Abstract
Introduction
Clinical trials employing regenerative treatment approaches have been challenged by (
Human central nervous system stem cells (HuCNS-SC) early-stage clinical trials in subjects suffering from neuronal ceroid lipofusinosis and Pelizaeus-Merzbacher disease have been completed without obvious safety issues while proving (ie, postmortem) survival of transplanted cells.2,3 In human SCI, for patient safety, regulatory agencies restrict the initial target population to participants living with chronic motor complete thoracic SCI where in case of any treatment-emergent adverse events causing the neurological level of injury (NLI) to ascend, the distance to the cervical cord would be sufficient to prevent any loss to upper extremity function.
Clinical translation faces many hurdles and premature termination of spinal cord stem cell–based trials is one of these many challenges with 7 of 15 cell-based SCI trials suspended or terminated in the period from 2003 to 2016. 4 For example, no reports could be found for the prematurely terminated GRNOPC1 trial (NCT01217008; human embryonic stem cell–derived oligodendrocyte progenitors) that introduced stem cell transplantation in patients with subacute (7-14 days after incidence) complete paraplegia. It is likely that the financial burden associated with functional and safety assessments, especially for the long-term follow-up of study participants, contributed to the lack of later stage trials exploring therapeutic efficacy of stem cell transplantation in humans. 5 It is imperative to ensure follow-up of trials even after early termination and report findings as a contribution to learnings for the field of stem cell transplantation in SCI (eg, current report).
Here we report 6 years’ safety and preliminary efficacy data of the transplantation of HuCNS-SC into the thoracic spinal cord in patients with chronic motor complete SCI (Figure 1). The study seamlessly incorporated a short-term (STFU, year 1) and long-term (LTFU, years 2-6) follow-up study designed to distinguish between effects of (
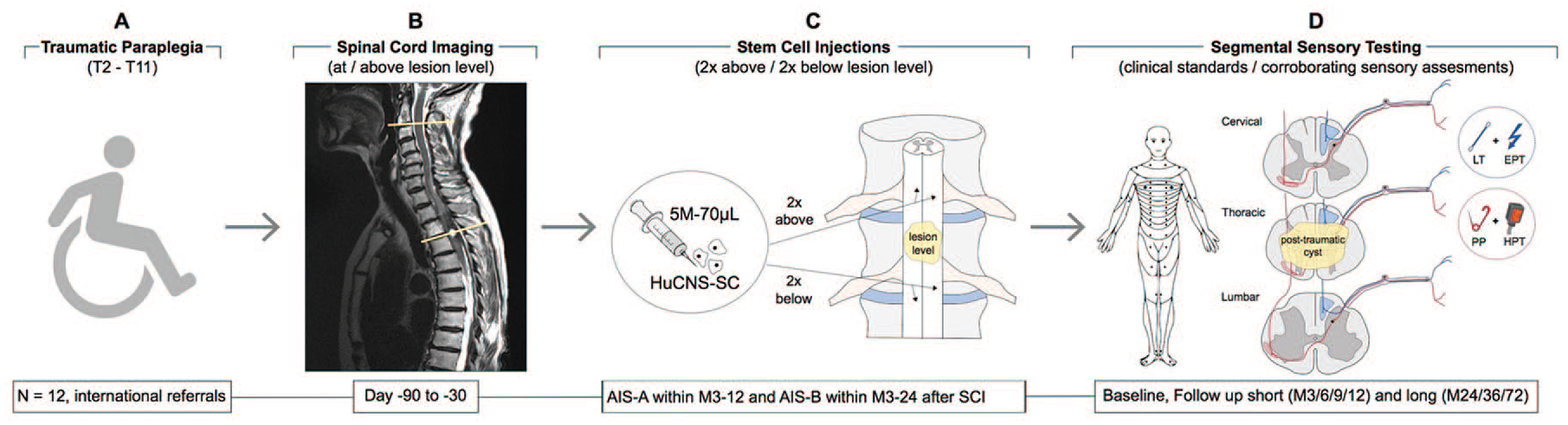
(A) This open-label, single-dose study involved allogeneic human central nervous system stem cells (HuCNS-SC) transplantation in 12 subjects with chronic thoracic spinal cord injury (SCI) for AIS A and AIS B subjects. (B) Magnetic resonance imaging at and above the level of lesion confirmed injury characteristics for eligibility and safety. (C) Twenty million HuCNS-SC cells divided into 4 microinjections were directly injected into the spinal cord immediately above (2×) and below (2×) the site of injury. An immunosuppression regimen lasting for 9 months was performed to support cell engraftment. (D) Subjects were monitored before and after transplantation (short-term follow-up initially daily for 7 days, on day 14, day 28, and at 3-month intervals for 1 year after transplantation, complemented by an additional long-term follow-up for 5 additional years). AIS, American Spinal Injury Association Impairment Scale.
Methods
Patients
The study protocol and all amendments were approved by related ethics committees and regulatory agencies in accordance with the Declaration of Helsinki. Eleven males and 1 female, aged 19 to 53 years, with an average age of 35 years for those with a complete injury (American Spinal Injury Association Impairment Scale [AIS] A) and 30 years for those with sensory preservation (AIS B), and a chronic, traumatic, single thoracic SCI between T2-T11 SCI and preserved sacral nerve function (confirmed by nerve conduction studies) were included in the trial. The time from injury to cell transplantation occurred between 5 and 24 months with the average being 7.9 and 13.6 months for AIS A and B participants, respectively. After the sponsor, principal investigator (PI), and data monitoring committee (DMC) reviewed the first 4 months of safety data for the initial 3 patients with complete injuries (AIS A), patients with sensory incomplete paraplegia (AIS B) were allowed for accrual until total enrolment reached 12 patients. Patient demographics are included in Table 1A. Key exclusion criteria included anatomical transection of the spinal cord confirmed by magnetic resonance imaging (MRI), unresolved traumatic brain injury, spinal column instability, contraindications for MRI or immunosuppression, and secondary medical complications that would preclude safe participation.
Patient Demographics and Safety Assessments.
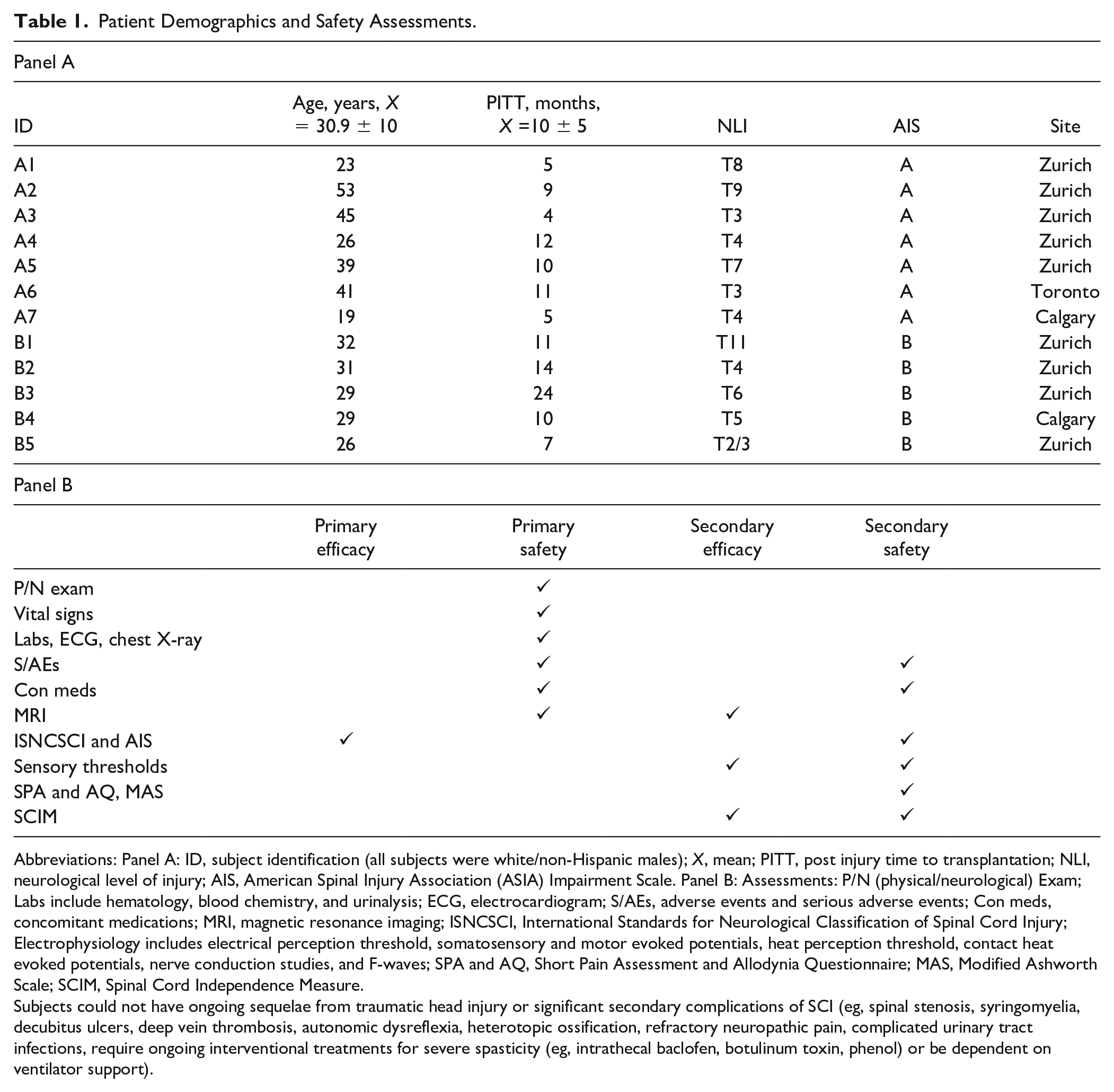
Abbreviations: Panel A: ID, subject identification (all subjects were white/non-Hispanic males);
Subjects could not have ongoing sequelae from traumatic head injury or significant secondary complications of SCI (eg, spinal stenosis, syringomyelia, decubitus ulcers, deep vein thrombosis, autonomic dysreflexia, heterotopic ossification, refractory neuropathic pain, complicated urinary tract infections, require ongoing interventional treatments for severe spasticity (eg, intrathecal baclofen, botulinum toxin, phenol) or be dependent on ventilator support).
Immunosuppression
Immunosuppressive agents were administered to optimize HuCNS-SC cell engraftment. A 9-month course of tacrolimus started 3 days pretransplantation (target blood levels of 5-10 ng/mL, accompanied a 28-day course of mycophenolate mofetil (MMF; 500 mg/d) that was started 2 days prior to surgery. On the day prior to transplantation, dexamethasone (2-4 mg/ intravenous/oral, 4 times a day) was initiated for 5 to 10 days to complete this study’s immunosuppression protocol.
Neurosurgery
Twenty million HuCNS-SC cells were administered through an open neurosurgical procedure with direct, ultrasound-guided injection into the spinal cord immediately above and below the site of injury as determined by preoperative spinal MRI as previously described. 6
Safety Assessments
Safety assessments included SCI specific (International Standards for Neurological Classification of Spinal Cord Injury [ISNCSCI], Short Pain Assessment and Allodynia Questionnaire, Modified Ashworth Scale, and spinal MRI), nonspecific physical and neurological examinations, as well as standard clinical assessments and adverse events (AEs) and serious adverse events (SAEs) monitoring (Table 1B).
MRI Acquisition
All participants at the University Hospital Balgrist were scanned with a 3-T MRI scanner (Siemens MAGNETOM Skyra-fit or Verio) applying a high-resolution T1-protocol (MPRAGE) 7 including the cervical cord (C1-C5). 8 The thoracic scan protocol was optimized to reduce metal artifacts and consisted of T2- and T1-weighted sagittal fast-spin echo sequences, axial T2-weighted fast-spin echo sequences, and gadolinium-enhanced sagittal fast-spin echo sequences (Gadovist, Bayer Pharma). The baseline scans before treatment were acquired at 9.67 ± 6.36 (mean ± SD) months after injury, and the 12-/24-/36-month follow-up scans at 22.11 ± 6.27, 33.75 ± 6.61 and 51.38 ± 10.94 months after injury, respectively. Of the 9 Zurich patients, 1 was lost to LTFU and 7/8 remaining patients showed a high-intensity T2-weighted signal at the lesion site at all time points. One patient showed no such posttraumatic cystic alteration, but presented with a collapsed, heavily atrophied spinal cord (Figure 4). Four of 8 patients did not show well-demarcated tissue bridges at any time point, albeit there was continuation of the cord envelope.
Statistical Analysis
There was no planned hypothesis testing with regard to safety or biologic activities as the number of subjects was based on gaining first data about safety and preliminary effectiveness to support future clinical trials.
Results
Patients
The study was conducted at 3 specialized spinal cord injury centers (Zurich n = 9; Calgary n = 2; Toronto n = 1) from 2011 to 2020 (Figure 2). STFU assessments (first year) were performed at least monthly for all patients and LTFU assessments (years 2-6) were undertaken at all scheduled and nonscheduled study contacts (up to 72 months in all patients but one). One subject (B2) declined further on-site follow-up beyond 12 months posttransplantation but did agree to telephone follow-up until 60 months posttransplantation. At month 12, one subject (A7) experienced an emerging psychiatric condition unrelated to the transplanted cells and was followed up after the month-12 visit by remote correspondence (eg, telephone and email) until month 60.
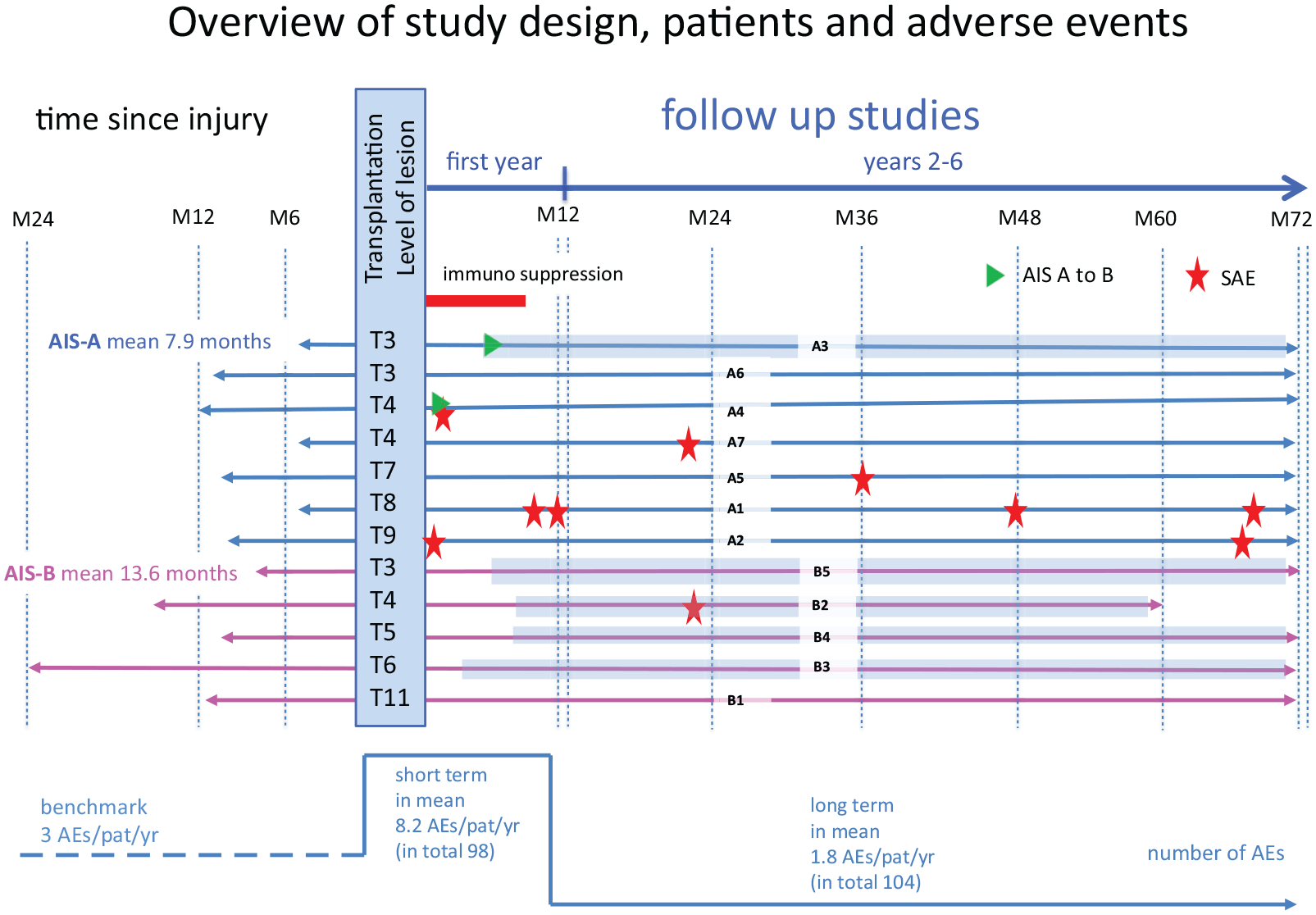
In total, 12 subjects (11 male; 7/5 AIS A/B, respectively) with a single traumatic T3-T11 injury (time from injury to cell transplantation from 5 to 24 months (AIS A
Adverse Events
In the first year, 4 SAEs occurred in 3 subjects requiring either prolonged hospitalization (cerebrospinal fluid leak at 11 days posttransplant and a moderate pseudomeningocele at 13 days posttransplant) or rehospitalization (constipation and UTI at 49 weeks posttransplant). For the final 2- to 6-year follow-up, 6 SAEs were recorded in 10 patients (Table 2 and Figure 2).
Adverse Events (AEs) and Serious Adverse Events (SAEs). a
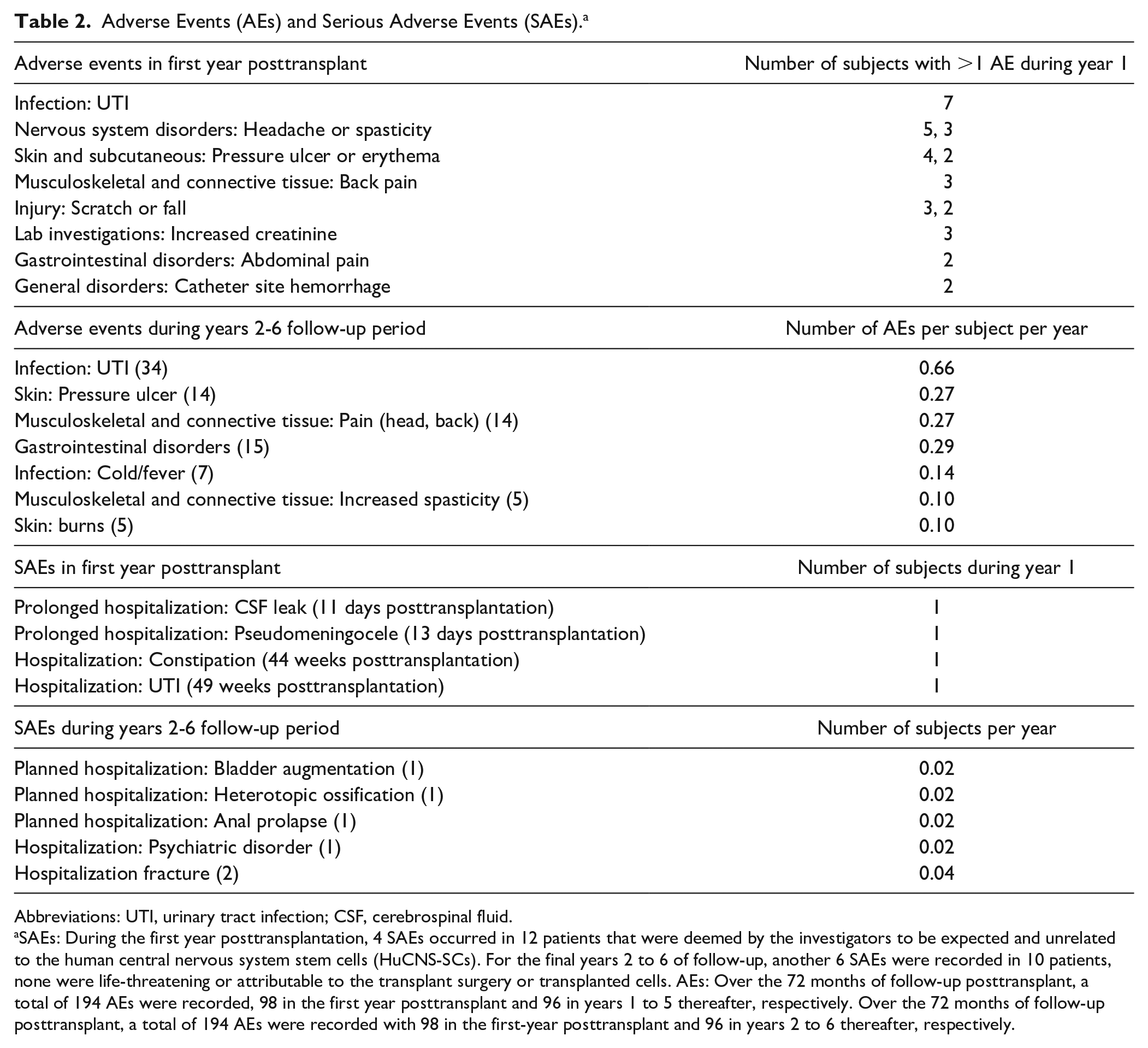
Abbreviations: UTI, urinary tract infection; CSF, cerebrospinal fluid.
SAEs: During the first year posttransplantation, 4 SAEs occurred in 12 patients that were deemed by the investigators to be expected and unrelated to the human central nervous system stem cells (HuCNS-SCs). For the final years 2 to 6 of follow-up, another 6 SAEs were recorded in 10 patients, none were life-threatening or attributable to the transplant surgery or transplanted cells. AEs: Over the 72 months of follow-up posttransplant, a total of 194 AEs were recorded, 98 in the first year posttransplant and 96 in years 1 to 5 thereafter, respectively. Over the 72 months of follow-up posttransplant, a total of 194 AEs were recorded with 98 in the first-year posttransplant and 96 in years 2 to 6 thereafter, respectively.
First-year AEs, related to SCI or other inter-current illness (50%), resolved without sequelae and none were attributed to the transplanted HuCNS-SC cells. Secondary to the transplant surgery, there were 9 first-year AEs (9%) considered mild (n = 6) or moderate (n = 3), including dizziness, bleeding at the bladder catheter site, ulnar nerve compression, postoperative pain, and a transient increase in spasticity over 1 day.
In the 2 to 6 years of follow-up, each of 10 patients reported on average 1.75 AEs per year. Compared with a convenience sample of 1063 Swiss paraplegic SCI residents, the number of treatments sought for concomitant health conditions (eg, pain, spasticity, infections [urinary and pulmonary], fractures, bowel/bladder problems, and pressure ulcers) was a mean of 3 over 1 year. 9
Twelve of 98 first-year AEs attributed to immunosuppression (12%) were considered mild, except in 2 instances designated as moderate (“increase in white blood cell count” and “headache,” both resolved without requiring specific actions). Forty-seven UTIs accounted for 24% of AEs reported over 6 years of follow-up (13/98 during 1 year of STFU and 34/96 during 5 additional years of LTFU). Subjects reported an average of 1.1 UTIs in the first-year postsurgery and 0.66 UTIs over the next 5 years, which is in the range of reported UTI frequencies. 10
Pain assessment did not reveal any allodynia at any visit in 8 of 12 subjects. Allodynia emerged in 1 subject at month 3 and in another subject at month 6 that was still reported at the final visit of the STFU at 12 months. Two remaining subjects reported allodynia intermittently, but only 1 continued to report it at the final month-12 visit. The LTFU study documented 14 AEs described as musculoskeletal pain (eg, head, back) in 8 of 12 subjects (67%). No additional subjects reported novel instances of allodynia or hyperalgesia in the years 2 to 6 LTFU study.
Although there were 4 reports of transient increased spasticity, no subject registered permanent worsening of spasticity when assessed with the Modified Ashworth score.
Preliminary Efficacy
Combined primary and exploratory efficacy measures revealed consistent preliminary efficacy throughout the study in 5 subjects (AIS A = 1; AIS B = 4; Figure 2). These 5 subjects exhibited neurological improvement in more than 1 mode of neurological function after transplantation that was maintained over at least the first year of follow-up. Modes of emerging neurological function included segmental recovery of light touch (LT), pin prick (PP), or deep anal pressure (DAP) starting at 3 months posttransplant. The emergence of LT and PP within these subjects occurred at a time point beyond the first 6 months after SCI (mean = 7.8 months postinjury), when spontaneous natural recovery in SCI patients is considered low. 11 Two of the 7 AIS A patients developed some sensation below the lesion, that is, subject A3 underwent AIS A to B conversion at month 6 posttransplant (or 10 months postinjury), while subject A4 converted on day 1 after transplantation surgery with no subsequent robust changes in clinical or electrophysiological assessment. The latter was not deemed a response to the intervention.
Although some of the 5 AIS B patients showed subtle sensory improvements, none acquired motor function below the level of injury, that is, none converted to AIS C. One AIS A and 4 AIS B patients showed a narrowing of the zone of injury with respect to "sensation.
Subjects exhibiting improvements in clinical testing (eg, LT and PP) also showed concordant quantitative sensory testing results (electrical perception threshold [EPT] and heat perception threshold [HPT], respectively) except for subject B2 (T4) where slight EPT discordance occurred several segments below the level of the lesion at T7 and T10 starting at month 6 posttransplantation (Figure 3). EPT was not assessed at one Canadian site and only for some subjects in the other Canadian site and therefore EPT was not recorded in subject B4. Improvement in LT was maintained over 1 year of follow-up in 1 AIS B subject (B2) before being lost to follow-up. Improvements were maintained in 3 additional AIS B subjects (B3, B4, B5) up to the last visit where a LT assessment was completed at 48 months.
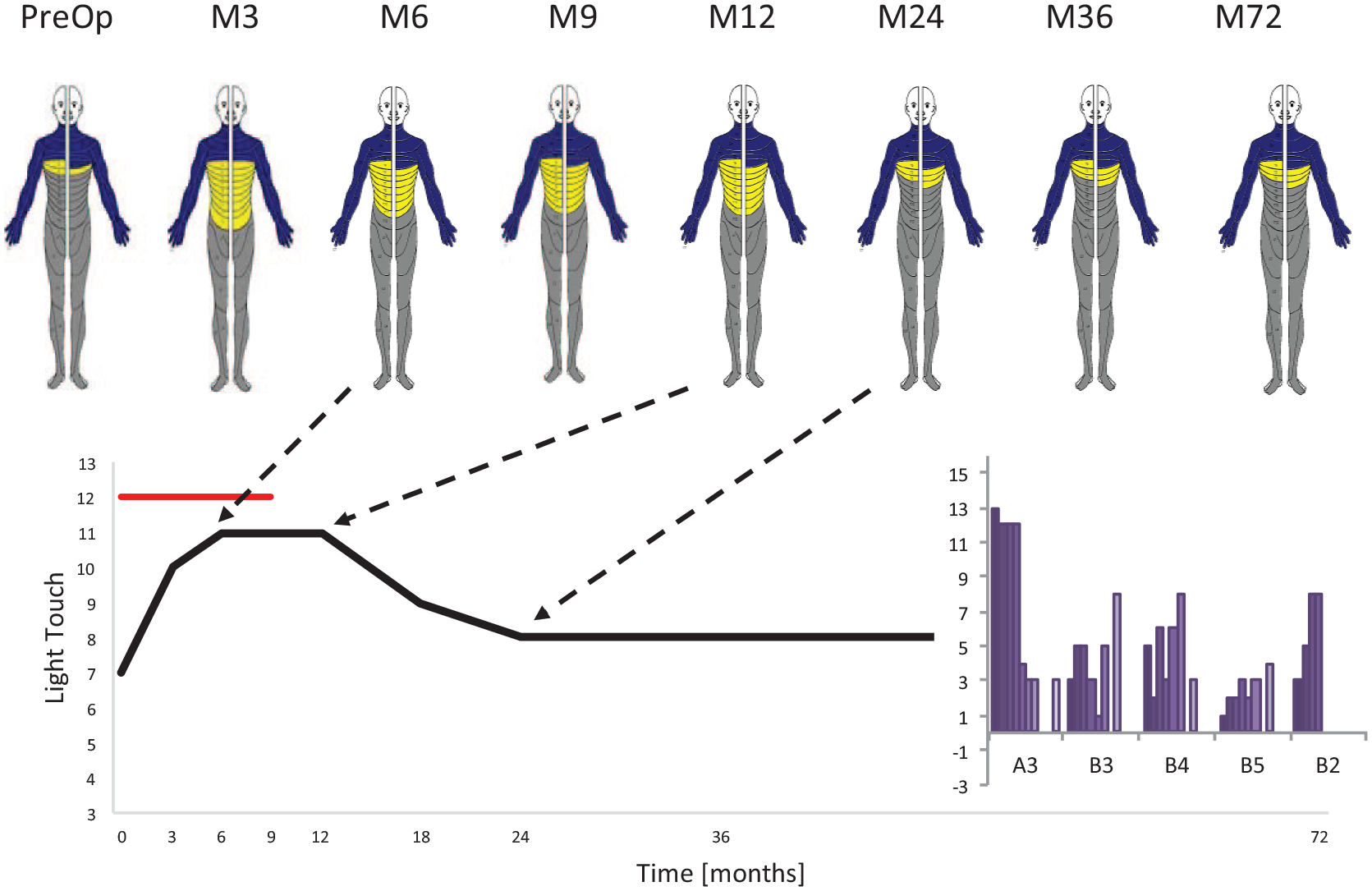
Individual case study in subject A3 and the overall light touch (LT) changes (overlaid histogram) of the responders. Blue/yellow/gray dermatomes represent normal/partial/absent sensation. The nonresponders had in mean no LT changes compared to in mean LT changes of +7.16 in the responders. Compared with control subjects (EMSCI data of mean LT changes 6-12 months after injury: thoracic AIS A n = 258, mean −0.25 ± 4.3; AIS B n = 17, mean 1.1 ± 3.2) the LT changes in the responders were above historical benchmark values. None of the nonresponders reported any decline in sensory functions.
Subject A3 showed extensive LT improvements following transplantation (improved dorsal column function in both LT and EPT) across T4-12 that eventually, after cessation of immunosuppression, was almost completely lost while he continued to remain AIS B up to 6 years posttransplantation (Figure 3).
No changes were detected in the Modified Ashworth Scale (MAS), Spinal Cord Independence Measure (SCIM-III, or the SF-36 Medical Outcomes Study [MOS] 36-item Short Form health survey.
Neuroimaging
The pretreatment
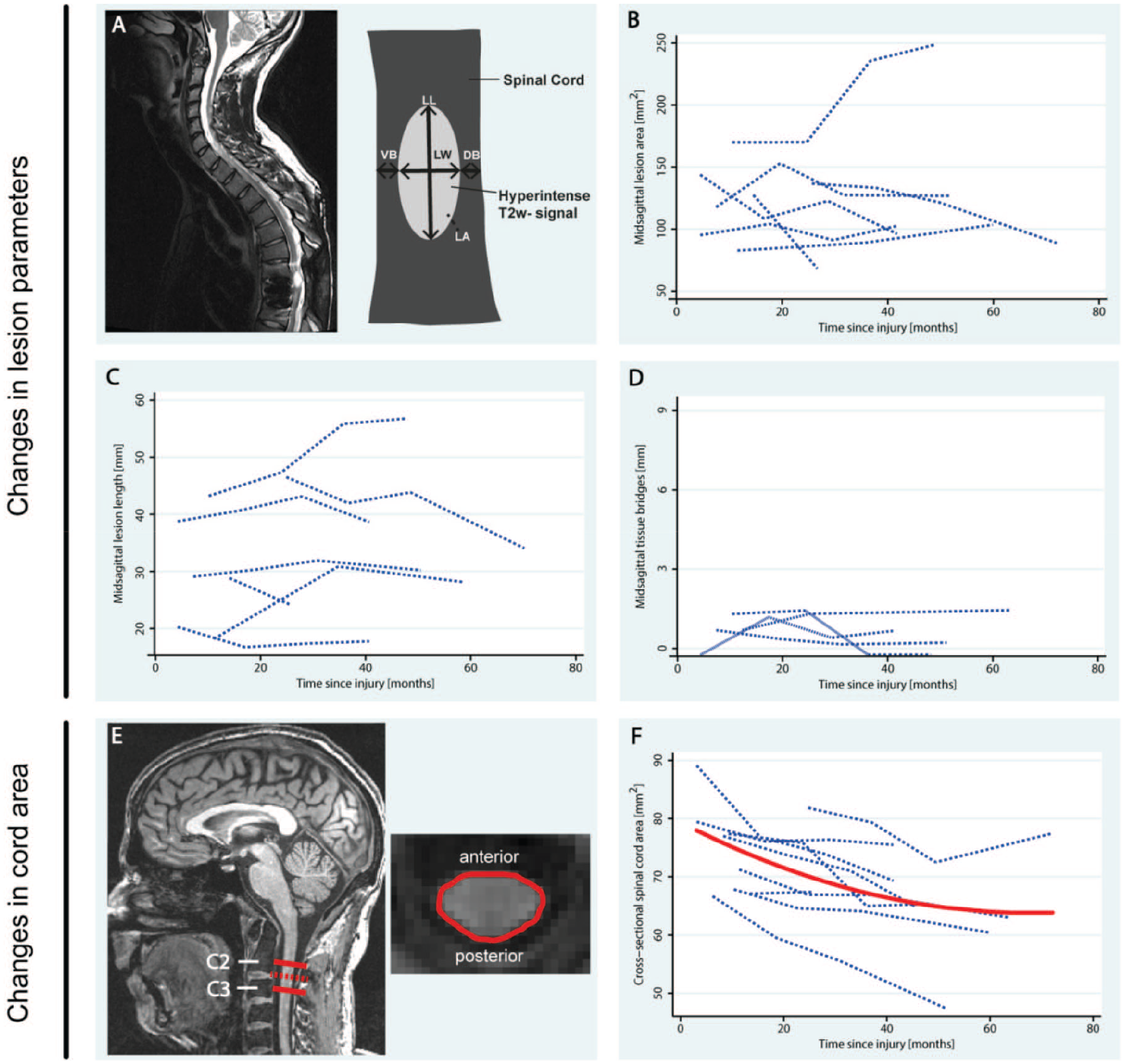
Changes in lesion parameters (A-D) and cross-sectional spinal cord area (E-F) before and up to 36 months after stem cell treatment. (A) T2-weighted scan and scheme showing the parameters analyzed. (B) Change in midsagittal lesion area over time. (C) Change in midsagittal lesion length over time. (D) Change in midsagittal width of tissue bridges over time. (E) T1-weighted scan showing the area analyzed. (F) Change in cross-sectional spinal cord area at the level of C2/C3 over time. The red line indicates the significant, fitted model. DB, dorsal tissue bridges; LA, lesion area; LL, lesion length; LW, lesion width; VB, ventral tissue bridges.
At baseline, lesion width was 6.76 ± 0.91 mm (n = 7, range: min/max 5.32/7.96 mm), and the width of tissue bridges 0.78 ± 0.58 mm (n = 4, range: min/max 0/1.41mm), which remained unchanged (lesion width:
At baseline, cross-sectional spinal cord area (SCA) was 75.30 ± 7.68 mm2 (range: min/max 66.67/89.12 mm2) and showed a significant decrease of 0.467 mm2 per month (
Discussion
This first-in-human study of allogeneic HuCNS-SC transplantation in chronic thoracic SCI subjects revealed promising safety for targeting even the damaged spinal cord. There was no clinical deterioration of function within the injected or adjacent myelomeres above and below the level of lesion, detection of tumors over 6 years of follow-up, or deterioration of spinal cord function subsequent to the surgical implantation. Assessments of standard clinical neurological and functional outcomes complemented by detailed assessments of segmental sensory function and neuroimaging demonstrated the value of an advanced protocol design in such a pioneering translational trial. HuCNS-SC transplantation was well tolerated with anticipated AEs that resolved without sequelae and were consistent with medical conditions commonly observed in individuals with SCI. 13 The required immunosuppression did not increase infections, specifically UTIs. 14 Preliminary efficacy of HuCNS-SC transplantation was observed both adjacent and below the thoracic NLI. Interestingly in 1 AIS A subject, withdrawal of immunosuppression as defined by the trial protocol, was followed by a remarkable loss of gained function. The remaining 4 participants who experienced improved sensory function remained stable for their remaining follow-up period.
Adverse Events
Patients suffering from chronic SCI remain vulnerable to secondary medical complications and rehospitalization rates of ~33% following discharge from rehabilitation due to UTI, skin lesion and respiratory dysfunction have been reported in representative patient cohorts. 13 Comparatively, the reasonable moderate rate of posttransplantation AEs observed in the current HuCNS-SC SCI study represented a promising AE profile with only a single incidence of rehospitalization due to UTI (1/12 or 8.3%). 15 Also, the incidence of neuropathic and musculoskeletal pain in the current study (25% and 67%, respectively) was as anticipated, and no unexpected courses in frequency and intensity of neuropathic pain were observed. 16
In addition, immunosuppression AEs were within the spectrum commonly reported for tacrolimus and mycophenolate mofetil administration (mainly laboratory abnormalities) and showed no effect on the incidence of specifically UTI. 14 Similarly, the type, severity and rate of surgically related S/AEs were in the expected range for similar spine stabilization surgeries without cell transplants. 17
Adverse Events Related to Stem Cells
HuCNS-SC cells have been evaluated in preclinical safety and toxicology Good Laboratory Practice studies with no evidence of harm in brains, spinal cords, or peripheral organs after intracerebral and spinal cord administration in any species studied. Specifically, neither an extra-CNS distribution nor tumor formation after subcortical and intraventricular HuCNS-SC cell transplantation was detected. More than 60% of the engrafted cells differentiated along the oligodendrocyte lineage and showed evidence of re-myelination of some host axons. Furthermore, allodynia did not increase in animals transplanted with HuCNS-SC cells. 3 Importantly, histological analysis revealed that transplantation of HuCNS-SC did not contribute to glial scar formation and or differentiation into glial fibrillary acidic protein-positive (GFAP+) astrocytes. 18 Accordingly, the 6 years observation period post HuCNS-SC transplantation in human thoracic spinal cord did not reveal signs or symptoms of tumor formation or aberrant changes in post-traumatic cyst formation by means of MRI. The observed safety is comparable to other studies performing intraspinal cell transplantations in humans that primarily relied on basic clinical examinations (ie, ISNCSCI) with a rather short follow-up regimen. 19 It should be noted that these safety findings can only be applied to transplantation of HuCNS-SC cells in the human spinal cord.
Preliminary Efficacy
Recovery in Thoracic SCI
The challenge in early clinical trials is detection of subtle but reliable changes both in the interest of safety (even of minor harmful effects) and efficacy (minimal improvements that could be enhanced to reach clinical meaningfulness in subsequent clinical trial phases). 20 Many SCI databases do not include sensory function across trunk segments and with no key testable muscles available at the trunk, reporting on the recovery of sensory function post-SCI is often neglected. However, in these databases some sensory function recovery can be indirectly interpreted when looking at the conversion from AIS A to B that occurs in about 7.7% by ~48 weeks after traumatic thoracic SCI. 21 The latter occurred in 2 AIS A patients while in 1 subject (A4), the conversion occurred immediately after surgery and was therefore unlikely due to the cell transplantation. Overall, levels of independence by means of SCIM and lower limb motor scores did not change in the patient population. This is different from a previous trial of expanded autologous bone marrow mesenchymal stromal cells (MSC) injected into the spinal CSF compartment in chronic paraplegia reporting dose-dependent clinical improvements in all participants, including motor recovery in at least 50% of subjects.22,23
It is important to note that the subsequent cervical HuCNS-SC trial reported some improvements in UEM scores and GRASSP strength component at final follow-up for those who were randomized to receive these human CNS-derived neural stem cells. 24 Unfortunately, the true impact of HuCNS-SC could not be proven due to premature termination of the trial by the sponsor leaving findings underpowered.
Segmental Sensory Changes
Combined tests of dermatomal LT/PP sensations and corroborating EPT/HPT revealed a higher responsiveness for consecutive changes in AIS B versus A designations. Four of 5 AIS B and 1/7 AIS A showed improved responses of sensory function associated with dorsal column tracts (concordance of LT and EPT recordings) and to a lower extent of the spinothalamic tracts (concordance of PP and HPT recordings). Dorsal column and spinothalamic changes were registered immediately below the NLI and caudally across lumbar and sacral segments. These changes recorded beyond the usual time course for natural recovery following transplantation (ie, 3 months post) and were sustained out to at least 1 year up to 6 years. Findings of segmental improvements were also reported by Curtis et al 25 in a study of 4 paraplegic chronic SCI patients using human-spinal-cord derived stem cells followed up to 27 months. These findings may be suggestive of biological activity resulting from stem cell transplantation that supersedes the improvements as reflected by natural recovery data (eg, EMSCI data).21,26
Assuming that these changes are biological in nature, remyelination might be the mechanism of action based on the preclinical findings. 27 Speculated mechanisms of action for cell-based interventions are likely to be combinatorial of neuron regeneration, promotion of remyelination by donor cells and/or neuroprotection or plasticity resulting from administration of trophic molecules. 28 The nonuniform manner in which these changes occur may reflect the overlap between adjacent dermatomes 29 and central spinal pathways close to the lesion site. Having 80% (4/5) AIS B and 14% (1/7) AIS A subjects register some subtle neurological improvement following the stem cell transplant and given this promising feasibility, further pre/clinical studies aiming at fostering the engraftment, survival and functional integration may be warranted. 8
Although immunosuppression is thought to improve stem cell transplant survival, the precise mechanism of action is not well understood, especially given the ongoing “paradox of chronic neuroinflammation, systemic immune suppression, autoimmunity after traumatic chronic spinal cord injury”. 30 An example of a possible immunosuppression related response was revealed in subject A3 (transplanted at 5 months postinjury) who experienced significant light touch gains over 8 dermatomal segments that reversed over 6 segments starting 3 months after the termination of immunosuppression (Figure 3). However, no patterns emerged across the 12 subjects in their responses to the termination of immunosuppression.
Neuroimaging
More sensitive (qualitative and quantitative measures) outcomes such as high-resolution MRI mapping of CNS plasticity is evolving and may be able to reveal plastic reorganization in both the spinal cord and brain. 31 These changes may not immediately relate to the improvement of outcome measures but could indicate signs of neuroplasticity as proof of concept worth following for eventual potential clinical benefit. 32 This also emphasizes the need for, as yet unavailable, biomarkers to track transplanted cells in human SCI translational research.33,34 Recent advances in ultra-high-resolution structural and functional MRI mapping in the cord and brain may be a means to complement clinical readouts and increase the sensitivity of translational trials.35,36 Here we applied measures of cervical spinal cord areas that have been shown in several studies to be responsive to spinal cord damage with secondary and continuing changes even over several years after injury.7,36,37 The consecutive reduction of spinal cord area is attributed to neuronal degeneration and demyelination indicating ongoing responses of the nervous system to the focal and singular spinal cord damage in patients with otherwise no clinical signs of deterioration. Measuring spinal cord areas above the level of lesion did not reveal remote changes deviating from changes that are normally seen within the cord and which are indiscernible by clinical means. 38 The observed decline was of a similar magnitude as observed in a cohort of SCI patients without treatment. 12
In summary, this study represents the first-in-man investigation study of HuCNS-SC in chronic SCI. The small study size and open-label design reflect the pilot nature of this investigation, but the comprehensive assessments up to 72 months after transplantation showed promising feasibility and safety in complete and incomplete SCI. Conclusions regarding the preliminary efficacy of HuCNS-SC need to be interpreted with care due to the open label study design. No sustained deterioration was recorded in any subject. Based on the safety profile established in this study for individuals with thoracic SCI, a larger scale, randomized study in people with cervical SCI was approved in the United States (NCT02163876). The latter phase II trial, although prematurely terminated by the sponsor, accordingly confirmed short-term safety (ie, up to 1-year posttransplantation) also at the cervical cord. 39
Limitations
This study was restricted to patients with thoracic SCI due to safety concerns and with no adjacent segmental key muscles available for assessment and monitoring, functional motor changes were noticeably absent from the results obtained in this study. Additionally, the translated preclinical to human dose was scaled back for a margin of safety, further diminishing the potential to see changes in larger diameter motor neurons compared to sensory cells. Fully translated human doses in the cervical and low thoracic cord with accessible key muscles for assessment of motor changes is required for further investigation.
Footnotes
Acknowledgements
We thank A. Prusse and I. Krüsi for excellent study nurse support, R. Pfiffner for retrieving all neurophysiological data and assistance for EP analysis, and P. Scheuren and L. Suter for technical support for the manuscript. We thank Dr J. Steeves for critical review of the manuscript. All data are stored and accessible at the Spinal Cord Injury Center Balgrist, Zurich, Switzerland.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The initial study (short-term follow-up of the first year; STFU) and part of the second study (long-term follow-up; LTFU) was funded by Stem Cells Inc, Newark, California, USA. After dissolution of Stem Cells Inc, 2016Q3 grants from Wings for Life (WfL), and the Bright Oceans Corporation (BOCO) enabled completion of the LTFU, compilation of the full set of monitored data (initial Phase I/IIa STFU trial and LTFU study), data analysis, and manuscript completion.
