Abstract
Background
Combinatory intervention such as high-frequency (50-100 Hz) excitatory cortical stimulation (ECS) given concurrently with motor rehabilitative training (RT) improves forelimb function, except in severely impaired animals after stroke. Clinical studies suggest that low-frequency (≤1 Hz) inhibitory cortical stimulation (ICS) may provide an alternative approach to enhance recovery. Currently, the molecular mediators of CS-induced behavioral effects are unknown. Brain-derived neurotrophic factor (BDNF) has been associated with improved recovery and neural remodeling after stroke and thus may be involved in CS-induced behavioral recovery.
Objective
To investigate whether inhibitory stimulation during RT improves functional recovery of severely impaired rats, following focal cortical ischemia and if this recovery alters BDNF expression (study 1) and depends on BDNF binding to TrkB receptors (study 2).
Methods
Rats underwent ECS + RT, ICS + RT, or noCS + RT treatment daily for 3 weeks following a unilateral ischemic lesion to the motor cortex. Electrode placement for stimulation was either placed ipsilateral (ECS) or contralateral (ICS) to the lesion. After treatment, BDNF expression was measured in cortical tissue samples (study 1). In study 2, the TrkB inhibitor, ANA-12, was injected prior to treatment daily for 21 days.
Results
ICS + RT treatment significantly improved impaired forelimb recovery compared with ECS + RT and noCS + RT treatment.
Conclusion
ICS given concurrently with rehabilitation improves motor recovery in severely impaired animals, and alters cortical BDNF expression; nevertheless, ICS-mediated improvements are not dependent on BDNF binding to TrkB. Conversely, inhibition of TrkB receptors does disrupt motor recovery in ECS + RT treated animals.
Introduction
According to the American Heart Association (2009), 1 stroke is the leading cause of disability within the United States. Motor impairments are some of the most common disabilities and most persistent following stroke. 2 Recovery of motor function is heterogeneous, but several studies conclude that the more severely impaired an individual is early after stroke, the poorer their recovery is likely to be long term.2,3 A promising treatment approach to enhance motor recovery is to combine physical rehabilitation with electrical or magnetic stimulation of the ipsilesional motor cortex.4,5 Specifically, research studies indicate that epidural excitatory cortical stimulation (ECS) combined with rehabilitative practice with the impaired limb greatly improves motor recovery compared with rehabilitation alone in primates, rats, and humans.6-13 However, if initial impairments are severe, ECS does not further enhance motor recovery compared with rehabilitative training (RT) alone in preclinical stroke models. 10
Over the past decade, several studies indicate that contralesional inhibitory cortical stimulation (ICS) may be effective when ipsilesional ECS is not. 14 There is growing evidence that contralesional ICS may provide an effective alternative approach to enhance behavioral recovery and is an alternative strategy to manipulate ipsilesional excitability to improve motor outcome. Low frequency (≤1 Hz) repetitive transcranial magnetic stimulation (rTMS) of the nonstroke cortex can induce neural inhibition and reduce spasticity and hemiparesis in chronic stroke subjects.14,15 Inhibiting the unaffected motor cortex using 1-Hz rTMS enhances motor performance of the paretic hand in patients with subcortical chronic stroke and severe postoperative impairments. 16 Thus, it is possible that ICS over the nonstroke motor cortex combined with forelimb RT may improve recovery more effectively than ECS or RT alone when initial impairments are severe. While it is not known in these studies, and is debatable within the field, whether the high-frequency 100 Hz and slow frequency of 1 HZ produced excitatory and inhibitory neural effects, respectively, we use this nomenclature throughout the article.
Despite promising studies suggesting that brain stimulation may augment rehabilitative training after stroke, the underlying molecular mechanisms are unknown. It is likely that brain-derived neurotrophic factor (BDNF) may play a key role in recovery of function following stroke and also may be involved in brain stimulation related enhancement of recovery. In clinical studies, a reduction in BDNF in the acute phase after stroke is a factor for poor prognosis in terms of functional status of patients. 17 Animal studies demonstrate that inhibition of BDNF using the injection of antisense oligodeoxynucleotides impairs functional recovery and disrupts motor cortex reorganization. 18 BDNF binding to the tropomyosin-related kinase B (TrkB) receptor has a synergistic role in motor learning in that the effectiveness of rehabilitation is reliant on a certain threshold of BDNF expression. 19 Motor cortical reorganization is reliant on TrkB’s downstream effects on synaptic plasticity, which is vital for recovery of function 20 While there is evidence that BDNF is necessary for many forms of brain plasticity and BDNF has been strongly implicated in stroke recovery, it is not known whether BDNF is involved in CS-induced motor recovery, although some studies do suggest a role. Brain stimulation can increase activity-dependent expression of BDNF.21,22 Previous studies found that daily 5-Hz rTMS for 5 days improves BDNF-TrkB signaling in rats by increasing the affinity of BDNF for the TrkB receptors that results in greater density of TrkB receptors within the cortex. 21 Together, these studies suggest that BDNF is an essential molecule for motor improvement following stroke, is important to rehabilitative training–related motor improvements and cortical reorganization, and may be enhanced through some forms of brain stimulation.
In the following studies we compared the behavioral effects of ECS over the lesion cortex or ICS over the nonlesion cortex during RT in a rat model of stroke in which postoperative impairments were severe. To begin to uncover possible neurobiological mechanisms underlying CS-related recovery of function we investigated whether BDNF expression was altered after treatment (Study 1) and if BDNF binding to its tyrosine-phosphorylated (TrkB) receptor was necessary to induce motor recovery.
This study has 2 central aims: (study 1) compare the effectiveness of ICS + RT, ECS + RT, and RT alone to enhance motor recovery after stroke in rats with severe impairments and (study 2) to investigate if BDNF is a primary mediator of CS-induced behavioral recovery.
Methods
Experimental Overview
All rats were tamed by gentle handling and trained to reach with their preferred forelimb on the single-pellet reaching (SPR) task until they reached a minimum criterion of 50% successful reaches (Figure 1). Ischemic lesions were induced contralateral to the preoperatively trained forelimb and electrodes were implanted according to treatment group assignment. After 3 days of recovery, all animals underwent rehabilitative training (RT), which consisted of practice with the paretic limb on the SPR task 6 days per week for 3 weeks, concurrent with treatment group stimulation protocols. After 3 weeks of treatment, animals were euthanized and tissue was collected for BDNF enzyme-linked immunosorbent assay (ELISA) analysis. In study 2, we administered either ANA-12 (N-[2-[(2-oxoazepan-3-yl)carbomoyl]phenyl]-1-benzothiophene-2-carboxamide) or dimethyl sulfoxide (DMSO)-saline (vehicle) injections 36 hours poststroke and daily 3 hours before each rehabilitation session (Figure 1).
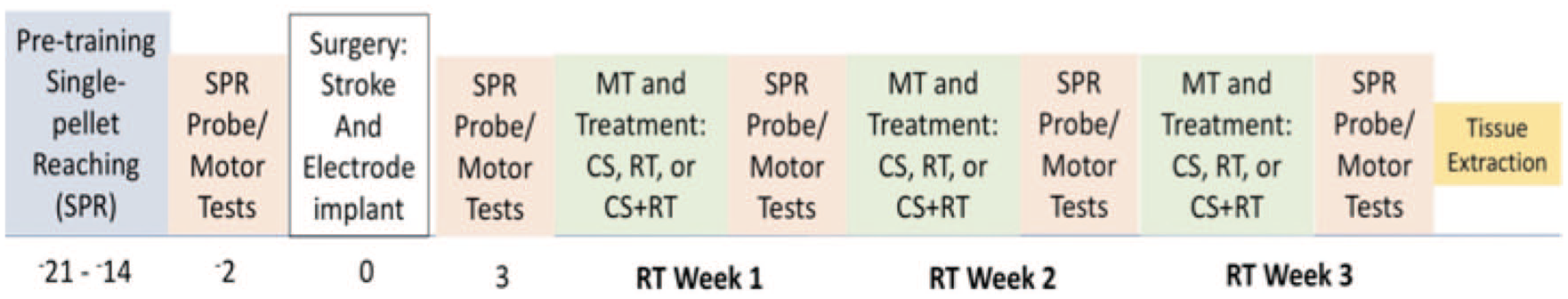
Timeline of study. Animals were trained on the single-pellet reaching (SPR) task to criterion and then assessed using this task before surgeries. Animals received stroke induction and postoperative performance assessments began 3 days after surgery in which the SPR test was given. Animals received rehabilitation for 21 days.
Animals
In study 1, 76 (4-9 months old, Envigo) and in study 2, 37 (4-month old, Envigo) Long Evans male rats were housed in pairs, received water ad libitum and were kept on a 12:12 hour light:dark cycle. Rats were maintained on a moderate restricted diet (15-20 g per day) to motivate performance on the reaching task. Surgeries were performed at 4 or 9 months. Animals were assigned randomly to treatment conditions with the exception that they were carefully matched for presurgery performance and initial severity of lesion induced impairments, assessed on the SPR task. All work was done in accordance with the Medical University of South Carolina Animal Care and Use Committee guidelines.
In study 1, animals were divided into the following 3 groups: (1) 100 Hz, excitatory stimulation over the stroke cortex concurrent with RT (ECS + RT n = 18), (2) 1 Hz, inhibitory stimulation over the stroke cortex concurrent with RT (ICS + RT, n = 19), (3) RT alone (NoCS + RT, n = 29). In study 1, 10 animals died during surgery or before the completion of the study.
In study 2, animals were divided into 6 groups: (1) excitatory stimulation + impaired forelimb RT + ANA-12 (ECS + RT + ANA-12, n = 5); (2) inhibitory stimulation + impaired forelimb RT + ANA-12 injection (ICS + RT + ANA-12, n = 5); (3) no stimulation + impaired forelimb RT+ ANA-12 (NOCS + RT + ANA-12 n = 5); (4) excitatory stimulation + impaired forelimb RT + VEHICLE (ECS + RT + VEH, n = 7); (5) inhibitory stimulation + impaired forelimb RT + VEHICLE injection (ICS + RT + VEH, n = 5); and (6) no stimulation + impaired forelimb RT + VEHICLE (NoCS + RT + VEH n = 5). In study 2, 5 animals died during surgery or before the completion of the study.
Surgical Procedure
Induction of Ischemia
Unilateral ischemic damage of the primary motor cortex (MC) was created by applying endothelin-1, a vasoconstricting peptide (American Peptide, Inc), to the cortical surface of the forelimb area of the sensorimotor cortex.23,24 Animals were anesthetized with an intraperitoneal injection of ketamine (110 mg/kg; Labcorp) and xylazine (70 mg/kg; Heartland Vet Supply). All animals received buprenorphine (0.5 mg/kg subcutaneously; Med-Vet International) prior to incision for pain. To induce severe reaching impairments, rats were placed in a stereotaxic apparatus, a midline incision was made and a craniotomy was performed over the motor cortex opposite the trained limb (Figure 2). Four- and 9-month old rats, received a craniotomy at 0.5 to 1.5 mm posterior and 3 to 4.5 mm anterior to bregma and 2.0 to 5.0 mm lateral to midline 7 and 3 or 4 µL of endothelin-1 (ET-1; 80 μM, 0.2 μg/μL in sterile saline; American Peptide, Inc) was topically applied to the exposed MC. For all surgeries, ET-1 was applied at approximately 1 μL/min, with a 2-minute wait between each 1μl of ET-1 application.7,10 After the final 1 µl of ET-1, the brain was left undisturbed for 10 minutes followed by electrode implantation.
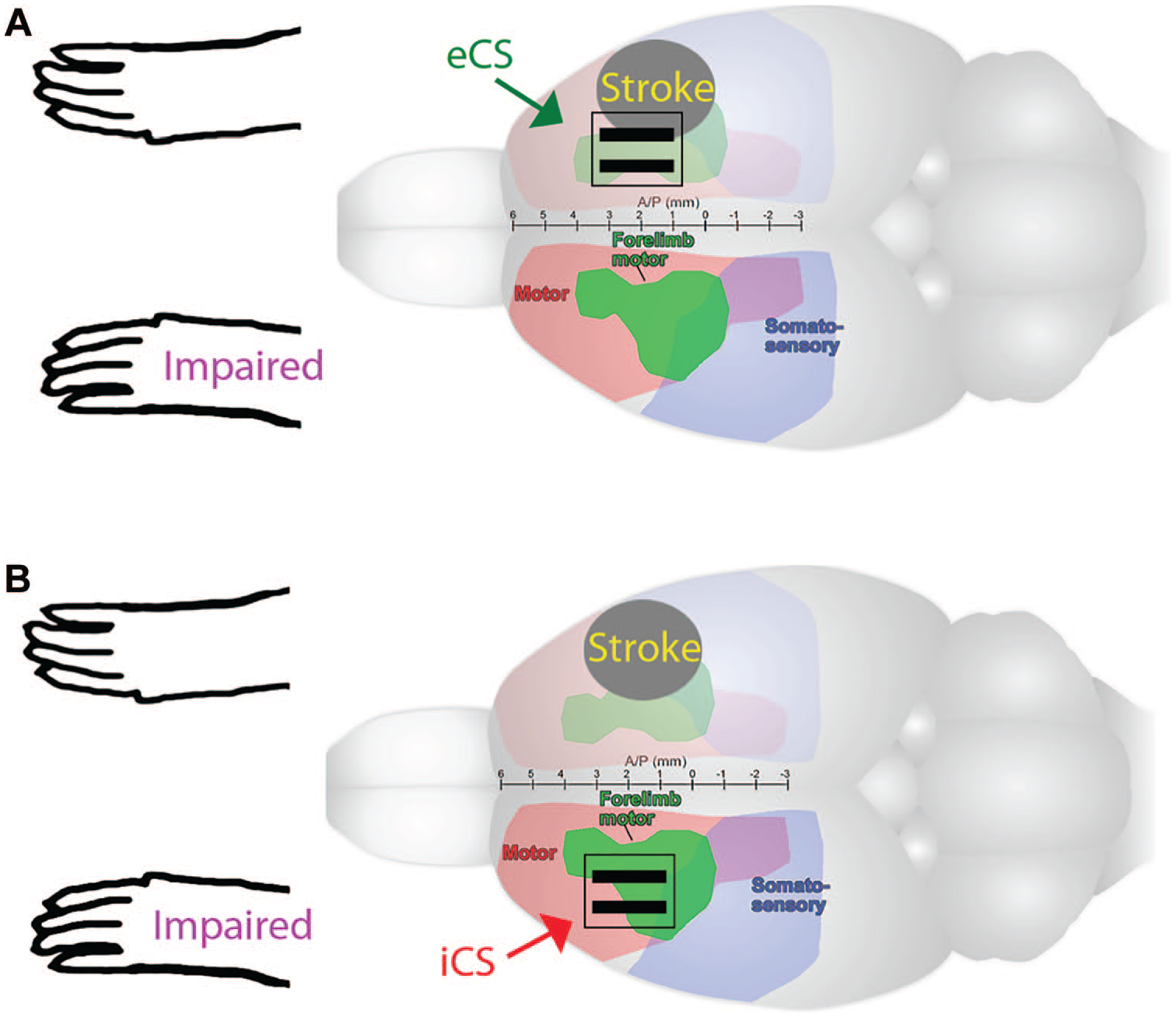
Electrode placement and cortical punches. Strokes were induced via topical application of endothelin-1, a vasoconstricting peptide, over the motor cortex opposite the preferred reaching limb. (A) Then the craniotomy was enlarged and a bipolar epidural electrode was placed over peri-injury motor cortex (eCS) or (B then a craniotomy was made over the forelimb area of the noninfarcted motor cortex and a bipolar epidural electrode was implanted (iCS). Three-millimeter cortical punches medial/lateral to the stroke area were obtained. Cortical punches were obtained on the M1 area of the motor cortex. For ECS and ICS, this is where the electrode is placed.
Lesion Severity
The intention was to create motor impairments that correlated with lesion size to determine whether inhibitory stimulation would improve forelimb motor recovery in either moderately or severely impaired young adult animals. Stroke size (determined by craniotomy coordinates) was not a reliable determinant of motor impairment. Animals, despite lesion area, resulted in both severe and moderate behavioral impairments as assessed with the SPR task. Several studies have determined that lesion size and lesion severity do not always correlate, making behavioral poststroke outcomes a more reliable predictor of functional recovery. Similar to previous studies, 10 experimental lesion size differences alone do not necessarily result in quantifiable differences in reaching impairments (at least as performed in these studies). At the end of the study, treatment groups were further divided based on forelimb impairments demonstrated on the SPR task analyzed treatment effects on these subgroupings. An impairment score was calculated with the formula: (PostOP − PreOP)/PreOP × 100. Severe impairments were defined as an 81% to 100% decrease in reaching success. Separating animals by their impairment levels based on their postoperative reaching success instead of craniotomy size and volume of ET-1 administered was determined to be a sensitive means divide animals into different levels of forelimb reaching impairments.
Electrode Implantation
Each bipolar electrode consisted of 1-mm wide by 3-mm long platinum strips mounted on a 3-mm by 3-mm silicone plate (Plastics One, Inc). The electrode was placed on the dura surface of the MC and positioned in order to optimize ability to evoke contralateral motor movements.
For all ECS and NoCS groups, the initial craniotomy was enlarged ~1 mm rostrally and medially to expose ipsilesion MC. The electrode contacts were positioned parallel to midline over remaining caudal and rostral forelimb and other motor areas of the MC (Figure 2).7,8 Platinum strips were oriented approximately parallel to midline (Figure 2).
Following all electrode placements, any exposed cortex was covered with a piece of gel-film. To secure the electrode to the skull, skull screws (Plastics One, Inc) were placed surrounding the craniotomy to create additional bonding surface. GC FugiCEM2 (GC America) and dental acrylic (Wave SDI Limited) and dental cement (GC America, Inc) were applied to both the skull and screws. After implantation animals were sutures and then given 3 mL of warm Ringers solution (subcutaneously) (Baxter Inc) during recovery. Animals were placed on heating pads (Sunbeam) after surgery and were given dampened standard food chow.
Rehabilitative Training Task and Forelimb Function Test
Single-Pellet Reaching Task
The SPR task has been described in detail previously.7,8,25,26 After shaping to determine limb preferences, rats were trained to reach with the preferred limb through a narrow window to retrieve a banana-flavored food pellet (45 mg, Bio-Serve) from a well 1 cm from the opening.
In both studies, rats were trained to reach a limb through a narrow window, grasp a pellet, bring the pellet back into the cage, and bring it to their mouths—a successful reach. Rats were evaluated for their demonstrated preferred reaching limb and then were trained, for approximately 3 weeks with that limb. Reaching performance was measured as the percent success of successful reaches [(total successes/total number reach attempts) × 100]. Prior to surgery, animals were trained for 30 trials over 15 minutes daily, until they achieved an average of 40% to 60% success rate. A trial began with the placement of a banana pellet in a well and ended when the animal either grasped the pellet and brought it to their mouth for consumption (successful reach), or dropped the pellet before reaching mouth (drop), or failed to grasp the pellet by knocking the pellet off the tray or missing the pellet after 5 consecutive attempts (misses). After each trial, a single banana pellet was dropped into the front of the chamber to “reset” the animals reaching posture.
Postsurgery, rats were allowed to recover for 3 days. On poststroke day 4, and then weekly, impaired forelimb function was assessed in probe trials. Probe trials consisted of assessing the average reaching success during 30 trials or 15 minutes of SPR. Data presented are the group mean success rates from weekly probe trials and were calculated as: (No. of successful pellet retrievals/No. of reaches) × 100.
On postsurgery day 5, rats practiced reaching with their impaired forelimb in the SPR task, a well-established form of rehabilitative forelimb training 27 for 6 d/wk for 3 weeks. Rehabilitative training trials consisted of 60 trials or 20 minutes, whichever came first.
Stimulation Parameters
Cortical Electrical Stimulation During Rehabilitative Training
Four days after lesion induction, excitatory or inhibitory stimulation was delivered for 21 days during practice on the rehabilitation SPR task. All animals were attached to stimulator cables and placed into the reaching chambers. The stimulation groups in all studies received stimulation that was delivered at 50% of that week’s movement threshold.7,9 Epidural stimulation was delivered as a train of bipolar, continuous pulses at 100 Hz for ECS or 1 Hz for ICS groups. Electrical stimulation was delivered through bipolar strip electrodes, as described above. For the high-frequency 100-Hz stimulation current was delivered as a train of continuous biphasic, charge balanced, and asymmetric pulses every 104 μs. Each biphasic, square pulse delivered current every 100 μs (first phase) and then the voltage was off for 9900 μs (second phase). Stimulation amplitudes were adjusted as needed to accommodate changes in movement thresholds. NoCS rats were attached to the stimulator cables, but no current was delivered.
Assessment of Movement Thresholds
The intensity of current delivered during RT was set at 50% of the lowest amount of current needed to induce involuntary motor movements or the motor threshold (MT). To assess MT, animals in the ECS and ICS treatment groups were attached to electrode leads, then they were placed into a transparent cylinder and electrical current was delivered in 3-second trains of 1 ms, 100-Hz bipolar current pulses, which were gradually increased by 5% increments until an involuntary movement was observed in the contralateral paw, head, or neck. The lowest current level to evoke a movement was recorded as that week’s MT. The weekly current level delivered during RT for that week was set at 50% of that week’s MT. In our hands, 1-Hz bipolar current pulses could not elicit involuntary motor movements. Thus, to keep consistency and uniformity in the way motor thresholds were obtained, we set ICS current thresholds based on the same parameters used for the ECS.
BDNF Enzyme-Linked Immunosorbent Assay
In study 1, After 21 days of RT (24 days postlesion) and within 1 hour of the last probe trial, all rats were anesthetized with an overdose of 0.8 cm3 Euthasol (Virbac Solutions) (400 mg/mL) and decapitated when breathing stopped. The right and left motor cortex regions were extracted and placed in vials and stored at −80 °C. Two cortical punches (3 mm each) were obtained from each hemisphere. To assess the concentration of BDNF in ipsilesion and contralesion motor cortex, the concentration of BDNF was measured using ELISA kits (R&D System). 28 The data are normalized to the control rat brains.
ANA-12 Injections
In study 2, animals received intraperitoneal injections of either ANA-12 (0.5 mg/kg in 17% DMSO in sterile saline, intraperitoneal, Sigma) or vehicle (17% DMSO in sterile saline, intraperitoneal) 4 hours prior to the brain stimulation and rehabilitation session. Animals were injected with an ANA-12 at a dose of 0.5 mg/kg solution of ANA-12 in DMSO because this was found to an optimal dose to induce inhibition of TrkB receptors in the brain for rats. 29 Injections were given 4 hours before rats performed the reaching rehabilitative task to allow for the maximum levels of cortical TrkB inhibition of 30%.
Statistical Analysis
All data are reported as group means with ± standard error of the mean (SEM). Data were considered statistically significant at P ≤ .05. Forelimb reaching success during probe trials on postischemia day 3 (postoperation) and after every treatment week (weeks 1, 2, and 3) was analyzed using repeated analysis of variance (rANOVA; IBM SPSS Statistics Data Editor Version 18.2). Post hoc comparisons (Tukey) were used to assess weekly differences between groups. Data from the BDNF ELISA for each hemisphere (ipsilesional and contralesional hemispheres) were analyzed in SPSS using independent t tests to compare each treatment to age-matched, nonlesion controls.
Results
Study 1: The Comparison of ECS + RT, ICS + RT, and RT Alone on Forelimb Recovery and BDNF Concentrations
Forelimb Functional Recovery
As seen in Figure 3A, all groups were trained with their preferred limb to criterion prior to ischemic damage. Following unilateral ET-1 lesion opposite the preferred limb, all groups demonstrated a significant impairment defined here as successfully reaching at less than 20%. There was a significant interaction effect of Group × Week, F(6, 189) = 2.363, P ≤ .05). We observed a main effect of week, F(3, 189) = 100.631, P ≤ .05, suggesting that all groups improved over the course of treatment. There was also an effect of group, F(2, 63) = 5.126, P ≤ .05. Post hoc tests indicate that ICS is overall a more effective treatment compared to ECS + RT (P ≤ .05) and noCS (P ≤ .05). As indicated by post hoc analysis, ICS + RT enhanced motor recovery compared with ECS + RT at week 1 (P ≤ .05), at week 2 (P ≤ .05) and both ECS + RT (P = .05) and NoCS + RT (P ≤ .05) at week 3. In these severely impaired animals, ICS + RT greatly enhanced reaching performance with the impaired forelimb compared with NoCS and ECS groups.
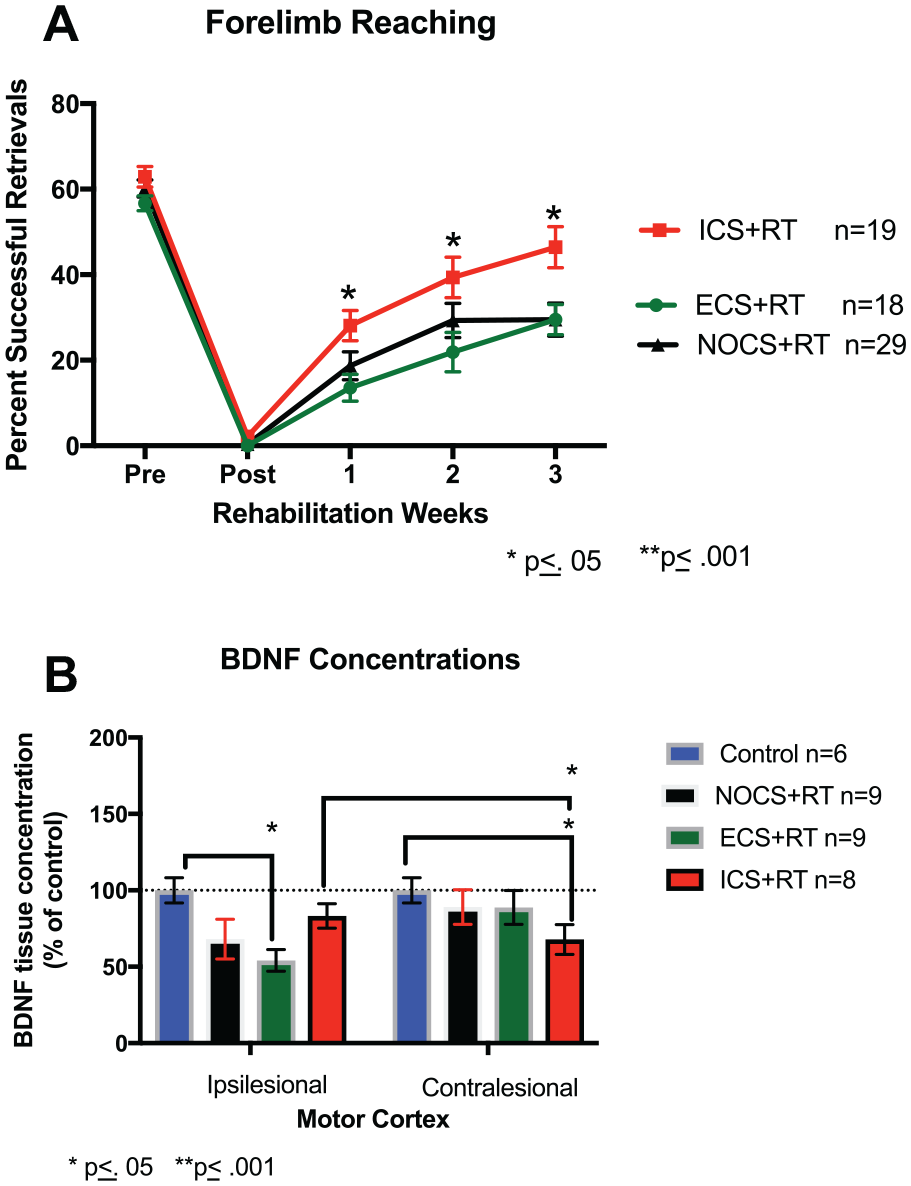
(A) percent successful retrievals and recovery scores ICS + RT improved more than ECS + RT at week 1 (P = .021), at week 2 (P = .035), and both ECS + RT (P = .026) and NoCS + RT (P = .013) at week 3. When all severely impaired animals are combined, ICS in this model of stroke demonstrates an additive treatment for animals that exhibit greater poststroke functional deficits. (B) Ipsilesional and contralesional BDNF concentrations cortical punches (3 mm) were taken from the primary motor and penumbra areas from stroke and nonstroke hemispheres. Concentrations of BDNF for severely impaired 4- and 9-month-old rats combined. One-way analysis of variance revealed a significant ipsilesional difference between control and ECS + RT (P = .0001). While not significant, there was a trend for an ipsilesional difference between control BDNF levels and NoCS + RT BDNF levels (P = .061). There was also a significant contralesional difference between control and ICS + RT (P = .022). There was also a significant difference between Ipsilesional ICS + RT levels and contralesional ICS + RT levels (P = .045). RT, rehabilitative training; ICS, inhibitory cortical stimulation; ECS, excitatory cortical stimulation; NoCS, no cortical stimulation; BDNF, brain-derived neurotrophic factor.
Motor Cortical Stimulation Induces BDNF Changes in both the Ipsilesional and Contralesional Hemisphere
To determine if ICS + RT and ECS + RT differentially alter BDNF protein expression in the motor cortex, we performed an ELISA for BDNF (Figure 3B). After 3 weeks of treatment, there were no significant differences between ipsilesional ICS + RT BDNF levels and nonstroke control cortical BDNF levels, t(10) = 0.299, P > .05; indicating a possible role of ICS as an additive treatment to RT that increases BDNF levels to prestroke levels. However, ECS + RT treated animals had significantly reduced BDNF expression in the ipsilesional cortex compared with nonlesion controls, t(10) = −5.896, P ≤ .05, indicating ECS + RT’s inability to return BDNF levels to baseline after 3 weeks of treatment after stroke. In the contralesional cortex, ICS + RT resulted in a significant reduction in BDNF expression compared with controls, t(13) = −2.606, P < .05, indicating that ICS + RT may downregulate BDNF production in the contralesional hemisphere.
Study 2: Effects of TrkB Inhibition on CS- and RT-Related Motor Recovery
Forelimb Functional Recovery
ANA-12 inhibition
In study 2, we investigated whether BDNF binding to its TrkB receptor is a key mediator of CS or RT treatment in severely impaired animals. Prior to daily CS and RT treatment, animals were injected with either vehicle or ANA-12, a ligand that reduces BDNF binding to the TrkB receptor. We observed a main effect of week, F(3, 78) = 33.564 P ≤ .05, suggesting that all groups improved over the course of treatment. Overall, there were no statistically significant differences (rANOVA) between groups over the course of treatment weeks, F(15, 78) = 1.051, P ≤ .05.
TrkB inhibition does not affect forelimb motor recovery in rats receiving just rehabilitative treatment, F(3, 30) = 1.033, P > .05 (Figure 4A) or inhibitory stimulation treatment, F(3, 24) = 0.212, P > .05 (Figure 4B). For animals that received ECS, there was a deviation between animals that received either vehicle or ANA-12 injections, F(3, 24) = 2.087, P > .05 (Figure 4C). While there was no statistically significant group by week effects, there was a significant difference between groups on reaching success when tested at week 3 (P = .05) and an overall group effect, F(1, 8) = 5.827, P ≤ .05. A recovery score was calculated (week 3-postop) for animals receiving ECS. As seen in Figure 4D, treatment with ANA-12 significantly reduced recovery of reaching success with the impaired forelimb in groups that received ECS + RT treatment, F(1, 5) = 10.042, P ≤ .05. This suggests that BDNF may play a role in motor recovery in the presence of ECS.
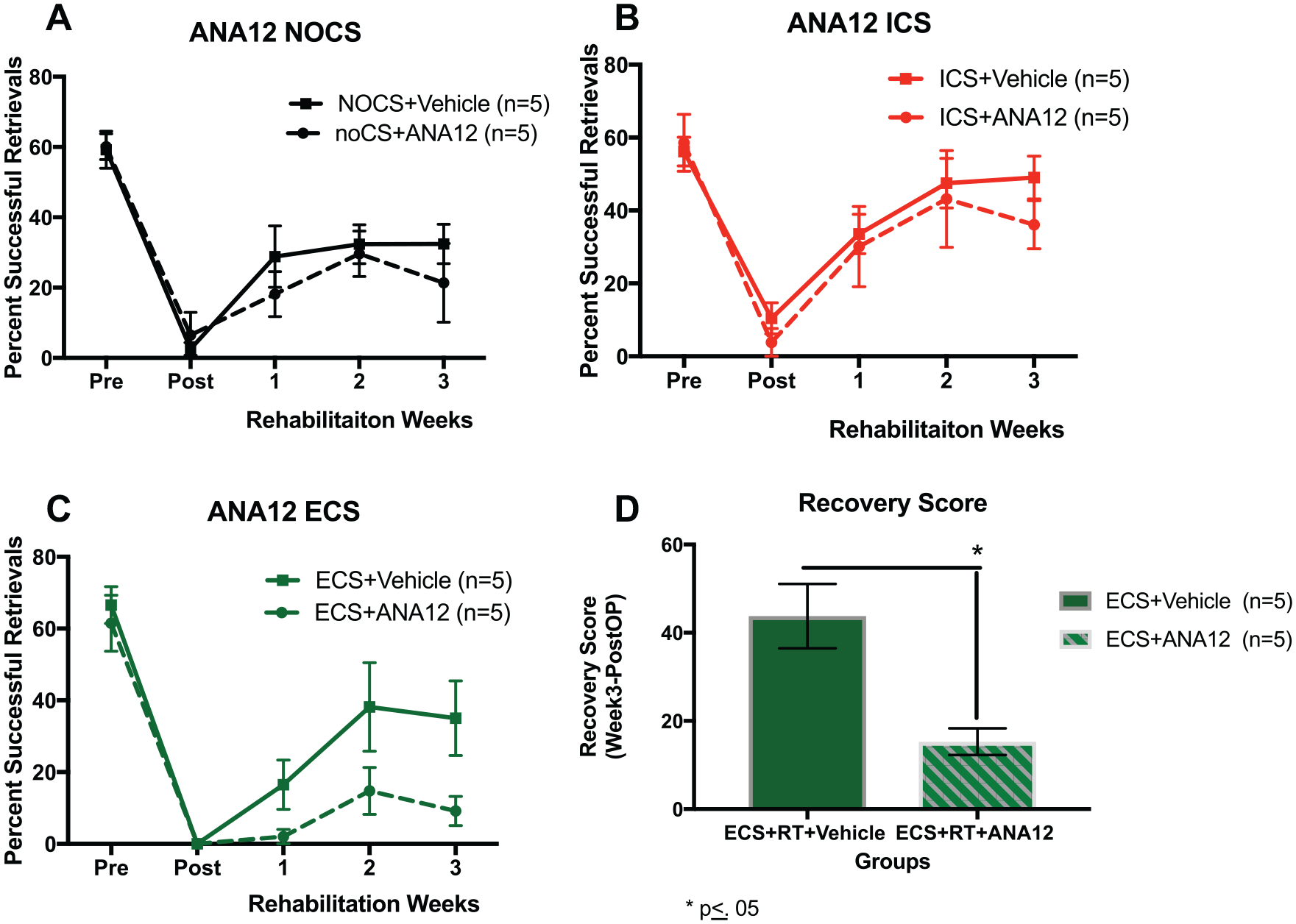
Percent successful retrievals and recovery scores: vehicle and ANA-12 animals. Rats received ECS or ICS given concurrently with the single pellet reaching task for 3 weeks following ischemic injury. ECS results in no added benefit in aged animals. (A) NoCS. There was no significant interaction effect of Group × Day, F(2, 9) = 4.32, P = .655, and there was no significant interaction of week (P = .347). (B) ICS. There was no significant interaction effect of Group × Day, F(2, 7) = 0.583, P = .570, but there was a significant interaction of week (P = .031). (C) ECS. There was a mean difference at week 3 between groups: F(1, 8) = 5.329, P = .05). (D) ECS mean difference. There is a mean difference at week 3 following ECS + RT between vehicle and ANA-12. A recovery score was calculated (week 3-PostOP for animals receiving ECS. There was a mean difference at week 3 between groups: F(1, 5) = 10.042, P = .025. RT, rehabilitative training; ICS, inhibitory cortical stimulation; ECS, excitatory cortical stimulation; NoCS, no cortical stimulation.
Recovery in all TrkB-inhibited (ANA-12) groups
We then compared the different CS treatment groups within the subgroup that received ANA-12 injections (Figure 5). Animals that received ECS + ANA-12 appear to be most detrimentally affected by a TrkB inhibition. Overall, there was a main effect of week, indicating that all groups improved over time, F(3, 36) = 9.952, P ≤ .05. While there was no significant interaction effect of Week × Group, F(6, 36) = 1.1062, P > .05, there was a group effect, F(2, 12) = 4.133, P ≤ .05, indicating a difference in ICs + RT treatment compared with ECS + RT. Post hoc comparisons (Tukey) revealed ICS + ANA-12 had greater motor recovery than ECS + ANA-12 (P ≤ .05).
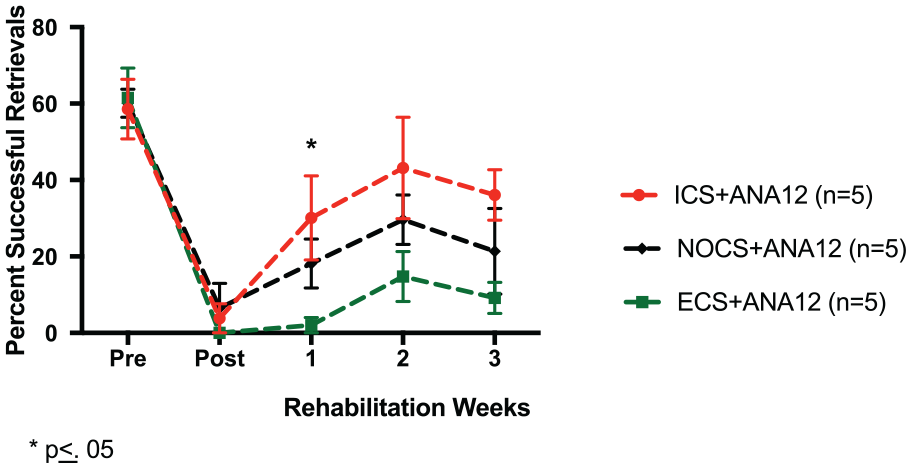
Percent successful retrievals: All ANA-12 groups. A comparison of the different CS treatment groups within the subgroup that received ANA-12 injections. At week 1, there was a significant difference between ICS + ANA-12 and ECS + ANA-12 (P = .05). Post hoc comparisons (Tukey) reveal no significant differences between NoCS + ANA-12 and ECS + ANA-12 (P = .311) or NoCS + ANA-12 and ICS + ANA-12 (P = .512) at week 1. ICS, inhibitory cortical stimulation; ECS, excitatory cortical stimulation; NoCS, no cortical stimulation.
Discussion
Overview
These studies investigated whether inhibitory or excitatory cortical stimulation would enhance the effectiveness of RT in animals with severe initial impairments after a unilateral, focal cortical stroke. As discussed in more detail below, delivering inhibitory (1 Hz) stimulation over the noninfarcted motor cortex concurrent with impaired forelimb reach training (RT) significantly improved motor recovery compared with RT alone or RT concurrent with excitatory stimulation, in severely impaired animals. Similar to previous findings, excitatory ipsilesion stimulation did not further enhance RT effectiveness in animals with severe motor impairments compared with RT alone. 10 Our data also suggest that BDNF expression in the lesion cortex was not altered by RT alone or inhibitory stimulation when combined with RT. Counterintuitively, inhibitory CS also resulted in significantly reduced BDNF expression in the noninfarcted motor cortex compared with lesioned cortex.
On the other hand, after 21 days of 100 Hz bipolar stimulation of the ipsilesion motor cortex, there is a significant reduction in BDNF expression in the remaining motor cortex compared with nonstroke controls. Inhibiting BDNF binding to its TrkB receptor, via the ANA-12 ligand, resulted in greater impairment in impaired forelimb reaching success only in subjects that received RT concurrent with ipsilesion 100-Hz stimulation.
As discussed more fully below, these results suggest that while BDNF may not be a key mediator of ICS enhancement of RT-related functional motor improvements.
One-Hertz, Inhibitory Stimulation in Severely Impaired Rats
We report that inhibitory stimulation over the noninfarcted motor cortex combined with impaired forelimb reach training (ICS + RT) resulted in a significant improvement in forelimb functional recovery compared with ECS + RT or RT alone. Animal studies have shown that the contralesional cortex may be highly involved in recovery of function, as indicated by greater contralesional cortical activation in strokes that produce more damage to the ipsilesional hemisphere.30,31 Previous studies also suggest that when the lesion is large, and thus likely to produce greater impairments, the contralesional hemisphere is important in reestablishing lost functions. 32 Following unilateral stroke, damage-related alterations in transcallosal inhibition can alter the balance of intercortical activity such that the ipsilesional cortex becomes hypoexcitable and the nonlesion cortex is hyperexcitable. 33 In humans, after a unilateral cortical stroke impaired limb activity results in a greater BOLD (blood oxygen–level dependent) signal in the nonlesion hemisphere compared with the lesion hemisphere. 34 Based on these prior studies, it is our hypothesis that 1-Hz stimulation over the contralesional cortex may improve the effectiveness of RT by reducing the nonlesion cortex hyperexcitability and thus reducing transcallosal inhibition of the stroke cortex. However, we did not measure cortical excitability in these studies and thus these are hypothetical explanations. Future studies are needed to investigate this potential explanation.
We found that 1-Hz CS delivered over the contralesional motor cortex during daily RT improved forelimb function and increased BDNF levels in the ipsilesional cortex to control, nonlesion levels. The ICS over the contralesional cortex likely releases ipsilesional inhibition, resulting in reduced activation of ipsilesional GABAergic interneurons and subsequently dis-inhibiting pyramidal cells. 35 This in turn may permit greater RT-induced activation of motor cortical neurons and thus may result in greater BDNF expression. It is also possible that BDNF is not related to the behaviors being tested or be related to glial cell expression. Glial cells in resting states release neurotrophic factors to help maintain homeostasis. It is possible that after injury, once glia become chronically activated, they no longer contribute to BDNF production, thus dampening any potential effects from ANA-12. However, this was not measured in these studies and further studies are needed. These studies do add to a growing body of literature attempting to elucidate when the contralesional hemisphere may provide a more positive role in stroke recovery.
BDNF as a Requirement for Poststroke Recovery
One of the questions we sought to investigate was the relationship between motor recovery, treatment, and BDNF levels in the motor cortical regions. We found that ICS + RT induced improvements in functional motor recovery which was related to changes in both ipsilesional and contralesional BDNF concentrations. Within the ipsilesional cortex, there is not a significant difference between ICS+RT and control (nonstroke animals) BDNF levels. On the other hand, we also found that contralesional BDNF levels in the ICS +RT group were reduced compared with contralesional control BDNF levels. With further investigation, it is plausible that inhibitory stimulation to the nonstroke contralesional cortex could be downregulating the over excitation within the cortex and thus BDNF production, resulting in greater excitation and BDNF synthesis in the ipsilesional cortex. This is important since previous studies have already reported that motor rehabilitative tasks are only effective in improving recovery when BDNF levels are increased. 36 Our findings fit with previous findings that relate motor recovery with increases in BDNF levels and further suggests that effective rehabilitative motor training increases BDNF concentration levels.
Inhibiting BDNF and Its Effects on Treatment
We investigated whether the slight elevations in BDNF concentrations and improvements in functional recovery after treatment would be diminished if BDNF binding to its TrkB receptor was inhibited by pretreating animals with the TrkB antagonist ANA-12. While we did not verify the rate of TrkB receptor antagonism in these studies, based on several previous investigations ANA-12 administered at this dose inhibits 30% of TrkB binding. We found that TrkB inhibition negatively influences animals receiving ECS + RT and not ICS + RT or NoCS + RT. It is possible that ipsilesional high-frequency stimulation delivered early after ischemia could exacerbate neuronal vulnerability in an area already undergoing cell death and dysfunction and may further reduce stroke-related declines in BDNF expression. 31 Thus, the combination of excitatory stimulation and reduced BDNF/TrkB signaling could produce a more detrimental environment and limit the ability for RT-related recovery to occur.
Alternatively, low-frequency stimulation over the contralesional (noninfarcted) cortex may not further exacerbate lesion-related cellular dysfunction. The contralesional hemisphere is not subject to the same diminished neuronal plasticity and neurotrophic factors that are in the ipsilesional cortex, making inhibition of TrkB not as detrimental in producing deficits in motor recovery in animals that are receiving stimulation to this area.
Conclusions
We hypothesized that unlike excitatory ipsi-infarct cortical stimulation, inhibitory stimulation combined with impaired forelimb rehabilitation would significantly improve motor recovery in animals with severe impairments. We also hypothesized that effective CS may affect or be related to cortical BDNF. Using a combination of sensitive behavioral tasks and assays to investigate the role of inhibitory stimulation on behavioral outcome following stroke, we report that inhibitory stimulation of the nonlesion motor cortex concurrent with forelimb rehabilitative training enhances forelimb motor recovery in severely impaired rats following experimental stroke. Our data also suggests that inhibitory stimulation alters BDNF cortical expression and may be related to ICS-related motor improvements. We also report that the addition of ipsilesion high-frequency stimulation combined with partial inhibition of TrkB receptors early after stroke, may exacerbate lesion induced motor impairments or inhibit RT-induced motor recovery. Together, these studies do suggest that ICS was more effective than ECS as an adjunctive treatment for stroke recovery. We also provide promising data supporting the role of BDNF in motor recovery and indications that the location and frequency of CS may play a role or be dependent on BDNF expression.
Footnotes
Authors’ Note
This article was prepared while DeAnna L. Adkins was employed at the Medical University of South Carolina. The opinions ex-pressed in this article are the author’s own and do not reflect the view of the National Institutes of Health, the Department of Health and Human Services, or the United States government. The two authors Aitana Rizzo and Kern Howard are undergraduates.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by the NIH BluePrint D-SPAN Award (F99/K00), and MUSC’s NIH training grants (R25 GM072643, TL1 TR001451 and TL1 TR000061) SK). This work is also supported by The Delaware-CTR ACCEL Institutional Development Award (IDeA) from the (NIGMS/NIH U54-GM104941), NC NM4R (P2CHD086844), and COBRE in Stroke Recovery (P20 GM109040) (DLA).
