Abstract
Background
People with multiple sclerosis (PwMS) frequently experience dizziness and imbalance that may be caused by central vestibular system dysfunction. Vestibular rehabilitation may offer an approach for improving dysfunction in these people.
Objective
To test the efficacy of a gaze and postural stability (GPS) retraining intervention compared to a strength and endurance (SAE) intervention in PwMS.
Methods
About 41 PwMS, with complaints of dizziness or history of falls, were randomized to either the GPS or SAE groups. Following randomization participants completed 6-weeks of 3×/week progressive training, delivered one-on-one by a provider. Following intervention, testing was performed at the primary (6-weeks) and secondary time point (10-weeks). A restricted maximum likelihood estimation mixed effects model was used to examine changes in the primary outcome of the Dizziness Handicap Inventory (DHI) between the 2 groups at the primary and secondary time point. Similar models were used to explore secondary outcomes between groups at both timepoints.
Results
Thirty-five people completed the study (17 GPS; 18 SAE). The change in the DHI at the primary time point was not statistically different between the GPS and SAE groups (mean difference = 2.33 [95% CI −9.18, 12.85]). However, both groups demonstrated significant improvement from baseline to 6-weeks (GPS −8.73; SAE −7.31). Similar results were observed for secondary outcomes and at the secondary timepoint.
Conclusions
In this sample of PwMS with complaints of dizziness or imbalance, 6-weeks of GPS training did not result in significantly greater improvements in dizziness handicap or balance compared to 6-weeks of SAE training.
Introduction
Multiple Sclerosis (MS) is a progressive neurological disease that results in a variety of symptoms including dizziness, unsteadiness, poor balance, fatigue, and pain.1 -6 Substantial research has been done to explore management strategies for many of these symptoms, yet the literature is limited regarding management of dizziness and balance related issues. More specifically, the examination of vestibular rehabilitation protocols for the management of these symptoms have only recently began to be explored.
Evidence suggests that cerebellar and brainstem pathology is common in people with MS (PwMS) and is closely linked with higher rates of disability.7,8 Presence of MS lesions in these areas are closely associated with vestibular related dysfunction as indicated by complaints of dizziness/vertigo, altered subjective visual vertical,9 -11 altered vestibular evoked myogenic potentials,12,13 altered performance on the vestibular dominant components of the sensory organization test,14,15 and other balance tests.16,17 In fact, these symptoms have been reported to occur in between 30% and 59% of PwMS.18,19 At minimum, these deficits contribute to motion sensitivity, dizziness, and oscillopsia, and at worst, they create problems with completing activities of daily living, can lead to social isolation, and lead to falls. Multiple studies have shown that falls and lack of balance are highly disabling in MS, and affect approximately 75% of individuals over the course of the disease.20,21 Furthermore, chronic dizziness and falls have a significant negative impact on quality of life.21,22
While there is clear evidence of the benefits of gaze and postural stability (GPS) training in individuals with peripheral vestibular system injury, 23 until relatively recently, no studies have examined the benefits of vestibular focused exercises to improve outcomes in PwMS. Furthermore, because of the scarcity of studies, the optimum dosing, exercise type, and duration of vestibular rehabilitation in MS remains unknown. Hebert et al 24 demonstrated significant improvements in outcomes including patient reported dizziness and scores of the sensory organization test following participation in a vestibular-based rehabilitation protocol. While this study provided strong evidence that vestibular training provided benefit relative to no intervention, a comparison of the efficacy and differential effect of 2 evidence based interventions (GPS training; strength and aerobic exercise training [SAE]) has not been performed. In order to focus on this gap in our research knowledge, the purpose of the current study was to examine the efficacy of these 2 interventions on measures of GPS reflecting the participation and activity level domains of the World Health Organization International Classification of Functioning, Disability, and Health (ICF). We hypothesized that (1) the intervention (GPS) group would experience greater improvement in DHI scores than the control (SAE) group from baseline to 6-weeks, (2) that the GPS group would experience greater improvement of secondary outcomes from baseline to 6-weeks, and (3) that the GPS group would maintain higher scores on all outcomes at 10-weeks (secondary timepoint) compared to the SAE group.
Methods
All study procedures were approved by the University of Utah Institutional Review Board (#00104298) and this study was reported in ClinicalTrials.gov (#NCT03521557).
Participant recruitment—Potential participants were recruited through advertising at the National Multiple Sclerosis Society, community multiple sclerosis clinics, and by screening patients seen at the University of Utah Neuroimmunology clinic between April 2018 and July 2020. Interested participants were screened for inclusion/exclusion criteria via telephone interview. Inclusion criteria required participants be between the ages of 20 and 75 years old, have a neurologist-diagnosed diagnosis of MS, 25 expanded disability status scale (EDSS) score of <6.0, current complaints of dizziness (>0 on the DHI), and/or at risk for fall (determined by >2 falls in the last year or Activity Specific Balance Confidence Scale <80). Participants were excluded from participation if they (1) had another or additional central or peripheral nervous system disorder other than MS, (2) otologic, cervical spine, or lower extremity injury in last 12 months that limited participation in exercise, (3) had peripheral vestibular pathology, (4) had internuclear ophthalmoplegia, (5) experienced MS exacerbation within 8 week of study enrollment, and (6) were already undergoing treatment for vestibular dysfunction (eg, pharmacological or physical therapy) and were unwilling to quit treatment during the study period.
Those participants meeting inclusion and exclusion criteria and willing to enter the study completed baseline assessment. Following baseline assessment participants were randomized using an a priori blinded randomization scheme. Randomization was performed by the unblinded study director. The randomization scheme was generated through Randomization.com (http://www.randomization.com) using permuted block randomization with block sizes ranging from 2 to 8. Of the 82 individuals screened, 41 were randomized.
Study Overview
This study used a single blinded, 2 group, randomized clinical trial design.
Qualifying participants were consented and completed a baseline assessment of GPS. Following the baseline assessment all participants were randomized to either the GPS or SAE group. All individuals then began 6-weeks of training at a frequency of 3×/week (18 visits total), during which they worked one-on-one with a trained research assistant.
Following completion of the training period, all participants returned for assessment by 2 blinded researchers at 2 distinct timepoints. The primary assessment timepoint occurred at a date within 7 days of completion of the final training day (~6 weeks from baseline), the secondary timepoint occurred 4 weeks later (~10 weeks following initiation of the study). During these assessments, participants completed the same battery of testing performed during the baseline assessment.
Intervention Programs (Independent Variables)
The GPS group completed a program tailored for the treatment of GPS deficits along with strengthening and endurance training. This program consisted of vestibular training exercises for GPS. 23 During each treatment session participants completed 15 minutes of gaze stability training and 15 minutes of postural stability training, with an additional ~45 minutes of lower extremity strengthening and cardiovascular endurance training. The SAE group completed ~45 minutes of lower extremity strengthening and cardiovascular endurance training, only. The endurance and lower extremity strengthening exercises used in both groups were chosen based on evidence supporting these interventions in PwMS.26 -29 Detailed information regarding both training protocols and generalized progressions are detailed in Loyd et al 30 and included in Supplemental Material 1 to 3.
Additionally, each participant in the GPS group was prescribed an individualized home exercise program (HEP) consisting of GPS exercises and walking. HEP prescription and dosing was specific to the individual’s function and to be performed 5 days per week. In general, participants were prescribed ~15 minutes each of both GPS training and ~15 to 30 minutes of walking. HEP adherence was recorded by the participant using a daily log. Without current parameters for dosing GPS training in PwMS, HEP dosing was based on recommendations from clinical practice guidelines for peripheral vestibular hypofunction. 23
Descriptive Variables
Descriptive variables collected for the study sample were age, sex, BMI, EDSS, duration of symptoms, fall history, race, smoking status, MS medical management, 25-foot walk time, and Modified Fatigue Impact Scale-5 Item (MFIS-5) scores.
Outcomes
The outcomes collected in this randomized clinical trial (RCT) were chosen to capture function across the domains of the WHO ICF with the primary outcome representing the participation domain. Additionally, the secondary outcome Activities-specific Balance Confidence (ABC) also represents the participation level domain, while secondary outcomes from the activity domain included the computerized dynamic visual acuity (cDVA) as a measure of gaze stability and the Functional Gait Assessment (FGA) as a measure of postural stability.
Primary Outcome (Participation Level)
Dizziness Handicap Inventory (DHI)—The DHI is a 25-item questionnaire completed by the participant that is scored on scale from 0 to 100, with 0 indicating no issues with balance or unsteadiness while 100 represents severe unsteadiness. Each item is given a response of “never,” “sometimes,” or “always,” which correspond with 0, 2, and 4 points, respectively. Each item states a situation across 1 of 3 sub-domains (Physical, Functional, Emotional) during which a person may experience problems with dizziness or unsteadiness in their daily life. The DHI has been found to be reliable in PwMS. 31
Secondary Outcomes (Participation and Activity Level)
Activities-Specific Balance Confidence scale (ABC)—The ABC is a 16-item self-report measure developed to identify a person’s confidence in their balance during performing different activities. Each item is given a percent confidence from 0% to 100% identifying how confident the person is that they would not lose their balance or become unsteady. All the items are averaged and the total score ranges from 0% meaning no confidence to 100% meaning total confidence. The ABC has been validated for use in PwMS. 31
Computerized Dynamic Visual Acuity (cDVA)—cDVA was performed using a head mounted inertial measurement unit which was paired with a 12″ tablet that displayed both static and dynamic acuity optotypes. Individuals were asked to properly identify the optotypes of increasing and decreasing size until the smallest identifiable optotype was identified in both static and right and left dynamic directions. Dynamic optotypes were displayed with head turns in either the right or left direction at greater than 160° per second. 32 cDVA socres are reported as logMAR difference from static to dynamic (logMAR diff). Variations of dynamic visual acuity measures have been tested in PwMS and found to have moderate test–retest reliability. 33
Functional Gait Assessment (FGA)—The FGA is a widely used and well accepted measure that assesses dynamic postural stability which assesses a variety of ambulatory tasks including normal walking, walking with head turns, stepping over an obstacle, stopping quickly and turning, walking with a narrow base of support, walking with eyes closed, walking backward, and ascending/descending stairs. The FGA has been reported to have excellent concurrent validity when compared to the Berg Balance Scale, timed up and go, and the ABC in a variety of balance impaired populations including those with MS, Parkinson’s disease, older adults, and vestibular disorders.34 -36 Each of these tasks is scored on a scale from 0 (unable to complete the task) to 3 (“normal” performance on the task), with a total possible score of 30 indicating perfect performance. When used in individuals with vestibular disorders the FGA has been shown to have excellent intrarater and interrater reliability. 35
Statistical Analysis
In order to calculate an a priori effect size, we utilized (1) data from our pilot research on GPS in persons with MS 37 and (2) spatial and temporal measures from previous vestibular research examining task specific training.38 -41 We estimated an effect size of d = 0.25 for the DHI with the correlation between baseline and follow-up measurements as .50. Twenty participants in each arm was predicted to provide us with greater than 80% power and 2-sided α = .05 to detect a difference between groups on the DHI. We targeted an enrollment of 50 individuals with MS in order to account for a smaller effect size than estimated and potential drop-outs and still ensure adequate power for the primary outcome.
Statistical analysis was performed using the Statistical Program for Social Sciences (IBM SPSS Statistics v26, IBM Corp., Armonk, New York, USA). Study data were collected and managed using REDCap (Research Electronic Data Capture) electronic data capture tools hosted at the University of Utah.42,43
Characteristics of the sample and each treatment group were summarized measures of central tendency (median, means, standard deviation) for continuous variables and counts with percentages for categorical variables. All data was analyzed to assure assumptions for parametric analysis were met.
We used restricted maximum likelihood estimation mixed effects models to examine changes in the DHI over time. Fixed variables included group assignment, assessment timepoint, the interaction between time and group assignment, and the baseline DHI score. The between group comparison of the DHI at 6-weeks was designated as the primary comparison. Treatment comparisons for the DHI at 10-weeks was interpreted as a secondary comparison and evaluation without adjustment for multiple comparisons. Random effects at the patient-level were modeled with variance components.
For secondary outcomes, similar mixed effects analyses were applied to evaluate the effects of the GPS intervention compared to SAE on the ABC, FGA, and cDVA adjusting for initial levels of the dependent variables. We analyzed all participants within the group to which they were assigned. For any outcomes with more than 10% missing data at follow-up, we ran the mixed models using multiple imputation datasets that included auxiliary variables to reduce the risk of bias due to missing measures. We used sensitivity analyses to address the robustness of our results. These were performed examining results including only individuals with complete data at 6-weeks.
Role of the Funding Source
This study was funded by the National Multiple Sclerosis Society, which had no role in the design or conduct of the study including: collection, management, analysis, or interpretation of the data; preparation, review, or approval of the manuscript; or decision to submit the manuscript for publication.
Results
From September, 2018 through May, 2020 a total of 82 persons were screened and 41 were enrolled (Figure 1). Mean age of participants was 53.9 (11.2) years and 32 (78%) were female. The average EDSS score was 3.6 (SD 1.0) and participants reported a median duration of symptoms of 20 (IQR 18.5) years (Table 1). Participants were randomly assigned to GPS (n = 20) or SAE (n = 21). Baseline DHI means were 41.8 (SD 16.1) and 41.9 (SD 24.2) points for the GPS and SAE groups, respectively.
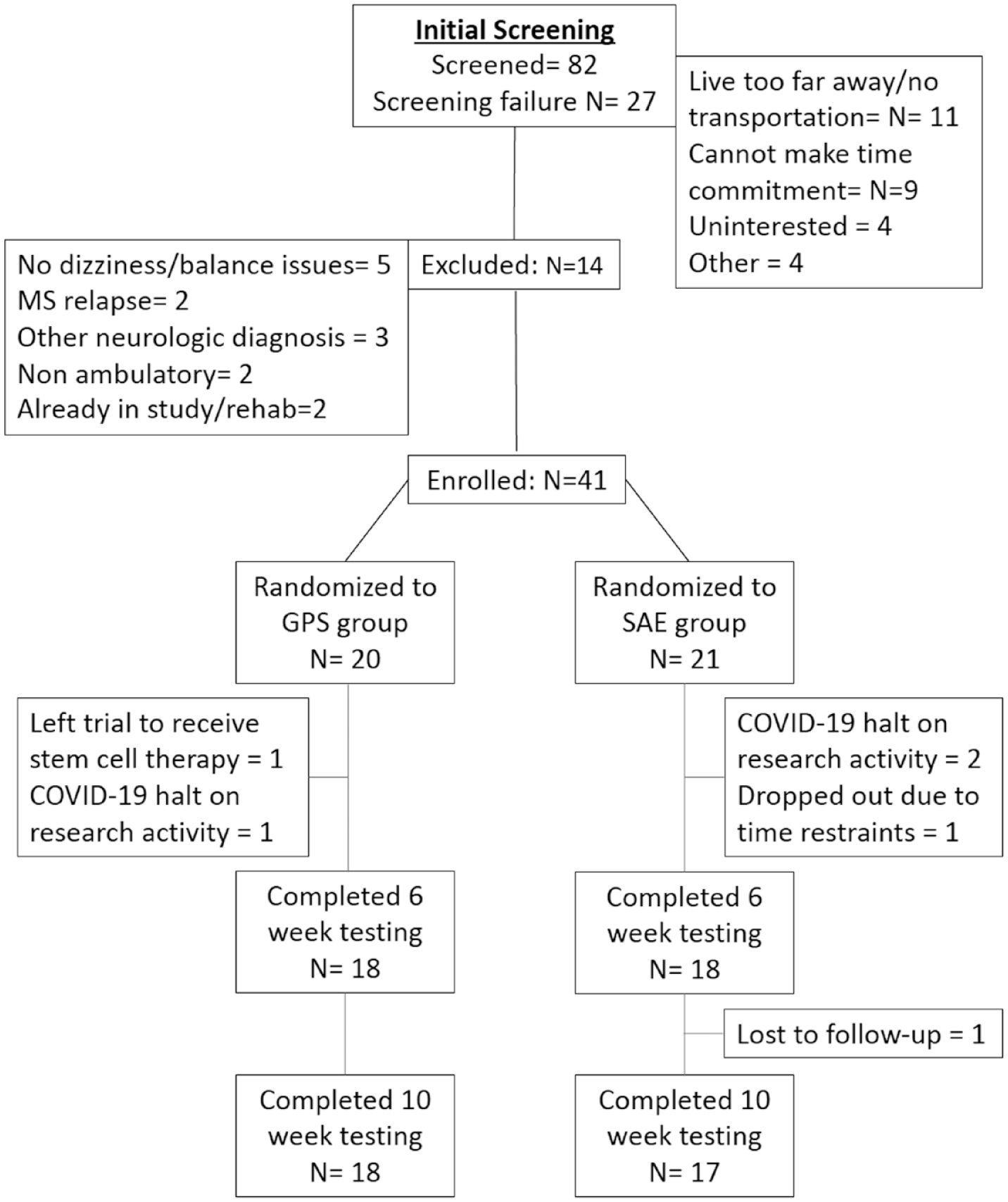
Consort diagram demonstrating the screening, enrollment, randomization, and study completion for both of the study groups.
Characteristics of the Entire Sample and Each Treatment Arm.
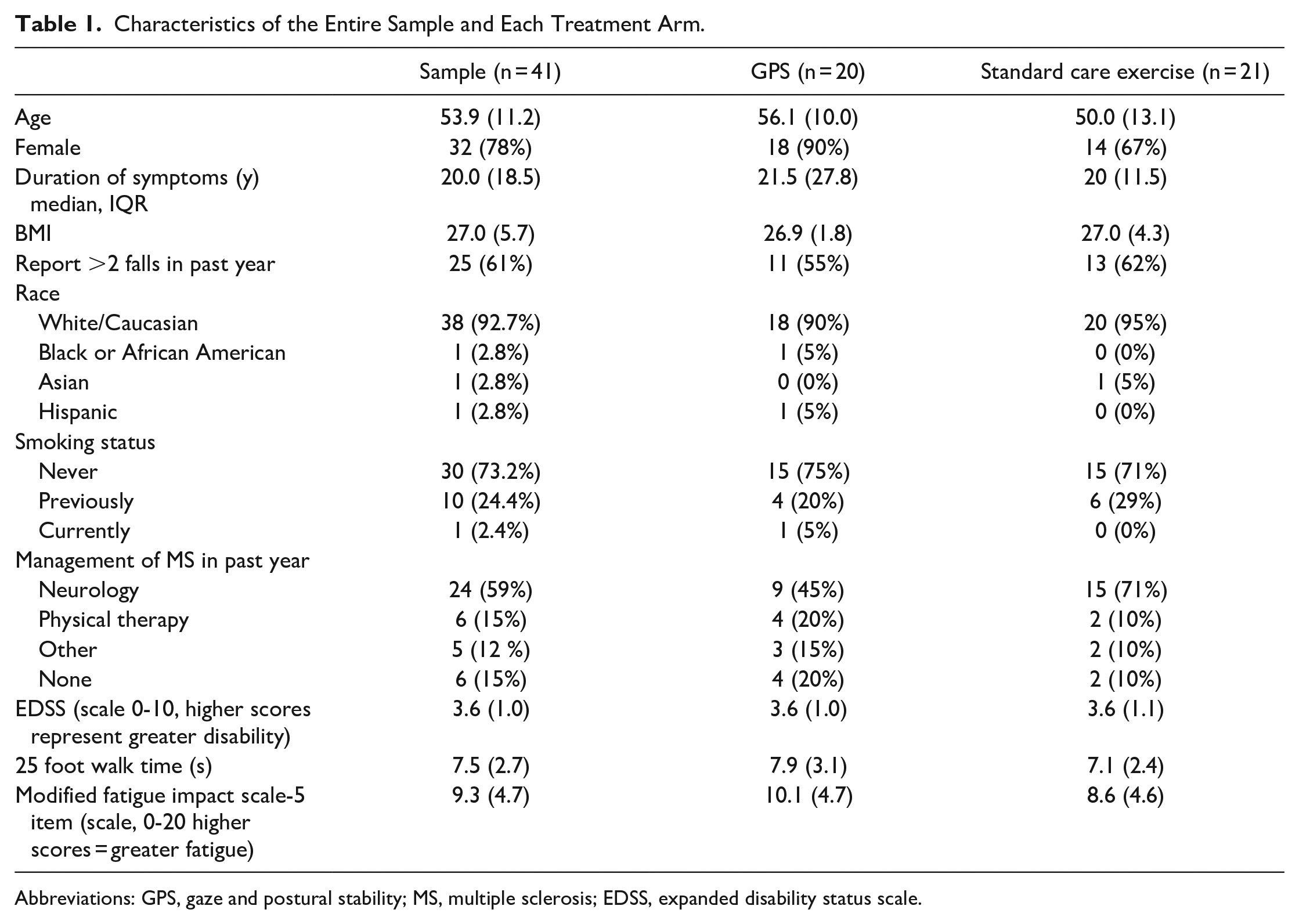
Abbreviations: GPS, gaze and postural stability; MS, multiple sclerosis; EDSS, expanded disability status scale.
Overall, 36 participants (88%) completed the 6-week assessment and 35 (85%) completed assessments at 10-weeks. No patients switched treatment groups. Of the 18 planned treatment sessions the average number of treatments attended in the GPS group was 17.7 while the SAE group was 17.1. Three individuals were screened for inclusion in the study and randomized but due to COVID-19 research restrictions imposed during March 2020, these participants were not able to participate in the study (Figure 1).
The average improvement over time across both groups was statistically significant for DHI, ABC, and FGA at both the primary and secondary timepoints (Table 2). However, the change in the primary outcome variable (DHI) over 6-weeks was not statistically significant between the GPS and SAE groups (Figure 2). Specifically, the mean DHI difference between groups at 6-weeks was 2.33 (95% CI −9.18, 12.85) and at 10-weeks −1.09 (95% CI −11.71, 9.53). The results were similar across ABC, cDVA, and FGA without any statistically significant change favoring one treatment over another (Table 2; Figures 3–5). Per protocol analyses were similar to the intention-to-treat results.
Primary and Secondary Outcomes.
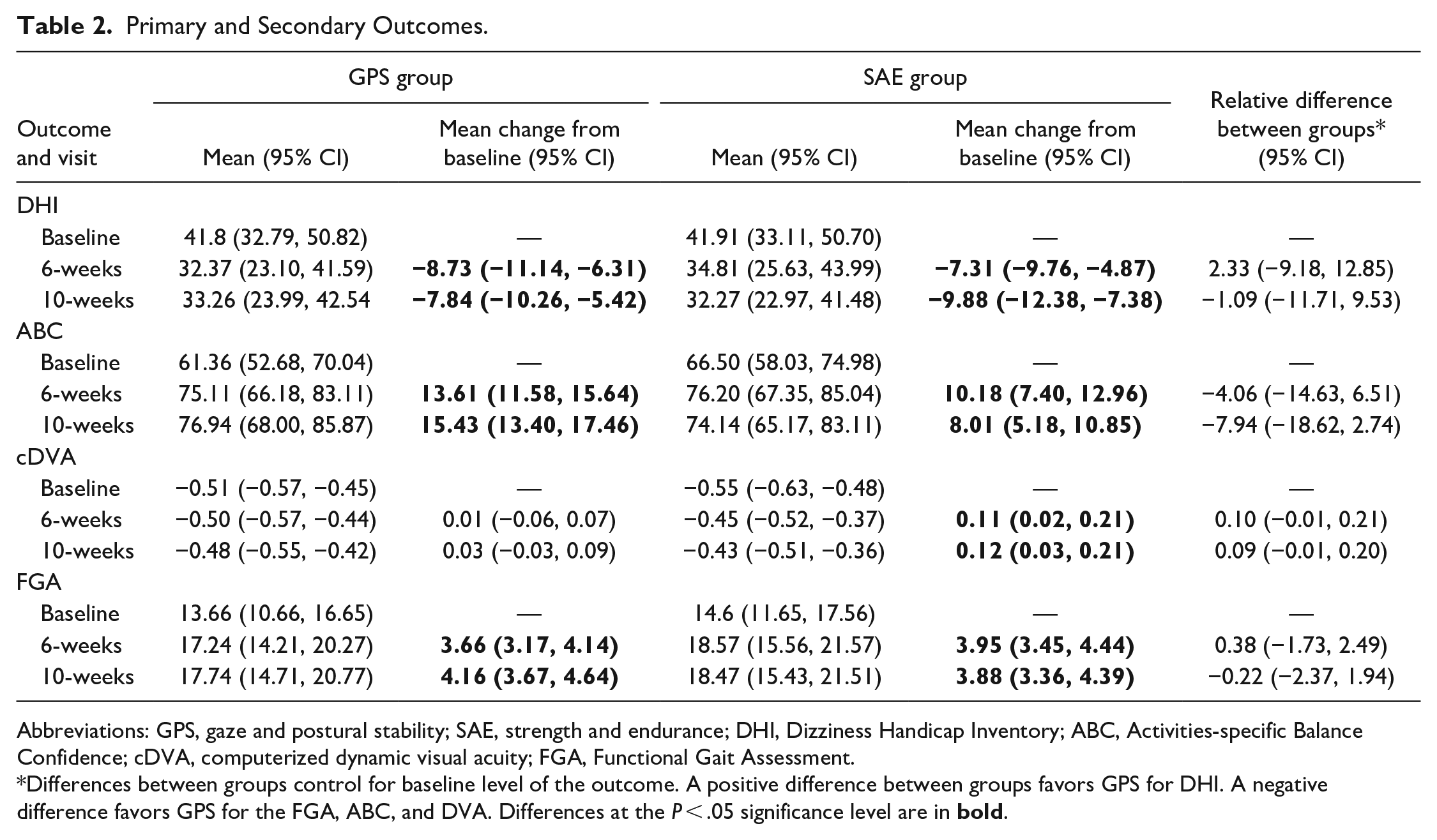
Abbreviations: GPS, gaze and postural stability; SAE, strength and endurance; DHI, Dizziness Handicap Inventory; ABC, Activities-specific Balance Confidence; cDVA, computerized dynamic visual acuity; FGA, Functional Gait Assessment.
Differences between groups control for baseline level of the outcome. A positive difference between groups favors GPS for DHI. A negative difference favors GPS for the FGA, ABC, and DVA. Differences at the P < .05 significance level are in bold.
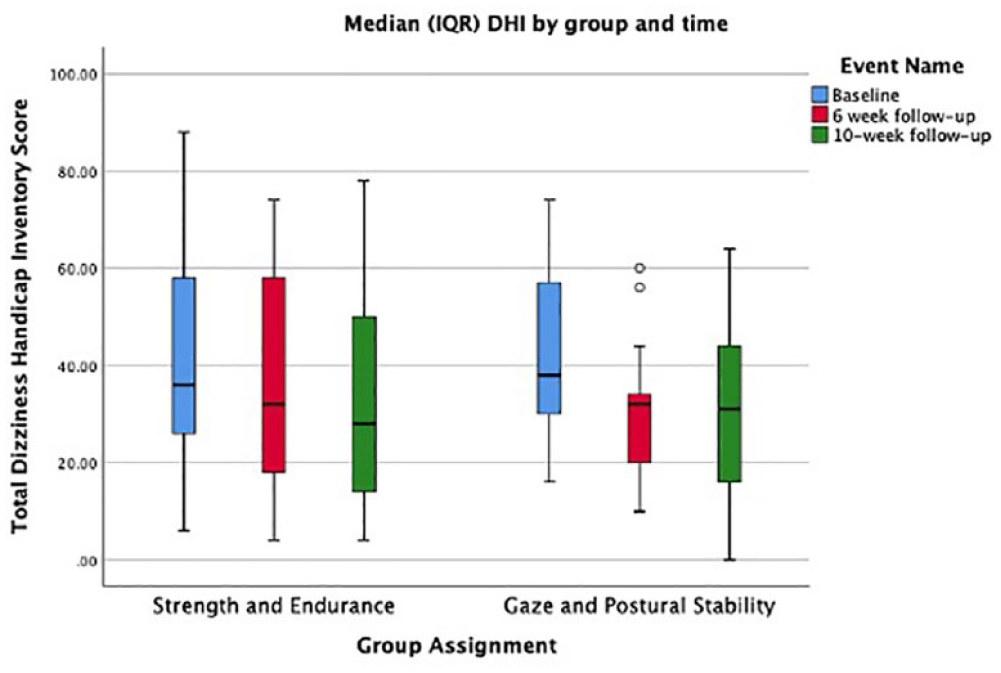
Box and whisker plot displaying the median, IQR, and minimum and maximum scores on the Dizziness Handicap Inventory (DHI) at baseline, 6-week, and 10-week timepoints for both the strength and endurance and gaze and postural stability groups.
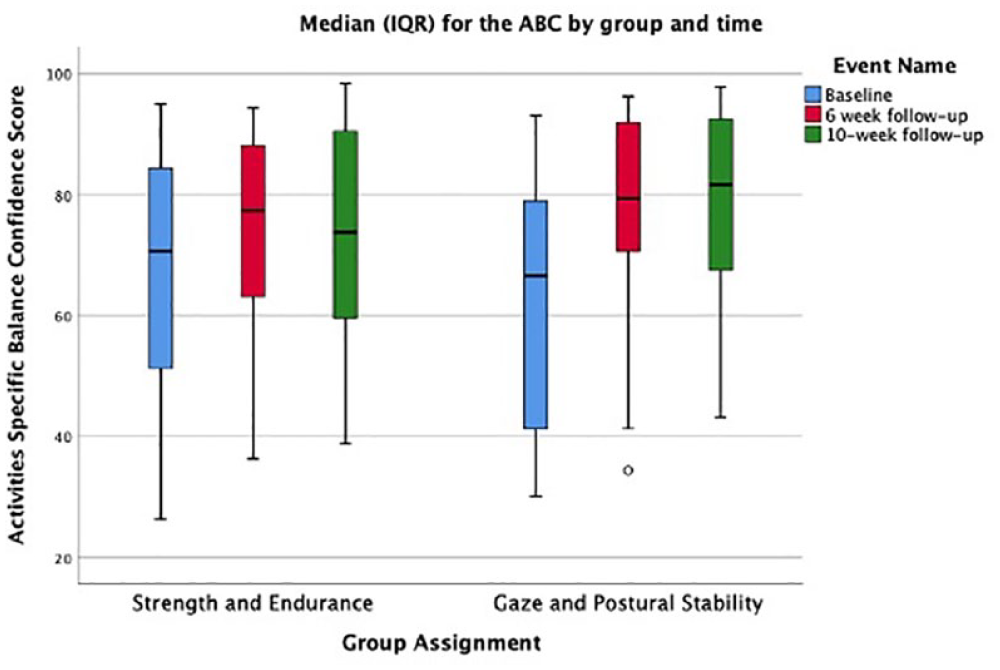
Box and whisker plot displaying the median, IQR, and minimum and maximum scores on the Activities-specific Balance Confidence (ABC) scale at baseline, 6-week, and 10-week timepoints for both the strength and endurance and gaze and postural stability groups.
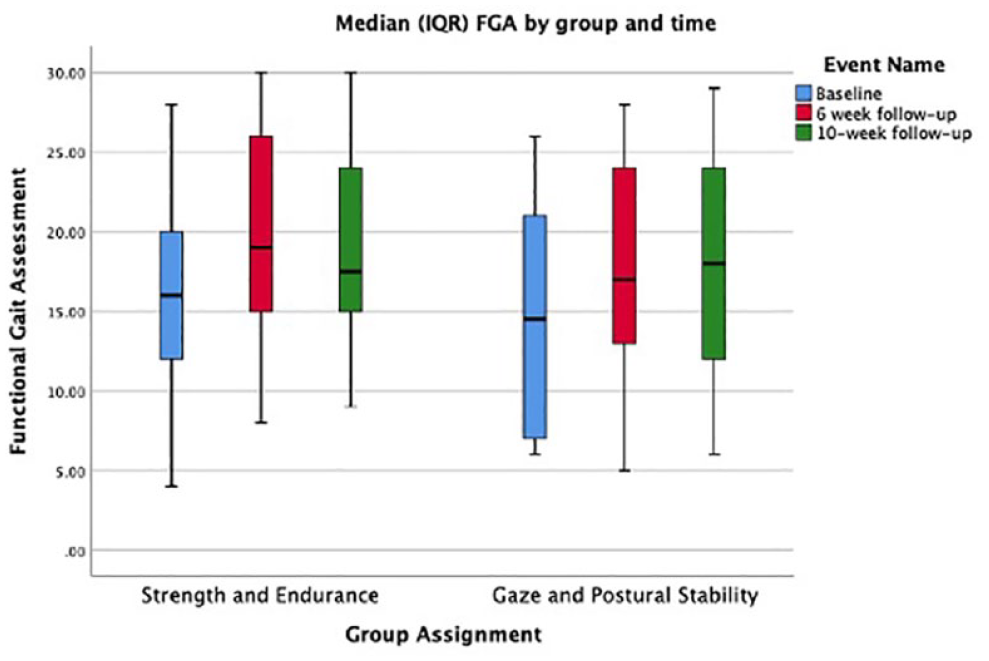
Box and whisker plot displaying the median, IQR, and minimum and maximum scores on the Functional Gait Assessment (FGA) at baseline, 6-week, and 10-week timepoints for both the strength and endurance and gaze and postural stability groups.
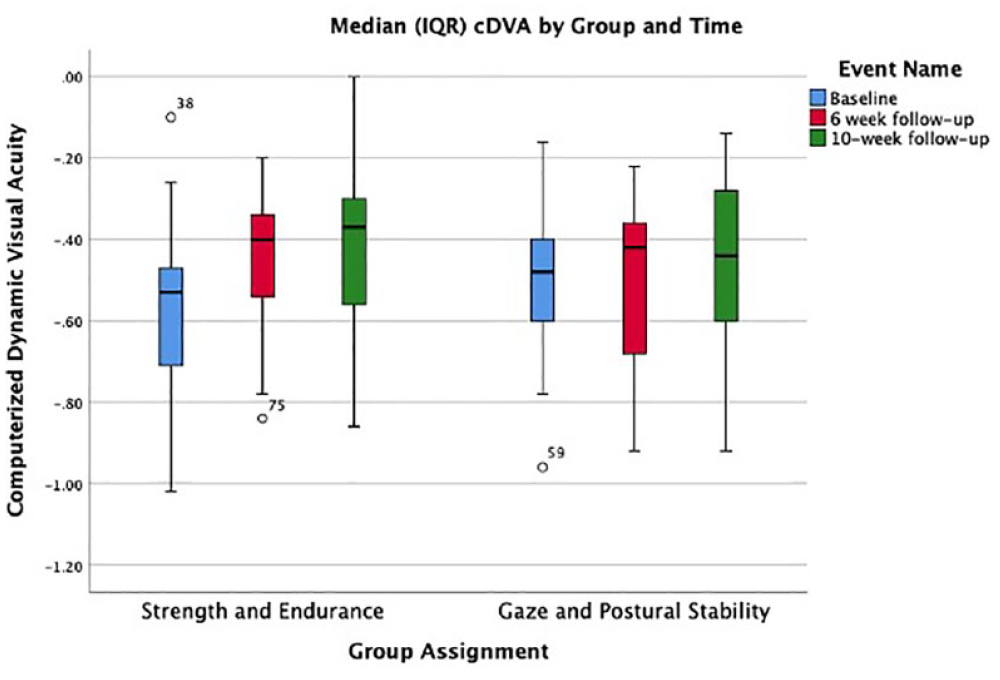
Box and whisker plot displaying the median, IQR, and minimum and maximum scores on the computerized dynamic visual acuity (cDVA) test at baseline, 6-week, and 10-week timepoints for both the strength and endurance and gaze and postural stability groups.
Discussion
This randomized trial was designed to compare the efficacy of a GPS training intervention to a SAE exercise intervention in PwMS. A significant group by time effect was not observed in the primary outcome (DHI) at the primary (6-week) or secondary (10-week) timepoints. Interestingly, significant time effects were observed at 6-weeks and 10-weeks for primary and secondary outcomes, indicating significant improvements in both groups as a result of each exercise program.
Gaze Stability
The gaze stability portion of the GPS intervention was based on evidence supporting the effects of gaze stability exercises to improve vestibulo-ocular reflex (VOR) gain or corrective saccade characteristics and/or diminish motion sensitivity.23,41 Such exercises are well supported for their use in people with peripheral vestibular hypofunction. 44 However, to date the use of these intervention approaches in the management of central mediated vestibular dysfunction, caused by MS, have been under studied. This is despite the fact that dizziness is a common and often-reported symptom in PwMS.4,37 Additionally, anatomic evidence of damage to brainstem vestibular pathways,12,45 and the presence of vestibular patterns in gaze instability have been observed in PwMS.24,33,37 Both groups demonstrated worse baseline scores on the cDVA and DHI than would be expected in a sample of neurotypical adults.46,47 Despite these baseline deficits in vestibular function, our primary directional hypothesis that the intervention group would demonstrate significantly greater improvements in DHI at 6-weeks compared to the SAE group was not supported. There was however a significant time effect, as both groups improved from baseline to 6-weeks (Figure 2). In fact, the effect size observed in the change in DHI score was 0.45 for the GPS group and 0.34 for the SAE group. These effect sizes are considered small to moderate, indicating that either form of exercise appeared to have clinical value at reducing dizziness, measured via the DHI.
The inability for the GPS intervention to drive significant change in DHI scores relative to the SAE group may be explained by several factors. First, unlike past studies exploring the use of vestibular rehabilitation interventions in PwMS, the current study used an active, exercise-based control intervention, as opposed to a true control (ie, non-intervention) group.2,24 The use of a high frequency, progressive program of exercise in the SAE group may explain improvements in the DHI. Basic strengthening and endurance based programs are commonly used, and have been shown to improve health outcomes, such as quality of life in PwMS.26 -29 Although the authors know of no previous work that has assessed the effects of a strengthening program on dizziness, these broad improvements may have carried over to balance and dizziness, measured in the DHI. Further, the DHI, while dizziness and unsteadiness focused, explores complex elements of quality of life across functional, emotional, and physical domains.48,49 Therefore, participation in either form of individualized exercise (vestibular specific or SAE) could lead to change. Secondly, as is common to research working with a heterogeneous population like PwMS, high levels of variability, both at baseline and at follow-up testing (Figure 6) may have contributed to a lack in group differences over time and points to the need for post-hoc examination of data to identify characteristics of those PwMS most likely to respond to vestibular based rehabilitation practices as well as larger or more refined samples (eg, more strict inclusion/exclusion criteria).
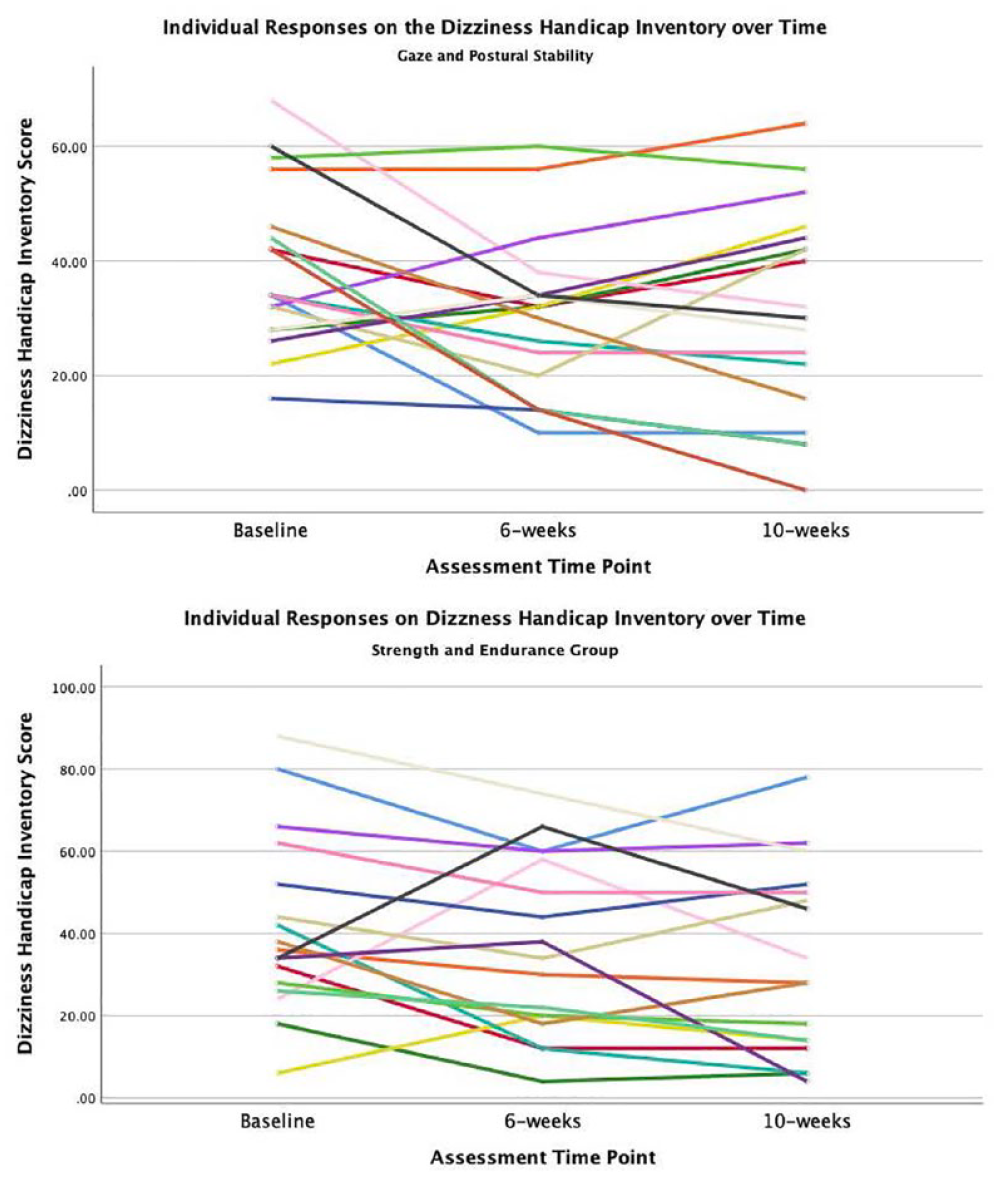
Displaying the individual Dizziness Handicap Inventory (DHI) scores from baseline to the 6- and 10-week timepoints for the strength and endurance and gaze and postural stability groups.
The secondary outcome used to assess gaze stability was the computerized measure of dynamic visual acuity. This measure is considered an activity level outcome and has been shown to correlate with gold standard measures of VOR function.44,50 Furthermore, this measure has been validated and is presented with population level norms in the NIH toolbox study. 32 Similar to what was seen in the assessment of DHI, the between group effect did not reach statistical significance (P = .06) (Figure 5). This result was among the most surprising findings, as the gaze stability portion of the GPS intervention is aimed at improving gaze stability through adaptation of the VOR, or compensation through the recruitment of compensatory saccades. However, previous studies have shown that head movement of any kind, rather than any specific type of exercise is responsible for driving change in cDVA in people with unilateral hypofunction. 51 This may explain the observed findings here as the majority of participants from both groups reported being relatively inactive prior to the study and both participated in weekly exercise that resulted in head movement. Furthermore, the nature of the cDVA measurement and the data presented here does not examine the physiologic changes (eg, improved VOR gain and saccade recruitment) that may have differed between groups, and will be explored further.
Postural Stability
Balance impairments are well documented in PwMS and an improvement in balance and reduction in falls remains a focal point of rehabilitation practice for these individuals. 52 Similar to the gaze stability training program, the postural stability paradigm used in this study was founded in progressive static and dynamic postural control exercises. Progressive balance training approaches similar to this have been used to improve postural control in people with vestibular impairment 23 and PwMS.53,54 Interestingly, in this sample both groups demonstrated relatively equivalent improvements in balance confidence (ABC score) (GPS = 14.2% vs SAE = 9.5%) over the 6-weeks of treatment (Figure 3). However, a medium to large effect size (Cohen’s d = 0.70) was observed in the GPS group while only a small to medium effect size was observed in the SAE group (Cohen’s d = 0.49). Similar to what was observed in the DHI, the intervention, designed to specifically target GPS, resulted in a slightly larger effect size, possibly reflecting the impact it has on patient reported function and perception of impairment or disablement. This lack of group differences was surprising, but mirrored that seen in the gaze stability outcomes.
Activity level changes in postural control were assessed using the FGA. 34 This measure was chosen to capture various elements of dynamic gait likely to influence patient function during daily activity. Furthermore, while the FGA has been used less frequently in the study of PwMS, it has elements designed to specifically challenge the vestibular system (eg, walking with horizontal and vertical head turns, walking with eyes closed) and likely to capture those people with a vestibular pattern of GPS dysfunction. 55 At baseline, mean values on the FGA were lower in the GPS (13.7) and SAE (14.6) groups than were reported previously (FGA = 19) in a sample of 326 people with vestibular dysfunction, 56 but more closely aligned with a sample of 87 individuals with MS (FGA = 14.9). 36 Following intervention, both groups demonstrated mean improvement in FGA scores of approximately 4 points. This is again lower than the mean improvement shown by Marchetti et al 56 through training in people with vestibular loss, but the effect size of this change would be considered moderate in the GPS and SAE groups (Cohen’s d = 0.52 and 0.57; respectively). Notably, the nature of the FGA measurement and the data presented here does not examine the physiologic changes (eg, improved sway metrics, head/gait stability during dynamic activities) that may have differed between groups.
10-Week Follow-Up
Across all primary and secondary outcomes one trend was observed in both groups—a slowing in improvement, but a general maintenance of gains from the 6-week timepoint to the 10-week timepoint. The 10-week assessment was used in this study to examine maintenance of potential gains over a follow-up period during which participants were provided with education, but no further instructions regarding exercise or physical activity. Therefore, the ability for both groups to maintain gains observed during the intervention period was a positive finding of the study. This is especially notable given the progressive, neurodegenerative process of MS. These findings do however point to the need for exercise programs, regardless of the type, that promote long-term patient adherence and maintenance. Allowing people to continue to participate in directed exercise programs or wellness programs that may lead to continued improvement and long term maintenance of gains in PwMS.
Limitations and Directions for Future Research
The current study was not without limitations. First, like in most rehabilitation related research, a true control is difficult to obtain. By design, we chose to compare 2 groups that received exercise interventions, therefore we were unable to examine the efficacy of the GPS intervention in relation to a true inactive control (ie, no exercise). However, the current study design does lend itself to examining the comparative efficacy of the GPS intervention to more commonly used SAE training. Second, daily physical activity measurements and prior level of function measures were not captured. This limited our ability to determine how exercise level prior to entering the study and exercise performed outside of the intervention protocol may of contributed to the results. Finally, considering the substantial amount of variability observed across the outcome measures and the known inherent variability in the presentation and progression of MS, a larger sample or more tightly controlled inclusion and exclusion criteria may have been warranted. Although we attempted to limit the variability of the sample by having inclusion criteria focused on dizziness and falls, these criteria were insufficient at preventing a substantial amount of subject heterogeneity on the outcome measures of interest. Future investigations in this area will utilize post hoc analyses to identify responders versus non-responders to vestibular based intervention.
Conclusions/Implications
In this sample, a progressive GPS intervention, based on principles of rehabilitation for people with peripheral vestibular hypofunction, was found to be statistically equivalent to a strength and aerobic exercise intervention at improving dizziness and balance measures in PwMS. Interestingly, both 6-week exercise programs led to significant improvements in the primary (DHI) and secondary (ABC and FGA) outcomes. Additionally, on average, benefits experienced during the training period were maintained at the 10-week timepoint in both groups.
PwMS who experience issues with dizziness and imbalance are likely to benefit from progressive exercises focused on improving elements of GPS as well as SAE. Future work should focus on determining if there are sub-groups of PwMS who are more likely to benefit from vestibular focused exercises. Identification of individual characteristics that predict responsiveness to specific interventions will allow for better personalization of care and lead to optimized outcomes in this complex patient population.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683221124126 – Supplemental material for Rehabilitation to Improve Gaze and Postural Stability in People With Multiple Sclerosis: A Randomized Clinical Trial
Supplemental material, sj-docx-1-nnr-10.1177_15459683221124126 for Rehabilitation to Improve Gaze and Postural Stability in People With Multiple Sclerosis: A Randomized Clinical Trial by Brian J. Loyd, Annie Fangman, Daniel Peterson, Eduard Gappmaier, Anne Thackeray, Michael Schubert and Leland Dibble in Neurorehabilitation and Neural Repair
Supplemental Material
sj-docx-2-nnr-10.1177_15459683221124126 – Supplemental material for Rehabilitation to Improve Gaze and Postural Stability in People With Multiple Sclerosis: A Randomized Clinical Trial
Supplemental material, sj-docx-2-nnr-10.1177_15459683221124126 for Rehabilitation to Improve Gaze and Postural Stability in People With Multiple Sclerosis: A Randomized Clinical Trial by Brian J. Loyd, Annie Fangman, Daniel Peterson, Eduard Gappmaier, Anne Thackeray, Michael Schubert and Leland Dibble in Neurorehabilitation and Neural Repair
Supplemental Material
sj-docx-3-nnr-10.1177_15459683221124126 – Supplemental material for Rehabilitation to Improve Gaze and Postural Stability in People With Multiple Sclerosis: A Randomized Clinical Trial
Supplemental material, sj-docx-3-nnr-10.1177_15459683221124126 for Rehabilitation to Improve Gaze and Postural Stability in People With Multiple Sclerosis: A Randomized Clinical Trial by Brian J. Loyd, Annie Fangman, Daniel Peterson, Eduard Gappmaier, Anne Thackeray, Michael Schubert and Leland Dibble in Neurorehabilitation and Neural Repair
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: National Multiple Sclerosis Society NMSS RG-1701-26763-Primary sponsor of the project, responsible for partial salary support for all authors, participant enrollment costs, other research costs. The lead author, Brian J. Loyd, received salary supported in part by the Foundation for Physical Therapy Research New Investigator Fellowship Training Initiative (NIFTI).
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
