Abstract
Background. Brain injury often causes severe motor dysfunction, leading to difficulties with living a self-reliant social life. Injured neural circuits must be reconstructed to restore functions, but the adult brain is limited in its ability to restore neuronal connections. The combination of molecular targeting, which enhances neural plasticity, and rehabilitative motor exercise is an important therapeutic approach to promote neuronal rewiring in the spared circuits and motor recovery. Objective. We tested whether genetic reduction of Src homology 2-containing phosphatase-1 (SHP-1), an inhibitor of brain-derived neurotrophic factor (BDNF)/tropomyosin receptor kinase B (TrkB) signaling, has synergistic effects with rehabilitative training to promote reorganization of motor circuits and functional recovery in a mouse model of brain injury. Methods. Rewiring of the corticospinal circuit was examined using neuronal tracers following unilateral cortical injury in control mice and in Shp-1 mutant mice subjected to voluntary exercise. Recovery of motor functions was assessed using motor behavior tests. Results. We found that rehabilitative exercise decreased SHP-1 and increased BDNF and TrkB expression in the contralesional motor cortex after the injury. Genetic reduction of SHP-1 and voluntary exercise significantly increased sprouting of corticospinal tract axons and enhanced motor recovery in the impaired forelimb. Conclusions. Our data demonstrate that combining voluntary exercise and SHP-1 suppression promotes motor recovery and neural circuit reorganization after brain injury.
Introduction
Brain injury often leads to loss of motor, sensory, and cognitive functions, which makes it difficult for affected patients to live a self-reliant social life. At the clinical and experimental level, lost motor functions often spontaneously recover owing to the compensatory reorganization of spared motor circuits, including the corticospinal tract (CST),1-3 brainstem-spinal tracts, 4 and cortico-rubral pathway. 5 We previously demonstrated in a mouse model of brain injury that the intraspinal rewiring of CST fibers is critical for functional recovery.2,6 The fibers sprout toward the denervated side of the cervical cord and make functional connections with propriospinal and segmental interneurons, which are the dominant spinal neurons that control the paralyzed limb muscles.1,2 However, because the rewiring is not sufficient for full motor recovery, treatments that promote both axonal sprouting and synapse formation in the appropriate target neurons are required.
Brain-derived neurotrophic factor (BDNF) and its receptor, tropomyosin receptor kinase B (TrkB), are important signaling components which mediate axon outgrowth, 7 spine formation,8-10 and reorganization of spared neural circuits. 2 We previously reported that axonal sprouting is enhanced by the exogenous overexpression of BDNF following mouse brain injury. 2 However, several researchers have expressed concern that treatment using neurotrophic factors also stimulates axonal sprouting or synapse formation in nonprecise targets, resulting in aberrant neural circuits. 11 To establish appropriate neural connections and promote functional recovery, the induction of synchronized neural activity, which is generated through exercise, is required. 12 Rehabilitative motor training is an important and effective method for the reformation of appropriate neural circuits after central nervous system (CNS) injuries. Indeed, various rehabilitative exercises have been shown to increase BDNF levels13-17 and improve functional recovery after brain injury.18-25 Specifically, we showed that bilateral movement training improves paretic forelimb function and enhances reorganization of spared neural circuits in mice. 3
We previously showed that inhibition of Src homology 2-containing phosphatase-1 (SHP-1), an intracellular cytoplasmic protein tyrosine phosphatase (PTP) that inhibits BDNF/TrkB signaling, promotes sprouting of CST axons. 6 These effects were mediated by the enhancement of BDNF/TrkB signaling and suppression of paired immunoglobulin-like receptor B (PirB), a receptor for myelin-associated inhibitors of axonal regeneration.26,27 In the present study, we hypothesized that genetic reduction of SHP-1 combined with rehabilitative exercises additively enhance CST rewiring and improve motor function following brain injury. We demonstrated that the combinational approach could promote plastic changes in the circuits and motor recovery.
Methods
Ethics
All experiments were conducted in accordance with the guide for the care and use of laboratory animals and were approved by the institutional review committee of Kindai University and Kanazawa Medical University.
Animals
C57BL/6J male mice (Japan SLC) and mice carrying the motheaten (me) mutation at the PTP nonreceptor type 6 (Ptpn6) locus, known as the Ptpn6me (Jackson Laboratory), were used. The mice with me mutation have severely impaired expression of SHP-1 (also termed PTP1C).28-30 Mice were propagated by mating +/+ with +/me heterozygotes, thereby producing +/+ and +/me offspring (Supplementary Methods). The +/+ mice were used as wild-type (WT) controls. Since the me/me mice develop systemic autoimmunity and have a shortened life span,
31
+/me mice were used in this study. The mice were each housed in a standard cage on a 12-hour light/dark cycle (lights on at 7:00
Brain Injury Models
The mice were anesthetized using medetomidine (0.75 mg/kg), midazolam (4.0 mg/kg), and butorphanol (5.0 mg/kg) and placed on a stereotaxic frame (Narishige). The skull was exposed with a midline skin incision, and the skull bone overlying the left motor cortex was removed. Hemicortical lesions with a depth of 1.0 mm were induced by cortical ablation of the left motor cortex using a pipette in 9- to 16-week-old male mice (WT, n = 109; +/me, n = 53).6,32 The injured area consisted of the rostral forelimb area (RFA) and caudal forelimb area (CFA) of the cerebral cortex. 33 Thereafter, the skull was replaced, the wound was sutured, and the mice were returned to the cages. Age-matched, sham-operated mice (n = 24) that underwent same surgical procedures without cortical ablation were used as controls.
Rehabilitative Exercises
The mice were randomly divided into 6 groups: nonexercised sham-operated mice (sham control), exercised sham-operated mice, nonexercised brain injury mice (control), nonexercised +/me brain injury mice, exercised brain injury mice, and exercised +/me brain injury mice.
The voluntary running exercise was slightly modified from a previous study. 34 WT and +/me mice were single housed in a cage with or without a running wheel (MK-713: Muromachi Kikai). Using the software in the computer connected to the running wheels (CompACT AMS Ver.3: Muromachi Kikai), the number of rotations of the running wheel placed in each cage was recorded during each 12-hour night cycle. The exercise was conducted for 2 weeks prior to brain injury to acclimatize the mice so that the number of rotations plateaus and was resumed at day 5 post-injury to day 28.
The rotarod running exercise was performed as described previously. 3 Briefly, the mice were placed on a rotating rod (diameter 30 mm) of a rotarod (LE8500: Panlab) that accelerated from 4 to 40 rpm. Both WT and +/me mice were trained for 30 minutes per day, 6 days per week, for 2 weeks before brain injury. The exercise was resumed at day 5 postinjury to allow the mice to recover from the surgery.
Real-Time Polymerase Chain Reaction
Total RNA was extracted from the contralesional cerebral cortex, including RFA, and CFA using TRIzol reagent (Invitrogen). The RNA was then reverse transcribed to synthesize first-strand cDNA by using ReverTra Ace qPCR RT Master Mix with gDNA Remover (TOYOBO). Real-time polymerase chain reaction (PCR) was then performed using oligonucleotide primer sets corresponding to the following mouse cDNA sequences: Shp-1, forward (GGACTTCTATGACCTGTACGGA) and reverse (CGAGCAGTTCAGTGGGTACTT); Bdnf, forward (TCATACTTCGGTTGCATGAAGG) and reverse (AGACCTCTCGAACCTGCCC); TrkB, forward (CTGGGGCTTATGCCTGCTG) and reverse (AGGCTCAGTACACCAAATCCTA); and Gapdh, forward (AACCTGCCAAGTATGATGACA) and reverse (GGAAGAGTGGGAGTTGCTGT). The cDNA samples were preheated at 98 °C for 2 minutes and subjected to 40 cycles of amplification (denaturation at 98 °C for 10 seconds, annealing at 60 °C for 10 seconds, and extension at 68 °C for 30 seconds) using KOD SYBR qPCR Mix (QKD-201, TOYOBO) and the QuantStudio 12K Flex Real-Time PCR System (Thermo Fisher Scientific). Relative mRNA expression was calculated by normalizing to Gapdh mRNA expression. The cycle threshold values were calculated using the ΔΔ cycle threshold method to obtain fold difference.
Immunohistochemistry
Mice were transcardially perfused with 4% paraformaldehyde in phosphate-buffered saline (PBS). The brains were dissected, postfixed overnight at 4 °C in the same fixatives, and immersed overnight at 4 °C in PBS containing 30% sucrose. The brain specimens were then embedded in Tissue-Tek OCT compound (Sakura Finetek) and kept frozen at −80 °C until use. Transverse 20-µm sections of cervical spinal cord were prepared using a cryostat (CM1510S, Leica Biosystems). For immunohistochemical analysis, the sections were rinsed in PBS, blocked with 5% bovine serum albumin and 0.3% Triton X-100 in PBS for 1 hour at room temperature, and then incubated with a primary antibody overnight at 4 °C. The primary antibody was rabbit anti-protein kinase Cγ (PKCγ; 1:500, sc-211, RRID: AB_632234, Santa Cruz Biotechnology), mouse anti-NeuN (1:2,000, MAB377, RRID: AB_2298772, Millipore), or rabbit anti-c-Fos (1:500, 2250S, RRID: AB_2247211, Cell Signaling Technology). The sections were washed in PBS and incubated with secondary antibodies for 1 hour at room temperature. Rhodamine Red-X anti-mouse IgG (1:1,000, 715-295-151, RRID: AB_2340832, Jackson ImmunoResearch Labs) or Alexa Fluor 488 anti-rabbit IgG (1:1,000, A-21206, RRID: AB_141708, Molecular Probes) was used as a secondary antibody. All images were acquired using fluorescent microscopes (BZ-X710, Keyence; ApoTome.2, Zeiss).
Histological Quantification of c-Fos-Positive Neurons
c-Fos-expressing neurons in the cervical cord were quantified at day 7 after brain injury in mice subjected to voluntary exercise (for 2 weeks preinjury and from day 5 postinjury) and those without exercise. To maintain constant conditions of locomotor stimulation among animals, the mice were perfused at 8:00
Anterograde CST Labeling
The anterograde tracer was injected as described previously. 6 Two weeks after brain injury, mice were anesthetized and placed on a stereotaxic frame. Small holes were made in the contralesional right CFA using a drill. To label uninjured motor CST, biotinylated dextran amine (BDA; 10% in PBS, Invitrogen), an anterograde tracer, was injected at a depth of 0.5 mm using a microsyringe tipped with a glass capillary (tip diameter: 70 μm) at the following locations within the CFA (0.6 µL/site): 1.0 mm lateral, 1.0 mm lateral and 0.5 mm anterior, 1.5 mm lateral and 0.5 mm anterior, and 1.5 mm lateral to the bregma. The injected area consisted of motor CST neurons. 35 After the injections, the wound was sutured, and the mice were returned to the cages. They were sacrificed 2 weeks after BDA injection (4 weeks after injury) by transcardial perfusion with 4% paraformaldehyde in PBS. The cervical cord was dissected, post-fixed in the same fixatives, and immersed overnight in PBS containing 30% sucrose. The cryosections were incubated for 4 hours in PBS supplemented with 0.3% Triton X-100, and then for 2 hours with Alexa Fluor 488-conjugated streptavidin (1:400, S11223, RRID: AB_2336881, Thermo Fisher Scientific) in PBS with 0.3% Triton X-100 at room temperature.
Retrograde Labeling of Segmental Interneurons
Three weeks after brain injury, the mice were anesthetized and their brachial muscles were exposed following a skin incision. To label segmental spinal interneurons, wheat germ agglutinin (WGA; 2% in saline, Sigma-Aldrich), a trans-synaptic retrograde tracer, 36 was infused into the biceps brachii and triceps brachii muscles of the impaired right forelimb at 5 sites per muscle (5 μL per muscle) using a microsyringe tipped with a glass capillary (tip diameter: 200 μm). After infusion, muscles and skin layers were sutured.
The mice were sacrificed 1 week after WGA injection (4 weeks after injury). Following the processes of BDA-staining, the sections were fixed for 15 minutes in 4% paraformaldehyde in PBS, rinsed in PBS, blocked using 5% bovine serum albumin and 0.2% Triton X-100 in PBS for 1 hour at room temperature, and incubated with a following primary antibody overnight at 4 °C: rabbit anti-lectin triticum vulgaris (1:5,000, T4144, RRID: AB_261669, Sigma-Aldrich), mouse anti-NeuN (1:2,000, MAB377, RRID: AB_2298772, Millipore), or guinea pig anti-vGlut1 (1:10,000, AB5905, RRID: AB_2301751, Millipore). The sections were washed in PBS and incubated with a secondary antibody for 1 hour at room temperature: Alexa Fluor 568 anti-rabbit IgG (1:500, A-11011, RRID: AB_143157, Molecular Probes), Rhodamine Red-X anti-mouse IgG (1:1,000, 715-295-151, RRID: AB_2340832, Jackson ImmunoResearch Labs), or Rhodamine Red-X anti–guinea pig IgG (1:1,000, 706-295-148, RRID: AB_2340468, Jackson ImmunoResearch Labs).
Quantitative Analysis of Sprouting in the Uninjured CST
Motor CST fibers projecting from the contralesional CFA to the denervated cervical cord gray matter were quantitatively analyzed by using fluorescence microscopy (BZ-X710, Keyence) at 200× magnification. The number of BDA-positive midline-crossing fibers was counted in serial transverse sections of the cervical cord (C4–7; 10 sections per cervical segment), and the value was normalized to the number of main dorsal CST fibers at C4 level (% crossing fibers).2,6 The number of BDA-positive fibers in the dorsal CST was determined from images captured by a fluorescent microscope (ApoTome.2, Zeiss) and ImageJ software (National Institutes of Health). The pixel axon intensity of 2.5-D image of the BDA-labeled CST fibers in the spinal gray matter was further measured using the ApoTome.2 software (Zeiss), and the intensity ratio between the denervated side and the healthy side was calculated for normalization.
Grid-Walk Test
The grid-walk test assesses an animal’s ability to accurately place its forepaws on the rungs of a grid during spontaneous exploration.
37
Mice were placed on a wire grid (30 × 30 cm) with 12-mm square holes and allowed to freely explore for 2 to 3 minutes; their performance was recorded using a video camera. The number of foot slips of the impaired right forepaw was assessed as a percentage of the first 100 steps. A foot slip was scored when the paw completely missed a rung or when the paw was correctly placed on a rung but slipped off while bearing body weight. The grid-walk test does not require training, but each animal was tested once before surgery to obtain baseline scores. This behavioral test was performed between 8:30
Rotarod Test
The rotarod is used to assess motor function and coordination in rodents. 38 Mice were placed on a rotating rod (diameter 30 mm) that accelerated from 4 to 40 rpm within 5 minutes. 39 Mice were trained twice a day for 2 days before injury. Timing stopped when the mouse fell off the rotating rod. The baseline value was scored as the mean of 3 trials 1 day before brain injury.
Statistical Analysis
All data are presented as means ± standard error of the mean (mean ± SEM). The motor function scores were compared using repeated-measures 2-way analysis of variance (ANOVA) followed by the post hoc Bonferroni test. The relationship between recovered motor function score and number of running wheel rotations per day was tested using Pearson’s correlation coefficient. All other results were compared using 1-way ANOVA followed by Tukey-Kramer test. All analyses were performed by the IBM SPSS statistics software version 25 (IBM Corp). P values less than .05 were considered statistically significant.
Results
Voluntary Exercise Decreases SHP-1 Expression in the Contralesional Cortex
First, we investigated SHP-1 expression in the cerebral cortex of mice with brain injury that underwent rehabilitative exercise. Expression was tested in the contralesional motor cortex, which contained spared corticospinal neurons that could contribute to motor recovery.2,6 The exercises were conducted for 2 weeks before the injury and resumed on day 5 postinjury. mRNAs were collected at day 7 for gene expression analysis (Figure 1A). We evaluated 2 running exercise routines, rotarod running and voluntary running on a wheel, which are bilateral movement trainings that possibly enhance CST rewiring and motor recovery (Figure 1B and C). 3 The approximate running distances on the rotarod and the wheel were 65 m and 5500 to 18 000 m per day, respectively. As we previously reported, 6 the expression of Shp-1 mRNA was increased in the contralesional motor cortex at day 7 post–brain injury (Figure 1D). We found that voluntary running attenuated the elevation of Shp-1 mRNA expression (Figure 1D). In contrast, rotarod running did not influence the expression of Shp-1 mRNA (Figure 1E). The data suggest that voluntary running with high amount of exercise may change SHP-1 signaling in the spared cortical region after the injury.
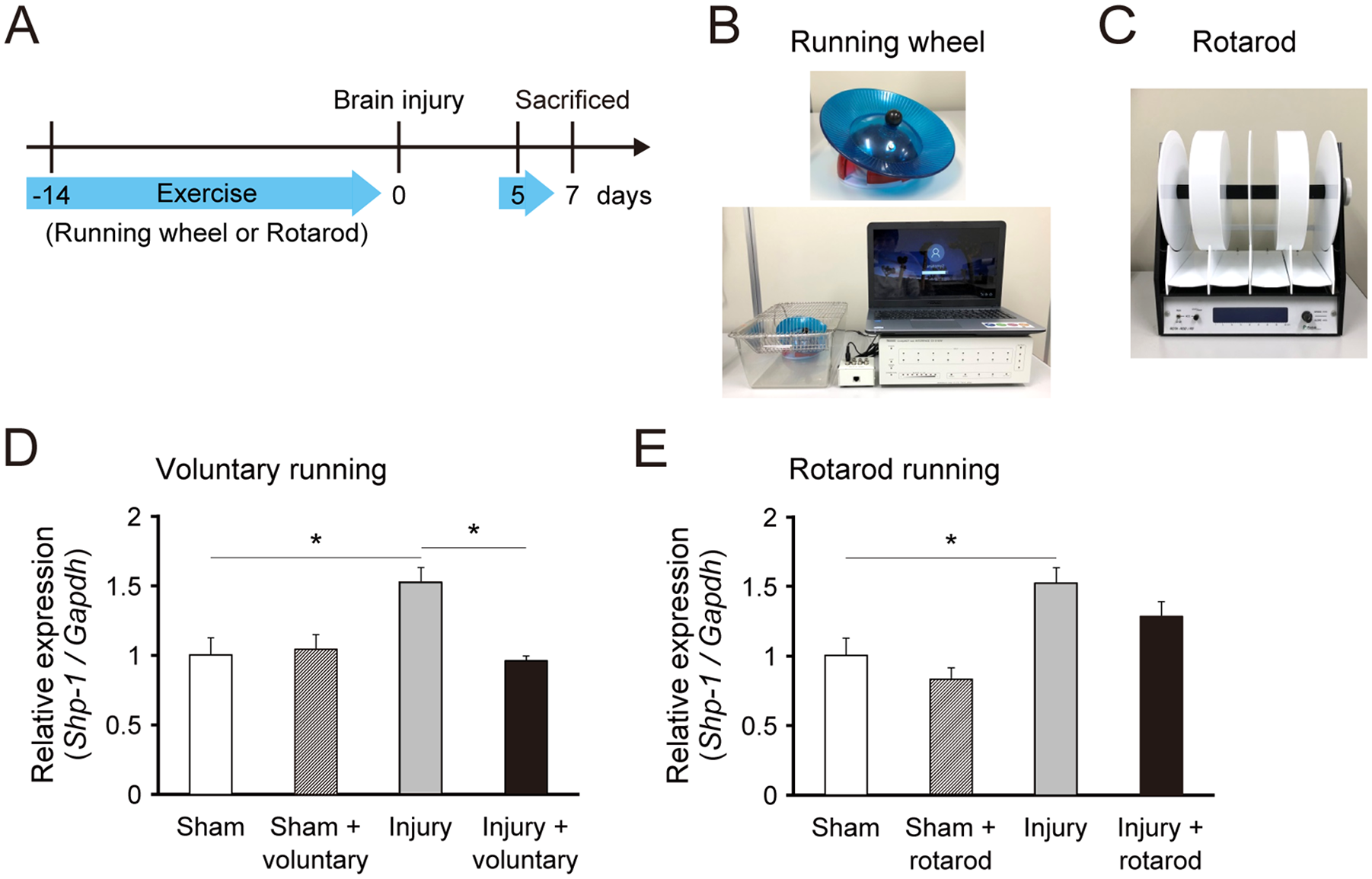
Voluntary exercise decreases SHP-1 expression in the contralesional motor cortex following brain injury. (A) mRNAs of the motor cortex were collected at day 7 postinjury in mice with/without exercise. The exercises were conducted over 2 weeks before the injury and resumed on day 5 after the injury. (B and C) The running exercise experiments were conducted using the running wheel (B) and the rotarod (C). (D and E) Relative expression of Shp-1 mRNA in the contralesional motor cortex on day 7 after injury in wild-type (WT) mice subjected to voluntary running (D) or rotarod running (E), quantified by real-time polymerase chain reaction (n = 3-4 in each group). Data are presented as means ± SEM. *P < .05 (1-way analysis of variance followed by Tukey-Kramer test).
We next examined the sequential changes of Shp-1 expression in voluntary running group. WT mice underwent voluntary exercise pre- and postinjury that was resumed at day 5 (Figure 2A). In nonexercise control mice, Shp-1 mRNA levels were significantly increased at days 1, 3, and 7 postinjury. The voluntary running attenuated the elevation at day 7, after the exercise was resumed (Figure 2B). This suggests that postinjury, but not preinjury, exercise would change SHP-1 expression.
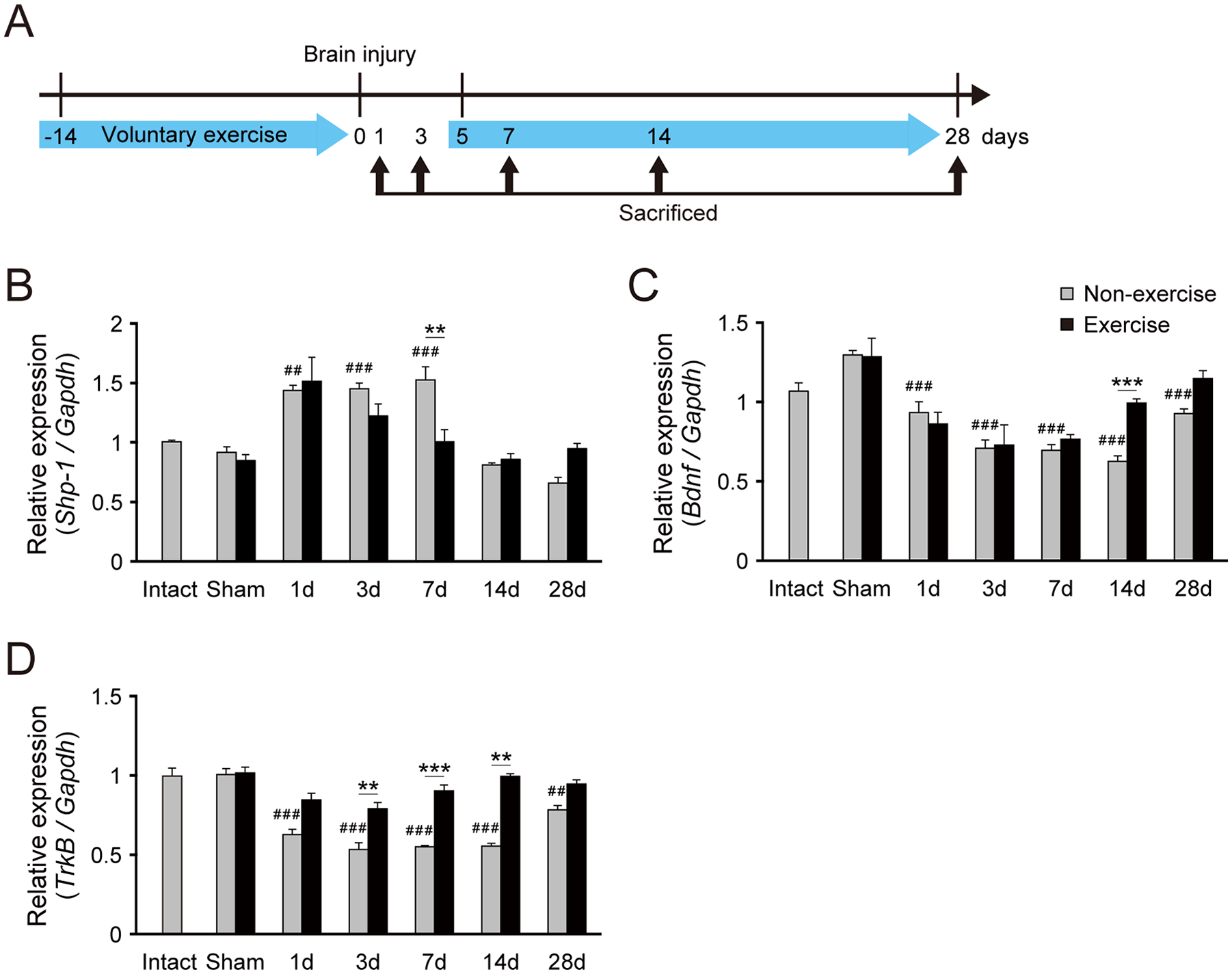
Voluntary exercise increases BDNF and TrkB expression and decreases SHP-1 expression in the contralesional motor cortex following brain injury. (A) mRNAs of the motor cortex were collected on days 1, 3, 7, 14, and 28 postinjury of mice with/without voluntary exercise on the running wheel. (B-D) Relative expression of Shp-1 (B), Bdnf (C), and TrkB (D) mRNA in the contralesional motor cortex, quantified by real-time polymerase chain reaction (n = 3-7 in each group). Data are presented as means ± SEM. **P < .01, ***P < .001 (1-way analysis of variance [ANOVA] followed by Tukey-Kramer test); ##P < .01, ###P < .001 (vs nonexercised sham, 1-way ANOVA followed by Tukey-Kramer test).
Voluntary Exercise Induces BDNF and TrkB Expression in the Contralesional Cortex
The altered Shp-1 expression after voluntary exercise led us to examine the effects of such exercise on BDNF and TrkB signals. We investigated Bdnf and TrkB expressions in the contralesional motor cortex from day 1 to 28. We found that Bdnf and TrkB mRNA levels were significantly decreased after injury in nonexercised mice (Figure 2C and D). However, voluntary exercise attenuated the decrease of Bdnf mRNA levels on day 14 after injury (Figure 2C). Similarly, voluntary exercise suppressed the reduction of TrkB mRNA levels on days 3, 7, and 14 after injury (Figure 2D). Taken together, the data suggest that voluntary exercise enhances BDNF and TrkB signaling in the spared motor cortex.
Voluntary Exercise Increases Neuronal Activity in the Cervical Cord
To determine whether the combination of voluntary exercise and SHP-1 reduction promote CST rewiring and motor recovery, we induced brain injury in WT and +/me mice. To ensure that the injury was similar among the mouse groups, we evaluated lesion volume in the brain and CST destruction in the cervical cord. Nissl staining of coronal brain sections on day 28 revealed that the injury led to complete destruction of the left CFA (Figure 3A). Lesion volume did not differ significantly among the nonexercised WT, non-exercised +/me, exercised WT, and exercised +/me mouse groups (Figure 3B). Immunostaining for PKCγ showed extremely low expression in the dorsal CST of the lesioned side on day 28 (Figure 3C). The CST destruction area did not differ among the groups (Figure 3D).
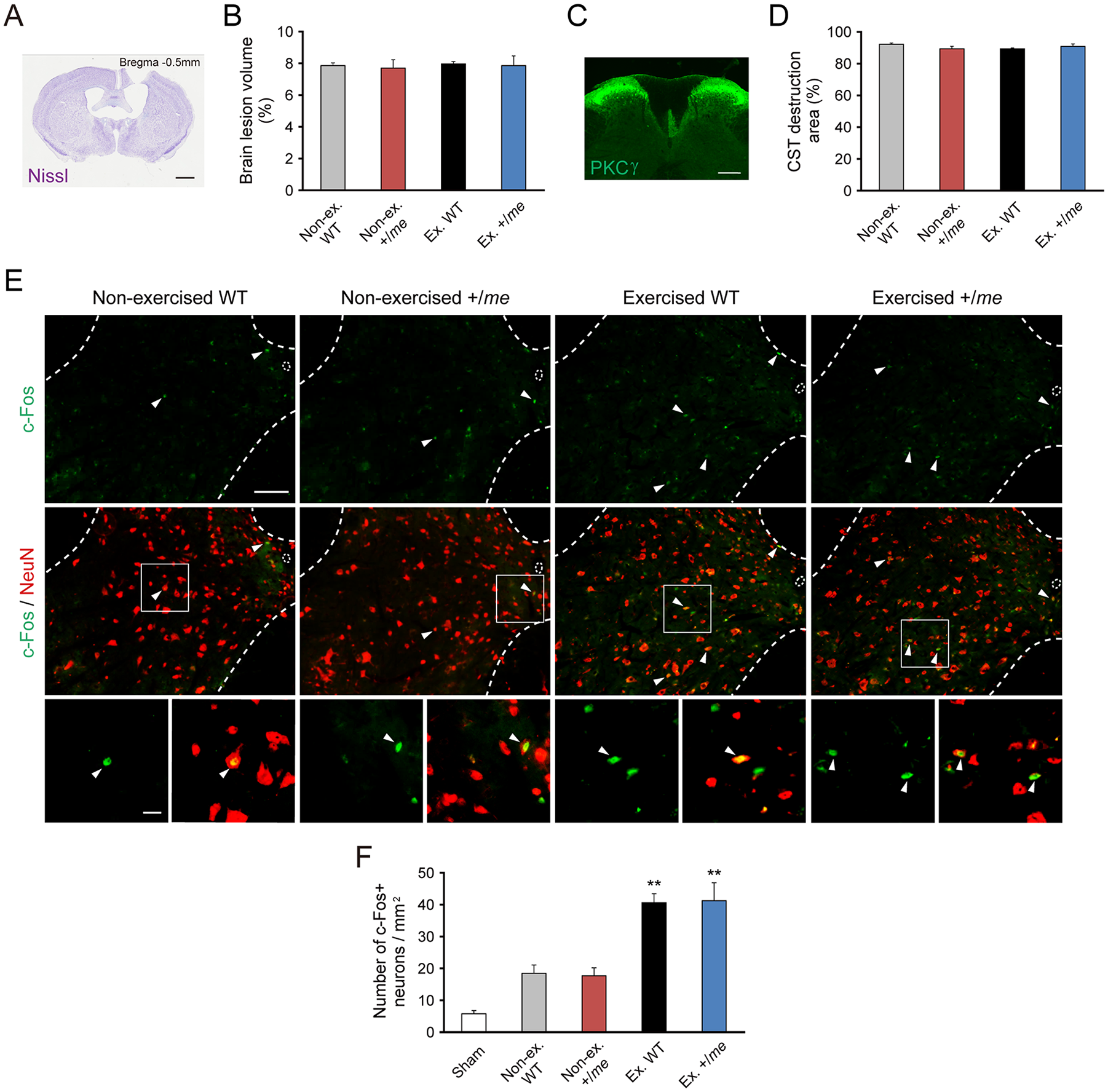
Voluntary exercise increases neural activity in the cervical cord following brain injury. (A) Nissl staining of a coronal section of an injured brain, showing the lesion in the left hemisphere (28 days after injury). Scale bar: 1 mm. (B) Quantitative data for the brain lesion volume (nonexercised wild-type [WT], n = 5; nonexercised +/me, n = 5; exercised WT, n = 4; exercised +/me, n = 5). Data are presented as means ± SEM. (C) Representative image of PKCγ immunostaining of the dorsal corticospinal tract (CST) in a transverse section of the cervical cord (day 28 postinjury). Scale bar: 200 μm. (D) Quantitative data on the extent of damage in the dorsal CST assessed by PKCγ immunohistochemistry (nonexercised WT, n = 5; nonexercised +/me, n = 5; exercised WT, n = 4; exercised +/me, n = 5). Data are presented as means ± SEM. (E) Immunostaining for c-Fos (green) and NeuN (red) in C6 of WT and +/me mice. Arrowheads indicate cells positive for both c-Fos and NeuN. Scale bars: upper and middle panels (low magnification), 100 μm; lower panels (high magnification), 20 μm. (F) The number of c-Fos positive neurons in Rexed laminae VII, VIII, and X on the denervated side at C4-C7 (Sham, n = 3; nonexercised WT, n = 5; nonexercised +/me, n = 5; exercised WT, n = 4; exercised +/me, n = 5). Data are presented as means ± SEM. **P < .01 (vs nonexercised WT, 1-way analysis of variance followed by Tukey-Kramer test).
We next examined the activity of neurons in the denervated side of the cervical cord (Rexed laminae VII, VIII, and X) in voluntarily exercised mice by assessing the expression of c-Fos, an activity-dependent intermediate-early gene (Figure 3E). The number of c-Fos positive neurons, colabeled with NeuN, was significantly higher in exercised WT and +/me mice than in non-exercised WT and +/me mice, while no significant differences were found between exercised WT and +/me mice (Figure 3F). This indicates that voluntary exercise increases neuronal activity in the cervical cord of both mouse strains.
Voluntary Exercise and SHP-1 Reduction Increase CST Sprouting
We then tested whether the combination of voluntary exercise and SHP-1 reduction increases CST sprouting. We injected BDA into the contralesional motor cortex to label spared CST in voluntarily exercised mice (Figure 4A). BDA-labeled CST axons had sprouted into the denervated side of the cervical cord in exercised WT and +/me mice 4 weeks after the injury (Figure 4B). The number of sprouting axons from the uninjured CST that had crossed the midline to the denervated side was significantly higher in exercised WT mice than in nonexercised WT mice, and it was even higher in exercised +/me mice (Figure 4C). We further compared the amount of innervating CST fibers in the denervated side with that in the healthy side (Figure 4D). The intensity of BDA-labeled CST fibers was significantly increased in the denervated side of exercised +/me mice at the C4 level (Figure 4E).
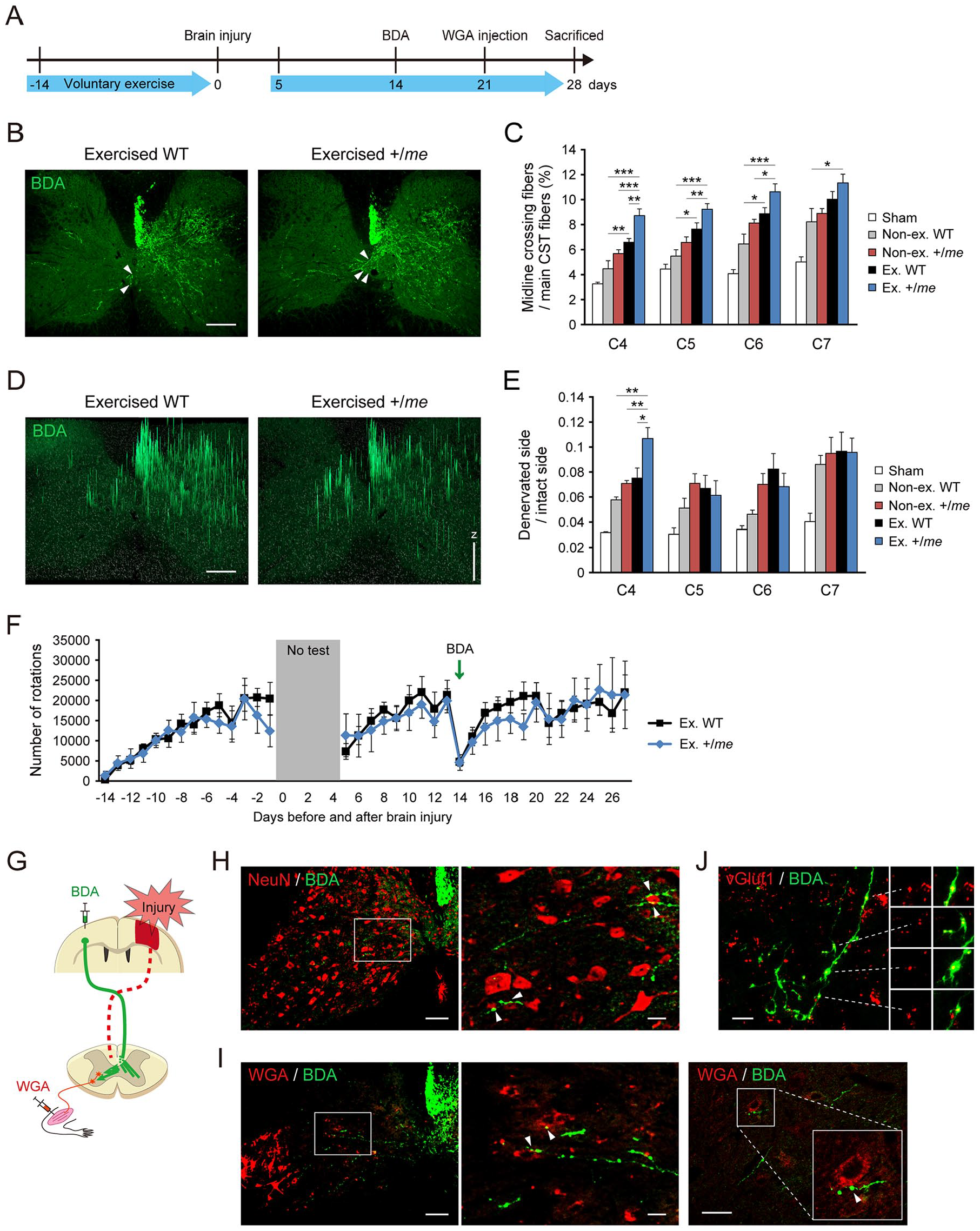
Combination of SHP-1 genetic reduction and voluntary exercise promotes corticospinal tract (CST) sprouting following brain injury. (A) The experimental protocol for biotinylated dextran amine (BDA) and wheat germ agglutinin (WGA) injections. BDA was injected into the contralesional motor cortex 2 weeks after brain injury. WGA infused into the impaired right forelimb muscles 3 weeks after the injury. (B) BDA-labeled CST axons (green) in transverse cervical cord sections from exercised wild-type (WT) and +/me mice (day 28 postinjury). Arrowheads indicate midline-crossing CST fibers. Scale bar: 200 µm. (C) Quantitative data on the number of CST axons crossing the midline (Sham, n = 4; nonexercised WT, n = 6; nonexercised +/me, n = 7; exercised WT, n = 9; exercised +/me, n = 5). Data are presented as means ± SEM. *P < .05, **P < .01, ***P < .001 (1-way analysis of variance [ANOVA] followed by Tukey-Kramer test). (D) Pixel axon intensity of 2.5-D images of BDA-labeled CST fibers (green) in the cervical cord of exercised WT and +/me mice at day 28 postinjury, constructed using ApoTome.2 software. Scale bar: 200 µm. Z projection: fluorescence arbitrary unit. (E) Quantitative data on the ratio of CST axon pixel intensity on the denervated side to that on the intact side (Sham, n = 3; nonexercised WT, n = 3; nonexercised +/me, n = 5; exercised WT, n = 5; exercised +/me, n = 5). Data are presented as means ± SEM. *P < .05, **P < .01 (1-way ANOVA followed by Tukey-Kramer test). (F) The number of rotations of the running wheel per day over 6 weeks in WT and +/me mice subjected to CST labeling after brain injury (WT mice, n = 9; +/me mice, n = 5). Voluntary exercise was not performed during the first 4 days after injury (gray shaded box). (G) Schematic illustration of tracing experiments of CST and spinal interneurons. Brain injury to the sensorimotor cortex (red) destroys the CST on one side (dotted lines). BDA, an anterograde tracer, was injected into the contralesional motor cortex to label spared CST axons. WGA, a trans-synaptic retrograde tracer, was injected into the impaired forelimb muscles to label premotor spinal interneurons. (H) Immunostained images of BDA-labeled CST fibers (green) and NeuN-positive cervical cord neurons (red) in the denervated side of exercised +/me mice at day 28 postinjury. Scale bars: left panel (low magnification), 100 μm; right panel (high magnification), 20 μm. (I) Images of BDA-labeled CST fibers (green) and WGA-labeled segmental interneurons (red) in exercised WT mice at day 28 postinjury. Scale bars: left panel (low magnification), 100 μm; middle and right panel (high magnification), 20 μm. Arrowheads indicate contacts between CST axons and neurons with bouton structures. (J) Images of BDA-labeled CST fibers (green) and vGlut1-labeled presynaptic vesicles (red) in the denervated side of exercised +/me mice at day 28 postinjury. Scale bar: 20 μm.
The amount of voluntary exercise was measured over a period of 6 weeks in WT and +/me mice that had received BDA injection. The number of running wheel rotations was not significantly different between WT and +/me mice either before or after brain injury, even after BDA injection (Figure 4F), indicating that the observed increase in CST sprouting in +/me mice is not dependent on increased exercise.
We next examined whether the sprouting CST fibers form neuronal connections with spinal interneurons. In this experiment, the CST from the contralesional motor cortex was traced using BDA. Subsequently, WGA was injected into the forelimb muscles of the impaired side to label motor neurons and premotor spinal interneurons (Figure 4G). BDA-labeled sprouting CST axons contacted NeuN positive spinal neurons with bouton structures (Figure 4H). We also found that CST axons also contacted WGA-labeled segmental interneurons in exercised WT and +/me mice with bouton structures (Figure 4I, Supplementary Figure 1A). Most of the boutons in CST fibers were labeled with vGlut1, indicating that they were presynaptic structures (Figure 4J, Supplementary Figure 1B).2,40 Although the efficiency of trans-synaptic labeling of WGA was variable among animals and it was difficult to quantify the number of connections among the groups, the results indicate that uninjured CST axons reorganize to form an intraspinal circuit in exercised WT and +/me mice.
Combination of Voluntary Exercise and Reduction in SHP-1 Enhances Recovery of Motor Function
Finally, we assessed whether functional recovery is enhanced by the combination of voluntary exercise and genetic reduction of SHP-1 (Figure 5). We used the grid-walk test to analyze functional recovery of impaired right forelimb. On day 7 after injury, mice in all groups showed marked deficits in their ability to grasp the wire and accurately place the impaired forelimb. Thereafter, the exercised WT and nonexercised +/me showed some improvements in the scores (Figure 5B). The exercised +/me mice exhibited further improvement compared with other groups (Figure 5B). We also used the rotarod test to assess motor functions. Although voluntary exercise increased the baseline pre-injury performance, the exercised +/me mice increased total time on the rotating rod after the injury, compared with the others (Figure 5C). Taken together, these results indicate that SHP-1 reduction and voluntary exercise have synergistic effects on functional recovery after brain injury.
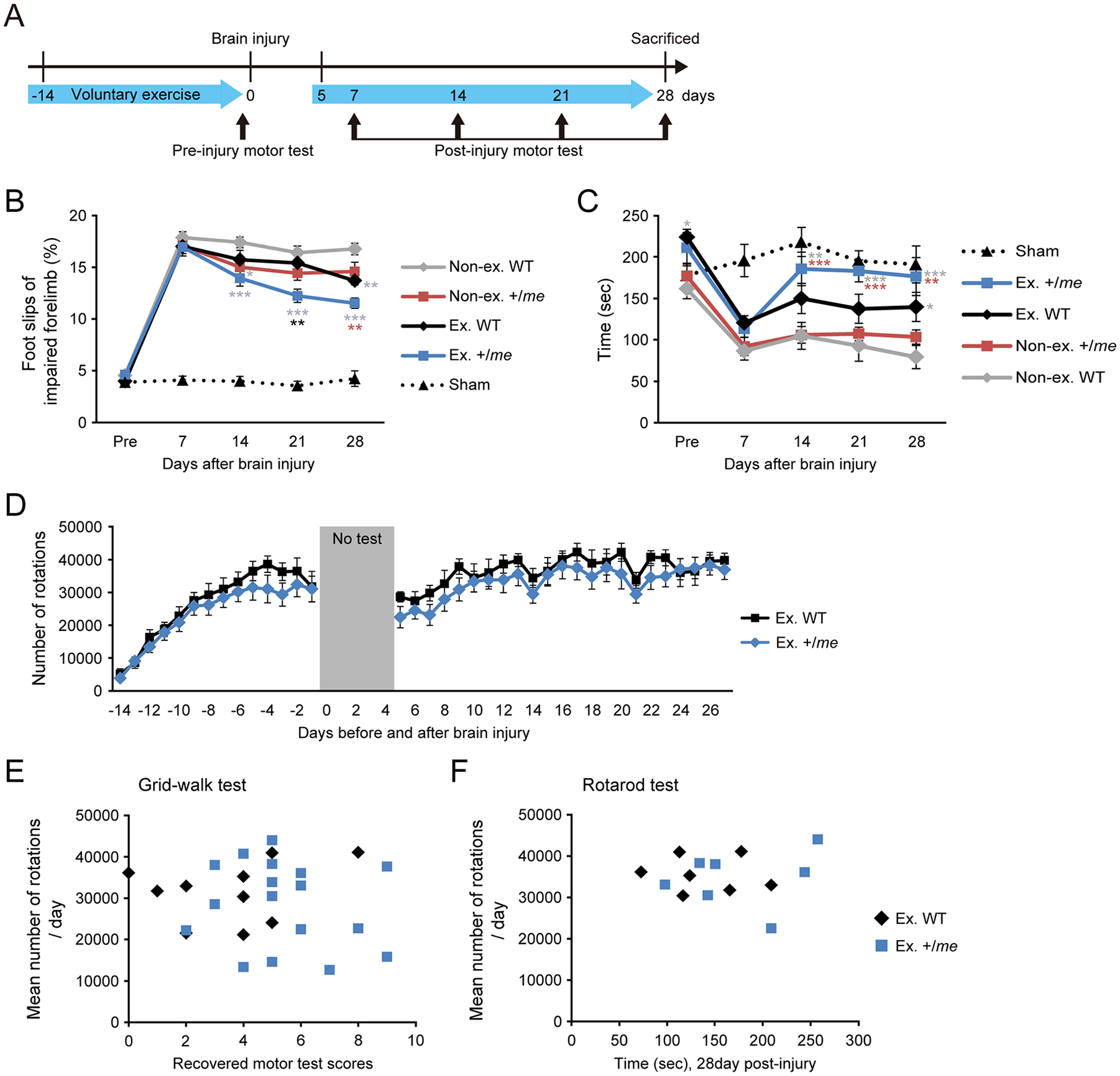
SHP-1 genetic reduction and voluntary exercise enhance motor recovery following brain injury. (A) Motor tests were conducted in wild-type (WT) and +/me mice with/without voluntary exercise once per week postinjury. (B) Scores from the grid-walk test pre- and postinjury (Sham, n = 4-10; nonexercised WT, n = 16-20; nonexercised +/me, n = 12; exercised WT, n = 10-11; exercised +/me, n = 16-17). (C) Scores from the rotarod test pre- and postinjury (Sham, n = 4; nonexercised WT, n = 7; nonexercised +/me, n = 12-13; exercised WT, n = 8; exercised +/me, n = 7-8). Data are presented as means ± SEM. *P < .05, **P < .01, ***P < .001 (2-way repeated-measures analysis of variance followed by Bonferroni test; the color of asterisk indicates the compared group in statistical analyses). (D) The number of rotations of the running wheel per day over 6 weeks in WT and +/me mice (WT, n = 10-11; +/me, n = 17). (E) The correlation between the recovered motor scores (score on day 28 subtracted from that on day 7) of the grid-walk test and mean amount of voluntary exercise per day before and after brain injury (exercised WT, n = 10; exercised +/me, n = 17). Exercised WT: r = 0.250, P = .486; exercised +/me: r = −0.164, P = .529 (Pearson correlation coefficient). (F) The correlation between the total time of the rotarod test at day 28 postinjury and mean amount of voluntary exercise per day (exercised WT, n = 7; exercised +/me, n = 7). Exercised WT: r = −0.109, P = .816; exercised +/me: r = 0.195, P = .675 (Pearson correlation coefficient).
The amount of voluntary exercise in mice used in the motor tests was assessed by measuring the number of running wheel rotations. The numbers were not significantly different between WT and +/me mice either before or after brain injury (Figure 5D). The daily rhythms of running activity were also examined in both groups. They were not significantly different between WT and +/me mice, in which the number peaked at 8:00
We finally evaluated the correlation between the amount of activity and motor recovery in individual mice. There were no correlations between the mean amount of voluntary exercise per day and the recovered scores in the grid-walk test (score on day 28 subtracted from that on day 7), and rotarod test (Figure 5E and F).
Discussion
Our results demonstrated that genetic reduction in SHP-1 and voluntary running exercise promote sprouting of CST axons and motor recovery after brain injury. We thus propose that the combination of molecular targeting and rehabilitative exercise is an effective therapeutic approach to improve motor functions following CNS injuries.
Compensational rewiring of axons is an important plastic change to restore circuits and functions following injuries.1-5 However, the functional restoration is often restricted by associative reactions, abnormal synergic muscle movements, and hypertonus such as spasticity. These abnormal motor outputs are caused by aberrant and maladaptive regrowth of axons.11,41 Indeed, some studies that used genetic approaches to promote axonal elongation showed that motor functions were not recovered but instead, they were exacerbated.11,42,43 Thus, to develop effective therapeutic approaches, it is critical to know how to connect growing axons to appropriate target neurons.
Behavioral training that evokes synchronized activity in neurons helps form adequate connections. One study reported that task-oriented active exercise enhances reorganization of neural circuits and promotes motor function recovery after CNS injury. 44 In contrast, biased exercise training restored a specific motor function while leading to abnormalities in other motor functions. 45 Balanced and active training may thus be important in the recovery of multiple motor functions. Consistent with this theory, a bilateral, voluntary running promoted CST axon sprouting and functional recovery in the present study. Although the exercise was started before the injury to acclimate the mice to the wheels, which altered some baseline parameters such as TrkB expression and preinjury rotarod performance, postinjury exercise changed SHP-1 expression, which would contribute to the sprouting and recovery.
In adult mice, motor CST axons mainly innervate the intermediate and ventral gray matter of the cervical cord (Rexed laminae V, VI, VII, and VIII). 46 Following brain injury, sprouting fibers of spared CST connected to spinal interneurons in laminae VII and VIII of the denervated side; these neural connections played a pivotal role in motor recovery after brain injury. 2 Importantly, our present study demonstrated that rehabilitative training (with SHP-1 reduction) promoted CST sprouting and that the axons formed anatomical connections with neurons in the same area of the spinal cord. Hence, our rehabilitative approach would likely promote proper neuronal connections and activities required for functional recovery. The results also showed some differences in the amount of sprouting fibers among the cervical levels. For example, the innervation was increased at C4 level in response to exercise and SHP-1 reduction. Some target neural components, for example, propriospinal neurons, 2 may be involved at this level.
Surprisingly, in our analyses, there were no correlations between the motor scores and the amount of voluntary exercise. Presumably, even the minimal amount of exercise undergone by the tested mice was sufficient to promote motor recovery. Our results also suggested that the large amount of exercise in some mice did not hinder motor recovery. With regard to the qualitative aspects of training, the forced running with low amount exercise (rotarod) and voluntary running with high amount exercise (running wheel) led to different SHP-1 responses in the spared cortical regions. If decreased SHP-1 responses indicate better regrowth of axons and dendrites, voluntary exercise may lead to better functional recovery. It should be noted that the rotarod might be insufficient to induce SHP-1 expressional changes due to the lower amount of running. Although the total distance of running in the forced and voluntary exercise conditions were not normalized in this study, it would be interesting to examine the differences in neuronal and behavioral output between the types of exercise, as well as to ascertain the underlying mechanisms of differences, which may be related to activity patterns of neurons and/or motivation, depression, stress, and so on.47,48
Our previous report demonstrated that SHP-1 expression is predominantly localized in neurons in the adult mouse brain. 6 SHP-1 is recruited to PirB on binding to myelin-associated glycoprotein (MAG), an inhibitor of axonal outgrowth, and is required for MAG-induced neurite growth inhibition. 26 SHP-1 also inactivates TrkB though dephosphorylation. 26 Since BDNF/TrkB signaling plays a pivotal role in axonal outgrowth 7 and CST rewiring, 2 suppression of SHP-1 would facilitate axon remodeling by enhancing BDNF/TrkB signaling concomitantly with suppressing myelin-related inhibitory signals. Accordingly, we previously demonstrated that suppression of SHP-1 promotes axonal sprouting following brain injury. 6 In the present study, we observed that voluntary exercise attenuated the increase of SHP-1 expression and suppressed the decrease of BDNF and TrkB expression in the contralesional cortex, resulting in increased sprouting of CST axons. The results suggest that voluntary exercise has cumulative effects that suppress inhibitory signals and enhance axon growth signaling in the spared brain regions. Although the mechanism by which exercise decreases SHP-1 expression remains unclear, the neuronal activity induced by rehabilitative training may control SHP-1 expression. It requires additional experiments to examine the mechanistic changes in BDNF/TrkB and the downstream signaling including SHP-1 at the protein levels in response to exercise and SHP-1 reduction. Understanding the signaling pathway of SHP-1 may lead to develop better approaches to enhance plastic changes.
Conclusions
The present study demonstrated that treatment with a combination of SHP-1 reduction and voluntary running exercise following brain injury, enhances CST axon sprouting in the cervical cord and promotes functional recovery more than exercise alone. Although voluntary exercise induced motor recovery, there was no correlation between the amount of exercise and the degree of recovery. The neuronal and physiological mechanisms which underlie the decrease in SHP-1 is unclear. Hence, to develop therapeutic strategies for CNS injuries, further multidirectional analyses should be conducted to ascertain the underlying neuronal and molecular mechanisms that facilitate circuit reorganization and to refine the combinational approaches with training.
Supplemental Material
NNR_34_6 – Supplemental material for Combinational Approach of Genetic SHP-1 Suppression and Voluntary Exercise Promotes Corticospinal Tract Sprouting and Motor Recovery Following Brain Injury
Supplemental material, NNR_34_6 for Combinational Approach of Genetic SHP-1 Suppression and Voluntary Exercise Promotes Corticospinal Tract Sprouting and Motor Recovery Following Brain Injury by Takashi Tanaka, Tetsufumi Ito, Megumi Sumizono, Munenori Ono, Nobuo Kato, Satoru Honma and Masaki Ueno in Neurorehabilitation and Neural Repair
Supplemental Material
Rev._NNR_Supplemental_material – Supplemental material for Combinational Approach of Genetic SHP-1 Suppression and Voluntary Exercise Promotes Corticospinal Tract Sprouting and Motor Recovery Following Brain Injury
Supplemental material, Rev._NNR_Supplemental_material for Combinational Approach of Genetic SHP-1 Suppression and Voluntary Exercise Promotes Corticospinal Tract Sprouting and Motor Recovery Following Brain Injury by Takashi Tanaka, Tetsufumi Ito, Megumi Sumizono, Munenori Ono, Nobuo Kato, Satoru Honma and Masaki Ueno in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We are grateful to Mr S. Muramoto (Kanazawa Medical University) for providing a conducive environment and support experiments. We would also like to thank Dr S. Ohtsuka and Y. Kaneko (Research Support Center, Kanazawa Medical University) for technical assistance with genetic SHP-1 suppression mice.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by the Japan Society for the Promotion of Science, a Grant-in-Aid for Scientific Research (KAKENHI: grant 15K16377 and 17K13079, 15K06790), and the Kindai University Research Grant (T.T.); AMED-CREST (JP19gm1210005) and KAKENHI 17H04985 (M.U.).
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
