Abstract
Background. Kinematic abundance permits using different movement patterns for task completion. Individuals poststroke may take advantage of abundance by using compensatory trunk displacement to overcome upper limb (UL) movement deficits. However, movement adaptation in tasks requiring specific intersegment coordination may remain limited. Objective. We tested movement adaptation in both arms of individuals with chronic stroke (n = 16) and nondominant arms of controls (n = 12) using 2 no-vision reaching tasks involving trunk movement (40 trials/arm). Methods. In the “stationary hand task” (SHT), subjects maintained the hand motionless over a target while leaning the trunk forward. In the “reaching hand task” (RHT), subjects reached to the target while leaning forward. For both tasks, trunk movement was unexpectedly blocked in 40% of trials to assess the influence of trunk movement on adaptive arm positioning or reaching. UL sensorimotor impairment, activity, and sitting balance were assessed in the stroke group. The primary outcome measure for SHT was gain (g), defined as the extent to which trunk displacement contributing to hand motion was offset by appropriate changes in UL movements (g = 1: complete compensation) and endpoint deviation for RHT. Results. Individuals poststroke had lower gains and greater endpoint deviation using the more-affected compared with less-affected UL and controls. Those with less sensorimotor impairment, greater activity levels, and better sitting balance had higher gains and smaller endpoint deviations. Lower gains were associated with diminished UL adaptability. Conclusions. Tests of condition-specific adaptability of interjoint coordination may be used to measure UL adaptability and changes in adaptability with treatment.
Introduction
Impaired upper limb (UL) function is one of the most disabling consequences of stroke that persists into the chronic phase. 1 An important underlying deficit that may impact effective UL use is an impaired ability to adapt movement to unexpected changes in the environment. For example, Baniña et al 2 showed that, compared with healthy controls, individuals with mild poststroke UL impairments used less elbow extension and more trunk movements (ie, flexion) when reaching rapidly toward a target within arm’s reach while avoiding unexpected obstacles in the hand path. Although these individuals were considered to be well recovered based on standard clinical assessment, higher order motor function (eg, obstacle avoidance involving rapid adaptation of arm movement) was impaired and resulted in subtle motor deficits (ie, critical timing deficits) that were related to decreased self-reported activities of daily living such as opening a drawer or a car door. Similar deficits in motor adaptability or motor problem solving 3 in subjects with chronic stroke have been attributed to difficulties in rapidly adapting elbow and shoulder interjoint coordination when reaching to targets placed beyond arm’s length. 4
Excessive trunk movements, although often considered compensatory, represent a general ability of the nervous system to exploit the kinematic abundance of the body5,6 that allows a reaching task to be performed using different combinations of joint rotations. The latter also refers to the concept of motor equivalence initially described by Lashley. 7 Almost 80% to 90% of individuals with chronic moderate-to-severe stroke incorporate up to 33% more trunk movement8 -10 and/or combined shoulder abduction and elevation (ie, arm-plane) movements11,12 even when reaching for targets placed at or within arm’s length. However, some subjects with severe stroke were unable to use the arm-plane compensation adaptively to reduce reaching errors compared with people with mild-to-moderate stroke and healthy controls. 12 Similarly, Tomita et al 13 found that more severely affected patients who used compensations for reaching while standing were less able to adapt elbow-shoulder interjoint coordination to maintain the same reaching trajectories when a trunk perturbation occurred. Thus, the presence of motor compensations in patients with stroke may interfere with the ability to adapt movement in different situations. Compensations and deficits in interjoint adaptability should be considered together in order to understand how the organization of complex movements is affected by stroke, when designing treatment interventions to improve UL function.
Specific adaptive strategies have been described for hand stabilization and UL reaching tasks involving trunk movement (ie, when bending forward while reaching for a cup). For example, hand trajectory remains invariant in terms of direction, curvature, velocity, and error when reaches are made with or without trunk movement.14-16 The ability to maintain endpoint position or trajectory invariant occurs through adaptive motor-equivalent modifications of all UL joint rotations. These modifications combine 2 spatial movement synergies: an arm-transport synergy that coordinates the arm joints to bring the hand to the target and a compensatory arm-trunk synergy that decreases the influence of trunk motion on the hand position by additional arm-joint changes.14 -16 Such compensatory (or adaptive) synergies have been described for other movement tasks such as eye-head coordination provided by the vestibulo-ocular reflex, with the purpose of nullifying the gaze shift elicited by head rotations. 17 The ability to maintain the endpoint position or trajectory has been quantified using a gain measure. For maintaining the endpoint position, the gain ranges from 1 to 0, such that, if the hand position remains stationary despite the trunk movement, then the amount of forward trunk displacement is completely neutralized by an equal amount of backward hand displacement, resulting in a gain of 1. However, if some or all of the trunk movement is transmitted to the hand, the gain will be less than 1 or 0. High gains have been reported in healthy individuals (0.85-0.87),18 -20 while individuals with unilateral vestibular deficits,18,19 deafferentation, 21 and Parkinson’s disease 22 had lower gains (0.47-0.67), indicating reduced movement adaptability. For reaching movement, the ability to combine the 2 synergies has been expressed as an error score of ~2 cm in healthy individuals,19,20 but has not been measured in people with neurological deficits.
Analysis of reaching synergies provides information on underlying motor control and movement adaptability mechanisms and how they may be affected by stroke. Gaining a better understanding of underlying motor deficits related to more complex reaching tasks commonly used in daily life activities can provide a better focus of remedial therapy for individuals with stroke and may lead to better rehabilitation outcomes. We hypothesized that subjects with stroke would have decreased movement adaptability reflected by lower stationary hand task (SHT) gains and higher reaching hand task (RHT) endpoint displacement differences in the more-affected compared with the less-affected limb and nondominant limbs of controls (Hypothesis 1). We also hypothesized that the SHT gain and RHT difference measures would reflect the capacity for UL adaptability in individuals with different levels of stroke severity (Hypothesis 2). Preliminary results have appeared in abstract form. 23
Methods
Participants
Sixteen individuals with poststroke UL hemiparesis and 12 healthy controls participated in a cross-sectional study. Sample size was estimated from preliminary data on differences in gain scores between healthy and stroke subjects based on t tests using G*Power 3 24 with the following parameters: effect size = 1.2, α = 0.05, 80% power. The sample size was 12 individuals/group. Anticipating the potential for fatigue from participation in repeated sessions and the large number of reaching trials, we accounted for a 33% dropout rate. Individuals with stroke were included if they (1) had sustained a stroke ≥6 months previously, (2) were aged 40 to 80 years, (3) scored 3-7/7 on the arm sub-scale of the Chedoke McMaster Stroke Assessment, 25 and (4) had no stroke-related dementia (>22/30 on Montreal Cognitive Assessment). 26 Exclusion criteria were the following: (1) cerebellar lesions influencing coordination; (2) other neurological, orthopedic, or muscular disorders interfering with reaching ability; and (3) dizziness (Dizziness Handicap Inventory ≥30) 27 to avoid confounding influences of vestibular deficits on movement performance.
Age-equivalent healthy controls were recruited from the community if they had no orthopedic or neurological impairments interfering with task performance, or dizziness. Participants signed consent forms approved by the institutional review board of the Center for Interdisciplinary Research in Rehabilitation of Greater Montreal. The stroke group underwent a clinical assessment prior to the kinematic assessment. Both groups participated in one or more sessions in which arm-trunk coordination was assessed using kinematic analysis.
Clinical Assessment (Stroke)
Upper limb and trunk impairment and activity were assessed by an experienced physiotherapist uninvolved in the kinematic assessment. The Composite Spasticity Index (CSI) 28 was used to assess elbow spasticity. The CSI measured the excitability of the biceps brachii tendon reflex, wrist clonus, and passive elbow flexor resistance to a moderate velocity stretch (~100°/s). Scores of 0 to 9, 10 to 12, and 13 to 16 indicate mild, moderate, and severe levels of spasticity, respectively. 28 Motor impairment was assessed with the UL section of the Fugl-Meyer Assessment (FMA) 29 assessing voluntary movement, reflex activity, grasping, and coordination. Scored on 66 points, higher scores indicate better UL function. Sensation (/12) and proprioception (/24) were also scored on the FMA scale. The Reaching Performance Scale for Stroke (RPSS) 30 was used to assess impairments and motor compensations related to reach-to-grasp performance for objects placed within and beyond arm’s length. Six movement components—trunk, shoulder and elbow movement, endpoint smoothness, and prehension quality—were scored on 4-point (0-3) scales (18 points/task). Limitations in UL activity levels were quantified using the Chedoke Arm and Hand Activity Inventory (CAHAI-9), 31 measuring movements of different UL joints and grasp formations on a 63-point scale. CAHAI-9 scoring distinguishes between the less- and more-affected arms. The Ottawa Sitting Scale (OSS) 32 assessed trunk use during seated activities involving mobility within and outside the base of support. The OSS evaluates activities performed with feet supported and unsupported for a maximum of 20 points/condition.
Kinematic Analysis: Arm-Trunk Coordination Assessment
Prior to recording, a calibration procedure was done to quantify the maximal hand displacement (hm) that could be attributed to trunk movement alone. A rigid foam form, strapped to the trunk, positioned the upper arm in slight abduction and flexion with the elbow flexed to 80°, and the index finger in the midline. hm was computed as the total hand displacement when the subject maximally flexed the trunk.
Arm-trunk coordination was assessed using the SHT and the RHT (Figure 1). For both tasks, participants sat in an armless chair with their feet supported. The target was placed at the height of the sternum at a distance of 66% of the subject’s arm length (measured from the acromion process to the index fingertip). Arm and trunk movements from 7 wired active markers placed on anatomical landmarks (index fingertip (endpoint), wrist, lateral epicondyle of elbow, ipsilateral and contralateral acromions, and sternal notch) and the target were recorded with a 2-bar Optotrak Certus system (Northern Digital Corp, Waterloo, Canada) at 100 Hz for ~4 to 6 seconds per trial.
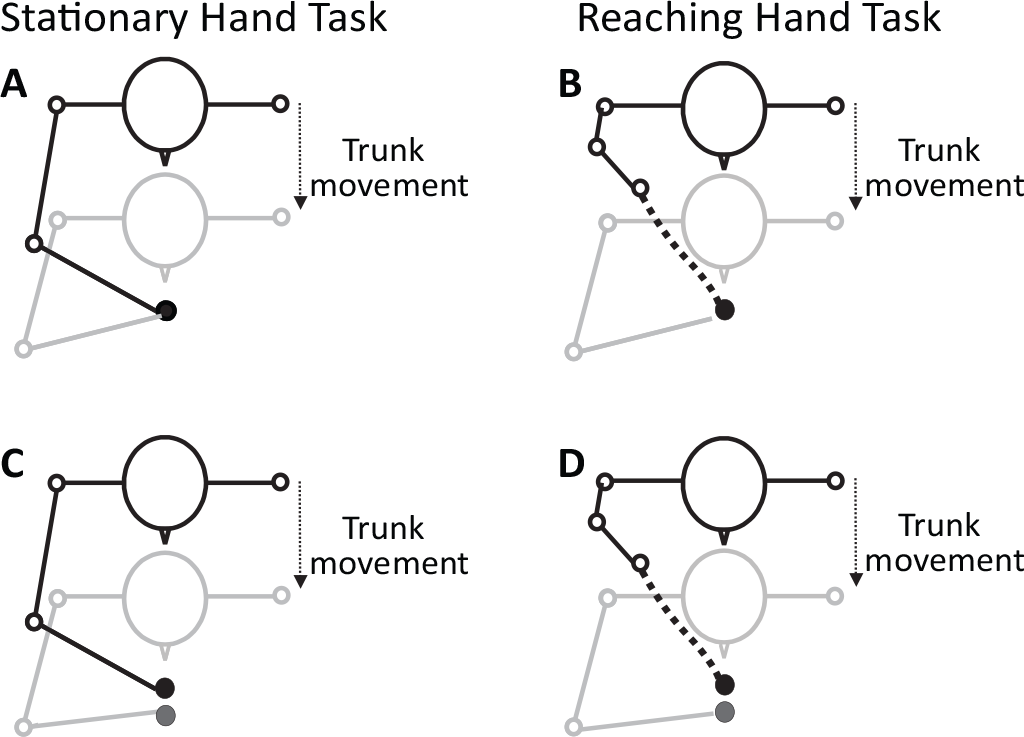
Illustration of the starting (black lines) and final (gray lines) body segment positions needed to achieve a gain of 1 for the Stationary Hand Task (A) or an endpoint error of 0 cm for the Reaching Hand Task (B). In these examples, the trunk displacement is fully compensated for by appropriate changes in shoulder and elbow movements. Lower panels illustrate cases in which the gain is less than 1 for the Stationary Hand Task (C) or the endpoint error is greater than 0 for the Reaching Hand Task (D). In C and D, lower values reflect incomplete compensation of the trunk forward motion by elbow and shoulder motion.
For the SHT (Figure 1A), subjects first placed their finger over the target position. At an auditory “go” signal, they closed their eyes and leaned their trunk forward as fast as possible while holding their index finger stationary over the remembered target position (free-trunk trials), with their arm and forearm maintained in the horizontal plane at the height of the sternum. The task was repeated 40 times with each arm, for a total of 80 trials.
For the RHT (Figure 1B), the hand was initially placed on a foam platform positioned on the chair seat next to the ipsilateral greater trochanter. At the “go” signal, subjects simultaneously leaned their trunk forward while reaching upwards and forwards toward the remembered target with eyes closed (free-trunk trials) as fast as possible. Forty trials per arm were recorded.
For both tasks, in 40% of randomly chosen trials, trunk movement was unexpectedly blocked (blocked-trunk trials) prior to the beginning of the task by an experimenter holding a 10-cm-wide strap attached to the torso at the mid-sternal level. These trials were used as catch trials for SHT and control trials for RHT to determine the amount of hand displacement in the absence of trunk movement.
Data Analysis
Data analysis was performed with custom programs written in MATLAB (Mathworks, Natick, MA). Data were interpolated with a third-order spline function to correct for missing markers during data collection and filtered with a low-pass 10 Hz Butterworth filter. Endpoint and trunk tangential velocities were computed from x, y, z displacements of the fingertip and sternal markers, respectively. Movement time was computed from movement onset and offset of the arm or trunk, defined as the first time at which endpoint or trunk tangential velocity exceeded or fell below 10% of its peak value for >50 ms. Late corrections were not considered. Endpoint error was calculated as the root mean square error of the endpoint from the target position at movement offset, and expressed as positive values. Shoulder abduction/adduction in the horizontal plane was computed where the arm positioned at 90° lateral abduction was 0°. The elbow angle in the horizontal plane was defined by vectors between acromial, elbow, and wrist markers, where a fully extended arm equaled 180°. Shoulder flexion/extension angle in the vertical plane (for RHT only) was defined by vectors between acromial and elbow markers relative to the trunk vertical where the arm alongside the body was 0°. Trunk pitch (flexion/extension) in the sagittal plane was computed, where the initial vertical position was 0° and positive values indicated forward flexion.
For the SHT, the arm-trunk gain (g) in the horizontal plane was the primary outcome measure. The amount of adaptive arm and trunk motion affecting final hand position was evaluated as g = 1 − (h/hm), where h was the average forward deviation of the hand in free-trunk trials, and hm was the maximal change in endpoint position if the trunk motion was fully transmitted to the hand, as determined in the calibration procedure. A score of g = 1 indicated that elbow flexion and shoulder horizontal adduction fully compensated for trunk motion resulting in no hand movement from the initial position (h = 0). Conversely, a score of g = 0 indicated that no compensation occurred (h = hm), and the hand moved with the trunk. Interjoint coordination was quantified as the slope of the linear orthogonal distance regression of the elbow-shoulder angle-angle plot in the horizontal plane. A slope of ±1.0 indicated that both joints contributed equally to the task, while slopes >1.0 or <1.0 indicated that the movement predominantly involved one joint. 33
For the RHT, the gain was expressed as the normalized difference (ie, error) in endpoint final positions in the free- and blocked-trunk trials. For comparisons between groups, absolute differences (d) were normalized by expressing them as a function of the ratio (r) of the participant’s arm length to the group mean arm length. Thus, for each subject, the normalized difference (dn) was dn = r·d. Interjoint coordination was quantified as above for elbow extension-shoulder horizontal adduction (horizontal plane) and elbow extension-shoulder flexion (sagittal plane).
Statistical Analysis
For greater equality of between-group comparisons and to avoid the confounding effect of UL dominance on task performance,34,35 data from both arms of stroke subjects were only compared to nondominant arms of controls. Descriptive statistics highlighted main demographic characteristics. Shapiro-Wilk and Levene’s tests verified assumptions of normality of data distributions and homogeneity of variances respectively.
For Hypothesis 1, SHT endpoint differences and gains as well as RHT arm peak tangential velocities, movement times and endpoint differences of the more-affected UL were compared with those of the less-affected and nondominant ULs of controls using appropriate nonparametric statistics (Mann-Whitney U, Wilcoxon Z) due to the ordinal nature of the data and failure to meet normality assumptions, or parametric statistics (t tests, ANOVA). For each task, slopes of the elbow-shoulder interjoint coordination of the more-affected limb were compared to values for the less-affected limb and the nondominant limb of controls with ANOVAs after verification of homogeneity of variance assumptions using Levene’s tests. Effect sizes (ES) for all significant comparisons were estimated with Cohen’s d for parametric comparisons and by dividing the Z value by the square root of the sample size for non-parametric comparisons. 36
For Hypothesis 2, the strengths of association between SHT gain/RHT differences, ranges of shoulder and elbow movements, and clinical measures were calculated using Spearman’s ρ, where correlation coefficients ≤0.39, 0.40 to 0.79, and ≥0.80 represented mild, moderate, and strong associations, respectively. 37 Least-squared differences post hoc tests were used for multiple comparisons. Analyses were conducted using SPSSv21.
Results
Twenty-eight participants (control: n = 12, age: 62.5 [SD 13.2] years, 5 females; stroke: n = 16, age: 60.1 (SD 10.2) years, 4 females, Table 1) completed all clinical and kinematic assessments. Participants with stroke had mild levels of UL motor impairment (FMA = 54.7 [SD 11.2]) and moderate levels of activity limitations (CAHAI-9 = 45.2 [SD 15.2]).
Demographic Characteristics and Clinical Scores of Stroke Subjects.
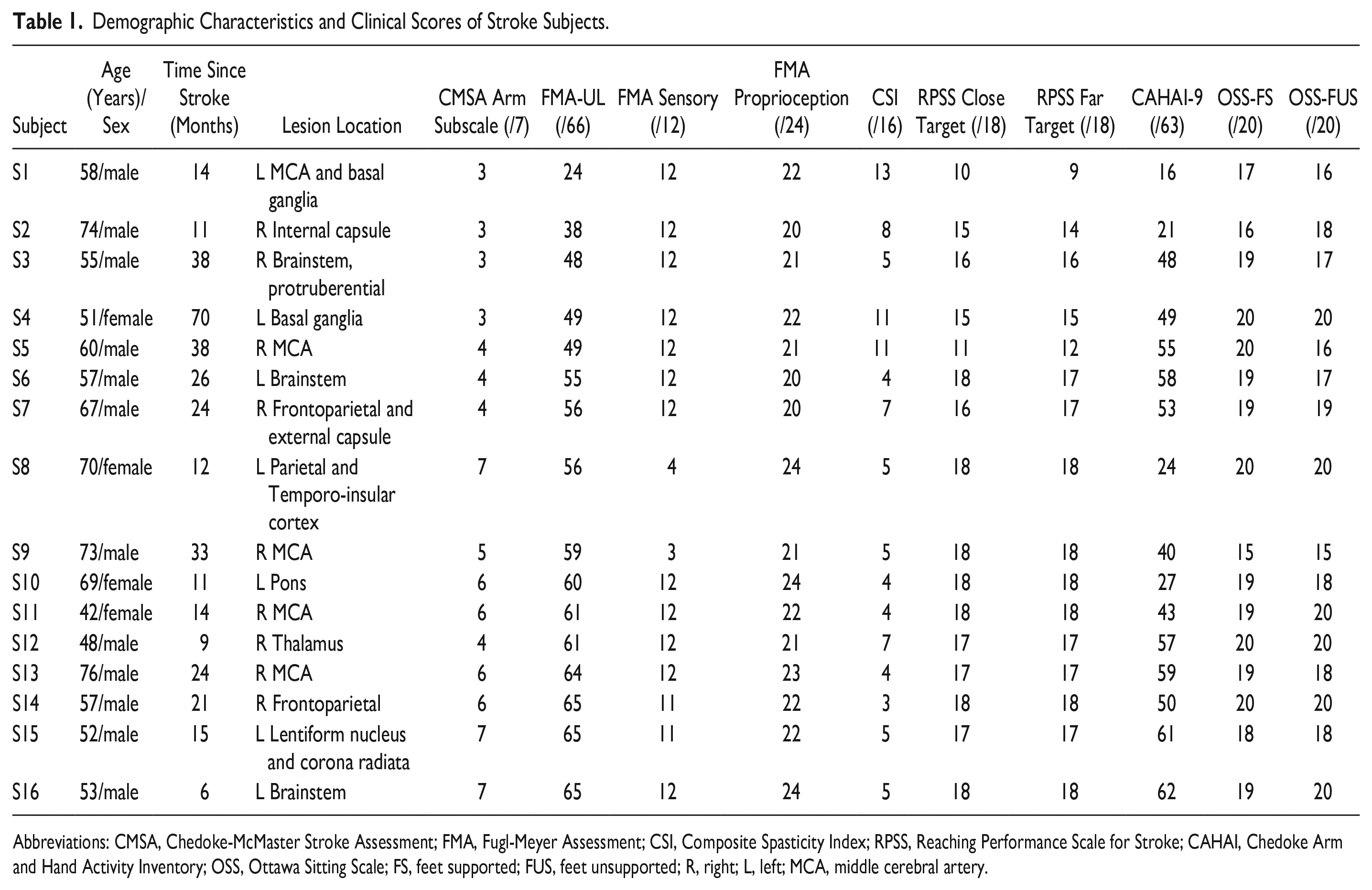
Abbreviations: CMSA, Chedoke-McMaster Stroke Assessment; FMA, Fugl-Meyer Assessment; CSI, Composite Spasticity Index; RPSS, Reaching Performance Scale for Stroke; CAHAI, Chedoke Arm and Hand Activity Inventory; OSS, Ottawa Sitting Scale; FS, feet supported; FUS, feet unsupported; R, right; L, left; MCA, middle cerebral artery.
Stationary Hand Task
Trunk peak tangential velocities, trunk pitch angles, and endpoint movement times in the free-trunk condition were similar between groups and arms (Table 2). The trunk was effectively blocked by the harness in both groups (Control <5°, Stroke <1°; Table 2). Endpoint error in the free-trunk condition for the more-affected UL in the stroke group was almost double that of the less-affected arm (Wilcoxon Z = −2.999, P = .003; ES = −0.53) and the nondominant UL of the control group (Mann-Whitney U = 38.000, Z = −2.693, P = .006; ES = −0.50; Table 2).
Secondary Kinematic Variables (Mean [SD]) of the Stationary Hand Task and the Reaching Hand Task a .
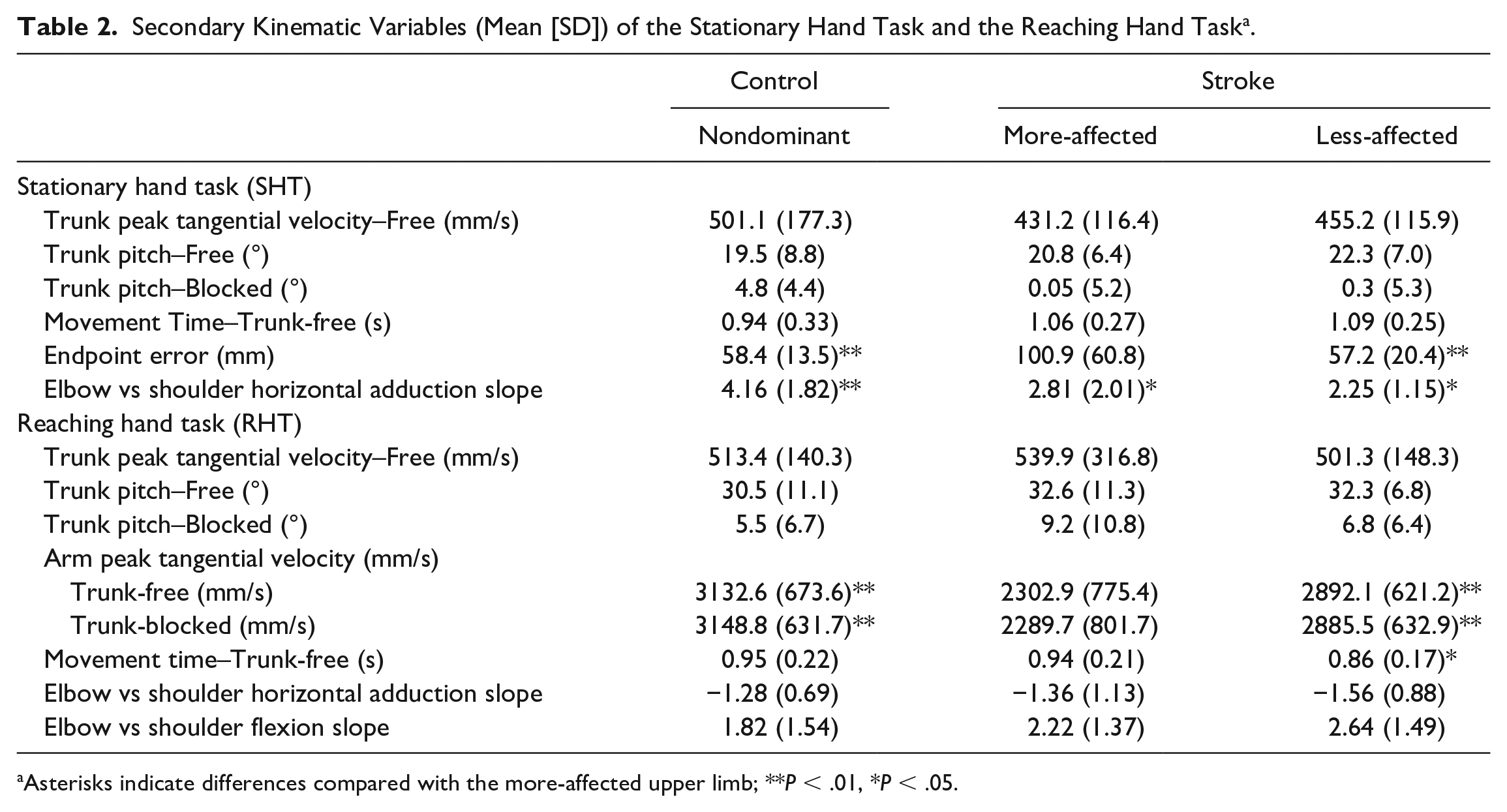
Asterisks indicate differences compared with the more-affected upper limb; **P < .01, *P < .05.
SHT Gain
Individuals with stroke had lower gains for the SHT performed using the more affected UL (0.51 [SD 0.19]) compared with both the less-affected UL (0.68 [SD 0.13]; Wilcoxon Z = −2.929, P = .003; ES = −0.52; Figure 2A) and the nondominant limb of controls (0.72 [SD 0.12]; Mann-Whitney U = 33.00, Z = −2.693, P = .003; ES = −0.51). Gains of the less-affected and control ULs were similar.
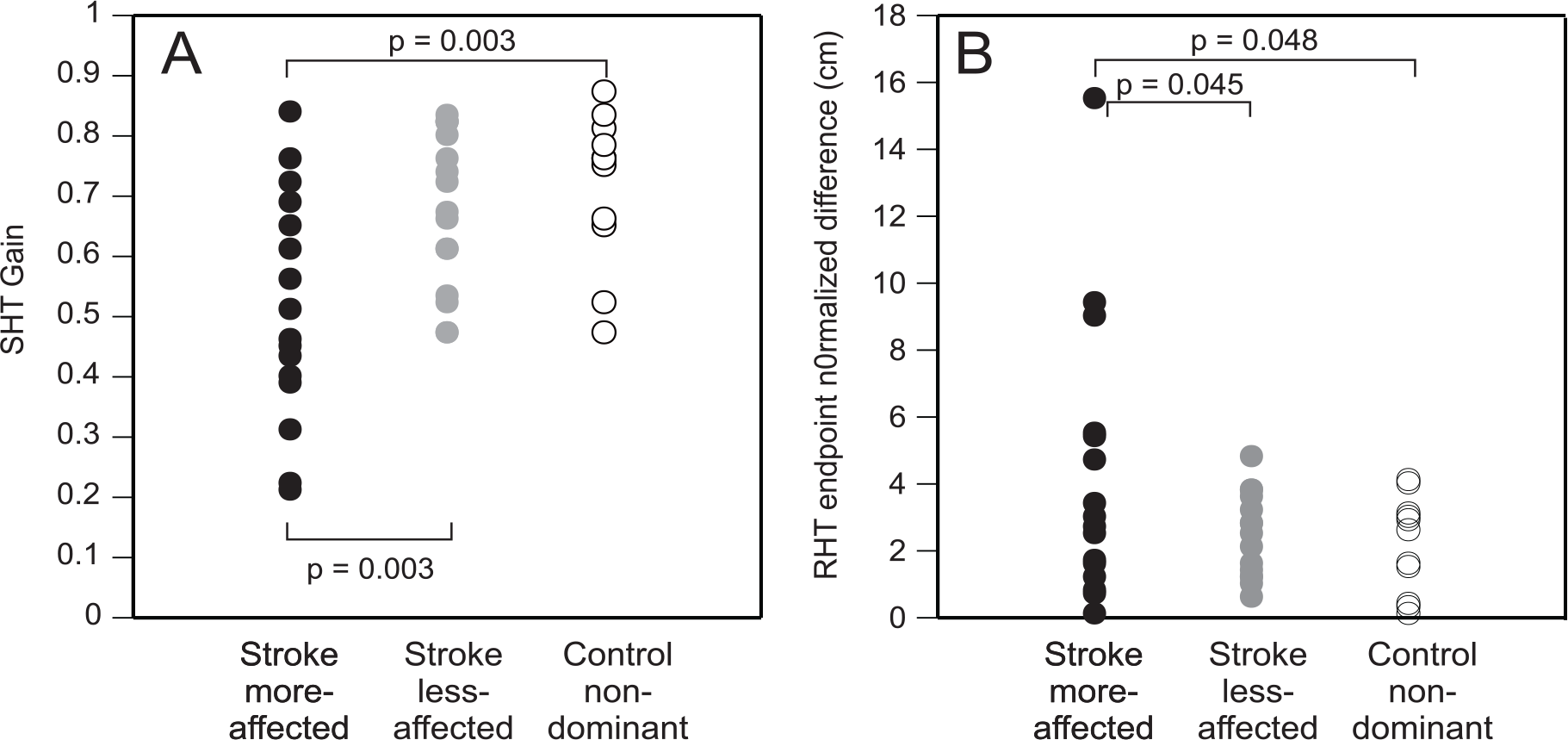
Individual values of the more-affected (black symbols) and the less-affected arms (gray symbols) of the stroke group and the nondominant arm of the control group (open symbols) of the gains of the Stationary Hand Task (SHT, A) and the endpoint normalized difference values of the Reaching Hand Task (RHT, B).
Elbow-shoulder horizontal abd/adduction slopes were lower for both arms of the stroke group compared with controls (F2,1 = 12.081, P = .037; ES = 0.16; Table 2). Gains were not correlated with elbow-shoulder slopes for either arm of the stroke subjects or for the nondominant arm of controls. However, gains were correlated with the range of shoulder horizontal adduction used (ρ = 0.641, P = .025) in controls and with the range of elbow movement used for the less-affected arm of participants with stroke (ρ = −0.508, P = .044).
Reaching Hand Task
As for the SHT, trunk peak tangential velocities and pitch angles in the free-trunk trials were similar between groups for the RHT, and the trunk was effectively blocked by the harness (Table 2). Although reaches with the more-affected UL of the stroke group were slower than the less-affected UL (t15 = 2.174, P = .046), movement time was not different from that of the control group using the nondominant UL (Mann-Whitney U = 58.5, Wilcoxon Z = −1.742, P = .082; Table 2). The arm peak tangential velocity for the trunk-free trials was lower for the more-affected compared with the less-affected UL in the stroke group (Wilcoxon Z = −3.296, P = .001; ES = −0.58) as well as the nondominant UL of controls (Mann-Whitney U = 25.00, Z = −2.999, P = .003; ES = −0.57; Table 2). Similar results were obtained for the trunk-blocked trials, where the arm peak tangential velocity was lower for the more-affected compared with the less-affected UL of the stroke group (Wilcoxon Z = −2.999, P = .003; ES = −0.53) and the nondominant UL of controls (Mann-Whitney U = 25.000, Z = −3.296, P = .001; ES = −0.62).
Endpoint Normalized Difference
The endpoint normalized difference (error) for the more-affected UL (4.24 [SD 4.09] cm) was greater than the error for the nondominant UL of controls (2.36 [SD 1.47] cm; t26 = −2.076, P = .048; ES = −0.42; Figure 2B) and for the less-affected UL (2.48 [SD 1.24] cm; t15 = −2.192; P = .045; ES = −0.52; Figure 2B).
Neither elbow-shoulder horizontal add/abduction slopes nor elbow-shoulder flexion slopes differed between arms (Table 2). Slopes of both relationships tended to be steeper in both arms of subjects with stroke compared with controls suggesting that the task was done with relatively more elbow than shoulder movement. However, neither angle-angle slopes nor joint ranges of motion were correlated with endpoint difference scores in either group or arm.
Correlations With Clinical Scores
In the stroke group, for the SHT with the more-affected UL, individuals with higher gains had higher scores on the FMA and RPSS for both close and far targets (moderate positive correlations) and lower CSI scores (moderate negative correlation; Table 3). Similarly, for the RHT, individuals with less error had higher FMA and RPSS scores for both close and far targets (strong negative correlations) and lower CSI scores (moderate positive correlation). In addition, individuals who made lower errors had higher CAHAI-9 scores and OSS scores for the unsupported condition (moderate negative correlations). Performance on either task was not correlated with demographic factors or sensory deficits.
Results of Correlational Analyses Between Gain and Normalized Endpoint Difference (Error) With Clinical Assessments.
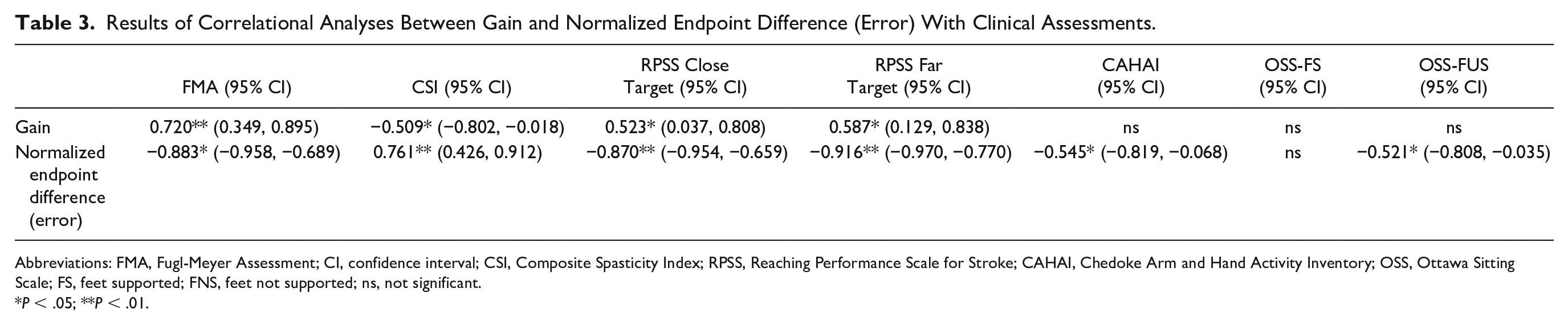
Abbreviations: FMA, Fugl-Meyer Assessment; CI, confidence interval; CSI, Composite Spasticity Index; RPSS, Reaching Performance Scale for Stroke; CAHAI, Chedoke Arm and Hand Activity Inventory; OSS, Ottawa Sitting Scale; FS, feet supported; FNS, feet not supported; ns, not significant.
P < .05; **P < .01.
FMA flexor (Figure 3A) and combined synergy (Figure 3B) subscores were moderately correlated with SHT gains, while combined synergy subscores were moderately negatively correlated with RHT endpoint normalized difference (Figure 3C). However, 10 individuals with full scores on the flexor and combined synergy subscales had a wide range of SHT gains (0.22-0.84) and RHT differences (0.11-9.01).
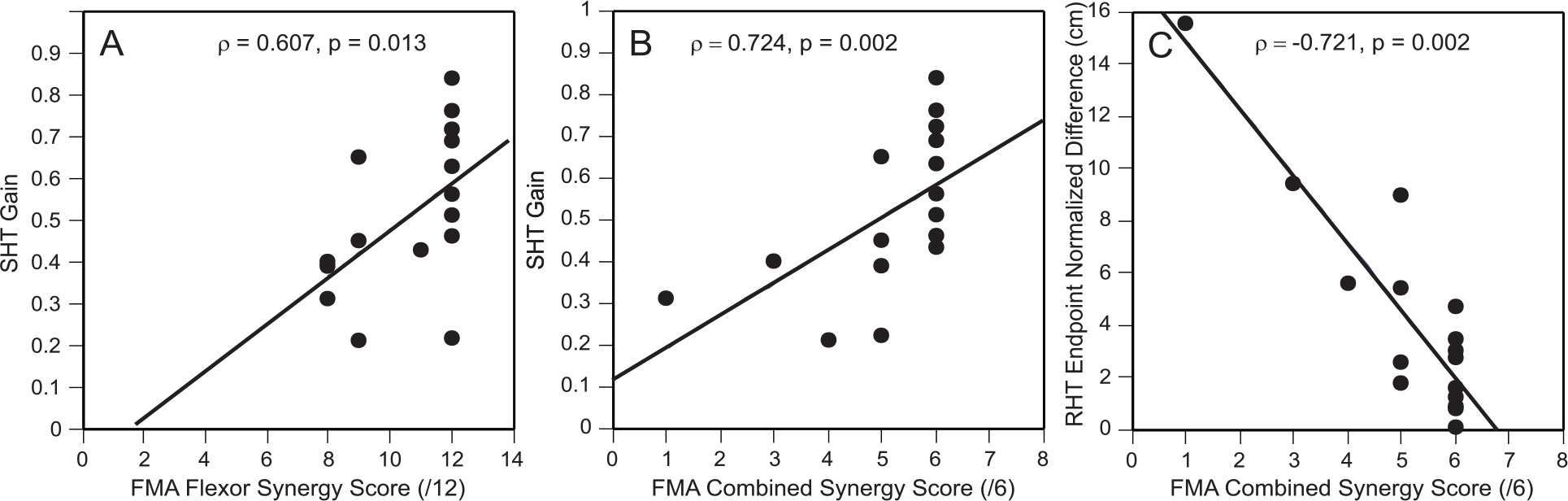
Scatterplots of the correlation between FMA flexor synergy subscore and SHT gain scores (A), FMA combined synergy subscores and SHT gain scores (B), and FMA combined synergy subscores and the RHT endpoint normalized difference (C). The gray open circles refer to correlations involving the SHT gain scores and the black open circles refer to correlations involving the RHT endpoint normalized difference.
Discussion
The capacity for movement adaptability based on motor equivalence was assessed in an arm-trunk coordination task in both arms of individuals with stroke and in the nondominant arm of age-equivalent controls. The tasks chosen to identify motor equivalence in this study involve the coordination of 2 functionally independent synergies.14 -16 One synergy produces arm movement to the target (the reaching synergy) and the other coordinates trunk and arm movements to diminish the influence of the trunk on the arm endpoint trajectory (the compensatory synergy). This coordination applies equally to the SHT and RHT tasks used in this study. Individuals with stroke had impaired performance using the more-affected arm. They could only partially compensate for the added movement of the trunk when trying to hold their hand stationary over a target or while reaching.
Lower gains and endpoint precision of movements made with the more-affected arm indicate that the stroke group had deficits in the ability to find motor equivalent solutions by adapting arm-trunk coordination in both tasks. Although the timing and extent of trunk recruitment was similar between groups (Table 2), individuals with stroke did not completely compensate for the influence of the trunk movement on the endpoint position (SHT) or arm motion (RHT) by adaptive modifications in elbow and shoulder movements. The differences in elbow-shoulder slopes between the more-affected arm of the stroke subjects and the controls suggests that subjects with stroke tended to use more shoulder horizontal adduction than elbow flexion for the SHT and more elbow than shoulder movement for the RHT. However, SHT gains and RHT endpoint differences did not depend on any particular elbow-shoulder interjoint coordination pattern in either group or arm. This supports the Bernsteinian notion that the nervous system does not reproduce a single “optimal” movement pattern to achieve a task but that new task solutions are found each time a movement is made. 5 Our results that the subjects with stroke had deficits in the capacity to adapt UL movement are consistent with those reported previously for reaching tasks9 -13 and extend these observations of reduced adaptability to UL movements involving the trunk.
Factors proposed to contribute to compensatory modifications of UL joint angles in response to trunk motion include sensory feedback (ie, proprioceptive, cutaneous, vestibular), 38 trunk stability, cognitive (movement planning deficits) and motor impairments. Overall, our study participants did not have severe cutaneous and/or proprioceptive deficits (Table 1). In addition, we found no correlations between sensory and proprioceptive assessment scores and gain or normalized error scores. Previous studies have investigated the role of sensation (ie, deafferented patients) 21 and vestibular lesions18,19 on the arm-trunk coordination (SHT) test. Patients without cutaneous sensation and proprioception but who had intact vestibular function maintained their ability to adapt movements using compensatory arm-trunk coordination in the absence of vision. However, patients with unilateral vestibular lesions had marked deficits in adapting arm-joint motion to maintain the hand position or to stabilize the reaching trajectory when trunk motion was imposed.18,19 In addition, disruption of the vestibulospinal system using galvanic vestibular stimulation 39 resulted in low gains and greater endpoint normalized differences in healthy subjects. Thus, vestibular signals evoked by head motion following trunk flexion may play a role in the initiation, maintenance, and modification of compensatory arm-trunk coordination. However, it is unlikely that vestibular insufficiency contributed to the lower scores in our subjects with stroke since only participants with no self-reported vestibular symptoms (as assessed using the Dizziness Handicap Inventory) were included. Nevertheless, a better understanding of the role of the vestibulospinal system in the mediation of the arm-trunk synergy in subjects with stroke may be gained using galvanic vestibular stimulation. Future studies could also further examine the role of minor sensory and/or proprioceptive deficits, not identified by usual clinical scales, on the disruption of movement adaptability.
All participants also had good trunk stability (relatively high scores on the OSS) and had no deficits in temporal coordination of arm and trunk segments. We instructed subjects to place their feet on the ground during the task and no correlation with the OSS score for the feet supported condition was found.
It is unlikely that motor planning deficits played a role in impaired compensatory modifications since participants did not have severe cognitive deficits and gain and endpoint normalized difference measurements on the less-affected side were not different from controls. However, moderate to strong correlations between clinical assessments and gain and endpoint normalized difference scores indicate that motor impairments contributed to the inadequate UL joint modifications in response to trunk movement.
Despite moderate correlations between FMA flexor and combined synergy subscores and SHT and RHT values, a large number of participants without abnormal synergies could have a wide range of SHT gain and RHT difference scores (see Figure 3). This suggests that lower gains and difference scores were more likely due to deficits in motor equivalence but does not rule out the influence of the presence of abnormal synergies.
Clinical Implications
Our results in individuals with stroke are similar to those reported previously for participants with other pathologies such as deafferentation, 21 parkinsonism, 22 and vestibular insufficiency 19 and suggest that movement adaptability and the capacity for motor equivalence is reduced after central nervous system pathology. Previous studies have identified differences in UL and trunk kinematics between healthy controls and individuals with stroke using both the more- and less-affected ULs.33,40-42 However, identifying disorders in coordination and adaptability has received less attention. 43 In the clinic, coordination is assessed primarily by scales that measure the time to complete a series of movements and subjectively score movement quality. 44 Recent studies suggest that the use of time to perform the finger-to-nose test may be a psychometrically sound variable to assess the coordination between movements of the arm joints,44,45 demonstrating good construct validity with kinematic measures of interjoint coordination and the ability to discriminate between mild and moderate-to-severe levels of motor impairment. However, the finger-to-nose test does not identify the contribution of compensatory trunk movement to task performance. The arm-trunk coordination tests used in this study provide indications of not only the ability to functionally integrate movements of different body segments but also of whether the individual is able to use the additional degrees of freedom adaptively. The gain and endpoint normalized difference measures had moderate to strong correlations with clinical measures and could distinguish between the more- and less-affected ULs. The tasks used in our study can be valuable additions to measure intersegmental arm-trunk coordination in addition to the timed component of the finger-to-nose test, in keeping with recent recommendations of the Second Stroke Rehabilitation and Recovery Roundtable to measure UL movement quality based on motor control principles. 46 Clinicians with access to motion analysis equipment can measure the gain using a reduced set of 3 markers on the endpoint, trunk, and target. Alternatively, clinicians can subjectively rate the ability of patients to maintain the hand position (SHT) or make accurate pointing movement (RHT) while moving the trunk. Such a scale of SHT performance has been developed 47 based on observational kinematics 48 and is currently being validated for clinical use.
This study extends previous findings of intersegmental coordination deficits in neurological patients for both the more-affected and less-affected ULs of individuals with stroke. Our results suggest that kinematic measures, obtained during the performance of specific skilled tasks, may be sensitive indicators of higher-order motor control deficits in individuals with stroke. Thus, they can provide complementary information to that provided by clinical motor impairment measures49,50 about higher-order motor control deficits that may limit further recovery of UL skilled movement.
Limitations
The generalizability of our findings is limited due to the small sample size. Specifically, the small sample size may have led to an increase in variability. The moderate-to-strong correlations with clinically assessed UL motor impairment provides some preliminary support for the discriminant validity of these tests. For the RHT, the computation of the shoulder flexion angle with respect to room coordinates rather than to actual trunk position may have resulted in an underestimation of the actual angle. Our sample mainly included individuals with mild-to-moderate stroke. To more completely estimate the ability of the SHT and RHT to discriminate between people with different motor impairment levels, future studies may test individuals with moderate-to-severe levels of UL motor impairment, investigate the relationship between gain measures and the resolution of pathological synergies during recovery, as well as the relationship between improvements in gain measures and UL skill levels. Finally, our methodology did not include caloric or perceptual testing to rule out vestibular or perceptual disorders 51 that may have affected the results.
Conclusions
Individuals with chronic mild-to-moderate poststroke UL hemiparesis had reduced motor adaptability for reaching tasks involving deliberate trunk movement. Results suggest that assessing intersegmental arm-trunk coordination in addition to UL interjoint coordination may provide more information about latent higher-order motor deficits important for motor recovery that may be targeted in therapy to improve UL function.
Footnotes
Acknowledgements
We acknowledge the study participants, Rhona Guberek and Vira Rose for their assistance with patient recruitment and assessment, and Dr Valeri Goussev for programming assistance.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by a Heart and Stroke Foundation Center for Stroke Recovery Catalyst Grant (HSFCSR PT-59562) to M.F. Levin, H. Sveistrup, and H.M. Finestone. M.F. Levin held a Canada Research Chair in Motor Recovery and Rehabilitation. H. Sveistrup was a Career Scientist with Ministry of Long-Term Health and Care, Ontario. S.K. Subramanian was supported by a Focus on Stroke Post-Doctoral Fellowship awarded by the Heart and Stroke Foundation of Canada. K. Haentjens received a summer studentship from the Canadian Stroke Network.
