Abstract
Traumatic brain injury (TBI) is a major cause of morbidity and mortality in the United States, with advanced age being one of the major predictors of poor prognosis. To replicate the mechanisms and multifaceted complexities of human TBI and develop prospective therapeutic treatments, various TBI animal models have been developed. These models have been essential in furthering our understanding of the pathophysiology and biochemical effects on brain mechanisms following TBI. Despite these advances, translating preclinical results to clinical application, particularly in elderly individuals, continues to be challenging. This review aims to provide a clinical perspective, identifying relevant variables currently not replicated in TBI animal models, to potentially improve translation to clinical practice, especially as it applies to elderly populations. As background for this clinical perspective, we reviewed articles indexed on PubMed from 1970 to 2019 that used aged animal models for studying TBI. These studies examined end points relevant for clinical translation, such as neurocognitive effects, sensorimotor behavior, physiological mechanisms, and efficacy of neuroprotective therapies. However, compared with the higher incidence of TBI in older individuals, animal studies on the basic science of aging and TBI remain remarkably scarce. Moreover, a fundamental disconnect remains between experiments in animal models of TBI and successful translation of findings for treating the older TBI population. In this article, we aim to provide a clinical perspective on the unique attributes of TBI in older individuals and a critical appraisal of the research to date on TBI in aged animal models as well as recommendations for future studies.
Introduction
Traumatic brain injury (TBI) is a major cause of death and disability in the United States. Based on recent surveillance data from the Centers for Disease Control and Prevention, in 2013, there were approximately 2.8 million people treated for TBI-related injuries, including 2.5 million emergency department visits (excluding hospitalizations, deaths, and transfers), 282 000 hospitalizations, and 56 000 deaths. 1 Incidence of TBI displays a bimodal age distribution, with peaks in adolescents to young adults (15-25 years of age) and older adults (65 years of age and older).
Historically, elderly individuals have accounted for a disproportionate fraction of TBI-related injuries. In 2013, those aged ≥65 years accounted for about 20% of the emergency department visits, 44% of hospitalizations, and 37% of TBI-related deaths. Furthermore, those aged ≥75 years comprise the largest proportion of hospitalizations and deaths compared with any other age group. 1 Of note, TBI rates have increased in all age groups over the period from 2007 to 2013 (total = 638.8 to 884.2 per 100 000, an increase of 38.4%), but the 2 groups with highest percentage increases were those aged 65 to 74 years (494.1 to 777.0 per 100 000 population, an increase of 57.3%) and those aged ≥75 years (1265.2 to 2232.2 per 100 000 population, an increase of 76.4%). Whereas in the younger age group men are 3 times more likely than women to sustain TBIs, for ages greater than 65 years, the incidence is roughly equal in men and women. However, the incidence of TBI has decreased with age in men who are 70 years and older compared with an increase for women aged 70 to 89 years. 2
In 2003, the aggregate charges for treating a principal diagnosis of TBI in patients aged 65 years and older exceeded $2.2 billion. 3 The National Institute on Aging projects that between 1990 and 2020, the population aged 65 to 74 years will grow 74% under middle series projections, whereas the population <65 years will grow only 24%. 4 With greater life expectancy, there is significant potential for long-standing financial impact of geriatric TBIs, both to survivors and to society at large. As the population continues to age, the costs for care and rehabilitation of those living with TBI have the potential to become a staggering drain on societal resources.
For those who survive TBI, advanced age is one of the strongest predictors of adverse outcome.2,3,5 Older adults are more likely to have complicated TBIs, even with TBIs that would otherwise be classified as mild, with a Glasgow Coma Scale score (GCS) of 13 to 15. For example, elderly patients with GCSs of 13 to 15 had a significantly higher rate of intracranial hemorrhage, at 16% versus 5% for younger adults. 5 Older adults are more likely to be hospitalized after TBI, have extended hospitalizations, and be severely disabled, and a greater proportion are discharged to a nursing facility. Older adults are disproportionately affected in terms of decreased quality of life resulting from temporary or permanent cognitive, physical, and psychosocial disability. TBI in older adults is more likely to be associated with poor memory, dependence, and slowness of thinking when compared with younger adults with TBI. 6 Finally, TBI also increases the risk of new-onset depression in older adults. 7
Beyond the direct impact of TBI on neural function, there is growing evidence that in individuals aged 65 years or older, even mild TBI results in an increased risk for the development of dementia, lowers the age of onset, and alters clinical manifestations. 8 The literature has described the association of TBI and dementia. 9 Most studies suggest that a single moderate to severe TBI is the major known environmental risk factor for Alzheimer disease, imparting a 2- to 4-fold increased risk. 10 TBI has also been suggested to be a risk factor for other neurodegenerative disorders associated with cognitive impairment and dementia, such as frontotemporal dementia, Parkinson disease, and amyotrophic lateral sclerosis. Finally, the age at which TBI occurs is directly related to the probability of developing dementia and amyloid-β plaques. 10
At a time when the population continues to age, pharmacological trials for treating TBI have yielded disappointing results. For example, the use of progesterone for treating TBI acutely was based on a long history of study of its neuroprotective effects in animal models of TBI. 11 After encouraging results in phase II trials, more than 2000 patients were enrolled in 2 large multisite phase III trials. One of these was ended prematurely for futility, whereas the other continued to completion but showed no clinical benefit on the GCS at 6 months.12,13 Although there is still debate regarding the most effective strategies for conducting multisite phase III studies for TBI, it is clear that despite strong evidence in preclinical models, translating efficacy to clinical populations remains a major hurdle.
The Problem: Paucity of Preclinical TBI Studies in Aged Animals
It is not surprising that the majority of TBI clinical trial participants are relatively young because underrepresentation of older participants in clinical trials is a widespread problem.14,15 Often, phase III trials have arbitrary upper age limits, and older adults are frequently excluded on the basis of polypharmacy and comorbid conditions. 16 Recently, in the ProTECT III progesterone trial, although there were no specific exclusion criteria for older individuals and participants were included up to 94 years of age, the median age was 35.
Despite the high incidence of TBI in older individuals, preclinical studies have focused almost exclusively on relatively young participants. To summarize the extant literature, we performed a search using PubMed for preclinical animal models of TBI. Because TBI studies of nonhuman primates are exceedingly rare at any age, and other large animal models of TBI (eg, pigs) have not used aged individuals, this review was restricted to rodents. The following keywords were used: rodent-, rats-, mice-, brain injuries-, aged-, elderly-, and animal model. References from the included studies were also screened. Original research utilizing mice or rats in studies of TBI from 1970 to July 2019 was included. Information pertaining to the TBI injury model, species, age, sex, neuropathological and physiological results, and functional outcomes were tabulated.
To restrict this review to the studies most relevant to TBI in aging, we excluded studies that did not include animals at least 20 months of age. Although any model of the relationship between rodent and human age is rather arbitrary, for simplicity, human age equivalence is often estimated based on a simple proportion of the total lifespan.17-19 The median lifespan for the rat and mouse strains in the included studies ranged from 24 to 30 months. Thus, when human age equivalence is estimated as a fraction of the total lifespan, a mouse or rat at 20 months is equivalent to a human in their mid-50s to mid-60s. We excluded methods articles, review articles, in vitro studies, ischemic and hemorrhagic stroke models, and other models not specifically designed to mimic TBI (eg, cut/stab brain injury, cryogenic lesions, excitotoxic lesions).
Surprisingly, this search resulted in only 33 articles over this period that included rats or mice at least 20 months of age (Table 1). If the criterion age was reduced to 18 months, only 4 additional studies were found (italicized references in Table 1). Thus, only a tiny fraction of animal model studies of TBI have been conducted in aged animals. The paucity of such studies and the slow translation to clinical practice of the findings highlight a vital need to assess the current research scope of TBI in animal models and their relevance to the elderly population. This perspective article aims to identify relevant variables of TBI in older individuals that are currently inadequately replicated in animal models, so that we may suggest new directions for research and ultimately improve translation to clinical practice.
Aged Animal Studies of TBI.
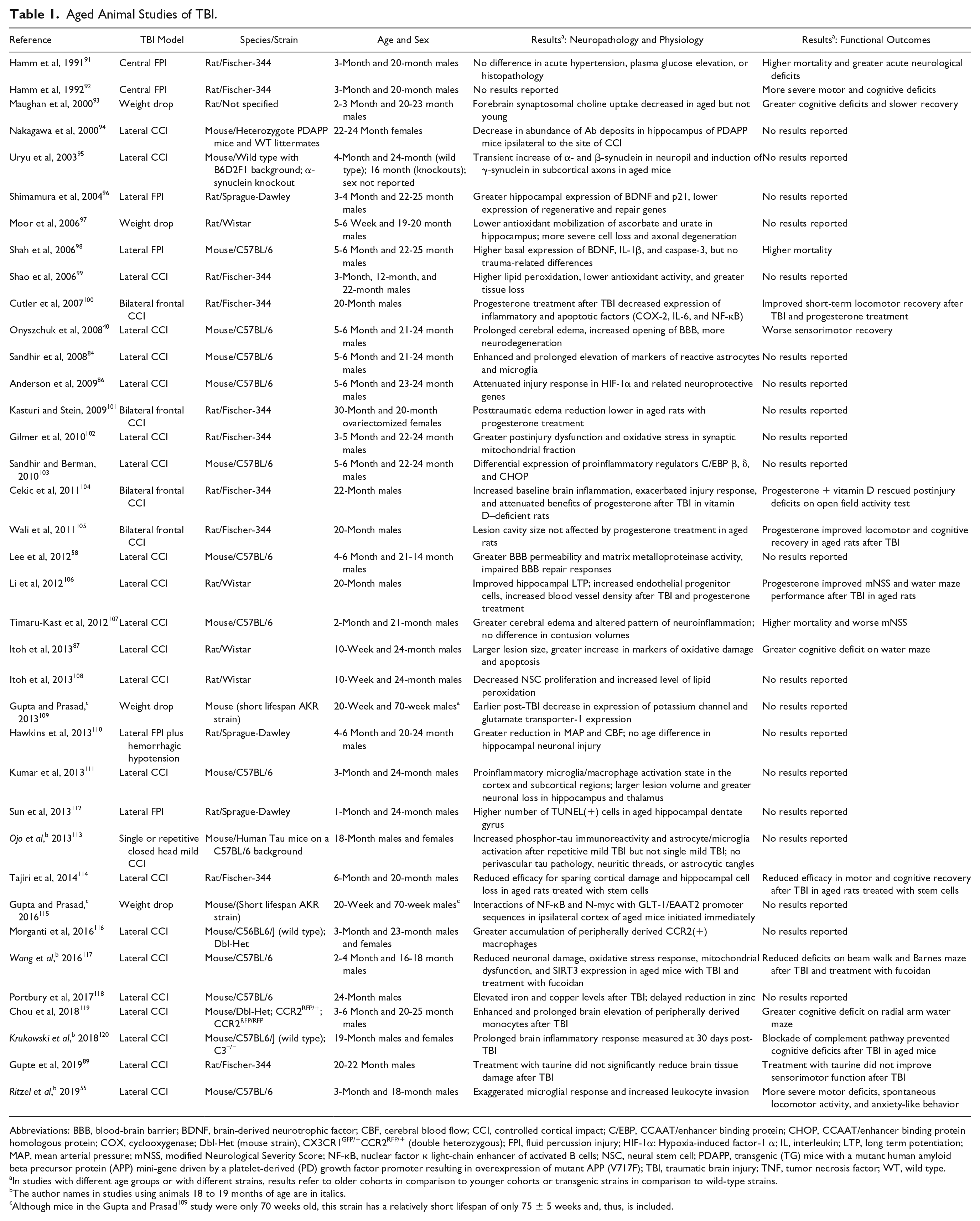
Abbreviations: BBB, blood-brain barrier; BDNF, brain-derived neurotrophic factor; CBF, cerebral blood flow; CCI, controlled cortical impact; C/EBP, CCAAT/enhancer binding protein; CHOP, CCAAT/enhancer binding protein homologous protein; COX, cyclooxygenase; Dbl-Het (mouse strain), CX3CR1GFP/+CCR2RFP/+ (double heterozygous); FPI, fluid percussion injury; HIF-1α: Hypoxia-induced factor-1 α; IL, interleukin; LTP, long term potentiation; MAP, mean arterial pressure; mNSS, modified Neurological Severity Score; NF-κB, nuclear factor κ light-chain enhancer of activated B cells; NSC, neural stem cell; PDAPP, transgenic (TG) mice with a mutant human amyloid beta precursor protein (APP) mini-gene driven by a platelet-derived (PD) growth factor promoter resulting in overexpression of mutant APP (V717F); TBI, traumatic brain injury; TNF, tumor necrosis factor; WT, wild type.
In studies with different age groups or with different strains, results refer to older cohorts in comparison to younger cohorts or transgenic strains in comparison to wild-type strains.
The author names in studies using animals 18 to 19 months of age are in italics.
Although mice in the Gupta and Prasad 109 study were only 70 weeks old, this strain has a relatively short lifespan of only 75 ± 5 weeks and, thus, is included.
Challenges to Modeling TBI in Animals
TBI in either humans or nonhuman animal models induces a complex pathophysiological cascade that results in structural damage and functional deficits caused by both primary and secondary injury mechanisms. 20 The primary insult results from direct mechanical injury by penetration or rapid acceleration-deceleration that may result in a brain contusion at the site of injury, widespread shearing of axons, vascular injury, and/or focal damage opposite the side of impact (contracoup injury). Primary insult may lead to other sequelae, including subdural hematoma from ruptured bridging blood vessels, decreased blood flow following increased intracranial pressure, infarction, and brain edema caused by increased permeability of cerebral blood vessels.21,22 Secondary injury manifests over a period of hours to days 23 after the primary injury and is the result of an additional cascade of metabolic, cellular, and molecular events that ultimately lead to brain cell death, tissue damage, and degeneration. 24 Many biochemical derangements occur, including perturbation of cellular calcium homeostasis, increased free radical generation, mitochondrial dysfunction, inflammation, 25 and progressive atrophy of gray and white matter. 24 These events may result in functional deficits, neurobehavioral disorders, impaired memory and executive function, premature cognitive decline, and premature mortality.
Modeling TBI sequalae in aged animals is challenging from a variety of perspectives, including the mechanics of the injury model, species-specific physiological changes with aging, the interaction of comorbidities seen in human TBI patients, and the feasibility of inducing clinically relevant brain injuries in aged animals with respect to cost and availability of aged cohorts, survivability after injury, postinjury care, and ethical considerations. As in any animal model, balancing construct validity and experimental control of relevant variables requires some compromise. Nevertheless, the current models are limited, especially with respect to replicating conditions in older populations. The following paragraphs summarize some of the key issues in TBI modeling in aged animals.
Animal Models of TBI
To study the multifaceted complexities of human TBI, animal models were initially developed to replicate certain pathological components of clinical brain trauma. 26 Most animal studies of TBI utilize carefully controlled procedures carried out under sterile surgical conditions and general anesthesia. Although less common, closed-head injuries also are sometimes modeled in animals to more closely mimic clinical conditions,27,28 and some of these models utilize conscious animals, though no such studies have been conducted in aged animals. 29 Animal models of TBI have enabled detailed evaluation of TBI pathology 22 and functional outcomes as well as the biomechanical, biochemical, and molecular mechanisms underlying brain injury. 30 Improving our understanding of the complex molecular and cellular cascades initiated by brain injury has been a primary focus that has resulted in numerous important insights into brain injury mechanisms and potential targets for treatment.
Simulating all aspects of TBI in a single animal model is not feasible, and thus, animal models of TBI are selective in their focus. 30 Although larger animals may more closely model the mechanical properties of human brain injury, and some models (eg, nonhuman primates) may more closely reflect neurophysiological processes and functional impairments seen in humans, rodent models are still by far the most frequently used in TBI research. However, larger, gyrencephalic brains better mimic the biomechanical and physiological conditions of human brain injury. It has been suggested that the presence of gyri influences movement of the brain within the skull and that gyrencephalic brains experience more deformation and mechanical stress, especially at the depths of the sulci. Brain mass also affects angular acceleration and shearing forces. Although rodent models have provided critical information regarding mechanisms of cell death, inflammation, and regenerative responses to injury, the limitations of small lissencephalic brains in the context of modeling human TBI are many. Large animal TBI models were once common through the 1980s, but they were virtually abandoned primarily because of ethical considerations. However, there has been recent interest in larger animal models, particularly in pigs. 31 Translation of results to clinical populations will require renewed consideration of these large animal models for TBI in general and particularly for developing therapeutic options for TBI in older populations.
Benefits of rodent models include their modest cost, small size, lower regulatory burden, availability of a variety of genetically modified strains, and well-characterized outcome measurements. 32 In addition, the relatively short life span of 2 to 3 years for most mouse and rat strains is a particular benefit for aging research. Of the numerous rodent brain injury models, the most frequently utilized are fluid percussion injury (FPI), controlled cortical impact injury (CCI), impact acceleration injury, and blast injury. Of these, only FPI and CCI have been used in studies of aged animal models. These 2 models, in particular, are advantageous for human translation because it has been shown that they result in delayed seizures, paralleling the long-term clinical problem of posttraumatic epilepsy. But some post-TBI clinical phenomena, such as long-lasting coma, typically are not replicated in rodent TBI models. 33
FPI Model
In the FPI model, after exposure of the skull through a craniotomy, the insult is inflicted by a pendulum striking the piston of a reservoir of fluid to generate a pressure pulse onto the intact dura. 34 The craniotomy may be made either centrally around the midline or laterally over the parietal bone. Severity can be altered by varying the height from which the pendulum is dropped, producing a larger or smaller pressure pulse. The advantages of FPI for modeling TBI are based on its ability to produce brief displacement and deformation of brain tissue, resulting in both focal and diffuse injury, intracranial hemorrhage, blood-brain barrier (BBB) disruption, brain swelling, and progressive gray and white matter damage, similar to human TBI. 35 Disadvantages of the FPI model include the use of anesthesia, complicating the interpretation of the injury pathology, the requirement of a craniotomy, and the inability to replicate skull fracture and widespread contusions, often seen in moderate to severe TBI in humans. Also, mortality is higher in this model compared with other injury models.
CCI Model
In the CCI model, after exposure of the skull through a craniotomy, the injury is delivered by direct impact to the intact dura, causing deformation of the underlying cortex. A pneumatic or electromagnetic impact device is used to drive a rigid impactor onto the dural surface. This model results in acute subdural hematoma, axonal injury, BBB disruption and edema, cortical tissue loss, neurobehavioral dysfunction, and even coma. 35 CCI offers precise control of mechanical factors, such as velocity, angle, and depth of impact, which vary the severity of the injury and the resulting histological and functional outcomes. This better control over injury parameters results in somewhat lower mortality compared with the FPI model. It has been emphasized that CCI should not be used at high severity levels, injuring a large portion of the cerebral hemisphere, because in the clinical setting, such an injury would not be compatible with survival. 33 Finally, like the FPI model, because a craniotomy is required, skull fracture cannot be replicated in the CCI model. Also, anesthesia is required, thus complicating interpretation of pathology.
Impact Acceleration (Weight Drop) Model
Another widely used model of TBI that thus far has not been used to study aged animals is impact acceleration (weight drop). To model closed head injury, the skull of the animal is exposed to a free-falling guided weight, which generates neuronal and axonal pathology concomitant with BBB disruption 36 particularly throughout the major white matter tracts of the cerebrum and brainstem. 4 Severity can be altered by adjusting the mass of the weight and the height from which it falls. The sequelae of impact acceleration resemble the clinical conditions of human diffuse TBI caused by motor vehicle accidents or falls. 36 Given that falls are the leading cause of TBI in older adults, this model would seem to have adequate face validity for studying TBI in aged animals. However, the greater difficulty in controlling impact parameters makes this model less than ideal. As in FPI and CCI, anesthesia is also required.
CHIMERA Model
One major disadvantage of current TBI models with respect to replication of the biomechanics of injuries caused by falls and motor vehicle accidents is that head motion is typically constrained. In particular, rotational forces are not integrated into the model. Recently a Closed-Head Impact Model of Engineered Rotational Acceleration (CHIMERA) was developed at the University of British Columbia. 37 In this model, the mouse head is supported on a foam pad in a supine position, with Velcro straps around the torso. Impact from a piston deflects the head, and after impact, the head returns to its original position. TBI induced with the CHIMERA model in mice produced loss of righting reflex; neurological, motor, and cognitive deficits; anxiety-like behavior; diffuse axonal injury; and microgliosis in white matter. Development of such models that better replicate the biomechanics of injuries seen in older adults are needed.
Blast Exposure Model
A focus on the movement of the brain within the skull in closed head injuries has led to nonimpact TBI models 22 of blast exposure. However, the relevance of blast injury models for TBI in older individuals is marginal because it does not replicate the type of mechanical injury seen in falls and motor vehicle accidents.
In all these rodent models of TBI, functional deficits can be assessed with a variety of cognitive and sensorimotor behavioral tasks. Cognitive impairment is frequently measured via the Morris water maze, Y maze, and radial-arm maze to determine spatial/contextual deficits, and novel object recognition and avoidance tests are used for learning and memory. 38 Sensorimotor impairments can be measured with the bilateral tactile adhesive removal test, skilled forelimb use (staircase test), Rotarod test, beam walk, grid walk, and grip and reaching tests.39,40
Physiological Changes With Aging Relevant for Recovery After TBI
Normal aging is associated with cellular, molecular, anatomical, and physiological changes. An organ system’s ability to effectively regulate and maintain its physiological reserve is paramount to adapting appropriately to stresses. Diseases and injuries in aging are associated with the reduced ability to properly restore a new steady state that is appropriate for the new set of conditions, 41 with increased maladaptive responses to internal and external stresses. In the brain, the hippocampus has been identified as a key component of the stress response, sensing increased glucocorticoid levels and relaying information to the hypothalamus in a negative feedback loop 42 to maintain homeostasis following perturbance. This decrease in function of the feedback loop worsens following TBI because of selective hippocampal cell death and is associated with cognitive impairment. 43 Understanding how decreased physiological reserve in older individuals might affect neurocognitive recovery following injury is lacking, and this area warrants more research.
Molecular and cellular changes that occur in the brain during normal aging may also naturally contribute to a delayed recovery postinjury. 44 Aging leads to a decline in dopamine receptor densities associated with declines in cognitive and motor performance. 45 Furthermore, expression of neurotrophic factors and neurotransmitters may frequently be altered in the aging brain. In particular, changes in brain-derived neurotrophic factor and serotonin may act in concert to regulate neural plasticity, neurogenesis, and neuronal survival in multiple brain regions. 46 Other factors that have been implicated include calcium dysregulation affecting multiple signaling pathways adversely influencing cellular physiology, molecular functions, and cell structure 47 ; mitochondrial dysfunction leading to the endogenous production of reactive oxygen species and apoptosis 48 ; and an increase of monoamine oxidase that is associated with the liberation of free radicals, 49 leading to apoptosis.
Systemic physiological changes that occur during aging may further predispose older individuals to an exacerbated pathophysiological response to brain trauma. Integration between brain function and peripheral metabolism is required for homeostasis and proper adaptations to aging, physical activity, or environmental stressors. Increasingly, it is being recognized that disruptions in these brain/periphery links likely play a critical role in the brain’s response to traumatic injuries. Likewise, alterations in centrally mediated processes after TBI can result in peripheral metabolic changes (in cardiac muscle, skeletal muscle, and the gut) affecting functional outcomes (activity, gait, motor control, postural adaptation) and posttraumatic morbidity.50-56 Examples of age-associated changes that could interfere with brain/periphery metabolic links include the following: (1) reduction in total body water and decreased blood volume leading to a decline in cerebral blood flow, which could exacerbate cell damage and/or death; (2) decreased elasticity and increased rigidity of arterial vasculature blunting the carotid baroreceptor sensitivity, thus increasing the risk of end organ damage 57 ; (3) BBB disruption that is exacerbated postinjury, 58 resulting in more severe traumatic edema, decreased cerebral perfusion pressure, and ischemia; (4) progressive brain atrophy leading to increased space in the cranial vault for accumulation of blood 59 ; and (5) increased adherence of dura to the skull and increased susceptibility of bridging veins to shearing, 3 leading to a greater chance of subarachnoid hemorrhage and/or subdural hematoma formation.
Comorbidity Effects on Aged TBI Outcomes
It is estimated that 80% of all adults aged 65 years and older have at least 1 chronic condition, and 50% have at least 2 conditions, 5 compared with 28% of younger adults with a single medical condition. 60 Thus, an elderly person with a TBI has a greater likelihood of chronic prescription medication use for these long-standing comorbid conditions compared with the younger TBI population. The Centers for Medicare and Medicaid Services report that the most common chronic conditions among Medicare beneficiaries (aged 65 years and older) in the United States were the following: hypertension (58%), high cholesterol (45%), cardiovascular disease (31%), arthritis (29%), and diabetes (28%). Although it is known that these factors negatively affect clinical recovery, they are generally not modeled in animal studies of TBI in aging.61-63 The following examines how common comorbid conditions might negatively affect outcomes in older adults with TBI.
Hypertension
The incidence and prevalence of most cardiovascular disorders increases with age, and cardiovascular disease is the leading cause of death and major disability in adults ≥75 years of age. 64 In a retrospective review of comorbidities in older TBI patients, hypertension was found to be the most prevalent condition. 65 Chronic hypertension in elderly individuals has been shown to disturb the BBB, which leads to changes in passage of molecules into the brain, cerebral edema, white matter changes, astrocyte activation exacerbating the neuroinflammatory cascade, and eventual cerebral ischemia and neurodegeneration.66,67 This is pertinent given the fact that brains of aged mice that were injured via moderate lateral CCI had a more prolonged postacute opening of the BBB than younger mice, with delayed resolution of cerebral edema and greater neurodegeneration that resulted in significant sensorimotor dysfunction. 40
Cardiovascular Disease
Cardiovascular disease, which includes coronary heart disease, peripheral arterial disease, heart failure, valvular heart disease, and stroke, becomes more prevalent with age. 68 Up to 66% of adults aged 75 years or older in the United States and Europe take daily aspirin and/or other antiplatelet drugs 69 to prevent or treat a myocardial infarction, which has a nearly 7-fold higher prevalence among individuals aged 65 to 74 years relative to those aged 35 to 44 years. 68 Intracranial bleeding has been found to progress in 10% of elderly TBI patients on clopidogrel, and these patients were 3 times more likely to be discharged to long-term inpatient facilities. TBI patients on clopidogrel had a 14 times higher mortality rate as compared with those not on antiplatelets. 70
In 2005, atrial fibrillation was estimated to affect 2.2 million Americans. 68 Warfarin is frequently used for the prevention of thromboembolic events in atrial fibrillation, but it has also been linked to an exacerbation of secondary injury in elderly individuals following a TBI. Those on warfarin with minor head injuries (GCS ≥ 13) are 2.7 times more likely to suffer from intracranial hemorrhage, 71 a condition that is strongly associated with 30-day mortality, regardless of age, sex, or coexisting illnesses. 72
Cerebrovascular Reactivity
It is well known that impaired autoregulation and cerebrovascular reactivity (CR) are major risk factors for poor functional outcome after TBI. 73 Several indices have been used to measure CR, including cerebral blood flow velocity, middle cerebral artery cross-sectional area, mean arterial pressure, the pressure reactivity index, pulse amplitude index, and RAC (correlation between pulse amplitude and cerebral perfusion pressure). The most common protocol to examine CR is response to hypercapnia. Importantly, in a multivariate study of 358 TBI patients, subdural hematomas, thickness of subarachnoid hemorrhage, and age were strongly associated with abnormal CR, more so than the gross injury burden. 73 Whereas several animal studies have examined CR in TBI models,74,75 very few studies have examined CR end points in aged animal models. In one rare study of CR in aged rats, CR to hypercapnia decreased, but there were differences between Wistar and Fischer 344 strains. 76 This is a topic that is ripe for further investigation.
Diabetes Mellitus
Diabetes mellitus (DM) prevalence continues to grow across the age spectrum, but its comorbidities and associated mortality are higher in the elderly population. According to Caspersen et al, diagnosed and/or undiagnosed DM affects 10.9 million adults in the US aged 65 years and older, with a projection to reach 26.7 million by 2050.77,78 In a registry-based study, the relative risk of low falls in adults with insulin-treated DM was nearly twice the risk in those without insulin-treated DM. 79 Of those older adults who fall, up to 10% experience a significant injury such as TBI. 80 Again, this is important given that falls are the most common cause of TBI in older adults and a single fall is a major risk factor for subsequent falls. Given their lower reserve, older adults who experience repetitive TBIs experience poorer cognitive and functional recovery following each subsequent injury, with an increased risk of fatal fall-related TBI. Severe TBI with DM has a higher mortality (14%) compared with severe TBI without DM (8.2%), and TBI with insulin-dependent DM has a higher mortality rate (17.1%) than non–insulin-dependent DM. 81 Insulin deficiency may contribute to the increased mortality after TBI, 81 or it may be a result of hyperglycemia, especially persistent hyperglycemia, which has been shown to play a detrimental role in the secondary complications following TBI. Hyperglycemia increases blood viscosity and diffuse small vessel disorders, which results in ischemia and hypoxia in the brain tissues. In addition, hyperglycemia has the potential to induce disruption of the BBB, accelerate cerebral vasospasm, exacerbate edema of endothelial cells, and impair glial cell function. 81
Aged Animal Studies of TBI: Advances and Limitations
Studies of TBI in aged animals, although limited to a small fraction of basic TBI research, have nonetheless led to important advances in our understanding of how aging interacts with complex cellular and molecular pathways of secondary injury. For example, animal studies have elucidated the critically important role of neuroinflammation in both acute pathological and reconstructive response of the brain to injury, similar to the neuropathology in clinical populations. 82 Older brains appear to undergo a homeostatic shift toward a proinflammatory state, a phenomenon termed inflamm-aging. This state describes the low-grade, chronic, systemic inflammation in the absence of overt infection (“sterile” inflammation) and is considered a highly significant risk factor for both morbidity and mortality in elderly individuals 83 following brain injury. TBI studies in aged rats and mice have noted that compared with adult subjects, there was delayed recovery along with prolonged activation of microglia and astrocytes in the hippocampus, with chronic activation of microglia being linked with increased neuronal loss 84 ; higher baseline expressions of cytokines, chemokines, and iNOS that was associated with exacerbated responses to injury 85 ; and a reduced expression of genes in the hypoxia-inducible factor-1 α pathway that exhibit neurotrophic and neuroprotective properties. 86 Thus, it appears that a heightened inflammatory response may contribute to the delayed recovery postinjury. These preclinical findings have formed a critical foundation for future research. However, we note that the absence of clinical variables in studies to date could limit effective translation of findings to human TBI. For example, aspirin and acetaminophen are 2 anti-inflammatory medications frequently used in the geriatric population. How might these medications influence TBI in older individuals? Future studies of common anti-inflammatory drugs in aged animal models will be important to determine potential effects of these treatments on the inflammatory response and TBI outcomes.
TBI studies in aged animals have also revealed that worse outcomes in older subjects may be linked with free radical–induced cell death following a depletion of mitochondrial antioxidant enzymes. Itoh et al 87 demonstrated that in the cerebral cortex and hippocampus of aged rats, significantly higher levels of free radicals are produced when compared with younger adults after TBI. 85 The free radicals induced neuronal and glial cell membrane peroxidation and DNA damage, causing apoptotic cell death and ultimately cognitive impairment. 87 It was also observed that in the focal ischemic area, free radical production was increased by facilitation of the arachidonic acid (AA) cascade. 87
Nonsteroidal anti-inflammatory drugs such as ibuprofen and acetylsalicylic acid (aspirin) are nonselective inhibitors of cyclooxygenase, a key enzyme in the AA metabolic pathway. As previously mentioned, because of the increased risk of cerebrovascular disease with aging, long-term antithrombotic therapy is common, and the most widely prescribed antithrombotic medication is aspirin. In 2016, it was reported that nearly 40% of US adults older than 50 years use aspirin for the primary or secondary prevention of cerebrovascular disease. 88 Consequently, it may be of great importance to determine how inhibition of the AA pathway affects free radical production in the context of TBI in older individuals.
Overall, experimental animal models have clearly been valuable in advancing our understanding of pathophysiological events related to TBI and essential for the identification of novel therapeutic targets. A recent study from our group showed that treatment with taurine, an anti-inflammatory, antioxidant, and anti-apoptotic therapy that had shown promise for TBI in young adult rats, did not show a similar degree of neuroprotection in elderly rats. 89 This highlights the likely influence of age on therapeutic efficacy and the critical need for inclusion of aged animal models in the drug development pipeline for TBI.
To date, the findings from aged animal studies have not specifically contributed to improved clinical care, treatment, or rehabilitation of TBI in the geriatric population. We posit that an important reason for this may be an inadequate replication of clinical variables in aged animal studies. Preclinical studies of TBI are carefully designed for reproducibility, to apply a standardized injury across subjects with age, sex, diet, living conditions, genetic background, and injury parameters all carefully controlled. 35 In the laboratory, heterogeneous injury has the potential to adversely affect statistical significance of results. On the other hand, limiting so many clinically relevant variables that are known to affect human TBI outcome(s) may limit clinical translation. Certain comorbid conditions that frequently accompany TBI and worsen outcomes have been implemented in adult animal models of TBI. Dual TBI and hypoxia models have been developed in FPI, impact acceleration, and CCI, 22 and hypoxia-and-hypotension models have been applied in FPI and CCI. 35 However, such combined neurotrauma studies have not yet been implemented in aged animals. Developing these types of studies in aged subjects to determine interactive effects (eg, between age and hypoxia, age and diet) may better recapitulate TBI pathophysiology in older humans posttrauma.
Recommendations for Aged Animal Studies of TBI
Animal models serve an essential role in TBI research, allowing elucidation of complex cellular and molecular mechanisms of injury and the identification of targets for therapy. For a variety of practical experimental reasons, these studies have primarily focused on young adult male rodents, and research that emphasizes issues particular to TBI in the aged population continues to be meager. Inbred strains of rodent species are extremely valuable as research subjects because of reduced genetic variability. 90 However, the commonly used strains display a limited range of pathology with regard to age-associated comorbidities that exist in humans. For example, normally aged mice do not develop neurodegenerative pathologies and have low prevalence of cerebrovascular disease. Experimental manipulations, such as dietary manipulations, are required to model these conditions in the laboratory.
There is a clear need to improve current practices in regard to the effects of brain injury, particularly in a population that is one of the most vulnerable to the debilitating consequences: the elderly population. It is our belief that the development of an interdisciplinary approach that strongly considers clinical perspectives during the preclinical research phase is vital to an improvement in translational research. It is clear that older age negatively influences outcome after TBI, and the study of TBI in aged animal models remains an area of considerable opportunity for future research. 3 With the elderly population contributing greatly to the epidemiology of TBI, modifications to existing preclinical research practices are imperative to bridge the divide between preclinical studies and clinical application.
From the meager body of evidence on TBI in aged animals, it would appear self-evident that our ability to translate animal findings to aged clinical TBI populations would benefit from several extensions of current models:
Utilize more aged animals in TBI research: Generally, this includes rats and mice older than 20 months. Use a range of “old” animal ages to better understand heterogeneity in the geriatric population.
Utilize injury models that replicate additional mechanical properties of TBI in older humans: This should include studies that model concussion and mild-to-moderate TBI, representing the most common type of clinical injury.
Model TBI in combination with comorbidities frequently seen in older humans: This includes comorbidities such as hypertension, DM, cardiovascular disease, and impaired CR. Some of these comorbidities exist in inbred and transgenic animals, but others will need to be induced in the laboratory setting via environmental conditions, such as those that predispose animals to obesity and inactivity. Modeling multiple comorbidities should also be considered.
Model TBI in combination with chronic medications most commonly taken by older individuals: This might include preinjury antiplatelet therapy (eg, aspirin) that have been linked to worse outcome after TBI.
Study the TBI response over a comprehensive period from acute to chronic post-TBI period.
Develop better age-specific functional outcome measures for aged animals.
Include biomarkers as outcome measures in animal studies that are translatable to humans: Imaging end points that can validate age-related changes in CR, tissue perfusion (arterial spin labeling), brain connectivity (diffusion tensor imaging, resting-state connectivity), and brain metabolism (magnetic resonance spectroscopy) will be particularly useful.
Consider the use of larger animal models of TBI, including animals with gyrencephalic brains, such as sheep, pigs, and nonhuman primates.
Use both sexes in aged animal studies of TBI.
Translation of preclinical research to clinical application continues to be challenging. A variety of animal models of TBI have been developed to reproduce different elements of human TBI. Improving translational success may require remodeling of existing approaches to animal TBI research based on real-world clinical experience, including increased attention to effects of interacting variables and comorbidities on morbidity, mortality, and recovery. Our hope is that the recommendations presented, if fully implemented into research practice for this understudied population, will ultimately translate to improved patient care and effective strategies to improve outcomes and quality of life for older patients with TBI.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by funding from the National Institutes of Health Grants R01 NS030853 (RJN) and R21 NS091920 (JLH).
