Abstract
Stroke has become one of the main causes of visual impairment, with more than 15 million incidences of first-time strokes, per year, worldwide. One-third of stroke survivors exhibit visual impairment, and most of them will not fully recover. Some recovery is possible, but this usually happens in the first few weeks after a stroke. Most of the rehabilitation options that are offered to patients are compensatory, such as optical aids or eye training. However, these techniques do not seem to provide a sufficient amount of improvement transferable to everyday life. Based on the relatively recent idea that the visual system can actually recover from a chronic lesion, visual retraining protocols have emerged, sometimes even in combination with noninvasive brain stimulation (NIBS), to further boost plastic changes in the residual visual tracts and network. The present article reviews the underlying mechanisms supporting visual retraining and describes the first clinical trials that applied NIBS combined with visual retraining. As a further perspective, it gathers the scientific evidence demonstrating the relevance of interregional functional synchronization of brain networks for visual field recovery, especially the causal role of α and γ oscillations in parieto-occipital regions. Because transcranial alternating current stimulation (tACS) can induce frequency-specific entrainment and modulate spike timing–dependent plasticity, we present a new promising interventional approach, consisting of applying physiologically motivated tACS protocols based on multifocal cross-frequency brain stimulation of the visuoattentional network for visual field recovery.
Humans are highly dependent on their sense of vision to interact with the surrounding world. Not surprisingly, the loss of visual function has a dramatic impact on an individual’s quality of life and independence.1,2 It definitively constitutes a widespread public health problem, with an estimation of approximately 314 million people suffering from vision impairment. 3 Blindness and visual impairment can have multiple potential origins—for example, glaucoma, macular degeneration, cataract, and others. Cortical visual field deficit is a well-known complication of stroke, with a reported incidence of 20% in acute stroke patients. 4 Despite diverse therapeutic attempts, no clinically proven efficient treatment is available to restore vision in these patients. The present article reviews the underlying mechanisms supporting visual retraining and describes the first clinical trials that applied NIBS combined with visual retraining. As a further perspective, it gathers the scientific evidence demonstrating the relevance of interregional functional synchronization of brain networks for visual field recovery, especially the causal role of α and γ oscillations in parieto-occipital regions. We finally present a new promising interventional approach, based on multifocal cross-frequency brain stimulation of the visuoattentional network for visual field recovery.
Stroke: A Cause of Visual Deficit
It is known that 45% of the lesions involve the occipital lobes and 32% the optic radiations, leading to drastic visual field losses. Spontaneous improvement is reported in approximatively 50% of these patients within the first month of injury. 5 Later on, up to 40% of patients will still have permanent visual deficits in the chronic stage.6,7 Homonymous hemianopia, the most common type of visual field deficit after stroke, consists in for the loss of the same half of the visual field for both eyes. It occurs after a lesion of the retrochiasmal visual pathways (ie, lesions of the optic tract, the lateral geniculate nucleus, the optic radiations, and the cerebral visual (occipital) cortex 8 (Figure 1).
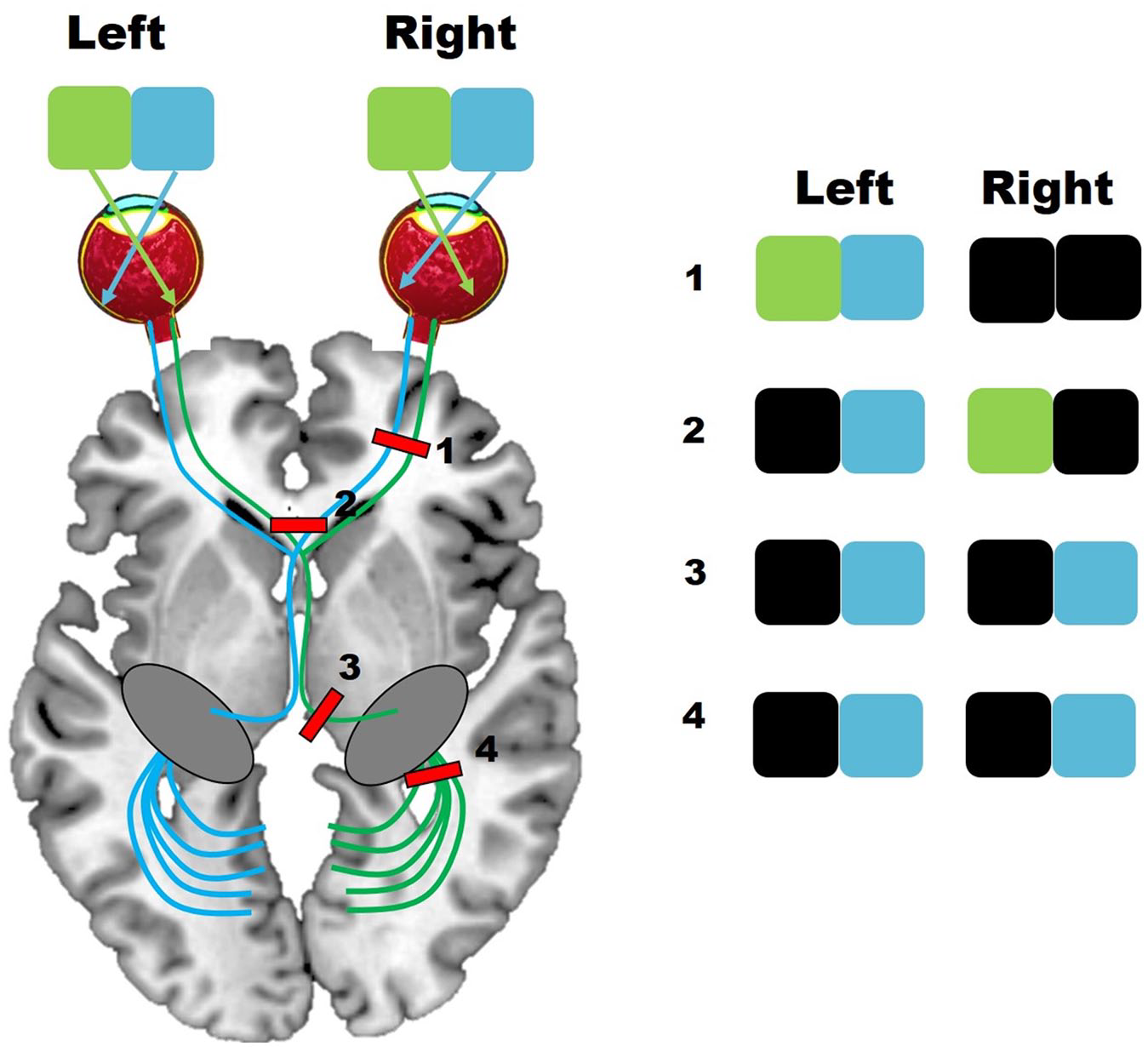
Schematic of the visual pathway in the brain. Lesions at different levels of the visual pathways (1-4) cause different functional vision deficits. Black boxes reflect the location of the visual deficit.
Visual field deficits are associated with impairment of reading, visual searches, driving, or navigating in complex environments.9,10 It has unavoidably negative implications for the quality of life11,12 and rehabilitation, being even an important predictor of functional status on discharge from stroke rehabilitation units. 13 Hence, visual field defects should be given as much attention as hemiparesis during rehabilitation for patients with stroke. However, for a long time, the visual system was thought to have poor capacities for recovery after injury. 14 Even nowadays, the main clinical options offered to patients are compensatory rather than restorative. For instance, they include optical aids, e.g., prisms that are monocularly or binocularly fitted to patients to restore peripheral vision.15,16 The idea is to artificially expand the visual field, so that parts of the visual world that would naturally fall into the blind field now appear in the intact field. These optical aids appear to improve visual functioning,17-19 but also induce central double vision (diplopia), which is obviously uncomfortable and hampers the patients. Of interest, monocularly fitted sector prisms have been introduced, allowing visual field expansions without central diplopia. 20 However, randomized controlled trials are missing to properly evaluate the functional relevance of such devices. Alternatively, eye movement therapy approaches attempt to improve visual performance using specific training paradigms based on visual search strategies, saccadic eye movements, or reading training, beyond others.21-23 Efficacy is moderate, 24 but changes in eye movement parameters, including increased amplitude of reading saccades into the damaged blind hemifield are linked to improvements in text reading speed. 25 Once more, these strategies rely on developing adaptive behavior rather than restoring the damaged pathways or reorganizing the impaired network. They actually do not induce any significant reversal of visual deficits induced by the cortical damage itself. However, recent evidence in animals and humans showed long-term visual improvements even after the early postlesion phase, associated with specific brain reorganization patterns. This might serve as a basis for the development of new evidence-based and biologically inspired therapeutic strategies to enhance functional reorganization with consecutive visual restoration.
Mechanisms of Recovery and Restorative Therapies
Besides removing the main source of feedforward inputs to higher visual areas, as well as feedback information to subcortical areas (eg, the superior colliculi), damage to V1 also induces retrograde degeneration in retinotopically corresponding regions of the lateral geniculate nucleus and sometimes even in associated areas of the retina, as shown in animals.26,27 However, in most patients with postchiasmatic lesions, the injury is incomplete. This allows some forms of residual visual function28,29 and might enable some plastic changes supporting partial recovery of function.
Among the early plastic mechanisms that have been proposed, an acute disinhibition of GABAergic inputs in the perilesional tissue occurs in the early stage postinjury.30-33 These nonspecific postlesional changes in neuronal responsiveness 34 might indeed support spontaneous recovery during the first few months postinjury, together with the resolution of inflammation and edema surrounding the lesion and later on via the reactivation of partially damaged perilesional tissue.35,36 This set of cellular changes leads to increased receptive field size and neuronal excitability around the lesion site. 37 After this early postlesion phase, further reorganization in the adult visual system has been finally demonstrated in adult animal studies, in which a part of V1 was deafferented via retinal lesions,38,39 although few studies have also failed to report this V1 reorganization.40,41 These findings led to the idea that further degrees of recovery could be achieved via sustained visual stimulation. Likewise, several groups have developed computer-based visual retraining approaches in an attempt to restore vision within the damaged field of view using flicker stimulation, 42 neural restoration training, 43 or optometric vision therapy. 44 A well-known approach, called blindsight training, focuses on exercising the unconscious perceptual functions deep inside the blind hemifield and relies on the recruitment of extrastriate visual pathways, or the blindsight pathways. 45 Visual attributes sensitive to blindsight are presented in the blind field simultaneous to temporal and spatial cues.46,47 An alternative approach called border training aims at restoring striate pathways by retraining vision at the border of the scotoma.48,49 The rationale is that responding to many thousand stimuli in this residual border region of the scotoma, the border zone may shift and, thus, reduce the field of blindness. Visual field restitution therapy (VRT; NovaVision Inc, Boca Raton, FL) is one example of border-training. It consists of a computer-based stimulation program presenting suprathreshold light stimuli along the border zone of the scotoma. 48 Although there are controversies regarding its potential mechanisms,50-54 VRT has been shown to provide a 5° expansion of the residual visual field on average. 55
In summary, repeated visual stimulation might help activate preexisting intact cortical islands within V1 and perilesional spared V1 neurons or reactivate part of the damaged V1 cortex. Alternative pathways can also be involved such as interhemispheric connections or extrageniculo-calcarine pathways to more effectively transfer information directly to higher visual areas such as V4 or V5. 56 But even when applied for repeated sessions, these therapies show a modest effect on the size of the residual field of view, and overall, they fail to provide significant and long-term benefits for the everyday life of patients.56,57 Therefore, there is a pressing need to develop innovative rehabilitation techniques able to maximize the treatment outcomes. Recently, noninvasive brain stimulation (NIBS) protocols have emerged in neurorehabilitation.58-60 Applied to visual field defects, they might also help counteract the poor outcomes of these conventional therapies.
Enhancing Restorative Therapies With NIBS
Whereas NIBS has been widely used in patients to recover motor functions after stroke,59,61,62 only a few clinical studies have applied brain stimulation to patients in conjunction with visual training. Even less research has been conducted in the context of visual field defects, as compared with other visual deficits such as amblyopia63-66 or myopia,67,68 all consisting of transcranial electrical stimulation studies. Nevertheless, the first proof-of-principle studies suggest that NIBS might be a promising add-on approach for neurorehabilitation of visual field deficits69-71 (see Table 1 for an overview of these first proof-of-principle studies).
Summary of the Studies Investigating the Combination of NIBS With Visual Training.
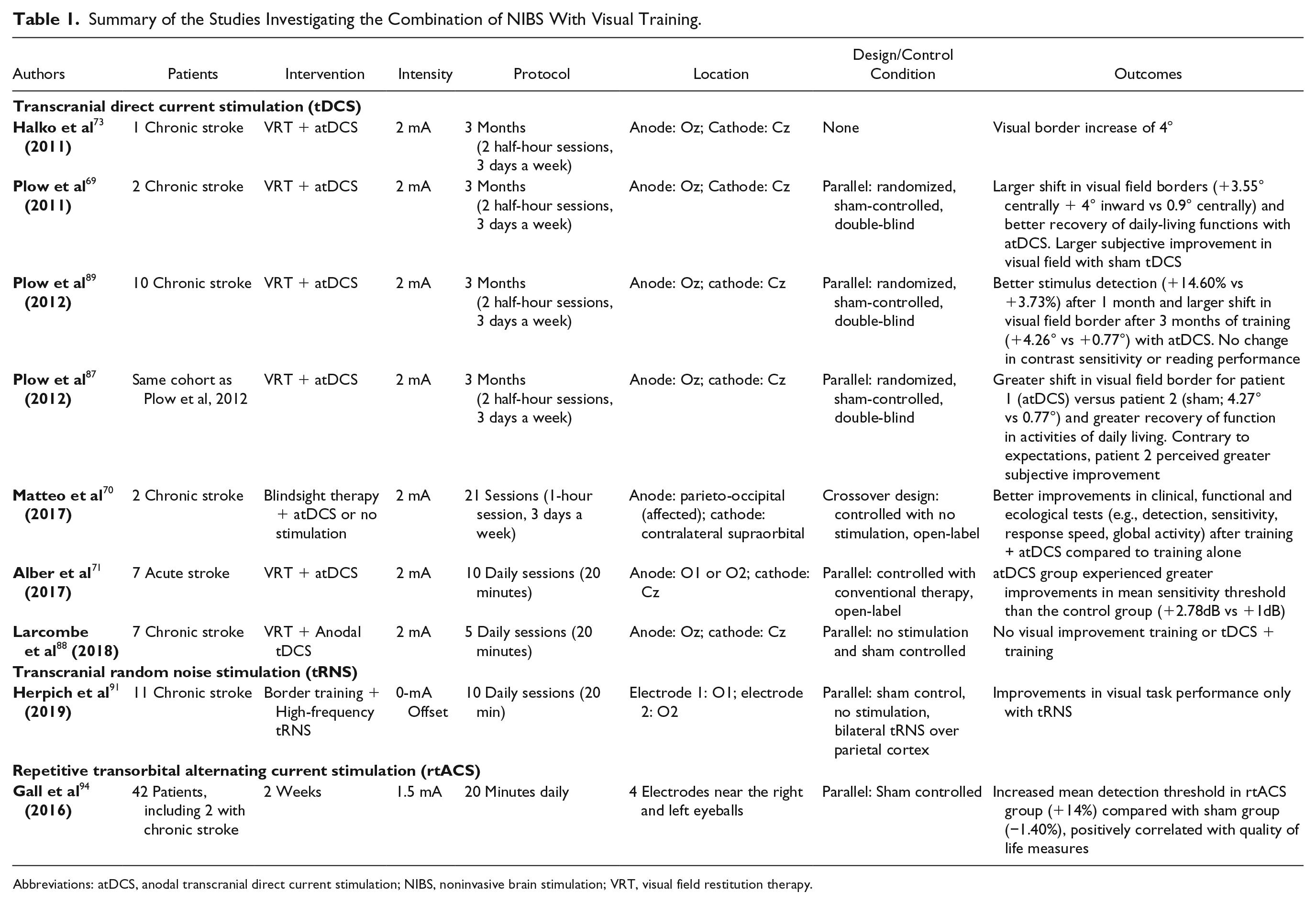
Abbreviations: atDCS, anodal transcranial direct current stimulation; NIBS, noninvasive brain stimulation; VRT, visual field restitution therapy.
Transcranial Direct Current and Random Noise Stimulation
Transcranial direct current stimulation (tDCS) delivers a steady current to the brain through saline-soaked sponge electrodes. 72 The classical montage for visual field recovery is the following, the anode electrode is placed along the midline, over the Oz position (based on the 10-20 EEG coordinate system), and the cathode is placed along the midline, over the Cz position. 73 Using this setup, results on healthy volunteers showed bidirectional changes in excitability of the visual cortex, as indicated by visual-evoked potentials (VEPs), after anodal and cathodal tDCS (increase and decrease, respectively).74-76 Behaviorally, significant anodal tDCS effects on visual contrast sensitivity, especially in low signal-to-noise conditions, has been reported, 77 whereas performance using suprathreshold stimuli was unchanged 78 (see also Behrens et al 79 and Kraft et al 80 ). These effects are rather short-lasting, most likely explained by long-term potentiation/depression (LTP/LTD)-like effects as revealed by pharmacological studies in humans81,82 and early animal work.83,84 However, the reliance on the interhemispheric competition model of recovery to design tDCS protocols to either promote the ipsilesional hemisphere or downregulate the contralesional hemisphere showed disappointing results for a list of different functions and might not be optimal.62,85,86
A couple of reports described the effects of combining visual retraining paradigms with anodal tDCS of the occipital cortex.69,70,71,73,87,88 The first single case study showed a visual border increase of 4° after 3 months of training (2 half-hour sessions, 3 days a week), based on VRT combined with concurrent tDCS. In addition, functional magnetic resonance imaging (fMRI) measurements during a motion discrimination task revealed perilesional activity at baseline, which shifted toward the occipital cortex after training. This “reactivation” was spatially congruent with the location of the highest electrical field induced by tDCS. 73 The same group next applied VRT and tDCS (1 sham, 1 active) in 2 chronic stroke patients with right side hemianopia. Perimetry revealed a larger shift in visual field borders in the patient receiving active tDCS. This was reported with better recovery of daily-living functions within this case study. However, the patient with sham stimulation perceived greater subjective improvement in the visual field. 69 The same protocol was applied to a larger sample size of patients (n = 12). 89 Again, the subjective perception of visual field change was larger in the VRT/sham group despite significantly greater expansion in the visual field for the VRT/active tDCS group. Measures of quality of life did not change for either group.
In the same cohort, a complex sequential pattern of improvement was found with respect to the sham group. Improved visual accuracy was found to be accelerated with real tDCS (ie, occurring within the first month), whereas the 3° extension of the visual field took place only at the end of the 3 months of training, suggesting that the beneficial translation to the affected visual field is a longer process. 87 Interestingly, Alber et al 71 demonstrated the applicability and acceptance of the treatment in the early stage poststroke (approximately 66 [±50] days poststroke). The absence of a parallel, double-blind, sham-controlled design largely limits the conclusions, but these authors reported a larger improvement in visual field in the tDCS/VRT group compared with historical controls. 71
Additionally, studies have combined “blindsight training” with tDCS. Matteo et al 70 showed, in a 2-case crossover report, better scores in clinical instrumental, functional, and ecological assessments after parietal-occipital tDCS combined with blindsight rehabilitation compared with rehabilitation alone. 70 Recently, however, Larcombe et al 88 demonstrated no behavioral improvement in 7 patients after 5 days of training to a motion direction discrimination task combined with anodal tDCS. 88 This study specifically targeted the geniculo-extrastriate pathway, and the fMRI results showed in particular a decrease in activity in the area hMT+. This finding could reflect a normalization of the commonly reported overactivation of the ipsilesional cortex when moving stimuli are presented to the sighted hemifield. 90 The causal link with a potential long-term visual improvement needs to be evaluated.
Recently, another form of DCS has been tested in 5 patients with chronic cortical blindness because of a lesion in V1. 91 Transcranial random noise stimulation (tRNS) consisted here in frequencies of alternating current ranging from 101 to 640 Hz with electrodes located bilaterally over the primary visual areas. tRNS was compared with sham stimulation and no stimulation in patients. The results showed that tRNS enhanced perceptual learning after only 10 days of training, whereas sham stimulation and no stimulation did not alter the learning curve. The effects persisted for 6 months after the end of the intervention. Interestingly, the authors also included healthy controls and further compared the effects of bilateral visual tRNS, bilateral parietal tRNS, bilateral anodal tDCS, sham stimulation, and no stimulation. They reported that anodal tDCS hampered perceptual learning, whereas tRNS boosted task performances compared with the 3 other conditions that were pulled together. Although this pilot study is promising, these data leave a few open questions that need to be addressed in future works. For instance, what are the exact mechanisms driving the enhanced perceptual learning after tRNS or to what extent do these behavioral effects translate into functional benefits and visual field recovery in patients. Interestingly, tRNS combined with perceptual learning has also been recently introduced to treat amblyopia, a neurodevelopmental disorder characterized by several visual deficits. Positive effects were reported after high-frequency tRNS on a trained and untrained visual function (contrast sensitivity and visual acuity). 92 One proposed mechanism to explain tRNS effects is stochastic resonance, implying that the neural noise induced by hf-RNS could enhance the weak response of neurons receiving input from the amblyopic eye during training.
Overall, the clinical trials combining transcranial electriacl stimulation (tES) with visual retraining in patients with hemianopia consist of case reports or small cohort studies, which sometimes lack proper sham controls. Although the interim results are promising, the suggested additional benefit of tES needs to be further assessed in larger samples of patients and well-controlled study designs.
Repetitive Transorbital Alternating Current Stimulation (rtACS)
Transorbital stimulation of the brain has been tested within few ophthalmological diseases, including stroke.93,94 rtACS consists of delivering small electrical currents to the eyeball area to stimulate the visual pathways to the striate cortex. 95 During rtACS, frequency ranges are applied between low-α and flicker fusion frequencies (ie, able to induce a constant perception of light instead of distinct flickering phosphenes). EEG power spectra analysis revealed increased α power at occipital sites.96,97 However, it should be noted that, in these studies, sham and active groups were not completely matched in etiologies and sample sizes or the conditions were not matched in terms of sensation of flickering light in response to rtACS.
Transorbital stimulation also seems to modulate functional connectivity. Bola et al 98 suggested that short- and long-range α coherence correlated with better detection abilities and processing speed after 10 days of active rtACS compared with sham rtACS. A large clinical observational study on rtACS that included 446 patients with optic nerve lesions showed that a 10-day course of rtACS improved visual acuity and increased the size of visual fields by 7.1% and 9.3% in the right and left eyes, respectively. Again, the EEG spectral profile computed from the 24 available data sets showed increased α power after rtACS. 95 This study was an open-label trial with no control group. Therefore, the results have to be interpreted within these limitations. A recent 7-T fMRI study on 1 stroke patient revealed that after 10 days of rtACS treatment, visual activity (activation size and coherence) in both hemispheres was changed with respect to different visual stimuli, suggesting that rtACS might modulate neuronal activity. 99
So far, only 3 randomized controlled studies, with questionable sham conditions, have shown that rtACS can improve vision restoration in patients with chronic optic nerve damage while inducing EEG aftereffects. rtACS in rats suggests that it might be efficient at an early stage of the disease, in particular to prevent retinal axons from degenerating.100,101 However, minimal activity of the residual visual system is required for rtACS to activate the brain and be therapeutically efficient. 102 Of note, there is an inevitable shunting of currents in the skin near the cones and rods of the retina that might alter the perception of flashes by eliciting phosphenes. 103 These statements, together with the limited level of evidence in patients and recent challenging articles 104 warrant further work to better understand the underlying effects of rtACS and eventually improve its effects. Furthermore, though rtACS seems to improve patients suffering from vision deficits after optic nerve damage, 97 the effects are less clear in patients suffering from hemianopia caused by focal brain lesions. 101
In summary, 3 distinct NIBS approaches have been tested so far in rather monocentric studies with smaller samples of patients with hemianopia (tDCS, tRNS, and rtACS). This might bear a risk of positive results bias. Therefore, further confirmatory studies are essential to determine the potential of these interventions for clinical application (see Figure 2). The level of evidence is insufficient for clear recommendations or a proper clinical deployment. Confirmatory, larger-sample, controlled, randomized, and double-blinded trials are needed to prove the beneficial role of the current NIBS protocols in visual rehabilitation to bring them from bench to bedside into the daily clinical world and to determine to what extent longer-term (weeks to months), clinically relevant effects in daily life can be reached. Furthermore, a better consideration of how the intact and partially damaged visual system processes information is necessary to optimize stimulation parameters and improve visual functions.
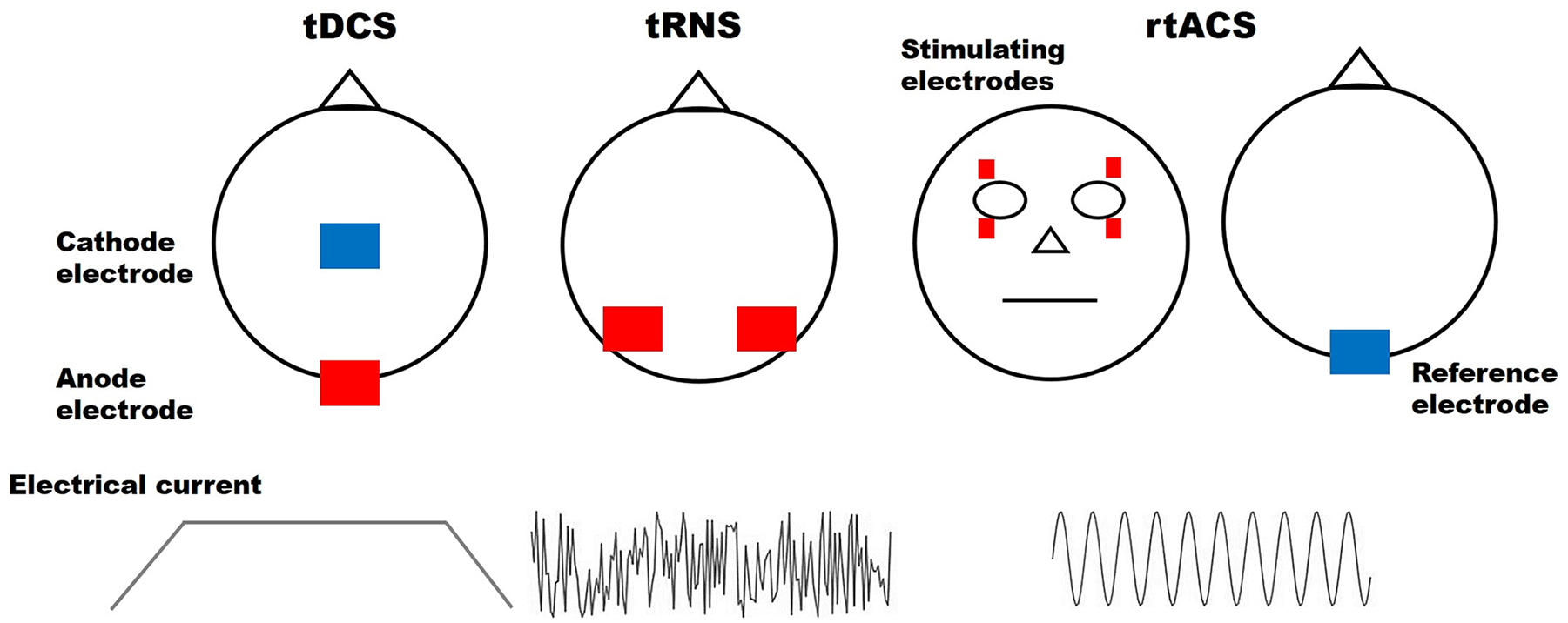
Conventional noninvasive brain stimulation approaches used for visual field recovery.
Dynamical and biologically informed stimulation protocols might provide larger effect sizes and longer-lasting benefits and might further reduce potential side effects. In the following section, we will introduce the theoretical and conceptual grounds of a new brain oscillatory–based approach for NIBS to restore vision.
Modulating Oscillatory Activity Through Individualized non-NIBS to Restore Vision
The Oscillatory Signatures of Visual Perception
Rhythms in neural activity are ubiquitously measured in the brain, with oscillatory patterns associated with specific cognitive operations. 105 Modulation of α power (7-13 Hz) has been consistently shown during visual processing. For instance, when attention is directed to internal representations (eg, during visual imagery), α power increases with attention.106-109 In contrast, when attention is directed to external visual events, α power decreases in the visual cortex.110,111 This release of inhibition (ie, suppression of α-band activity or event-related desynchronization) is assumed to reflect better information processing and to be driven by inputs originating from higher-order areas.112,113 Complementarily, γ-band oscillations are thought to modulate perception by driving visuospatial attention, as shown in humans 114 and monkeys. 115 Cognitive hypotheses about the functional role of γ oscillations in the visual cortex posit that they mediate perceptual binding, attention, or feedforward signaling. 116
A number of models have been proposed to describe how oscillatory coupling properties might mediate the control of signal flow among anatomically connected networks, allowing selective communication. 117 Among them, the group of Wolf Singer obtained experimental evidence supporting the binding by synchrony hypothesis, stating that coordinating mechanisms are provided by the synchronization of neuronal activity by phase locking of network oscillations. 118 Later the communication through coherence theory (Fries 119 ) stipulated that interregional communication is established when the oscillatory activities between neuronal pools oscillate with a stable phase difference.119,120
Complementarily, the gating by inhibition theory 107 suggests that the interareal communication is established by actively inhibiting the pathway not required for the task. This would essentially be associated with regional-specific inhibition mediated by α activity modulation,107,113,121,122 which in turn, allows an increase in γ power in the regions that are relevant to the task. In an attempt to reconcile the 2 last concepts, Bonnefond et al 123 suggested that interareal communication is mediated by nested oscillations. Precisely, phase synchronization of oscillations at lower frequencies (<20 Hz) controls the transfer of information. The cross-frequency interaction with higher γ rhythms (>40 Hz) acts as a temporal frame to express specific neuronal firing. 123
Studies on the role of synchronized oscillations in visual attention and visual perception provide further information into how synchronized low-frequency activity (α and β oscillations) might mediate top-down feedback and how γ activity might reflect feedforward interactions (see Figure 3A).124-128 In nonhuman primates, Richter et al 129 recently used Granger causality measures to demonstrate that elevated top-down α/β-band inputs directly promote feedforward stimulus–induced γ-band processing, leading to enhanced detection of the selected stimulus with a specific phase relationship.
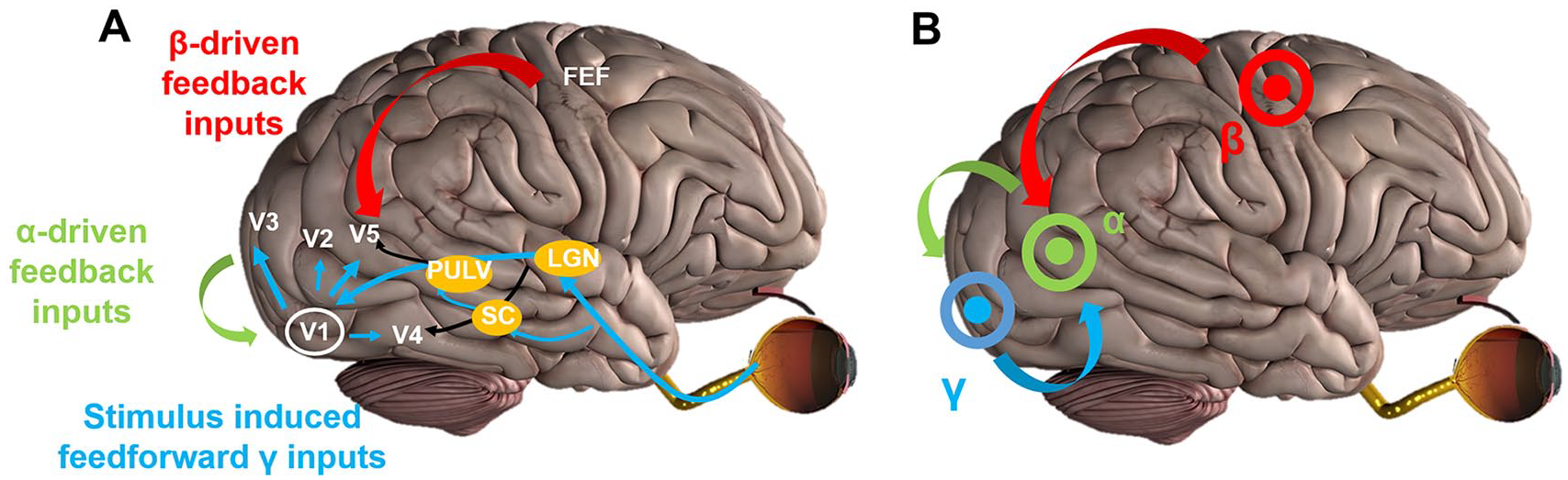
A. Simplified cross-frequency interactions within the visual system. B. The novel multisite cross-frequency oscillatory-based model to restore visual functions.
In conclusion, α-γ interactive oscillations are orchestrated by coupling properties specific to the visual processing steps. These findings indeed demonstrate the behavioral relevance of cross-frequency interactions in the visual system. Given the functional hierarchy derived from the interregional interactions in the α and γ bands, we think that promoting the visuoattentional network by selectively stimulating α/β top-down neural inputs from associative visual areas to V1 while entraining γ bottom-up inputs via cross-frequency interactions might lead to improvement of processing of visual inputs. Ultimately, this might significantly support retraining in patients with hemianopia and drive functional recovery to a much better level.
A New Oscillatory Based Stimulation Framework to Restore Vision in Patients
Concretely, we propose a new paradigm based on coentrainment of interregional oscillatory activity to resynchronize and reorchestrate feedforward and feedback interactions using multifocal cross-frequency transcranial alternating current stimulation (tACS) to enhance residual visual functions and promote recovery in stroke patients (Figure 3). tACS is a unique tool to noninvasively modulate oscillatory activity by sending sinusoidal currents through the scalp.130,131 For instance monofocal occipital tACS at an individual α-frequency has been shown to selectively enhance oscillatory EEG activity in the α-band. 132 tACS appears to be well suited to not only modulate local oscillatory activity, but also long-range connections in a frequency-specific manner, relevant to visual perception. 116
Recent data suggest that vision deficits secondary to a stroke might not only be caused by the primary tissue damage, but also by a change in interregional communication or functional synchronization in the underlying brain networks.98,133-135 These abnormalities appear to be localized within the visuoattentional network and to affect specific frequency bands involved in visual processing, such as α oscillations. 136 In a patient with homonymous hemianopia, γ-band oscillations in the occipitoparietal region have been associated with awareness of a near-threshold stimulus in the blind hemifield, attesting that there are still some residual top-down attentional driving interactions. 137 Then, based on the electrophysiological data reviewed above, our proposal relies on an accurate mixture of brain signals, at the correct frequencies and timings, to achieve an efficient transmission of information. Precisely, 1 minimal configuration would include 2 entries. Primary visual areas would receive sinusoidal currents at high frequency using a high-definition electrode montage to promote feedforward inputs. 138 Stimulating these relatively close areas might be optimally done using a 2-center surround electrodes combination, driven by 2 synchronized 2-channel stimulators, ensuring a good spatial separation of the electric field patterns. Time-locked with an optimal phase lag to the first stimulator, a second one would target higher-order visual areas with high-dimension (hd)-tACS and apply low-frequency signals to promote feedforward interactions. A third stimulated area could be the frontal eye field, known to exert an impact on the attentional modulation of stimulus-induced γ oscillations via high-β oscillations.128,139 In summary, this orchestration of oscillatory signals might be possible using multisite tACS, ideally with distinct stimulators and each stimulated area having its own electrode reference, allowing good focality and better control for the stimulation parameters, 140 each node applying the relevant oscillatory signals in a phase-dependent manner (see Figure 3B).
Further Development: Toward Individualized Closed-Loop Approaches
A further step toward biologically motivated NIBS is a better prediction of individual tACS effects. This can be done on the level of determining biomarkers (eg, degree of structural integrity in the visuoattentional network after stroke) to phenotype each specific patient (eg, to predict whether the patient will respond to the intervention or not). But prediction of response can also be determined in relation to the ongoing brain activity and its actual state when the bursts of tACS are applied. To this end, online electrophysiological recordings might provide valuable feedback on the systemic consequences of the intervention and enable one to take into account the predicted individual response to brain stimulation to ultimately personalize the stimulation parameters.141,142
To individualize as many tACS parameters as possible (eg, location, intensity, phase), the most straightforward alternative is to develop customized profiles of electrical activity (recorded, for example, through EEG) linked to a specific function. 143 These profiles ought to be composed of a wealthy range of metrics of oscillatory activity going all the way from voltage amplitude, power density, and so on, or phase relationships such as intertrial phase consistency, toward more complex measurements, such as connectivity expressed through coherence, Granger causality, and so on.142,144 The occurrence or the combination of one or several of these metrics could eventually serve as a driving informer of the stimulation. The act of informing-controlling the stimulation might function through diverse mechanisms (eg, trigger like, dynamic adapter of oscillations, power/phase compensation), but ultimately, what remains fundamental is the objective over passing the temporal flaws and steadiness assumptions from open-loop systems. 142 Under this premise, a closed-loop system could actually be nourished not only by a single electrophysiological loop, but eventually also by a combination of several loops, depending on the parameters used to inform and the parameters that need to be controlled. For instance, a loop based on behavioral performances could take into account the dynamics of the sensorimotor, attentional, or executive brain circuits requested in the carrying out of a certain task, thus being able to modulate the stimulation. Another alternative would be the inclusion of an automatic adjustment based on a computational model of electromagnetic field dispersion of the stimulation. 145
However, a substantial list of challenges must be addressed before the implementation of any of these systems. Especially because, from an engineering point of view, the optimization of codes, routines, and models that take into account the huge sensibility and variability of biological systems represents an enormous scientific effort, which is tailored to the experimental context. An explicit example of this is the quest to remove the artifact produced by the electrical stimulation on the EEG recordings without taking out enormous quantities of electrophysiological signals of interest. Although attempts have been made, none has been successfully proved nor reproduced.13,137,146 Some attempts of open-loop tACS or closed-loop protocols went around the artifact issue by using short durations of stimulation (a few tACS cycles) and then recorded the immediate EEG aftereffects.147,148
Conclusions
A new promising target to treat visual field defects is the application of multifocal, synchronized oscillatory activity mediating visuoattentional processing, which appears to be altered after a postchiasmatic lesion. Considering that cross-frequency phase synchrony between α phase alignment on higher visual areas and local γ oscillations in the primary visual cortex underlies the incorporation of stimulus representations into the focus of attention, we propose a novel multisite cross-frequency tACS protocol to restore the α/γ coupling in patients following a stroke affecting the visual system.
Furthermore, it becomes more and more clear that “one suits all” therapy concepts in medicine are not optimal. 59 Personalized NIBS strategies tailored to individual patients using individual spectral signatures (but also lesion site and size etc), for instance, might enhance the interventional effects significantly. Thus, individualized, multifocal and cross-frequency brain stimulation interventions can be designed and even applied in a state-dependent or closed-loop fashion to maximize visual recovery.
Footnotes
Authors’ Note
Estelle Raffin and Roberto F. Salamanca-Giron contributed equally to this work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research has been funded by the Bertarelli Foundation (Catalyste BC77O7 to FCH & ER), by the Swiss National Science Foundation (PRIMA PR00P3_179867 to ER), and by the Defitech Foundation (to FCH).
