Abstract
Background. Although the effect of rehabilitation is influenced by aspects of the training protocol, such as initiation time and intensity of training, it is unclear whether training protocol modifications affect the corticospinal projections. Objective. The present study was designed to investigate how modification of initiation time (time-dependency) and affected forelimb use (use-dependency) influence the effects of rehabilitation on functional recovery and corticospinal projections. Methods. The time-dependency of rehabilitation was investigated in rats forced to use their impaired forelimb immediately, at 1 day, and 4 days after photothrombotic stroke. The use-dependency of rehabilitation was investigated by comparing rats with affected forelimb immobilization (forced nonuse), unaffected forelimb immobilization (forced use), and a combination of forced use and skilled forelimb training beginning at 4 days after stroke. Results. Although forced use beginning 1 day or 4 days after stroke caused significant functional improvement, immediate forced limb use caused no functional improvement. On the other hand, a combination of forced use and skilled forelimb training boosted functional recovery in multiple tasks compared to simple forced use treatment. Histological examination showed that no treatment caused brain damage. However, a retrograde tracer study revealed that immediate forced use and combination training, including forced use and skilled forelimb training, increased corticospinal projections from the contralesional and ipsilesional motor cortex, respectively. Conclusions. These results indicate that although both very early initiation time and enhanced skilled forelimb use increased corticospinal projections, premature initiation time hampers the functional improvement induced by poststroke rehabilitation.
Keywords
Introduction
Rehabilitation is the most commonly used treatment for chronic stroke patients due to many evidences demonstrating its safety and effectiveness. 1 However, functional recovery is often incomplete even when intensive rehabilitation is carried out. Although preclinical studies showed neurorestorative effects of newly developing therapies such as stem cell therapy 2 or anti-NogoA immunotherapy, 3 none of them has been approved in clinical setting. Thus, improving effectiveness of rehabilitation remains an essential strategy to attain better recovery after stroke. 4 Since effect of rehabilitation is affected by modification of rehabilitation protocol as well as patient’s age, comorbidity, and size and location of infarct, guideline to design optimal rehabilitation protocol is required to maximize their efficacy. Nevertheless, it has not been accomplished due to lack of sufficient evidences.
Neural plasticity is heightened during the critical period of the early poststroke recovery phase. And the greatest gains in recovery occur in this period through activity-dependent neural network remodeling. 5 Therefore, there is a general consensus that more intensive rehabilitation initiated in earlier recovery phase would cause better functional outcome, and which was actually supported by many clinical trials.6,7 For example, a clinical trial that compared the functional improvement after constraint-induced movement therapy (CIMT) initiated in early (3-9 months after stroke) and delayed phase (15-21 months after stroke) demonstrated that early CIMT induces greater functional improvement (EXCITE Stroke Trial). 8 However, some important questions remain to be clarified. Although it is generally accepted that early rehabilitation caused better functional outcome than later one, how early rehabilitation should begin is still controversial because previous studies demonstrated very early rehabilitation has a potential to increase damage to the ischemic penumbra. 9 Recent big multicenter randomized controlled trial also reported that the higher dose, very early mobilization protocol was associated with a reduction in the odds of a favorable outcome at 3 months. 10 Similarly, in the regard of training intensity, whereas dose-response relationship has been repeatedly evaluated by comparing total time for therapy,6,7 a single-blind phase II trial of CIMT (Very Early Constraint-Induced Movement during Stroke Rehabilitation [VECTORS] study) reported that higher intensity CIMT resulted in less motor improvement at 90 days compared to lower intensity CIMT and traditional upper extremity therapy, 11 suggesting that too intense training could deteriorate functional recovery. To determine proper rehabilitation regimen, understanding of biological events underlying unfavorable effect induced by too early and intensive training is necessary. However, although previous studies have demonstrated that the integrity and plasticity of corticospinal projections are fundamental for rehabilitation-induced functional recovery after stroke,12-14 it is not known how modifications of the rehabilitation protocol affect these projections.
The purpose of the present study was to examine how modifications of the rehabilitation protocol, and in particular of time of initiation (time dependency) and affected forelimb use (use dependency), impact functional recovery by rehabilitative training after severe cortical stroke and how corticospinal projections is also affected by these modifications.
Materials and Methods
Animals
We used a total of 75 adult male Fisher 344 rats (9 weeks old at the beginning of training; 160-180 g; Charles River Laboratories Japan, Inc). All rats were paired and housed in a temperature-controlled vivarium on a 12-hour:12-hour light:dark cycle. To motivate rats for the pellet reaching task, food intake was moderately restricted throughout the study to maintain body weight at 80% of ad libitum feeding weight. Water was available ad libitum. All experimental procedures were in accordance with National Institutes of Health regulations and approved by the Animal Research Committee of Kawasaki Medical School.
Experimental Design
Rats were randomly divided into 6 groups as indicated in Figure 1A; Control: n = 16, Forced use (Immediate): n = 9, Forced use (day 1): n = 8, Forced use (day 4): n = 9, Forced nonuse (day 4): n = 9, and Forced use + skilled forelimb training (SFT) (day 4): n = 10 (Figure 1A). Forced use and forced nonuse of the affected forelimb were applied by immobilization of unaffected forelimbs and affected forelimb, respectively. After completion of acquisition training for the single-pellet reaching task, rats underwent photothrombotic stroke in the caudal forelimb area (CFA) and rostral forelimb area (RFA) (Figure 1B). Forced use or forced nonuse was carried out for 3 weeks beginning immediately, 1 day, or 4 days after stroke. Behavioral tests were conducted to assess both skilled motor function (single-pellet reaching and staircase tests) and general motor function (rotarod and cylinder tests). Prestroke skilled motor performance was evaluated on the 3 consecutive days before stroke. Poststroke motor performance was evaluated for 3 weeks, 5 days a week from 24 days after stroke. General motor performance was evaluated on the day before stroke and 24 and 42 days after stroke (Figure 1A). Three to 4 days after final behavioral testing, all rats received an injection of Fast Blue into the dorsal column (C2) and Retrobeads into the contralesional side of gray matter (C7-8) (Figure 1F). Rats were number-coded, and behavioral assessments and data analyses were carried out by an examiner who was blinded to the experimental groups.
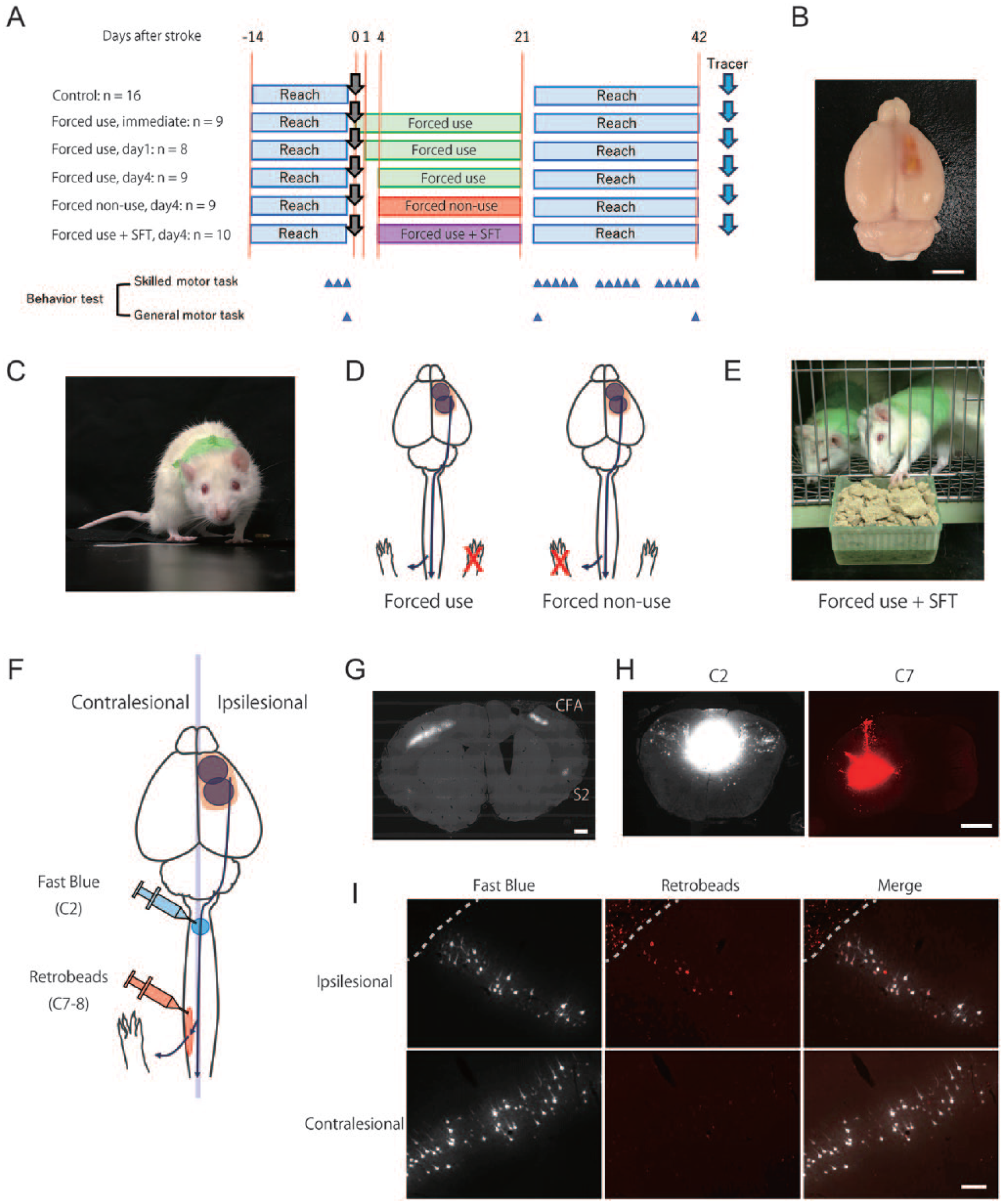
Experimental design. (A) Experimental groups and schedule. (B) Photothrombotic Infarct. Scale bar = 5 mm. (C) Picture of a rat receiving forelimb immobilization. (D) Modification of forelimb immobilization. While forced use immobilized ipsilesional forelimb (red cross) to completely rely on affected forelimb (left panel), forelimb nonuse immobilized contralesional forelimb to completely rely on unaffected forelimb (right panel). (E) Picture of combination of forced use and SFT. (F) retrograde tracer injections. Fast Blue (blue circle) was injected into the dorsal column of upper cervical cord (C2) and Retrobeads (red circle) was injected into contralesional gray matter of lower cervical cord to label total corticospinal neurons and corticospinal neurons projecting to spinal area which control affected forelimb movement, respectively. Infarct is indicated with black circle and peri-infarct tissue is indicated with orange area. (G) Images of brain section labeled with Fast Blue. Fast Blue–positive cells are observed in CFA and secondary sensory area. Scale bar = 1 mm. (H) Images of Fast Blue injection (left panel) and Retrobeads (right panel). Scale bar = 1 mm. (I) Images of corticospinal neurons labeled with Fast Blue and Retrobeads. Scale bar = 200 μm. CFA, caudal forelimb area; S2, secondary sensory area; SFT, skilled forelimb reaching training.
Photothrombosis
Photothrombotic stroke was induced as described previously. 14 Rats were anesthetized with an intraperitoneal injection of ketamine hydrochloride (60 mg/kg; Ketalar; Daiichi-Sankyou, Tokyo, Japan)/xylazine hydrochloride (6.0 mg/kg; Selactar; Bayer Yakuhin, Osaka, Japan). The skull over the RFA and CFA was carefully thinned (center; RFA: 3.0 mm lateral, 2.0 mm rostral from bregma; CFA: 3.5 mm lateral, 2.0 mm caudal from bregma; radius: 2.0 mm) and illuminated for 10 minutes. During the first minutes of illumination, Rose Bengal (15 mg/kg; 184-00272; Wako Pure Chemical, Osaka, Japan) was injected via tail vein catheter. Stroke was induced in the RFA first, then in the CFA by repeating same procedure.
Behavioral Testing and Rehabilitative Training
Single-Pellet Reaching Task
The single-pellet reaching task was carried out as previously described.14,15 Rats received training on 5 sequential days per week. Rats carried out the single-pellet reaching task using a Plexiglas reaching box with a tall narrow window (25 cm high × 1 cm wide) and a 2-cm-wide shelf positioned 2 cm above the floor (order-made; Kagawa Kikai, Tokyo, Japan). Single sucrose pellets (Dustless Precision Pellets 45 mg; F06233; Bioserve, Flemington, NJ, USA) were initially placed within tongue distance, then at gradually greater distances. After shaping the reaching behavior, rats performed 50 training trials per day for 2 weeks. Rats were allowed to reach until they successfully retrieved, missed, or knocked away the food. Because the animals compensate for their affected forelimb by making more reaching attempts, the first reaching success rate was the most sensitive value for motor deficits after stroke. 16 We therefore used the first reaching success rate for the motor performance score in the present study, calculated in the following way: success rate (%) = (number of trials in which the pellet was retrieved on the first reach attempt / number of trials) × 100. Testing sessions were performed as 50 trials with a maximum time of 10 minutes. Prestroke motor performance was calculated by averaging the results of the last 3 days prior to stroke. Poststroke motor performance was evaluated for 3 weeks, 5 days a week from 24 days after stroke. Rats were excluded if prestroke motor performance was lower than 30%.
To analyze the involvement of compensatory movement in single-pellet reaching task recovery, we performed movement element analysis as described previously14,17 with slight modification. A reach was broken down into 6 movement elements: (1) Advance, (2) Pronate, (3) Grasp, (4) Supinate 1, (5) Supinate 2, and (6) Release. Each of these 6 movement elements were rated on a 2-point scale: ‘‘0,” movement was normal; and ‘‘1,” movement was abnormal or absent and total score of the reach was calculated. Reaching movement ratings were obtained from the video recordings taken in the last session on day 42 after stroke. Scoring was done on the first three successful reaches and the average of three scores was defined as abnormal movement score. We exclude 12 rats that could not perform a successful reach. Because most of rats (8/9) in Forced nonuse group could not perform a successful reach, movement element analysis was not carried out in this group.
Staircase Test
The staircase apparatus (made to order; Kagawakikai, Takamatsu, Japan) consisted of a chamber with a central platform for the rat to climb onto and a set of 8 steps on either side. Three sucrose pellets were placed in a well on each step. Rats were placed in the chamber for 10 minutes and the number of pellets consumed and number of steps reached (number of steps in which the number of pellet is less than 3) were recorded. For acquisition of the skill, rats received sequential training 5 days per week for 1 week. The results of the 3 days prior to stroke were averaged and used for prestroke performance.
Rotarod Test
The animals were placed on a rotating wheel accelerating from 4 to 40 rpm within 5 minutes (ENV-574; Brain Science Idea. Co., ltd. Osaka, Japan). The time rats remained on the rotating wheel was measured and averaged from 3 repeated trials. Rats were habituated for 2 days prior to surgery and baseline performance was determined from the day before stroke.
Cylinder Test
The cylinder test was used to assess asymmetries in forelimb use. Rats were placed into a clear plastic cylinder (height, 27 cm; diameter, 17 cm) for 5 minutes, and the number of times each forelimb was placed on the cylinder wall was counted. Performance was scored as the probability of impaired forelimb use (impaired forelimb use/total forelimb use).
Forelimb Immobilization
Forelimb immobilization was performed as described previously 11 with slight modification (Figure 1C and D). Briefly, rats were anesthetized by sevoflurane (Sevofrane: 857150095; Abbvie GK, Tokyo, Japan) and the unaffected forelimb (forced use) or affected forelimb (forced nonuse) and upper torso were wrapped in elastic bandaging tape (Vetrap: 1404; 3M, Maplewood, MN, USA). The upper torso of control group was wrapped in the same way, but they were allowed to use both forelimb. Two or three rats were housed in a cage so that the animals could groom each other. Immobilizing bandages were reattached under light sevoflurane anesthesia when the rats removed it, but the bandages were rarely torn if they were not removed in the first hours after the immobilization procedure. The activity and appetite of the rats were carefully monitored, and the tightness of the bandage was adjusted by making a small cut if a rat showed any sign of restraint stress. Rats in the forced use + SFT group were housed in a wire mesh cage with a food tray (4 cm long, 7 cm wide, 4 cm deep) that was filled with mashed food pellets every 12 hours to train skilled forelimb use (Figure 1E). The overall amount of food pellets was adjusted so that total food consumption (mashed pellets when reaching the tray + usual food pellets) was the same in all groups. While the rats did not actively reach for the mashed pellets in the first couple of days, their motivation increased during the first week. Usually, rats actively reached for about 30 minutes after the food tray was refilled. Although we did not measure individual activity during the skilled forelimb training, we observed that few or no pellets from the tray were consumed during the first couple of days after stroke, but that about half of the rats’ daily consumption stemmed from the tray from the second week onward (Supplementary Figure 1).
Retrograde Labeling of the Spinal Projection Neurons
Retrograde labeling was carried out using 2 types of retrograde tracers, Fast Blue (1.25% suspension in distilled water with 2% dimethyl sulfoxide; 17740-1; Polysciences, Inc. Warrington, PA, USA) and Retrobeads (Lumafluor, Durham, NC, USA). Rats were anesthetized and maintained under a constant rate infusion of ketamine hydrochloride (48 mg/kg/h; intravenously) and laminectomy was performed at the cervical spinal level from C5 to C7. To label total corticospinal neurons, 1 injection of 400 nL Fast Blue was injected at the middle of the cervical spinal cord (spinal level of C2; Figure 1F-I) with the needle tip positioned at a depth of 1.3 mm with a flow rate of 200 nL/min. To label corticospinal neurons projecting to the spinal area innervating affected forelimb, Retrobeads was injected in the contralesional side of cervical spinal cord (spinal level of C7-8; Figure 1F-I). Five injections (200 nL/injection) were made with the needle tip positioned at 0.65 mm lateral to midline at a depth of 1.4 mm, at 1-mm intervals. One week following tracer injection, rats were perfused with 4% phosphate buffered paraformaldehyde. 20 coronal sections (20 μm thick) were cut at 500-μm intervals from the rostral-most tip of the cerebral cortex to 10 mm caudal (approximately 5.3 to −4.7 mm anterior to the bregma in a brain atlas). Images were taken with an all-in-one fluorescence microscope (10× objective: BZ-X700; Keyence, Osaka, Japan) and cells labeled with retrograde tracers were identified with MetaXpress Imaging Analysis V2.0 (Molecular Devices, Sunnyvale, CA, USA). Our preliminary study showed that the RFA and the CFA could be clearly distinguished by their border at 3.0 mm from the rostral-most tip of the cortex (Supplementary Figure 2). We thus defined the RFA as the motor area 0 to 2.5 mm posterior to the rostral-most tip of the cortex, and the CFA as the motor area 3.0 to 10.0 mm posterior to the rostral-most tip of the cortex. Although the location of the RFA and the CFA might be shifted after stroke, the 2 areas were indistinguishable, since most corticospinal neurons were destroyed. Thus, we used the same area definitions for the histological analysis in the present study. Since the CFA and the secondary sensory area (S2) were clearly separated by a blank space (Figure 1G), these 2 areas were easily distinguishable after stroke. The number of cells in each area was counted and multiplied by the section intervals to estimate overall cell numbers.
Assessment of Lesion Volume
A total of 18 coronal sections (20 μm thick) at 500-μm intervals from the rostral-most tip of the cerebral cortex to 9 mm caudal (approximately 5.3 to −3.7 mm anterior to the bregma in a brain atlas) were used for the lesion volume assessment. Since infarct tissue produced by photothrombotic stroke could be clearly distinguished without any additional staining, we analyzed the pictures taken for the tracer study to assess lesion volumes. The remaining tissue of each hemisphere excluding the ventricle and the infarct area were measured with Image J software, and volumes were calculated by multiplying the area by the section interval. Brain damage was calculated using the following formula:
Statistics
All data are reported as mean ± standard error of the mean (SEM). One-way analysis of variance (ANOVA) was performed to compare the histological measures and abnormal movement score assessed by movement element analysis. For post hoc testing, Tukey’s multiple comparison tests were used. We compared behavioral performance in multiple groups using two-way repeated-measures ANOVA and Tukey’s multiple comparison tests. The success rate of the single-pellet reaching test was also analyzed using a linear mixed model analysis and effect size calculation. We used random intercepts to control for repeated measurements in the linear mixed model analysis. Correlations between behavioral and histological values were analyzed using Pearson’s linear-regression method. A statistically significant difference was defined as a P value <.05. All statistical analyses except for effect size were carried out using GraphPad Prism Version 7.0 (GraphPad Software Inc. La Jolla, CA, USA). Linear mixed model analysis and the effect size calculation were performed using SPSS statistics 23 (IBM, Armonk, NY, USA). Complete statistical measures are provided in Supplementary Tables to make the data more accessible for future meta-analysis.
Results
Number of Animals Analyzed and Excluded
In this study, a total of 75 rats were used. Following the behavioral procedures, a total of 13 rats were excluded due to prestroke success rate less than 30% in single-pellet reaching test. One rat died as a result of the surgical procedure. The remaining 61 rats were included in the study (Figure 1A). In the tracer studies, 10 rats were excluded due to unsuccessful injection (eg, tracer leakage in the ipsilesional spinal cord or white matter).
Too Early Initiation Reduces the Benefits of Forced Limb Use
To precisely evaluate the effect of forced limb use on functional recovery, both general and skilled motor functions were examined using multiple motor tests (general motor test: cylinder test and rotarod test; skilled motor test: single-pellet reaching test and staircase test). Motor function was repeatedly evaluated for 3 weeks to assess influence on the relearning of skilled motor task and persistence of the effects. In the first analysis, we assessed time dependency of forced limb use in the functional recovery by comparing forced use beginning immediately, 1 day or 4 days after stroke (Figure 2 and Supplementary Table 1). In general motor test, although there was a tendency for the earlier forced use condition to lead to greater disuse of the contralesional forelimb on day 24 after stroke, no significant difference was detected by either cylinder test (Figure 2A; group effect: P = .272: two-way repeated-measures ANOVA; Control vs Forced use [immediate]: P = .530; Control vs Forced use [day 1]: P = .706: Tukey’s test) or rotarod test (Figure 2B; group effect: P = .131: two-way repeated-measures ANOVA). However, forced use beginning 1 day or 4 days after stroke significantly improved functional recovery in single-pellet reaching test, while immediate forced use did not show significant effect (Figure 2C; interaction: P < .0001; group effect: P < .0001: two-way repeated-measures ANOVA). Post hoc test comparing success rate in each time point revealed that there was no difference between groups on the first testing day after stroke (Figure 2D; day 24; Control vs Forced use [immediate]: P = .977; Control vs Forced use [day 1]: P = .987; Control vs Forced use [day 4]: P < .961; Tukey’s test). But forced use beginning 1 day or 4 days after stroke gradually improved success rate and caused significantly higher success rate in the last testing session (Figure 2D; day 42; Control vs Forced use [immediate]: P = .147; Control vs Forced use [day 1]: P = .004; Control vs Forced use (day 4): P < .0001; Tukey’s test), suggesting forced use improved efficiency of relearning. We also carried out movement element analysis to verify whether increased success rate was caused from compensation. Interestingly, the element analysis revealed that only forced use beginning 4 days after stroke decreased abnormal movement scores on the last testing session (Figure 2E; Control vs Forced use [immediate]: P = .997; Control vs Forced use [day 1]: P = .739; Control vs Forced use [day 4]: P = .024; Tukey’s test). The analysis of the abnormal movement scores for each movement element revealed that forced use beginning 4 days after stroke improved mainly supination movements during reaching behavior (Supplementary Figure 3 and Supplementary Table 1). This result suggests that whereas forced use beginning 4 days after stroke improved success rates by normalizing reaching movements, increased success rates in rats treated with forced use beginning 1 day after stroke might have resulted from compensatory movements. In the staircase test, forced use did not cause any significant change on the number of consumed pellets or the number of steps reached in either ipsilesional or contralesional forelimb (Figure 2F-H; the number of consumed pellets; contralesional: group effect: P = .896; ipsilesional; group effect: P = .495; Figure 2I-K; the number of steps reached; contralesional: group effect: P = .672; ipsilesional; group effect: P = .579: two-way repeated-measures ANOVA).
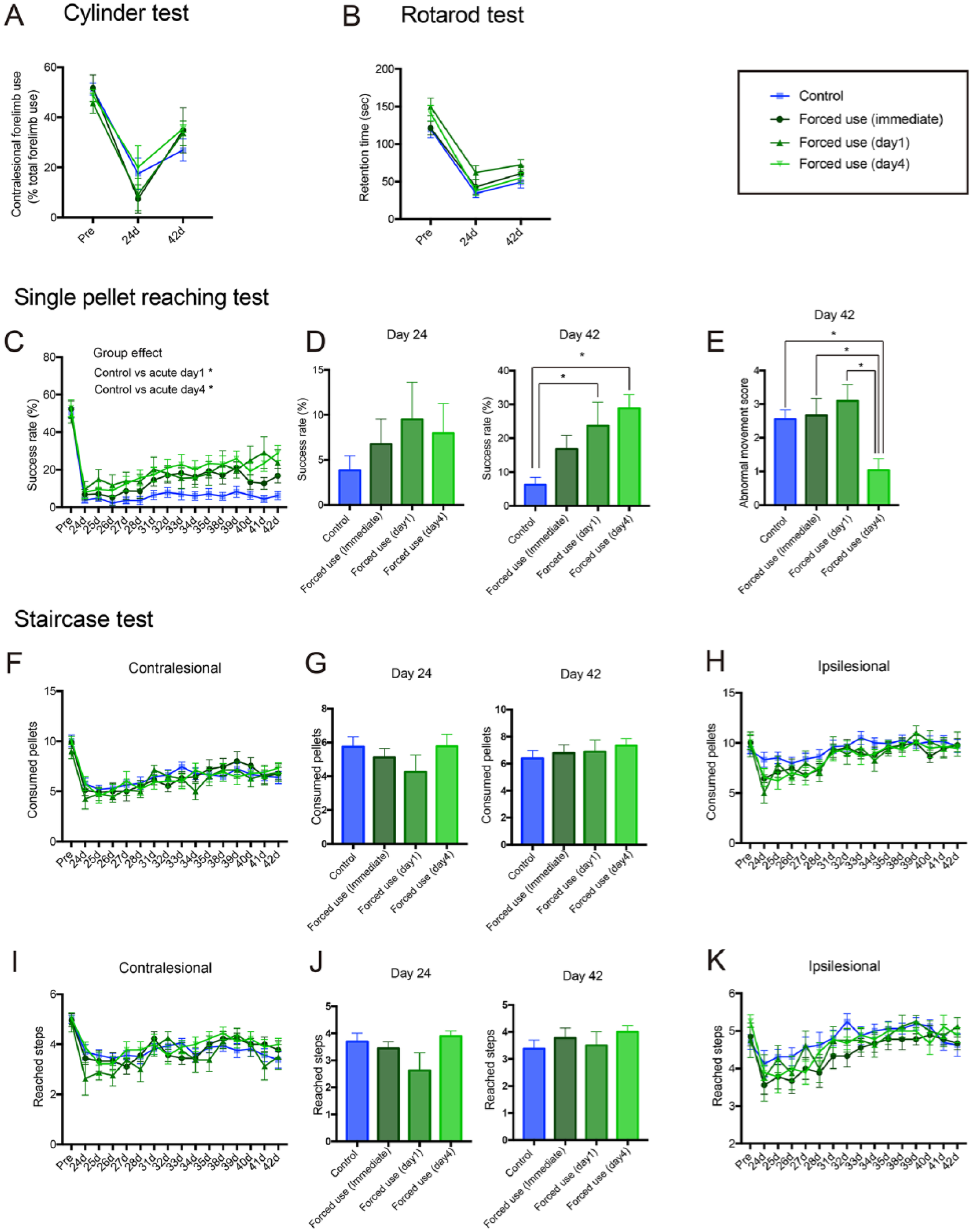
Forced use of affected forelimb improves functional recovery in a time-dependent manner. Forced use does not affect results of the cylinder test (A) and rotarod test (B). Results of single-pellet reaching test (C-E) and staircase test (F-K). In addition to the time course results (F, H, I, K), the results of day 24 and day 42 are presented separately (D, G, I) to clearly show the effect of forced limb use before (day 24) and after (day 42) continuous skilled forelimb testing. (C) Time course change of success rate. (D) Success rate on day 24 and day 42 after stroke. (E) Results of movement element analysis. Forced use beginning 1 day or 4 days after stroke) gradually improved success rate during recovery period (C), which resulted in significantly higher success rate on day 42 after stroke compared to control group (D). Abnormal movement was normalized by forced use (day 4) but not by forced use (Immediate) and forced use (day 1) (E). (F, H, I, K) Time course change of motor performance. (G, J) Motor performance on day 24 and day 42 after stroke. Forced use did not affect the number of consumed pellets (F-H) or the number of reached steps (I-K) in either contralesional (F, G, I, J) or ipsilesional side (H and K). Tukey’s multiple comparison: *P < .05.
Enhanced Skilled Forelimb Use Boosts Forced Use–Induced Functional Recovery
In the second analysis, we investigated whether intensity of affected forelimb use alters the functional recovery after stroke by comparing rats forced to completely rely on their affected (forced use) or their unaffected (forced nonuse) forelimb (Figure 1D). We further set another group in which rats carried out skilled forelimb training in their home cage during their forced use training (Figure 1E: forced use + SFT) to investigate the effect of enhanced skilled forelimb use (Figure 3 and Supplementary Table 2). Whereas forced nonuse caused lower reliance on the affected forelimb in cylinder test on day 24 after stroke (Figure 3A), we did not find significant change in general motor test (Figure 3A and B; Cylinder test: group effect: P = .441; Rotarod: group effect: P = .357: 2-way repeated-measures ANOVA). On the other hand, forced use and forced use + SFT caused significantly higher success rate in single-pellet reaching test compared with nontreated control (Figure 3C; Interaction: P < .0001; group effect: P < .0001: 2-way repeated-measures ANOVA). While forced use and forced use + SFT showed similar success rate (Figure 3D; day 42; Control vs Forced use: P < .0001; Control vs Forced use + SFT: P < .0001; Control vs Forced nonuse: P = .8242; Tukey’s test) and abnormal movement score (Figure 3E; Control vs Forced use: P < .0053; Control vs Forced use + SFT: P = .0376; Tukey’s test) in the last session, only forced use + SFT caused higher success rate in the first session after forced use treatment (Figure 3D; day 24; Control vs Forced use: P = .959; Control vs Forced use + SFT: P = .022; Control vs Forced nonuse: P = .975; Tukey’s test). Moreover, forced use + SFT increased number of consumed pellets in staircase test (Figure 3F; Interaction: P = .4276; group effect: P = .0002: two-way repeated-measures ANOVA). Significant increase of consumed pellets was observed from the first testing session to the last testing session (Figure 3G; day 42; Control vs Forced use: P = .675; Control vs Forced use + SFT: P = .0024; Control vs Forced nonuse: P = .941; Tukey’s test). Forced use + SFT also increased the number of steps reached in the last testing session (Figure 3J; day 42; Control vs Forced use: P = .414; Control vs Forced use + SFT: P = .022; Control vs Forced nonuse: P = .915; Tukey’s test), which indicates extension of reaching distance. In contrast to forced use, forced nonuse did not improve functional recovery but slightly worsen the motor performance in staircase test in both ipsilesional and contralesional forelimb (Figure 3F-K).
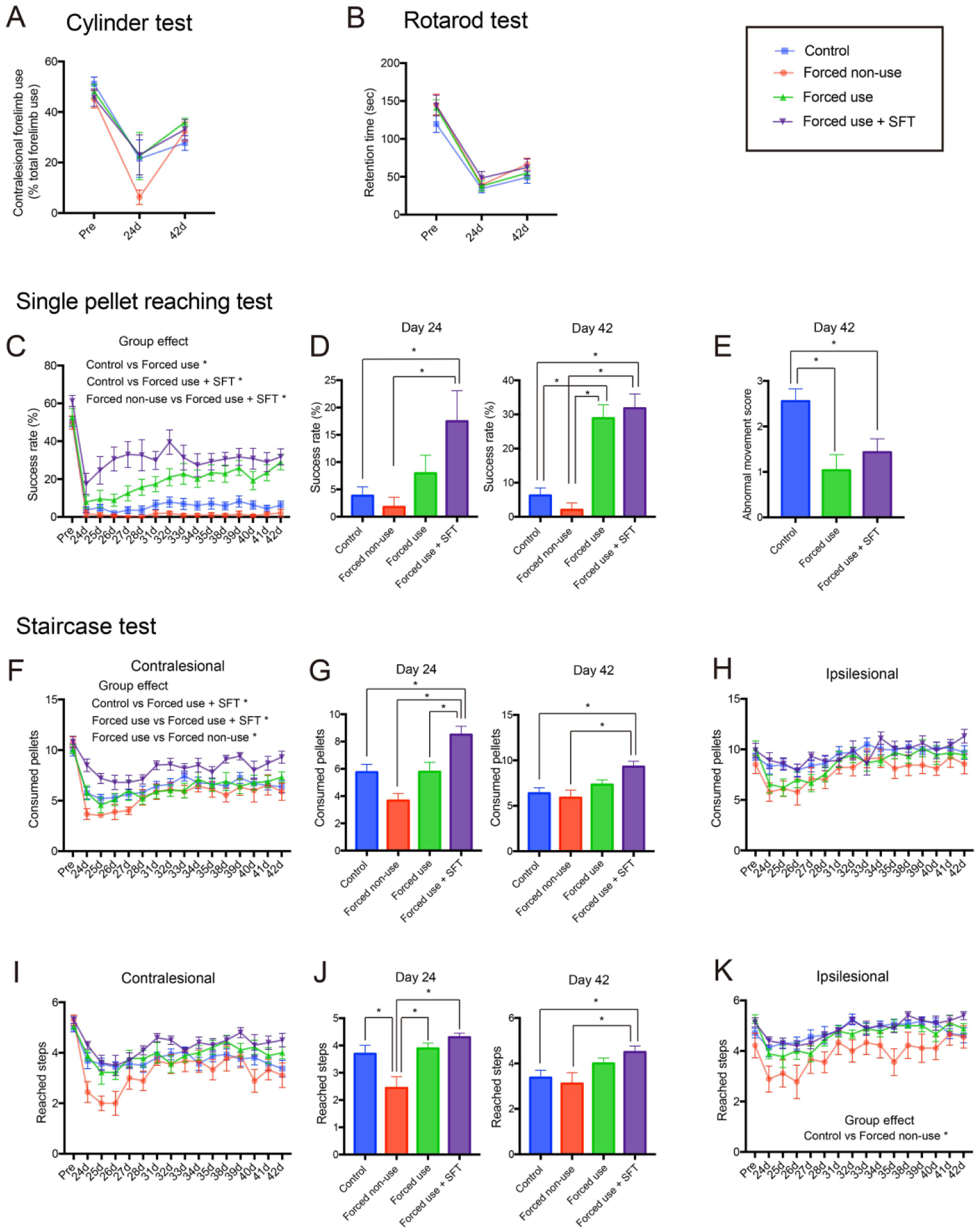
Forced use of affected forelimb improves functional recovery in a use-dependent manner. (A and B) Results of general motor test. Forced use did not affect results of the cylinder test (A) and rotarod test (B). Results of the single-pellet reaching test (C-E) and the staircase test (F-K). In addition to the time course results (F, H, I, K), the results of day 24 and day 42 are presented separately (D, G, I) to clearly show the effect of forced limb use before (day 24) and after (day 42) continuous skilled forelimb testing. (C) Time course change of success rate. (D) Success rate on day 24 and day 42 after stroke. (E) Results of movement element analysis. Forced use and forced use + SFT improved both success rate (C and D) and normalized skilled reaching movement (E) compared to control group. While forced use improved functional gain during recovery period, forced use + SFT improved success rate from the first test after completion of forced use treatment. (F-K) Results of staircase test. (F, H, I, K) Time course change of motor performance. (G, J) Motor performance on day 24 and day 42 after stroke. Although simple forced use did not improve motor performance in staircase test, forced use + SFT significantly increased the number of consumed pellets (F and G) and the number of reached steps (I and J) in contralesional forelimb compared with control group. On the other hand, forced nonuse showed slightly worse motor performance in both contralesional and ipsilesional forelimb compared with control. Tukey’s multiple comparison: *P < .05. SFT, skilled forelimb reaching training.
Linear Mixed Model Analysis for the Effect of Different Rehabilitation Protocols on Functional Recovery After Stroke
Although the repeated-measures ANOVA is often used to analyze data collected in a repeated measures study design, multilevel modeling techniques are more informative than ANOVA because they characterize both group-level (nomothetic) and individual-level (idiographic) effects and yield a more complete understanding of the phenomena under study. 18 Thus, we also analyzed the success rate in the single-pellet reaching test after rehabilitation (days 24-42) using linear mixed model analysis to better understand the effect of different rehabilitation protocols (Supplementary Table 3). In the linear mixed model analysis, we found that only a combination of forced use and skilled forelimb training significantly increased the baseline success rate, which was determined by the success rate in the first test, (Control vs Forced use + SFT: P < .001); these results indicate that the combination training improved the success rate without repeated testing sessions. On the contrary, forced limb use without combination training increased the linear slope whenever forced use was initiated (Forced use [immediate]: P = .003; Forced use [day 1]: P < .001; Forced use [day 4]: P < .001; vs Control). These data indicate that forced limb use increased the recovery rate during the repeated testing period, regardless of the initiation time. Notably, the linear slope was smaller if forced use training was initiated at an earlier time period after stroke (Forced use [immediate]: 0.449; Forced use (day 1): 0.624; Forced use [day 4]: 0.797). Consequently, the rats that had forced use initiated immediately after stroke did not make a significant recovery in the last testing session (P = .105: post hoc tests with Bonferroni-adjusted P values). Consistent with these data, the effect size in the final outcome was smaller in the rats in which forced limb use was initiated at an earlier time period (effect size: day 42; Forced use [immediate]: 0.220; Forced use [day 1]: 0.294; Forced use [day 4]: 0.561; Forced use + SFT: 0.593; Forced nonuse: 0.067).
Forced Use Increases Corticospinal Projections in a Time- and Use-Dependent Manner
To elucidate the biological basis of time- and use-dependent effect of rehabilitation, we carried out histological examination (Figures 4 and 5 and Supplementary Table 4). First, we validated brain tissue damage induced by photothrombotic stroke. Despite worse functional recovery in early forced use and enhanced functional recovery induced by additional skilled forelimb training, no significant change was detected in the remaining tissue volume in ipsilesional and contralesional hemisphere or brain damage (Figure 4A and B; brain damage; P = .157: one-way ANOVA), suggesting that the effect of training modification in functional recovery is not associated with tissue damage or neuroprotection. Then, we evaluated changes in the corticospinal projections. In the current study, we injected Fast Blue in the dorsal column at C2 and Retrobeads in the contralesional gray matter at C7-8, so that we can assess the number of total corticospinal neurons and corticospinal neurons projecting to the contralesional lower cervical cord which innervate affected forelimb (Figure 1F). In this tracer study, we did not find significant changes in the number of Fast Blue–positive cells in the motor cortex (RFA + CFA: Figure 5A; ipsilesional; P = .055; contralesional; P = .819: One-way ANOVA), RFA (Figure 5C; ipsilesional; P = .164; contralesional; P = .801: one-way ANOVA), CFA (Figure 5E; ipsilesional; P = .134 contralesional; P = .796: one-way ANOVA) or S2 (Figure 5G; ipsilesional; P = .069; contralesional; P = .480: one-way ANOVA). These data indicate that forced use did not affect survival of corticospinal neurons after stroke, consistent with the brain damage data. In contrast to Fast Blue–positive cells, the number of Retrobeads-positive cells was increased in the ipsilesional motor cortex (Figure 5B; Control vs Forced use + SFT: P = .009: Tukey’s test) and CFA (Figure 5F; Control vs Forced use + SFT: P = .019: Tukey’s test) in the rats treated with combination of forced use and skilled forelimb compared with control rats. Surprisingly, the number of Retrobeads-positive cells was also increased in the contralesional CFA in the rat treated with immediate forced limb use (Figure 5F; P = .048: Tukey’s test). The correlation analysis revealed that success rates in the single-pellet reaching test on day 42 after stroke were significantly correlated with Retrobeads-positive cells in the ipsilesional motor cortex (Supplementary Figure 4A; R2 = .143, P = .0062: Pearson’s linear regression), but not in the contralesional motor cortex (Supplementary Figure 4B; R2 = .0318, P = .2105: Pearson’s linear regression).
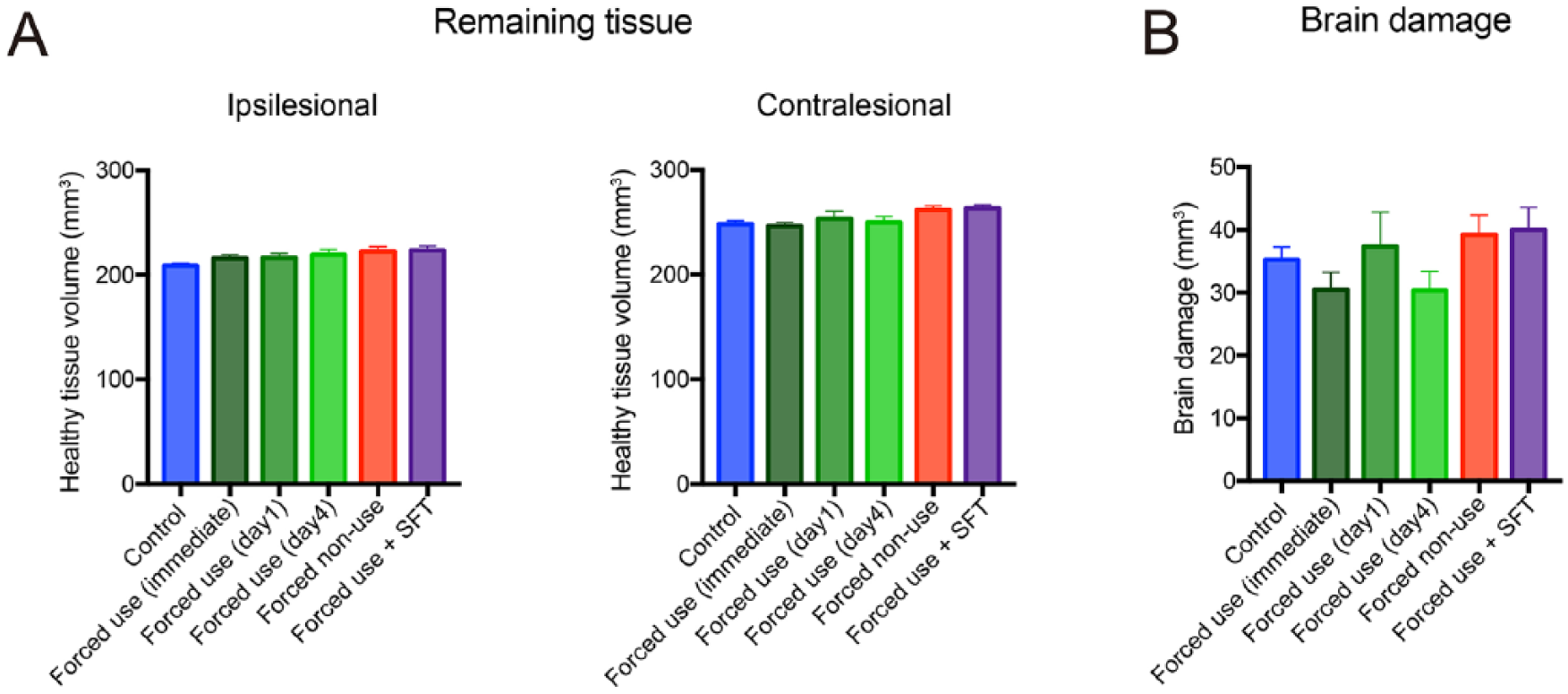
Forelimb immobilization does not affect tissue damage induced by photothrombotic stroke. (A) Remaining tissue volume in the ipsilesional and contralesional hemispheres. (B) Brain damage in each group. Neither forced use nor forced nonuse affected remaining tissue volume or brain damage. SFT, skilled forelimb reaching training.
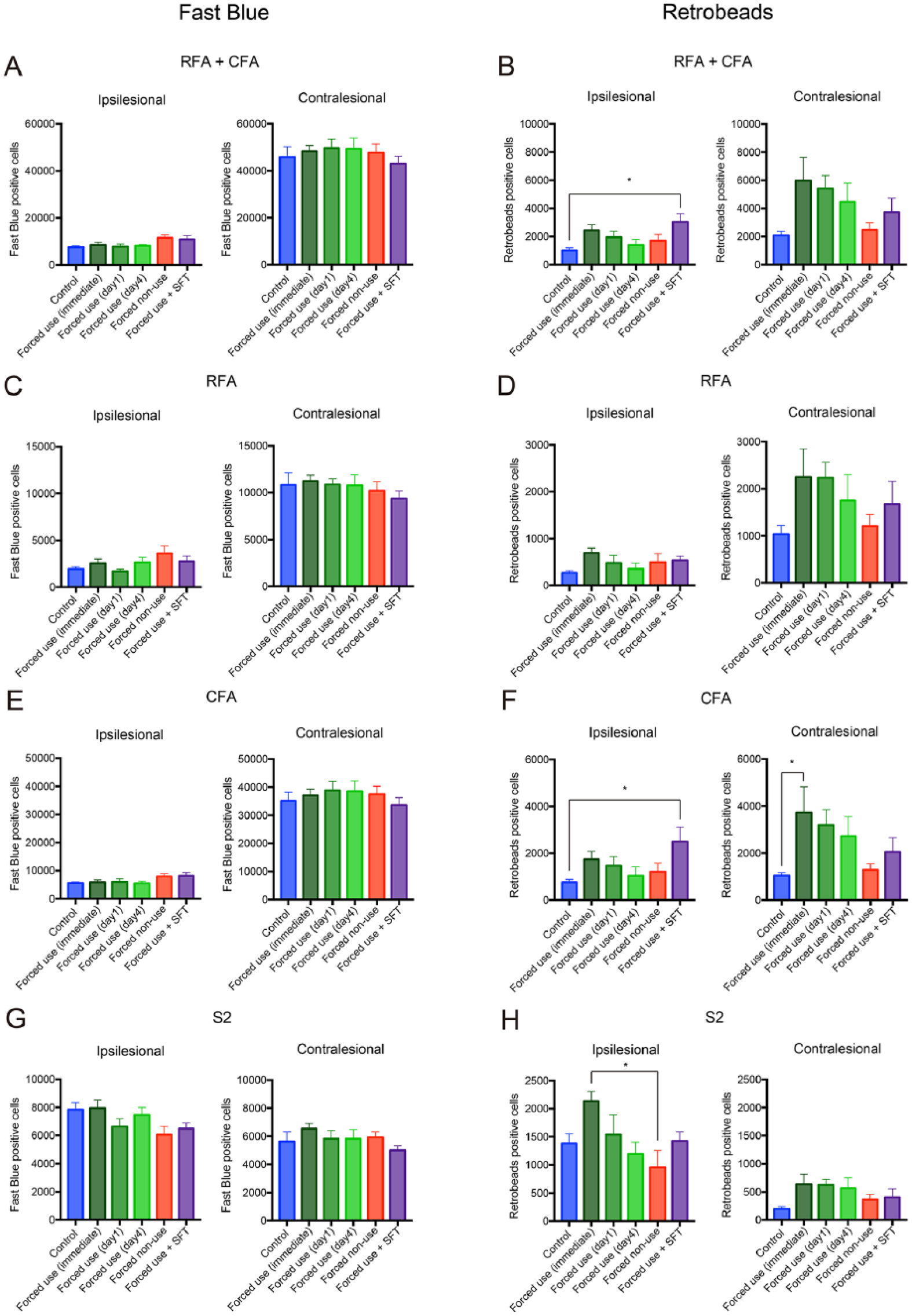
Forced use of affected forelimb increases corticospinal projections in a time- and use-dependent manner. (A, C, E, G) The number of Fast Blue–positive cells in the motor cortex (RFA + CFA: A), RFA (C), CFA (E) and S2 (G). (B, D, F, H) The number of Retrobeads-positive cells in the motor cortex (RFA + CFA: B), RFA (D), CFA (F) and S2 (H). Whereas no difference was observed in the number of Fast Blue–positive cells, forced use (immediate) and forced use + SFT increased the number of Retrobeads-positive cells in the ipsilesional and contralesional CFA compared with control group (F), respectively. The scales are different for each brain area, while the same scales are used for the identical areas in the ipsilesional and contralesional hemispheres. Tukey’s multiple comparison: *P < .05. CFA, caudal forelimb area; RFA, rostral forelimb area; S2, secondary sensory area; SFT, skilled forelimb reaching training.
Discussion
Although many studies demonstrated that delayed initiation of training reduces the rehabilitation-induced functional recovery, recent big clinical trial showed that very acute mobilization caused unfavorable outcome after stroke.
10
In the current study, we found that whereas forced limb use beginning 1 day or 4 days after stroke improved success rates in the single-pellet reaching test, immediate forced use did not lead to significant functional improvement. Furthermore, in contrast to forced use beginning 4 days after stroke, which decreased abnormal compensatory movements, forced use beginning 1 day after stroke did not normalize such abnormal movements. The abnormal movement score for each movement element revealed that forced use beginning 4 days after stroke decreased mainly supination movements during reaching behavior. Similar improvements were observed in spontaneous recovery
17
or recovery induced by skilled forelimb training.
14
In addition to the 2-way repeated-measures ANOVA, we also performed a linear mixed model analysis to better understand the behavioral changes induced by different rehabilitation protocols. It revealed that although forced limb use can improve the functional recovery during repeated testing session initiated at any time after stroke, rats that had forced use initiated at an earlier time period after stroke had smaller increases in recovery rate. The Stroke Recovery and Rehabilitation Roundtable recently defined the hyperacute phase in which cell death expansion is prominent as the time period between 0 and 24 hours after stroke, and the acute phase in which strong inflammatory responses occur as the time period between 1 and 7 days after stroke.
19
According to this definition, our results indicate that functional improvements induced by forced limb use initiated in the hyperacute phase is inferior to that in the acute phase. Previous studies reported negative effects of rehabilitation beginning at a very early phase after stroke. Risedal et al
20
reported that rehabilitation with an enriched environment starting 7 days after middle cerebral artery occlusion (MCAO) resulted in better functional recovery compared with rehabilitation starting 24 hours after stroke. Several studies have also demonstrated adverse effects of the very early initiation of forced limb use. For example, Kozlowski et al
21
demonstrated that forced use of the contralesional forelimb after electrolytic lesion in the sensorimotor cortex increased the lesion volume and exaggerated sensorimotor functions as assessed by several asymmetry tests. Similarly, Bland et al
22
reported that immediate forced use after MCAO increased infarct volume and led to a deterioration of functional outcomes as assessed by forelimb placing tests and the grid walk test. In contrast to these studies, the current study shows that immediate forced use affects neither brain damage nor the number of total corticospinal neurons. Some studies also reported that immediate forced limb use after photothrombotic stroke affects sensorimotor function without significant effects on lesion volume.23,24 Since a previous study suggested NMDA (N-methyl-
Although the present study demonstrated that very early initiation hampers the rehabilitation-induced functional recovery, it is not clear when rehabilitation should begin in order to attain maximum recovery. Biernaski et al 30 reported that although enriched rehabilitation initiated 5 days after MCAO had a significant effect on both functional recovery and dendritic growth, it was less effective when started 14 or 30 days after MCAO. Similarly, Ishida et al 31 also demonstrated that forced limb use beginning 1 day after subcortical hemorrhage improved functional recovery and dendritic growth, but was ineffective if started 17 days after stroke. Consistent with previous behavior and morphological studies, Carmichael et al 32 reported that the expression of growth promoting genes increased 3 to 7 days after stroke, while the expression of growth-inhibiting genes increased approximately 14 days after stroke; these results indicate that neuronal plasticity is heightened from 3 to 14 days after stroke. These data suggest that rehabilitation beginning at approximately 4 days after stroke seems to be the optimal time to obtain maximum advantage of the enhancement in neural plasticity in order to minimize adverse effects.
In contrast to immediate forced use, a combination therapy of forced use and skilled forelimb reaching training promoted functional recovery and increased corticospinal projections, while the rats prohibited to use their affected forelimb showed worse motor function than control rats. Furthermore, motor performance on the single-pellet reaching test on day 42 after stroke was significantly correlated with Retrobeads-positive cells in the ipsilesional motor cortex. In the present study, motor performance was continuously investigated using the single-pellet reaching test as well as the staircase test. These motor tests also served as motor training, to verify the effect of treatment on the relearning of motor skills. Since the efficacy of motor training after stroke depends on timing, forced use + SFT might induce better functional recovery of motor performance, because rats in this group started their skilled forelimb training earlier than those in other groups. However, our previous study 12 demonstrated that intensive training on the single-pellet reaching task (250 trials/day) beginning 4 days after stroke failed to improve functional recovery as tested on a trained motor task (task-specific recovery). These results indicated that skilled forelimb training alone cannot induce functional recovery after severe stroke, which destroys most corticospinal neurons, even if intensive treatment begins early after stroke. Therefore, it is not very likely that the combination of forced use and skilled forelimb training induced better functional recovery because of the earlier initiation of the skilled forelimb training. These results thus indicated that poststroke functional recovery is dependent on the use of the affected forelimb, and that additional skilled forelimb training can boost functional recovery induced by forced use. Several studies reported beneficial effect of combination training. Jeffers and Corbett 33 demonstrated that combination of enriched environment and skilled forelimb training improves functional recovery in single-pellet reaching task whereas single therapy with either enriched environment or skilled forelimb reaching training does not cause significant recovery. DeBow et al 34 reported better functional recovery and reduced lesion volume after striatal hemorrhagic stroke using a combination therapy involving forced limb use and a skilled forelimb reaching training. These results indicate that additional rehabilitative training could heightened poststroke neural plasticity and improves functional recovery. Since the efficacy of rehabilitation is strongly affected by the intensity of training,35,36 it is likely that increased affected forelimb use induced by additional training resulted in better functional recovery. In the current study, the rats with combined forced use and skilled forelimb training consumed more than one-third of the total food by reaching with the affected forelimb in the second and third week after stroke (Supplementary Figure 1), compared with other rats who did not undergo any skilled forelimb training during the three weeks after the stroke. Axonal sprouting of the corticospinal tract depends on the activity of corticospinal neurons. 37 Thus, additional skilled forelimb training might also promote axonal sprouting of the corticospinal tract by increasing the neural activity of the surviving corticospinal neuron. In fact, Wiersma et al 38 reported that chondroitinase ABC therapy induced greater axonal remodeling of corticospinal neurons in the peri-infarct area when combined with high intensity rehabilitation than when combined with low intensity rehabilitation. Although, a clinical trial of CIMT suggested harmful effects caused by too intense training, 11 additional skilled forelimb training did not lead to any adverse effects, such as increased brain damage, in the current study. Since previous study reported that poststroke stress increases infarct volume and exaggerate functional recovery, 39 training-induced stress might be a factor which determines whether rehabilitative training is beneficial or not. Ke et al 40 reported voluntary exercise with running wheel increased hippocampal BDNF concentration but forced exercise on motor-driven treadmill failed to increase BDNF concentration associated with higher serum corticosterone level. Sun et al 41 also reported gradually increased training intensity causes higher functional recovery than stably low and high training intensity associated with higher BDNF production and less plasma corticosterone concentration. In the current study, additional skilled forelimb training was carried out voluntarily in the rats’ home cage, and training intensity was decreased during the first week after stroke when neuronal vulnerability was high. Wahl et al 42 reported that activity levels in an enriched environment that were reduced 1 to 2 weeks after stroke compared with 3 to 4 weeks correlated with systemic inflammation. The authors suggested that stroke-induced fatigue is a biologically purposeful reaction enabling the formation of new circuits. Stroke-induced fatigue may also contribute to the prevention of negative effects caused by too intensive training during the very early period after stroke. Because the ideal time point to start a respective therapy must be determined in order to potentiate beneficial effects and avoid neutral or even negative ones, 43 future studies exploring various time courses for different combination therapies would be beneficial for the development of optimal rehabilitative protocols.
Conclusion
The current study demonstrated that enhanced skilled forelimb use restores corticospinal projections and boosts rehabilitation-induced functional recovery. On the other hand, too early initiation of training reduces benefits of rehabilitation despite of increased corticospinal projections. These data highlight that training program, especially initiation time must be carefully designed to maximize the efficiency of rehabilitation because heightened neural plasticity in very acute phase after stroke could cause also harmful effects such as vulnerability to the excite toxicity and aberrant neural plasticity.
Supplemental Material
Supplementary_figure_1 – Supplemental material for Very Early Initiation Reduces Benefits of Poststroke Rehabilitation Despite Increased Corticospinal Projections
Supplemental material, Supplementary_figure_1 for Very Early Initiation Reduces Benefits of Poststroke Rehabilitation Despite Increased Corticospinal Projections by Naohiko Okabe, Naoyuki Himi, Emi Nakamura-Maruyama, Norito Hayashi, Issei Sakamoto, Toru Hasegawa and Osamu Miyamoto in Neurorehabilitation and Neural Repair
Supplemental Material
Supplementary_figure_2 – Supplemental material for Very Early Initiation Reduces Benefits of Poststroke Rehabilitation Despite Increased Corticospinal Projections
Supplemental material, Supplementary_figure_2 for Very Early Initiation Reduces Benefits of Poststroke Rehabilitation Despite Increased Corticospinal Projections by Naohiko Okabe, Naoyuki Himi, Emi Nakamura-Maruyama, Norito Hayashi, Issei Sakamoto, Toru Hasegawa and Osamu Miyamoto in Neurorehabilitation and Neural Repair
Supplemental Material
Supplementary_figure_3 – Supplemental material for Very Early Initiation Reduces Benefits of Poststroke Rehabilitation Despite Increased Corticospinal Projections
Supplemental material, Supplementary_figure_3 for Very Early Initiation Reduces Benefits of Poststroke Rehabilitation Despite Increased Corticospinal Projections by Naohiko Okabe, Naoyuki Himi, Emi Nakamura-Maruyama, Norito Hayashi, Issei Sakamoto, Toru Hasegawa and Osamu Miyamoto in Neurorehabilitation and Neural Repair
Supplemental Material
Supplementary_figure_4 – Supplemental material for Very Early Initiation Reduces Benefits of Poststroke Rehabilitation Despite Increased Corticospinal Projections
Supplemental material, Supplementary_figure_4 for Very Early Initiation Reduces Benefits of Poststroke Rehabilitation Despite Increased Corticospinal Projections by Naohiko Okabe, Naoyuki Himi, Emi Nakamura-Maruyama, Norito Hayashi, Issei Sakamoto, Toru Hasegawa and Osamu Miyamoto in Neurorehabilitation and Neural Repair
Supplemental Material
Supplementary_table – Supplemental material for Very Early Initiation Reduces Benefits of Poststroke Rehabilitation Despite Increased Corticospinal Projections
Supplemental material, Supplementary_table for Very Early Initiation Reduces Benefits of Poststroke Rehabilitation Despite Increased Corticospinal Projections by Naohiko Okabe, Naoyuki Himi, Emi Nakamura-Maruyama, Norito Hayashi, Issei Sakamoto, Toru Hasegawa and Osamu Miyamoto in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We appreciate the advice of the central research center section of Kawasaki Medical School for their help with animal testing. We also acknowledge and thank Dr Andy Lin (Institute for Digital Research and Education at UCLA) for his instructive advice on statistical analysis.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

