Abstract
Background. Few studies have addressed early out-of-bed mobilization specifically in acute intracerebral hemorrhage (ICH) patients. Patient benefit in such cases is unclear, with early intervention timing and duration identical to those in standard care. Objective. We investigated the efficacy of an early mobilization (EM) protocol, administered within 24 to 72 hours of stroke onset, for early functional independence in mild-moderate ICH patients. Methods. Sixty patients admitted to a stroke center within 24 hours of ICH were randomly assigned to early mobilization (EM) or standard early rehabilitation (SER). The EM group underwent an early out-of-bed mobilization protocol, while the SER group underwent a standard protocol focusing on in-bed training in the stroke center. Intervention in both groups lasted 30 minutes per session, once a day, 5 days a week. Motor subscales of the Functional Independence Measure (FIM-motor; primary outcome), Postural Assessment Scale for Stroke Patients, and Functional Ambulation Category (FAC) were evaluated (assessor-blinded) at baseline, and at 2 weeks, 4 weeks, and 3 months after stroke. Length of stay in the stroke center was also recorded. Results. The EM group showed significant improvement in FIM-motor score at all evaluated time points (P = .004) and in FAC outcomes at 2 weeks (P = .033) and 4 weeks (P = .011) after stroke. Length of stay in the stroke center was significantly shorter for the EM group (P = .004). Conclusion. Early out-of-bed mobilization via rehabilitation in a stroke center, within 24 to 72 hours of ICH, may improve early functional independence compared with standard early rehabilitation. Clinical Trial Registration: NCT03292211.
Introduction
In an intensive care unit (ICU), early mobilization, implementing mobility within 72 hours of ICU admission, is used with the intention of maintaining or restoring musculoskeletal strength and function, and potentially improving overall functionality.1,2 Recently, early mobilization was defined as early out-of-bed activities of daily living (ADLs), based on the concepts of very early mobilization in stroke patients 3 and associated enhanced functional outcomes.4-6 Very early mobilization with higher-frequency sessions in an out-of-bed mobility-based training protocol within 24 hours of stroke onset may be associated with a worse outcome at 3 months poststroke,7,8 but most studies examined the beneficial effects of early mobilization commencing within 24 to 72 hours of stroke.4,9-12 However, early mobilization within 24 to 72 hours might also worsen any poststroke hypertension and increase the risk of rebleeding, resulting in early deterioration after acute intracerebral hemorrhage (ICH). 13 Most clinicians harbor greater concerns about the early mobilization of patients with hemorrhagic rather than ischemic stroke.14,15 Furthermore, ICH is associated with a different pathophysiology and acute management strategy than cerebral infarction, and it is possible that early mobilization may have different effects in patients with ICH rather than ischemia. However, there have been relatively few findings in stroke intervention trials of early mobilization involving participants with hemorrhagic stroke.16,17 Most previous studies did not stratify for stroke subtype at randomization or prespecify expected subgroup effects. 18 Therefore, the evidence base for early mobilization in hemorrhagic stroke patients has been weak.
Physiologically, early mobilization may have benefits, such as fewer complications from immobility, maintenance of lean body mass, attenuation of secondary muscle weakness, and reduced loss of cardiovascular fitness due to stroke-related inactivity. 19 Additionally, current concepts of biological recovery after brain injury suggest a narrow window of opportunity for brain plasticity and repair. 20 Also, if the brain does indeed remodel itself, based on experience, 21 then early task-specific training may well contribute to improved recovery. 22 Preclinical studies showed that early rehabilitation promoted the growth of neuronal dendrites and astrocyte processes, and increased astrocyte density after ICH.23,24 Moreover, early enhanced rehabilitation promotes dendritic complexity, reduces the number of degenerating perihematoma neurons, and causes changes in growth factors that aid in neuroprotection but do not change iron toxicity and inflammation after ICH.25,26 Although the exact mechanism by which early mobilization improves outcomes is not known, the aforementioned findings provide a biological basis for early training in humans after ICH.
One previous study specifically examined early mobilization within 3 days of spontaneous ICH: patients were directly admitted to neurology wards or rehabilitation units, and none of them had been admitted to stroke centers or ICUs. 27 It is not uncommon for ICH patients to be managed in stroke centers or ICUs rather than general units; however, most studies investigating early mobilization in stroke centers or ICUs tended to focus on cerebral ischemia.4,28-30 To test whether the very early mobilization protocol might be widely applicable to patients with stroke, the inclusion criteria in AVERT (A Very Early Rehabilitation Trial) were broad, and included lack of restriction regarding stroke subtype (infarct or hemorrhage). 7 However, the AVERT protocol was characterized not only by early mobilization within 24 hours of stroke onset but also by a significantly greater frequency (≥3 times) and total duration of mobilization sessions than the usual care protocol. 7 Because there is insufficient evidence to recommend intensive training, such as longer session duration or higher frequency intervention within the first few days of stroke, 11 and considering the staff workload involved with higher frequency intervention, it is important to understand whether early mobilization improves outcomes, even if intervention time and daily session duration are similar to those in standard care. Therefore, we conducted a study with stringent selection criteria and rigorously controlled the intensity of early mobilization to determine the merits of early mobilization in poststroke rehabilitation, specifically in ICH patients during the acute period.
In previous trials of early mobilization, most investigators used the modified Rankin Scale (mRS) 3 months after stroke as a primary outcome measure; further, no progressive information during the acute phase was available, and the data interpretability and implications of mRS classification as dead or dependent (mRS score 3-6) versus independent (mRS 0-2) were limited.31,32 Among ADL measures for stroke patients, the Functional Independence Measure (FIM) is a widely used functional performance instrument developed specifically for inpatient acute rehabilitation. 33 Therefore, the aim of this randomized, controlled study was to investigate if an early out-of-bed mobilization protocol started within 24 to 72 hours of stroke onset (compared with an early standard rehabilitation protocol), and with intervention time and daily session duration similar to those in standard care, would lead to improvements in functional independence measures during the early period in patients with acute ICH. We hypothesized that early mobilization rather than early standard rehabilitation protocol in a stroke center, but with intervention time and daily session duration similar to those in standard care, would result in greater benefits in early functional outcomes.
Methods
Study Design
This was a prospective, assessor-blinded, randomized controlled trial with 2 parallel groups monitored for 3 months. The study was conducted at the stroke center of the National Taiwan University Hospital (NTUH) in Taipei, Taiwan, from September 2017 to November 2018. ICH stroke patients admitted to the stroke center were screened and consecutively enrolled after meeting the eligibility criteria for intensive antihypertensive treatment (primarily intravenous nicardipine infusion). 34 The study was approved by the institutional review board of NTUH (201706073RINB) and registered at ClinicalTrials.gov (trial registration ID: NCT03292211). Written informed consent was obtained from all participants before randomization. All research procedures were conducted in accordance with ethical standards of the Declaration of Helsinki.
The primary endpoint was total score on the motor subscale of the FIM (FIM-motor) assessed 3 months after stroke. To the best of our knowledge, no previous study has investigated the effect of early mobilization after acute stroke on the FIM-motor scale. Study sample size calculations were based on a lower estimate of 17 points for minimum clinically important difference (MCID) in FIM-motor assessment between the 2 groups at all follow-up points. 35 Twenty-seven subjects per group would be required, assuming a standard deviation of approximately 30% in a 2-tailed test,36,37 with an α level of .05 and a power level of 80%. A total of 60 patients were included to allow for a dropout rate of approximately 10%.
Patients and Recruitment Process
Patients presented to the stroke center at NTUH ≤24 hours after ICH were considered for the study. Inclusion criteria were: a first-time primary ICH with unilateral hemiparesis/hemiplegia confirmed via computed tomography; no contraindications to mobilization (early intervention) within 72 hours of stroke onset (based on the medical team’s clinical judgment, systolic blood pressure (SBP) <160 mm Hg at rest, resting heart rate <130 bpm, oxygen saturation >92% without supplementation, and no hydrocephalus before intervention); a National Institutes of Health Stroke Scale (NIHSS) score of <20 at admission 38 ; complete prestroke independence in ADL; and age 20 to 80 years.39-41
Exclusion criteria were secondary ICH due to trauma, surgery, hemorrhagic transformation from stroke, or an underlying mass; living alone and unable to complete the baseline survey because of serious aphasia, language difficulties, or cognitive deficit; other medical conditions, including severe heart failure, acute coronary syndrome, active gastrointestinal blood loss, or lower-limb disorders, preventing early mobilization; and an inability to provide informed consent or previous enrollment in another intervention trial. In addition, we excluded patients with rapid early deterioration of symptoms within 24 hours of stroke, that is, patients with a ≥4-point change in NIHSS score 42 ; those who underwent immediate surgery; and those with a concurrent diagnosis of rapidly deteriorating disease (eg, terminal cancer).
Recruitment Process and Randomization
Study participants were randomized into 1 of 2 groups and underwent different rehabilitation protocols: early mobilization within 24 to 72 hours of stroke onset (EM group); or standard early rehabilitation (SER group). A permuted-block randomization procedure at a 1:1 ratio and with a block size of 4 was used to generate an allocation sequence. The details of group allocation were placed into opaque, sealed envelopes secured by the research staff. As each patient entered the trial, the next envelope in the sequence was opened, indicating which treatment the patient would receive. Assessors collecting the study data were unaware of treatment allocations throughout the trial.
Interventions
Early Mobilization
Participants in the EM group underwent the early mobilization protocol in the stroke center, within 24 to 72 hours of ICH onset; the protocol consisted of out-of-bed mobilization (including unsupported sitting on the edge of bed, standing with/without hand support, and stepping while standing) 43 as soon as practicable, and based on the therapist’s ability to provide maximum assistance and fully guarantee patient safety. All physical therapies in the EM and SER groups were provided for 5 days a week, with 1 session per day and 30 minutes per session. 44 The entire EM intervention was carried out within this time frame, and the overall rehabilitation duration did not differ between groups.
The EM protocol in the stroke center involved out-of-bed, task-specific activities. The first out-of-bed activities consisted mainly of sitting unsupported on the edge of the bed, via rolling and then sitting up for a minimal duration (the first sitting duration was 15 minutes), 45 and then standing with/without hand support for approximately 3 to 5 minutes whenever possible. The use of a lifter was not allowed. Initially, if a patient was able to stand for only short periods, or was even unable to stand, the first EM activity was modified to sitting on the side of the bed for a total of 15 to 20 minutes whenever possible. However, it was necessary to perform the standing intervention once a patient had acceptable leg or trunk strength against gravity, or once a patient could perform this intervention with a device or with assistance from the therapist. While sitting on the edge of the bed, sitting-balance activities were used to stimulate trunk control. Furthermore, while in the standing position, standing programs focused not only on attempting to stand well but also on prewalking activities, such as weight shifting, stepping in place, and side steps along the bed. Close monitoring of blood pressure and heart rate was conducted.
While sitting for the first time, patients with any signs of low tolerance, based on neurologic worsening (of current or new neurologic deficits), vagal reactions (bradycardia or nausea), SBP >180 mm Hg, or a >30 mm Hg symptomatic decrease in blood pressure, were put back to bed. Rest was allowed at any time, if needed, but was not considered when calculating the total training duration. Three physical therapists (PTs) carried out the treatment and intervention. These therapists were provided with explanations of the EM protocol and practiced the protocol for 2 weeks before implementing it in the study.
Standard Early Rehabilitation
SER focused on training in bed and was performed in line with physical therapy provided at the NTUH stroke center, and based on international guidelines for the traditional approach to physical therapy for ICU patients.46,47
In the SER group, out-of-bed mobilization was started as soon as possible after ICU discharge. While intending to restrict early out-of-bed mobilization, sitting upright in bed up to an angle of 45 degrees was acceptable. The SER program consisted of an intervention regularly used at the NTUH stroke center. The intervention was performed by a PT, and each patient’s relatives, with guidance from the PT, and comprised: bed exercises, including joint range-of-motion, bridge, straight leg-raising, and stretching exercises; facilitation techniques; and functional training, in which patients were instructed to practice repetitive and systematic tasks, such as rolling (as per the Bobath concept) or sitting with support in bed.48,49 A PT conducted the standard progression of exercises, as subjects’ muscle strength improved; the progression comprised increasing the number of repetitions and range of movement, adjusting the speed of movement, and decreasing the rest interval between exercises. Functional exercises, based on motor relearning, were also adjusted and implemented according to the ability and progress of each participant.
Outcome Measures
After randomization, a baseline assessment of demographic and disease characteristics, and a medical history were conducted for each participant. Outcome measures were collected at baseline (preintervention), and at 2 weeks, 4 weeks, and 3 months after stroke onset. The primary outcome variable was FIM-motor score for ADL function at 3 months after stroke onset. 50 Tasks and capacities in the motor domain of the FIM were self-care tasks (eating, grooming, bathing, upper-body dressing, lower-body dressing, and using the toilet), sphincter control (bladder and bowel management), transfers (from bed or a chair to the toilet, bath, or shower), and locomotion ability (walking or wheelchair mobility, and stair climbing). Each item was rated from 1 (requiring complete assistance) to 7 (completely independent). The sum of results for the 13 items yielded a motor score with a range of 13 to 91. Post hoc outcomes from the FIM-motor assessment were limited to the “self-care tasks”, and “transfers” plus “locomotion ability” subscales (maximum scores of 42 and 35, respectively) to understand whether EM alone influenced lower-extremity function.
Secondary outcomes included the Postural Assessment Scale for Stroke Patients (PASS), 51 the Functional Ambulation Category (FAC), 52 and the mean duration of stay in the stroke center. The PASS contains 12 four-level items of varying difficulty with a maximum score of 36 (0-36, with the highest score being best) for assessing a patient’s ability to maintain or change a given lying, sitting, or standing posture. Psychometric properties of the PASS have been reported as satisfactory in stroke patients during the first 3 months after stroke. 51 The FAC further assesses ambulatory ability, with required support during gait, without considering the use of aids and is rated on a 0 (nonfunctional) to 5 (independent) scale. Patients can perform the test with a cane but without an orthosis. Navigating stairs and indoor walking for a 10-m distance are needed to discern between categories 4 and 5. The test has demonstrated good reliability, validity, and sensitivity to change during the first 6 months after stroke. 53 The duration of stay in the stroke center was defined as the time from admission to being declared medically fit for discharge by the senior ICU clinician. All adverse events, including neurologic deterioration, hypotension, falls, and line dislodgements, during the intervention periods were recorded.
Statistical Analyses
Results were analyzed using Statistical Package for the Social Sciences version 22.0 (IBM SPSS Statistics, Armonk, NY, USA). We assessed all patients who underwent at least 1 treatment session (intention-to-treat principle), and intent-to-treat analyses were used for all outcome comparisons. Descriptive statistics were expressed as mean (standard deviation, SD) for continuous variables, and as median (interquartile range, IQR) for ordinal variables. Number (n) and percent (%) were used for categoric variables. All continuous variables were first assessed for normality using the Shapiro-Wilk test and transformed when necessary to meet the assumption of normal distribution. Descriptive statistics were used to define relevant patient characteristics.
For total FIM-motor and PASS scores, we explored the associations between potential factors and outcomes of interest using a linear mixed model. This analytical model factors in each participant’s varying initial performance and allows for adjustments to be made by inserting, into the analytical model as covariates, initial values from the evaluation scales that correspond to different dependent outcomes. In the preliminary regression analysis, we treated group (EM vs SER), time (baseline, 2, weeks, 4 weeks, and 3 months poststroke), and the group × time interaction term as fixed effects, and baseline of the outcome of interest as a random effect. If no statistically significant difference was found in the interaction term, the subsequent analysis would not put the interaction term in the model. Additional analyses to estimate scores for the FIM-motor self-care activities and transfers plus locomotion subscales were completed to improve interpretation of the results. The Mann-Whitney U test was used to identify any significant differences in FAC measurements between the 2 groups. The independent-samples t test was used to identify any significant differences in mean duration of stay in the stroke center. All hypothesis tests used a significance level of P < .05.
Results
A CONSORT (Consolidated Standards of Reporting Trials) diagram is shown in Figure 1. Sixty patients were randomized into 2 groups, with equal numbers (n = 30) in each group. After 3 months’ follow-up, there were 2 dropouts in each group due to family reasons, but all patients were included in the final analysis. Participant characteristics at study enrollment are shown in Table 1, which summarizes demographic and clinical characteristics and outcome measures, which were equivalent in the 2 groups except for 1 outcome measure: mean time to first mobilization after symptom onset (defined as time elapsed from admission to achieving out-of-bed unsupported sitting) was 51.60 hours (SD 14.15) in the EM group and 135.02 hours (SD 33.05) in the SER group (P < .001). At baseline, the 2 groups were similar regarding age, height, weight, gender, hematoma volume, stroke site, lesion location, initial NIHSS score, NIHSS upper-extremity and lower-extremity subscale scores, ICH score 39 at admission, initial FIM-motor score, PASS score, and FAC level. In addition, the median number of treatment sessions per group during the acute phase was similar: EM 7.0 (range 2-18) versus SER 6.5 (range 2-19) (P = .988 between groups). None of the participants in either group experienced adverse effects, including neurologic deterioration, hypotension, falls, or line dislodgements, during early rehabilitation or mobilization.
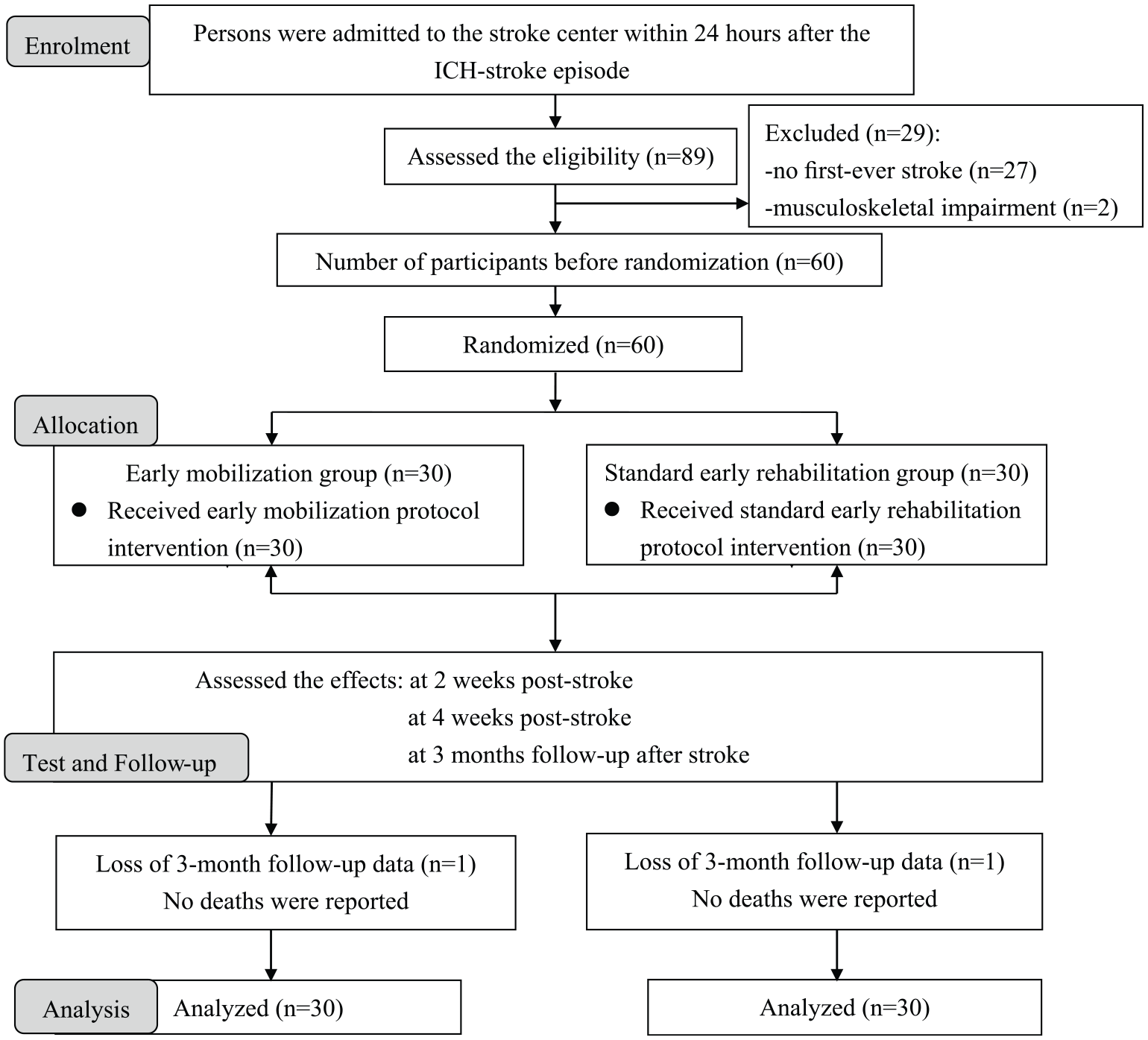
CONSORT diagram.
Participant Characteristics.
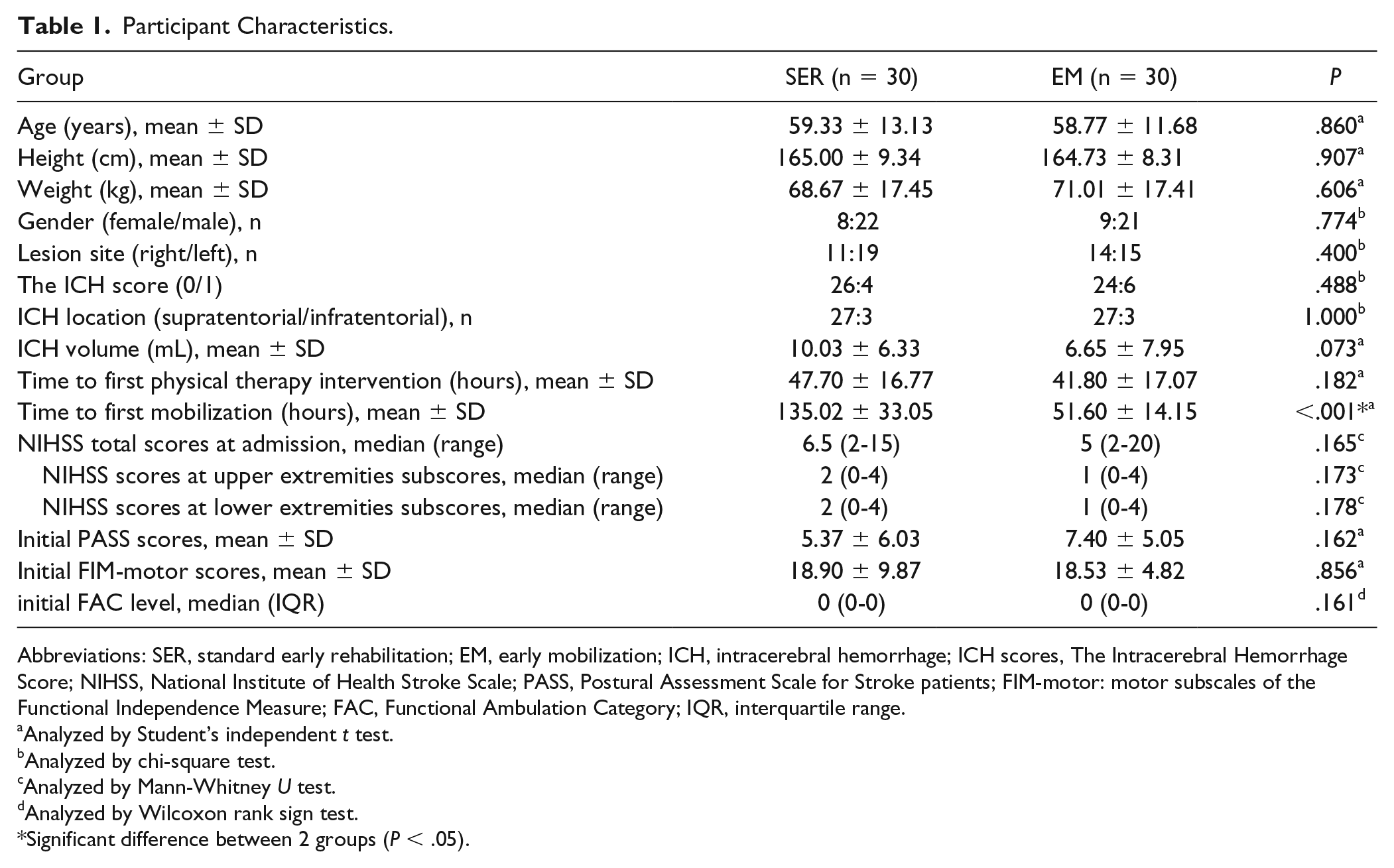
Abbreviations: SER, standard early rehabilitation; EM, early mobilization; ICH, intracerebral hemorrhage; ICH scores, The Intracerebral Hemorrhage Score; NIHSS, National Institute of Health Stroke Scale; PASS, Postural Assessment Scale for Stroke patients; FIM-motor: motor subscales of the Functional Independence Measure; FAC, Functional Ambulation Category; IQR, interquartile range.
Analyzed by Student’s independent t test.
Analyzed by chi-square test.
Analyzed by Mann-Whitney U test.
Analyzed by Wilcoxon rank sign test.
Significant difference between 2 groups (P < .05).
Table 2 shows total FIM-motor score, FIM-motor self-care tasks subscale score, FIM-motor transfers plus locomotion subscale score, and total PASS score in the 2 groups at baseline, and at 2 weeks, 4 weeks, and 3 months after stroke. In the preliminary regression analysis for total FIM-motor scores, including group, time, and the group × time interaction term as fixed effects, and baseline FIM-motor scores as a random effect, no statistically significant difference was found in the interaction term (F = 0.004, P = .949). Similar results were noted for the FIM-motor self-care tasks subscale (F = 0.231, P = .631), for the FIM-motor transfers plus locomotion ability subscales (F = 0.016, P = .901), and for PASS score (F = 0.235,P = .628). Thus, subsequent analysis did not include the interaction term in the model. We found that group effects were significant at all evaluation times for FIM-motor total score (F = 8.859, P = .004), FIM-motor self-care tasks subscale score (F = 8.868, P = .005), and FIM-motor transfers plus locomotion ability subscale score (F = 4.997, P = .029). However, no significant group effect was observed for PASS scores at all evaluation times, although PASS scores did improve significantly over time (time effect: F = 130.66, P < .001; Table 2).
The Total Scores of the FIM-Motor and PASS Measures at Baseline and Each Follow-up Assessment in Both Groups. a
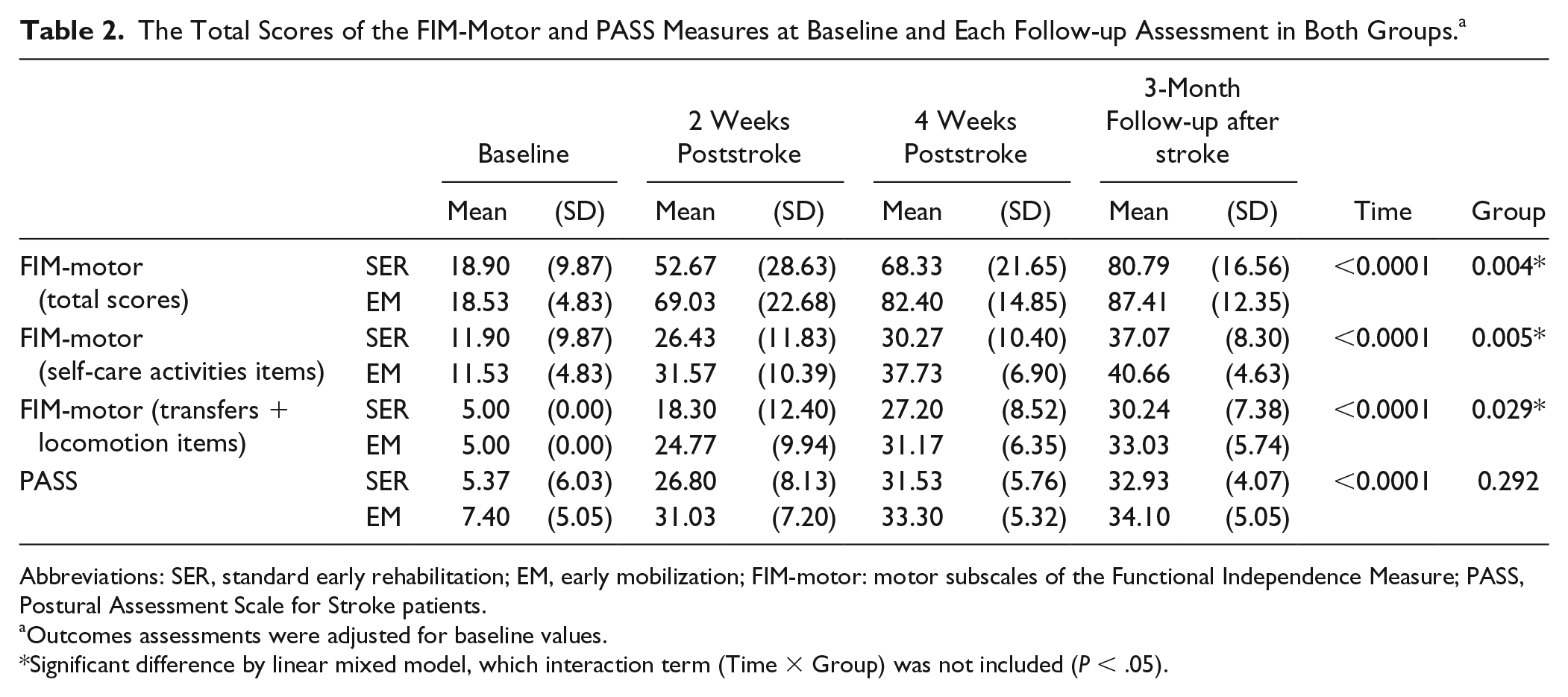
Abbreviations: SER, standard early rehabilitation; EM, early mobilization; FIM-motor: motor subscales of the Functional Independence Measure; PASS, Postural Assessment Scale for Stroke patients.
Outcomes assessments were adjusted for baseline values.
Significant difference by linear mixed model, which interaction term (Time × Group) was not included (P < .05).
The other secondary outcome measure, FAC, showed a significant increase in independent walking in the EM group at 2 weeks (P = .033) and 4 weeks (P = .011) after stroke (Table 3). In addition, mean duration of stay in the stroke center was significantly less in the EM group than SER group (86.22 ± 41.31 vs 119.2 ± 44.44 hours; P = .004).
The Level of the FAC Measure at Each Follow-up Assessment in Both Groups.
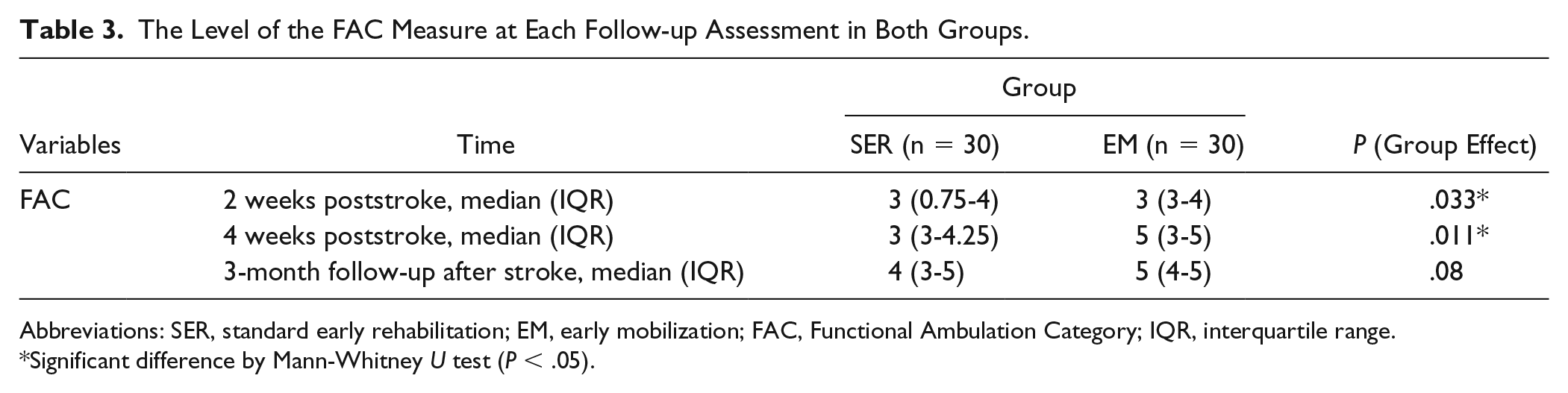
Abbreviations: SER, standard early rehabilitation; EM, early mobilization; FAC, Functional Ambulation Category; IQR, interquartile range.
Significant difference by Mann-Whitney U test (P < .05).
Discussion
This randomized controlled trial is, to the best of our knowledge, the first to test an EM protocol with standard intervention time and session frequency, as in the standard care protocol (dose-matched therapy), specifically in acute ICH patients in a stroke center and starting within 24 to 72 hours of stroke onset. Our results demonstrated that the EM versus SER protocol produced significantly greater FIM-motor scores, and may accelerate ADL restoration within 3 months of ICH. Patients who underwent the EM rather than SER protocol after ICH also had a shorter duration of stay in the stroke center and reported significantly greater walking independence within 4 weeks of stroke onset.
Regarding evidence supporting early mobilization and specific to ICH patients, one previous study showed that ICH patients admitted to neurology wards or rehabilitation units within 48 hours, and who underwent very early rehabilitation, had significantly greater ADL independence 6 months after stroke, compared with patients who started rehabilitation >7 days after stroke. 27 However, the aim of the current study was to compare the efficacy of early rehabilitation, implemented within 48 hours of stroke, with rehabilitation implemented ≥7 days post-stroke, rather than to compare 2 different protocols started at the same time. 27
In the prespecified dose-response analysis of AVERT, trends disfavored “early” (time from stroke onset to first out-of-bed mobilization), more intensive rehabilitation group with hemorrhagic stroke when compared with usual care, which resulted in improved functional outcomes. 54 However, a follow-up analysis suggested that the specific frequency and duration of mobilization sessions needed to be optimized to avoid the harmful effects of aggressive early mobilization. 54 Furthermore, patients in AVERT who had ICH were more likely to be excluded than recruited. 17 Moreover, the definition of “early” differed from that in our study: our mean time to first mobilization in the EM group was 51.6 hours, which was longer than the 22.4 hours for the usual care group in AVERT. 7 Since the starting time or parameters of the intervention protocol, including treatment duration and frequency of out-of-bed activities, differed from those in previous studies,17,55,56 and may have affected the outcomes independently, direct comparison is difficult. However, our results allow the design of characteristics for early intervention during 24 to 72 hours after ICH; these characteristics can be used in treatment programs to improve the efficacy of mobilization protocols in stroke centers.
In other critically ill patients in ICUs, early mobility has been linked to improved functional status. 57 In the present study, total FIM-motor score and FIM-motor subscale score for self-care tasks highlighted important benefits within 3 months after stroke: That is, an EM protocol started within 24 to 72 hours of stroke onset promoted greater ADL independence in mild-moderate acute ICH patients. Indeed, functional recovery from a stroke event occurs through a combination of spontaneous and experience-dependent processes. 20 In our study, active and unsupported sitting out of bed constituted early mobilization, with the opportunity to stand. The EM protocol used in our study may enhance motor experiences or provide greater stimulation than an SER protocol. Moreover, we found early recovery of walking independence in the EM group, for which a similarly significant group difference was evident regarding FIM-motor subscale score for transfers plus locomotion ability at 4 weeks after stroke. This suggests that patients who receive an EM protocol rather than standard care may be more active once transferred to an ordinary ward. Some previous results showed that ambulation distance at ICU discharge was greater in patients who received early active mobilization in the ICU, compared with patients for whom mobilization was started after ICU discharge.58,59 In addition, shorter ICU stays have been reported in adult patients with primary neurologic injury receiving progressive EM therapy. 60 In the present study, while the EM protocol was implemented to reduce stroke impact, associated reductions in duration of stay in the stroke center were also observed.
Besides the lack of significant differences in postural stability at all evaluation time points, we also found no differences between the 2 groups in FAC-level outcomes at 3 months poststroke. We cannot rule out the possibility that other intensive training at the sub-acute stage might have achieved similar improvements in walking ability. However, the ICH patients in our study had relatively low NIHSS and ICH scores at admission. Lower NIHSS scores indicate fewer neurologic deficits, and a previous study found that paresis severity was a predictor of mobility 30 days after stroke. An ICH score <3 predicts a good outcome at 30 days, which is defined as an mRS score of ≤2. 39 Consistent with the above, most participants in the present study had good posture control after the acute phase and had walking recovery at 3 months poststroke; thus, it may have been difficult to detect further progress regarding posture control outcomes because of a ceiling effect. Another probable reason is that the study was underpowered to detect such long-term differences in walking ability; each group was of relatively small size.
Thus, a limitation of the present study was that the small number of patients may have limited statistical power, thereby increasing the risk of type II errors. The estimated population size was 54 participants (based on known evolution of functional independence on the FIM-motor scale) required to observe a 17-point difference between groups. However, our EM protocol, which moved ICH patients out of bed within 24 to 72 hours of stroke onset, was better defined due to small patient numbers. It would now be useful to explore impact of the EM protocol in specific ICH populations, who may have been excluded from previous trials. As another limitation, the mechanism by which the EM protocol is effective during the acute stage cannot be adequately explained because outcomes in the present study were determined at a functional level. Further, this sample of study participants does not reflect the whole ICH population in ICUs. Our sample was characterized by patients in a stroke center with mild stroke, high motor control, a relatively young age, and a relatively low mortality rate. As such, further related studies are needed with more objective and larger designs and covering a larger spectrum of disabilities.
In conclusion, early mobilization in a stroke center within 24 to 72 hours of stroke onset, specifically in patients with mild or mild-moderate ICH, and using an EM protocol with standard intervention time and session frequency, may be more effective than standard early rehabilitation in achieving functional independence within 3 months of stroke.
Footnotes
Acknowledgements
The authors express gratitude to the Statistical Center of NTUH. We also thank staff at the NTUH stroke center (4D1) for supporting this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the National Taiwan University Hospital (NTUH.107-003774).
