Abstract
Introduction
Transplantation of stem/progenitor cells offers a novel and exciting possibility for repairing the injured spinal cord (ISC) through replacement of damaged cells, neuroprotection, or the creation of an environment conducive to regeneration by endogenous cells. Among the stem cells that could be used for this purpose, bone marrow (BM)–derived mesenchymal stem cells (MSCs) are one of the best candidates because they are relatively easy to isolate, can be expanded rapidly in vitro,1-3 and differentiate into multiple cell types,4-8 including neuron-like cells. 9 Several recent reports concerning the neural differentiation potential of MSCs suggest that with appropriate neural induction protocols, MSCs could produce mature neuron-like cells that exhibit multiple neuronal properties and traits, such as an action potential, synaptic transmission, secretion of neurotrophic factors and dopamine, and demonstration of a spontaneous postsynaptic current.10-16 Moreover, direct or intravascular MSC delivery is associated with functional improvements in experimental models of central nervous system (CNS) injury and disease.17-20
Recently, we developed a new method for efficient generation of neural-like cells from human BM-derived MSCs (hMSCs). 21 The aim of this study was to determine whether transplanted neurally induced (NI) hMSCs can (1) survive and differentiate in the ISC of rats, (2) improve tissue and white matter preservation, and (3) promote functional recovery.
Methods
Expansion of hMSCs
Human MSCs (frozen at passage 1) were provided by Tulane University Center of Gene Therapy. According to the product specification sheet, human BM aspirate was drawn, and mononuclear cells were separated using density centrifugation. The cells were plated to obtain adherent human MSCs, which were harvested when cells reached 60% to 80% confluence. These specimens were considered passage zero (P0) cells. These P0 cells were expended, harvested, and frozen at passage 1 (P1) for distribution. Prior to release, 2 trials of the frozen P1 cells were analyzed over 3 passages for colony forming units, cell growth, and differentiation into fat, bone, and chondrocytes (at P2 only). These characterized hMSCs from P1 were expanded and used for neural induction protocols.
Neural Induction
Neural induction was performed by the method described recently. 21 Briefly, hMSCs were exposed to 200 nM trichostatin A (inhibitor of histone deacetylases), 3 µM RG-108 (DNA methyltransferase inhibitor), 300 µM 8-BrcAMP (highly stable, biologically active form of cAMP), and 1 µM Rolipram (inhibitor of phosphodiesterases) in a medium consisting of NeuroCult/N2 supplemented with 20 ng bFGF. After 2 weeks of treatment, cells were used for transplantation.
Spinal Cord Injury and Transplantation Procedures
All animal surgeries were performed in accordance with guidelines established by the Medical College of Wisconsin and Zablocki Veterans Affairs Medical Center Veterinary Medical Unit. For these studies, Sprague-Dawley female rats (200-250 g body weight) were anesthetized using intraperitoneal ketamine (75 mg/kg) and medetomidine (0.5 mg/kg). Rats were placed prone on an operating table covered with a warming blanket. The dorsal midthoracic region was shaved and prepped with Betadine. Using a sterile technique, an incision was made over the midthoracic region, and a subperiosteal dissection was performed. Then, 3 spinal-level laminectomies (T7-9) exposed the underlying spinal cord. Hemostasis was obtained with Surgical Gelfoam and bone edge waxing. The NYU Impactor was used to produce a consistent, uniformly severe injury (10 g drop from a height of 25 mm directly onto the dura at level T8). Following impact, the wound was closed in layers. Postoperatively, all animals were given 1 dose of enrofloxacin (10 mg/kg subcutaneously). Subcutaneous lactated Ringer’s solution (12 mL) was provided and made available as needed (50 mL/kg). Bladder expression was performed manually twice per day until the animals were able to void independently.
One week after SCI, the rats were randomized to receive NI hMSCs, hMSCs, or phosphate-buffered saline (PBS). From each group, 9 animals were for used for behavioral studies and 10 for stereological and immunohistochemical studies. For the transplant surgery, rats were reanesthetized and, using a sterile technique, the SCI site was reexposed. NI hMSCs were loaded into a 25-µL Hamilton syringe at a concentration of 100 000 cells/10 µL. Under microscopic visualization, the cells were stereotactically injected into the spinal cord on either side of the midline and 1 mm rostral and 1 mm caudal to the injury site. Four 2.5-µL injections delivered a total of 100 000 NI hMSCs to each spinal cord. After injection, the surgical site was closed in multiple layers, and the animals were allowed to recover with analgesia and postoperative care, as described above. All animals received the immunosuppressant Prograf (50 mg/kg) on a daily basis. All groups received Prograf in order to account for any neuroprotective effects of the immunosuppressant.
Behavioral Testing
Individuals involved in assessing functional recovery of the animal subjects were blinded with regard to treatment. Locomotor function was evaluated using the BBB Locomotor Recovery Scale for open-field walking. 22 Behavioral tests for thermal allodynia were performed before and after SCI in both forelimbs and hindlimbs, as previously described.23,24 The response to thermal stimulation was measured by latency of forelimb and hindlimb paw withdrawal to radiant heat of 55°C. Briefly, rats were placed in a Plexiglas cylindrical container over a radiant heat source, and latency to response was recorded. An average of 3 trials was recorded, and nonresponders were removed from the hot plate after 60 s. Brisk paw withdrawal with or without accompanying supraspinal reflexes, such as head turning, paw guarding, licking, biting, or vocalization, were considered positive responses to the thermal behavioral testing. 24
In all groups, behavioral scoring was performed prior to injury, after injury, prior to transplantation, and then weekly for 12 weeks posttransplantation. Effects of treatment were assessed using 2-way analysis of variance (ANOVA) followed by post hoc Tukey’s analysis with a significance level of
Immunohistochemistry and Histological Assessment
After 24 hours and 1, 2, 4, and 12 weeks, animals were given an overdose of nembutal and perfused intracardially with 0.9% PBS followed by 4% paraformaldehyde in PBS. Spinal cords were dissected and the injury site at T8 identified. The cervical C6-T1 region was identified by the origin of the median-ulnar forelimb nerve complex from the spinal cord at C7-T1 vertebral levels. The spinal cord was cut into segments that included the T8 region with 2-mm uninjured cord rostral and caudal to the injury and the cervical cord C6-T1. These segments were postfixed in 4% paraformaldehyde/PBS at 4°C for 1 h and cryoprotected in 30% sucrose/PBS overnight at 4°C. To eliminate or reduce autofluorescence, 2 reagents—sodium borohydride and Sudan Black B (Sigma)—were used. To this end, postfixed cryostat cross-sectioned samples were immersed in ice cold PBS supplemented with 1 mg/mL sodium borohydride for 40 minutes. After this, samples were washed in PBS, permeabilized 5 minutes with 1% bovine serum albumin (BSA), 2% goat serum, and 0.5% Triton X-100 in calcium magnesium free (CMF)-PBS and incubated for 1 hour with one of the following primary antibodies in the same solution (except for 10-fold less Triton X-100): human antimitochondrial antibodies (monoclonal, 1:400, Millipore), polyclonal anti-Sox2 (1:800, Chemicon), polyclonal anti-GFAP (1:1000, Novus Biologicals, Inc, Littleton, CO), and polyclonal anti-B3T (1:1000, Covance, Princeton, NJ). After 3 washes of 5 minutes in PBS, the sections were incubated in PBS containing 1% BSA, 2% goat serum, and one of the following secondary antibodies: Texas Red–conjugated goat antimouse IgG and FITC-conjugated goat antirabbit. Nuclear counterstaining was achieved with DAPI or Topro. Cells were then treated with Sudan Black (0.3% in 70% ethanol) for 1 minute, rinsed in PBS, and coverslipped with Fluoromount G mounting medium (Electron Microscopy Sciences, Hatfield, PA). A Nikon inverted microscope equipped with a color digital camera Spot II (Diagnostic Instruments, Inc, Sterling Heights, MI) or BioRad confocal microscope were used to capture representative images. Metamorph software (Universal Imaging, Downingtown, PA) were used for cell counts.
For histological assessments of white matter sparing, 20-µm transverse spinal cord sections of rats that were killed humanely at 12 weeks after SCI were stained with eriochrome cyanine (to stain myelinated white matter) and Eosin/Phloxine (to visualize cells and gray matter) as described.25,26 Stained sections were viewed on an optical microscope to ensure that all samples contained the entire lesion extent and to identify the epicenter of injury. The section containing this epicenter was defined visually as the one with the smallest visible rim of spared myelin and with the largest cystic cavity. The eriochrome cyanine–positive area (white matter) was assessed using the MetaMorph color threshold tool. The percentage of spared white matter in each section was calculated by dividing the white matter area by the total cross-sectional area and multiplying by 100. The percentages of spared white matter for 13 evenly spaced sections 1 mm apart (picked up from approximately 3 mm rostral and caudal to the epicenter of the lesion) were summed, and means and standard errors were calculated for each treatment group (PBS, hMSCs, and NI hMSCs). One-way ANOVA and a Fisher’s LSD (least significant difference) post hoc were used to determine significant differences between groups.
To assess the volume of cystic cavities, the 13th transverse spinal cord sections with an equal distance (500 µm) spanning ± 3 mm from the epicenter were used. The total estimated volume was calculated using Cavalieri’s principle. The individual subvolumes were obtained by multiplying the cavity area by the distance between sections, and the subvolumes were summed to generate the total volume of cystic cavities (∑n [cystic cavity areas × intersection distance], n = number of sections analyzed). Lesion cavity volumes expressed as a percentage of the volume of the spinal cord T8 segment (3 mm rostral and caudal to the lesion epicenter) were calculated by dividing the cystic cavity volume by the spinal cord T8 segment volume and multiplying by 100. For statistical analysis, the group means were compared with 1-way ANOVA and Fisher’s LSD post hoc. Mimics 8.11 3D cord modeling software was used for 3-dimensional reconstruction of lesion cavities.
Results
HMSCs grown in neural induction medium for 2 weeks produced cells that were positive to several neural stem cell, neural progenitor, and mature neuronal markers (Figure 1). At this stage, NI hMSCs were used for transplantation studies.
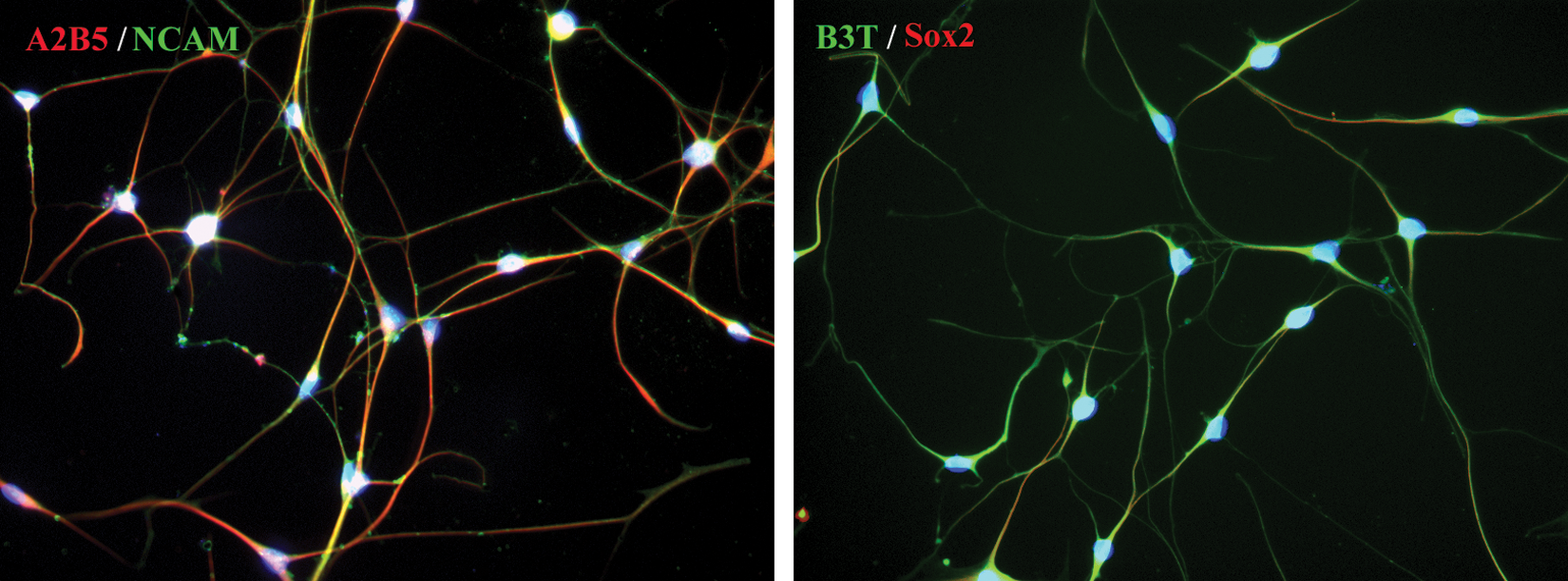
Expression of several neural markers in NI hMSCs grown for 2 weeks in neural induction medium
Assigned BBB scores reflected the near complete loss of hindlimb motor function that was observed in all groups by 1 week after injury and also prior to transplantation with NI hMSCs, hMSCs, and PBS. After the intraspinal transplantation procedure, hindlimb function improved gradually in all groups irrespective of what was used for transplantation. Motor recovery that consisted of hindlimb weight support and consistent hindlimb stepping was significantly different at 2 to 12 weeks postrecovery in the group that was transplanted with NI hMSCs when compared with the control groups that received hMSCs and PBS (Figure 2).
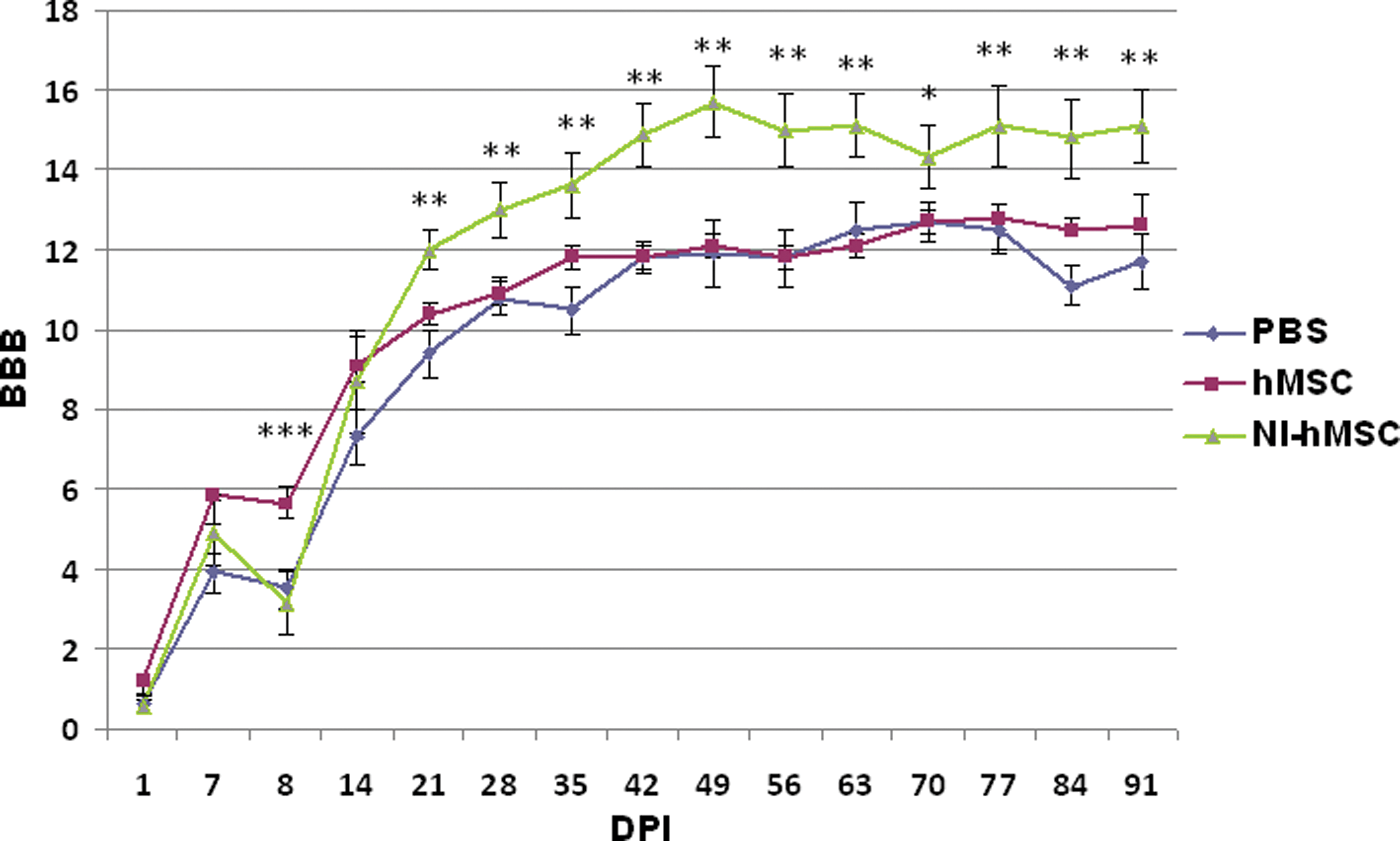
Locomotor recovery (BBB) scores for the post–spinal cord injury behavioral analysis. The asterisks and double asterisk indicate significant differences between the NI hMSC–transplanted group compared with the PBS and PBS + HMSC groups, respectively. The triple asterisk indicates significant differences between the hMSC-transplanted group and the PBS group
Sensory function was assessed by measuring the latency of limb withdrawal using a hotplate test. Withdrawal latencies for affected hindlimbs were slightly reduced in animals that were transplanted with NI hMSCs compared with those treated with vehicle (Figure 3A) at posttransplant weeks 3, 4, 5, 7, 8, and 9 but significantly different only at 12 weeks posttransplantation. What was important was that no abnormal pain responses were observed in the forelimbs of rats that were transplanted with NI hMSCs (Figure 3B).
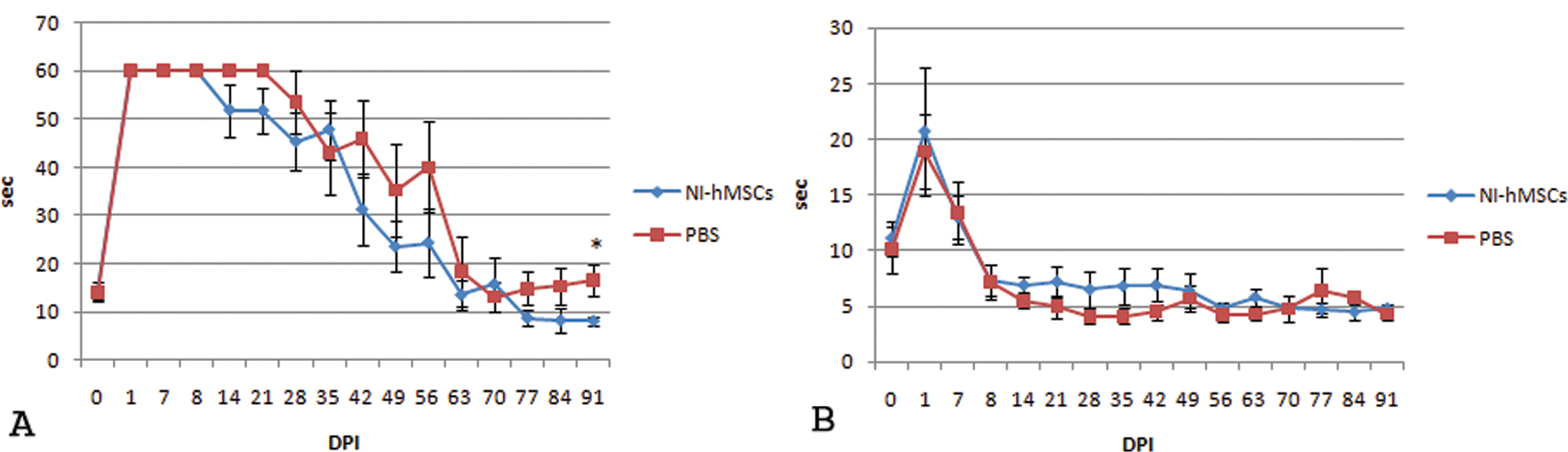
Hindlimb (A) and forelimb (B) behavioral responses were recorded as paw withdrawal latencies to the thermal stimulation of 55°C
Immunohistochemistry data showed that NI hMSCs survived at posttransplantation weeks 1 to 12. Analysis of the spinal cord slices of animals treated for 2 weeks revealed that 85% of the cells that survived were positive to B3T (Figures 4A, 4B, 4C, and 4D). A small percentage of cells (2%) was positive to GFAP (Figure 4E) and 5% to Sox2 (Figure 4F). By 12 weeks, the number of surviving cells declined to 15% to 20% of that at week 2, and only 10% of cells that survived were positive to B3T (Figures 4G, 4H, and 4I). Histological studies of spinal cord sections at specified distances rostral and caudal to the epicenter demonstrated that at the epicenter and 1 mm caudal and rostral from it the percentage of the eriochrome cyanine–positive spared white matter was significantly larger in the NI hMSC–treated group than in the PBS group (Figures 5A and 5B). Although there was no significant difference between naïve hMSCs and PBS groups, there was a modest trend for increased white matter sparing in the hMSC-treated versus PBS-treated spinal cords (Figure 5B).
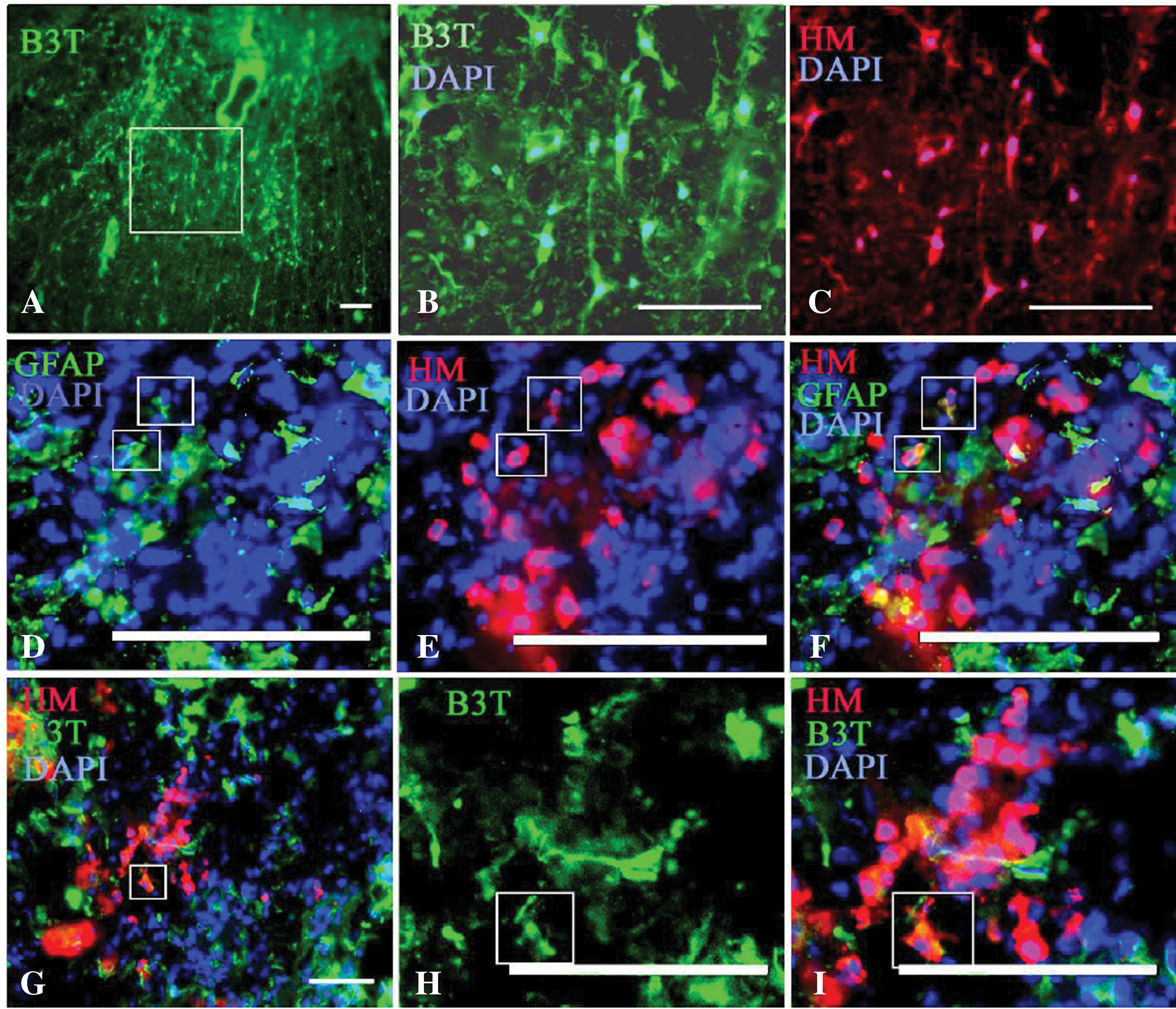
Transplanted NI hMSCs survived 2 weeks after transplantation and expressed several neural markers such as B3T (A-D), GFAP (E), and Sox2 (F). By 12 weeks, the number of surviving cells declined to 15% to 20% of that at week 2, and only 10% of survived cells were positive to B3T (G, H, I). The images H and I are higher magnifications of G
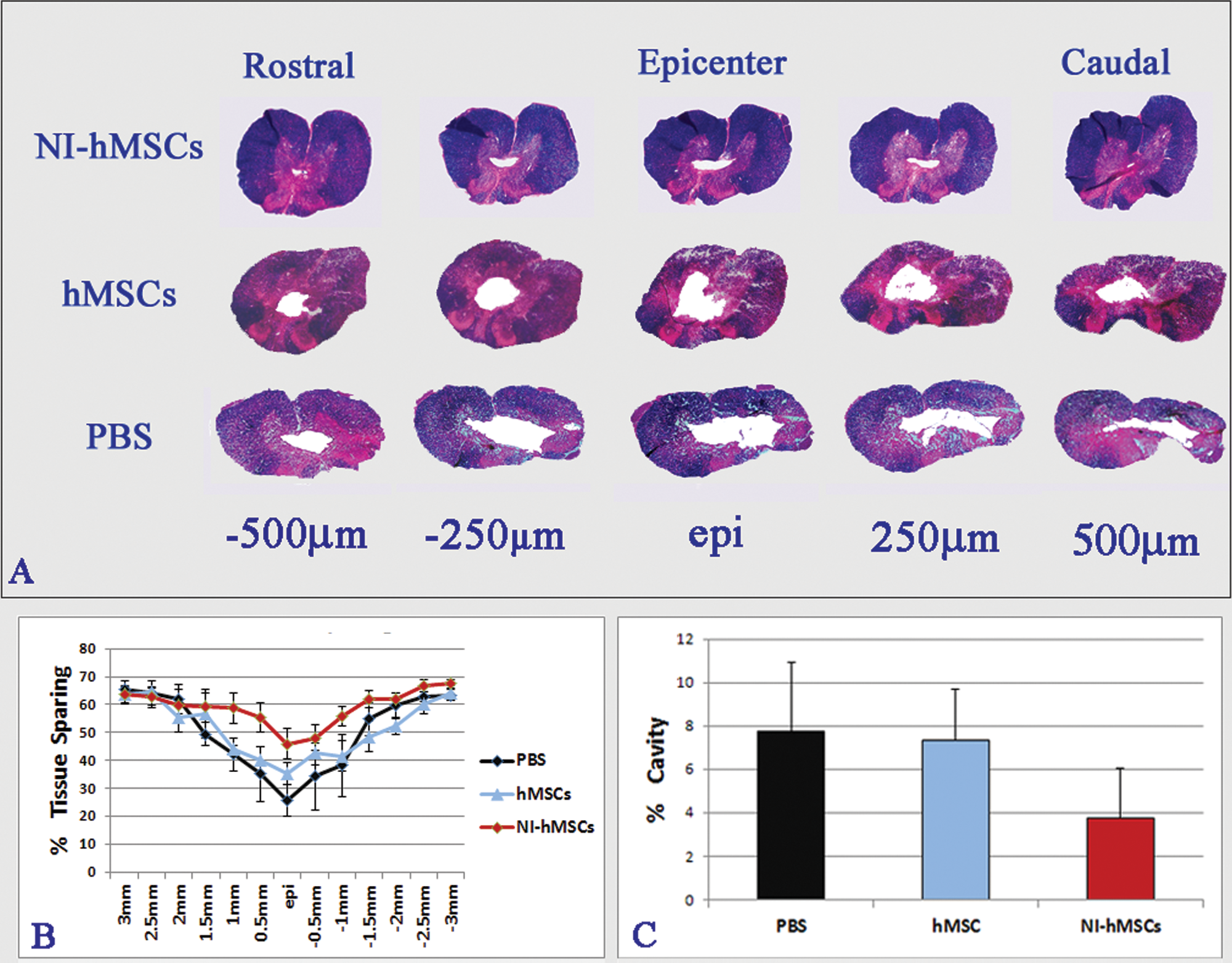
Analysis of white matter sparing and lesion cavity volumes in NI hMSC-, hMSC-, and PBS-treated groups: (A) Representative spinal cord cross-sections extending 500 µm rostral and caudal from the lesion epicenter. (B) Graph of the percentages of spared white matter through the entire T8 spinal cord segment. (C) Graph representing comparison of the volumes of lesion cavities
Stereological assessments of ISC tissues demonstrated a modest reduction in the percentage of cystic cavities in the NI hMSC–treated and hMSC-treated groups when compared with the PBS group (Figure 5C, Figure 6). Although no statistically significant difference was found between groups (Figure 5C), the difference found between NI HMSCs and PBS was very close to the significance level adopted in the study (

Representative 3-dimensionally reconstructed images of the lesion cavities through T8 ISC segments of NI hMSC- and PBS-treated animals
Discussion
Several hypotheses to account for MSC therapeutic benefits have been suggested, including neuroprotective effects from release or stimulation of growth factors and cytokines, the induction of neovascularization and axonal sprouting, modulation of immune response, and the replacement of damaged cells.18,27-29 Several in vitro neural induction protocols have been suggested.9,10,15,30-43
We developed an efficient method for generation of neural-like cells from BM-derived MSCs. 21 The novelty of this approach is simultaneous reactivation of pluripotent and neural genes by exposing MSCs simultaneously to inhibitors of DNA methylation and histone deacetylation and pharmacological agents that increase cAMP levels. Neural-like cells generated with this methodology exhibit numerous traits of neural cells and with further differentiation in appropriate conditions produce different neuronal and glial phenotypes.
Results presented in this study demonstrated that transplanted NI hMSCs survive, differentiate, and significantly improve locomotor recovery of severe ISC rats. Transplantation also reduced the volume of cavity and increased sparing of white matter as compared with the controls. The poor survival of NI hMSCs after 12 weeks of transplantation could be explained by type of transplant, which in this case was discordant xenograft (transplant between distantly related species). The level of immune response, and hence the risk of an immune rejection response, increases in magnitude when moving from an autologous transplant, to a syngeneic transplant, to a matched allograft, to a near-matched allograft, to an unmatched allograft, to a concordant xenograft, and finally to a discordant xenograft. Therefore, we can speculate that the rate of NI hMSCs survival and thus their beneficial therapeutic effect could be higher if NI hMSCs are used as autografts.
Thus, hMSCs neurally modified by this methodology may provide an alternative source of autologous adult stem cells to replace damaged neural cells in the injured or diseased CNS and provide support to CNS tissue.
Footnotes
Acknowledgements
We thank Christy Stadig, Kyle Stehlik, and Brad Peterson for technical assistance.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
This work is supported by AOSpina North America, Bryon Riesch Paralysis Foundation, VA Medical Research, and the Department of Neurosurgery of the Medical College of Wisconsin.
