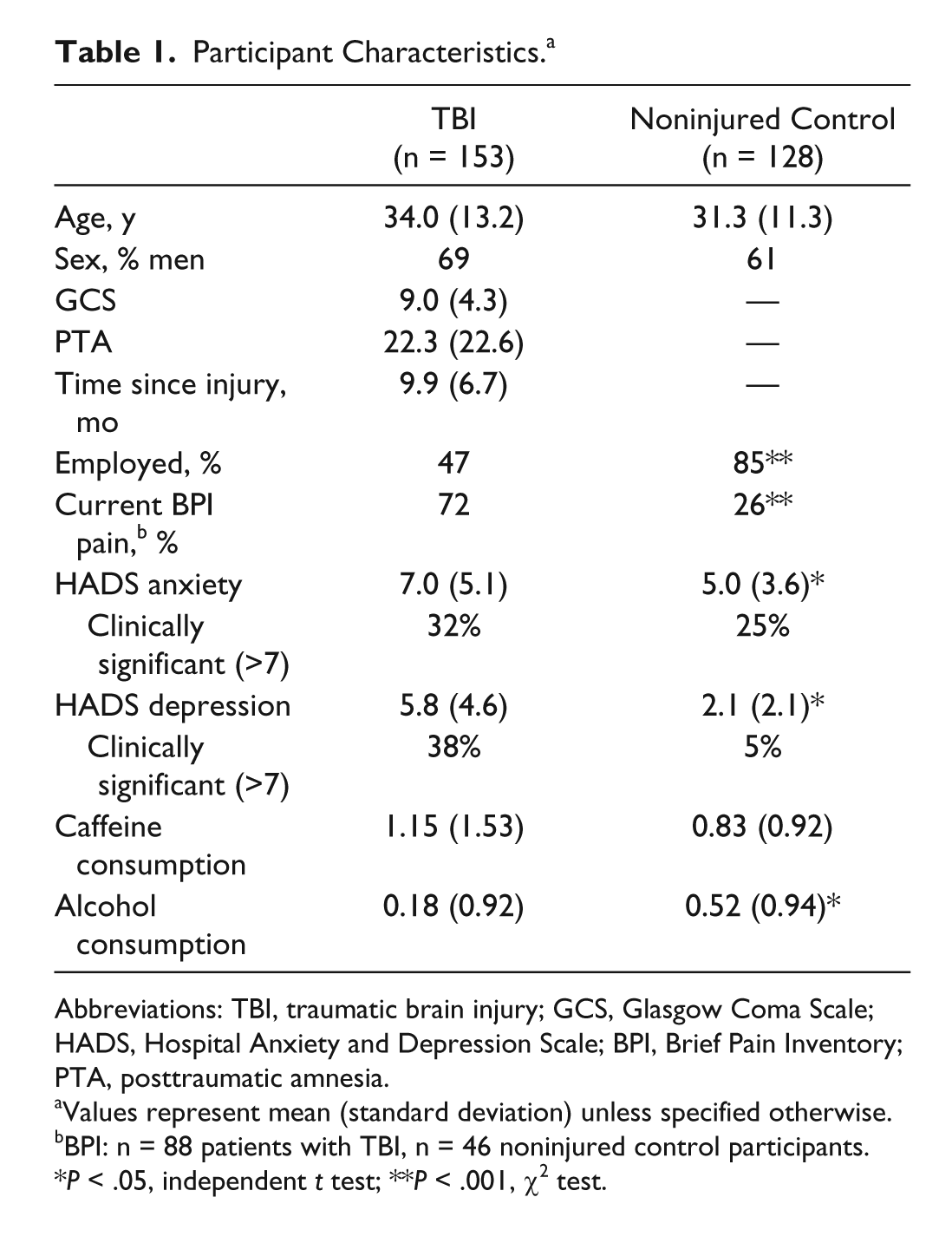
Participants in the current study included 153 patients with TBI and 128 noninjured control participants (Table 1). There was no significant difference between the groups in terms of age or sex (P > .05), although the TBI group were more likely to be taking medication (38.6%) than the control group (19.3%), were significantly less likely to be employed, and were more likely to be experiencing pain symptoms. Medication use in the TBI group included antidepressants (11%), anticonvulsants (9%), antiepileptics (3%), regular pain medication (16%), and other (14%).
Participant Characteristics. a
Abbreviations: TBI, traumatic brain injury; GCS, Glasgow Coma Scale; HADS, Hospital Anxiety and Depression Scale; BPI, Brief Pain Inventory; PTA, posttraumatic amnesia.
Values represent mean (standard deviation) unless specified otherwise.
BPI: n = 88 patients with TBI, n = 46 noninjured control participants.
P < .05, independent t test; **P < .001, χ2 test.
In terms of injury characteristics, based on duration of PTA, 4% of the participants with TBI had a mild injury (PTA < 24 hours), 22% had a moderate injury (PTA 1-7 days), 49% had a severe injury (PTA 8-28 days), and 25% had a very severe injury (PTA > 4 weeks). Causes of injury included motor vehicle accidents (74%), pedestrian accidents (8%), assaults (2%), falls (11%), and other (5%). Brain computed tomographic data, available for 94% of participants with TBI, revealed abnormalities in 69% of scans, in frontal (40%), temporal (24%), parietal (21%), occipital (12%), medial/limbic (11%) regions, and diffuse axonal injury in 13% of scans. Seventy percent of the participants with TBI also sustained physical injury at the time of TBI.
The participants with TBI reported significantly higher scores for anxiety and depression on the HADS than the noninjured control group (Table 1). For the TBI group, anxiety was associated with increasing time since injury (n = 151, r = 0.19, P = .02). In contrast to the previous study, 5 there was no significant association between anxiety and injury severity on the GCS (n = 153, r = 0.11, P = .18). Time since injury was not associated with severity of depressive symptoms (n = 151, r = 0.14, P = .09). There was no difference between the groups in daily caffeine consumption, although the noninjured control group reported consuming more alcohol on average than the participants with TBI.
Sleep Symptoms
Sleep Quality and Daytime Sleepiness
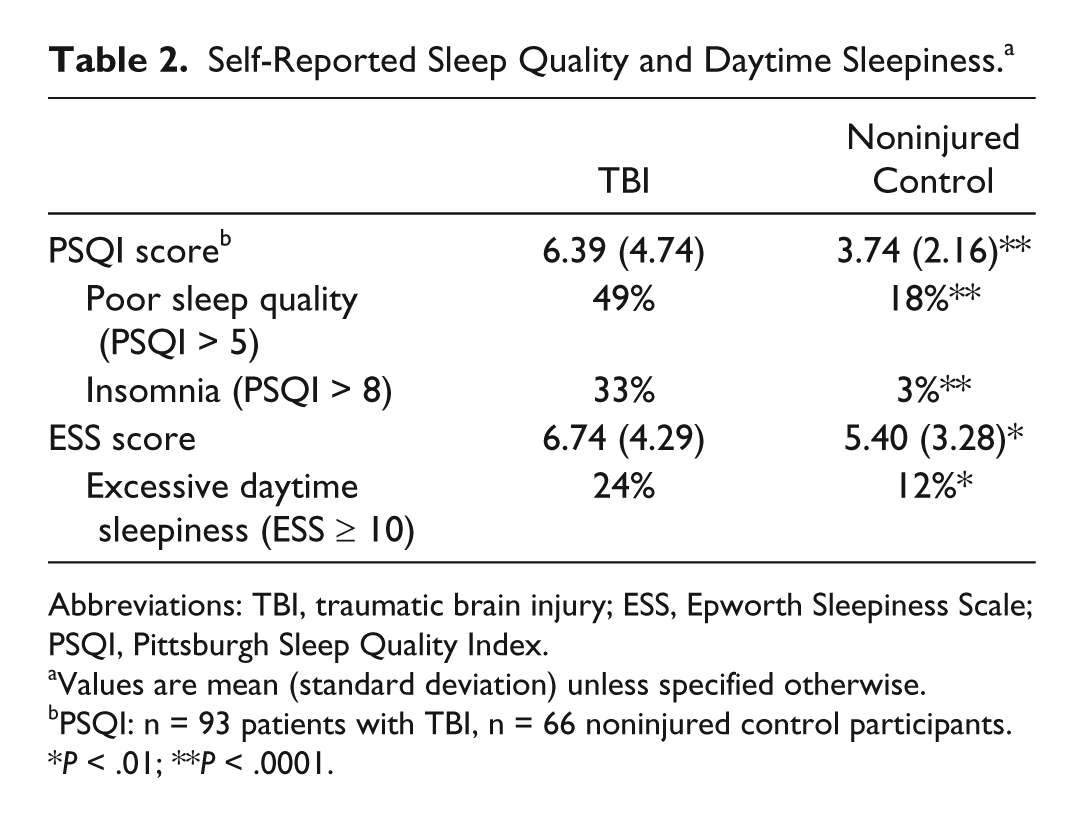
Table 2 shows group differences in self-reported sleep quality and daytime sleepiness. The PSQI was completed by 61% (n = 93) of the TBI participants and 51% (n = 66) of the noninjured control participants in the current study. There was no significant difference in demographics (age, sex) or injury characteristics (PTA, GCS) between participants with TBI who completed the PSQI and those from our previous sample (n = 63) 5 who did not complete the PSQI (P > .05). As seen in Table 2, the TBI group reported significantly poorer sleep quality than the control group. A significantly higher proportion of participants with TBI was classified as having poor sleep quality (PSQI > 5) and symptoms suggestive of insomnia (PSQI > 8). Additionally, sleep quality—determined by responses on the General Sleep Questionnaire asking about how participants “felt” and how “rested” they were each morning—was found to be significantly poorer in the TBI group (M = 5.20, SD = 2.17, n = 144) relative to the noninjured controls (M = 4.68, SD = 1.84, n = 114;), t(256) = 2.07, P = .04). A similar proportion of participants in the TBI (45%) and non injured control (47%) groups identified factors that they believed restricted their sleep. Factors most frequently identified by the TBI group included pain (25%), worries (6%), and work or chores (5%), compared with work or chores (18%), children (7%), and light (6%) in the noninjured control group. With regard to daytime sleepiness, TBI participants reported significantly higher levels of daytime sleepiness relative to noninjured controls, and a significantly higher proportion of the TBI sample reported excessive daytime sleepiness (ESS ≥ 10; Table 2).
Self-Reported Sleep Quality and Daytime Sleepiness. a
Abbreviations: TBI, traumatic brain injury; ESS, Epworth Sleepiness Scale; PSQI, Pittsburgh Sleep Quality Index.
Values are mean (standard deviation) unless specified otherwise.
PSQI: n = 93 patients with TBI, n = 66 noninjured control participants.
P < .01; **P < .0001.
Sleep Changes Following Injury
A significantly higher proportion of participants with TBI (80%, n = 144) reported changes to their sleep since their injury in comparison with the control group in the past 3 months (30%, n = 114), χ2(1) = 65.29, P < .0001. Figure 1 displays the difference between groups in the frequency of reported changes to sleep following injury (for the TBI group) and in the past 3 months (for the noninjured controls).
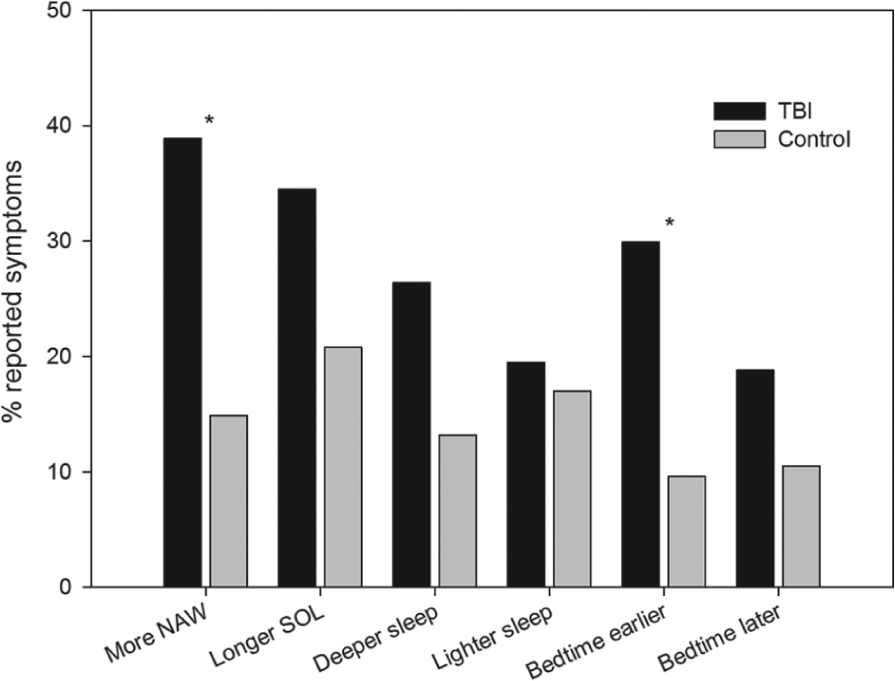
Frequency of self-reported changes to sleep in both TBI and noninjured control groups. Abbreviations: TBI, traumatic brain injury; NAW, nighttime awakenings; SOL, sleep onset latency.
Self-Reported Sleep Timing
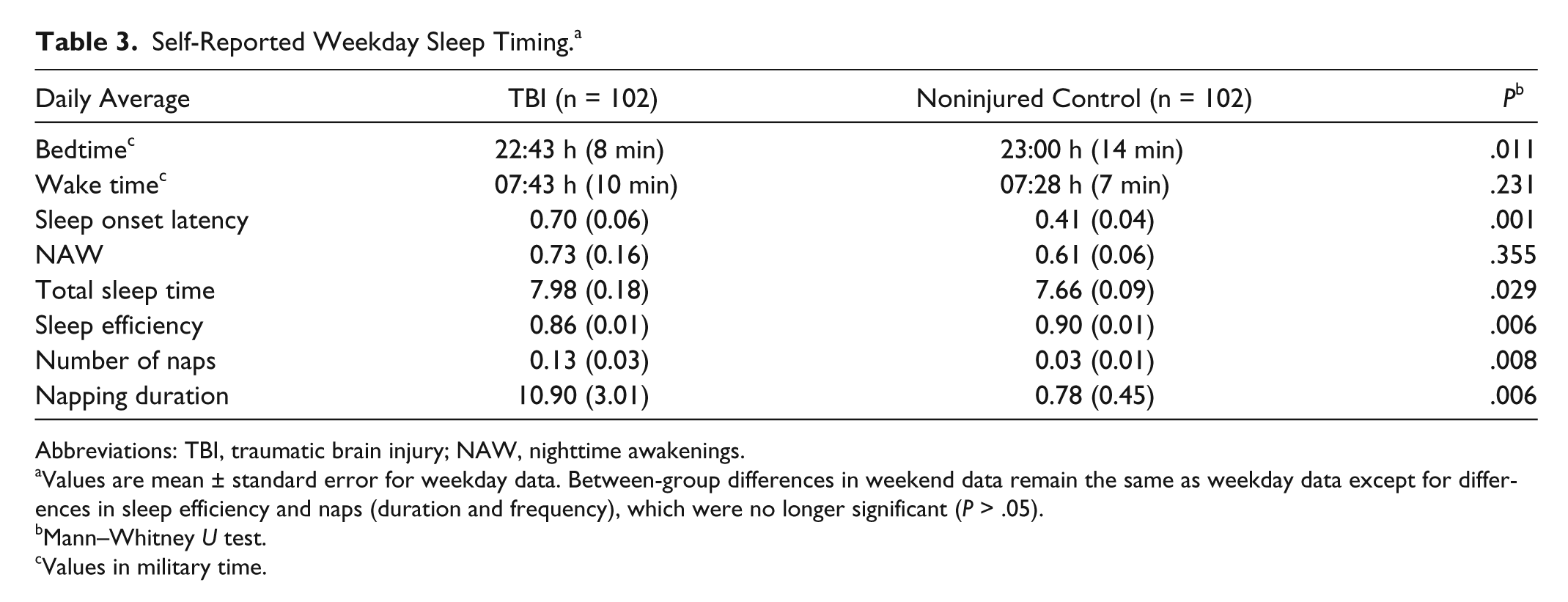
Group differences in self-reported sleep timing on weekdays are displayed in Table 3. Compared with noninjured controls, participants with TBI reported longer sleep onset latency, poorer sleep efficiency, and longer durations of daytime napping. In addition, they reported earlier bedtimes, greater total sleep duration, and more frequent daytime napping during weekdays.
Self-Reported Weekday Sleep Timing. a
Abbreviations: TBI, traumatic brain injury; NAW, nighttime awakenings.
Values are mean ± standard error for weekday data. Between-group differences in weekend data remain the same as weekday data except for differences in sleep efficiency and naps (duration and frequency), which were no longer significant (P > .05).
Mann–Whitney U test.
Values in military time.
Role of Secondary Factors in Sleep Symptoms
The associations between sleep symptoms and participant demographics and injury characteristics are displayed in Table 4. Only those sleep symptoms that differed significantly between groups were included in analysis. Older age was associated with earlier bedtime, less sleep duration, and increasing daytime sleepiness. Increasing time since injury was also associated with greater daytime sleepiness and poorer sleep quality in this group. With regard to injury severity, more severe injuries (on the GCS and PTA) were associated with increasing average sleep duration.
Significant Correlations Between Sleep Symptoms a and Demographic/Injury Characteristics of Patients With TBI.
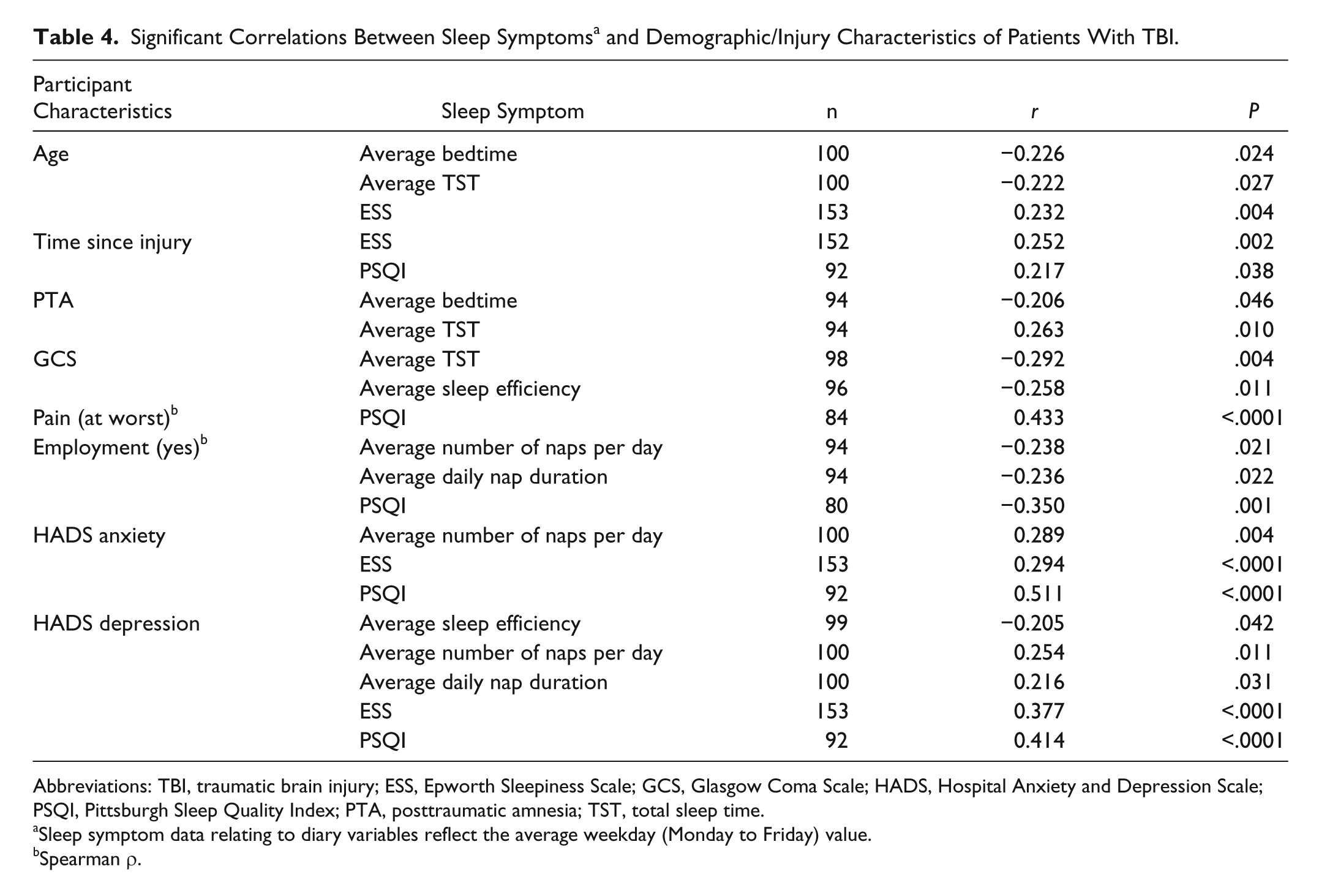
Abbreviations: TBI, traumatic brain injury; ESS, Epworth Sleepiness Scale; GCS, Glasgow Coma Scale; HADS, Hospital Anxiety and Depression Scale; PSQI, Pittsburgh Sleep Quality Index; PTA, posttraumatic amnesia; TST, total sleep time.
Sleep symptom data relating to diary variables reflect the average weekday (Monday to Friday) value.
Spearman ρ.
The impact of several secondary factors, namely employment status, level of pain, and mood, on group differences in sleep symptoms was also investigated (Table 4). Being in employed was associated with reduced daytime napping and nap duration, as well as increased sleep quality. The possibility that the group differences in sleep (diary) patterns during weekdays might be associated with differences in employment status was examined using 3-way mixed-model analysis of variance. We found no significant interaction effects of day, group, and employment status for any of the sleep diary measures, indicating that employment status did not affect sleep diary parameters differently between the groups for weekdays or weekends (P > .05).
Given that participants with TBI reported significantly greater levels of pain severity on the BPI than the noninjured control group, the role of pain in sleep symptoms was also examined further. As seen (Table 4), increased pain severity was associated with poorer sleep quality but no self-reported sleep (diary) outcomes (P > .05). Furthermore, in participants with TBI, increased pain severity was associated with greater anxiety (n = 87, r = 0.38, P < .0001) and depression (n = 87, r = 0.40, P < .0001). There was no association between pain severity and sleep quality, anxiety, or depression in the noninjured control participants (P > .05). The association between mood and sleep symptoms was also investigated. As displayed in Table 4, increased anxiety and depression were both found to be associated with increased daytime sleepiness, poorer sleep quality, and greater average number of naps per day. Increased depressive symptoms were additionally related to longer napping duration and poorer sleep efficiency.
Finally, a linear regression was conducted to identify predictors of sleep quality in patients with TBI. Pain, anxiety, and depression were selected as potential predictors of sleep quality in the TBI group. Preliminary analysis revealed no violation of the statistical assumptions. Pain (β = 0.268, P = .009) and anxiety (β = 0.369, P = .006), but not depression (β = 0.055, P = .670), were found to account for 32.8% of the variance in sleep quality, F(3, 79) = 12.84, P < .0001.
Discussion
This study has confirmed and extended the findings of Parcell et al 5 in highlighting the high rates of self-reported sleep changes in this sample of individuals with predominantly moderate to severe TBI, as compared with age- and sex-matched healthy controls. The TBI group reported significantly poorer sleep quality than the noninjured control group on the PSQI, with almost half of the sample obtaining scores indicative of poor sleep quality, as compared with 18% of controls, and a third obtaining scores indicative of insomnia, as compared with only 3% of controls. They also showed significantly higher levels of daytime sleepiness, with 24% reporting clinically significant excessive daytime sleepiness as compared with 12% of controls. This difference had only approached significance in the previous study by Parcell et al. 5 As in the study by Parcell et al, 5 sleep diaries revealed longer sleep onset latency, poorer sleep efficiency, and longer durations of daytime napping in participants with TBI, but in this larger sample they also reported significantly earlier bedtimes, greater total sleep duration, and more frequent daytime napping during weekdays than controls.
Examination of factors associated with sleep disturbance revealed that, as in the general population, older age was associated with a decline in some sleep parameters. Greater injury severity was associated with longer total sleep times. Unlike the previous study by Parcell et al, 5 those with milder injuries did not show higher levels of anxiety or depression and, perhaps as a result of this, milder injury was not associated with greater sleep disturbance in this study. Rather, it appears that more severe injuries increased the drive for sleep. Longer time postinjury was associated with poorer sleep quality and greater daytime sleepiness, suggesting that sleep disturbances generally worsened over time. Although employment status did not affect sleep diary parameters differently between the groups for weekdays or weekends, those who were working took fewer daytime naps during the week and reported better sleep quality.
Presence of both anxiety and depression was associated with poorer sleep quality, increased napping, and greater daytime sleepiness, and in the case of depression, poorer sleep efficiency. Pain was also associated with poorer sleep quality but not with other sleep diary parameters, although 25% of the TBI group identified pain as a factor they believed restricted their sleep. Pain was also associated with greater anxiety and depression. Pain and anxiety independently and significantly contributed to overall sleep quality. None of these relationships were evident in the control group.
In demonstrating the associations between sleep disturbance, mood, and pain, the study findings are consistent with those of other previous studies in addition to that of Parcell et al, 5 including that of Ouellet et al 6 and with the findings of a recent study by Fogelberg et al 41 who also found that anxiety, depression, and pain had a significant impact on self-reported sleep quality on the PSQI in a group of patients assessed 1 year following TBI. Using the cutoff of PSQI > 5, they determined that 44% of their sample had significant sleep problems, similar to the 49% in the current sample. The direction of these associations remains ill-defined. Symptoms of TBI, including mental slowness, fatigue, lethargy, emotional blunting, or emotional lability, 42 overlap with the symptoms of both depression and sleep disturbances, whereas irritability, poor concentration, and restlessness may be associated with TBI, anxiety, and sleep disturbance. While depression and anxiety each causes sleep disturbance, so too the experience of chronic sleep disturbance may cause lowering of mood and/or development of anxiety. Whichever the direction of this association, which may become clearer with monitoring in individual cases, these findings highlight the importance of assessing and addressing pain, anxiety, and depression when treating TBI-related sleep disturbances. The association of mild TBI with anxiety and depression, as demonstrated in studies by Parcell et al 5 and others, suggests that for individuals with mild TBI these factors may contribute to reported sleep disturbances.
The present study has provided additional detail regarding the nature of sleep changes relative to a demographically similar control group. The TBI group reported higher daytime sleepiness level, earlier bedtime, and longer total sleep time, which would be consistent with increased sleep need and sleep drive. However, the TBI group also reported increased sleep onset latency, poorer sleep quality, and more nighttime awakenings. Multiple interacting factors, including neurologic damage, behavior (daytime napping), mood, and pain, may contribute to this profile of sleep complaints. Perhaps the experience of greater daytime sleepiness leads to an increased tendency to nap, which in turn contributes to poor nocturnal sleep quality. We suggest that, in addition to treating anxiety, depression, and pain, it may be important to ensure that individuals with TBI regulate their napping to minimize the disruption of nighttime sleep. Understanding the increased need for sleep and accommodating it within the injured person’s lifestyle represents another important aspect of this adjustment.
These findings highlight the need for “objective” assessment of sleep in order to elucidate the factors contributing to subjectively reported sleep changes. The hypothesis that individuals with TBI experience an increased drive for sleep is supported by the finding of increased slow wave sleep in polysomnographic studies reported by Parcell et al 24 and Shekleton et al 43 after controlling for anxiety and depression. It also supports the contention that hypersomnia occurs following TBI, possibly caused by a loss of hypocretin neurons.13,14,18,19 Recent studies have also found lower levels of evening melatonin in people with TBI, which may contribute to the increased sleep onset latency and nighttime awakenings. There has been less evidence in support of changes in timing of dim light melatonin onset.43,44 Further research regarding the pathophysiological factors underpinning the self-reported sleep changes is important to further drive the development of efficacious treatments.
This study has some limitations. Individuals with TBI may not be as accurate as healthy individuals in recalling the quality and timing of their sleep. Moreover individuals with anxiety and insomnia may report greater sleep disruption than is evident on objective recording. 45 Both groups required regular reminders to complete the diary, and one cannot rule out the possibility that the recordings were affected by recall bias. The use of actigraphy or better still, polysomnography, as an objective monitor of sleep patterns was unfortunately not feasible in such a large study. Another recent study by these authors has suggested that actigraphy may be a useful supplement to self-report measures. 46
In summary, this study has contributed a large body of evidence confirming a subjectively reported decline in sleep quality and increased daytime sleepiness in individuals with TBI compared to healthy controls, with greater injury severity associated with a need for longer sleep time and most likely associated with greater need for naps. Increased sleep onset latency is also apparent. Pain, anxiety, and depression contribute significantly to poorer sleep quality. These findings should be used to guide the development of comprehensive treatment strategies that address the multiple, interrelated factors contributing to sleep disturbances in the TBI population.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JLP, DLP, KS, and MR report no conflicts of interest. SMWR reports that he has served as a consultant through his institution to Vanda Pharmaceuticals, Philips Respironics, EdanSafe, The Australian Workers’ Union, Rail, Bus and Tram Union, and National Transport Commission, and has through his institution received research grants and/or unrestricted educational grants from Vanda Pharmaceuticals, Takeda Pharmaceuticals North America, Philips Lighting, Philips Respironics, Cephalon, and ResMed Foundation, and reimbursements for conference travel expenses from Vanda Pharmaceuticals. His institution has received equipment donations or other support from Optalert, Compumedics, and Tyco Healthcare. He has also served as an expert witness and/or consultant to shift work organizations.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The projects described in the article received funding from the National Health and Medical Research Council (Project ID 334002) and Monash University.
