Abstract
Background and Objective. The question of the best therapeutic window in which noninvasive brain stimulation (NIBS) could potentiate the plastic changes for motor recovery after a stroke is still unresolved. Most of the previous NIBS studies included patients in the chronic phase of recovery and very few in the subacute or acute phase. We investigated the effect of transcranial direct current stimulation (tDCS) combined with repetitive peripheral nerve stimulation (rPNS) on the time course of motor recovery in the acute phase after a stroke. Methods. Twenty patients enrolled within the first few days after a stroke were randomized in 2 parallel groups: one receiving 5 consecutive daily sessions of anodal tDCS over the ipsilesional motor cortex in association with rPNS and the other receiving the same rPNS combined with sham tDCS. Motor performance (primary endpoint: Jebsen and Taylor Hand Function Test [JHFT]) and transcranial magnetic stimulation cortical excitability measures were obtained at baseline (D1), at the end of the treatment (D5), and at 2 and 4 weeks’ follow-up (D15 and D30). Results. The time course of motor recovery of the 2 groups of patients was different and positively influenced by the intervention (Group × Time interaction P = .01). The amount of improvement on the JHFT was greater at D15 and D30 in the anodal tDCS group than in the sham group. Conclusion. These results show that early cortical neuromodulation with anodal tDCS combined with rPNS can promote motor hand recovery and that the benefit is still present 1 month after the stroke.
Introduction
Although the usefulness of classical physical therapies to promote the recovery of upper limb function after a stroke is well established, 1 additional therapeutic approaches are clearly needed to enhance it. In this context and based on functional imaging data providing insight into both the pathophysiology of stroke-induced cortical network disturbances and a better understanding of the mechanisms underlying neuromodulation, noninvasive brain stimulation (NIBS) appears to be an interesting option as an add-on intervention.
Among NIBS techniques, transcranial direct current stimulation (tDCS) is a safe,2,3 painless, and inexpensive method that induces prolonged cortical excitability modifications in humans through a combination of glutamatergic and polarity-driven alterations of resting membrane mechanisms, resulting in LTP/LTD-like synaptic changes. 4 These changes are NMDA-receptor dependent and mediated by secretion of brain-derived neurotrophic factor in rodent M1 slices. 5
Several studies, mainly performed in the chronic or subacute recovery phase after a stroke, have reported a tDCS-induced functional motor improvement of the paretic hand (chronic6-9 and subacute9-11). Various tDCS strategies that follow a model of interhemispheric rivalry between the damaged and the intact hemispheres have been tested to promote motor recovery and suggest that motor recovery might be facilitated by upregulating the excitability of the affected motor cortex through anodal tDCS6,7,12 or by downregulating the excitability of the intact motor cortex through cathodal tDCS,6,7,12 or, more recently, by the use of bihemispheric anodal/cathodal tDCS.13-17 Besides NIBS, repetitive peripheral nerve stimulation (rPNS) has been proposed to enhance motor recovery: in chronic stroke patients, 2 hours of stimulation of the median nerve, 18 the median + ulnar nerves combined, 19 or the median + ulnar + radial nerves combined 20 was able to transiently improve the paretic hand performance and this was also observed in the acute or subacute phase. 21 Finally, the combination of central and peripheral stimulation has been successfully used to enhance motor recovery in chronic poststroke patients. 22
However, the question of the best NIBS strategy remains open. Some studies have tried to address it7,23,24 but have failed to reach a consensus probably because of the different methodologies used (crossover or parallel studies, single or multiple sessions) and different outcome measures.
The question of the best therapeutic window in which NIBS could potentiate the plastic changes for motor recovery is still unresolved. Animal models show that, after focal ischemic damage, there is a brief, approximately 3- to 4-week window of heightened plasticity. 25 Analogously, almost all recovery from impairment in humans occurs in the first 3 months after stroke, 25 although functional motor improvement can be obtained later.
In the present double-blind controlled pilot study, we investigated the effect of 5 daily sessions of anodal tDCS, applied over the ipsilesional primary motor cortex (M1) combined with rPNS, on the time course of the paretic hand motor recovery in patients in the acute phase of recovery after an ischemic stroke. Behavioral outcome measures associated with transcranial magnetic stimulation (TMS) study were recorded at baseline and repeated at the end of the intervention, at 2 and 4 weeks’ follow-up.
The rationale for targeting ipsilesional motor regions was that previous functional neuroimaging studies had shown reactivation of intact portions of the ipsilesional motor cortex to be associated with better outcome after stroke.26,27 The anodal-stimulation-induced behavioral gains in chronic stroke are associated with a functionally relevant increase in activity within the ipsilesional primary motor cortex. 28
We chose the radial nerve because the aim was to improve the recovery of wrist extension, which is crucial for the recovery of skilled hand prehension.29,30 Our hypothesis was that NIBS intervention proposed during the acute phase after a stroke, as add-on treatment to the classical rehabilitation program, could potentially enhance motor recovery.
Patients and Methods
Patients
Inclusion criteria were the following: first-ever, single, unilateral hemispheric ischemic stroke within 4 weeks with mild to moderate motor deficit. Exclusion criteria were the following: cortical infarct with large hand/wrist M1 involvement, major depression or other severe psychiatric comorbidity, alcohol abuse, TMS contraindications. 31 The study was performed in accordance with the Declaration of Helsinki, registered in the ClinicalTrials.gov Web site (no. NCT01007136) and approved by the local ethics committee (CPP Sud Ouest et Outre-mer 1). Written informed consent was obtained from all participants before their inclusion in the study.
Transcranial Direct Current Stimulation
tDCS was applied with the anode placed over the ipsilesional motor cortex (M1) at the hotspot of the extensor carpi radialis muscle (ECR, previously determined by the baseline TMS study, see below) and the cathode over the contralesional supraorbital region. Anodal stimulation was delivered by a Magstim Eldith DC stimulator plus through a pair of 35 cm2 saline-soaked sponge surface electrodes.
For the active condition, patients received 5 consecutive daily sessions of 1.2 mA anodal tDCS for 13 minutes each, while for the sham condition, the stimulation (same site and same parameters) was turned off after 60 seconds of stimulation.
Repetitive Peripheral Nerve Stimulation
Repetitive electrical (DIGITIMER DS7A) stimulation (5 Hz) was delivered to the radial nerve through bipolar round brass electrodes (2 cm diameter) placed in the spiral grove of the paretic side and was applied at the same time as the real or sham tDCS stimulation. It was applied similarly in both active and sham conditions for 13 minutes. The intensity of the rPNS was adjusted to be below the threshold for direct M response (0.7 × MT).
Outcome Measures
The Jebsen Taylor Hand Function Test (JHFT) was used as the primary outcome measure 32 (with exclusion of the writing task). It measures hand function in real-life activities by evaluating the time required to perform various tasks (turning cards, lifting objects, feeding simulation, stacking checkers, moving light and heavy cans). As secondary outcomes, we used the Hand Dynamometer (maximum grip force of the hand, DYN) (JAMAR), the Nine Hole Peg Test (9HPT), the Hand Tapping test (HTap, number of palm taps on a mechanical hand tapping for 10 seconds), and the Upper Limb Fugl-Meyer (ULFM) scale. 33
All the tests were performed 3 times with the affected hand, and the mean of 3 trials was taken as the result—except for the 9HPT, which was performed only twice.
Corticomotor Excitability
Using TMS, we determined the presence or absence of a motor evoked potential (MEP) and resting and active motor thresholds (RMT, AMT) in the affected motor cortex. TMS was delivered using a Magstim 200 stimulator (Magstim Co, Whitland, UK) through a 70 mm figure-of-eight–shaped magnetic coil, held with the handle pointing backward at approximately 45° from the sagittal midline, over the cortical ECR Hotspot on the affected hemisphere. Electromyographic (EMG) activity was recorded from Ag–AgCl surface disposable electrodes pasted on the skin over the muscle belly of the paretic ECR muscle, with the reference electrode placed between EMG and radial nerve stimulation electrodes. During the experiments, EMG activity was continuously monitored with visual (oscilloscope) feedback to ensure complete relaxation at rest. The optimal scalp position for the coil was defined as the site of stimulation that consistently yielded the largest ECR MEP.
Rest and Active Motor Threshold
The resting motor threshold (RMT) was defined as the minimum TMS intensity (measured to the nearest 1% of the maximum output of the magnetic stimulator) required to elicit an MEP of at least 50 µV in the relaxed ECR in at least 5 of 10 trials with an intertrial interval of 6 seconds.
The active motor threshold (AMT) was defined as the minimum intensity required to elicit at least 5 out of 10 MEPs of at least 200 µV in the ECR during a weak voluntary contraction of the ECR (10% to 20% of maximal isometric voluntary muscle contraction).
Study Design
Patients included in this double-blind, sham-controlled, pilot study first participated in a familiarization session in which they trained themselves on the motor tests 3 times with each hand. They were randomized in 2 parallel groups (anodal tDCS or sham tDCS group), without stratification regarding the severity of their baseline motor impairment. The randomization list was created by the Clinical Research Center of Toulouse using Rand List Software V1.2 (Dat Inf GmbH; www.randomisation.eu), which provided 5 blocks of 4 patients, each balanced between the sham and active interventions. All the investigators were blinded to the patient’s allocation except the doctor who applied the stimulation. Each patient received 5 consecutive daily sessions of tDCS (anodal or sham), combined with rPNS on the paretic side. The peripheral and cortical stimulations were applied at the same time. Motor performance and TMS measures were obtained at baseline (D1) before the first stimulation session, at the end of the treatment (D5), and at 2 and 4 weeks’ follow-up (D15 and D30). The ULFM was not recorded at D15. The assessment of motor functions and the TMS study were performed by trained doctors, blinded to group assignment.
Conventional occupational therapy sessions (from 3 to 5 times per week) started at the in-patient stroke clinic, independently of the tDCS sessions, most often at D4 or D5, and lasted 30 minutes. After discharge, outpatient therapy sessions were given 3 to 5 times per week (45-90 minutes/session) until the end of the follow-up period. Therapists were blinded to group allocation.
Data Analysis
The comparison of the baseline clinical motor performance, TMS parameters, and therapy doses between groups were performed using unpaired t tests.
Because the baseline motor performance of the patients enrolled differed widely, we calculated the differences between baseline and D5, D15, and D30 data (Δt5, Δt15, Δt30) for each patient and each motor test. These Δt values were input to the general linear mixed-factors variance analysis (repeated measures ANOVA) with GROUP (anodal tDCS/sham tDCS), as the between-subject factor, and TIME (Δt5, Δt15, Δt30), as the within-subject factor, and baseline JHFT as covariate. The primary outcome for analysis was the mean change in motor performance as indexed by the decrease in time needed to perform the JHFT between baseline (D1) and respectively D5, D15, and D30. TMS data (RMT, AMT) were tested using repeated-measures ANOVA with GROUP as the between-subject factor and TIME (D1, D5, D15, D30) as the within-subject factor. Appropriate post hoc comparisons were carried out using a Bonferroni correction. Results are reported as means and standard deviations. Statistical significance refers to a 2-tailed P value <.05. Correlation between motor performance and electrophysiological parameters was investigated using the Pearson test.
Results
Forty-two acute ischemic stroke patients were consecutively assessed for participation (Figure 1). Twenty-two patients were excluded (6 for not meeting inclusion criteria, 11 for instability of motor deficits, and 5 declined to participate), leaving 20 patients randomized (mean age: 65 years ± 11 [range 37-82], 6 women). They had mild to moderate motor deficit at onset (Table 1; mean National Institutes of Health Stroke Scale [NIHSS]: 3 ± 1, range 1-6; mean ULFM score: 48 ± 2.5, range 29-60). They were heterogeneous with regard to lesion site (cortical and subcortical areas; imaging [DWI] data given in Table 2 supplemental data). Two patients in the sham group (nos. 1 and 2) and 3 in the tDCS group (nos. 3, 6, and 17) had white matter lesions (range of Fazekas & Schmidt’s score: 3-5). 34
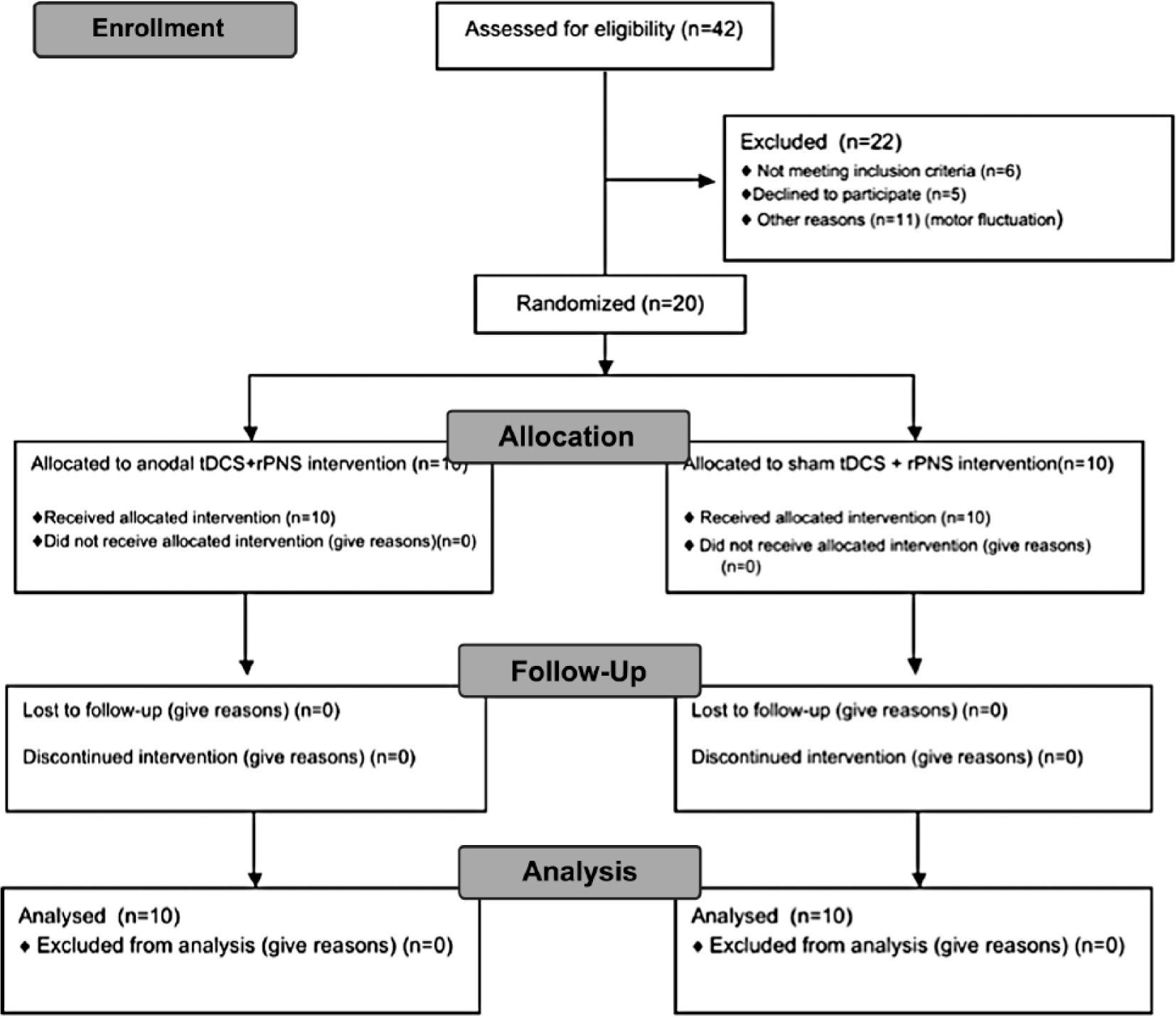
Study flow chart.
Demographic and Baseline Data. a
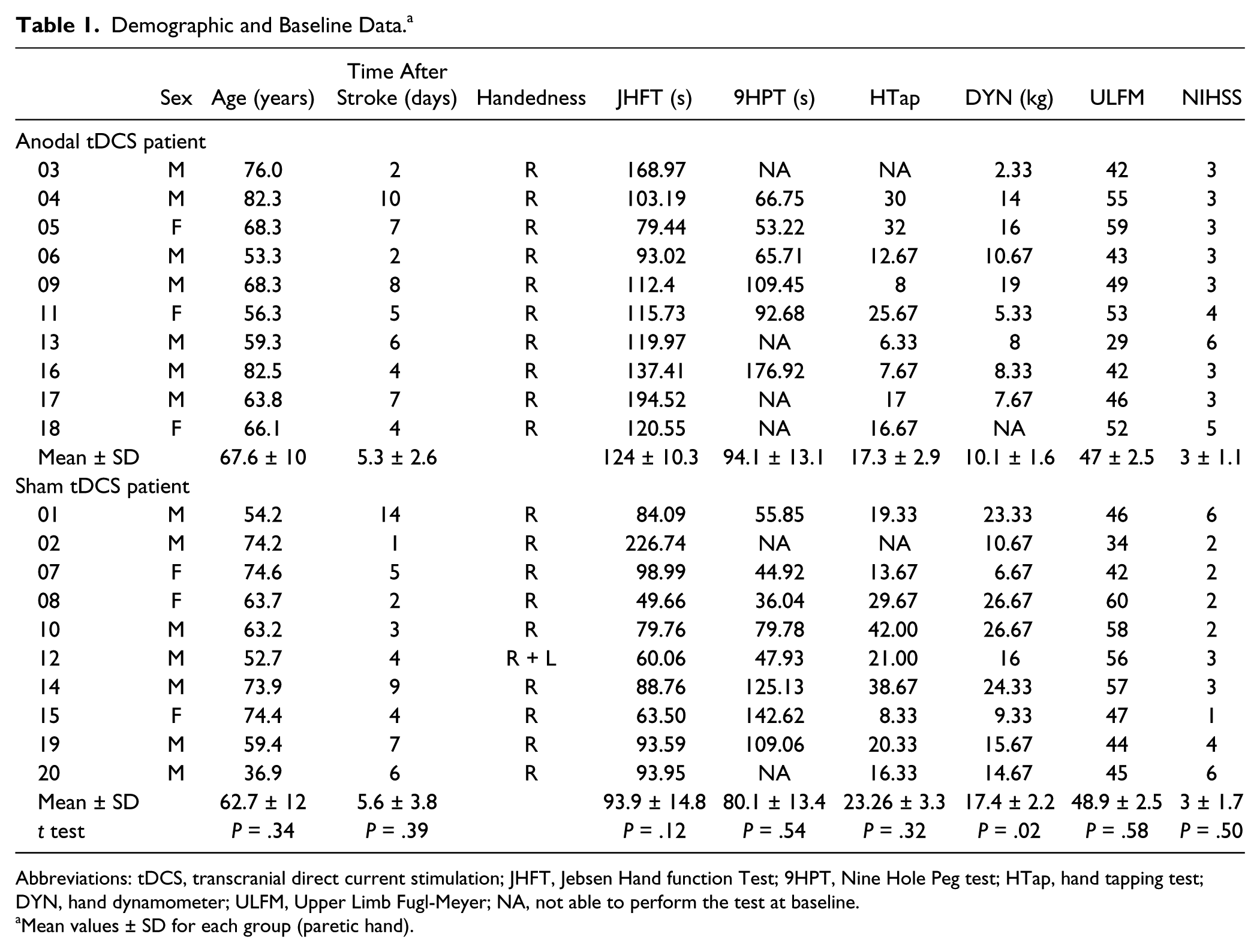
Abbreviations: tDCS, transcranial direct current stimulation; JHFT, Jebsen Hand function Test; 9HPT, Nine Hole Peg test; HTap, hand tapping test; DYN, hand dynamometer; ULFM, Upper Limb Fugl-Meyer; NA, not able to perform the test at baseline.
Mean values ± SD for each group (paretic hand).
Changes in Motor Performance of the Paretic Hand Compared to Baseline at the End of the Treatment and in the Follow-Up. a
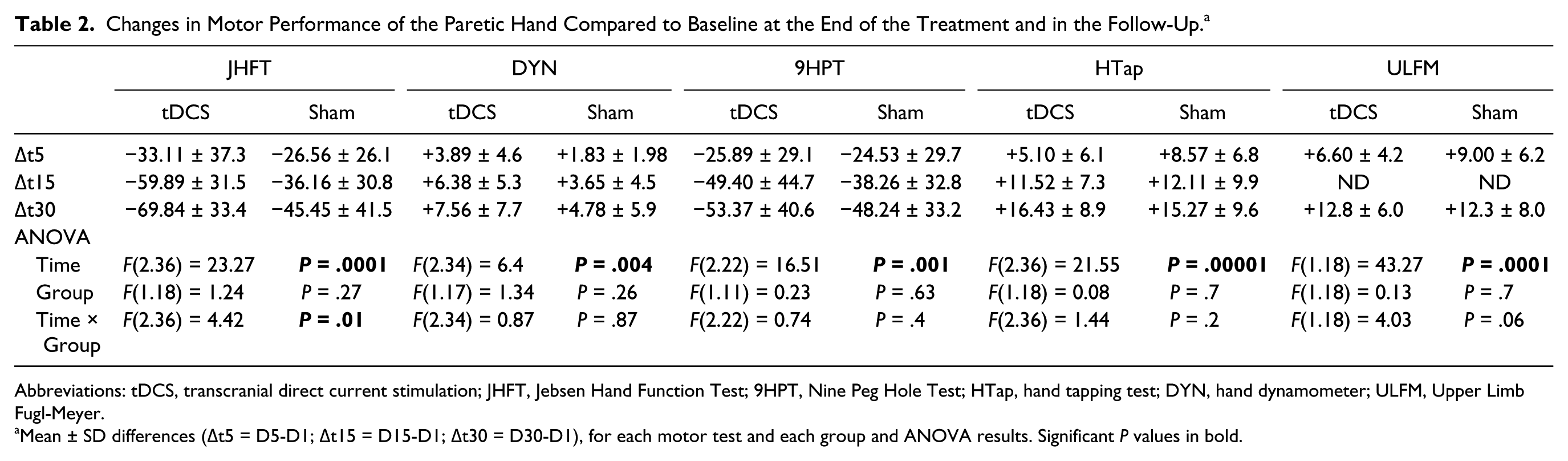
Abbreviations: tDCS, transcranial direct current stimulation; JHFT, Jebsen Hand Function Test; 9HPT, Nine Peg Hole Test; HTap, hand tapping test; DYN, hand dynamometer; ULFM, Upper Limb Fugl-Meyer.
Mean ± SD differences (Δt5 = D5-D1; Δt15 = D15-D1; Δt30 = D30-D1), for each motor test and each group and ANOVA results. Significant P values in bold.
The mean time between stroke and tDCS intervention was 5.5 ± 3 days. No adverse effects were reported except a tingling sensation at the site of the scalp electrodes at the beginning of the session. This was similarly observed in both groups of patients, who were thus unable to distinguish the tDCS from the sham session. Each group received the same amount of physical therapy during the follow-up (tDCS group: mean therapy dose: 17.5 ± 9 hours; sham group: 18 ± 13 hours; P = .8).
Demographic and clinical data of the 2 groups of patients are reported in Table 1. Despite our selective inclusion criteria (mild or moderate hand motor deficit), the range of individual baseline times to perform the JHFT was very large (49-226 seconds). No significant differences between the 2 groups were observed at baseline (Table 1), except for the grip test (P = .02).
Motor Function of the Paretic Hand
Motor performance improved significantly with time in both groups, with a progressive decrease of the time to perform the JHFT between D1 and D30 (Table 3). Analysis of the differences in the total time to perform the JHFT between baseline and D5 (Δt5), D15 (Δt15), and D30 (Δt30) showed a TIME effect (F[2.36] = 23.27, P = .0001), no GROUP effect (F[1.18] = 1.24, P = .27), but a significant TIME × GROUP interaction (F[2.36] = 4.42, P = .01). This interaction was explained by the fact that the amount of improvement was greater at D15 (P = .03) and D30 (P = .01) in the anodal tDCS group than in the sham tDCS group. Figure 2 illustrates the difference of time course of the mean Δt ± SD between the 2 groups of patients, which did not occur at D5, as expected, but occurred 10 days after the end of the intervention (D15) and was still present and more pronounced at D30. On average, compared to the sham group, the gain of motor performance in JHFT after the 5 days of anodal tDCS was expressed by decreases of 23 seconds at D15, 24 seconds at D30 (and 6 seconds at D5).
Electrophysiological Results. a
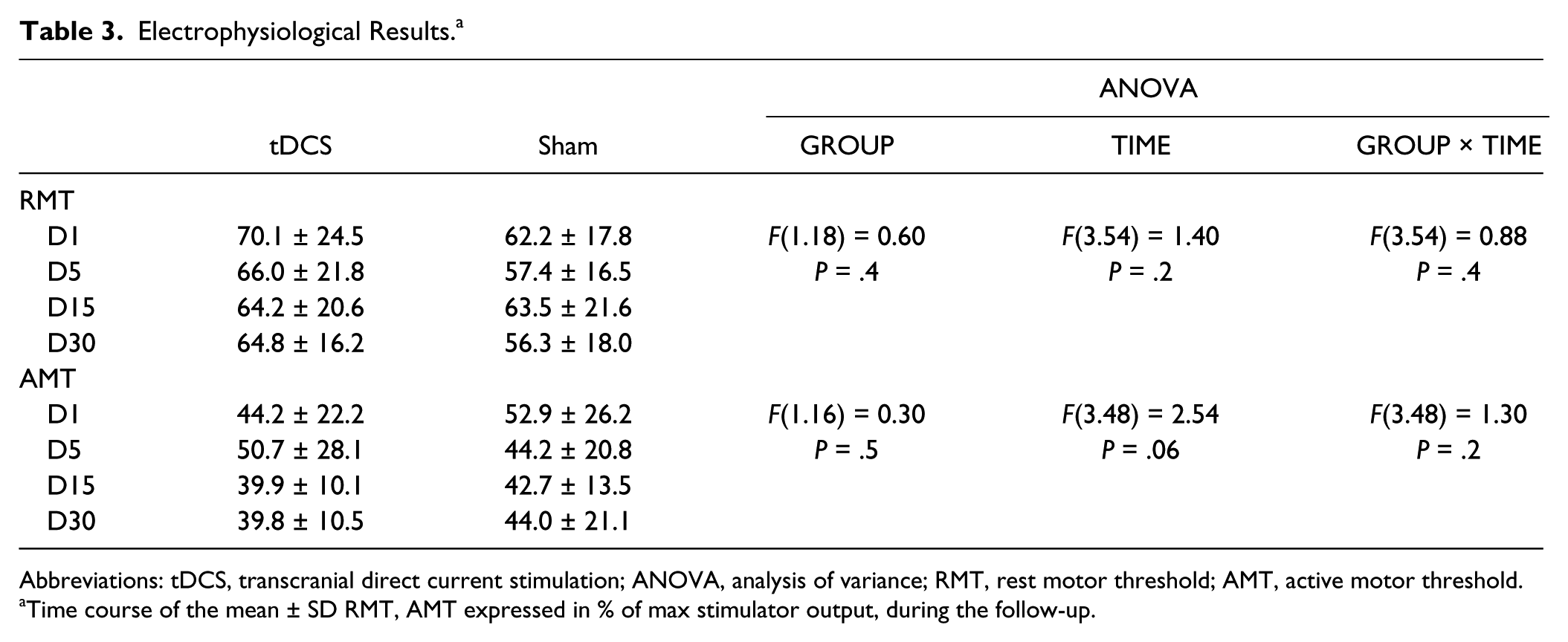
Abbreviations: tDCS, transcranial direct current stimulation; ANOVA, analysis of variance; RMT, rest motor threshold; AMT, active motor threshold.
Time course of the mean ± SD RMT, AMT expressed in % of max stimulator output, during the follow-up.
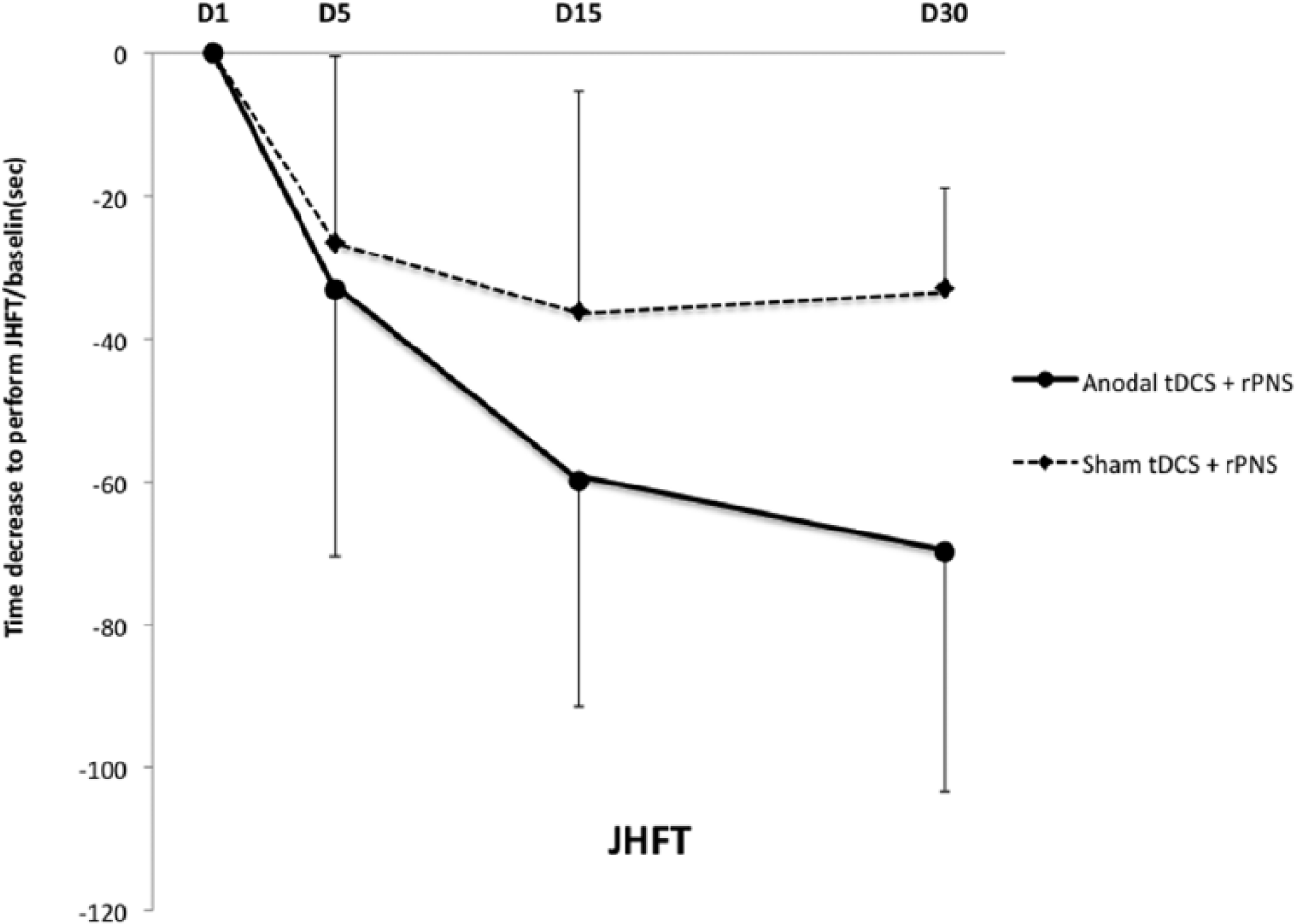
Time course of the mean (±SD) gain of motor performance of the paretic hand in JHFT compared to baseline in each group of patients.
Six patients (4 in the active group and 2 in the sham group) were unable to perform the 9HPT test at D1, and 2 patients (1 in each group) were unable to perform the HTap task at D1.
Similar improvement of motor performance with time was observed in both groups for the secondary motor outcomes (Table 3). A trend toward a gain of performance in the anodal tDCS group compared to the sham group for the 9HPT (n = 14, on average −11 seconds at D15 and −5 seconds at D30 to perform the task) and for the DYN test (on average gain of +3 kg at D15 and D30) were observed but without significant TIME × GROUP interactions. For the HTap test, the increase in the number of taps with time was in the same range in both groups, without any significant differences (n = 18). At D5, there was a trend toward greater gain on the Fugl-Meyer scale for the sham group than for the tDCS group (on average +2.4; NS), which was no longer observed at D30 (Table 3). The comparison of the mean sum of the Fugl-Meyer’s wrist and hand finger extension subscores (0-12), between tDCS + rPNS group and sham tDCS + rPNS group, did not show any significant difference at D1 (7.3 ± 3/8.7 ± 2.5), D5 (9.3 ± 2.5/10.5 ± 1.2), and D30 (10.7 ± 1.2/11.1 ± 1.2), respectively (P = .19, F = 1.79), but a significant improvement with time (P = .0001, F = 17.94) for both groups (without significant interaction), confirming that most of the patients improved significantly their wrist and finger extension at the end of the study.
Electrophysiology
TMS data are summarized in Table 4. At D1 and at maximal output of TMS, MEPs were absent in only 2 of the 10 patients in the tDCS group and in 1 patient of the sham group. One month after inclusion, MEPs were measurable in all patients except one.
The mean RMT at baseline was 70.10 ± 24.5% for the tDCS group and 62.2 ± 17.8% for the sham group (P = .4). There was a trend toward a slight decrease of RMT and AMT with time in both groups, which did not reach statistical significance. ANOVA did not reveal any significant TIME, GROUP, or TIME × GROUP interaction.
Baseline motor performance of the whole group was correlated with RMT measured at D1 (ULFM: r = −.58, P = . 007; HTap: r = −.58, P = .007; JHFT: r = .49, P = .03); the worse the motor performance was at inclusion, the higher was the RMT (Figure 3).
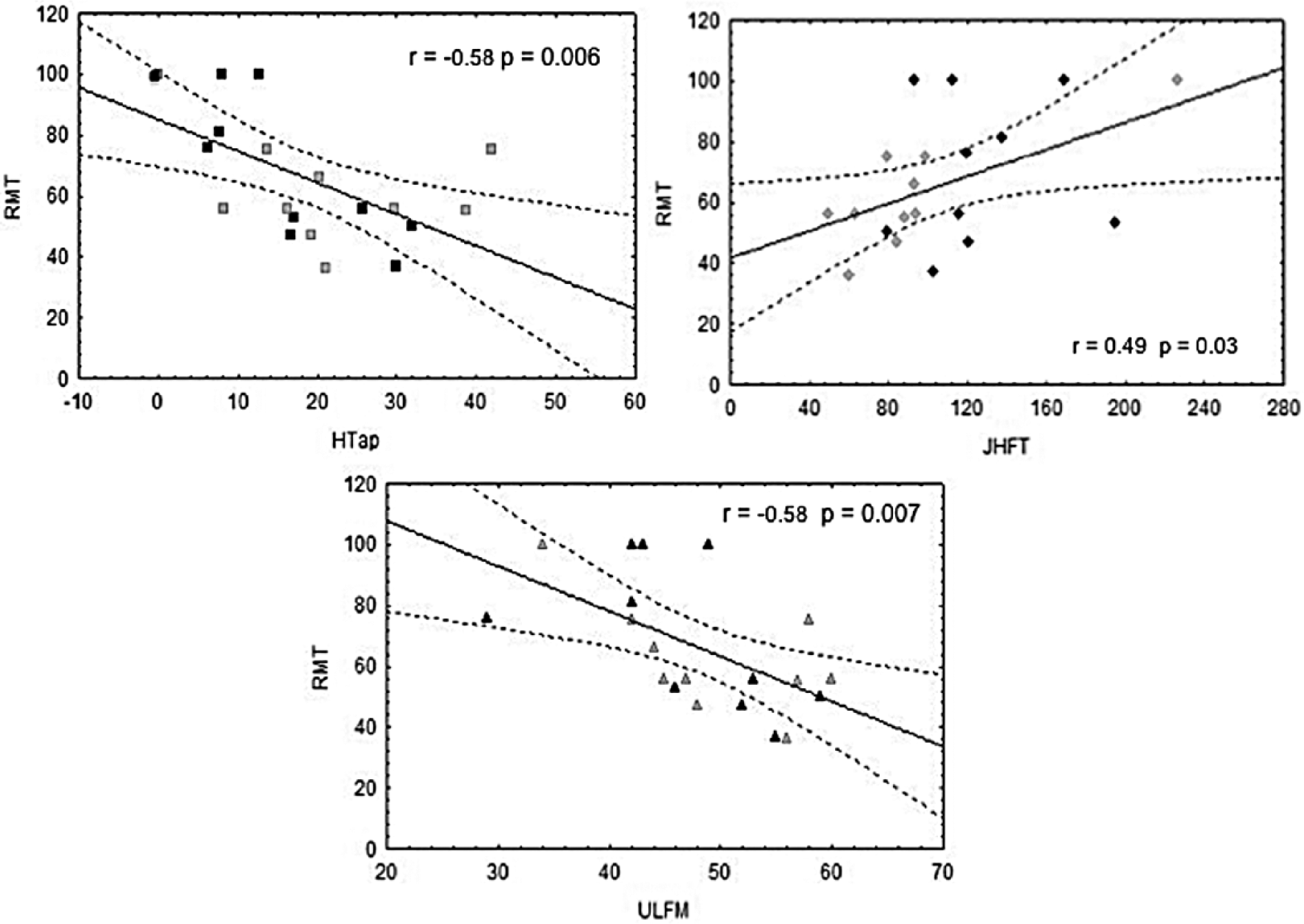
Baseline correlations between motor tests.
Discussion
The results of this pilot study, performed in the acute phase after stroke (on average 5 days), suggest that the time course of the motor recovery of the 2 groups of patients during the first month was different and was positively influenced by the intervention.
Our results confirm the good responsiveness of the JHFT, widely used in NIBS studies6,7,13,24,35 to reveal an improvement in functional hand motricity that reflects activities of daily life.36,37 The 9HPT has good validity, reliability, and sensitivity in assessing manual dexterity 38 but is feasible only in patients with a mild motor deficit. Six patients were unable to perform it at baseline and this could explain the lack of statistical difference between the 2 groups, in contrast to the JHFT, which was performed by all the patients at baseline. The Hand Grip test was easily achieved by all the patients at baseline but was less sensitive than the ecological JHFT. The lower sensitivity of the HTap test compared to JHFT could perhaps be explained by a greater influence of factors other than the severity of the motor deficit, such as age 39 or genetics, to explain the wide range of values observed at baseline (range 8-42). A significant positive effect of a single session of tDCS versus sham applied to stroke patients in the subacute phase was also reported in 2 previous studies, one with a different hand dexterity test (the Box and Block test 11 ) and the other with the 9HPT, 23 but without any significant effect on the grip force in the latter. The lack of a significant change in the ULFM score was not surprising considering the small size of our patient sample and the low sensitivity of this scale for mild or moderate hemiparesis. Despite a larger number of patients (25 × 2), Rossi et al 40 failed to demonstrate a significant positive effect of 5 sessions of anodal tDCS versus sham applied in the very acute phase (2 days) after stroke, using the ULFM score as primary outcome measure. These negative results were probably explained by broader inclusion criteria (with patients enrolled even if they had a severe motor deficit and a large lesion in M1) than ours (mild or moderate deficit without large involvement of M1). However, the lack of significant positive effect of the intervention on motor impairment assessed by the ULFM, HTap, and Hand Grip test contrasts with the significant positive effect on the JHFT assessing the functional hand motricity. These results suggest that the intervention-induced enhancement of motor recovery could facilitate functional upper limb adaptation strategies rather than “restitution” of motor force. Other mechanisms such an increased gain from physical therapy through a positive additional effect on motor learning cannot be completely ruled out even if intervention was not time-locked to the physical therapy.
It has previously been shown in chronic stroke that anodal tDCS increases excitability within the stimulated region 41 and increases task-related functional motor activity in ipsilesional M1 and in a number of interconnected secondary motor areas.28,42 It has been suggested that the bidirectional effects of NIBS over M1 on corticospinal excitability in humans might reflect long-term potentiation (LTP) and long-term depression (LTD)-like plasticity. 43 The NIBS-induced increase in corticospinal excitability might improve motor function by facilitating the volitional recruitment of corticomotor output neurons. 44 However, the mechanisms underlying the long-lasting anodal tDCS facilitatory posteffects are not completely elucidated, and the potential influence of the cathodal electrode over the contralateral prefrontal cortex is still unknown. 45 It is worth noting that tDCS increases BDNF secretion and synaptic plasticity in animals, 5 which could be a key mechanism underlying tDCS-induced improvements. 46
If these results suggest that a certain degree of recovery in the treated group was due to therapeutic intervention, several factors, other than the intervention itself, deserve to be discussed. Brain reorganization after stroke is a dynamic process, which differs considerably across patients depending on lesion location, time since stroke, severity of motor impairment, premorbid state, and genetics. Baseline mean motor performance, NIHSS, age, sex, and delay after stroke at inclusion, were not statistically different between the 2 groups (except for the grip force), so that the influence of these factors should be very mild. However, the absence of significant difference in the mean time required to perform the JHFT at baseline was not a guarantee that both groups were prognostically equal, given the large range of values and their unequal distribution between the 2 groups.
More intensive physical therapy in the tDCS group compared to the sham group could have contributed to the differences of recovery pattern observed. Although outpatient poststroke rehabilitation programs were delivered by different rehabilitation centers, the mean therapy doses (expressed in total number of hours during the follow-up) was similar in both groups. None of the patients included had poststroke depressive symptoms, severe spasticity, or shoulder pain, which also would possibly have influenced the results. More patients with cortical involvement (7/10) were recruited in the sham group than in the tDCS group (2/10), and this difference of lesion site may have influenced the quality of the recovery as previously shown after one session of 10 Hz rTMS in subacute and chronic stroke. 47 This difference did not have any impact on clinical baseline motor performances of the 2 groups that were comparable, except for the grip force, which was little stronger in the sham group than in the tDCS group. Two patients of each group had a cortical infarction involving to some extent the upper extremity of M1 (nos. 11, 16, 12, and 15). These lesions were very limited and the presence of an MEP at inclusion in most of the patients in both groups (8/10 tDCS group; 9/10 sham group, including these 4 patients) suggests a small impact of the lesion location on the excitability of the lesioned upper limb extremity M1 and/or cortico-spinal tract in this population. At least, both groups were well balanced in this regard.
The radial nerve stimulation alone was not expected to induce a large beneficial effect on wrist and finger extension and rather proposed as a “control” intervention in the sham group to facilitate recruitment and the patients’ acceptance of the protocol. In fact, previous studies have shown that rPNS should last at least 2 hours to induce moderate but measurable positive effects on the motor performance in chronic stroke patients.19,22,48 Because of the absence of a sham rPNS arm and the lack of selective outcome measuring the wrist and finger extension specifically, we are unable to estimate precisely the amount of facilitation induced by the radial nerve stimulation on the paretic wrist and finger extension (in comparison with the spontaneous motor recovery), and its relative contribution to the global better motor performances observed at JHFT in the group receiving the combined tDCS + rPNS intervention compared to the group receiving only the rPNS. Nevertheless, Fugl-Meyer’s wrist and finger extension subscore analysis confirms that most of the patients of both groups had a very good recovery of their wrist and finger extension on the paretic side at the end of the study. The 2-arm design of our pilot study did not allow either to compare the effect of combined tDCS + rPNS versus real tDCS alone so that the potential additive beneficial effects of the combined stimulation remain to be demonstrated in the acute phase. However, even if we cannot assess precisely the amount of motor improvement at JHFT, respectively, due to anodal tDCS, radial nerve stimulation, or their combination, our results are encouraging. The changes of corticomotor excitability induced by rPNS on motor cortical function may well involve a modulation of GABAergic interneurons that are somatotopically specific to the stimulated region,19,22,49 and which could potentiate the long-lasting anodal tDCS facilitatory post-effects on motor learning. Promising results have been reported in chronic patients with a clear additional benefit of the combined peripheral and cortical stimulation compared to the cortical or peripheral stimulation alone. 22
The tDCS-induced gain of motor performance in JHFT was measurable 10 days after the end of the 5 daily sessions but not on the last day of treatment (D5), contrary to what has been found in most of the multisession NIBS studies performed in the chronic phase of motor recovery.6,15,16,22,50 In the subacute phase, Kim et al 10 reported an improvement in the Fugl-Meyer score at 6 months follow-up but not 1 day after the end of 10 sessions of real tDCS compared to sham for 18 patients included. It is also likely that a more subtle beneficial effect of tDCS on recovery was not evident at D5 because of the small number of patients in each group.
Another important question is how long the tDCS post-effects actually last. In most of tDCS stroke studies, the follow-up of the patients is very short, not exceeding 15 days, except for two,9,10 with a follow-up of 6 months. Here, the beneficial additional effect of the tDCS observed 2 weeks after the first day of treatment was still present after a further 2 weeks, confirming that 5 days of NIBS treatment, proposed on average 5 days after the stroke, were enough to induce long-lasting enduring positive effects on motor recovery.
The presence of an ECR MEP in almost all the patients (except 3), on the first day of treatment (which is generally accepted as a marker of good prognosis for motor recovery51,52), suggests that, in most patients, the motor output was preserved. The absence of significant changes of RMT and AMT between baseline and D5 in the anodal tDCS group was in accordance with results observed in healthy subjects and suggests that tDCS neuromodulation is not enough to induce an MT shift. 53 The trend toward a decrease of RMT and AMT with time observed in both groups during the follow-up was in accordance with previous results.9,54-56 As in previous reports,55-57 confirmed recently, 54 we did not find any association between clinical improvement and measures of RMT, but we observed a good correlation at baseline between raised motor threshold and weak motor performance as assessed by ULFM, HTap, and JHFT. These correlations confirm that the RMT is closely associated with motor function and muscle strength in the paretic limb.55,58,59
Limitations
Although the results of this study are promising, it has a number of limitations, such as the small sample size, the differences in lesion side and sites, the lack of DTI white matter lesion load, and the lack of control arms in which either the therapeutic effect of tDCS alone or the spontaneous motor recovery (sham tDCS + sham rPNS) could be tested. In addition, most of the stroke patients included presented mild to moderate disability, and we do not know if such an intervention proposed in the acute phase could benefit more severely impaired patients.
In conclusion, this pilot study shows that 5 consecutive days of tDCS combined with rPNS proposed in the acute phase after a stroke is well tolerated, that it can promote the recovery of paretic hand performance, at least in patients with motor deficit that is not too severe, and that the benefit is still present 1 month after the stroke. Further trials performed in multiple centers with larger sample size, different designs, longer follow-up, different comparative NIBS doses and montages (anodal, cathodal, or bihemispheric), using markers predictive of behavioral response, 17 should be conducted in the acute or subacute phase in order to confirm that NIBS is able to speed up recovery, which could have a medico-economic impact in terms of the duration of rehabilitation programs.
Footnotes
Acknowledgements
The authors are grateful to Dr Tap for the statistical analysis and to Dr Samia Cheriet for technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from Fondation de l’Avenir (ET9-531), by INSERM (C09-27), the Clinical Research Center of Toulouse (CIC), and Toulouse University Hospital.
