Abstract
Background and Aims. White blood cell (WBC) and neutrophil counts (NC) are common markers of inflammation and neurological stroke damage and could be expected to predict poststroke outcomes. Objective. The aim of this study was to explore the prognostic value of early poststroke WBC and NC to predict cognition, mood, and disability outcomes at 3 and 12 months poststroke. Methods. Routine clinical analyses WBC and NC were collected at 3 time points in the first 4 days of hospitalization from 156 acute stroke patients. Correlations using hierarchical or ordinal regressions were explored between acute WBC and NC and functional recovery, depression, and cognition at 3 and 12 months poststroke, after covarying for age and baseline stroke severity. Results. We found significant increases in NC between <12 hours and 24 to 48 hours time points (P = .05). Hierarchical regressions, covaried for age and baseline stroke severity, found that 24 to 48 hours WBC (P = .05) and NC (P = .04) significantly predicted 3-month cognition scores. Similarly, 24 to 48 hours WBC (P = .05) and NC (P = .02) predicted cognition scores at 12 months. Increases in WBC and NC were predictive of increased cognition scores at both 3 and 12 months (positive recovery) though there were no significant associations between WBC and NC and disability or depression scores. Conclusions. Routine acute stroke clinical laboratory tests such as WBC and NC taken between 24 and 48 hours poststroke are predictive of cognition poststroke. It is interpreted that higher rapid immunological activation in the acute phase is an indicator for the trajectory of positive stroke recovery.
Acute focal brain ischemia is characterized by rapid decrease in oxygen and adenosine triphosphate (ATP) availability leading to impairment of ionic pumps compromise of basement membranes and cellular swelling. 1 Such neuronal excitotoxicity immediately induces a sterile inflammatory response mediated by damage associated molecular patterns, increased permeability of the blood-brain barrier, 2 and local infiltration by peripheral blood leukocytes. 3 Thus, the interaction of immune and inflammatory pathways has long been conceptualized as one of the mechanisms underlying long-term stroke recovery.4,5 Indeed, it has also been proposed that recovery of neurological function after stroke may be mediated by the initial infiltration of leukocytes into the infarcted area and/or with the associated immune and inflammatory response within the first to second week poststroke.5,6 However, general clinical blood tests such as the complete blood count or full blood examination 7 and component leukocyte or white blood cell (WBC) count that are prioritized following suspected stroke admission 8 are predominantly utilized to investigate the differential diagnoses presenting as stroke mimics such as the presence of infections or hypoglycemia 9 rather than as a marker of tissue injury, recovery, and prognosis.
Previous studies in poststroke populations have shown that persistent leukocytosis (WBC > 11 × 109 cells/L in adults) 48 hours after admission is associated with higher baseline scores on the National Institute of Health Stroke Scale (NIHSS) and worse functional recovery at discharge and longer lengths of stay (mean = 10.4 days vs mean = 6.6 days). 10 Other studies have also shown that even after controlling for age and baseline stroke severity, relatively elevated leukocyte count (regardless of reference range defined leukocytosis 11 ), is a predictor of poststroke NIHSS severity, modified Rankin scale scores (mRS), and stroke volume.12,13 Indeed, controlling for age and baseline stroke severity is extremely important to reveal changes in poststroke immune responses while addressing the natural progression of immunosenescence in elderly adults 14 and the large effect of acute stroke on the peripheral immune system. 15 However, knowing when the most clinically and scientifically valuable time points post stroke onset are to measure blood products such as WBC and neutrophil count (NC) for the prediction of poststroke recovery, has not yet been established. Studies examining the acute trajectory of poststroke WBC 16 and NC 17 have shown that cell counts increase rapidly within the first 0 to 2 days poststroke and then fall between 3 and 7 days. Indeed, both the timing and amplitude of acute poststroke immune responses have been found to correlate with stroke severity within 24 hours of symptom onset 18 and also recently have been reported to correlate with improvement on NIHSS scores over the first week. 19
A recent 2017 review and international consensus recommendation concludes that there is a relative lack of evidence for blood-based biomarkers to predict stroke outcomes and recovery, 20 when poststroke recovery is defined using the mRS disability and functional recovery scale. 21 Yet this measure does not include mood or cognitive outcomes, despite the fact that they are commonly reported as unmet needs by stroke survivors. 22 Recent studies examining the association of WBCs, neutrophils, and lymphocytes have determined that increased acute neutrophil-to-lymphocyte ratio is often predictive of poststroke depression. 23 Indeed, despite accumulating evidence for the use of acute routine investigations of blood products in the association and prediction of stroke outcomes, these measures have yet to be adopted by clinical guidelines. 24
The role of immune markers in depression and cognition regardless of stroke involves a complex pathophysiological system linking immune dysregulation, chronic inflammation, and neurobehavioral changes. 25 Previous meta-analyses have identified that increased levels of inflammatory mediators such as interleukin 1, interleukin 6, and C-reactive protein are associated with increased symptoms of depression. 26 Indeed, chronic inflammation has the potential to mediate neurotoxic response dysregulation of the glutamatergic system and upregulation of reactive oxygen and nitrogen species, with structural changes such as hippocampal and prefontal cortex matter loss. 25 Furthermore, there are sickness behavior similarities between subjects facing immune challenge (such as infection) and depression symptoms in many mammalian species. 27 The link between cognition and immunity has not been as well established as the physiological basis of immunity in depression. 27 Multiple large prospective studies examining elderly adults found both positive and nonsignificant associations between cognitive outcomes and leukocyte count28,29 or inflammatory markers.29,30 Studies in stroke immunity and cognition have identified that increases in myelin basic protein antibodies that respond to central nervous system antigens are associated with longitudinal cognitive decline.4,31
We aimed to explore the associations between measures of acute poststroke WBC and NC acquired within the first week of hospitalization and longer-term poststroke recovery at 3 and 12 months defined by clinical scales for functional recovery (mRS), depression (Montgomery-Åsberg Depression Rating Scale [MADRS]), 32 and cognition (Montreal Cognitive Assessment [MoCA]). 33 Investigation of predictive associations between routine blood tests conducted in the acute phase to clinical outcomes at 3 and 12 months poststroke was possible using the longitudinal START stroke cohort. 34 It was hypothesized that increases in hematological markers of immune function within 0 to 12 hours, 24 to 48 hours, and 3 to 7 days poststroke would be associated with poorer poststroke functional, cognitive, and depressive outcomes at 3 and 12 months poststroke. Furthermore, we investigated from the acute samples available, the optimal timing for blood draw and analysis that would be the most useful predictor of outcomes. Knowledge of an association between routine bloods tests, such as WBC and NC, combined with measures of recovery in the first year poststroke, may assist with longer term prediction and highlight the importance of treating early immunological dysfunction to reestablish immune and inflammatory homeostasis for better long-term outcome.
Method
Participants
Patients were recruited as part of START-EXTEND (Neuroscience Trials Australia: NTA 0901, NTA 0901, NCT01580839) 35 and START-PrePARE (NTA 0902) 34 cohort studies. These studies recruited 219 patients from a group of participating hospitals in Australia between 2011 and 2015. Briefly, the START-EXTEND study investigated the clinical characteristics of extension of the thrombolysis treatment window and included patients within 9 hours of stroke onset, and the START-PrePARE cohort allowed for recruitment of patients with stroke onset up to 3 days with the purpose of examining predictors of recovery, including the functional brain networks associated with longitudinal stroke recovery. 16 A subset of 156 participants (104 male and 41 female) aged 71 (SD = 12.78) from whom clinical scores and blood test results (Table 3) were available were selected for the current study.
Blood Tests
Routine blood tests for a range of hematological markers were conducted at participating hospital sites by onsite commercial pathology services. Venipuncture was conducted at baseline (<12 hours), 24 to 48 hours, and 3 to 7 days poststroke, as outlined in the START study protocols. 34 Data presented are drawn from electronic patient records and standardized to units recommended by the Royal College of Pathologists Australia. 11 Only total WBC and NC were consistently available from data obtained from the START cohort.
Baseline Demographics and Clinical Characteristics
Patient demographics were obtained by semistructured interview at baseline admission as part of inclusion in the START cohort studies. 34 Routine demographic and clinical assessments included age, gender, stroke risk factors, and regular blood pressure monitoring. Stroke severity was assessed at baseline using the NIHSS according to clinical trial protocols. 36 Baseline patient characteristics including risk factors and physiological measures were obtained.
Clinical Measures
A series of standardized assessments of neurological function, disability status, cognition, and depression were conducted at 3 months and 12 months poststroke by double-blinded trained health professionals and encompassed several domains of poststroke recovery. Descriptive details of clinical measures and scoring criteria are presented in Table 1.
Clinical Assessments Administered at 3 Months and 12 Months Poststroke.
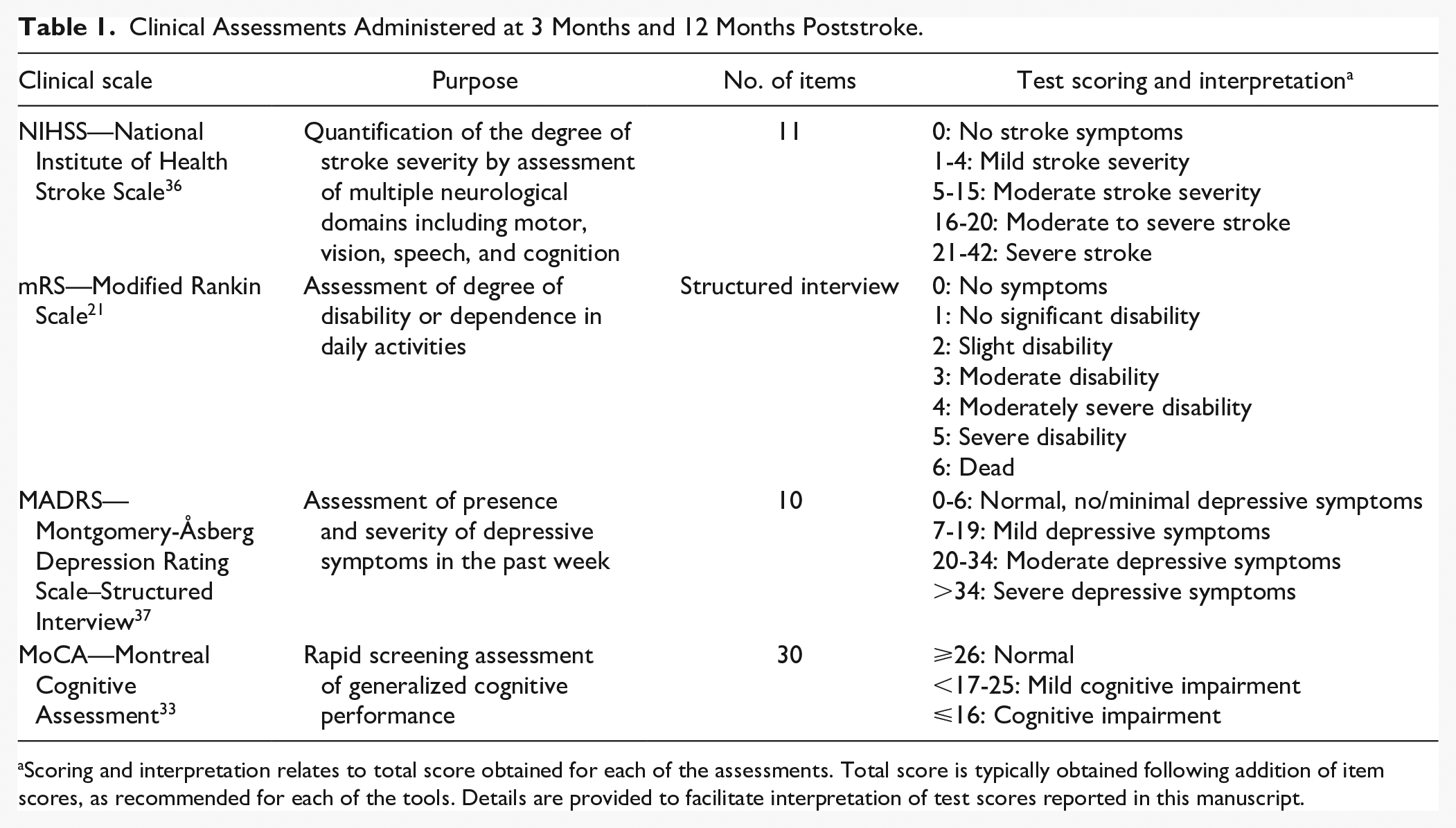
Scoring and interpretation relates to total score obtained for each of the assessments. Total score is typically obtained following addition of item scores, as recommended for each of the tools. Details are provided to facilitate interpretation of test scores reported in this manuscript.
The MADRS 37 is a measure of depressive symptoms and has been widely used in major depressive disorders 38 and poststroke research. 39 The MADRS was administered using the structured interview guide, which is reported to have higher reliability than the self-report method. 37 A score above 8 on the MADRS for depressive symptom screening has been proposed as the normative cutoff in stroke cases 32 for increased sensitivity (>0.80) and acceptable specificity (0.74) compared to the original recommended score of 12. 37 The mRS was used to describe functional disability of patients. 21
The MoCA 33 is a screening measure of multiple cognitive domains and has been shown to be more sensitive to poststroke cognitive impairment than the Mini-Mental State Examination. 40 Normative cutoffs to determine cognitive impairment in poststroke research have been suggested at 19 to 22 for acute administration and 20 to 27 in the chronic phase. 41
Data Analyses
A Pearson’s correlation analysis was first conducted to independently assess the associations between acute immune cell counts and longitudinal functional recovery, mood, and cognition status. A post hoc power analysis showed that our sample size (n = 156) was adequate to detect a medium correlation (r = .30) with a power of 0.8 and α level of .05 (n pwr = 84). Normality assessment using the Kolmogorov-Smirnov (K-S) test was conducted after outlier removal at ±>2.5 interquartile range (IQR). The K-S demonstrated that normality was within acceptable bounds for parametric tests. Repeated-measures ANCOVAs were used to test for significant differences between immune cell counts at 0 to 12 hours, 24 to 48 hours, and 3 to 7 days time points poststroke, and t tests were used to test changes between clinical scores at 3 and 12 months. Post hoc ANCOVA analyses were used to determine changes between blood test time points.
Pearson’s partial correlations were used to explore the associations between acute laboratory blood tests (ie, <12 hours, 24-48 hours, and 3-7 days) and mRS, MADRS, and MoCA at 3 and 12 months. To control for confounding factors such as age and baseline stroke severity (previously identified as being associated with WBC count and/or recovery42,43), these factors were entered as covariates in the partial correlational matrix to allow for examination of the unique contribution of WBC and NC to poststroke clinical recovery. Hierarchical regressions were conducted following significant partial correlations to determine the changes in explained variance from the addition of immune cell counts to stroke and severity predicting MADRS and MoCA scores at 3 and 12 months. As this was an exploratory study, an α level of .05 was used to detect statistical significance after adjusting for covariates, for each of the 3 outcomes.
Results
Baseline patient characteristics including risk factors are reported in Table 2. Based on individual patient 44 NIHSS scores (mean of 8.47, range = 0-32, SD = 6.49, median = 6, IQR = 3-6) the neurological severity of ischemic stroke in this cohort at the baseline assessment (<12 hours) was primarily mild to moderate. At 3 and 12 months poststroke, mean stroke severity was still in the mild range (Table 3). Quantification of recovery outcomes showed relatively mild to moderate clinical scores on the mRS 21 and MADRS 45 at 3 and 12 months (Table 3). However, the trajectory of recovery for cognitive outcomes poststroke was poor, with a significant decrease of MoCA scores from 3 months to 12 months post stroke (Mdiff = 0.96, t = 2.72, degrees of freedom [df] = 137, P = .001), with a small effect size (d = 0.23). Clinically, this represents a trend from cognitive functioning in the unimpaired range toward mild cognitive impairment. 46 Stroke severity symptoms assessed using the NIHSS were significantly increased between 3 months and 12 months poststroke (Mdiff = 0.64, t = 3.34, df = 180, P = .001, d = 0.21), indicating deterioration in neurological symptoms.
Patient Baseline Characteristics.
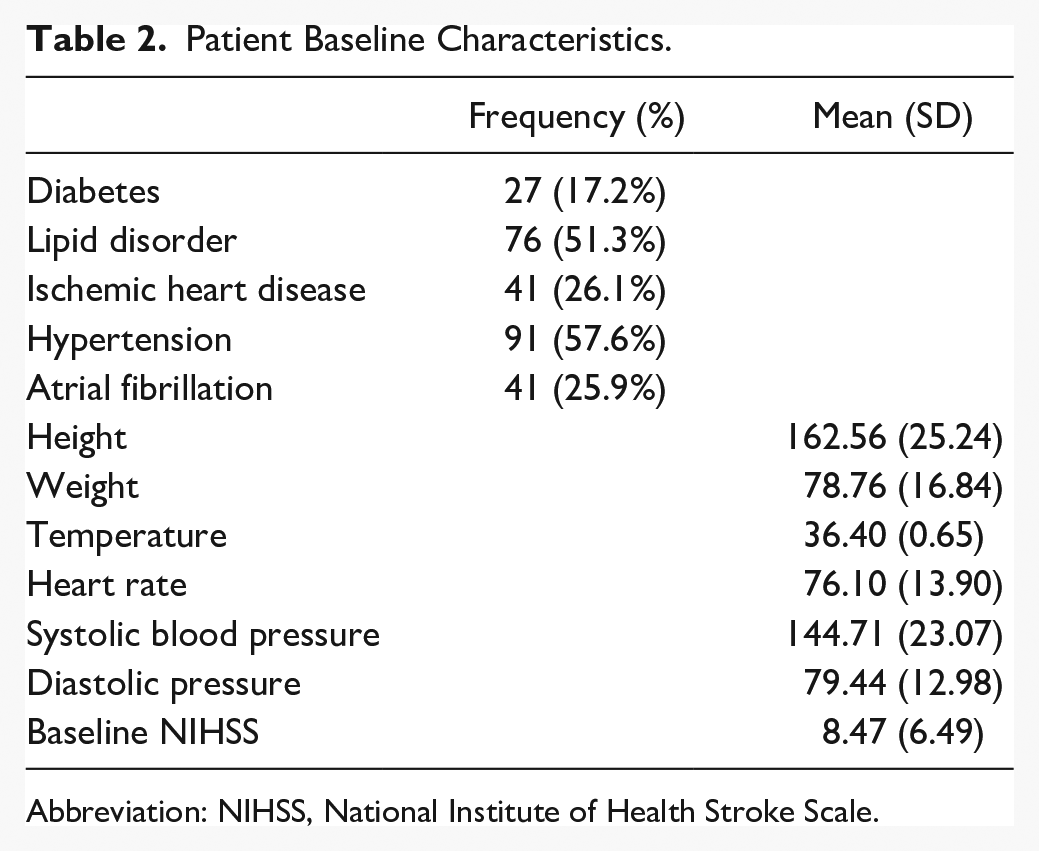
Abbreviation: NIHSS, National Institute of Health Stroke Scale.
Clinical Scores at 3 Months and 12 Months Poststroke (n = 157).
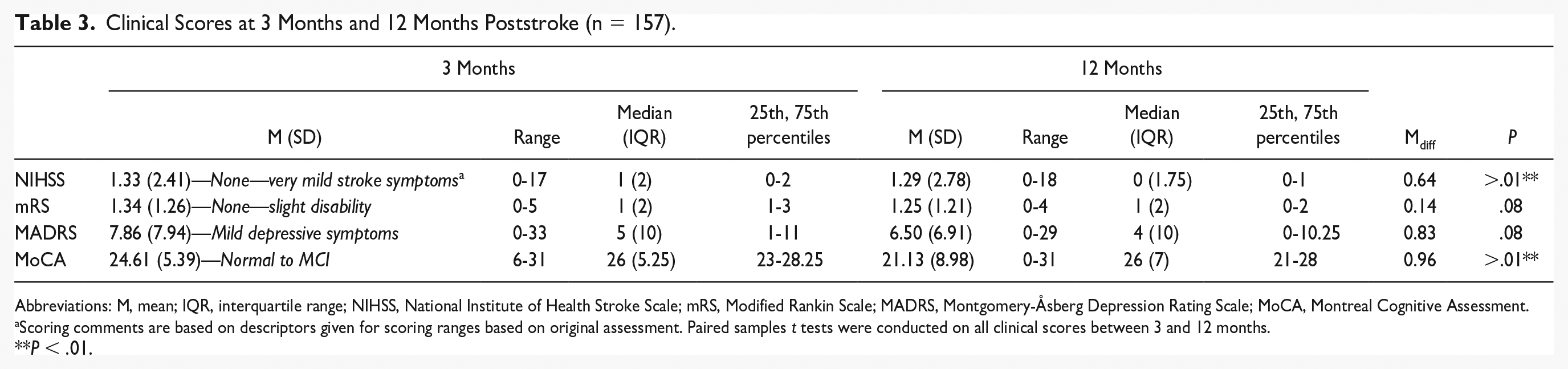
Abbreviations: M, mean; IQR, interquartile range; NIHSS, National Institute of Health Stroke Scale; mRS, Modified Rankin Scale; MADRS, Montgomery-Åsberg Depression Rating Scale; MoCA, Montreal Cognitive Assessment.
Scoring comments are based on descriptors given for scoring ranges based on original assessment. Paired samples t tests were conducted on all clinical scores between 3 and 12 months.
P < .01.
Details of acquisition, including the average time from stroke onset to blood draw for each time point, and counts of WBC and neutrophils at <12 hours, 24 to 48 hours, and 3 to 7 days, are provided in Table 4. WBC and NC showed an increasing pattern from <12 hours to 24 to 48 hours poststroke, followed by a trend back toward <12 hour levels at 3 to 7 days (Figure 1). A repeated-measures ANCOVA for NC at <12 hours, 24 to 48 hours, and 3 to 7 days, with age and stroke severity as covariates, was significant F(2, 76) = 4.52, P = .01, with post hocs showing that changes between <12 hours and 24 to 48 hours were significantly different (Mdiff = 0.88, 95% confidence interval [CI] = [−0.38, 1.78], P< .05).
Routine Blood Tests: Details of Acquisition and Counts at 0 to 12 Hours, 12 to 48 Hours, and 3 to 7 Days.
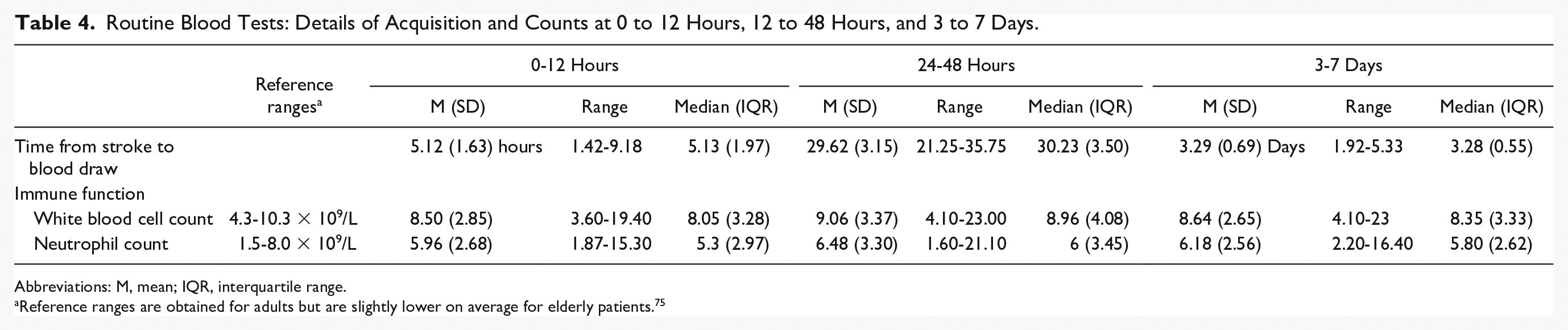
Abbreviations: M, mean; IQR, interquartile range.
Reference ranges are obtained for adults but are slightly lower on average for elderly patients. 75
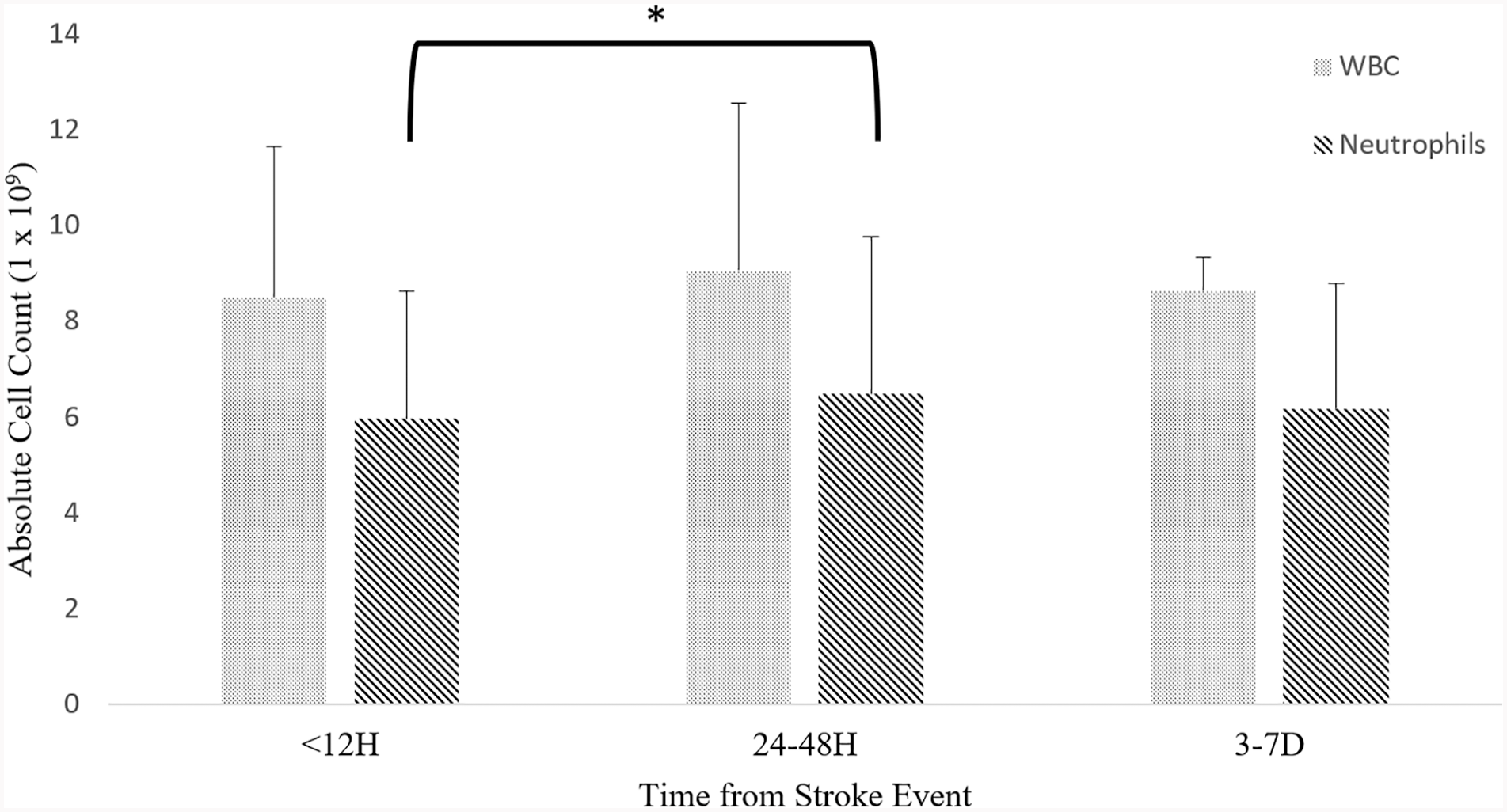
Trend of mean white blood cell count and neutrophil count at <12 hours, 24 to 48 hours, and 3 to 7 days. Error bars represent SD.
A partial correlation matrix was used to explore the relationship between the early blood tests at <12 hours, 24 to 48 hours, and 3 to 7 days and clinical measures at 3 months and 12 months, adjusted for age and baseline NIHSS severity score (Table 5). Depression symptoms as measured by the MADRS were not significantly associated with any acute blood WBC or NC counts.
Correlation Between Immune Cell Counts (Multiple Acute Times) and Clinical Outcome Scores at 3 Months and 12 Months Poststroke (Covaried for Age and Baseline Stroke Severity) a .
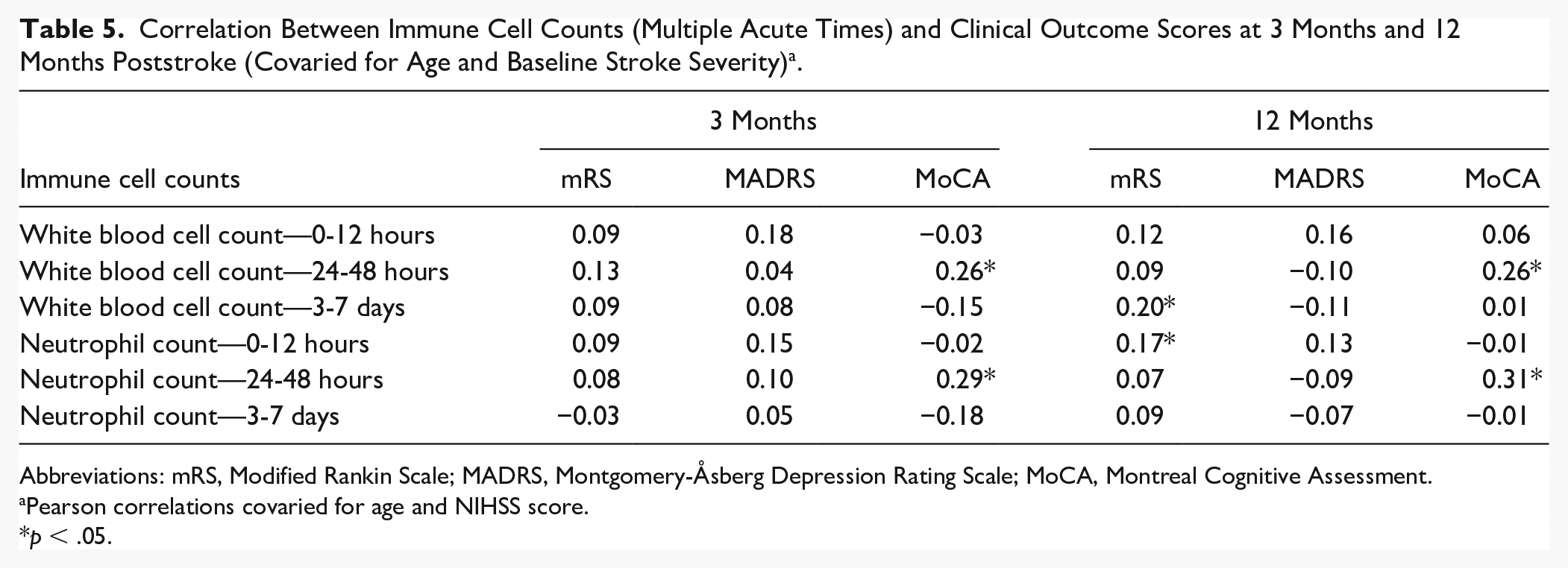
Abbreviations: mRS, Modified Rankin Scale; MADRS, Montgomery-Åsberg Depression Rating Scale; MoCA, Montreal Cognitive Assessment.
Pearson correlations covaried for age and NIHSS score.
p < .05.
As follow-up analyses to the significant correlations, hierarchical regressions were conducted between WBC and NC counts at <12 hours, 24 to 48 hours, and 3 to 7 days with 3-month (Table 6) and 12-month (Table 7) poststroke MoCA scores, with age and baseline NIHSS entered as covariates for all models in Step 1. In prediction of 3-month MoCA scores, the regression model with age and baseline NIHSS entered as Step 1, showed medium effect sizes (R2 = .20, F[2, 54] = 6.84, P = .002). The addition of 24- to 48-hour WBC counts (B = 0.49, P = .05) to Step 1 improved explained variance with a small effect size (R2 change = .06, F[1, 53] = 3.88, P = .05). Similarly, the addition of 24- to 48-hour NC counts (B = 0.45, P = .04) to Step 1 improved explained variance with a small effect size (R2 change = .06, F[1, 53] = 4.71, P = .04). In prediction of 12-month MoCA scores, the regression model with baseline NIHSS entered as Step 1 showed large effect sizes (R2 = .34, F[2, 56] = 14.70, P = >.001). The addition of 24- to 48-hour WBC counts (B = 0.74, P = .05) to Step 1 improved explained variance for 12-month MoCA scores with a small effect size (R2 change = .05, F[1, 55] = 4.08, P = .048). The addition of 24- to 48-hour NC counts (B = 0.45, P = .02) to Step 1 improved explained variance with a small effect size (R2 change = .06, F[1, 55] = 5.87, P = .02). As expected, age and baseline stroke severity predict a significant portion of 3- and 12-month MoCA scores, generally in the expected direction of cognitive decline. In contrast, given the unstandardized B of regressions models with the addition of immune cell counts, an increase of 1 × 109 WBC count and NC at 24 to 48 hours poststroke is predictive of an approximate 0.53 increase in MoCA scores at 3 and 12 months.
Hierarchical Regressions for White Blood Cell Count and Neutrophil Count at 24 to 48 Hours With 3-Month MoCA Scores, Controlled for by Age and Baseline National Institute of Health Stroke Scale.
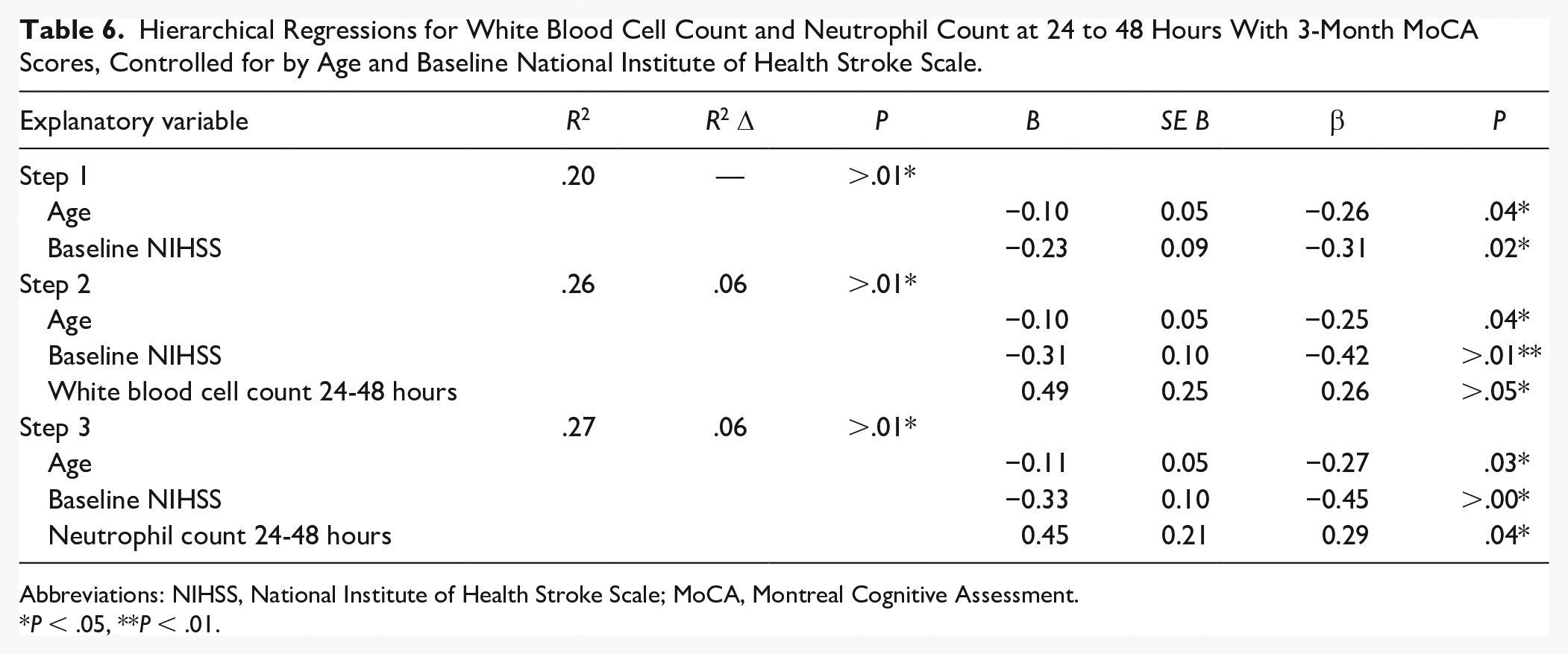
Abbreviations: NIHSS, National Institute of Health Stroke Scale; MoCA, Montreal Cognitive Assessment.
P < .05, **P < .01.
Hierarchical Regressions for White Blood Cell Count and Neutrophil Count at 24 to 48 Hours With 12-Month MoCA Scores, Controlled for by Age and Baseline National Institute of Health Stroke Scale.
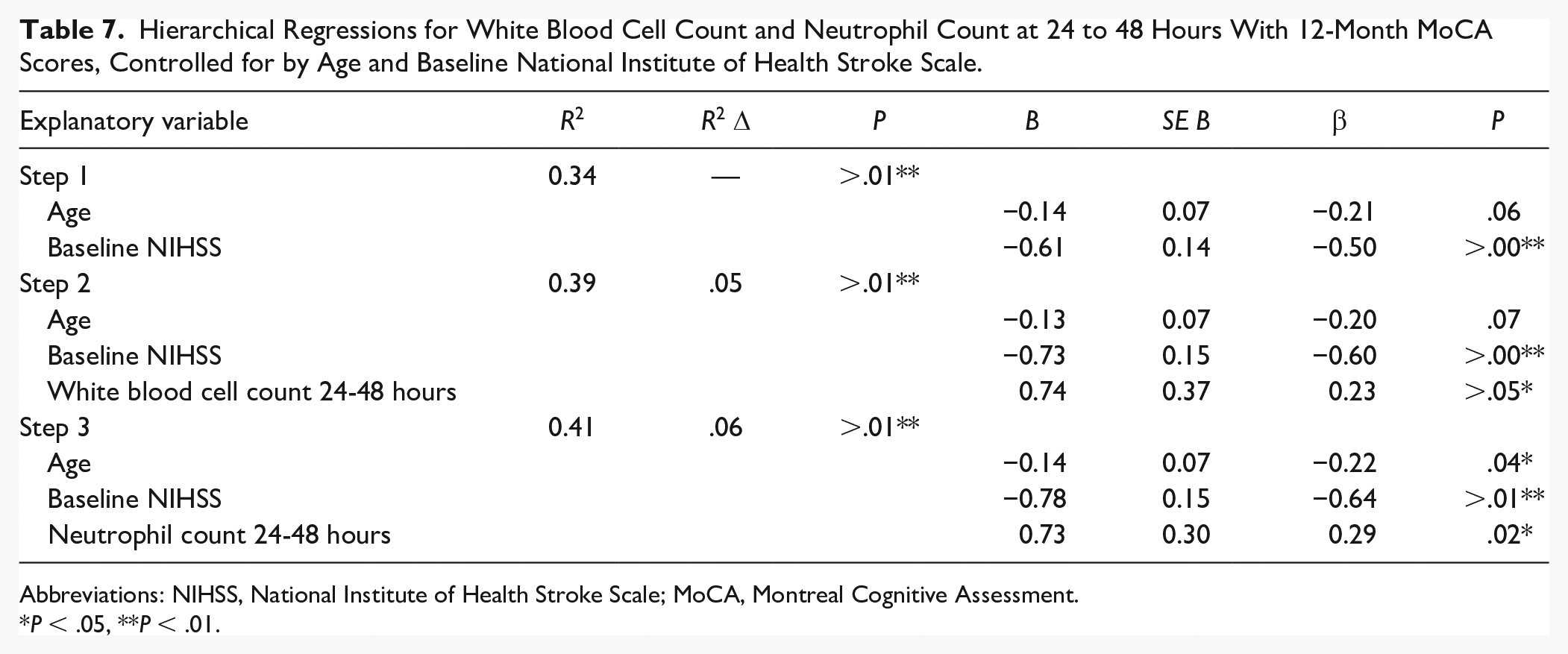
Abbreviations: NIHSS, National Institute of Health Stroke Scale; MoCA, Montreal Cognitive Assessment.
P < .05, **P < .01.
An ordinal regression, with age and baseline NIHSS as covariates, between WBC count at 3 to 7 days and mRS scores at 12 months found an odds ratio of 1.02, 95% CI = [0.89, 1.18], Wald χ2(1) = .09, which was not significant (P = .76). An ordinal regression was conducted with age and baseline NIHSS entered as covariates with NC at <12 hours predicting mRS scores at 12 months found a significant odds ratio of 1.13, 95% CI = [1.00, 1.27], Wald χ2(1) = 3.91, P = .048.
Discussion
In this longitudinal cohort study, we have investigated the associations between hospital-based hematological markers of immune function taken within 1 week poststroke and outcomes of clinical functioning and recovery at 3 months and 12 months post vascular event. A significant increase in NC was found between <12 hours and 24 to 48 hours timepoints. The partial correlations and hierarchical regressions revealed that elevations in poststroke WBC and NC at 24 to 48 hours are associated with better cognitive outcome at 3 and 12 months poststroke, independent of age and baseline stroke severity. Although the convergence of regression models showed that both WBC and NC are predictive of increased MoCA scores at 2 separate time points, these data should be interpreted with caution given the relatively weak but consistent effect sizes and small sample sizes. To date, there has only been a single study showing that WBC and/or NC early (within 24-48 hours) are correlated or predictive of better cognition at longitudinal time points such as 3 or 12 months poststroke. 47 Rather, as recently indicated, the majority of research has examined novel biomarkers such as interleukins and apolipoproteins in relation to poststroke cognitive impairment and recovery (see review in Wahul et al 48 ).
The significant regression of immune-related white blood cells to cognitive outcomes can be interpreted as evidence for the involvement of immune system activation following cerebral ischemia in acute poststroke having long-term effects on functional recovery. 2 As shown in the hierarchical regression models, baseline stroke severity and age account for a large amount of variance in relation to longitudinal MoCA scores and are in the expected direction of predicting cognitive decline. Indeed, demographics, functional factors, 49 and lesion characteristics 50 often predict worsened cognitive outcomes poststroke. Interestingly, the addition of 24- to 48-hour WBC and NC to these models significantly explained additional variance and while the regression models overall were predictive of cognitive decline, WBC and NC as individual predictors were found to be in the unexpected direction of predicting positive cognitive outcomes. Previous studies examining the link between blood-based biomarkers and poststroke cognition have largely focused on the association of inflammatory markers to impairment. 48 Increases in serum C-reactive protein have been reported to predict lowered global cognitive function within the first month of stroke. 51 Furthermore, prolonged erythrocyte sedimentation rates have been associated with cognitive decline. 52 However, few studies have described a blood-based biomarker for positive cognitive outcomes, especially where blood-based measures are acquired early in admission and investigated in association with long term cognition.
The biophysiological correlates of worsened poststroke outcome are usually understood in the context of ongoing peripheral and central nervous system inflammation 53 and improved outcomes, including cognition, are understood in the context of neural repair and plasticity. 54 Although it is difficult to speculate on the functional neuro-immunomodulation of inflammation and neural repair in the context of gross peripheral leukocyte counts, acute innate immune responses in stroke have been suggested to be both inflammatory and neuroprotective. 55 Indeed, more rigorous studies with better defined timing for blood draw and increased sample size representative of more severe stroke cases are required to replicate the positive associations between immune cell counts and cognitive function found here.
Our study did not find any significant correlations between acute immune cell count and depressive symptoms, when adjusted for age and stroke severity, despite previous research suggesting an inflammation- and immune-based pathogenesis for poststroke depression.56,57 The significant prediction of NC at <12 hours to mRS at 12 months is in agreement with previous studies that show an association between increased acute leukocyte counts and poor mRS functional recovery outcomes at both 3 months 58 and 12 months. 18 These findings may appear inconsistent with our results that found an association with increased WBC and NC with positive cognitive outcomes in the same cohort. Although functional recovery might be expected to be associated with cognition, the mRS is perhaps not sensitive enough to adequately capture cognitive function or neuropsychological impairments in stroke patients in comparison to the MoCA. 59
Our results also found that an increase in NC at <12 hours poststroke is predictive of poorer functional recovery at 12 months. An increasing NC from 0 to 48 hours poststroke has been previously reported 17 and may reflect changes in blood-brain barrier (BBB) permeability due to disassembly of the tight junctions by hypoxia-reoxygenation 2 and disruptions to both peripheral and central nervous system immune/inflammatory pathways (see review in Brouns and De Deyn 60 ). Although blood tests at 12 hours poststroke could not be compared to prestroke reference levels, this systemic change is confirmation of several known physiological responses to stroke such as penumbral excitotoxicity and perivascular edema 61 and resulting increased glymphatic perfusion. 62 Indeed, these results may be related to the molecular signaling timeline following neuronal oncosis driven by mitrochondrial and associated bioenergetic impairment (ie, reduced availability of ATP63,64). Indeed, acute edema and inflammation following stroke-induced excitotoxity would be expected to lead to previously activated autophagy and necrosis pathways, 65 and be associated with the abrupt upregulation of immune activity that has been predicted to occur around 24 to 48 hours poststroke in conjunction with BBB permeability and be related to peripheral innate immunity as pro-inflammatory cytokines and other endocrine responses influence leukocyte recruitment across a vulnerable BBB. 66 The timeline of leukocyte levels presented here closely resembles dynamic contrast-enhanced magnetic resonance image defined BBB permeability pattern within the first 3 days of stroke. 2 Previous studies within this acute stage have shown a phasic increase in permeability-surface area product from 6 to 48 hours while subsequently returning to 6 hour levels beyond 48 hours poststroke. 2 Similarly, the return of WBC and NC to <12 hour levels at 3 to 7 days may suggest a shift in the pathophysiological cell death cascade, including functions such as cell necrosis, vascular leakage, cerebral edema, and maintenance processes such as the clearance of cellular debris, ionic osmoregulation, and restoration of energy processes—processes that have been well described in animal models.5,63 The apparent reduction in WBC and NC may also be neuroprotective, as persistent antigen-specific immune cell recruitment and activity in the brain is predictive of poorer outcome and mortality. 4
Considering the ionic, biochemical, and molecular cascades accompanying stroke damage, 67 the leukocyte pattern observed may be reflective of the mild nature of stroke in this cohort and an ongoing trajectory to positive poststroke recovery that has not been observed in other studies. 42 Our regression analyses of acute immune cell counts predicting increased MoCA scores also suggest that trajectories of later poststroke recovery could be based on the extent of the early leukocyte response and the subsequent attempt to restore immune cell balance within the first week. The timing of the leukocyte infiltration response may also be indicative of the duration of BBB permeability, vessel occlusion, and therefore increased tissue damage, with longer duration strokes showing an earlier leukocyte infiltration. 68 On the contrary, poor poststroke recovery is commonly followed by continuing immunosuppression that may continue beyond 3 months.5,69 This trend toward immunosuppression supports the prevalence and timeline of poststroke infections such as upper respiratory tract infection, pneumonia, and urinary tract infections that commonly present toward the end of the first week.70,71 Indeed, the timeline for immunoreactivity seems to precede optimal trajectories for patients undergoing early physiotherapy rehabilitation at 15.6 days poststroke, followed by a period of 4 weeks of rapid functional recovery. 72
There are several limitations associated with the methodologies in this study. This study has examined a cross section of stroke patients obtained from a longitudinal stroke cohort study examining thrombolysis outcomes and stroke recovery outcomes: it is not inclusive of all patients as may be the case in national level data registries.34,35 The nature of the original inclusion criteria and ability to participate in a year-long study may bias the characteristics of the sample, in particular the mild stroke severity, depressive symptoms, and cognitive outcomes. Age and baseline NIHSS are factors that have been shown in previous studies to correlate highly with clinical measures at 3 months42,73 and thus were selected to adjust for in these analyses. It is acknowledged, however, that there are other baseline measures such as infection status or atherosclerosis in relationship to WBC and NC that could be addressed as covariates. Furthermore, there may be large variability in the amplitude of immune responses 17 given the wide acute blood sampling times achieved in this study. It is also noted that this study did not collect lymphocyte cell counts that could be used to explore the neutrophil to lymphocyte ratio which has been suggested to be a better measure of acute inflammation than immune cell counts alone. 74 In order to provide more robust evidence for predictive value of immune cell counts in stroke recovery, future studies should examine national- or hospital-level data registries with similar blood test protocols. Furthermore, sample sizes should be greatly increased, in contrast to the relatively low sample size used in this exploratory study.
Conclusion
Blood tests are routinely prescribed as an investigative tool in acute stroke management but are seldom used as clinical tools to predict poststroke recovery in the longer term. We have found that increased WBC and neutrophils count within the first week of stroke are consistently independent predictors of better cognitive outcomes at 3 and 12 months. In conjunction with the observed trends in immune cell counts, these data suggest that the degree and timing of the peripheral immune response in the acute phase is likely to play a role in setting the trajectory for long-term recovery. Further studies should be conducted on acute blood tests and their role in similar long-term clinical outcomes, with the goal of eventual integration into stroke guidelines and best practice.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge financial support for the conduct of the research from the Australian Commonwealth Scientific and Industrial Research Organisation (CSIRO) Flagship Collaboration Fund through the Preventative Health Flagship scheme, and support for the writing from the James S. McDonnell Foundation 21st Century Science Initiative in Cognitive Rehabilitation Collaborative Award (#220020413), NHMRC Program Grant: Saving Brain and Changing Practice in Stroke (#1113352), and La Trobe University Post-Graduate Scholarship from the Understanding Diseases research focus area awarded to VAN.
