Abstract
Keywords
Background
A common side effect of taxane chemotherapy (docetaxel/paclitaxel) is chemotherapy-induced peripheral neuropathy (CIPN). CIPN is a small fiber sensory predominant neuropathy developing in the hands/feet that worsens with increasing dose and duration. It affects the Aβ, Aδ, and C-fiber function involved in light touch and vibration sense, thermal detection, and thermal pain. Symptoms include hypoesthesia, dysesthesias, hyperalgesia, allodynia, and neuropathic pain. At present, there are few effective treatment options for CIPN.1-7
Risk factors for CIPN include previous chemotherapy, exposure to toxins, and genetic causes. 3 One potential risk factor is preexisting nerve health. 8 Mechanical entrapment neuropathies (such as carpal tunnel syndrome) and decreased neural excursion on nerve testing (such as postoperative nerve irritation from breast surgery) may sensitize the nerve to further injury with the chemotherapy drug and is termed the double crush injury or dual nerve disorder.9,10 Physical therapy is well established in orthopedics and plastic surgery for entrapment neuropathies, neuropathic pain, postoperative nerve repair, and regeneration.11-16 To improve nerve excursion across joints, improve pain, and decrease inflammation, therapists frequently add nerve gliding exercises to the treatment plan.16-18 Due to the success of nerve gliding treatment in orthopedic hand therapy, it was hypothesized that physiotherapy intervention prior to or during chemotherapy could improve the sensory symptoms of CIPN, reducing the burden of disease.
Accurate evaluation of sensory function necessitates quantitative sensory testing (QST). These are noninvasive tests used in pain research to clinically assess neuropathy with validity and reliability dependent on standardized methods and patient cooperation.19-22 It is used for sensory detection and pain thresholds for both mechanical and thermal stimuli and provides information on large myelinated (Aβ), small thinly myelinated (Aδ), and unmyelinated (C-fiber) function or dysfunction. The advantages of QST is that it is a repeatable, quantifiable response with a controlled stimulus that directly measures participants sensory experience. 23
Patient-reported symptoms and QST were used to evaluate the role for a physical therapy home program during chemotherapy for the prevention and management of CIPN. Participants were assigned to either treatment (physical therapy program) or control (treatment as usual) group. We also evaluated the possibility of a dual nerve disorder contribution to severity of symptoms by comparing sensory thermal and vibration QST results on the surgical arm to the nonsurgical side. Finally, we completed a nonrandomized subgroup analysis of those who remained “active” (reported general exercise >4 times per week on at least 3 out of 4 reassessment visits that was not part of treatment intervention) to less active participants to further observe the correlation between exercise and sensory preservation.
Methods
Participants and Eligibility
Stages I to III patients with breast cancer that attended oncology clinics at CancerCare Manitoba were eligible if they were receiving standard adjuvant taxane chemotherapy. Two different chemotherapy regimens were included: (1) docetaxel 75 mg/m2 and cyclophosphamide 600 mg/m2 given every 21 days × 4 cycles (TC) or (2) 5-fluorouracil 500 mg/m2, epirubicin 100 mg/m2, cyclophosphamide 500 mg/m2 given every 21 days × 3 cycles, followed by docetaxel 100 mg/m2 given every 21 days × 3 cycles (FECD).24-26 Participants were approached at their initial oncology visit to advise them of the study. Interested patients signed a consent to contact form. Participants were excluded if they had comorbid conditions causing peripheral neuropathic symptoms (including previous chemotherapy and diabetes) or patients not scheduled for one of the aforementioned taxane-based regimens. Prior history of mechanical nerve entrapment without clinical symptoms (ie, carpal tunnel syndrome) were included. In total, 80 patients were contacted to participate between November 2014 and January 2017. Sixty-one participants were enrolled, and 7 withdrew over the course of the study. Reasons for withdrawal included too ill to continue, too busy to continue, or rediagnosed as stage IV. Data collection was complete by October 2017 (n = 54). Six participants had received neoadjuvant chemotherapy or chemotherapy that differed from the TC or FECD regimes and were excluded from analysis because these regimes substantially affected the timing for surgery and, in some cases, the overall dose of taxane chemotherapy. Forty-eight participants were included for analysis. Sample size was estimated at 100 participants for 80% power to detect a 25% difference between groups one-sided (α = 0.05), that is, if there is a 60% incidence in any grade CIPN with no intervention, incidence with intervention would need to be reduced to 35%.
Protocol
This was a single-blind (blinded assessor) randomized controlled exploratory study testing the potential for nerve-specific home exercises to improve pain and function postsurgery during and after chemotherapy. Random numbers were drawn by a member of the team directly involved to assign participants to either treatment or control conditions to ensure concealed allocation of 1:1. Approval was obtained from both the Health Research Ethics Board (H:2014:281) at the University of Manitoba and the Research Resource Impact Committee (RRIC 2014-031) at CancerCare Manitoba. The study was registered with clinicaltrials.gov (NCT02239601).
Patient questionnaires and QST were used to evaluate the treatment effect of these exercises, the dual nerve disorder, and sensory preservation. Study participants attended the Pain Research Laboratory, College of Rehabilitation Sciences, University of Manitoba, for all nerve assessments.
Physical Therapy Treatment
Four visits with a physical therapist to develop a home exercise and education program were provided to the treatment group at the beginning of chemotherapy. Nerve gliding exercises were the focus of the home program and were completed 3 times daily requiring 5 to 10 minutes to complete. Nerve gliding exercises are theorized to help by elongating the nerve, restoring mobility, and decreasing neural edema by promoting axoplasmic flow.12,17 Participants were advised to complete these exercises throughout and after chemotherapy until the symptoms of neuropathy subsided.
Education was provided on how to manage symptoms of neuropathic pain, cold intolerance, and hyperalgesia. This included compression gloves, heated mittens, resting wrist splints and desensitization, and stereognosis exercises. Education was also provided for hypoesthesia symptoms including safety and protection. Stretching and range of motion exercises for the neck and upper limb and axillary webbing exercises were provided. All the information was contained in an education package. Only one follow-up phone call (6 weeks later) was provided by the physical therapist. The purpose of the call was to ask if they had questions about the exercises and encourage compliance. To avoid a possible treatment effect, no other intervention for neuropathy during or after chemotherapy was provided to either the treatment or control groups.
Outcome Measures
Primary Outcome Measures for Physical Therapy Intervention
The primary outcome measures related to function and quality of life.
Numeric Pain Rating Scale (NPRS) rated CIPN pain. The NPRS is an 11 point scale (0-10) ranking pain from 0, indicating no pain at all, to 10, indicating worst pain imaginable on each assessment visit, of the finger tips. This was to identify pain specific to CIPN. A change score of 2 is reported to be a clinically relevant change.
Disability of the Arm, Shoulder, and Hand (DASH) is a 30-item participant-reported questionnaire commonly used to gauge upper limb function. The DASH was chosen because of high test-retest reliability and the responsiveness and construct validity in patients with breast cancer over other quality of life measures.27,28 A minimal clinical important difference is a change score of 15.
Self-report version of Leeds Assessment for Neuropathic Symptoms and Signs (S-LANSS) is a 7-item patient-reported questionnaire and was used to confirm the presence of neuropathic pain. 29 The score ranges from 0 to 19, with a score above 12 indicative of neuropathic pain/symptoms. S-LANSS was chosen because of its specificity and accuracy in a cancer population.30,31 Participants were requested to answer specifically for the hands, not feet.
Secondary Outcome Measures for Physical Therapy Intervention
Vibration analysis testing for perception thresholds are specific to Aβ nerve fibers. The TSAII Vibration Sensory Analyzer (VSA3000; Medoc, Israel) was used. The pulp of the index finger lightly touched the sensor that delivered random and varying vibration amplitudes (0-130 µm at 0.1-4.0 µm/s) using the limits testing method on the VSA. The participant responds “yes/no” to sensing the vibration. Vibration perception was selected for its sensitivity and has been suggested to be the first clinical sign of CIPN and was tested bilaterally. 32
Pressure Algometry (Somedic AB, Sweden) is a hand-held device to measure pressure/pain thresholds. Participants were sitting with feet on the floor, and pressure was applied perpendicular to the quadriceps muscle (centered 18 cm proximal to the bent knee). Increasing pressure was applied on a single trial until the participant determined that the sensation has changed from a feeling of pressure to a feeling of pain and pressure (kPa) is recorded. When tested at a distant site from the source of pain, this test measured relative hyperalgesia. Lower pressure values (more sensitive to noxious stimuli) suggest impaired central pain and/or diminished descending inhibition pathway. The left quadriceps muscle was tested as a measure of relative hyperalgesia.
Hand dynamometry recorded grip strength in kilograms and was used as a measure of function (3 trials). The dominant hand was tested using the Jamar dynamometer (Patterson Medical) in the second handle position. All 48 participants were right handed.
Outcome Measures Assessing Dual Nerve Disorder
Thermal detection threshold (warm and cool) and thermal pain thresholds (hot and cold) measures Aδ and C-fiber function. The Neurosensory Analyzer (TSA-II, Medoc, Israel) thermode was attached to the cutaneous tissue on the palmar surface of the index and middle finger. Temperature is increased or decreased by 0.1°C increments until the patient presses a button indicating temperature detection or thermal pain. The test immediately stops as soon as the patient presses the button. The patient is always in control and is never at risk for tissue damage (temperature limits are set to vary only from 0°C to 50°C).
QST data from the TSA-II including thermal detection, thermal pain, and vibration sensory analyzer data from the physical therapy question were gathered bilaterally for left and right hands. The surgical arm was compared with the nonsurgical arm to evaluate whether sensory impairment was greater on the surgical/cancer side, possibly suggesting a dual nerve disorder.
Measuring Activity
Participants were asked about their level of exercise per week at each assessment visit. Measuring general activity levels throughout chemotherapy was important as it may be a confounding variable. Previous studies have correlated general exercise to preservation of sensation, and we wanted to be able to ensure that exercisers were relatively balanced between the groups. 33 Participants were considered active if they reported engaging in any form of moderate activity for 30 minutes at least 4 times a week (120 minutes/week consistent with recommended activity guidelines) on at least 3 out of 4 reassessment visits. 34 Types of reported activities included cycling, running, yoga, swimming, Zumba, dance classes, and tennis. Walking was included if it was at least 30 minutes at a moderate pace. In total, 10 participants from the control group and 5 participants from the treatment group were considered active. This subanalysis redefined the groups as “active” (n = 15) and “less active” (n = 33) for the comparison of both thermal and vibration QST data to observe an association with physical activity.
Assessment Visits
Five visits in total with the research coordinator were required over an average of 8 months at the Pain Research Laboratory, College of Rehabilitation Sciences, University of Manitoba. The first visit proceeded after the initial oncology visit confirmed chemotherapy was required. This primary assessment (visit 1) completed the informed consent and baseline nerve evaluation including subjective questionnaires and QST data. Prerandomized envelopes containing experimental group assignment were provided after initial assessment, and participants were advised not to disclose their assigned group to the research coordinator in order to maintain blinding. Standard care for the control group was to attend nerve reassessments at the pain research laboratory, but no exercises or appointments with a physical therapist were provided. Instructions in the treatment group envelope directed participants to arrange 4 sessions as soon as possible with the physical therapist assigned to the study prior to the start of chemotherapy (most often a 2-week window was available prior to the start of chemotherapy). If participants had questions, they were to contact the oncologist or co-investigator on the study.
Nerve reassessment date for visit 2 (mid-taxane chemotherapy) was scheduled based on participant chemotherapy treatment cycles and start date (timing for visit 2 varied between 7 and 18 weeks depending on the chemotherapy schedule). Visit 3 was post-chemotherapy. Visit 4 was 3 months post-chemotherapy, and visit 5 was the final assessment at 6 months post-chemotherapy. After the final evaluation participants disclosed whether they were treatment or control and received a $70 honorarium to offset transportation costs. Patients with residual symptoms allocated to the control group were then offered a physical therapy assessment and home exercise program tailored to their needs.
Data Analysis
Outcomes for the effect of the treatment and control groups were analyzed with a mixed models analysis, which accounts for repeated measurements, unequal time intervals, and data missing at random. Comparisons were made during follow-up. Linear mixed models were used to predict continuous outcomes when the assumption of normality was met. Quantile mixed models were used to predict ordinal outcomes or continuous outcomes where the assumption of normality was not met. More than half of the NPRS pain scores were 0, indicating “no pain.” Due to this floor effect, the pain scale score was dichotomized into 0 and 1+ (no pain or pain) for analysis. Logistic mixed models were used for predicting binary outcomes, and the results were marginalized using the approach by Hedeker et al, 35 which converts the “subject-specific” estimates to “population-averaged” estimates. Residual plots were used to evaluate the assumption of normality and to detect outliers. The assumption of linearity for continuous predictors was evaluated using restricted cubic splines. The predictors for all models were treatment variable and follow-up time. Analyses were run using the R project for statistical computing software version 3.4.1 and SAS 9.4. Specific programs for the models included nmle package (linear mixed), lqmm (quantile mixed), and NLMixed (logistic mixed).36,37
Results
Forty-eight stages I to III breast cancer participants scheduled to receive adjuvant chemotherapy were followed postsurgery until 6 months post-chemotherapy. Figure 1 provides a CONSORT diagram of the enrolment process. Table 1 illustrates the demographics of participants. The mean age was 61.5 years (range 37-78). There were no statistical differences in baseline demographics between the groups. Participants were followed for an average of 8.25 months (range 6.6-9.4 months). One mid-chemotherapy visit was missed in the treatment group as the participant was too sick to attend. This left 87 reassessment visits in the treatment group (n = 22) and 104 in the control group (n = 26).
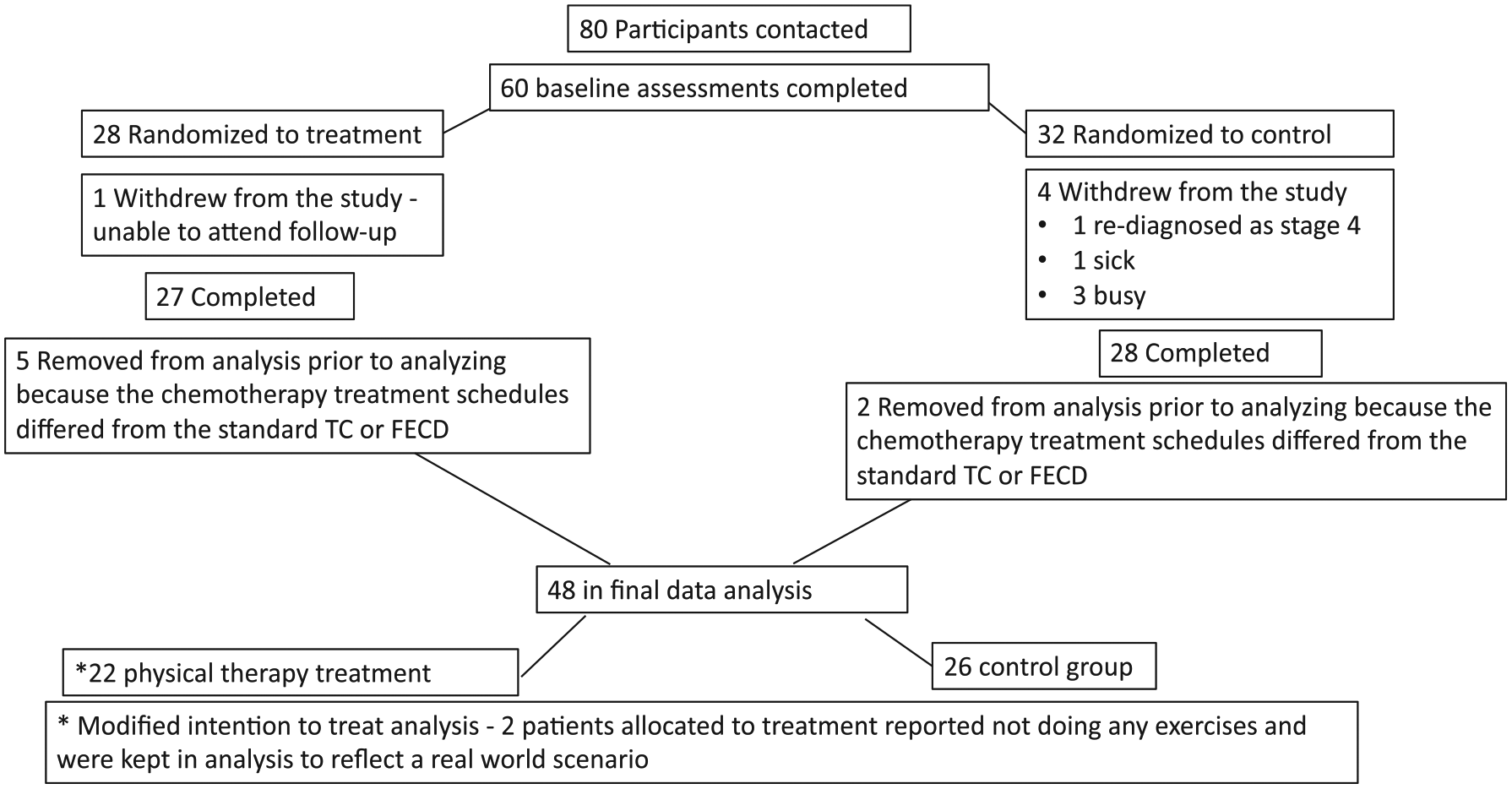
CONSORT diagram of enrollment. This figure pictorially represents the enrolment and withdrawal process for the study.
Demographics of Participants a .
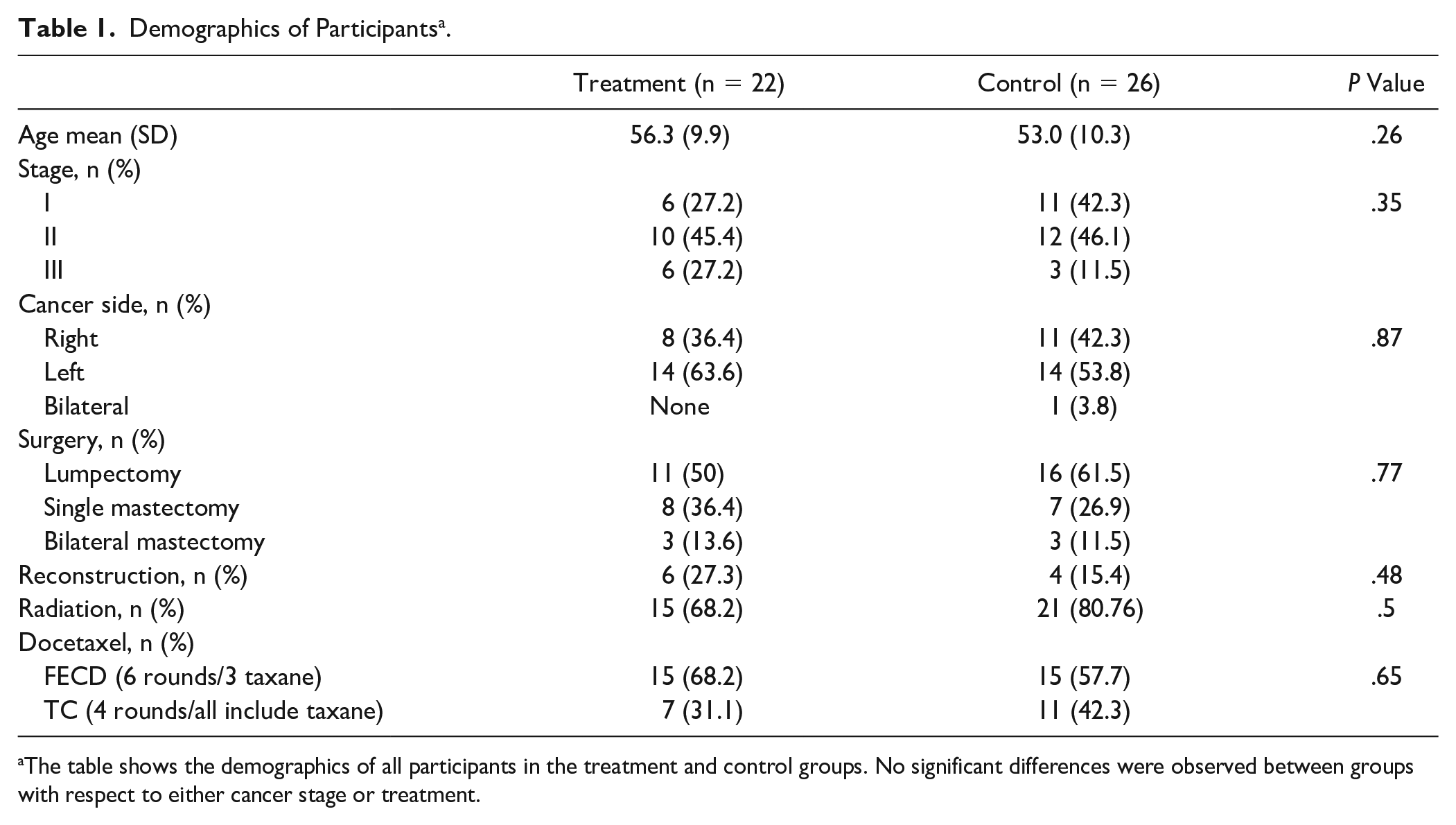
The table shows the demographics of all participants in the treatment and control groups. No significant differences were observed between groups with respect to either cancer stage or treatment.
Pain was defined as the NPRS of 1 or more (1+) on the 11-point scale. Thirty percent of participants in the treatment group had 1+ pain scores compared with 49% in the control group (odds ratio [OR] 0.41, 95% confidence interval [CI] 0.17-1.01;
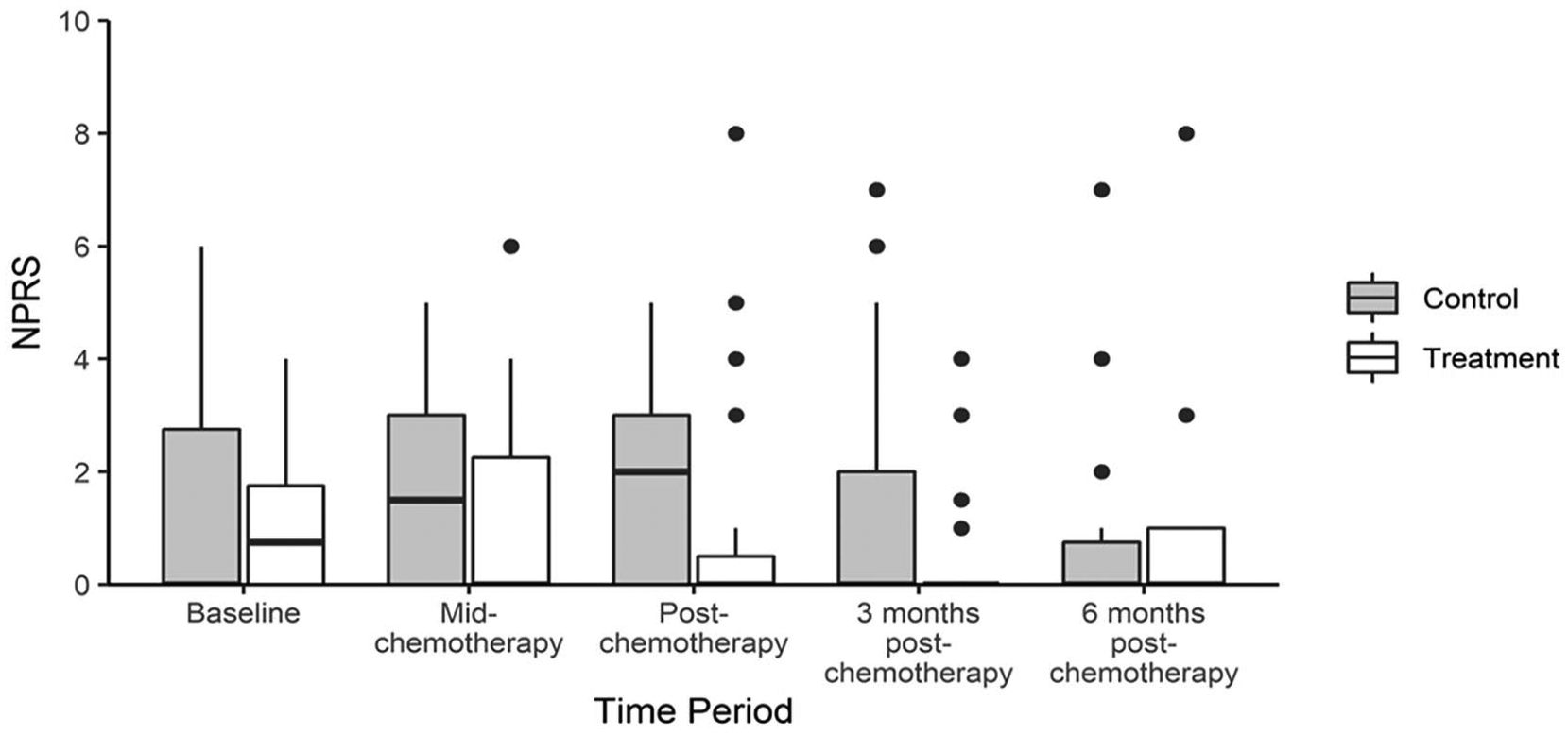
Numerical pain report over time. This graph represents raw pain scores and time trend for all participants (n = 48). Control participants (n = 22) are represented in grey and treatment participants (n = 26) in white. The box plots represent the median and IQR (interquartile range) with the whiskers representing the maximum values. The dots are the outliers (defined as Q3+1.5&*IQR or below Q1−1.5*IQR). The treatment group demonstrated clinically relevant decreases in pain scores (change score of 2 on the NPRS) post-chemotherapy and 3 months post-chemotherapy.
Physical Therapy Outcome Measures a .
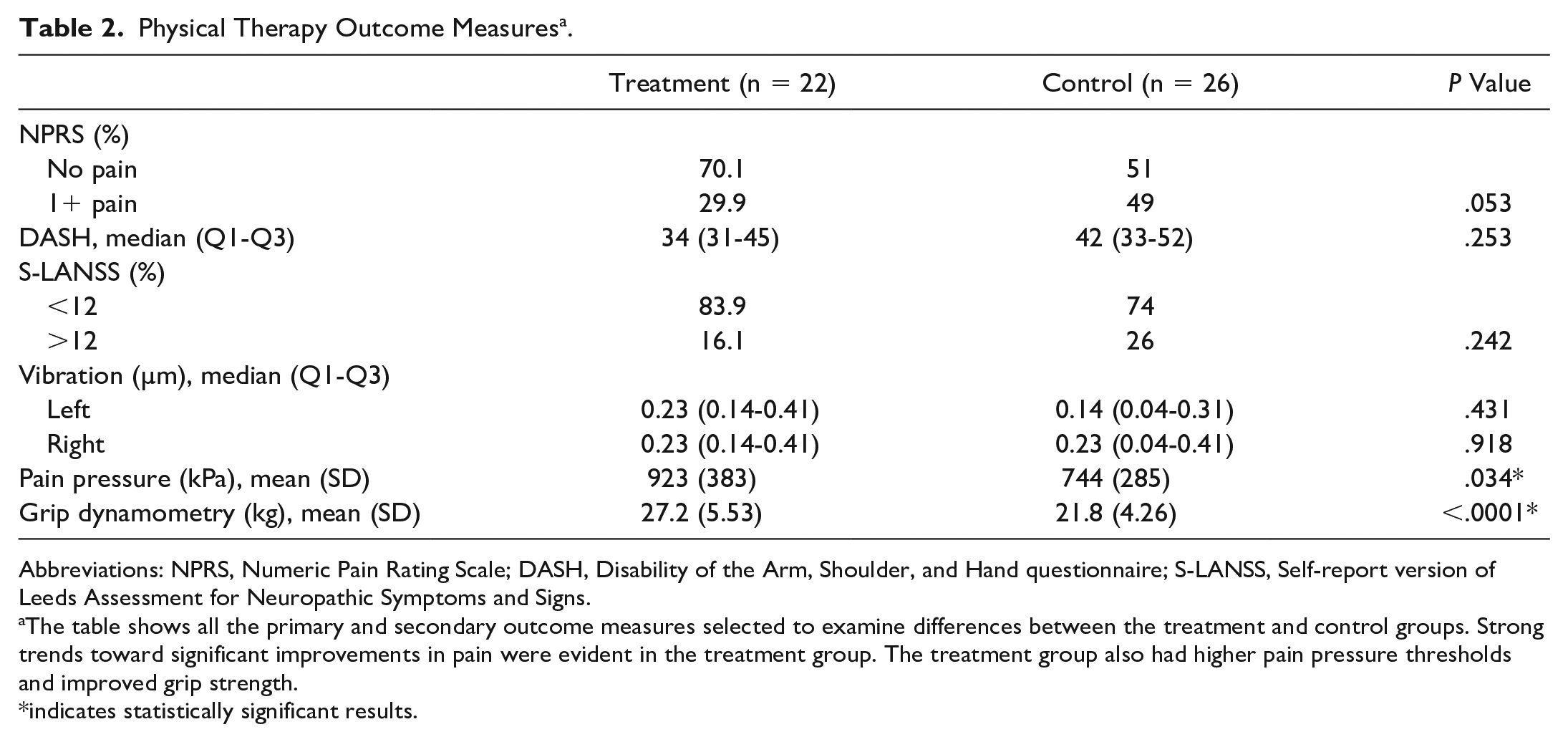
Abbreviations: NPRS, Numeric Pain Rating Scale; DASH, Disability of the Arm, Shoulder, and Hand questionnaire; S-LANSS, Self-report version of Leeds Assessment for Neuropathic Symptoms and Signs.
The table shows all the primary and secondary outcome measures selected to examine differences between the treatment and control groups. Strong trends toward significant improvements in pain were evident in the treatment group. The treatment group also had higher pain pressure thresholds and improved grip strength.
indicates statistically significant results.
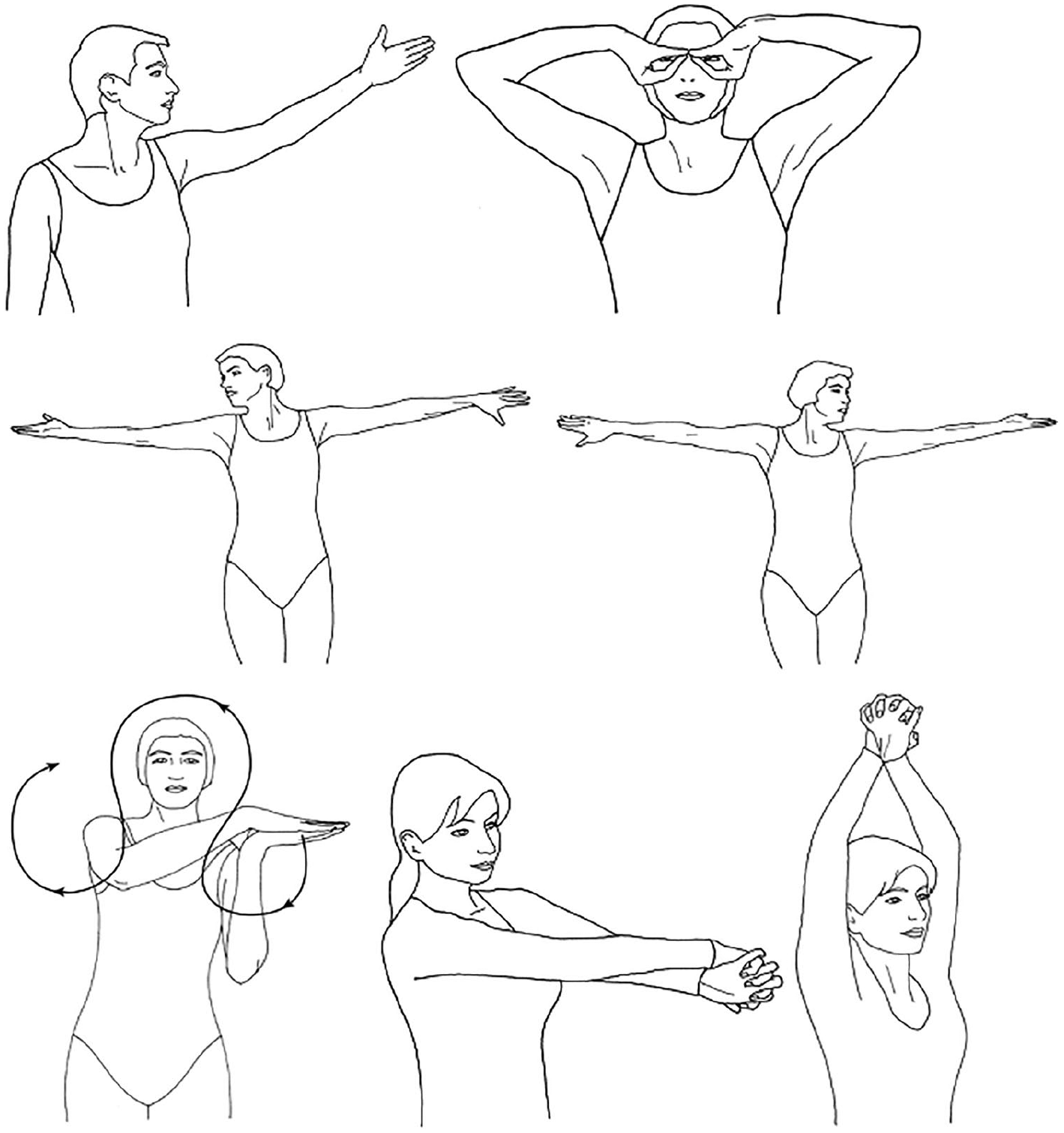
Nerve gliding exercise. Examples of some of the nerve gliding exercises prescribed. Reprinted with permission from David Butler, taken from The Sensitive Nervous System, 2000.
QST scores on the participants’ surgical upper extremity were compared to the nonsurgical upper extremity to assess whether a dual nerve disorder was evident from tumor excision, radiation combined with chemotherapy. Table 3 summarizes the results for the comparison of surgical side to nonsurgical side. The results indicate no differences for vibration perception, warm, cool, hot, or cold pain thresholds were found, suggesting that postoperative nerve irritation postsurgery does not contribute to a dual nerve disorder when combined with chemotherapy.
Dual Nerve Disorder (Surgery − Nonsurgery Scores) a .
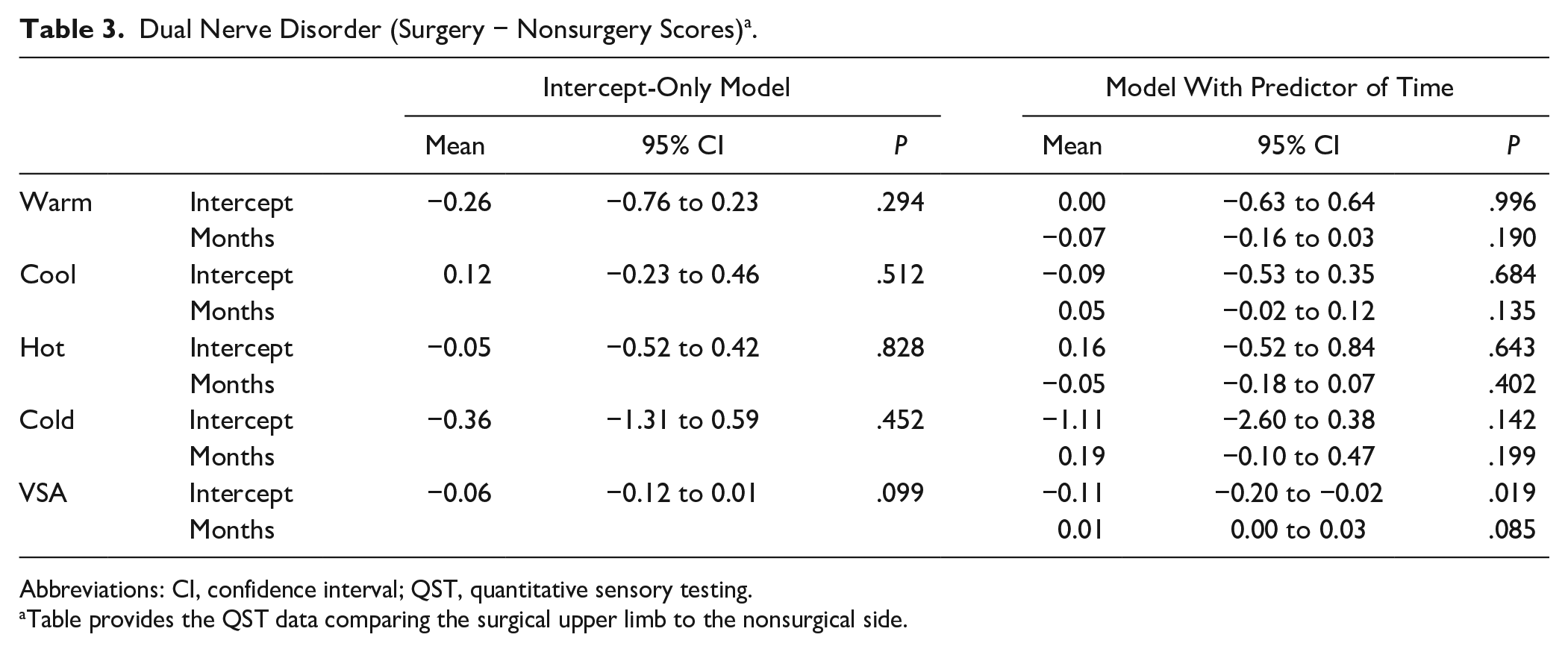
Abbreviations: CI, confidence interval; QST, quantitative sensory testing.
Table provides the QST data comparing the surgical upper limb to the nonsurgical side.
Finally, we evaluated the difference between patients who were more active compared with those who were less active. Table 4 illustrates the observation of significantly better vibration scores (Left
Participants Considered Active (With General Exercise) and Less Active (No Regular Exercise) a .
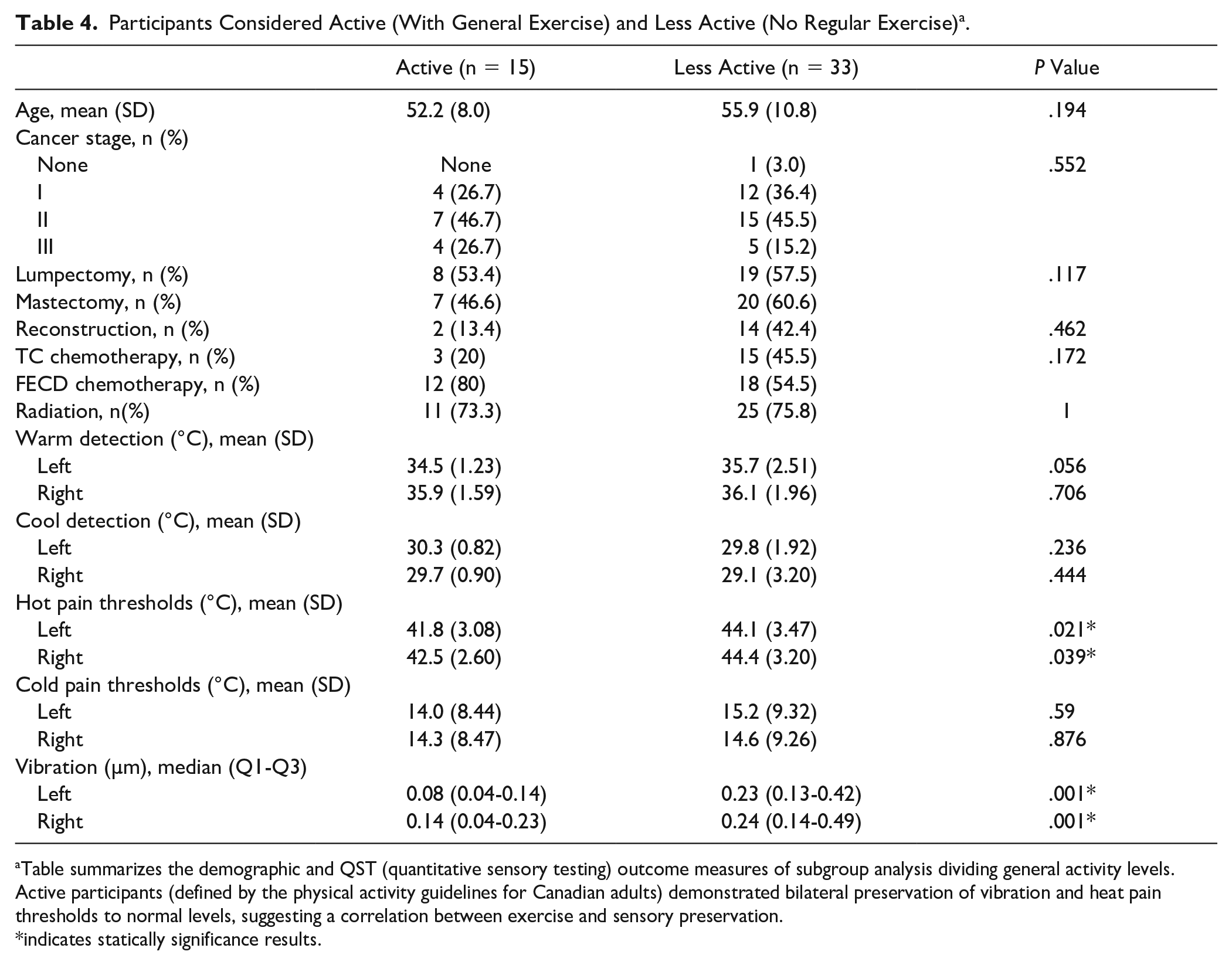
Table summarizes the demographic and QST (quantitative sensory testing) outcome measures of subgroup analysis dividing general activity levels. Active participants (defined by the physical activity guidelines for Canadian adults) demonstrated bilateral preservation of vibration and heat pain thresholds to normal levels, suggesting a correlation between exercise and sensory preservation.
indicates statically significance results.
Raw Data (Mean and SD) at Each Time Point.
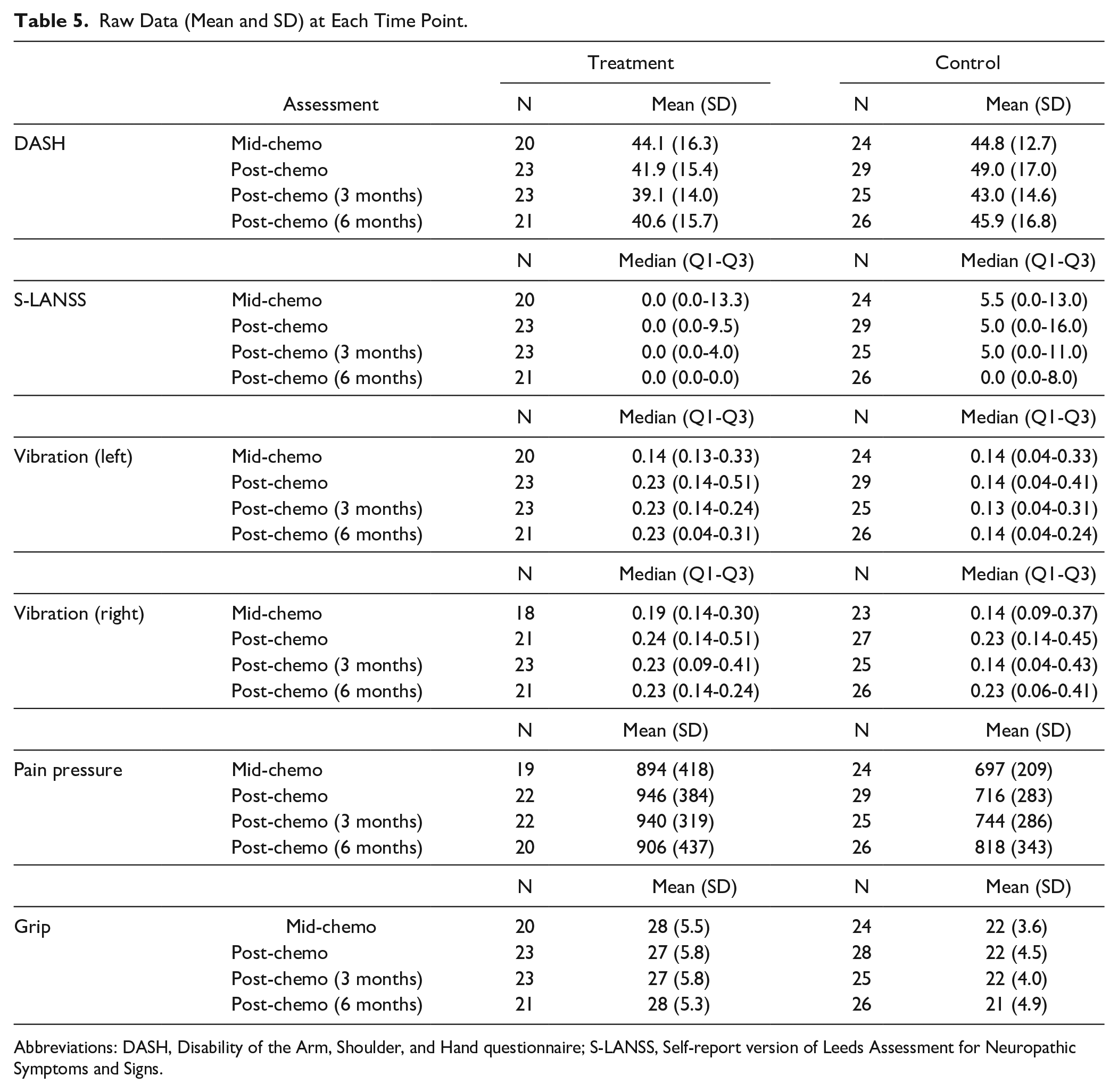
Abbreviations: DASH, Disability of the Arm, Shoulder, and Hand questionnaire; S-LANSS, Self-report version of Leeds Assessment for Neuropathic Symptoms and Signs.
Discussion
The physical therapy treatment group reported clinically relevant improvements (trending toward statistical significance) for CIPN pain on the NPRS, and statistically significant improvements in pain pressure thresholds, and grip strength compared to the control group. The type of pain patients reported varied throughout the trial and has been published previously. 38 Generally neuropathic symptoms were more likely to be reported during chemotherapy and decreased over time. Physical therapy treatment for nerve disorders is well established for entrapment, neuropathic pain, nerve repair, and regeneration.11-16 Treatment addresses both positive and negative sensory symptoms that are involved in nerve compression injuries, crush injuries and nerve lacerations to improve pain and function, regain diminished sensation, and normalize hypersensitivity/allodynia. 14 Conservative treatment with physical therapy includes nerve gliding exercises that have been shown to reduce neural edema and decrease pressure in the carpal tunnel and restore function and improve pain in the neck and arm.16,18 Immediate short-term hypoalgesia has been reported with nerve gliding exercises and may be the explanation for the reduction in reported pain in the treatment group. 39 Referral for a physical therapy assessment and home program prior to treatment for those undergoing chemotherapy may assist in improved symptoms and tolerance during chemotherapy treatment, especially when general exercise can be prescribed and adjusted throughout chemotherapy. 40
A study by Kelley and Jull 41 was one of the earliest to show sensitivity during movement in the neural tissue following breast cancer surgery, and others have replicated the results. 42 Sensitized neural tissues pre-chemotherapy may predispose the nerves to further trauma from chemotherapy and/or radiation, resulting in a double-crush syndrome or dual nerve disorder prolonging symptoms. From this, it was hypothesized that impaired thermal and vibration sense would result with the surgical side exhibiting impairments in QST. The results indicate no differences in vibration or thermal detection and pain thresholds between the arms. Too few participants had a history of either prior nerve damage or entrapment to allow for analysis based on previous history of nerve damage.
An epidemiological study on CIPN risk factors found a positive association between exercise on most days and decreased risk of neuropathy. 33 In our current study, general activity was not controlled or randomized, but we did ask each participant about exercise amount and frequency on each visit. We did not quantify exercise in a way that would calculate metabolic equivalents, which would have been useful and should be collected in future trials.
Our findings are consistent with observations found in diabetes research that a correlation is present between exercise and sensory preservation.43-46 Studies have correlated exercise to cutaneous nerve reinnervation in diabetic neuropathy,47-49 improved painful diabetic neuropathy symptoms, 50 improved gait, reaction time, and postural stability,51-53 and has demonstrated the safety of engaging in aerobic activity for a diabetic peripheral neuropathy population.54,55
Neuropathic pain research is uncovering the neuroimmune relationship and the association between inflammation and neuropathic pain to provide insight into the pathways and specific neurotrophic factors that exercise assists in modulating/normalizing to minimize symptoms.40,43
In CIPN, exercise improves strength and balance and minimizes CIPN symptom progression. 56 Having a normal body mass index and engaging in moderate to vigorous physical activity are associated with less CIPN symptoms in a breast cancer population exposed to taxane chemotherapy. 57 While our results should be considered exploratory and require confirmation, it is promising that many other studies are producing similar results supporting a role for exercise in all causes of neuropathy.
Limitations
Since we collected multiple outcome measures, it is possible that some variables deemed significant may be due to a random effect. However, the unilateral direction of improvements for all the quality of life variables in the physical therapy intervention group (NPRS, DASH, S-LANSS, functional grip strength) suggest a consistent and real effect. The bilateral effect of sensory preservation in the exercise analysis also suggests a correlation. We only recorded general activity in an informal way throughout the trial. Future trials need to record baseline activity levels of participants and need a standardized way to calculate METs (metabolic equivalents) due to the correlation between general exercise and reduction in CIPN symptoms. QST provides an objective stimulus but relies on a subjective patient response. Great care was taken to ensure standard wording and testing procedures. Despite being asked to record a daily log, only 3 participants completed the written diary of exercises despite being asked to record a daily log. Future studies should confirm exercise adherence. The visual analog scale as a continuous measure may have improved sensitivity over the NPRS. Finally, we chose to measure the hands even though CIPN is a bilateral polyneuropathy affecting hands and feet. This was for 2 reasons. First, patients with breast cancer receiving chemotherapy post tumor excision were thought to benefit most from nerve exercises in the upper limb. Second, QST and questionnaire assessments for both the hands and feet bilaterally were thought to be too time consuming and there was concern that the addition of the lower extremity would lead to a larger attrition rate.
Conclusion
To our knowledge, no study to date has used physical therapy nerve gliding exercises during chemotherapy for CIPN. The exploratory nature of this first study suggests physical therapy home exercise program administered postoperatively may improve pain for patients with breast cancer throughout chemotherapy. More studies are required that can evaluate whether these exercises might assist in minimizing dose reductions or treatment delay for the improvement of chemotherapy treatment delivery. Further research is needed to confirm the observation that general exercise can help with sensory preservation.
Footnotes
Acknowledgements
CancerCare Manitoba Foundation for grant support for this project.
Authors’ Note
The clinical trials registry was opened September 10, 2014, prior to ethics approval, recruitment, and our final protocol meeting. The primary outcome measure was change in quantitative sensory testing. Pain and function were secondary measures. Primary and secondary outcome measures were switched but the clinical trials database was only amended when recruitment was closed prior to data analysis (February 6, 2018). On September 19, 2019, the complete methods/analysis was entered in the clinical trials database. The clinical trials database now reflects NPRS, DASH, S-LANSS as primary outcomes and secondarily VSA, pain pressure, and grip strength.
All data, being stored for 7 years, have been wiped of all identifying information and are available to anyone that requests it.
Author Contributions
Elizabeth Andersen Hammond: PI, collected data, submitted ethics, assisted with grant writing and writing of the manuscript.
Karen Steinfeld: Developed the physical therapy program and administered the program to patients.
Pascal Lambert: Analyzed all the data.
Marshall Pitz: Grant writing, input to original protocol, assisted in writing the manuscript.
Barbara Shay: Elizabeth’s PhD advisor and had input in all aspects on grant, ethics, data collection, interpretation, and final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: CancerCare Manitoba Foundation.
Ethics Approval and Consent to Participate
Approval was obtained from both the Health Research Ethics Board (H:2014:281) at the University of Manitoba and the Research Resource Impact Committee (RRIC 2014-031) at CancerCare Manitoba.
Clinical Trial
The study was registered with clinicaltrials.gov (NCT02239601).
