Abstract
Background. In children with unilateral cerebral palsy (UCP), the fibers of the corticospinal tract (CST) emerging from the lesioned hemisphere are damaged following the initial brain injury. The extent to which the integrity of these fibers is restorable with training is unknown. Objective. To assess changes in CST integrity in children with UCP following Hand-and-Arm-Bimanual-Intensive-Therapy-Including-Lower-Extremity (HABIT-ILE) compared to a control group. Methods. Forty-four children with UCP participated in this study. Integrity of the CSTs was measured using diffusion tensor imaging before and after 2 weeks of HABIT-ILE (treatment group, n = 23) or 2 weeks apart without intensive treatment (control group, n = 18). Fractional anisotropy (FA) and mean diffusivity (MD) were the endpoints for assessing the integrity of CST. Results. As highlighted in our whole tract analysis, the FA of the CST originating from the nonlesioned and lesioned hemispheres increased significantly after therapy in the treatment group compared to the control group (group * test session interaction: P < .001 and P = .049, respectively). A decrease in MD was also observed in the CST emerging from the nonlesioned and lesioned hemispheres (group * time interaction: both P < .001). In addition, changes in manual ability correlated with changes in FA in both CSTs (r = 0.463, P = .024; r = 0.643, P < .001) and changes in MD in CST emerging from nonlesioned hemisphere (r = −0.662, P < .001). Conclusions. HABIT-ILE improves FA/MD in the CST and hand function of children with UCP, suggesting that CST fibers retain a capacity for functional restoration. This finding supports the application of intensive motor skill training in clinical practice for the benefit of numerous patients.
Keywords
Introduction
Over the past 2 decades, physical neurorehabilitation efforts have been extensively devoted to intensive motor skill training, aiming to improve functional abilities of children with cerebral palsy (CP). 1 Plastic changes within the gray matter–induced processes have been widely demonstrated in humans engaged in motor skill learning. 2 In children with unilateral CP (UCP), the impact of motor skill training has been demonstrated using transcranial magnetic stimulation (TMS)-based cortical mapping 3 and functional magnetic resonance imaging (fMRI).4-6 Furthermore, plastic changes of white matter tracts, notably entailing reorganization of the corticospinal tract (CST) projections to the spinal cord, are elicited in animal models of CP by motor skill training, albeit during a brief window of opportunity during development. 7 Though this window of opportunity is not precisely defined in humans, reorganization of the CST projections to the spinal cord is thought to occur during early infancy, 8 concomitant with potential reorganization of the structure 9 and myelination of CST fibers. 10 Despite the evidence of plasticity of CST projections suggested from animal studies,8,11 the possibility of improving structural connectivity of the CST through skill training has received scant attention in the management of children with CP.
Following the initial brain damage in children with UCP, 2 main features have been observed in the CSTs. First, depending on the timing, extent, and location of the lesion, the organization of the CST can be modified. The affected limb can therefore be controlled as in typically developing individuals by the contralateral hemicortex, but can also undergo a reorganization, whereby the affected limb may be controlled by the ipsilateral hemicortex (nonlesioned) or bilaterally. 12 Such changes in CST organization can be determined either through transcranial magnetic stimulation12,13 or by diffusion tensor imaging (DTI). 14 DTI has also emerged over the last decade as a key tool to explore white matter integrity.15-17
Second, the CST emerging from the lesioned hemicortex regardless of the CST organization presents a loss in white matter integrity as evidenced by DTI as a decrease in fractional anisotropy (FA) and an increase in mean diffusivity (MD) in its craniocaudally oriented fibers. 18 FA is a measure providing an insight of the directionality of water diffusion along white matter tracts, which is higher in organized and myelinated tracts. MD measures the rotationally invariant magnitude of water diffusion in brain tissue, used to measure structural brain integrity. The decrease of CST integrity is positively correlated with the loss of motor skill in the affected extremity,19-21 suggesting that CST damage might be a limiting factor for motor improvement after injury. Furthermore, while the CST in typically developing children continues to develop (ie, an increase in FA and decrease in MD) during childhood 22 —steep increase until 7 and then gradual slow increase until adulthood—it was recently observed that this maturation process was absent in the CST emerging from both the more affected and the less affected hemispheres of children with UCP. 18
Although motor skill training in healthy individuals induces gray matter changes,23,24 with potential impact on strength in intracortical connections 25 and cortical white matter architecture, 26 motor skill training has not been tested with regard to loss of integrity in the injured CST. Recent small sample pilot studies in children with UCP indicated possible modifications in the anisotropy/number of voxels of CST after training.5,6,27,28 This observation concurs with modified white matter anisotropy after motor skill training in mice,29,30 suggesting that CST structure might be restored by intensive motor skill training. Potentially, such changes could be associated with increased myelination produced by newly generated oligodendrocytes. 31
Based on this background, we aimed to assess the effects of intensive motor skill training on CST integrity in children with UCP. We hypothesized that motor skill training would increase FA and decrease MD in the CST of children with UCP.
Material and Methods
Participants
Forty-four children participated in this study at the Université catholique de Louvain, Brussels, Belgium, over the summer camp program from 2013 to 2019. Children were recruited from a center dedicated to the treatment of CP at the University Hospitals. All parents and children provided their written informed consent. The review board of the Université catholique de Louvain approved this study.
Inclusion criteria included the following: (1) children diagnosed with UCP, (2) aged 6 to 16 years, (3) an ability to grasp light objects and lift the paretic arm 15 cm above a table, (4) school level equal to that of typically developing peers (all in regular or mainstreamed education appropriate for age), and (5) ability to follow instructions and complete testing. Exclusion criteria were the following: (1) uncontrolled seizures, (2) botulinum toxin injections or orthopedic surgery in the previous/following 6 months, (3) visual problems likely to interfere with treatment/testing, and (4) all MRI contraindications.
The behavioral data of some children included in this article have been published elsewhere.32-34
Experimental Design
All children underwent 2 DTI scans 2 weeks apart, either before/after a HABIT-ILE (Hand-and-Arm-Bimanual-Intensive-Therapy-Including-Lower-Extremity) intervention (treatment group, n = 23) or without intensive treatment in between (control group, n = 18). The demographics of the children are presented in Table 1. Examiners blinded to the study purpose performed the MRI acquisitions, and the analyses were mainly automated. Part of analysis requiring human input was blindly performed (using code numbers).
Characteristics of the Children.
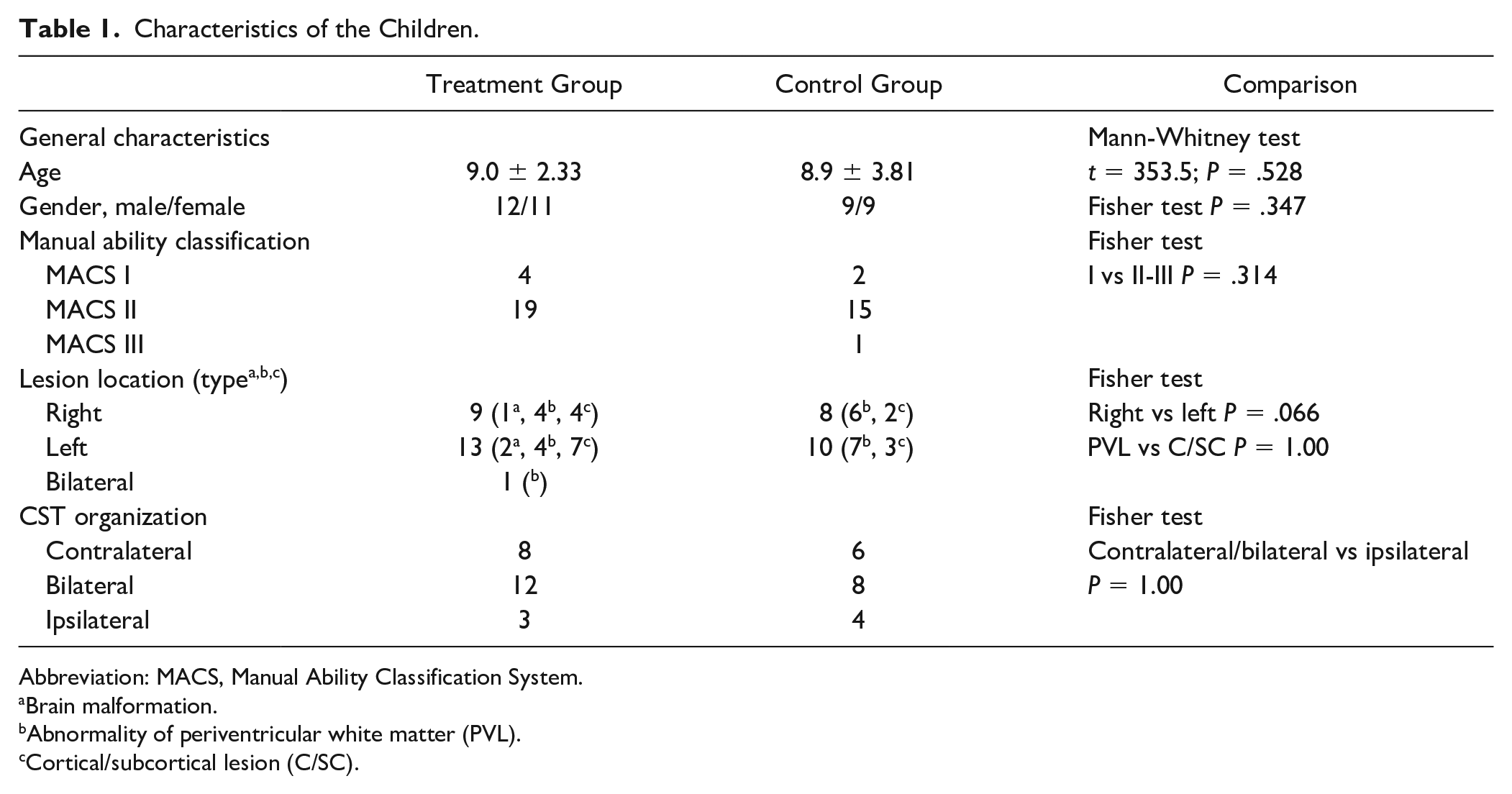
Abbreviation: MACS, Manual Ability Classification System.
Brain malformation.
Abnormality of periventricular white matter (PVL).
Cortical/subcortical lesion (C/SC).
We calculated the sample size based on the Box and Block changes observed in a HABIT-ILE intervention in children with UCP, 35 wherein there was a mean increase of 3 blocks in the more-affected hand after intensive intervention. This result indicated the requirement of at least 12 participants per group for a significance level of .05, 1 − β = 0.90, µ1 − µ2 = 3, and σ = 2.8. Considering a risk of losing 30% of the data due to MRI quality (eg, due to excessive movement), we recruited 18 participants per group.
Intervention
Children in the treatment group received 10 days of HABIT-ILE, representing in total 90 hours of treatment by motor skill learning 35 involving concomitant training of the upper extremities (UE) and the lower extremities (LE). We have described the HABIT-ILE intervention in detail in a methodological paper. 36 In brief, the HABIT-ILE consists of structured bimanual tasks (games and functional bimanual activities) in which difficulty of the motor task progressively increases. Concomitantly, postural/LE requirements also progressively increase in complexity as children successfully master the skills. More details on HABIT-ILE are available in the supplementary material.
Imaging
In each scanning session, the children underwent an MRI performed at 3 T with a 32-channel phased array head coil to record 3D heavily T1-weighted and DTI images using a Philips Achieva scanner (Philips Healthcare, Eindhoven, The Netherlands; sequence details in supplementary material).
The data were analyzed with BrainVoyager QX (Version 2.6.1, Brain Innovation, The Netherlands). To create MD and FA maps, as well quantify the number of voxels, we co-registered the DTI data with the 3D anatomy of the subject without normalization (native space). Two different analyses were subsequently performed to investigate changes in CST fibers: a focal analysis and a whole tract analysis.
Focal Analysis
In order to determine FA, MD, and the number of voxels of the CST fibers, we first created axial planes for each DTI sequence in each child, based on anatomical landmarks. The plane was centered on the CST fibers at the level of the mid pons, as visualized in a transverse plane passing through the emergence of the middle cerebellar peduncle. At this level, fibers of the CST were isolated from the cortico-ponto-cerebellar fibers. The plane extended between the anterior limit of the cerebral peduncle and the posterior limit of the CST, thereby avoiding the posterior cranio-caudal fibers of the cerebral peduncle, corresponding to the medial lemniscus. The lateral edges of the cerebral peduncle define the lateral limits. In the z-direction, the plane had a depth of 5 voxels width, of which 2 were in the cranial direction and 2 in the caudal direction from the transversal plane, passing through the middle cerebellar peduncle. We then selected voxels with a main cranio-caudal direction, reporting their number as well as their FA and MD value.
Whole Tract Analysis
This analysis was performed using as region of interest (ROI), a sphere (diameter: 6 mm) in the anterior part of the mid pons at the level of the middle cerebellar peduncle. Only fibers with a FA > 0.20 and a deviation angle <50° were included in the tracking of fibers. Voxels of the CST, mainly in the cranio-caudal direction, were subsequently selected to determine the FA and MD of each participant on the whole tract. The values provided for each participant is the mean value of FA and MD in cranio-caudal direction.
Determination of CST Patterns
As determination of CST patterns with DTI was previously shown to be consistent with that determined by TMS mapping, 14 the classification of CST patterns was performed using DTI. We made a deterministic tracking from voxels detected in the defined plane, aiming to reconstruct, track, and determine the CST organization. As for whole tract analysis, solely fibers with FA > 0.20 and a deviation angle <50° were considered for this tracking. Participants were classified as having 1 of 3 CST patterns, as follows: (1) ipsilateral, when 100% of fibers tracked to the cortex from both sides projected to the nonlesioned hemisphere; (2) bilateral, when fibers tracked from the affected CST projected toward both the lesioned and the nonlesioned hemisphere; or (3) contralateral, when >90% of fibers tracked from the affected CST ascended to the lesioned hemisphere. This cutoff was set since individuals without neurological lesions usually maintain a small number (≈10%) of fibers not crossing.37,38 Tracts were considered in the analysis to be CST of the lesioned or nonlesioned hemisphere, regardless of the side of the lesion.
Assessment of Functional Changes
Manual ability level of the children was defined using the Manual Ability Classification System (MACS). 39 The children’s ability ranged from a MACS level I (ie, handling objects easily and successfully) to MACS level III (ie, needing help to prepare or modify activities due to difficulties in manipulation).
Changes in unilateral dexterity were measured using the Box and Block test. 40 Manual ability was measured using the ABILHAND-Kids questionnaire.34,41 Performance and satisfaction in goals management were assessed through the Canadian Occupational Performance Measure (COPM). 42 Changes in walking abilities were tested using the 6-Minute Walk Test 43 and the ABILOCO-Kids. 44 These tests are widely used and present good psychometric properties to assess children with cerebral palsy, although the minimally clinical important change is not available for many of them.45,46
Statistics
We tested normality of the data using the Shapiro-Wilk test and homoscedasticity with the Fisher test. We performed a 2 (group) * 2 (test session) analysis of variance (ANOVA) for each variable with repeated measures on test session. The overall interaction (group * test session) allowed testing whether the second assessment differed between groups. We performed Newman-Keuls post hoc tests after an adjustment for multiple comparisons. We used a Pearson correlation (or Spearman in nonparametric conditions) to investigate the relationship between changes in FA and MD of the CST and functional changes, as well as between changes in FA/MD and age. Where multiple correlations were performed, a Bonferroni correction was applied on the P value threshold to avoid Type I error (adjusted P value for correlation between MRI changes and functional changes, P = .007; adjusted P value for correlation between MRI changes and age, P = .005). For functional tests, the number of children reaching a clinically meaningful degree of difference, calculated with the empirical rule of effect size (ERES, clinically meaningful if change >0.5 SD), is provided.
Results
Participants Flow and Characteristics
Participant flow is illustrated in Figure 1. At baseline, the characteristics (age, gender, lesion side, and time of injury) did not differ between the treatment group (n = 23) and the control group (n = 18; Table 1). However, 3 neuroimaging measures out of 14, as well as 3 functional assessments, differed between groups (see Tables 2 and 3, group comparison). Each child’s lesion was classified separately by a neuropediatrician and a neuroradiologist, based on Krägeloh-Mann and Horber’s classification. 47 In both groups, most children presented with abnormalities of periventricular white matter or cortical/subcortical lesions, while 4 children presented brain malformations. Three children in the treatment group and 3 children in the control group were excluded from neuroimaging analysis due to low image quality in 1 of the 2 MR sequences, and 3 children were not included in the analysis of Box and Block test due to lack of participation in 1 of the 2 testing sessions.
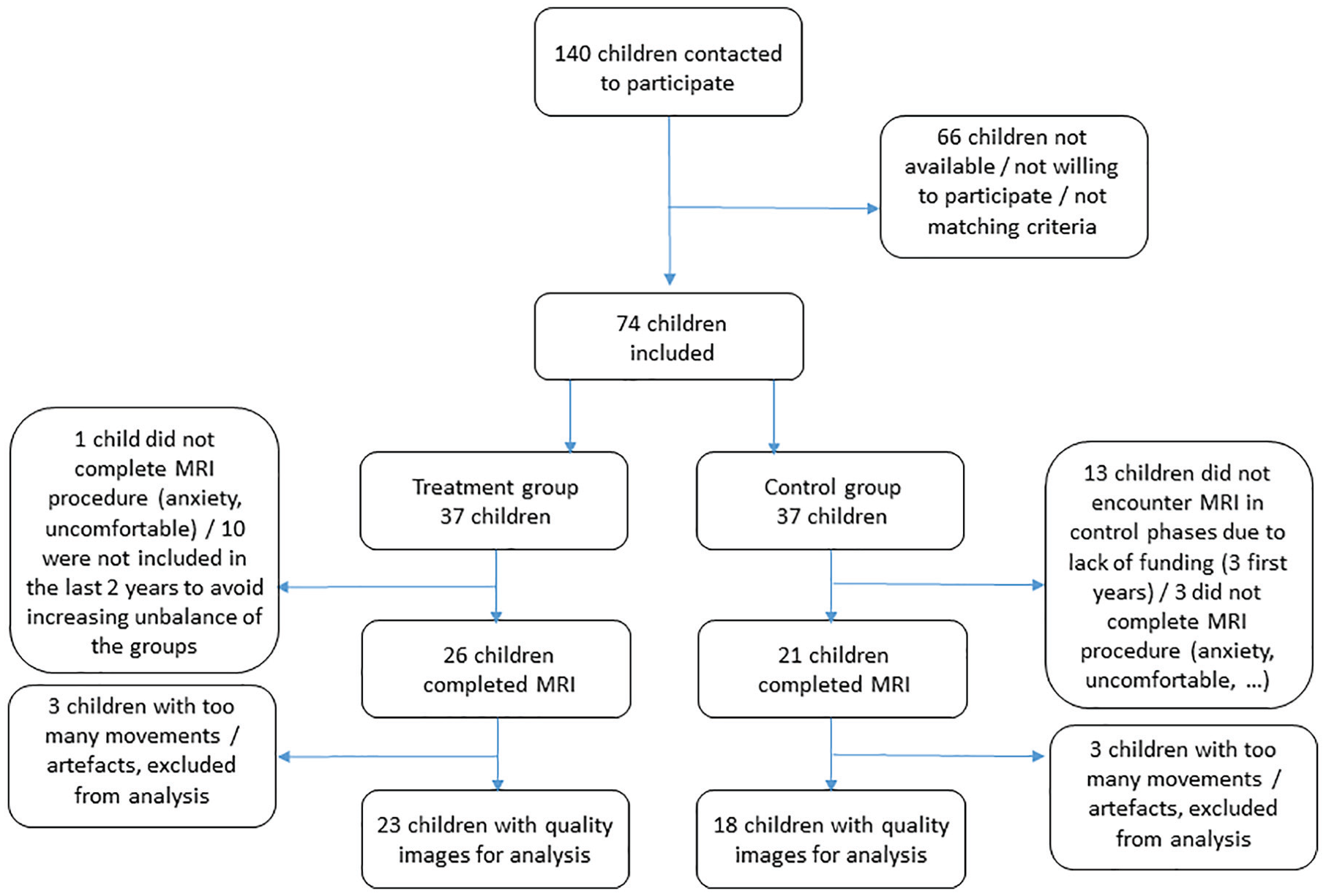
CONSORT flow diagram illustrating children flow (inclusion, exclusion, and withdrawal) during the study. Databases of Center dedicated to the treatment of CP at the University Hospitals were searched by the responsible MDs to identify children with UCP meeting the criteria. Of the 140 children initially identified who met criteria, 74 agreed to participate. They were each year randomized into a control or a treatment group. Over the 3 first years, children encountered MRI solely during treatment phases, due to lack of funding. This was partly compensated by excluding children of the treatment group over the last 2 years of data collection. In addition, 1 child in the treatment group and 3 in the control group did not tolerate the MRI procedure, and 3 children in each group were lost at analysis due to bad image quality. Finally, valid MRI data were obtained for 23 children in the treatment group and 18 children in the control group (demographic details in Table 1). Abbreviations: CONSORT, Consolidated Standards of Reporting Trials; UCP, unilateral cerebral palsy; HABIT-ILE, Hand and Arm Bimanual Intensive Therapy Including Lower Extremity, MD, medical doctor, MRI, magnetic resonance imaging.
Changes in Anisotropy, Mean Diffusivity, Voxels, and Functional Changes.
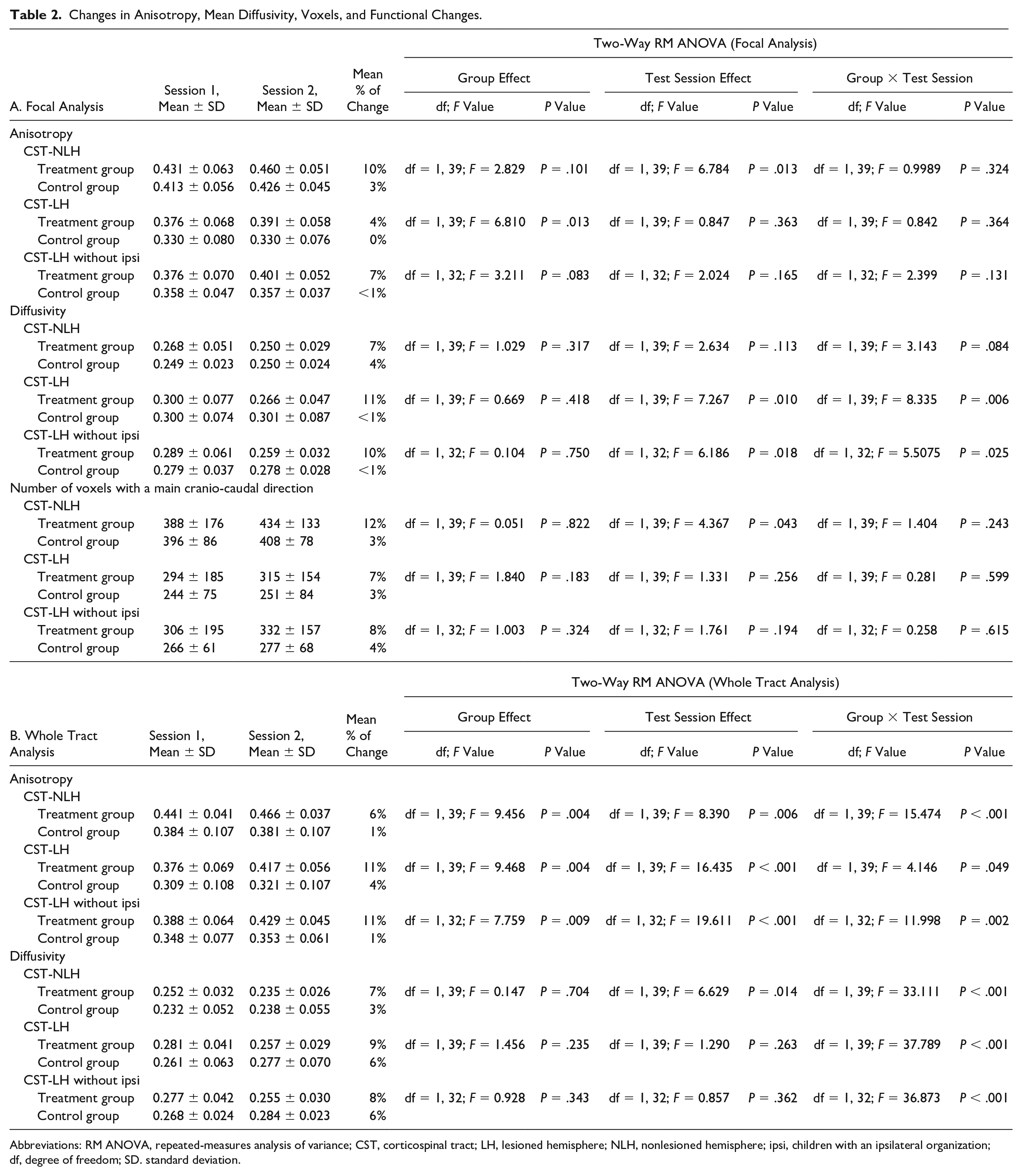
Abbreviations: RM ANOVA, repeated-measures analysis of variance; CST, corticospinal tract; LH, lesioned hemisphere; NLH, nonlesioned hemisphere; ipsi, children with an ipsilateral organization; df, degree of freedom; SD. standard deviation.
Changes in Functional Assessments.
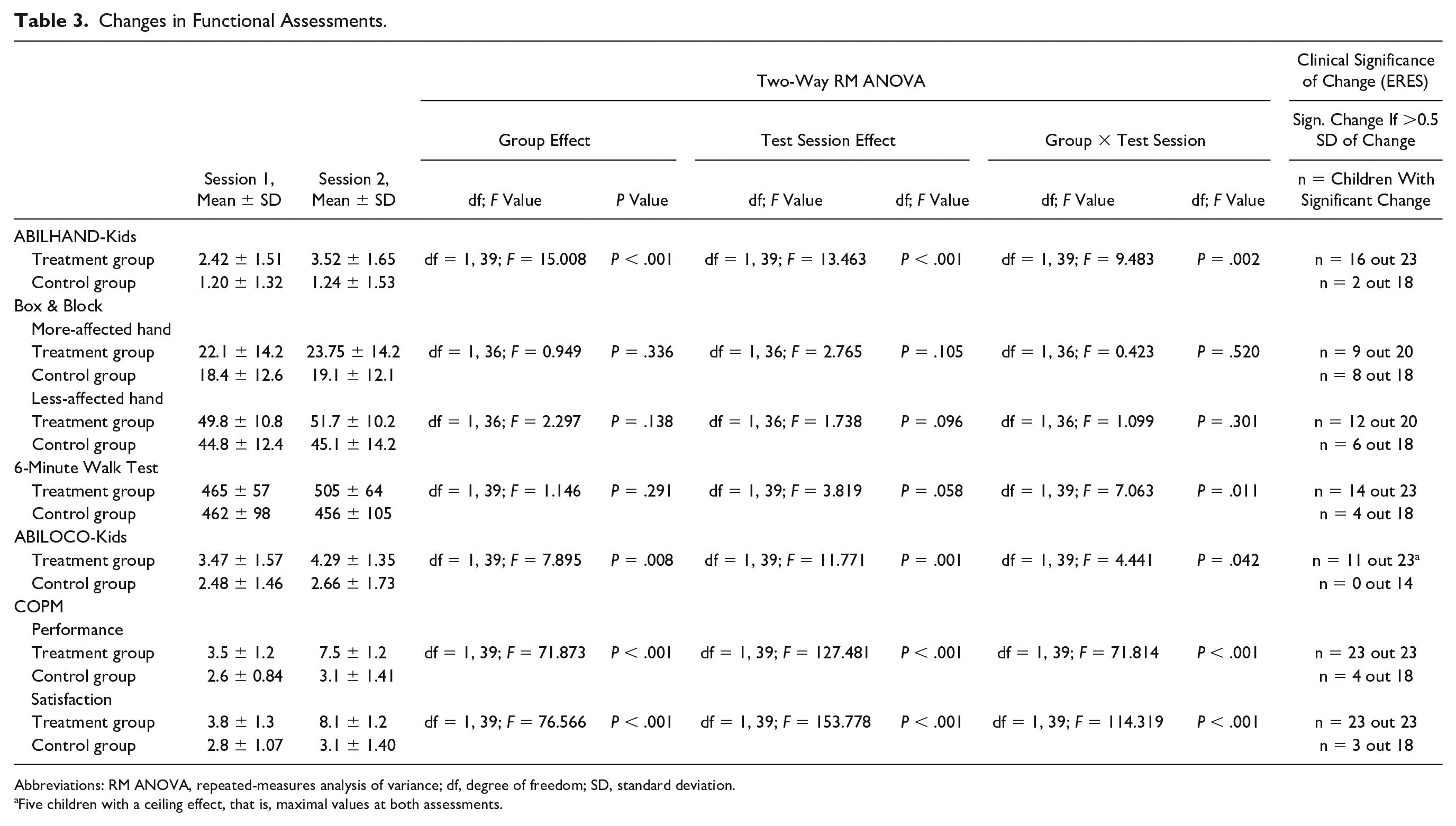
Abbreviations: RM ANOVA, repeated-measures analysis of variance; df, degree of freedom; SD, standard deviation.
Five children with a ceiling effect, that is, maximal values at both assessments.
Neuroplastic and Functional Changes
The results of this section are reported in Table 2 and illustrated in Figure 2.
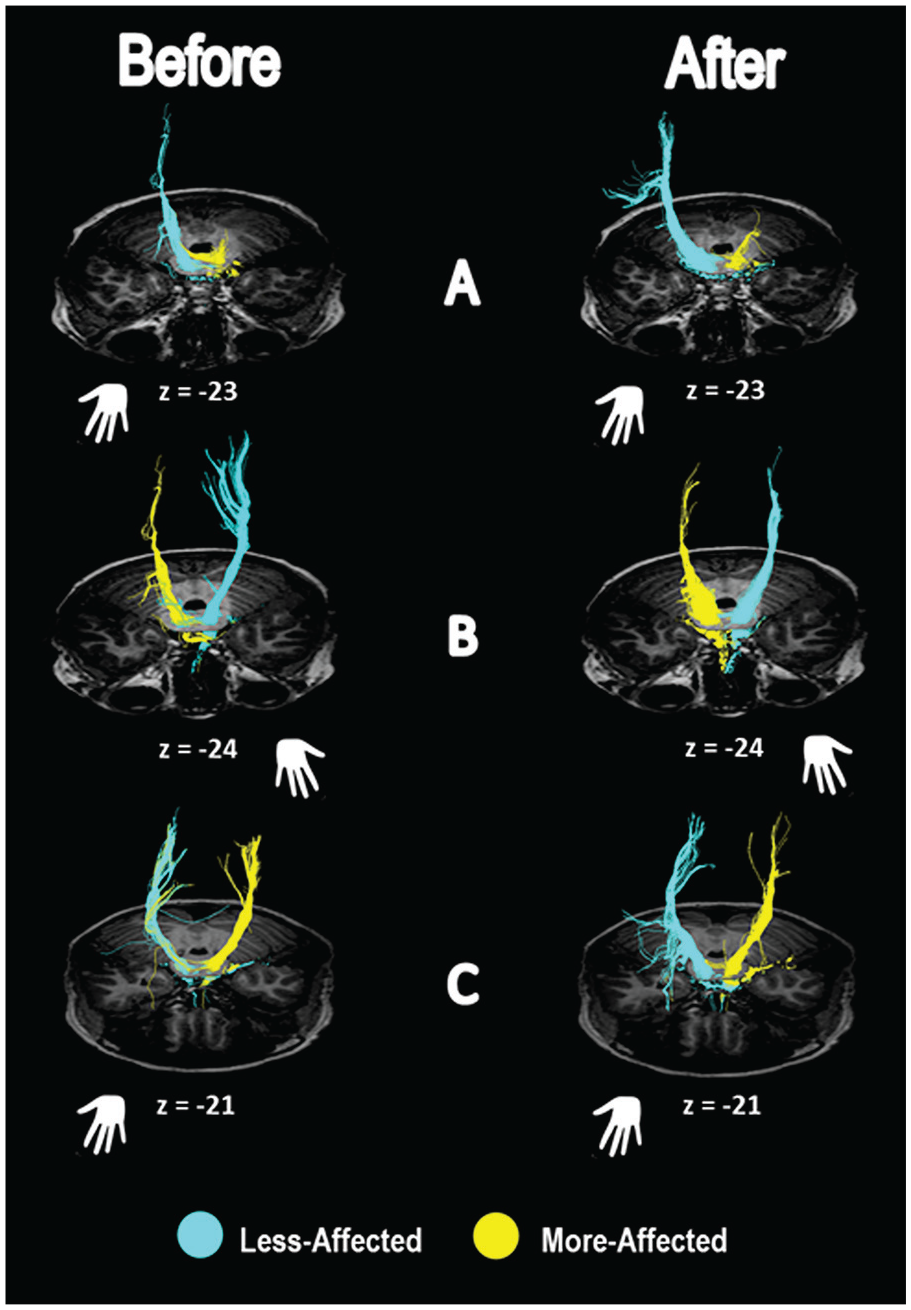
Examples of CST organization before and after intensive intervention in representative children with (A) ipsilateral organization, (B) contralateral organization, and (C) bilateral organization. Z coordinates in each child’s native space (151, 152, and 149) corresponded, respectively, to z = −23, z = −24, and z = −21 in Talairach’s coordinates. Blue color represents the CST emerging from the nonlesioned hemicortex; yellow color represents the CST emerging from the lesioned hemicortex. Abbreviation: CST, corticospinal tract.
Focal Analysis (Table 2A)
For the focal analysis, a main effect of test session (P = .010) and a group × test session interaction (P = .006) was observed only for the mean diffusivity in the CST emerging from the lesioned hemisphere (CST-LH). Post hoc tests showed that MD values were significantly lower in test session 2 compared to test session 1 for the treatment group only. A significant test session effect (P = .013) in FA was also observed in the CST emerging from the lesioned hemi-cortex (CST-LH). However, no group × test session interaction was observed (P = .324). There was a significant test session effect for the number of voxels detected with a main cranio-caudal direction in the CST-NLH (P = .043), although there was no group × test session interaction (P = .243).
Whole Tract Analysis (Table 2B, Figure 3)
A main effect of test session (P = .014) and a group × test session interaction (P < .001) was observed for the mean diffusivity in the CST emerging from the nonlesioned hemisphere (CST-NLH). Post hoc tests showed that for the treatment group only, MD values were significantly lower in test session 2 compared to test session 1. A group × test session interaction was also observed for the CST-LH (P < .001). Post hoc tests showed that MD values were significantly lower in test session 2 compared to test session 1 only for the treatment group.
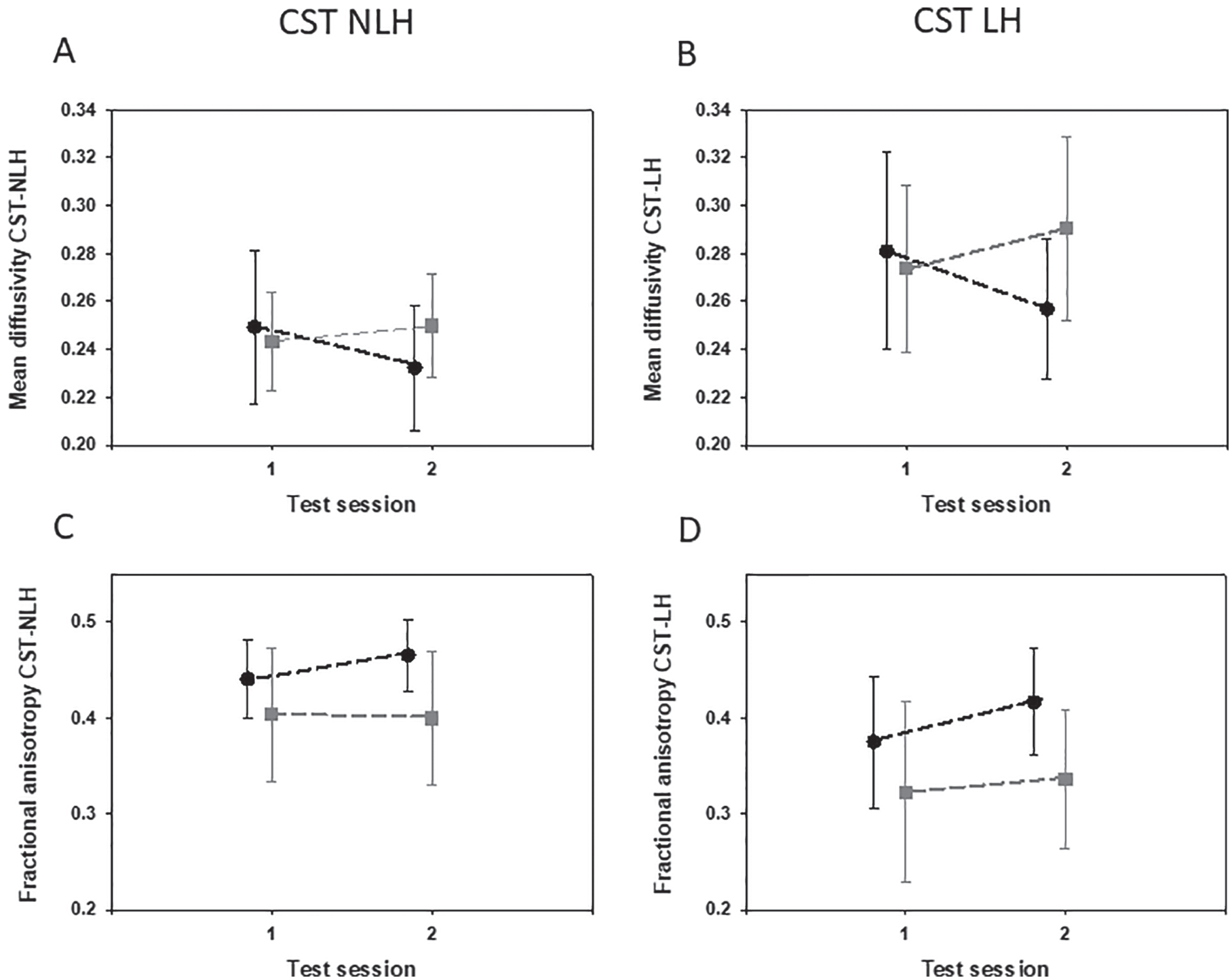
Mean diffusivity (MD, panels A and B) and Fractional anisotropy (FA, panels C and D) measured in the CST-NLH (left) and the CST-LH (right). Black dots represent results for the treatment group before and after intensive motor skill training. Gray squares represent the control group at first and second assessments (2 weeks in between). Error bars represent standard deviations. Abbreviations: CST, corticospinal tract, NLH, nonlesioned hemisphere, LH, lesioned hemisphere.
For FA, a main effect of test session (P = .006) and a group × test session interaction (P < .001) was observed in the CST-NLH. Post hoc tests showed that for the treatment group only, FA values were significantly higher in test session 2 compared to test session 1. A main effect of test session was also observed for FA in the CST-LH (P < .001) with a significant group × test session interaction (P = .049). This comparison was significant when the children with ipsilateral organization were excluded (test session effect P < .001; interaction P = .002). Post hoc tests showed that FA values were significantly higher in test session 2 compared to test session 1 only for the treatment group.
Functional Changes (Table 3)
A main effect of test session (P < .001) and a group × test session interaction (P = .002) was observed for the ABILHAND-Kids score. Post hoc tests showed that for the treatment group only, manual ability values were significantly higher in test session 2 compared to test session 1. For ABILOCO-Kids, a main effect of test session (P = .001) and a group × test session interaction (P = .042) was observed. Post hoc tests showed that for the treatment group only, locomotor ability values were significantly higher in test session 2 compared to test session 1. A group × test session interaction was also observed for the 6MWT (P = .011). Post hoc tests showed that walking distances were significantly higher in test session 2 compared to test session 1 for the treatment group but not the control group. Finally for the COPM, a main effect of test session was observed both for performance and satisfaction (both P < .001) as well as group × test session interactions (both P < .001). Post hoc tests showed that for the treatment group only, both performance and satisfaction scores were significantly higher in test session 2 compared to test session 1. No significant effects were highlighted on the Box and Blocks measure on either hand (all P > .096).
Correlations
Due to multiple correlations between each neuroimaging measure and the different functional tests, the P value significance threshold was set at P = .007. In the treatment group, the changes in the ABILHAND-Kids score correlated with the changes measured with our focal analysis in FA of CST-LH (r = 0.643, P < .001, n = 23) and presented a trend to correlate with CST-NLH (r = 0.468, P = .024, n = 23). Similarly, the change in MD of CST-NLH correlated with ABILHAND-Kids changes (r = −0.662, P < .001, n = 23). The change in the more affected hand in performance of the Box and Block test tended to correlate with the change of FA in the CST-LH (r = 0.570, P = .008, n = 20) and in the CST-NLH (r = 0.447, P = .032, n = 20). No correlation was observed between CST FA or CST MD changes and changes in the 6MWT, ABILOCO, or COPM (all P > .700).
When correlations between changes in functional assessments and neuroplastic changes were performed with our whole tract analysis, only a trend was observed between changes in the ABILHAND-Kids score and the MD change on the CST-NLH (r = −0.414, P = .049).
Measures obtained from our focal analysis showed in addition that the baseline FA of the CST-NLH correlated inversely with the change in FA of this tract: lower initial values predicted larger changes in FA after training (r = −0.665, P < .001, n = 23). A similar relationship was observed in the CST-LH (r = −0.609, P = .002, n = 23). This correlation was higher when performed exclusively in children with a bilateral or contralateral organization (r = −0.689, P < .001, n = 20). Similar relationships were observed between the baseline MD and the change in MD (r = −0.817, P < .001; r = −0.850, P < .001, for CST-NLH and CST-LH, respectively), as well as between the baseline voxels value and the change observed in voxels (r = −0.660, P < .001; r = −0.551, P = .006, for CST-NLH and CST-LH, respectively). Data from our whole tract analysis showed similar correlations although the strength of the correlations was slightly decreased: baseline FA × changes in FA were r = −0.509, P = .013; r = −0.541, P = .008, for CST-NLH and CST-LH, respectively; baseline MD × changes in MD were r = −0.595, P = .003; r = −0.500, P < .005, for CST-NLH and CST-LH, respectively.
The baseline MD measured with focal analysis in the CST-NLH was predictive of the changes in ABILHAND-Kids questionnaire (r = 0.639, P < .001). A trend between baseline MD and changes in ABILHAND-Kids scores was also observed in the whole tract data: r = 0.506, P = .014, and ρ = 0.456, P = .029, for the CST-NLH and CST-LH, respectively (Bonferroni-corrected threshold P = .007).
The baseline FA measured with focal analysis tended also to predict dexterity changes, with a trend between lower baseline FA in the CST-NLH and changes in the Box and Block test of the more affected hand (r = −0.513, P = .012, n = 23). A similar trend in the CST-LH was observed (r = −0.490, P = .017, n = 23).
The post-training changes in the CST-LH and the CST-NLH (focal analysis) were strongly correlated both for the FA (r = 0.730, P < .001) and for the number of voxels (r = 0.779, P < .001). The correlations in the control group between changes in CST-LH and CST-NLH for the FA (r = 0.009, P = .978) or number of voxels (r = −0.444, P = .065) were not significant.
In the treatment group, no correlations were found between age and neuroplastic changes in CST (corrected P threshold = .005; all P > .049).
Discussion
The aim of this study was to assess CST white matter changes in children with UCP after intensive motor skill training as compared to a nontreatment control group. We hypothesized that intensive motor rehabilitation would increase FA and decrease MD of the CST-LH in the treatment group only. We further hypothesized that changes in FA and MD would correlate with the improvement in hand motor function upon training. This hypothesis was supported by our results, not only in the CST-LH but also in CST-NLH, showing increased FA and decreased MD in CST fibers only after HABIT-ILE training. Furthermore, the changes in FA and MD correlated with improved dexterity in tests of hand motor function.
The increase of FA and decrease in MD in the treatment group after HABIT-ILE is consistent with recent pilot studies in children with CP5,6,27,28 and with results in rodents showing that motor skill learning alters white matter parameters.29,30 The latter rodent studies showed that motor skill training in the adult led to persistent structural changes in task-relevant white matter pathways that seemed attributable to learning-related increases in myelination. 29 Results of animal and cellular studies in vitro suggest that the motor learning–induced changes in FA/MD depend on increased task-related activity within the white matter.48,49 Accordingly, the amount of skill practice during the intensive therapy should correlate with the magnitude of white matter changes. If the changes in FA/MD observed in the CST in this study relates to structural changes in the myelin sheath around the CST fibers, this may increase the speed of saltatory conduction. The attribution of altered myelin levels to the response to HABIT-ILE is further supported by observations in rodents that learning complex motor skills occurs in conjunction with generation of new oligodendrocytes. 30 Transgenic mice unable to generate the transcription factor for myelin production in oligodendrocytes precursors are unable to learn complex motor skills. 30 In addition, Xiao et al 31 demonstrated that altered myelin expression in oligodendrocytes together with neuronal changes increased the efficiency of the pathway related with performance of the learned task.
The possibility to induce changes in white matter fibers of healthy humans has been highlighted in recent studies investigating highly skilled motor learning. For instance, a 6-week training of healthy adults in juggling altered cortico-cortical white matter pathways. 26 In addition, in piano players of all ages, the FA of various cortico-cortical white matter tracts correlates with the amount of music training. 50 However, only piano players who started playing during childhood showed distinct FA levels in the CST that correlated to the amount of training during childhood. Thus, it seems likely that plasticity of CST structure and FA may decline in the transition to adolescence, which would suggest that intensive interventions might be more effective during early childhood, although our data did not support this phenomenon in children with CP.
Present findings support the conjecture that the changes in FA and MD values arises from intensive practice. First, the change in the MD and FA correlated with improvement in hand function (ABILHAND-Kids and Box and Block scores), suggesting the activity-dependent aspect of this change. Second, while the FA/MD of the CST emerging from the lesioned hemisphere increased following intensive intervention in children with contralateral or bilateral CST patterns, this did not seem to appear in children with ipsilateral motor patterns, although their small number in this study does not allow drawing general conclusions. If demonstrated in larger groups of children with ipsilateral organization in the future, this discrepancy might suggest that solely white matter pathways used to mediate information to the effector muscles are likely to undergo changes in MD/FA during intensive motor learning intervention. Interestingly, as previously observed in studies investigating correlations between CST integrity and functional abilities,19,20 solely UE measures—and here UE changes—correlated with changes in DTI measures. This may suggest either that the motor skill learning aspect of the intervention is more developed in our intervention for the UE or that CST integrity and additional myelination is more linked to intensive stimulation of UE, which could suggest to favor in the future the integration of UE training in children with CP, even during LE-focused programs. It might also be that the tests we used—focused on capacity for LE testing—are influenced by different factors (balance, trunk control, etc) introducing a bias in the correlation measure. This should be further investigated with neuroimaging assessments in studies contrasting purely upper and purely lower extremities training. 51
The possibility of improving white matter integrity observed in our results is of major interest since a recent study in children with UCP aged 4 to 18 years old demonstrated that in contrast to typically developing children, children with UCP do not present a maturational process of increasing CST integrity. 18 The authors of this study reported also a decreased FA in both CSTs of children with UCP, which is supported by our measures since baseline measures of FA are far lower than the FA value expected for children above 6 years old (ie, around 0.5). 22 The absence of increase with age in these low initial values was attributed by Papadelis and colleagues to a possible perinatal disruption of CST myelination and axonal integrity. Our results suggest that intensive motor skill learning–based interventions may trigger this myelination process in both CSTs. The extent of the mean changes observed in DTI measures in our treatment group is higher than the natural variability of the measure, which was characterized in healthy adults: mean reproducibility errors for FA were comprised in the 2% to 4% range and for MD in the 2% to 6% range. 52 In contrast, the smaller changes sometimes observed in the control group of our study were systematically comprised in (or lower than) the error ranges (FA: 0% to 4%, MD: <1% to 6%). This has a high clinical relevance for children with UCP.
Furthermore, the correlation between MD/FA changes in the CST emerging from the lesioned and the nonlesioned hemisphere in the treatment group suggests that intrinsic physiological factors favoring increased CST myelination underlie the activity-induced changes in individual children. According to a recent proposal, increased myelination may depend on 2 distinct processes: an activity-independent process that occurs in the absence of glutamate, and an activity-dependent process occurring when glutamate release activates NDMA (N-methyl-D-aspartate) receptors in oligodendrocytes. 53 A switch between these 2 processes could be mediated by factors such as neuregulin (NRG) and brain derived neurotrophic factor (BDNF), both of which induce increased NMDA receptor expression in oligodendrocytes. 53 The interindividual differences in the magnitude of FA increase seen in our current sample suggests that despite the same amount of practice, myelination in some children may more depend on an activity-independent mechanism, or that the switching between these 2 processes may differ between individuals. For example, the activity-dependent secretion of BDNF is subject to both genetic and epigenetic factors that may potentially explain the substantial variability in the magnitude of the changes observed in children with CP. 54
Finally, this study confirms the interest of using CST anisotropy as a predictor of response to intensive intervention in children with CP. As already observed by Schertz and colleagues, the FA of the CST appears to be a good predictor of upper extremities functional changes. 55
Study Limitations
This study has several methodological limitations. First, although data were collected in the running of ongoing randomized controlled trials, due to limited funding resulting in the controls not being collected during the first years, the groups were unbalanced, leading to a protocol that is not fully randomized. Second, although a previous study pointed some discrepancies between fMRI activation and TMS for determination of CST organization, 56 we chose the use of an MRI indicator (DTI tracking) to define CST organization. This is consistent with a previous paper showing a good consistency between track reconstruction with DTI and TMS– but might have induce some variability in the pattern determination (perfect match between TMS and DTI in 85% of the participants). Third, due to the brain deformities, anatomical structures could not be matched with standard atlases needed for probabilistic tractography and we used thus only deterministic tractography from a unique precise area in the pons. Fourth, although the outcome measures allowed to highlight changes, the level of detail available from DTI analysis does allow to attribute these changes to either recovery or compensatory mechanisms. Fifth, although our sample size was based on a mean change of 3 blocks at the Box and Blocks test in the more affected hand in a previous HABIT-ILE trial, the mean change observed here was lower (around 2 blocks). This is probably explained by the larger standard deviation in the present study, including children with larger impairments (some children scored 0 or 1 block at the test) for which solely a 1 block (or no) improvement could be noticed. A few children presented also a slight decrease in the measure after intervention, which might be related to some fatigue. Finally, we used the ERES to describe clinically meaningful change, which might not be the more adapted method to assess it. A different criterion might have led to different results.
Conclusion
To our knowledge, this is the first study demonstrating that an intensive intervention can improve the integrity of CST fibers in children with UCP, as evidenced by increased FA values and decreased MD values. This improvement in fiber integrity, which correlated with better motor performance, suggests that CST fibers retain some capacity for plasticity in children with CP. Present results are encouraging to new efforts in neurorehabilitation research, and may call for a reassessment of clinical practice for treating CP, which is the most common pediatric motor disorder.
Supplemental Material
NNR918841_Supplementary_material_CLN – Supplemental material for Motor Skill Training May Restore Impaired Corticospinal Tract Fibers in Children With Cerebral Palsy
Supplemental material, NNR918841_Supplementary_material_CLN for Motor Skill Training May Restore Impaired Corticospinal Tract Fibers in Children With Cerebral Palsy by Yannick Bleyenheuft, Laurence Dricot, Daniela Ebner-Karestinos, Julie Paradis, Geoffroy Saussez, Anne Renders, Anne De Volder, Rodrigo Araneda, Andrew M. Gordon and Kathleen M. Friel in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
The authors thank Dr Paul Cumming for critical reading and editing.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
Author Contributions
Yannick Bleyenheuft, Andrew M. Gordon, and Kathleen Friel have contributed to the research design, acquisition of data, analysis and interpretation of the data, as well as the writing of the manuscript. Laurence Dricot and Rodrigo Araneda have contributed to the data acquisition, to the analysis and interpretation of the data, as well as the writing of the manuscript. Julie Paradis, Daniela Ebner-Karestinos and Geoffroy Sau have contributed to the acquisition of the data, as well as the writing of the manuscript. Anne De Volder and Anne Renders have contributed to the data analysis and provided a critical review of the manuscript. All authors approved the last version of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Fondation Van Goethem-Brichant (Prize 2012); KF and AG were funded by the NIH grants R03HD07315/K01NS062116.
Clinical Trials
Data were collected in the context of 2 clinical trials registered as NCT01700777 and NCT02667613.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
