Abstract
Background. The ability to adapt, a form of short-term motor learning, and retain this adaptation, is essential for rehabilitation and for day-to-day living. Yet little research is available on this topic in persons with multiple sclerosis (PwMS), particularly in relation to complex walking tasks. Objective. To determine the ability of PwMS to learn and retain a novel relationship between visual input and motor output—or visuomotor map—during visually guided walking. Methods. Nineteen PwMS and 17 healthy controls performed a precision walking task while adapting to prism lenses that altered the normal visuomotor map on 1 day, and again after a 1-week delay. The task required individuals to walk and step onto 2 targets without stopping. To quantify motor performance, we determined foot placement error relative to the targets. Results. PwMS with mild disability and healthy controls attenuated foot placement error over repeated trials when exposed to the novel mapping and demonstrated a similar rate and magnitude of adaptation in the first learning session. Both groups equally retained the adaptation 1 week later, reflected by reduced foot placement error and a faster rate of error reduction in that session. Conclusion. PwMS can learn and retain a novel visuomotor mapping during a precision-based walking task. This suggests that PwMS with mild disability have the capacity for short-term motor learning and retention, indicating that neural plasticity is preserved.
Introduction
Walking ability and visual function are major concerns among persons with multiple sclerosis (PwMS) and are noted as the most important attributes to functional independence and quality of life.1-3 Impairments in walking range from reduced speed to greater joint angle variability to changes in spatial-temporal step metrics.4-10 Many of these gait impairments are evident even in PwMS with minimal disability.4,6,7,9 Visual impairments are also common among PwMS, often secondary to optic neuritis.11,12 These factors, among other sensorimotor deficits, contribute to the high risk of falling in this population.13-15 For instance, variability in the location of foot placement while walking is significantly greater in PwMS classified as recurrent fallers compared to nonfallers as well as healthy controls. 8 Rehabilitation strategies may help improve motor function despite these issues, and the development of these interventions would likely benefit from a better understanding of motor learning in this population.
Spontaneous recovery of function and rehabilitation rely on the nervous system’s capacity for neural plasticity and its ability to adapt. Sensorimotor adaptation—one form of motor learning—involves modifying movement patterns based on altered sensory input and/or motor function as a result of neurological injury or novel environmental constraints.16,17 The brain may use the error between the predicted outcome of a movement and the actual observed outcome to drive the adaptation.16,18 This process occurs relatively rapidly over repeated movements to achieve a certain level of performance, and which we define here as short-term motor learning. When adaptation occurs because of neurological injury, it is desirable for both the patient and the therapist that performance gains are retained over days, weeks, months, and so on. Maintaining these abilities is thus important for PwMS to attain benefits from rehabilitation.
However, research on motor learning in PwMS is limited, has focused predominantly on upper limb or standing balance tasks, and has largely neglected longer-term retention. These studies have generally found that motor learning is preserved in PwMS when compared to healthy controls,19-24 with a few exceptions,21,25,26 despite many showing worse motor performance in PwMS on the specific task at baseline and/or throughout testing. For instance, Casadio et al 19 demonstrated that PwMS with no or minimal clinical disability are able to adapt to a velocity-dependent force field in a reaching task similarly to controls, although with greater movement variability. In addition, PwMS can learn an upper limb–based visuomotor tracking task to the same extent as controls. 23 Given the importance of visually guided walking for community ambulation, it is prudent to study motor learning in PwMS in this context as well.
The ability to coordinate visual information with movement is normally important to interact with the environment. Indeed, visually guided walking requires the brain to maintain an accurate relationship (or visuomotor mapping) between the perceived stepping location and the motor command necessary to direct the foot to that position on the ground with minimal error.18,27-29 This is crucial to safely negotiate changes in terrain and obstacles, where appropriate foot placement is paramount.30,31 Interestingly, PwMS with mild-to-moderate disability walk with greater head pitch downward and with the head and body center of mass positioned closer to the toe of the stance foot when in swing phase.32-34 These authors suggest that this strategy allows PwMS to better visualize foot placement.
In this study, to study motor learning in PwMS, we used prism lenses to alter the visuomotor mapping during a precision walking task that involved stepping to the center of targets. This led to errors in foot placement on initial exposure. Specifically, we tested the hypotheses that PwMS are able to adapt to, and retain over a 1-week period, a novel visuomotor mapping but will adapt more slowly and exhibit less retention compared to healthy controls. The ability to predict which patients are more likely to show greater motor learning may help clinicians prescribe and administer better rehabilitation protocols, both in terms of type and duration. Thus, we also tested the hypothesis that clinical measures used to characterize function in PwMS predict retention capability.
Materials and Methods
Participants
Nineteen PwMS (mean age = 42.1 ± 9.1 years, range = 22-54 years; 12 females; 17 relapsing-remitting type, 2 primary-progressive type) recruited from the Fraser Health Multiple Sclerosis Clinic (Burnaby, Canada) and 17 healthy controls of similar age and gender (mean age = 41.6 ± 11.0 years, range = 23-54 years; 10 females) recruited from the Simon Fraser University community and surrounding area participated in this study. We used a convenience sampling method to recruit both groups. The Fraser Health Research Ethics Board and Office of Research Ethics at Simon Fraser University approved this study, and all participants gave informed written consent before testing.
The inclusion criteria for PwMS included (1) clinically diagnosed multiple sclerosis based on the McDonald Criteria; 35 (2) free of another neurological or musculoskeletal disorder, or uncorrected vision problem that impaired walking; and (3) an Expanded Disability Status Scale (EDSS) score below 4.0. We chose to recruit PwMS with mild disability to minimize the impact of motor weakness interfering with their ability to perform the task. We included controls if they had no musculoskeletal or neurological disorder and no uncorrected vision problem.
Experimental Task and Protocol
Participants performed a precision walking task27-29 characterized by walking across a 6-m path and stepping onto the center of 2 targets (15 × 30 cm) without stopping (Figure 1A). The second target was located at a distance of 90% of the participant’s height of the greater trochanter from the floor and a 30° counterclockwise angle with respect to the plane of progression.
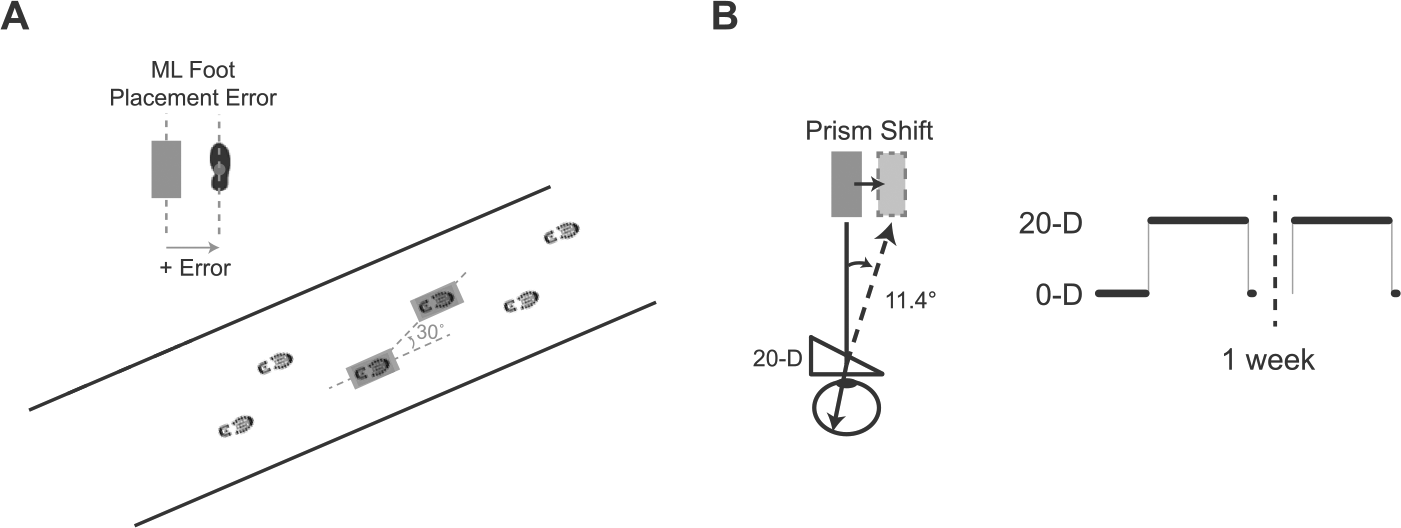
Experimental setup and protocol. (A) Schematic of the visually guided walking task. PwMS and controls walked and stepped onto 2 targets on the ground. Medial-lateral foot placement error, defined as the distance between markers on the foot and center of the targets, quantified performance. (B) A simulated view of the target through the prism lenses, and the perceived target shift for 20-diopter (20-D) lenses. In this protocol, subjects experienced 25 baseline trials, 60 adaptation trials (denoted by the step impulse in the illustration), and a single postadaptation trial on day 1. One week later, subjects experienced the same 60 adaptation trials followed by 5 postadaptation trials.
During the precision walking task participants wore goggles with either 20-D (11.4°) wedge prism lenses that shifted the visual perception of the target to the right with respect to its actual location (see Figure 1B), or flat lenses that did not shift perceived target position. The prism lenses altered the participant’s normal visuomotor mapping, inducing movement errors when walking and stepping onto the perceived location of the targets. The goggles reduced a portion of the visual field such that they forced participants to look only through the lenses. Participants knew the goggles altered their vision, but they did not know how. The prisms enabled us to study short-term visuomotor adaptation and retention in a motor behavior (ie, walking) typically impaired and a primary concern in PwMS.
In each trial, participants started at random anterior-posterior locations within a 1.8- to 3-m distance from the first target to prevent them from learning a specific stepping sequence and to ensure that the task remained under visual guidance. We provided several (standardized) instructions to participants. Specifically, we instructed participants to step in the medial-lateral center of the targets and to not stop until taking at least one step after the second target. Although difficult, it is possible to bypass the effects of the prisms via rapid corrections of leg/foot trajectory during the step to a target if a person moves very slowly. Thus, we also instructed participants to walk at a quick and constant pace to minimize the possibility of these so-called online corrections of leg/foot trajectory. Participants walked with an average speed (± SD) of 1.14 ± 0.31 m/s (PwMS) or 1.23 ± 0.24 m/s (controls), and we verified the absence of sudden changes in foot-marker trajectory during steps to the targets to confirm the lack of online corrections. We also told participants to look down to see their feet make contact with the targets but provided no specific instruction to look anywhere as they approached. Participants kept their eyes closed before each trial and again when walking back (under experimenter guidance) to the start position to avoid adaptation between trials.
Participants experienced 25 baseline trials (with flat, 0-D lenses), 60 adaptation phase trials (with 20-D lenses), and one postadaptation trial (with flat, 0-D lenses) in the first testing session (Figure 1B). In the second testing session 1 week later, participants experienced the same 60 adaptation trials followed by 5 postadaptation trials (with 0-D lenses). An Optotrak Certus motion capture camera (Northern Digital Inc, Waterloo, Ontario, Canada) recorded infrared-emitting diodes secured to the chest and bilaterally on each mid-foot at a sampling frequency of 100 Hz.
Clinical Measurements
PwMS also underwent a series of clinical assessments, completed within 6 weeks of the precision walking task. This included the latency—in both eyes—of visual evoked potentials (VEPs), where we used the value for the worst eye, and the Multiple Sclerosis Functional Composite (MSFC) assessment, which consists of the paced auditory serial addition test, the 9-hole peg test, and the timed 25-foot walk. 36 To assess functional mobility, PwMS performed the Six Spot Step Test (SSST), a timed walking test that involves kicking over a number of targets placed along a 5-m path, 37 and the Timed Up and Go (TUG) test. 38 To assess visuomotor coordination, we tested reaction time using the Dynavision D2 apparatus (Mode A; Dynavision International LLC, West Chester, OH), where participants press, as quickly as possible, targets that light up in different positions in front of them while standing. An experienced neurologist determined the EDSS score. Details are shown in Table 1.
PwMS Characteristics a .
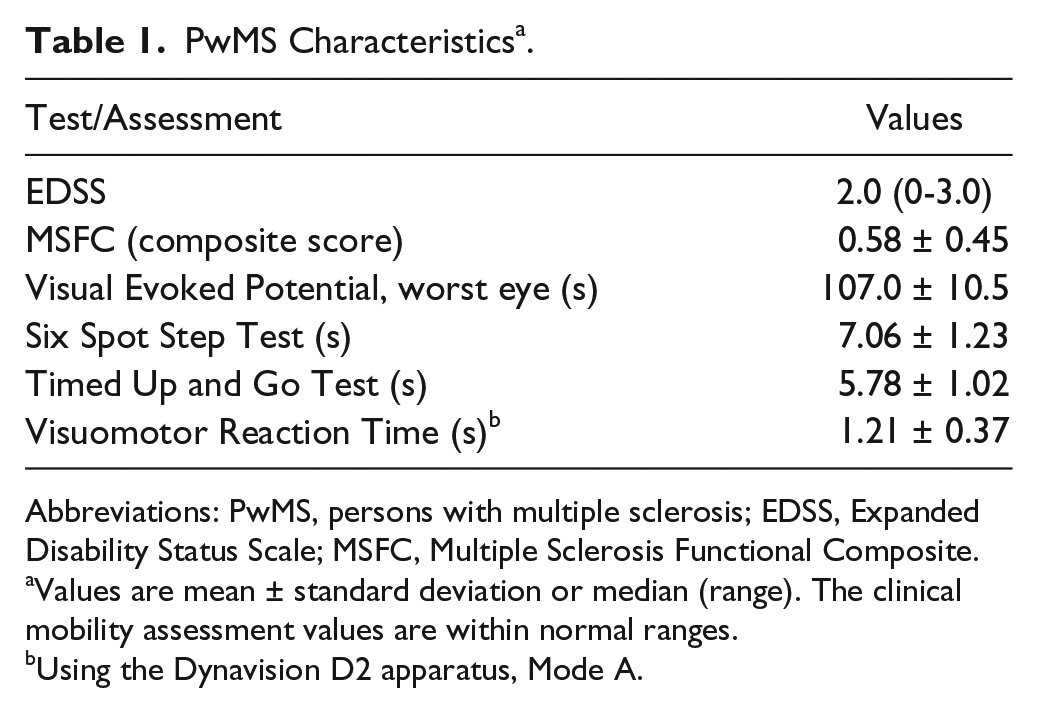
Abbreviations: PwMS, persons with multiple sclerosis; EDSS, Expanded Disability Status Scale; MSFC, Multiple Sclerosis Functional Composite.
Values are mean ± standard deviation or median (range). The clinical mobility assessment values are within normal ranges.
Using the Dynavision D2 apparatus, Mode A.
Data and Statistical Analysis
We analyzed data using custom-written MATLAB programs. First, we filtered kinematic data with a fourth-order low-pass Butterworth filter (cutoff frequency of 6 Hz). Next, we calculated foot placement on each target as the time at which point the foot-marker’s anterior-posterior velocity and acceleration profiles stabilized to zero. The medial-lateral distance between the foot position marker and the center of the target at this time point defined the medial-lateral foot placement error, and served to quantify adaptation across trials, and retention between testing sessions (Figure 1A). Positive values represent foot placement errors in the direction of the prism shift (ie, toward the right). We analyzed each target separately because our previous work found that the legs adapt and generalize differently,27,28 and the second target step is likely biased by the first. Here, we only report results of the step to target 1, although the results are similar for the step to target 2.
To determine differences in baseline performance on the precision walking task between PwMS and controls, we used separate 2-sample t tests after calculating the mean error and error variability (ie, standard deviation) across all baseline trials.
To determine whether PwMS adapt differently than healthy controls, we compared foot placement error at specific time points (or phases) during the first testing session between these groups using a 2-way (Group × Phase) repeated-measures ANOVA. We used the following phases: baseline (mean of last 10 baseline trials), first adaptation trial, early adaptation (mean of adaptation trials 2 to 8), late adaptation (mean of last 10 adaptation trials), and first postadaptation trial. We used Tukey post hoc tests for significant main effects and interactions.
We also quantified response time, a measure of adaptation rate. To calculate response time, we first smoothed individual subject’s adaptation phase data using a 5-trial moving average (with MATLAB’s movmean function). Next, we calculated the number of trials for each subject’s error to reach a plateau; we defined this plateau range as the mean ± SD of the last 10 adaptation phase trials. This measure captures how long it takes subjects to reduce movement errors in the adaptation phase and is similar to the methods of others. 39 We used separate 2-sample t tests to determine differences between groups on this measure on day 1.
To assess retention across 1 week, we compared first adaptation trial error, early adaptation error, and response time between days and groups using separate 2-way (Day × Group) ANOVAs, and Tukey post hoc tests when applicable. Our early adaptation error measure is similar to other research and described above;29,40,41 it focuses on the period of rapid early adaptation.
We performed separate Pearson correlations between the clinical measures and retention performance (change in early adaptation error and change in response time across days) on the precision walking task. We log transformed VEP and visuomotor reaction time since they did not fit a normal distribution. Finally, we performed a multiple linear regression analysis to determine which combination of clinical measures predicted retention performance using the same 2 measures described above as dependent variables. We first entered log(VEP), MSFC, TUG, SSST, log(visuomotor reaction time), and age as independent variables in each model. We removed variables one-by-one using backward elimination in order of descending P values until all remaining variables had P values <.1, and we obtained a significant overall model. We performed all statistical analyses using JMP 12 software with an α level of .05.
Results
Adapting to a Novel Visuomotor Mapping During Precision Walking
We found no differences in baseline mean foot placement error (PwMS = −7.2 ± 24.2 mm vs controls = 0.5 ± 13.5 mm; t29 = −1.2, P = .243) or error variability (PwMS = 20.2 ± 6.6 mm vs controls = 18.5 ± 5.1 mm; t33 = 0.85, P = .401) between PwMS and controls. Both groups demonstrated large initial foot placement error when exposed to the prism lenses in the adaptation phase but this error decreased over repeated trials. In the postadaptation phase, we found a large error in the opposite direction, suggesting that both groups stored the novel visuomotor mapping. These results are illustrated in Figure 2A. We found no significant Group main effect (F1,34 = 0.03, P = .871) or Group × Phase interaction (F4,136 = 0.9, P = .484). However, as shown in Figure 2B, we found a significant main effect of Phase (F4,136 = 359.6, P < .0001). Post hoc tests indicated greater error in the first adaptation trial compared to the rest (P < .0001). Error at baseline matched that in late adaptation though (P = .913). Furthermore, error differed significantly between early adaptation and the other phases (P < .0001). We found a similar effect for the postadaptation phase. Although both groups adapted to the prisms, it is possible that the rate at which they accomplished this differed. However, we found no differences between PwMS and controls for response time (t34 = 0.47, P = .638). This measure, which reflects the time it takes foot placement error to reach a plateau during the adaptation phase, is shown in Figure 2C.
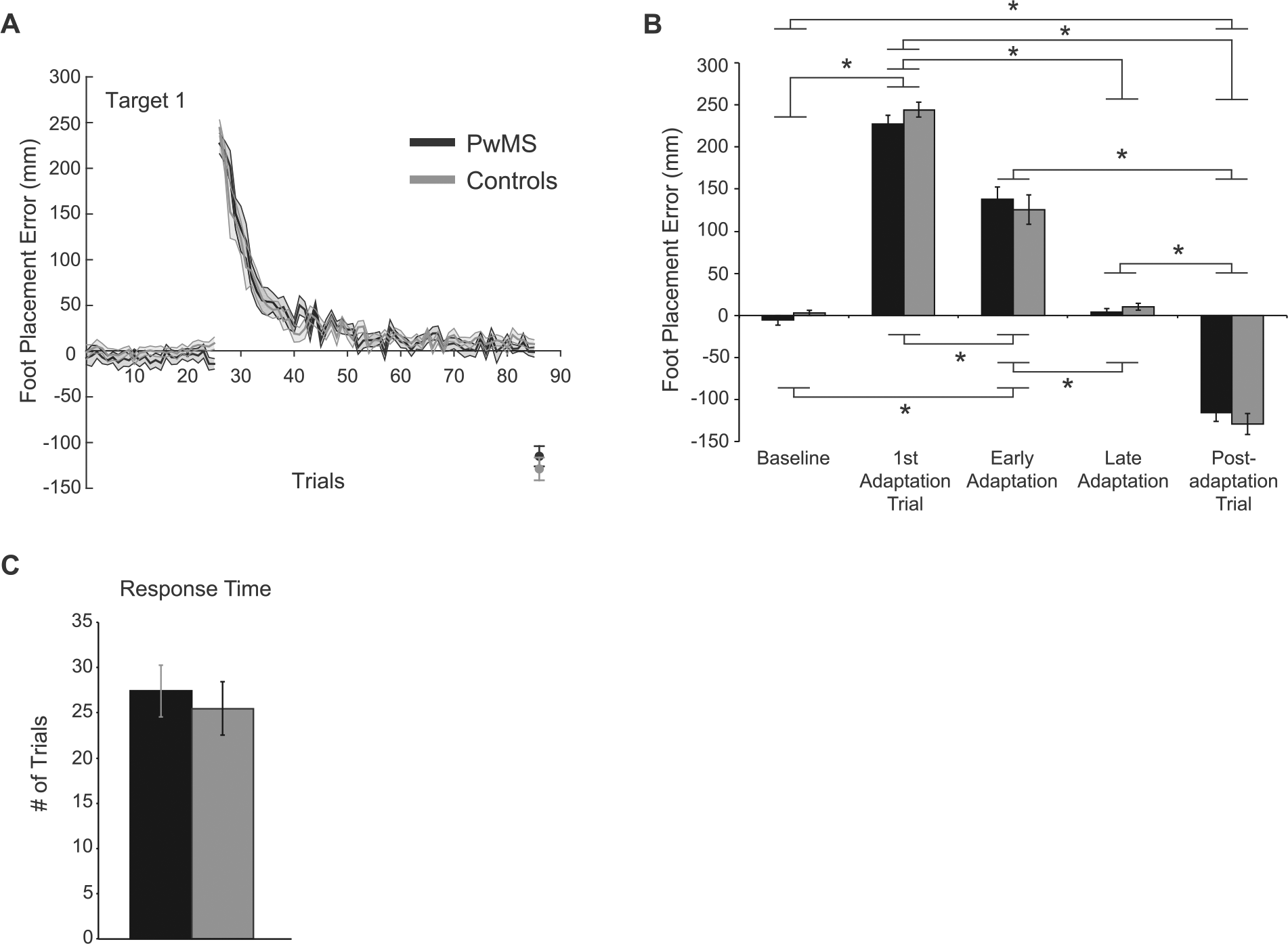
Adapting to a novel visuomotor mapping. (A) Group mean ± SE foot placement error related to target 1 during learning (day 1) for PwMS (black line) and controls (gray line) is shown. (B) Group mean ± SE foot placement error in different phases (baseline, first adaptation trial, early adaptation, late adaptation, postadaptation trial) for PwMS (black bars) and controls (gray bars). Asterisks indicate significant post hoc tests for the main effect of Phase (P < .05). (C) Group mean ± SE response time for PwMS (black bars) and controls (gray bars) are shown.
Retention of a Novel Visuomotor Mapping Over 1 Week
Figure 3A illustrates group mean foot placement error on days 1 and 2 for each group. In each case, error with relearning on day 2 is reduced. Specifically, we found a significant main effect of Day (F1,34 = 89.2, P < .0001) for first adaptation trial error (Figure 3B, left side), supporting the notion that both groups recalled the novel mapping at least to some extent. Additionally, Figure 3B (middle) shows reduced early adaptation error on day 2 (Day main effect: F1,34 = 44.3, P < .0001). The PwMS and controls demonstrated similar first adaptation trial error (nonsignificant Group main effect: F1,67 = 1.1, P = .296; nonsignificant Group × Day interaction: F1,34 = 0.1, P = .777) and early adaptation error (nonsignificant main effect of Group: F1,68 = 0.4, P = .518; nonsignificant Group × Day interaction: F1,34 = 0.2, P = .668). Response time decreased across days (main effect of day: F1,34 = 23.2, P < .0001) in both groups (Figure 3B, right side). However, we found no differences between the 2 groups for this measure (nonsignificant Group main effect: F1,63 = 0.28, P = .598; nonsignificant Group × Day interaction: F1,34 = 0.26, P = .615).
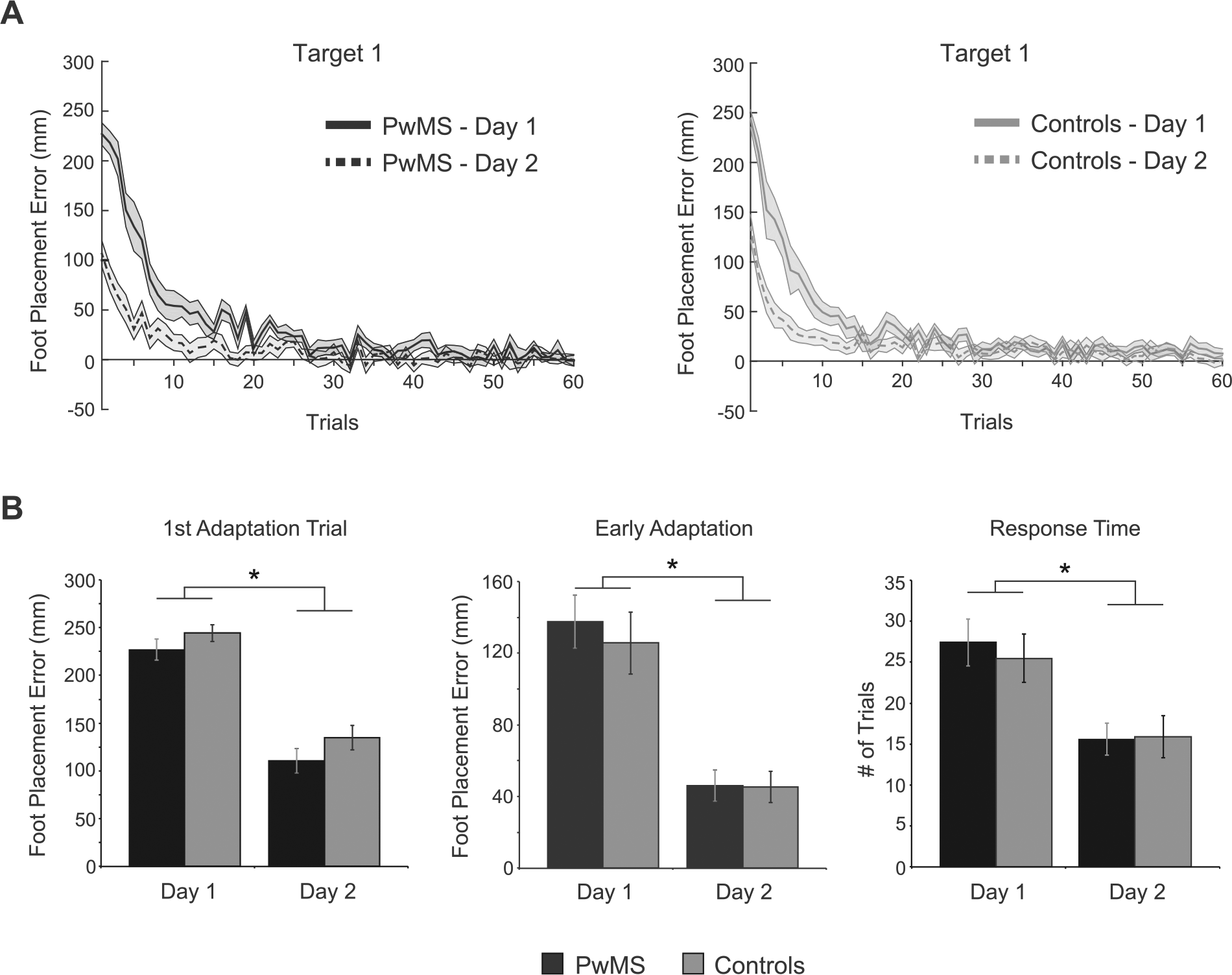
Retention of a novel mapping over 1 week. (A) Group mean ± SE foot placement error related to target 1 during learning (day 1) and relearning (day 2) for PwMS (left panel) and controls (right panel) is shown. (B) Group mean ± SE foot placement error for the first adaptation trial (left panel), early adaptation trials (middle panel), and response time (right panel) for PwMS (black bars) and controls (gray bars) across days are shown. Asterisks indicate significant post hoc tests for the main effect of Day (P < .05).
Relationship Between Clinical Measures and Motor Learning Performance
We performed Pearson correlations between our clinical measures (eg, SSST) and the change in performance on the precision walking task (based on our early adaptation error and response time measures). However, we found no significant relationships. We also found no significant multiple regression models.
Discussion
The ability to adapt in response to impairment and retain motor performance gains—key components of motor learning—is essential for rehabilitation and day-to-day living following neurological injury. Here, we exposed PwMS with mild disability and healthy controls to a novel visuomotor mapping induced by prism lenses during a precision walking task on 2 separate occasions. We found that PwMS and controls are affected by this initial novel visuomotor mapping to the same extent, they demonstrate a similar rate and magnitude of adaptation, and retain their ability to accurately control foot placement 1 week following a single learning session despite the effects of the prisms.
Short-Term Motor Learning and Retention Are Preserved in PwMS
PwMS reduced foot placement error similarly to controls as they adapted to a novel visuomotor mapping while walking. In fact, both groups reduced error to baseline levels. Furthermore, both groups demonstrated large foot placement error in the direction opposite to that of the prism shift (and compared to the error in the initial prism exposure) when wearing 0-D lenses in the postadaptation phase. This negative aftereffect occurs because the brain applies the new, adapted visuomotor mapping in the task even though it is no longer applicable in the postadaptation phase. This suggests that PwMS, and controls, not only learned the new mapping but stored it as well. Previous research also shows that PwMS adapt to force fields during reaching, 19 oscillating platforms while standing,20,21 and upper extremity-based isometric visuomotor tracking tasks 23 to the same extent as controls. In contrast to other studies,19-23,25 we found similar motor performance between groups at baseline, as reflected by mean foot placement error (and standard deviation), and during the adaptation phase, as reflected by the magnitude of error throughout. Disease severity is unlikely to explain the discrepancy with these other studies given that we tested PwMS with a similar range of EDSS scores. It is possible that stepping to ground locations is a highly practiced task and thus relatively simple. However, overcoming the novel visuomotor mapping induced by the prisms is challenging as evident in the error values and past research in this area. Nonetheless, the results of our study and those of others support the notion that short-term learning is maintained in PwMS with mild disability in a variety of motor tasks.
PwMS and controls demonstrated reduced foot placement error and adapted faster when exposed to the novel visuomotor mapping a second time, 1 week later. Unfortunately, few studies in MS have investigated retention.20,21,23 In one instance, PwMS retained, after 24 hours, learned improvements in the temporal aspect of maintaining standing balance on an oscillating surface platform, defined as a shift in the timing relationship between the minimum and maximum body center of mass displacement and platform displacement.20,21 These researchers argued that PwMS learned to predictively compensate for the repeating platform motion. 21 We recently demonstrated that adapting foot placement in our paradigm relies on the integration of predicted and actual visual feedback to estimate the state of the limb and issue an appropriate motor command to direct the foot to the target. 18 Taken together, these results suggest predictive motor control remains intact in PwMS.
It is somewhat remarkable that both groups retained the visuomotor mapping for at least 1 week following a single session. This is similar to our recent findings in young adults in this paradigm. 29 Although Tomassini et al 23 found retention after 2 weeks in their motor learning paradigm, PwMS and controls practiced each of those days, rather than a single session like in our work. Other studies tested retention only after a 24-hour delay. Future work should determine exactly how long PwMS can retain a learned motor behavior, an important consideration for the effectiveness of rehabilitation strategies.
Visually guided walking relies on cortical and cerebellar circuits. 42 These same brain regions are implicated in the process of adapting to altered visuomotor mappings caused by prism lenses.43,44 The fact that PwMS—at least high functioning individuals—are capable of adapting, and perhaps more important, retaining the stored adaptation, suggests that cortical and/or cerebellar plasticity is preserved. A range of studies using neuroimaging and neurostimulation techniques as well as different motor tasks support this notion.20,45,46
While we found intact motor learning in our sample with mild disability, one might expect low-functioning PwMS to show differences compared to controls. However, these individuals would likely struggle to properly perform our precision walking task, thus possibly confounding the results. Most motor learning studies in PwMS use a moderate to high-functioning cohort. The results are mixed when low-functioning PwMS are included and perform visuomotor tasks, with some studies failing to reveal learning in these individuals, 22 and others showing learning at comparable levels to controls. 23
In an effort to determine whether clinical measures can predict which individuals demonstrate better motor learning, as evidenced by retention of the novel visuomotor mapping, we performed a series of correlations and multiple regressions. However, we did not find significant relationships with any of our measures. There are at least 2 possible reasons for these results. First, the clinical measures may lack the necessary sensitivity, particular at the milder disability levels. Second, we tested a relatively small sample. Further research, with larger sample sizes, should explore the relationship of these and other clinical tests with motor learning performance in a variety of tasks.
Implications for Rehabilitation
The common occurrence of gait impairments in PwMS, even those with minimal disability,4,6,7,9 and the fact that variability of foot placement is associated with falling, 8 highlights the importance of gait training interventions for this population. Although specific stepping locations are not typically marked on the ground as in the present experiment, people often must direct their feet to desired locations to avoid hazardous terrain and obstacles. Thus, the ability to ensure a proper visuomotor mapping is applicable for general community ambulation. As such, incorporating walking tasks that stress the importance of visually guided foot placement into this gait training may be beneficial. Indeed, there is evidence that visual feedback improves walking ability in PwMS, as reflected by speed and stride length. 47 We suggest that altering vision, potentially through the use of prism lenses, might facilitate this gait training. The error in performance caused by this manipulation forces the nervous system to adapt, initiating the process of neural plasticity. Consequently, this may prime the appropriate neural circuitry for greater short-term motor learning and retention. Thus, training could involve practicing specific visually guided walking tasks with altered vision prior to other walking tasks with normal vision.
An important consideration is whether the type of motor learning observed in the present study in PwMS transfers to community ambulation. Although this is currently unknown, we previously found that university-aged adults transfer what they have learned in an identical precision walking task to stepping over obstacles,27,28 and others report that prism adaptation of walking trajectory transfers to reaching to a target. 48 A second consideration is that we altered the visuomotor mapping using a fixed visual disturbance created by prisms to study motor learning. The visual deficits seen in PwMS, though, typically relate to blurry or double vision. 12 Nonetheless, PwMS must at times adapt to altered vision, no matter the specific nature or variability of the change. Consequently, any practice that forces the individual’s nervous system to adapt may help improve its ability at later times.
Conclusions
To our knowledge, this is the first study to demonstrate that PwMS with mild disability can learn a novel visuomotor mapping in a walking paradigm, and retain this mapping for an extended period of time. Our results provide additional support for the notion that neural plasticity is possible in PwMS. Given the importance of walking ability and visual function to PwMS, future research should assess more impaired individuals and test motor learning ability with different visual manipulations, for instance, that better simulate blurry and/or double vision.
Footnotes
Acknowledgements
The authors thank Dr Kim Lajoie for help with data collection, Calvin Mok for help with performing clinical assessments, and Rodrigo Maeda for help with MATLAB programming.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Natural Sciences and Engineering Research Council of Canada (RGPIN-2014-04361) provides general funding for Dr Marigold’s research program, which covered minor expendables during the course of this study.
