Abstract
Objective. Leftward optokinetic stimulation (OKS) is a promising therapeutic approach for right-hemisphere stroke patients with left hemispatial neglect. We questioned whether the putative neural basis is an activation of frontoparietal brain regions involved in the control of eye movements and spatial attention. Methods. We used functional magnetic resonance imaging to investigate brain activations during OKS in acute right-hemisphere stroke patients (RHS, n = 19) compared with healthy control subjects (HC, n = 9). Based on neuropsychological testing we determined the ipsilesional attention bias in all RHS patients, 11 showed manifest hemispatial neglect. Results. In HC subjects, OKS in either direction led to bilateral activation of the visual cortex (V1-V4), frontal (FEF) and supplementary (SEF) eye fields, intraparietal sulcus (IPS), basal ganglia, and thalamus. RHS patients’ activations were generally reduced compared with HC. Nevertheless, leftward OKS bilaterally activated the visual cortex (V1-V4), FEF, SEF, IPS, and thalamus. The neural response to OKS was negatively correlated with patients’ behavioral impairment: The greater the individual attention bias/neglect the weaker the brain activations. Conclusion. In RHS patients, leftward OKS activates frontoparietal regions (FEF, IPS) that are spared from structural brain damage and functionally involved in both oculomotor control and spatial attention. This may provide a neural basis for the known therapeutic effects of OKS on hemispatial neglect. In acute stroke stages, reduced activation levels correlating with neglect severity indicate functional downregulation of the underlying dorsal attention network. Therefore, chronic RHS patients with less severe neglect after recovery of network disturbances may be more suitable candidates for OKS rehabilitation.
Introduction
Hemispatial neglect is a common cognitive disorder following unilateral stroke that represents a severe deficit of directed spatial attention and orientation. Patients fail to be aware of objects in the contralesional hemispace and do not orient spontaneously to the affected side. 1 As an underlying pathophysiology of neglect, it has been proposed that the spatial attention in neglect patients and their egocentric spatial reference is abnormally shifted toward the ipsilesional hemispace.2,3 In the acute stage, about 2/3 of all right-hemisphere stroke patients exhibit hemispatial neglect symptoms and in about 50% of these patients they persist up to the chronic stage.4,5 Hemispatial neglect is known to be a major prognostic factor for poor functional outcome after a stroke, 6 which has provoked an intensive search for effective treatment strategies.7,8
Repetitive optokinetic stimulation (OKS) counts among the few interventions that can induce enduring and functionally relevant positive effects in patients with left hemispatial neglect.9,10 In these patients, leftward OKS was able to significantly reduce neglect behavior, whereas rightward OKS even aggravated neglect symptoms slightly.11,12 OKS is assumed to reduce neglect behavior by reorienting the patients’ subjective straight ahead orientation (ie, the egocentric spatial reference) via overt shifts of gaze and covert attentional shifts toward the contralesional hemifield.9,12-14 While the neural basis in right-hemisphere stroke patients is still unclear, the assumption has been made that the effects are mediated by OKS-related activations of multiple brain regions involved in the control of smooth pursuit eye movements and saccades as the two components of an optokinetic nystagmus.9,15 Some of these oculomotor regions, such as the intraparietal sulcus (IPS) and the frontal eye fields (FEF), have been shown to be also activated during tasks requiring shifts of spatial attention, revealing almost a congruent overlap of attention- and eye movement–related activations 16 These frontoparietal regions (IPS, FEF) constitute a dorsal attention network that is usually spared from structural damage in hemispatial neglect patients. 17 However, its functional disturbance is correlated with neglect severity in the acute stage and its recovery is related to the remission of neglect in the chronic stage,18-20 which makes it an interesting target region for rehabilitation approaches.
Functional magnetic resonance imaging (fMRI) studies in healthy subjects provided first evidence that OKS can induce activations in these frontoparietal regions (FEF, IPS) as well as in the primary and associated visual cortices, insula, basal ganglia, cerebellum, and brainstem.21-25
The goal of our fMRI study was to investigate whether optokinetic stimulation is able to activate these cross-functional frontoparietal regions (FEF, IPS) in right-hemisphere stroke patients despite their structural brain damage and functional network disturbances. If so, this could provide a putative neural basis for the therapeutic effect of OKS on neglect symptoms observed in previous clinical trials.10-12,26 As opposed to the findings in healthy subjects, yielding almost symmetrical OKS-induced brain activations in both hemispheres independent of the stimulus direction,21,23 we expected that there would be direction-specific and hemispheric differences in right-hemisphere stroke patients due to their unilateral structural brain damage. Finally we asked whether patient characteristics, such as the severity of neglect behavior or the size of the stroke lesion, may predict the degree of OKS-induced neural responses (fMRI activation strength), in order to obtain a potential marker that would allow a priori identification of patients as responders or nonresponders for the OKS rehabilitation approach.
Materials and Methods
Participants
The experiment was approved by the Ethics Committee of the University of Lübeck (no. #12-064) and carried out in accordance with the Declaration of Helsinki. All participants gave their written informed consent.
We initially recruited 28 acute patients (mean age 69 ± 10 years, 39% female) from the Stroke Unit of the University Hospital in Lübeck, who were right-handed, had a first ever ischemic stroke in the right hemisphere, no other neurological disease, normal or corrected to normal vision and no contraindications for MRI.
As control subjects, we recruited 10 healthy right-handed, age-matched volunteers (68 ± 11 years, 60% female), who had no history of stroke or neurological complaints, normal or corrected to normal vision and no contraindications for MRI (healthy controls, HC).
Because of head motion artifacts during the fMRI investigation, limited cooperation (eyes closed, not following the stimuli) and a secondary (cerebellar) stroke detected by the MRI in 1 case, we had to exclude 9 RHS patients and 1 HC subject postenrollment (see flowchart in supplementary material s-figure 1). At the end, the data from 19 RHS patients (mean age 66 ± 10 years, 37% female) and 9 HC subjects (66 ± 12 years, 67% female) went into final analysis.
There was no statistically significant difference between the RHS and the HC group with respect to age, t(26) = −0.06, P = .95, and gender, t(26) = −1.48, P = .16.
In the patient group, the time since stroke onset ranged from 4 to 16 days with a median time of 7 days. According to the consensus paper of the Stroke Recovery and Rehabilitation Roundtable, 27 the acute phase of stroke extents over the first week, while the subsequent early subacute phase ranges from 1 week to 3 months after stroke onset. Since most of our patients were investigated in the acute or very early postacute stage of stroke, and for the sake of clarity, we report our patients throughout the text as acute stroke patients.
Further details on patients’ individual clinical characteristics can be found in Table 1.
Patients’ Clinical Characteristics and Neuropsychological Test Results. a
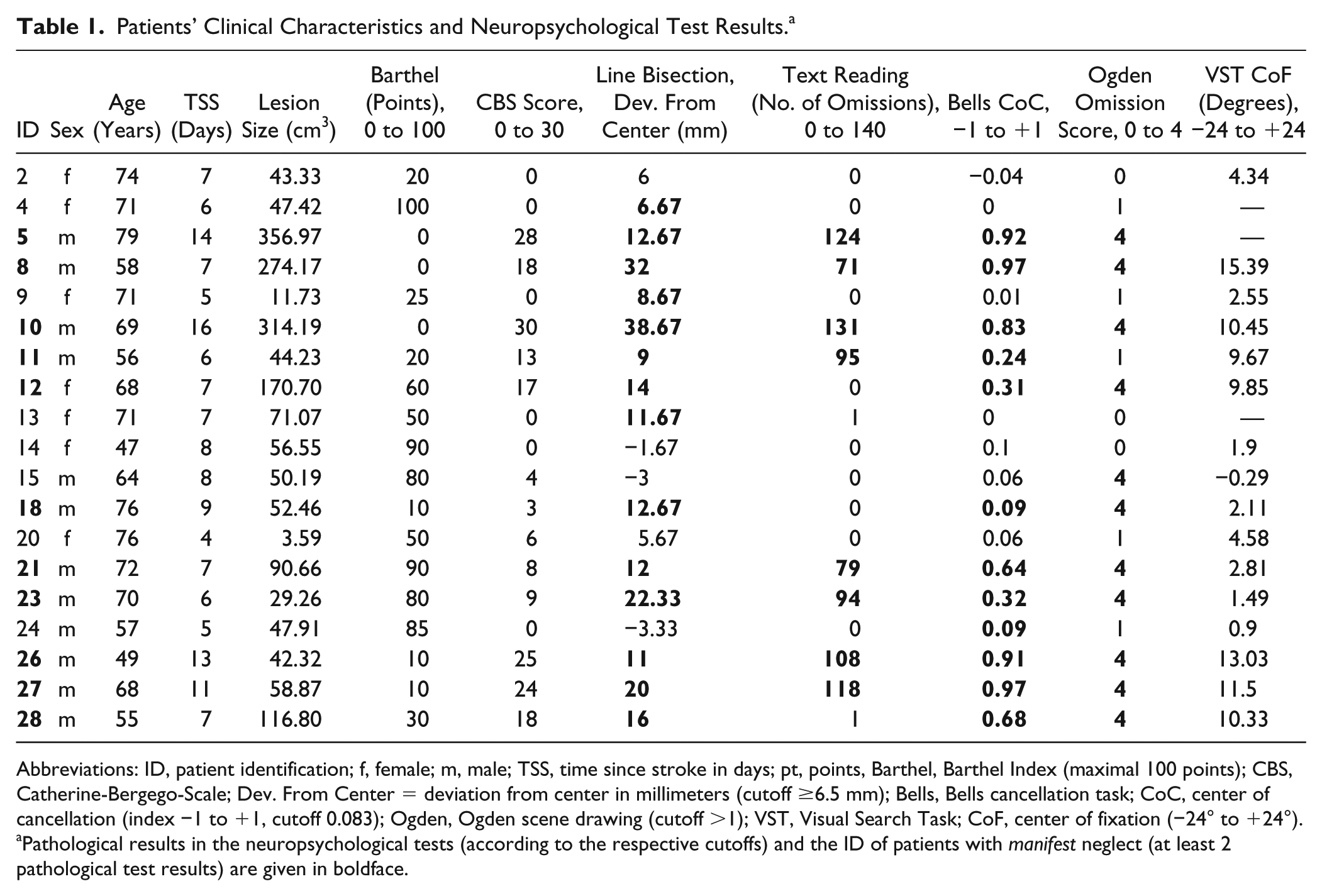
Abbreviations: ID, patient identification; f, female; m, male; TSS, time since stroke in days; pt, points, Barthel, Barthel Index (maximal 100 points); CBS, Catherine-Bergego-Scale; Dev. From Center = deviation from center in millimeters (cutoff ≥6.5 mm); Bells, Bells cancellation task; CoC, center of cancellation (index −1 to +1, cutoff 0.083); Ogden, Ogden scene drawing (cutoff >1); VST, Visual Search Task; CoF, center of fixation (−24° to +24°).
Pathological results in the neuropsychological tests (according to the respective cutoffs) and the ID of patients with manifest neglect (at least 2 pathological test results) are given in boldface.
Neuropsychological Test Battery for Spatial Neglect and Functional Disability Scales
All patients were assessed by the use of an established paper-and-pencil tests battery for spatial neglect.28,29 This test battery not only comprised the most sensitive subtests for spatial neglect, including the line bisection test, 30 text reading,30,31 the bells cancellation task, 32 and a figure copying task (Ogden-Scene), 33 it also allowed rapid assessment of the ipsilesional spatial attention bias in right-hemisphere stroke patients at the bedside.
We used previously determined cutoffs for pathological performance in the different subtests (see footnote in Table 1) 28 and defined patients to have manifest neglect according to previous studies,5,29 when they exhibited pathological performance in at least 2 of these tests, which applied to 11 patients in our cohort.
Patients were additionally introduced to a computerized visual search task outside the MR scanner, in which they had to search for a paperclip on a computer screen showing a naturalistic image of a desk (details on the procedure have been published elsewhere 34 ). This task has been proven very sensitive in detecting even mild subclinical signs of lateralized inattention, otherwise missed by established paper-and-pencil tests and clinical observation. 34 Eye movements were recorded in order to obtain information on an ipsilesional oculomotor/attention bias during overt shifts of spatial attention (as opposed to the rather reflexive pattern of an optokinetic nystagmus in the OKS task). Based on the horizontal fixation distribution of eye movements on the screen, we calculated the center of fixation (CoF), which corresponds to the median x-position on the screen from where 50% of all fixations were located on the left and 50% on the right.
Finally, functional independence in the activities of daily living was measured by the Barthel Index 35 and the investigator version of the Catherine-Bergego-Scale (CBS) 36 for a more neglect-specific functional assessment.
Statistical analyses were performed using the SPSS software package (Version 21.0; IBM Corporation, Armonk, NY).
Optokinetic Stimulation and Oculography in the Magnetic Resonance Scanner
Participants were laid comfortably in the MR scanner with ear protection and the head fixed to reduce head movement artifacts. The stimulus pattern was presented by a projector on a screen that could be seen by the participants via a mirror attached onto the head coil. The field of view was covering 37.5° horizontally and 21.7° vertically. Eye movements were recorded using a fiber-optic limbus tracking device37,38 with a sampling frequency of 500 Hz and a best spatial resolution of 0.2° of visual angle. A computer program (LabView, National Instruments, Austin, TX) was used to display the eye movement signals to check the participant’s performance online.
After the recording, the oculographic data from the participants were used to analyze the subtypes of optokinetic nystagmus (OKN). Based on its frequency, the participant’s OKN was assigned to reflect either a look (≤1 Hz) or a stare (>1 Hz) nystagmus or a combination of both types.39,40
The stimulus pattern consisted of randomly distributed red dots (n = 70) coherently moving on a black background with a velocity of 10 deg/s in either leftward or rightward direction (s-Figure 2 in the supplementary material). The stimulus was presented using Psychophysics Toolbox (Version 3.09)41,42 in Matlab (R2014b, MathWorks, Natick, MA). Participants were instructed to select one dot, to pursue it with the eyes until it disappears at the left (or right) edge of the display and then jump to a new dot on the opposite side. This instruction was chosen because such active tracking of an optokinetic stimulus induces a look-nystagmus, which has been shown to elicit significantly higher cortical activations in the fMRI 23 and to yield greater modulatory effects on the behavioral level 9 than passive viewing of the stimulus (stare-nystagmus).
The experiment was performed in a block design. Three different conditions were presented in a pseudo-randomized order: (a) leftward OKS (leftward motion of multiple dots), (b) rightward OKS (rightward motion of multiple dots), and (c) baseline (one central stationary dot). There were 18 activation blocks (9 leftward, 9 rightward) and 12 baseline blocks, each block had a duration of 18 to 24 seconds.
MRI Data Acquisition
Functional data, consisting of T2*-weighted echo planar imaging sequences, and structural images (T1-weighted 3D fast spoiled gradient echo sequence and T2 fluid-attenuated inversion recovery [FLAIR] sequence) were acquired using a 3.0 tesla Philips Achieva MRI scanner. More detailed information on scanning parameters can be found in the supplemental material (Section s1.1. MRI Data Acquisition).
MRI Data Analysis
For structural lesion analysis the lesion of every stroke patient was marked on their individual FLAIR or T1 image and normalized using cost function masking in SPM. Subsequent illustrations of lesion overlaps were created by MRIcron software (http://www.mricro.com/mricron). Detailed information on this procedure can be found in the supplementary material (Section s1.2. Lesion Analysis).
The functional MRI data were preprocessed and statistically analyzed using SPM 8 implemented in Matlab (MathWorks, Natick, MA). The preprocessing included slice time and motion correction, segmentation, coregistration, normalization, resampling, and smoothing. Cost function masking was implemented for coregistration of lesioned brains. Detailed information on preprocessing can be found in the supplementary material (Section s1.3. Preprocessing of Functional Data). After preprocessing all participants with head movement greater than 3 mm were excluded from the analyses. Statistical parametric maps were calculated for each participant on a voxel-by-voxel basis using a general linear model with one regressor for each of the 3 conditions. The following contrasts were defined: (a) leftward OKS versus baseline, (b) rightward OKS versus baseline, and (c) leftward OKS versus rightward OKS. After model estimation, the obtained first-level contrast images from each subject were entered into a second-level analysis using a 1-sample t test at each voxel for every group. Additionally, we performed 2-sample t tests at each voxel for the comparison of the 2 groups (RHS vs HC) using the first level contrast images from each subject.
A region of interest (ROI) analysis was performed for the comparison of leftward versus rightward OKS within the relevant oculomotor regions for the RHS group. One customized oculomotor network mask (OMN-ROI) was created using the blood oxygen level dependency (BOLD) activation that HC showed during leftward OKS as compared to the baseline, which constituted the condition with the strongest activation (see Figure 1a). This OMN mask included bilateral regions in the visual cortex (VC), frontal and supplementary eye fields, inferior and superior parietal lobule (including IPS), middle frontal gyrus, and the right-hemispheric cingulate eye field. The OMN-ROI mask also complied with the OKS-induced brain activations that were previously described in healthy participants.21-24 In the supplemental material, we further describe an analysis on the percentage of voxels within the OMN mask that were structurally damaged, that is, affected by the patients’ individual stroke lesions (Section s1.2 Lesion Analysis).
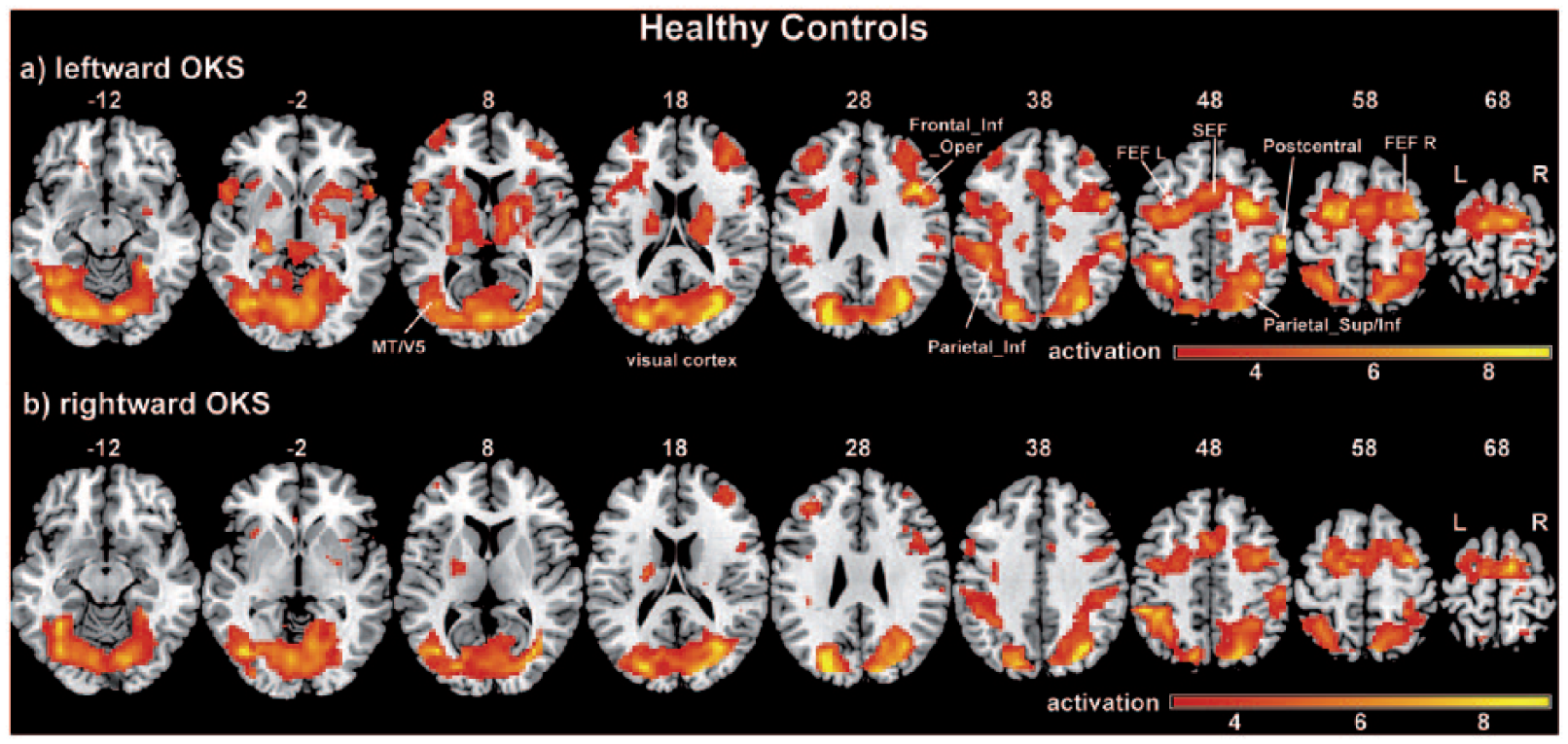
Activation maps of leftward and rightward optokinetic stimulation for healthy control subjects.
We additionally implemented an analysis of laterality effects by performing a paired t test with the flipped and non-flipped brain of every subject as described by Bense et al. 21
The results where visualized using MRIcron (http://www.mricro.com/mricron). Anatomical labeling was performed using xjView (http://www.alivelearn.net/xjview) and the Anatomy Toolbox. 43
If not stated otherwise all resulting maps are corrected for multiple comparisons using the false discovery rate (FDR) correction 44 and considered at a P value of .05 with a cluster size of 10 voxels.
Correlation of OKS-Related Brain Activation With Behavioral Data
The magnitude of leftward OKS-related brain activation was correlated with RHS patients’ behavioral data and lesion size. Therefore, the percentage of activated voxels within our customized OMN-ROI mask (see ROI analysis in the previous section) was extracted for all stroke patients using marsbar. 45 Spearman rank-order correlation coefficient and linear regression analyses where then computed for exploration of a causal correlation between the percentage of activated voxels with lesion size and different behavioral parameters (CBS score, center of fixation in the visual search task). The calculations were performed using the SPSS software package.
Results
Clinical, Neuropsychological, and Eye Movement data
Individual patient results of neuropsychological paper-and-pencil test battery, the computerized visual search task and functional disability scores are presented in Table 1 for every single patient and the lesion overlap for the RHS group is presented in supplementary s-Figure 3.
We could further analyze the eye movements of 13 RHS patients and eight HC subjects, recorded during the MR scanning procedure. The oculographic data of the other 6 patients and 1 HC subject did not allow a deeper analysis due to interfering artifacts in the scanner. Four RHS patients performed a mixture of look- and stare-nystagmus, the other 9 patients and all HC subjects exclusively performed a look-nystagmus (s-Table 2 and s-Figure 4 in the supplementary materials).
Brain Activation Patterns During Optokinetic Stimulation
Within-Group Analyses
Healthy Control Subjects
The activation results for the HC subjects are presented in Figure 1 and supplementary s-Table 3. The contrasts leftward OKS versus baseline (Figure 1a) and rightward OKS versus baseline (Figure 1b) showed nearly symmetrical significant BOLD activation clusters bilaterally in the VC (including V1-V4), the FEF, the SEF and the IPS (right occipital activation merging to right IPS) as well as in the thalamus, the putamen, and in the middle frontal gyrus. Furthermore, the right supramarginal gyrus were also activated during the stimulation in both directions. Leftward OKS elicited additional contralateral (right) activation of the CEF, ipsilateral (left) activation in the inferior and superior temporal gyrus and bilateral activation in the inferior frontal gyrus. Rightward OKS elicited additional bilateral activation in the middle frontal gyrus and activation in the right parahippocampal gyrus.
When directly contrasting leftward OKS and rightward OKS, in order to look for direction-specific effects of OKS, we did not find any significant cluster in the whole brain and in the ROI analyses.
There were also no hemispheric laterality effects, since contrasting left hemisphere activation versus right hemisphere activation during the different stimulation types did not exhibit any statistically significant clusters.
Right-Hemisphere Stroke Patients
The activation results for RHS patients during OKS in either direction are shown in Figure 2 (together with the structural brain lesions projected onto one brain template) and in Table 2.
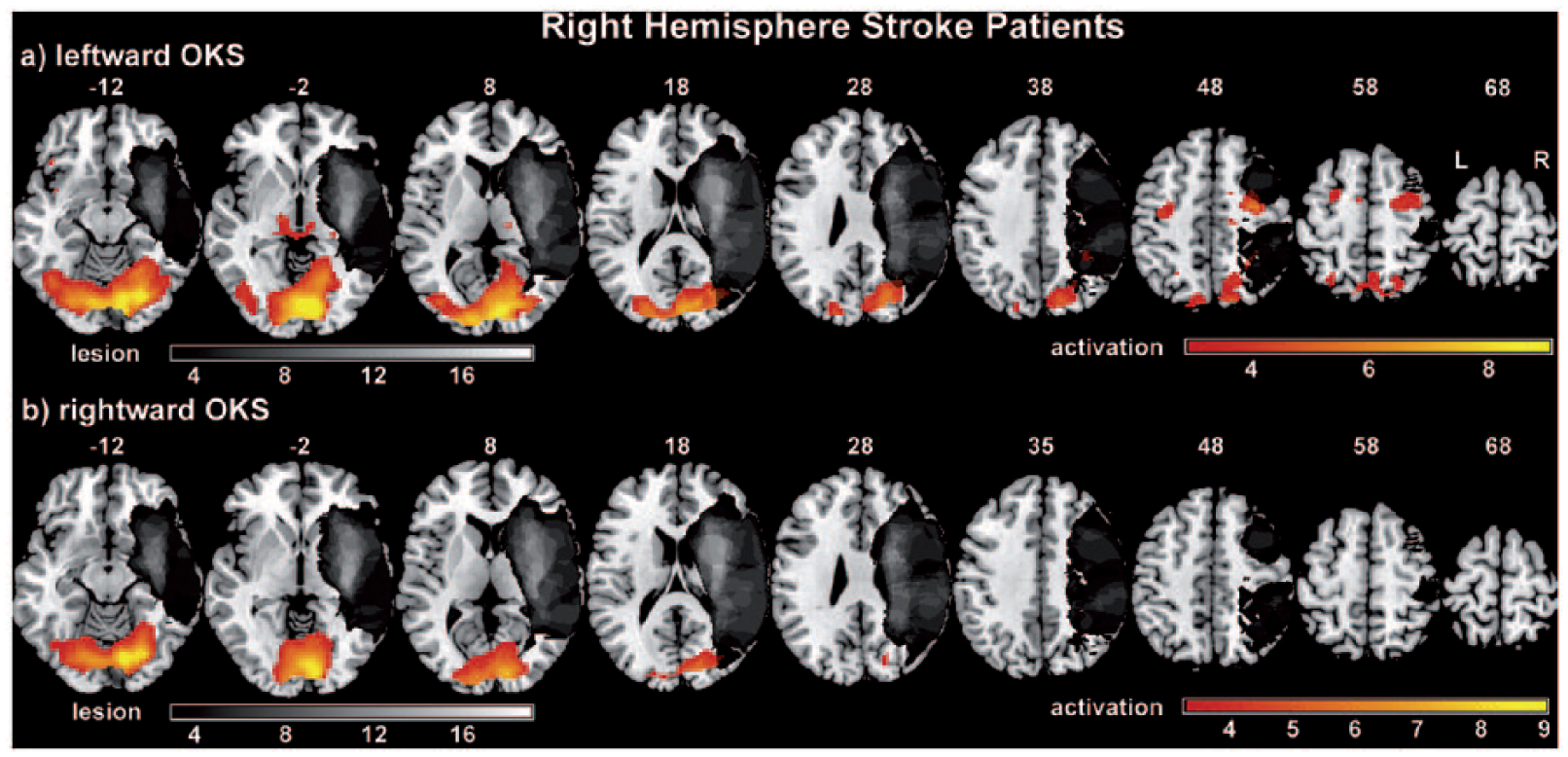
Activation maps of leftward and rightward optokinetic stimulation for right-hemisphere stroke patients.
Brain Activation in Right-Hemisphere Stroke Patients During Optokinetic Stimulation. a
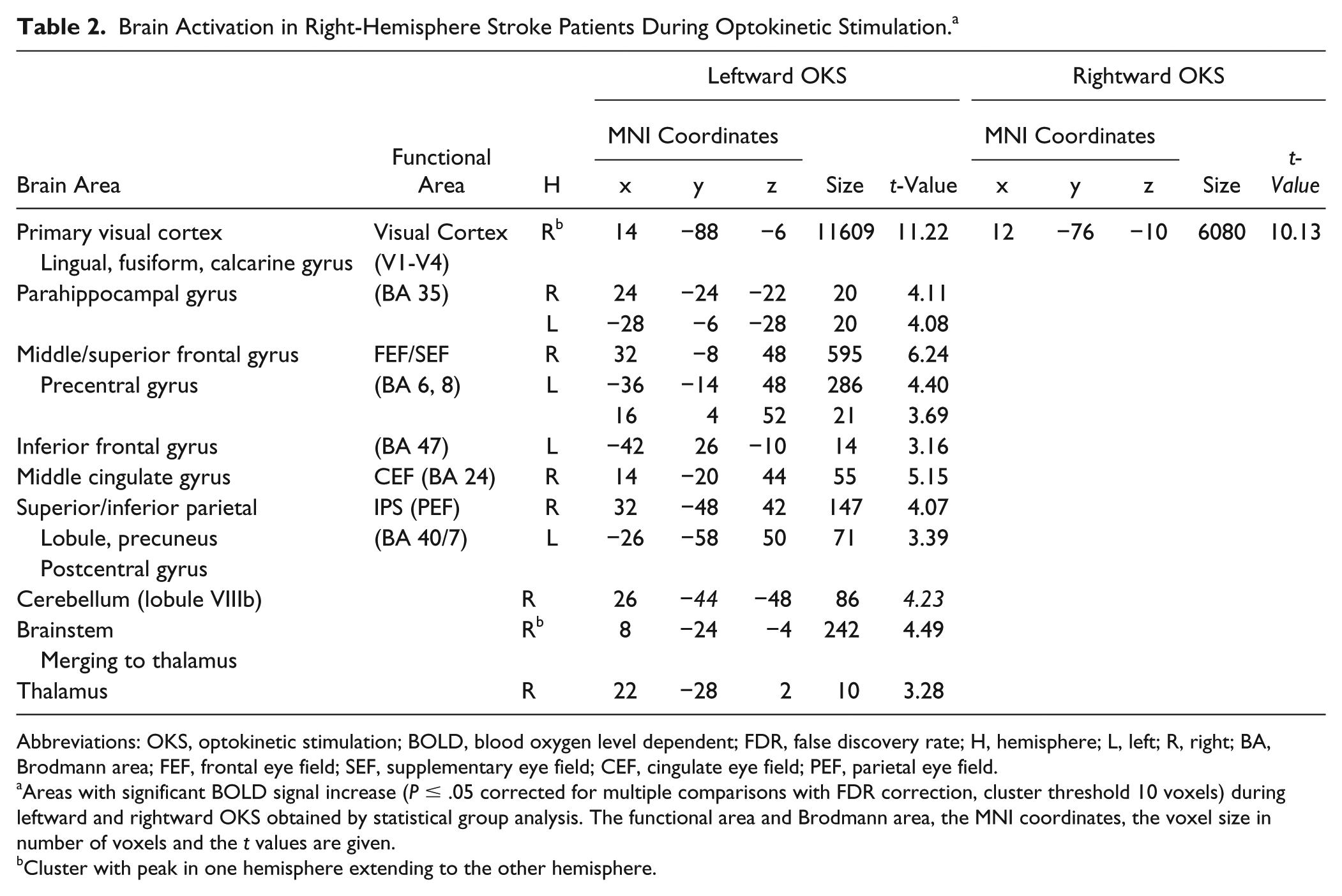
Abbreviations: OKS, optokinetic stimulation; BOLD, blood oxygen level dependent; FDR, false discovery rate; H, hemisphere; L, left; R, right; BA, Brodmann area; FEF, frontal eye field; SEF, supplementary eye field; CEF, cingulate eye field; PEF, parietal eye field.
Areas with significant BOLD signal increase (P ≤ .05 corrected for multiple comparisons with FDR correction, cluster threshold 10 voxels) during leftward and rightward OKS obtained by statistical group analysis. The functional area and Brodmann area, the MNI coordinates, the voxel size in number of voxels and the t values are given.
Cluster with peak in one hemisphere extending to the other hemisphere.
Leftward OKS (Figure 2a) led to bilateral activation in the visual cortex (V1-V4), the FEF, SEF, PEF, and parahippocampal gyrus. Activation of the CEF, the thalamus, and the brainstem were found in the right hemisphere. On the group level, significant activations during rightward OKS were only found for the primary visual cortex (V1-V4) (Figure 2b). However, on visual inspection of the individual contrast maps of the patients (s-Figure 6 in supplementary material), there are at least 9 patients who showed bilateral activation of the oculomotor regions in the rightward OKS condition as described above for leftward OKS (see also s-Figure 5 for comparison).
Congruently, we did not find any significant differences in the activations when directly contrasting the leftward and rightward OKS condition on the group level in the whole brain and in the ROI analyses.
Finally, there were no hemispheric differences (statistically significant clusters) when contrasting left hemisphere activation versus right hemisphere activation within the different stimulation conditions.
Between-Group Analyses
Healthy controls versus right-hemisphere stroke patients
The comparison of HC subjects and RHS patients for the condition leftward OKS versus baseline revealed significant stronger activation in HC subjects for both hemispheres in the visual cortex (V1-V4), the FEF, SEF, and CEF, the anterior cingulate cortex, the putamen and the middle frontal gyrus. Stronger activations for HC subjects were further found in the left hemisphere within the IPS and the medial frontal gyrus, and in the right hemisphere for the supplementary motor area, the pallidum, and the inferior parietal lobule (angular and supramarginal gyrus) (Figure 3a and s-Table 4).
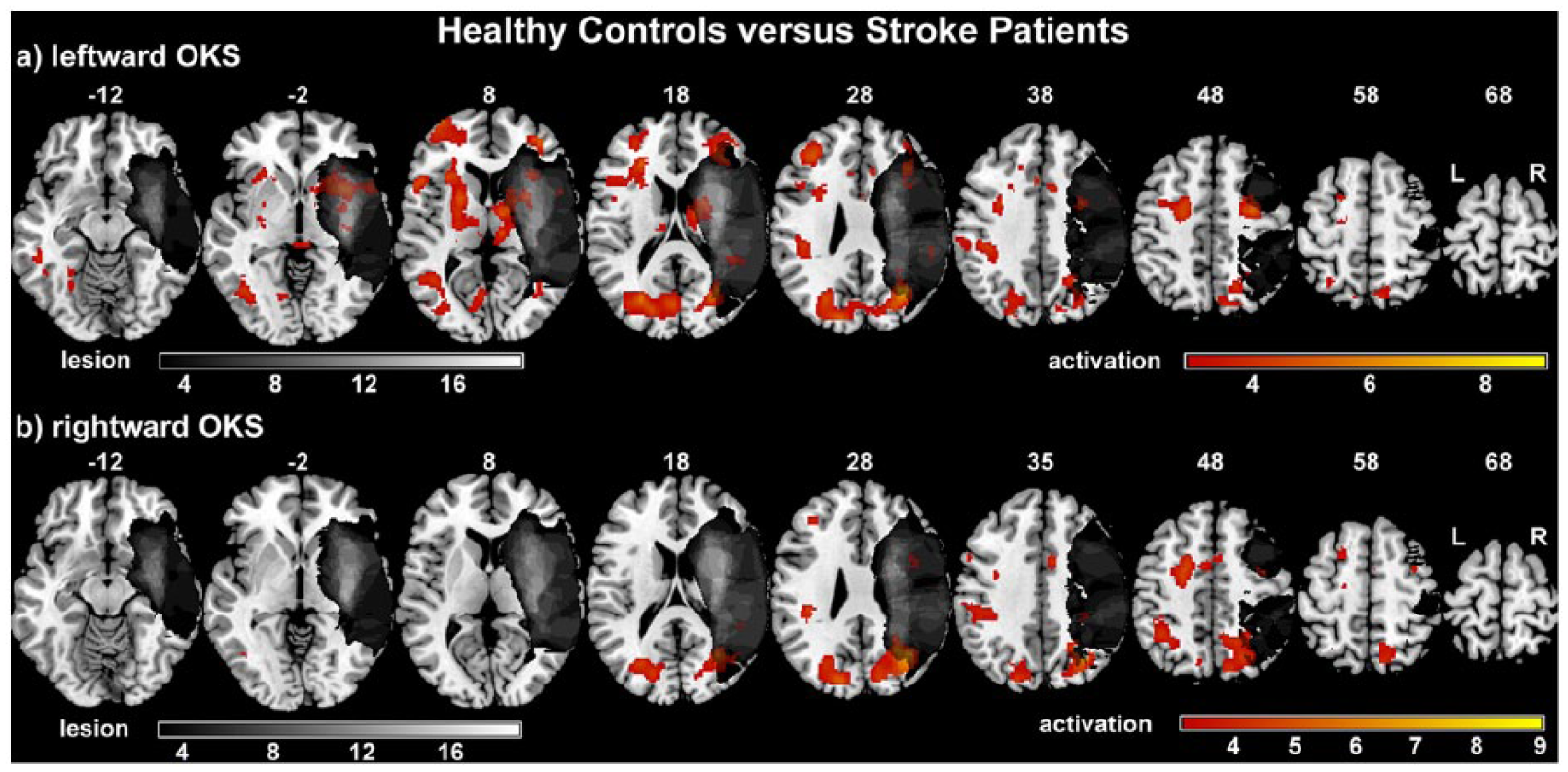
Between-group comparisons of optokinetic stimulation–induced brain activation.
For the corresponding contrast of rightward OKS over baseline, stronger activations in the HC group as compared with RHS patients were found for both hemispheres in VC (V1-V4), FEF, SEF, and middle frontal gyrus, for the left hemisphere in the IPS, the supramarginal gyrus and subgyral frontal lobe and for the right hemisphere in the postcentral gyrus (Figure 3b and s-Table 4).
For all the different within-group and between-group analysis we did not find any significant decrease of activation.
Single-Subject Analyses and Correlation of Brain Activation With Behavioral Data
Our individual RHS patients differed with regard to lesion size, neglect-related functional impairment and the oculomotor/attention bias (Table 1). We therefore repeated analyses of OKS-related brain activation on a single-subject basis and correlated their activations with individual patient characteristics.
For every single RHS patient, the brain lesion and the OKS-related brain activations are presented in the s-Figure 5 and s-Figure 6 of the supplementary material. These maps reflect the range between no significant brain activation (eg, patient F05), isolated VC activation (eg, patient F08) and a widespread activation of the VC and oculomotor regions (FEF, SEF, IPS, CEF) (eg, patient F21). Individual results on the exact percentage of structurally damaged voxels within the OMN-ROI mask are provided in s-Table 1 in the supplementary material.
To determine reasons for the interindividual differences, we performed correlation and linear regression analyzes between brain activation (measured in percentage of activated voxels during leftward OKS in the customized OMN-ROI) and behavioral parameters. Results of the Spearman rank-order correlation and linear regression indicate a significant negative correlation of patients’ brain activation with neglect-related functional disability as reflected by the CBS score (rs = −0.58, P = .009, n = 19) and with the pathological oculomotor/attention bias as indicated by the center of fixation in the visual search task (rs = −0.58, P = 0.019, n = 16). Scatterplots for these parameters are shown in Figure 4. Notably, neither the absolute lesion size (P = .79) nor the percentage of damaged voxels within the OMN-ROI (P = .76) had a significant impact on the strength of brain activation.
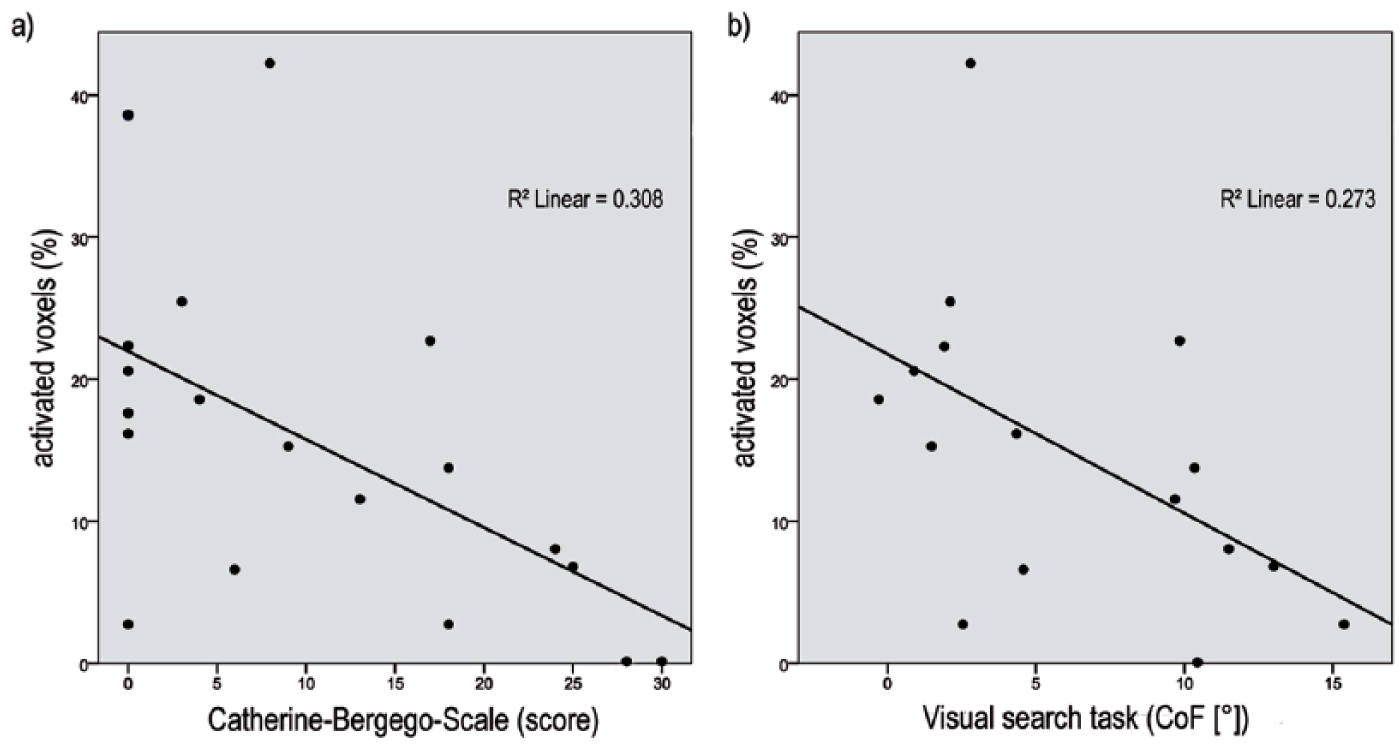
Correlation of brain activation with behavioral parameters.
Discussion
OKS-Induced Brain Activations in RHS Patients Compared With HC Subjects
This is the first study that investigated fMRI brain activations in acute right-hemisphere stroke patients (RHS), with or without hemispatial neglect, compared to healthy control subjects (HC) during leftward and rightward optokinetic stimulation (OKS).
In HC subjects, the optokinetic stimulus in either direction sufficiently activated the visual cortex (V1- V4), the cortical eye fields (FEF, SEF), the parietal cortex (IPS), the basal ganglia, and the prefrontal cortex in both hemispheres. The activations found are in line with previous studies and encompass those brain regions that are functionally involved in a visually induced (optokinetic) nystagmus: visual stimulus perception, following the stimulus with a smooth pursuit eye movement until its disappearance, and execution of a redirecting saccadic eye movement toward a new stimulus on the opposite side of the visual field.21,23,25 In accordance with previous studies, we did not find a hemispheric laterality effect or a direction-specific effect for the different OKS directions in our HC subjects.21,46
Despite the structural brain damage in our RHS patients, leftward OKS led to similar, mostly bilateral activations in cortical oculomotor regions as in HC subjects: the visual cortex (V1-V4), the cortical eye fields (FEF, SEF), and the intraparietal sulcus (IPS).
For rightward OKS, there was a lack of significant clusters in the mentioned oculomotor regions, apart from bilateral activations in the visual cortex. We doubt, however, that these results from the group-analysis indicate a real failure of the stimulus or a loss of smooth pursuit function toward the ipsilesional hemifield in our RHS patients. 47 These explanations are ruled out by our oculographic data on preserved nystagmus generation during the fMRI experiment and the single-subject analyzes that showed significant rightward OKS-induced activations in some of the RHS patients. The negative group result for rightward OKS rather reflects the problem of reduced statistical power due to the high variance in a heterogeneous patient group. In support of that, the direct comparison between leftward and rightward OKS did not reveal significant differences with regards to the strength of brain activations in our RHS patients. Hence, despite the different results in the group analyses for either condition, we cannot necessarily infer from our data a direction-specific effect, that is, that leftward OKS has greater impact on brain activations than rightward OKS in RHS patients.
In line with the results from the HC subjects but in contrast to our expectations, a laterality effect (hemispheric difference) was also not revealed in our RHS patients. Despite their structural brain damage of one hemisphere, there were no significant differences in the functional activation between the 2 hemispheres for either OKS direction. One explanation might be that the OKS task specifically activated cortical regions in RHS patients that laid outside the lesioned areas of the right hemisphere, which will be discussed in the following subsection.
Structural Integrity and OKS-Induced Activation of Dorsal Attention Network Regions in RHS Patients
Beside the structural stroke lesions in ventral temporo-insular brain regions of our RHS patients, leftward OKS was able to activate dorsal frontoparietal brain regions including the IPS and FEF. This is in accordance with the observation that structural lesions in the majority of patients with hemispatial neglect affect those brain regions that constitute the ventral attention network in the right hemisphere, that is, the temporoparietal junction between the inferior parietal lobule and the superior temporal gyrus and the ventral frontal cortex, including the inferior and middle frontal gyrus.17,19 In the same patients, the regions belonging to the dorsal attention network, including the motion-sensitive visual association areas (V3a, MT), the IPS and the FEF, are usually spared from the structural damage 19 . The fronto-parietal regions FEF and IPS have been shown to have a cross-functional role in controlling both visuospatial attention (covert attention shifts) and oculomotor processes (goal-directed eye movements). 16 The functional hybrid and structural integrity of these regions may be the preconditions why an OKS training can influence attentional disorders such as hemispatial neglect in RHS patients via the induction of an optokinetic nystagmus.11,12,15 This issue will be further discussed in the last paragraph of the discussion.
Influence of Individual Patient Characteristics on OKS-Induced Brain Activations
Taking into account the natural heterogeneity of RHS patients with their different sizes and locations of stroke lesions as well as their individual degrees of spatial attention deficits and functional impairments, we performed additional analyzes on OKS-induced activations in dependence of individual patient characteristics. Given a strong relationship between fMRI brain activations and behavior, finding a clinical marker that predicts the neural response (strength of activation) to OKS in RHS patients, could also help to identify patients who are more likely to benefit from OKS training during rehabilitation. We investigated different individual parameters (lesion size, the oculomotor/attention bias during visual search, and the CBS score as an indicator of neglect severity in the activities of daily living) to correlate with the strength of OKS-induced brain activation. Among these parameters, it was not the lesion size but the degree of the ipsilesional attention bias and the neglect-related functional disability that significantly correlated with the strength of OKS-induced brain activations. The higher the degree of neglect-related functional disability and the stronger the ipsilesional attention bias, the smaller were the RHS patients’ brain activations during OKS.
We questioned whether this finding could be attributed to a mere lack of cooperation with deficient OKN generation or the large structural brain damage in the more impaired patients with stronger neglect. A previous study showed, that only the look-nystagmus, induced when subjects actively followed the moving stimulus with their eyes, led to significant activations in cortical oculomotor regions, whereas passive OKN (stare-nystagmus) failed to elicit significant signals. 23 Our eye tracking data of online oculography in the scanner, however, showed that all of our patients performed a look- or at least a combination of look- and stare-nystagmus during the OKS task, ruling out a general lack of compliance or inability to perform the task. An alternative explanation would be an artificial reduction of the fMRI BOLD signal due to the structural brain damage, especially in large stroke lesions. This assumption is firstly challenged by the fact, that lesion size per se was not correlated with the number of activated voxels during OKS. Furthermore, the percentage of lesioned voxels in the OMN-ROI was rather small in the individual patients and it did not correlate with the number of activated voxels in the mask.
Hence, if it cannot be explained by a lack of cooperation in the OKS task or the structural lesions, we may have to attribute the findings of weaker activations in the more impaired patients with a stronger ipsilesional attention bias to specific functional disturbances of the brain in the acute stage of stroke, which will be discussed in the next subsection.
A Potential Neural Basis for OKS Rehabilitation Effects
As discussed above, our study provides first evidence that OKS in RHS patients is able to activate these structurally intact frontoparietal regions (FEF, IPS) that have been shown to constitute not only important cortical oculomotor hubs but also core regions of the dorsal attention network. The fact, that patients’ attentional deficits and related functional impairments, rather than the structural lesion, predicted the strength of OKS-induced brain activations, is in accordance with the finding that neglect patients reveal a functional downregulation of the structurally intact dorsal attention network in their acute stage of stroke.19,20 In support of that, Ramsey et al 48 and Umarova et al 49 have previously shown a reduced fractional anisotropy of the attention networks in another cohort of acute neglect patients. Although not directly tested, it is likely, that our acute RHS patients, in particular those with hemispatial neglect, had such a functional downregulation of their dorsal attention network. This may have led to reduced activations of the cross-functional regions FEF and IPS during the OKS-induced generation of smooth pursuit eye movements and saccades. This hypothesis is further supported by the finding that our acute RHS patients showed generally reduced activations of these frontoparietal as compared with the HC subjects.
However, if the OKS-induced brain activations are especially weak in RHS patients with more severe neglect, how does this comply with the previous reports on beneficial therapeutic effects of OKS in neglect patients?9,11,15,50 And what is the basis for the assumption that a reduced fMRI activation of oculomotor/attention network regions during OKS could indicate a weaker therapeutic effect of repetitive OKS as a rehabilitation intervention for neglect?
Results from previous studies provide some evidence for a general relationship between neglect behavior and task-induced fMRI activation in the relevant cortical regions. Corbetta and colleagues 51 revealed that acute neglect patients, asked to perform covert attention shifts during a Posner task variant, showed a reduction of task-induced fMRI activations in the crucial cortical region of the right superior parietal lobule (posterior IPS, parietal eye field) and that this significantly correlated with the behavioral deficit (proportion of missed targets). Another study by Thimm et al 10 showed, that a 3-week OKS training in neglect patients led to an improvement of performance in a neglect test battery and that this amelioration of neglect symptoms was associated with an increase of neural activity during an fMRI spatial attention task in frontal and parietooccipital regions involved in visuo-spatial attention processes. Hence, we would infer from these studies that reduced fMRI activations in core regions of the oculomotor/attention networks are at least correlated (if not even causally related) with behavioral impairments in neglect patients and with their change under repetitive OKS training. Taking these fMRI-behavior correlations into account, we regard the reverse conclusion as plausible, that patients with no or strongly reduced fMRI activations of oculomotor/attention network regions during OKS may benefit less from OKS rehabilitation.
We have to emphasize, however, that all of our patients were recorded in the acute stage of stroke, within the first two weeks after symptom onset. As described above, these patients may have had an acute functional downregulation of the dorsal attention network, which may have prevented a stronger OKS effect. In analogy, our previous interventional clinical trial on OKS (in combination with hemifield eye patching) was negative for neglect patients in the early poststroke phase. 29 Studies, that showed beneficial effects of repetitive leftward OKS on neglect behavior, were performed in chronic stroke patients.9,10,15,50 We know from previous fMRI studies investigating neglect patients longitudinally,20,51 that a breakdown of the dorsal attention network in the acute stage of stroke is followed by a network recovery at the chronic stage. 20 At this stage, OKS may then be able to better stimulate the frontoparietal regions with their hybrid function of oculomotor and attentional control and thereby support neglect recovery during rehabilitation.15,50
Support comes from recent fMRI studies in chronic neglect patients (usually 6 months after the stroke) that investigated the influence of other neglect interventions, that is, virtual reality training,52,53 motor imagery, 54 or intense neuropsychological rehabilitation. 55 Together with an improvement of performance in the respective task, they revealed an increase of fMRI brain activations in bilateral dorsal frontal/premotor cortex,52,55 an increase of functional connectivity (resting-state fMRI) between right frontal eye field and left intraparietal sulcus, and an activation of the left supplementary motor area (containing the SEF) during motor imagery of the affected left hand, which correlated with visual imagery capacity and time since stroke.
Limitations
There are several limitations of our study that we have to mention before drawing the conclusions. First, the final sample size of our cohort was quite small, partly due to the post-hoc removal of patients’ fMRI data after checking for quality parameters such as head motion and cooperation. Nevertheless, to our knowledge this study with 19 RHS patients with or without neglect is still one of the bigger studies investigating task-related fMRI in this cohort. Second, our findings may not be generalized to RHS patients in the chronic stage because all of our patients were investigated during their acute stage of stroke. This was mainly due to the structural setting of a university medical center but is certainly a caveat, especially since interventional studies, showing beneficial effects of OKS on neglect behavior, were mostly performed in chronic neglect patients at rehabilitation units. Third, despite the controlling for patients’ cooperation via online-oculography in the MR scanner, we cannot fully rule out that weak brain activations in some patients may have been due to reduced motivation and cooperation during the OKS task that required active tracking of the stimulus in order to elicit an optokinetic look-nystagmus.
Conclusions
This is, to our knowledge, the first study that showed bilateral fMRI brain activations in occipital (V1-V4) and frontoparietal (FEF, SEF, IPS) brain regions of RHS patients (with and without hemispatial neglect) as induced by a leftward OKS. Structural integrity of these regions outside the right hemispheric stroke lesion as well as their cross-functional role for oculomotor control and spatial attention processes within the dorsal attention network may provide the neural basis for the previously described therapeutic effects in neglect patients after repetitive leftward OKS. Our study further revealed that acute stroke patients with more severe neglect and higher functional impairment respond less to the optokinetic stimulus, which may indicate that OKS is less appropriate as a rehabilitation intervention for this subgroup and more promising for chronic RHS patients with less severe neglect.
Supplemental Material
Supplemental_Material – Supplemental material for Brain Activations During Optokinetic Stimulation in Acute Right-Hemisphere Stroke Patients and Hemispatial Neglect: An fMRI Study
Supplemental material, Supplemental_Material for Brain Activations During Optokinetic Stimulation in Acute Right-Hemisphere Stroke Patients and Hemispatial Neglect: An fMRI Study by Janina von der Gablentz, Inga Könemund, Andreas Sprenger, Wolfgang Heide, Marcus Heldmann, Christoph Helmchen and Björn Machner in Neurorehabilitation and Neural Repair
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Deutsche Forschungsgemeinschaft (MA5332/1 and MA5332/3-1 to B.M.). The funding source had no involvement in the study design, collection, analysis and interpretation of data.
Ethical Approval
All procedures performed in this study were in accordance with the ethical standards of the institutional research committee (Ethics Committee of the University of Lübeck, no. #12-064) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed Consent
Informed consent was obtained from all individual participants included in the study.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
