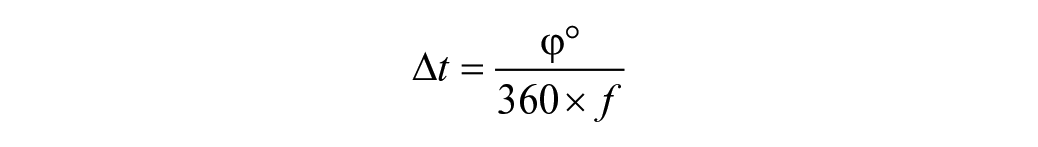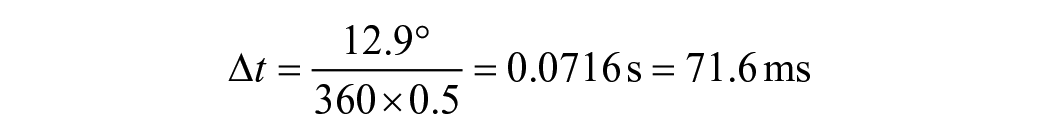Abstract
Aim. To compare reactive balance in people with multiple sclerosis (MS) with healthy controls and to examine the ability of people with MS to adapt their reactive balance and retain training effects. Data Sources. Electronic databases (PubMed, EMBASE, PsychINFO) and reference lists of included articles from inception to February 25, 2019. Study Selection. Case-control and intervention studies that assessed reactive balance using mechanical perturbations in people with a confirmed diagnosis of MS. Results. Meta-analyses of 9 studies (n = 342) showed that people with MS have significantly worse reactive balance than healthy controls (standardized mean difference [SMD] 0.78, 95% CI 0.44-1.11, P < .0001, I2 = 47%). Specifically, people with MS have greater center of mass displacements (SMD 0.41, 95% CI 0.05-0.77, P = .02, I2 = 9%) and longer response times (MD (ms) 31.45, 95% CI 19.91-42.98, P < .0001, I2 = 75%) in response to standing perturbations than healthy controls. Subsequent meta-analyses revealed training comprising repeated exposure to perturbations improved response times (P < .001) and training effects on response times could be retained for 24 hours (P < .001) in people with MS. Conclusions. Reactive balance assessments can highlight functional impairments related to falls in people with MS, and perturbation training can acutely improve reactive balance control and such improvements can be retained for 24 hours in this population. Systematic review registration number: CRD42019126130.
Keywords
Introduction
Multiple sclerosis (MS) is an immune-mediated disease that causes widespread demyelination of the central nervous system (CNS), including areas responsible for balance. 1 It is estimated that MS affects more than 2 million individuals worldwide, 2 with its prevalence observed to follow a geographical distribution peaking in North America and Western Europe. 3 People with MS have slowed somatosensory conduction and impaired central integration, leading to a loss of postural control. 1 As a result, they have a high risk of falls, with prospective studies estimating a >60% fall prevalence in people with MS over 3 months.4,5 Risk factors for falling in MS include physiological4,6-8 changes such as impaired dexterity, reduced lower limb strength, increased postural sway, poor vision, muscle weakness, numbness, and fatigue, as well as cognitive factors such as poor concentration, memory problems, and poor executive function.3,4,6-9 Those with higher Expanded Disability Status Scale (EDSS) scores fall more, with fall risk peaking at EDSS scores between 4.0 and 6.0. 9 Falls are a leading cause of bone fractures, 8 fear of falling,6,8 and activity curtailment,6,8 resulting in deconditioning, further falls and worsening disease prognosis.
Several studies have shown that people with MS have poor static balance control regardless of the task complexity or availability of sensory inputs such as vision as compared to their healthy counterparts. 10 As a result, interventions that target balance and functional training have been commonly used in clinical practice and research, and clinical trials have shown static balance can be improved in people with MS. 11 Balance control in daily life, however, also requires reactive strategies in response to postural perturbations like trips and slips. 12 Reactive balance refers to a person’s ability to maintain postural balance in response to a mechanical perturbation. It is triggered by detection of a postural threat, followed by a fast-acting reflex (response latency of 30-40 ms) that stiffens the joints, and longer latency reflexes (response latency of 70-90 ms) that are functionally more relevant.13,14 Impairment in reactive balance has recently been proposed as a key mechanism of frequent falls in MS, 15 and some studies have shown that people with MS have impaired responses to perturbations.16-20 However, these studies have used heterogeneous methods to induce reactive balance, and reactive balance impairment in MS has not been systematically quantified.
In both healthy young and older adults, reactive balance has been shown to be “adaptable” in that it can be rapidly improved by repeated exposure to perturbations. These adaptations may result from the CNS acquiring new sensorimotor strategies to enhance stability and cope with external challenges. In healthy people, these adaptations can be retained for up to 12 months, reducing the incidence of falls.21,22 One study has also reported that people with MS may have similar adaptability, 16 but more robust evidence is needed.
The purpose of this systematic review was to examine and quantify reactive balance in people with MS and their ability to adapt and retain reactive balance training effects. The quantification of reactive balance, its adaptability and retention in this population may assist in the development of neurorehabilitation strategies to prevent falls.
Methods
Registry of This Systematic Review Protocol
This review was written in accordance to the Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) guidelines. 23 A protocol was prospectively registered with the International Prospective Register of Systematic Reviews (PROSPERO) (registration number CRD42019126130).
Literature Search Strategy
Three electronic databases (PubMed, EMBASE, PsychINFO) were searched for articles published from their inception to February 25, 2019. Medical Subject Headings (MeSH) terms and key words that could identify studies on reactive balance in MS were chosen. The full search strategy can be found in the Supplementary Appendix A. There were no language restrictions applied during the initial search, and the reference lists of included articles were also screened for relevant articles.
Inclusion Criteria
Case-control studies and intervention studies conducted in any settings were included. The target population included participants with a confirmed diagnosis of MS. Studies that recruited participants with symptoms suggestive of MS but with no formal diagnosis were excluded. Studies that assessed reactive balance using mechanical perturbations (eg, platform surface translation) were included but studies using internal perturbations (eg, stepping or standing up) or nonmechanical perturbations (eg, sensory) were excluded. Primary outcomes were reactive balance measures in response to mechanical perturbations (eg, margin of stability, center of mass [CoM] displacement, response onset latency, step length, and number of steps to reach stability). Study protocols, conference abstracts, and articles published in languages other than English were excluded.
Screening Process and Data Extraction
Titles and abstracts of studies were retrieved using the search strategy as well as those obtained from additional sources were screened independently by 2 authors to identify studies that potentially met the inclusion criteria outlined above. The full texts of these potentially eligible studies were then independently assessed for eligibility by 2 authors. Any disagreements were resolved through discussion with a third author. A standardized form was used to extract data from the included studies for assessment of study quality and evidence synthesis. Extracted information included study population, participant demographics, study setting; reactive balance assessment protocol; details of the intervention (if applicable); study design; outcomes at pre- and postintervention; information for assessment of the risk of bias. One author extracted data, and any ambiguities were resolved through discussion with 2 other authors as necessary. Missing data required for meta-analyses were requested from original study authors.
Methodological Qualities
A customized methodological quality scale was designed to assess the internal, statistical, and external validity of the included studies with regard to the conceptual definition of the present review. These items are presented in detail in Supplementary Appendix B. One point was assigned for each quality criterion fulfilled (Supplementary Appendix B). However, if a criterion was not relevant (eg, the study did not include healthy controls), no score for this criterion was given. A combined quality score was not calculated due to unequal criterion weights. Quality assessment was performed independently by 2 authors and in case of disagreement, a third author was consulted.
Statistical Analyses
Analyses were conducted using Review Manager (RevMan, V.5.3.; The Nordic Cochrane Centre, The Cochrane Collaboration). Where studies used the same type of outcome measure, the results were pooled using a random-effects meta-analysis, with 95% confidence intervals (95% CI) and 2-sided P values. The pooled mean differences (MDs) for continuous outcomes with the same measurement units and standardized MDs (SMDs) for continuous outcomes with different measurement units were calculated. Heterogeneity between the studies in effect measures was assessed using both the χ2 test and the I2 statistic. An I2 value greater than 50% is considered indicative of substantial heterogeneity. P values <.05 were considered statistically significant.
Results
Studies Included
Our selection process is described in Figure 1. The initial search yielded 129 articles, which were screened with their titles and abstracts. Of these, 26 full-text articles were obtained, and 11 articles were considered eligible after applying the selection criteria. Of these, 2 articles reported findings from the same study. Thus, a total of 10 studies were included in our review. Two original study authors were contacted and provided necessary details and/or unpublished data.
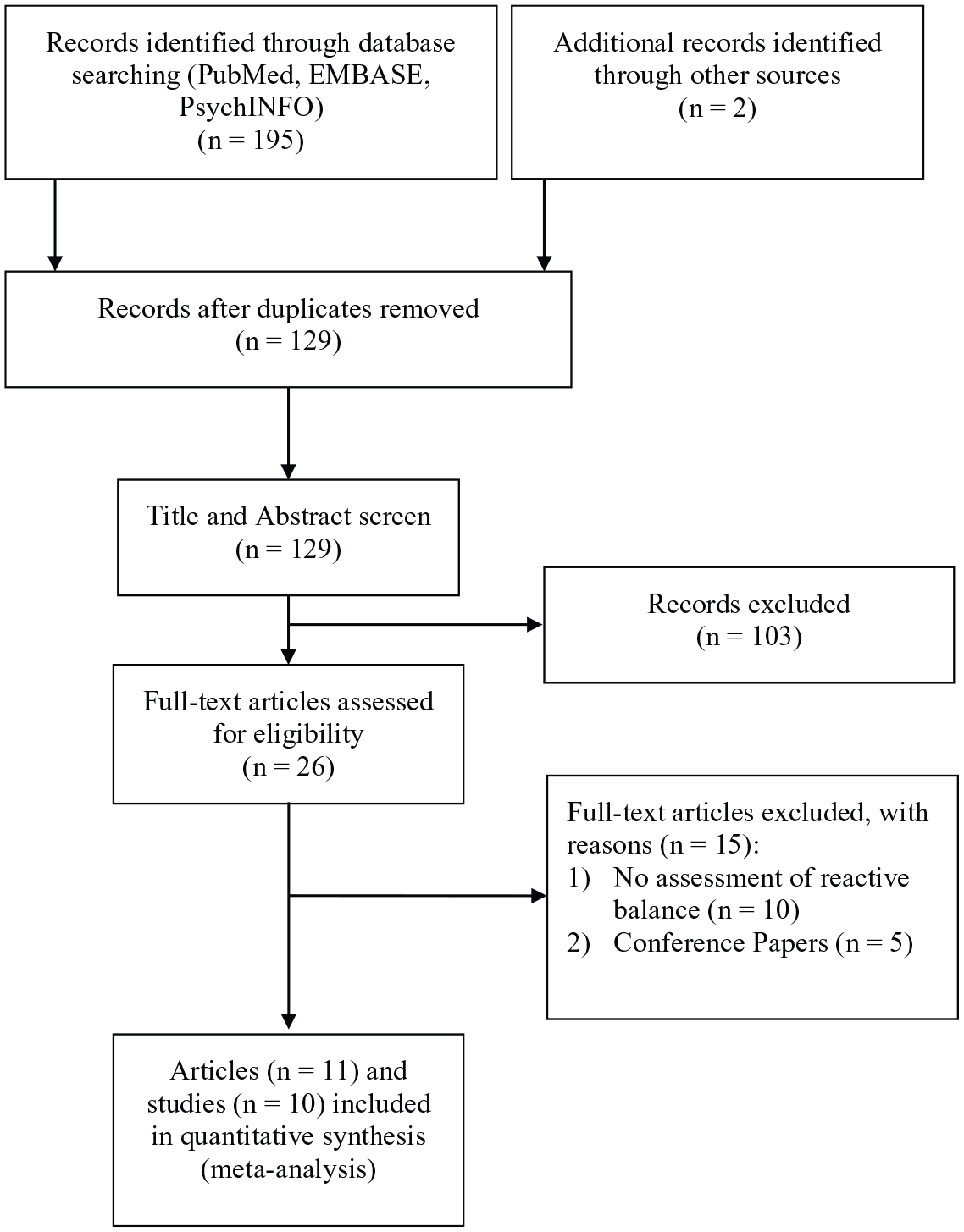
Flowchart of the study selection process.
Description of Included Studies
The summarized methodology of the included studies is presented in the Supplementary Appendix C. Eight studies were cross-sectional studies,15-17,19,20,24-26 and 3 studies were intervention studies.18,27,28 Ten studies compared participants with MS with healthy control participants,15-20,24-26,28 and 3 studies studied the acute effect of reactive balance training in MS either with or without a comparison group of healthy controls.18,27,28 Two of these later studies examined retention of the training effect over 2 days in MS and healthy control participants.18,28
To evoke reactive balance, 6 studies used support surface translations,15,17,18,20,25,28 2 used pendulum weights,16,27 1 used a push and release test, 24 1 used a pulley-based system, 26 and 1 used a toe up rotation of the support surface. 19 All perturbations were applied while participants were standing still. One intervention study used repeated ball throwing exercises as training over 1 day. 27 Two intervention studies induced their training effects by repeated exposure to external perturbations over 5 trial blocks on one day and conducted posttraining testing in one trial block the following day to examine short-term retention.18,28 In these intervention studies, Gera et al 18 administered continuously oscillating feet-in-place perturbations on a standing platform, while Van Liew et al 28 administered a fast translating standing platform to induce a stepping response in their participants. Reactive balance parameters reported in the studies included CoM displacement, response onset latency, step length, and number of steps to recovery. Response onset latency parameters included muscle activation onset, step initiation times and time to peak center of pressure displacements. For Gera et al, 18 with the knowledge that the platform oscillated at a fixed frequency of 0.5 Hz sinusoidally, onset latency was converted from relative phase using the equation:
where t represents time in seconds, ϕ° represents the relative phase in degrees, and
We interpret this result as the CoM lagging an average of 71.7 ms behind the platform as it is oscillating, of which reflects the onset latency component of reactive balance in the participants of this study.
Methodological Quality
Results of the methodological quality are presented in the Supplementary Appendix C. In terms of internal validity, fatigue (common in MS) was minimized in 1 study by conducting all experiments in the morning, 18 and in 5 studies by providing breaks.16,20,24,27,28 The perturbation methods used were standardized except in 1 study, 24 unpredictable in timing except in 2 studies,18,27 but were not unpredictable in magnitude in 6 studies16,19,24-27 and not unpredictable in type or direction in 8 studies.16-18,20,24-27 In terms of statistical validity, 1 study 28 did not report values that could be used to calculate effect sizes. In terms of external validity, 1 study 17 did not specify the eligibility criteria, and 5 studies16,17,19,27,28 had sample sizes smaller than 30.
Impact of MS on Reactive Balance
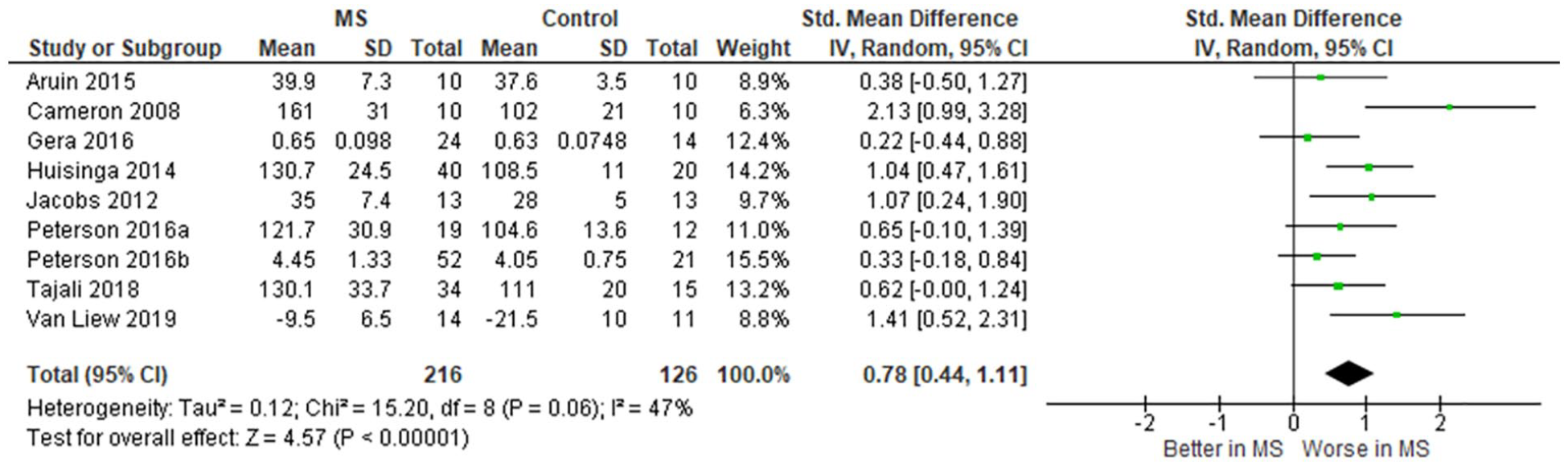
Nine studies (n = 342)15-20,25,26,28 comparing participants with MS and healthy controls in terms of reactive balance assessed by either CoM displacement, margin of stability response or onset latency were included in the meta-analysis (Figure 2). These results show that reactive balance is significantly worse in people with MS (SMD 0.78, 95% CI 0.44-1.11, P < .0001, I2 = 47%).
MS–Healthy Control Comparisons for Specific Reactive Balance Outcome Measures
MS–healthy control comparisons for specific reactive balance measures are shown in Figure 3. CoM displacement was significantly worse in people with MS compared with healthy controls (SMD 0.41, 95% CI 0.05-0.77, P = .02, I2 = 9%)—4 studies (n = 157).15,16,18,19 Onset latency response times were significantly longer in people with MS compared with healthy controls (MD [ms] 31.45, 95% CI 19.91-42.98, P < .0001, I2 = 75%)—9 studies (n = 342).15-20,25,26,28 Finally, step lengths during recovery after loss of balance were (nonsignificantly) shorter in people with MS compared with healthy controls (MD [cm] −8.34, 95% CI −18.50 to 1.83, P = .11, I2 = 81%)—2 studies (n = 98).15,28
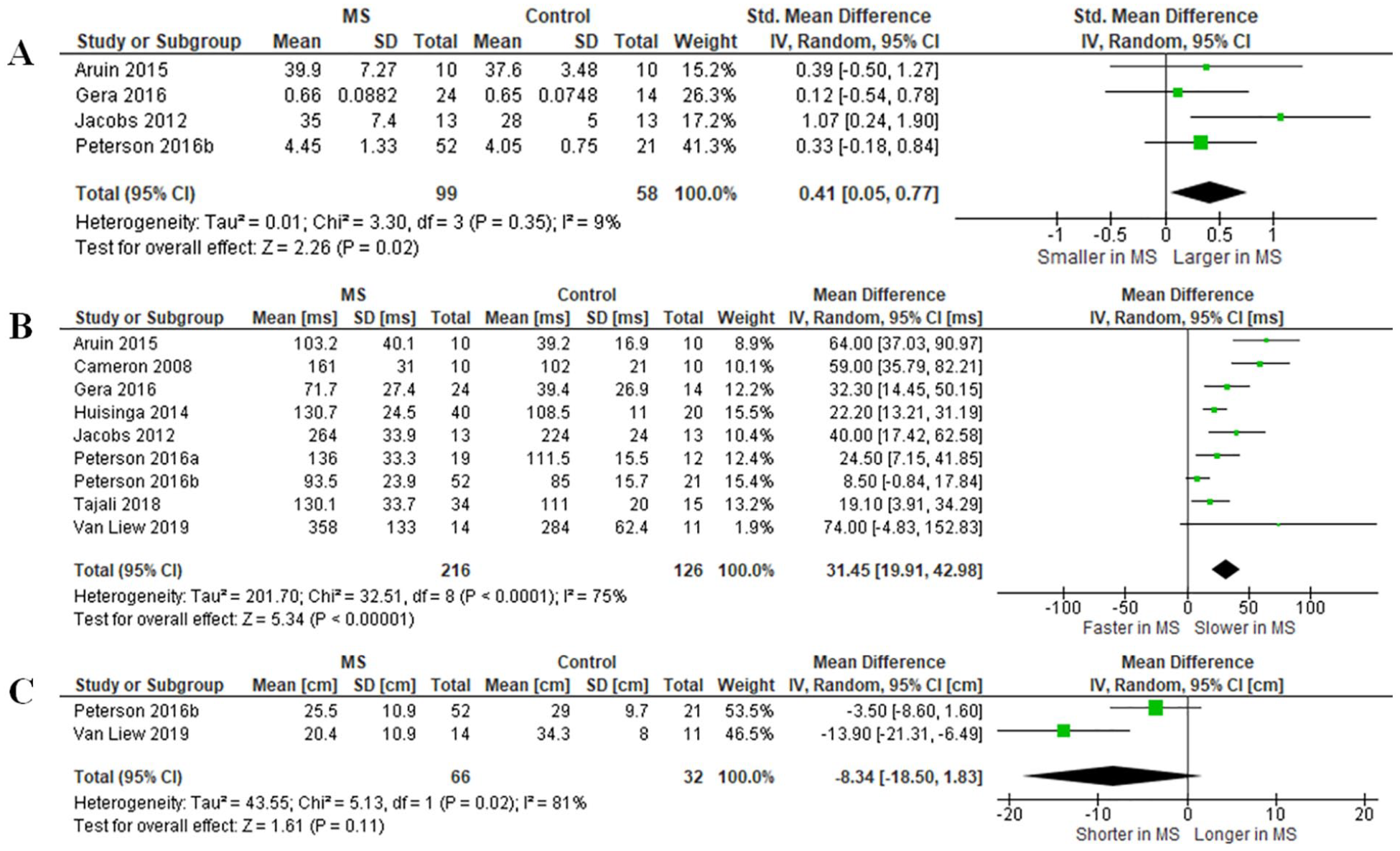
Comparisons between (A) center of mass (CoM) displacement, (B) onset latency, and (C) step length between participants with multiple sclerosis and healthy controls. Onset latency parameters included muscle activity onset (Aruin 2015 [tibialis anterior (TA), vastus lateralis (VL), and rectus abdominis (RA) combined], 16 Cameron 2008 [medial gastrocnemius (MG)], 17 Huisinga 2014 [TA], 25 Peterson 2016a [MG], 15 Peterson 2016b [TA], 20 and Tajali 2018 [rectus abdominis (RA), rectus femoris (RF), and TA combined] 26 ), step latency (Van Liew 2019) 28 and time to peak center of pressure displacement (Jacobs 2012 19 and Gera 2016 28 [relative phase lag in degrees converted to time shift between platform and body]).
Acute Effect of Training in Participants With MS and Healthy Controls
Two studies (n = 63)18,28 contrasting the acute effect of training on onset latency response times between participants with MS and healthy controls were included in the meta-analysis (Figure 4). Acute training significantly improved onset latency response times in both participants with MS (MD [ms] 36.11, 95% CI 17.61-54.60, P = .0001, I2 = 0%) and healthy controls (MD [ms]: 33.25, 95% CI 12.31-54.19, P = .002, I2 = 0%). There was no significant between-group training effect difference (P = .84).
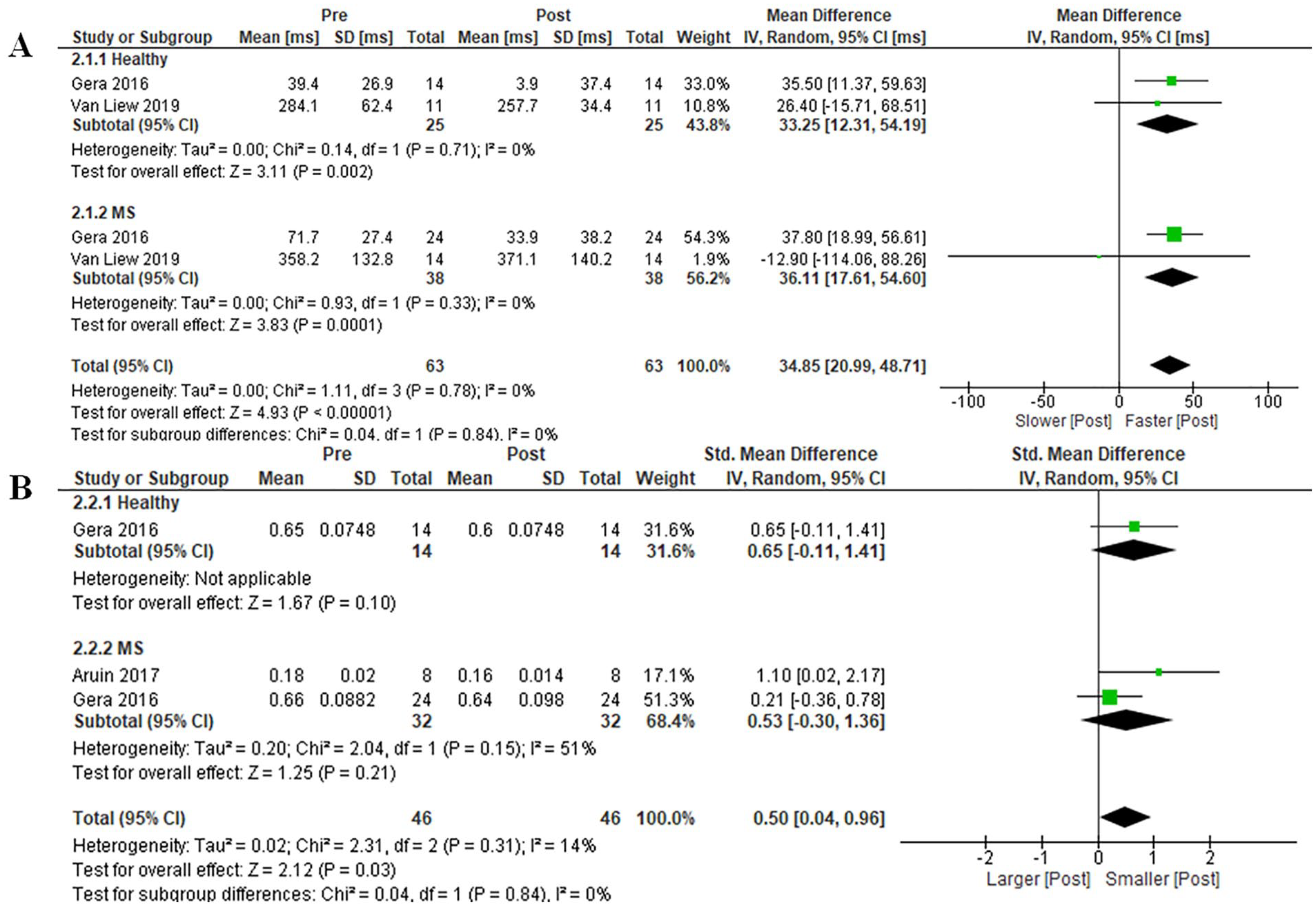
Acute training effects on (A) onset latency and (B) center of mass displacement in people with multiple sclerosis and healthy controls.
Two studies (n = 46)18,27 reporting acute effects of training on CoM displacements were included in the meta-analysis. Acute training did not significantly improve CoM displacement in either participants with MS (SMD 0.53, 95% CI −0.30 to 1.36, P = .21, I2 = 51%) or healthy controls (SMD 0.65, 95% CI −0.11 to 1.41, P = .10). There was no significant between-group training effect difference (P = .84).
Retention of Training in Participants With MS and Healthy Controls
Two studies (n = 63)18,28 contrasted retention of training on onset latency response times between participants with MS and healthy controls (Figure 5). On day 2 of training, onset latency response times were significantly improved in both participants with MS (MD [ms] 39.71, 95% CI 25.21-54.21, P < .0001, I2 = 0%) and healthy controls (MD [ms] 28.94, 95% CI 11.19-46.69, P = .001, I2 = 0%). There was no significant subgroup difference between the 2 groups (P = .36).
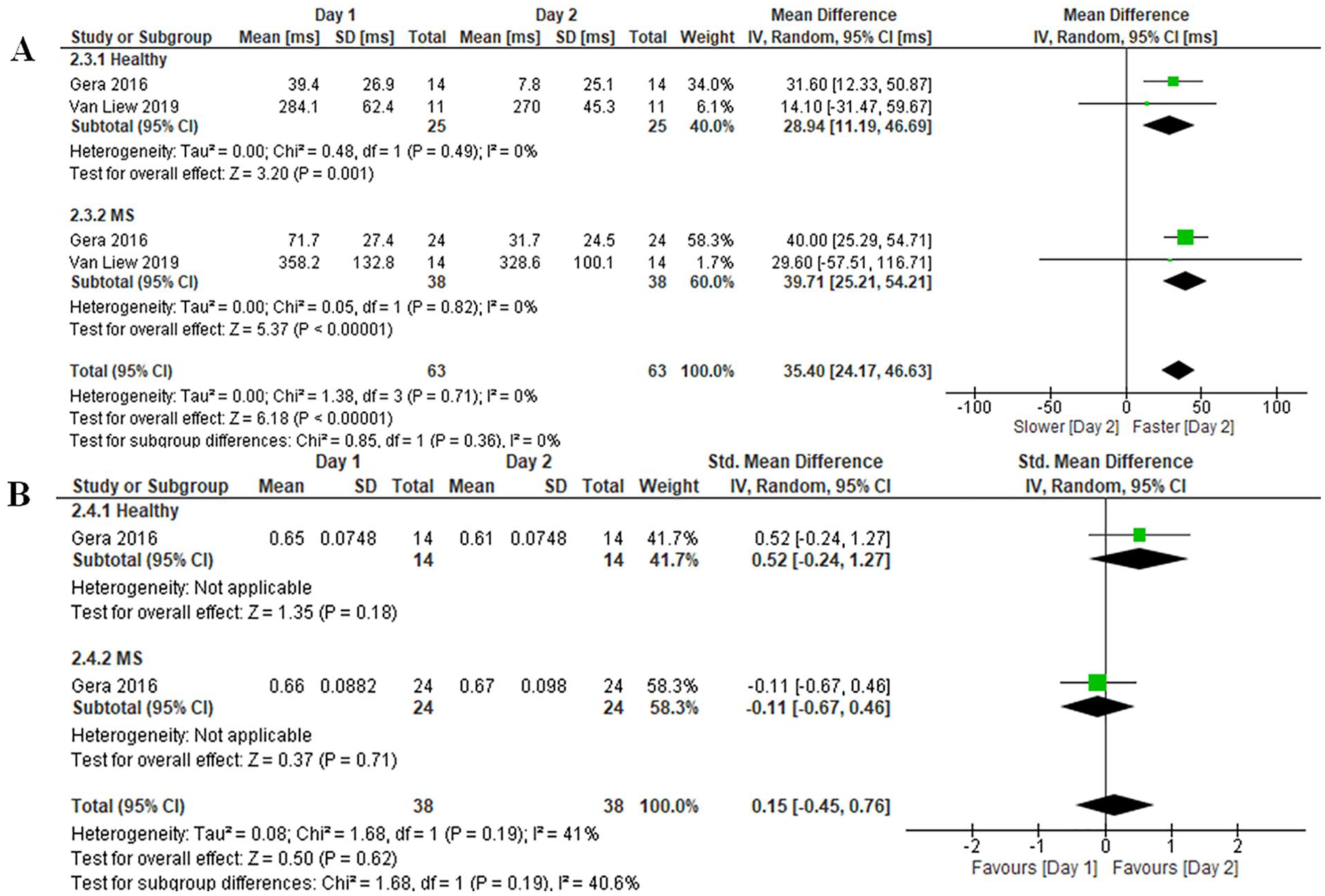
Retention of training effects on (A) onset latency and (B) center of mass displacement in people with multiple sclerosis and healthy controls.
One study (n = 38) 18 contrasted CoM displacement outcomes between MS and healthy controls. On day 2 of training, CoM displacement outcomes were not significantly improved in either participants with MS (SMD −0.11, 95% CI −0.67 to 0.46, P = .71) or healthy controls (SMD 0.52, 95% CI −0.24 to 1.27, P = .18), with no significant subgroup difference between the 2 groups (P = .19).
Subgroup Analyses
Subgroup analyses of the reactive balance parameters stratified by participants’ disability status (mean EDSS <4 vs Mean EDSS 4+), type of perturbation (perturbation to base of support [BoS] vs onset latency measurements [electromyography, EMG vs non-EMG] vs perturbations to CoM) used in the studies are presented in Supplementary Appendices E, F, and G. The MS samples were categorized based on gait impairment, that is, average EDSS scores <4 and ≥4.
Onset latency response times in studies with average EDSS scores <4 MD (ms) and those with average EDSS scores ≥4 were 39 ms (95% CI 25-52, P < .0001, I2 = 61%) and 15 ms (95% CI 2-29, P = .02, I2 = 77%), respectively. This significant subgroup difference (P = .02) indicated the between-group difference (MS vs healthy controls) was larger in the studies with average EDSS scores <4 compared with the studies with average EDSS scores ≥4 (Appendix E). However, when studies that used non-EMG measurements were excluded, this subgroup difference was no longer significant (P = .06). All other reactive balance parameters showed no significant differences between the subgroups (Appendix F).
Stratified by EMG and non-EMG measurements of onset latency, between-group differences (MS vs healthy controls) among studies using EMG (MD [ms] 29.35, 95% CI 15.47-43.24, P < .0001, I2 = 82%) and studies using non-EMG methods (MD [ms] 31.45, 95% CI 19.27-43.63, P < .0001, I2 = 0%) were similar (Appendix G). This subgroup difference was not statistically significant (P = .82).
Sensitivity Analysis
A sensitivity analysis was conducted in which 1 study 18 that used continuous perturbations as opposed to a single discrete perturbation was excluded. This revealed no changes in the findings with respect to reactive balance (SMD 0.85, 95% CI 0.50-1.20, P < .0001, I2 = 45%), CoM displacement (SMD 0.52, 95% CI 0.09-0.95, P = .02, I2 = 13%) and onset latency (MD [ms] 31.66, 95% CI 18.82-44.51, P < .0001, I2 = 78%). A sensitivity analysis for the intervention studies could not be conducted because there were only 2 studies18,28 in this group.
Discussion
Reactive Balance Outcomes
Our review quantitatively synthesized the evidence on reactive balance in people with MS, and their ability to rapidly adapt and retain training effects. Our findings demonstrate that people with MS have significantly larger peak CoM displacements (SMD 0.41) and worse reactive balance as determined by all reactive balance outcome measures when compared with healthy controls (SMD 0.78). These findings complement the findings of a recent systematic review, 10 which found that people with MS have significantly larger CoM displacements during static postural balance tests, and indicate that people with MS also have reduced ability to regain their stability when exposed to external perturbations such as a sudden displacement of the support surface15,18,19 or to CoM. 16 The larger peak CoM displacements in response to a perturbation may result from an inappropriate scaling of corrective responses against the perturbation and a prolonged response latency in people with MS which allows the CoM to deviate further. This aligns with our findings that people with MS required approximately 30 ms longer than healthy controls (Figure 3B) to detect and initiate balance recovery responses.
The prolonged response times in people with MS can be attributed to the pathways involved in generating automatic postural reflexes, which consist of the initial part of compensatory postural corrections. Automatic postural reflexes include short-latency (SL), medium-latency (ML), and long-latency (LL) components that are part of a transcortical loop that produce a response to an external perturbation. 29 SL responses tend to be nonfunctional (eg, a monosynaptic stretch reflex) and last around 50 ms. 30 ML and LL responses are functional and take longer, as they convey proprioceptive and sensory information up the dorsal column 31 into the brainstem. LL responses continue to travel to the primary somatosensory cortex in the postcentral gyrus of the parietal lobe. Here, the information is processed, and a subsequent motor command is transmitted from the motor cortex down the corticospinal tract to produce a postural response. 30 In our subgroup analysis, mean muscle activation (EMG) onset latency values in the MS participants ranged from 94 to 161 ms (Appendix G). This suggests that these are ML and LL responses, that is, the first physiological response functionally relevant for responding to the external perturbations. This is consistent with previous work by Diener et al, 32 who showed that people with probable MS had significantly delayed LL responses, that is, >164 ms.
Related research 17 has also found that there is approximately a 10-ms difference in spinal conduction times between people with MS and healthy controls, providing evidence for reduced nerve conduction velocities of people with MS due to demyelination. In addition, reduced tactile and proprioceptive sensitivity, and reduced functional connectivity such as the cortico-cerebellar and cortico-striatal motor loops 33 in people with MS may also contribute to the prolonged onset latency. These findings suggest that MS delays sensory transmission, central processing and subsequent motor commands to the effector muscles, leading to the slower response times and reactive balance deficits in this group. As such, people with MS appear to be unable to respond efficiently to external perturbations, producing shorter steps (MD [cm] −8.34) and requiring more steps to recover. 15
Adaptability to Training
Our meta-analysis also demonstrated that people with MS retain the adaptability and significantly improve their response times (MD [ms] −36.11) after being exposed to repeated standing perturbations (25-35 trials). This concurs with findings from a previous meta-analysis 34 on the effect of repeated perturbation training on reactive balance in younger and older healthy adults (SMD +1.22). This was also supported by a recent study of 13 people with MS by Yang et al, 35 which was published after the completion of our review search. This study found that during five repeated slip-like perturbations induced by sudden reverse rotations of a treadmill belt, dynamic stability significantly improved through feedforward mechanisms including reduced step length and flat foot landing prior to slip onset. 35
Motor control adaptation relies on 2 main mechanisms; feedback (reactive) and feedforward (predictive). 36 In the feedback mechanism, sensory input from the periphery is processed, and the brain learns to adapt to these environmental challenges by fine tuning its motor commands. 36 On the other hand, in the feedforward mechanism, the brain uses prior knowledge or experience to predict sensory consequences through repeated exposure to environmental challenges, and fine tune its motor commands and preset the corticospinal excitability to respond to these perturbations efficiently.36-38 Among the 3 studies included in the meta-analyses for acute training effects, Gera et al 18 (forward and backward surface translation in sinusoidal oscillation) and Aruin et al 27 (stopping a pendulum) used perturbations that were predictable in 2 of the 3 elements, that is, magnitude, timing, and type/direction (Supplementary Appendix D). This would have allowed participants to learn and anticipate the magnitude and type of perturbations in the repeated trials. Thus, it is likely that “feedforward” is the main mechanism for the improved adaptability observed in the participants.
Our findings therefore suggest that motor adaptability—especially the feedforward mechanism—is intact in people with MS. This strengthens perturbation training’s position as a form of neurorehabilitation for people with MS. However, the acute improvements in CoM control through repeated standing perturbations in both people with MS and healthy control were only of moderate size (CoM displacement SMD +0.65 vs +0.53) and did not reach statistical significance. It may also indicate that the training dose (25-35 trials) in a single session is not sufficient to improve reactive control of CoM in response to sudden perturbations. The nonsignificant finding in this regard also relates to the small sample size of the included studies, thus further intervention studies with greater sample sizes are required to examine training effects in people with MS.
While feedforward and feedback balance control mechanisms are both required in daily activities, 12 future research should investigate training of the “feedback” mechanism which does not rely on prediction of a possible perturbation. This requires exposure to perturbations that remain highly unpredictable even in repeated trials and may require a greater training dose in multiple sessions.39,40 In our training analysis, only Van Liew et al 28 used perturbations unpredictable in magnitude, timing and direction that may be suitable to train reactive balance via a feedback mechanism. We also saw heterogeneity in perturbation methods. Thus, more research is required to determine the optimal methodology to train reactive balance in people with MS.
Retention of Training Effects
Finally, our meta-analyses demonstrated significant retention of the acute training effect for 24 hours in both people with MS and healthy controls in terms of response times. These results are encouraging, as it shows that a dose of 25 to 35 repeated perturbation trials18,28 can produce significant adaptation which can be retained in people with MS. Previous studies trialing a single session of repeated slip 21 or trip-like 41 perturbations in healthy older adults showed that participants were able to retain the most training effects after 6 to 18 months with additional benefit of booster sessions (e.g. every 3 months). 42 Such long-term retention is likely attributable to the previous knowledge of the perturbation and its consequences that remained to influence the motor command and muscle synergies based on a feedforward model. 36 While it is possible that people with MS also can also retain the training effect for longer than 24 hours, this must be determined in future studies. The promising findings in our meta-analysis therefore highlight the need for more studies to determine long-term retention, optimal training doses and frequency to maximize the training effects and prevent falls in people with MS. In addition, as the studies included in this review mostly focused on perturbations to standing balance, future studies could investigate how reactive balance training during gait, such as exposure to trips and slips, affects reactive balance outcomes in people with MS.
Limitations
Our systematic review and meta-analysis were conducted following the PRISMA guidelines, including prospective registration of the review protocol and the acquisition of unpublished data. However, we acknowledge some limitations. First, there is the possibility that we may have omitted relevant articles due to different terminologies used for reactive balance. Second, many of the included studies had small samples, with 5 studies16,17,27,28,43 having fewer than 30 participants. Third, there was substantial heterogeneity in our analyses (Figure 2), possibly due to the differing study methodologies. Fourth, although previous studies have indicated cerebellar lesions might impair relearning motor programs and affect the feedforward mechanism,44-46 our review findings do not provide evidence of cerebellar involvement. Finally, the included studies involved participants with relatively low EDSS scores (<5), which reduces the generalizability of our findings to people with greater MS disease severity.
Clinical Implications
Our findings demonstrate that reactive balance is significantly impaired in people with MS. Therefore, a reactive balance assessment may assist in the clinical assessment of balance control and fall risk in people with MS. Moreover, as exposure to repeated perturbations can rapidly improve reactive balance in people with MS (primarily via feedforward mechanisms), reactive balance training may complement existing neurorehabilitation interventions to maximize their efficacy in preventing falls in this population. The feasibility and possible efficacy of neurorehabilitation using repeated perturbations in other clinical populations such as those with Parkinson’s disease and stroke have also been suggested and require further research. 47
Conclusion
People with MS have impaired reactive balance as compared with the general healthy population. Our findings indicate that repeated perturbation training can significantly improve reactive balance parameters and these effects can be retained for 24 hours. Further studies are needed to determine the dose-response relationship of such a training method in people with MS, and to understand the underlying neurophysiological mechanisms responsible for motor learning and retention in this population.
Supplemental Material
Supplementary_materials – Supplemental material for Reactive Balance Adaptability and Retention in People With Multiple Sclerosis: A Systematic Review and Meta-Analysis
Supplemental material, Supplementary_materials for Reactive Balance Adaptability and Retention in People With Multiple Sclerosis: A Systematic Review and Meta-Analysis by Mohamed Suhair Bin Mohamed Suhaimy, Yoshiro Okubo, Phu D. Hoang and Stephen R. Lord in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We thank the authors who kindly provided the unpublished data and/or clarifications for this systematic review and meta-analysis.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.