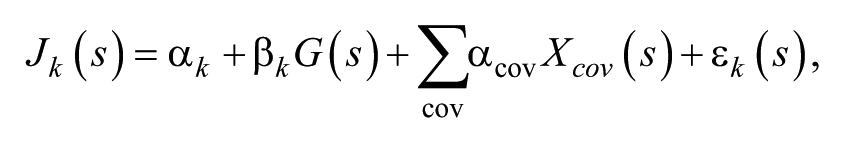Abstract
Background. Subcortical infarcts can result in verbal memory impairment, but the potential underlying mechanisms remain unknown. Objective. We investigated the spatiotemporal deterioration patterns of brain structures in patients with subcortical infarction and identified the regions that contributed to verbal memory impairment. Methods. Cognitive assessment and structural magnetic resonance imaging were performed 1, 4, and 12 weeks after stroke onset in 28 left-hemisphere and 22 right-hemisphere stroke patients with subcortical infarction. Whole-brain volumetric analysis combined with a further-refined shape analysis was conducted to analyze longitudinal morphometric changes in brain structures and their relationship to verbal memory performance. Results. Between weeks 1 and 12, significant volume decreases in the ipsilesional basal ganglia, inferior white matter, and thalamus were found in the left-hemisphere stroke group. Among those 3 structures, only the change rate of the thalamus volume was significantly correlated with that in immediate recall. For the right-hemisphere stroke group, only the ipsilesional basal ganglia survived the week 1 to week 12 group comparison, but its change rate was not significantly correlated with the verbal memory change rate. Shape analysis of the thalamus revealed atrophies of the ipsilesional thalamic subregions connected to the prefrontal, temporal, and premotor cortices in the left-hemisphere stroke group and positive correlations between the rates of those atrophies and the change rate in immediate recall. Conclusions. Secondary damage to the thalamus, especially to the left subregions connected to specific cortices, may be associated with early verbal memory impairment following an acute subcortical infarct.
Keywords
Introduction
One systematic review has suggested that 31% to 55% of patients with stroke experience memory deficits within 3 months of the first infarct. 1 Moreover, working (verbal and visuospatial) and episodic memory were more regularly diminished in stroke victims. 2 Verbal and visuospatial memory deficits have been reported to occur in stroke patients with a cortical lesion. 3 This finding is unsurprising because several cortical areas play essential roles in memory function. Verbal and visuospatial memory require encoding and consolidating verbal and visuospatial information for subsequent retrieval and depend primarily on regions within the left and right hemispheres of the brain, respectively. 4 The medial temporal lobe system has been identified as being important for encoding, consolidation, and retrieval functions. 4 Frontal regions within the left hemisphere of the brain might also be involved in verbal memory encoding. 5 It has been reported that patients with subcortical lesions located in the basal ganglia, internal capsule, or corona radiata in the left hemisphere experienced poor verbal memory performance.6-8 Moreover, poor verbal memory, but not visuospatial memory, could be predicted by lesion characteristics, for example, lesions located in the left hemisphere and large lesions. 6 Interestingly, why do these patients with nonstrategically located infarct still experience poor verbal memory performance? The verbal memory impairment resulting from secondary hippocampal atrophy has been reported in the chronic phase of middle cerebral artery (MCA) infarcts with cerebral cortex involvement.9,10 However, the potential underlying mechanism of early verbal memory impairment in patients with an acute subcortical infarct in the area of the MCA remains largely unknown.
Typically, structural brain studies of cognition have focused on cortical regions with less attention paid to any modulatory role of the thalamus. Philp et al 11 found that verbal memory performance in normal individuals correlated with the volume of the left-thalamic region defined by anatomical connectivity with the temporal lobe, with larger thalamic volumes corresponding to superior word recall. In addition, damage to the thalamus, especially to the left anterior part, has been reported to cause verbal memory dysfunction in patients with thalamic infarction by interrupting the thalamo-cortical circuit.12,13 In patients with unilateral MCA infarcts, secondary degeneration in the ipsilesional thalamus, characterized by hyperintensity on conventional T2-weighted images, was found a few weeks after stroke.14,15 However, it is unclear whether secondary damage to the left thalamus, which is connected to cortical regions that play key roles in verbal memory performance, contributes to early verbal memory impairment. Recently, deformation-based statistical shape analysis has been proposed to quantitatively assess region-specific shape changes (atrophy or expansion) of subcortical structures restricted to different subregions. This can characterize a spatial profile of the disease-spreading pattern within that structure as well as the temporal evolution dynamics.16,17 Deformation-based shape analysis has been successfully applied to the study of subcortical and ventricular structures in various neurodegenerative diseases.16,17 Thus, in the present study, we first conducted whole-brain segmentation-based volumetric analyses, in terms of both group comparisons and correlation analyses to verbal memory, so as to determine whether the thalamus is the key structure of interest. Next, we performed statistical shape analyses to identify region-specific shape abnormalities in the ipsilesional thalamus, in terms of both group differences and memory associations.
Methods
Participants
This prospective study was approved by the institutional review board of The First Affiliated Hospital of Sun Yat–Sen University. Each participant provided written informed consent. A total of 59 patients were included in the study from October 2011 to August 2016. Inclusion criteria were as follows: (1) admission within 7 days of the onset of symptoms; (2) a first onset and single subcortical infarct within the territory of the MCA identified by magnetic resonance imaging (MRI); and (3) no intracranial or extracranial artery occlusion on ultrasound and magnetic resonance angiography. Exclusion criteria were as follows: (1) prohibited medical implants that were contraindicated for cerebral MRI; (2) comorbid neurological disease (eg, epilepsy, Parkinson’s disease); (3) Mini-Mental State Examination (MMSE) score on admission ≤23 for >6 years of education, ≤20 for ≤6 years of education, or ≤17 for 0 years of education 18 ; (4) evidence of possible depression (Center for Epidemiologic Studies Depression Scale score ≥16), 6 psychiatric disease, traumatic brain injury; or (5) any other neurological deficits (eg, aphasia). We also recruited 30 healthy controls (16 women and 14 men; median age: 55 years).
Experimental Design
The protocol involved a 12-week longitudinal investigation during which patients with stroke underwent acquisition of MRI data 3 times after stroke onset: within 1 week after symptom onset (W1, <7 days), at the end of week 4 (W4, 28 ± 4 days), and week 12 (W12, 84 ± 4 days). The control participants underwent only 1 session of MRI acquisition as well as neurological and cognitive testing.
Behavioral Tests
Cognitive function was evaluated prior to the MRI examination at each time point by a trained neurologist (SX) using the MMSE and the Chinese version of the Rey Auditory Verbal Learning Test (RAVLT). 9 This test comprises a list of 12 nouns presented to the participant on 3 trials. Recall was tested immediately after each trial, and the total number of correctly remembered words in all 3 trials was taken as the score for immediate recall. After a delay of 30 minutes, the patient was asked to recall again as many words as possible (long-delay recall). The RAVLT is widely used among neuropsychologists and has good reliability and validity. 6 In addition, the test allows a person to encode, consolidate, store, and retrieve verbal information. 19 The neurocognitive batteries used in this study were completed in clinically feasible examination times and did not exhaust patients, which would lead to lower scores that may not reflect their typical abilities. The Reliable Change Index (RCI) was used to determine whether a change observed in a participant is clinically significant and can define improvement or deterioration on cognitive tests in stroke patients.20,21 RCI is calculated by dividing the amount of change in RAVLT scores between 2 measurement time points by the standard error of the difference between the 2 test scores. A score outside the range of −1.96 to 1.96 reflects a significant change.20,21
MRI Data Acquisition
The 3D T1-weighted imaging (WI) data were acquired using a 3.0-T scanner (Tim Trio, Siemens, Erlangen, Germany) with the following scan parameters: repetition time/echo time/inversion time/number of excitations/flip angle = 2530 ms/3.45 ms/1100 ms/1/7°, 192 slices, 256 × 256 matrix dimensions, and 1 × 1 × 1 mm3 voxel size.
Support Vector Regression-Based Lesion Symptom Mapping (SVR-LSM) Analysis
Lesion masks were created by manually tracing stroke damage on the T1-weighted images in native space using MRIcron (version 6.6.2013). 22 All lesion masks were verified by 2 board-certified neurologists (GL and JZ) and then warped to the Montreal Neurological Institute space using the Statistical Parametric Mapping software (SPM8; http://www.fil.ion.ucl.ac.uk/spm). SVR-LSM, a multivariate lesion-symptom mapping approach, 23 was used to investigate the correlation between lesion location and the change rates of verbal memory scores within 12 weeks after stroke after controlling for age, sex, education, and lesion volume, using MATLAB R2014a. Only voxels damaged in at least 10% of patients were included in the analysis. Probabilistic maps created using 1000 permutations of the RAVLT scores were thresholded at P <.001.
Whole Brain Segmentation
Before segmentation, lesion tracings were employed to mask damaged tissue to achieve correct segmentation. All T1-weighted images were automatically segmented using the online platform MRICloud (http://www.mricloud.org) 24 through a T1-WI segmentation pipeline, which is built on the diffeomorphic multi-atlas likelihood-fusion algorithm in the framework of the random deformable template model.25,26 In this study, a set of 50 adult brain atlases (median age = 53 years) were used. Those 50 atlases are freely accessible at the MRICloud platform. The whole brain was segmented into 289 structures, which could be divided into 5 levels of granularity according to their ontological relationships. In this study, we used a median level of granularity—that is, level 3, wherein the whole brain was segmented into 54 structures. We chose this level because it matches the radiologists’ readings relatively well, and it has high segmentation reliability. 27 Details about the segmentation method can be found in the supplemental material.
Shape Processing
As shown below, only the thalamus survived the whole-brain–based volumetric statistical analyses. Also driven by plausible hypotheses, we conducted shape analysis on the thalamus. We first used an established approach16,28 to create a triangulated surface contouring the boundary of each thalamic segmentation (left and right separately). This method has been successfully used to investigate subcortical structural shapes in neurodegenerative disorders. 16 Details about the shape processing method can be found in the Supplemental Material. In brief, we first manually created a triangulated model surface, ensuring sufficient smoothness and correct anatomical topology. Large deformation diffeomorphic metric mapping (LDDMM) was then applied to deform the model surface, resulting in a close approximation of the contouring surface of the segmented volume. These resultant surfaces are the target shapes whose vertex-wise morphometrics were later analyzed.
The vertex-wise shape morphometrics of a target shape were quantified using a diffeomorphism connecting a common template shape to the target shape for the left and right thalamus. The common template shape of the thalamus in each hemisphere was obtained from the collection of all controls’ surfaces via an established Bayesian template estimation algorithm. 29 The LDDMM surface algorithm 30 was then used to obtain a diffeomorphism carrying the common template surface to each target surface. A scalar field was later computed from each of these diffeomorphisms—that is, the Jacobian log-determinant of the diffeomorphism, indexed at each vertex of the common template surface. This scalar field quantifies the factor by which the diffeomorphism expands or shrinks the vertex-based localized surface area in the target relative to the template. A positive value indicates that the target surface has an expanding localized surface area relative to the template, whereas a negative value suggests decreasing localized surface area in the target. This scalar field is the shape deformation marker that we subsequently analyzed.
Group Comparison Analysis
Group differences between every 2 of the 3 time points were statistically analyzed using a multivariate linear regression model:
where k indexes features, s indexes subjects, Jk(s) denotes the volumetric data of the kth structure or the shape deformation marker at the kth vertex of subject s, G(s) is the group indicator [G(s) = 0 for one group and G(s) = 1 for the other group in the comparison], Xcov(s) is the covariate information (baseline age, sex, and intracranial volume), and ε
k
(s) is Gaussian noise:
Correlation Analysis
We analyzed the association between the change rate of verbal memory and that of volumetric atrophy of structures that survived the aforementioned group comparison analysis (left and right hemispheres separately). We first estimated the rate of change in the volume of each structure via a linear regression using volumes at all 3 time points (W1, W4, and W12). The rate of change of the memory score was similarly computed. We next factored out the effects of sex, age, intracranial volume, lesion volume, and education and then performed correlation analysis, which was quantified by the Pearson product-moment correlation coefficient (PCC). The approach for the shape correlation analysis was similar, except that it was conducted at every vertex of the thalamus that survived the group comparison analysis. Multiple correlation correction was conducted again at a 5% FDR.
Template Subdivision
There are multiple subregions in the thalamus, and each of them contributes to distinct functions. We divided our bilateral thalamic shapes into 7 subregions based on a predefined atlas using an approach described previously. 16 The thalamic subregions were assigned connections to cortical regions according to the most likely diffusion tensor imaging (DTI) tractography. The 7 thalamic subregions are defined as those connected to the posterior parietal cortex, premotor cortex, primary motor cortex, occipital cortex, sensory cortex, prefrontal cortex, and temporal cortex. 31
Statistical Analyses
In the comparisons of participants’ demographic data, baseline characteristics, and behavioral test scores, categorical data were assessed using the Pearson χ2 or Fisher exact tests (when the expected number was ≤5), numerical data were assessed using the Mann-Whitney U test, and repeated measures tests were used for longitudinal within-group comparisons. Post hoc analyses were performed using the Bonferroni correction for multiple comparisons. All analyses were performed using SPSS 16.0 for Windows software (SPSS, Chicago, IL), and the significance level was set at P <.05.
Results
Participant Characteristics and Behavioral Evaluation
A total of 9 patients were excluded from the analyses because of distortion on MRI data caused by movement (n = 6) or early scan termination (n = 3). This resulted in a final study sample of 50 patients (28 left hemisphere and 22 right hemisphere; Figure 1). Demographic information, clinical characteristics, and behavioral test scores for all participants are detailed in Table 1. No significant group differences were found in age, sex, and vascular risk factors at baseline. As shown in Supplemental Tables A and B, 21 (75%) and 20 (71.4%) patients with left-hemispheric stroke experienced deteriorations in immediate recall and long-delay recall at W12 after stroke. Nevertheless, most patients with right-hemispheric stroke remained relatively stable or had improved verbal memory function. As shown in Supplemental Figure and Table 1, the left-hemispheric stroke patients performed significantly worse on both immediate verbal recall and long-delay recall at W12 compared with both W1 (all P < .01) and control participants (all P < .01). Patients with right-hemisphere stroke did not experience any verbal memory deteriorations, as indicated by the absence of significant changes on scores of immediate recall and long-delay recall at W12 relative to scores at W1 and those of controls.
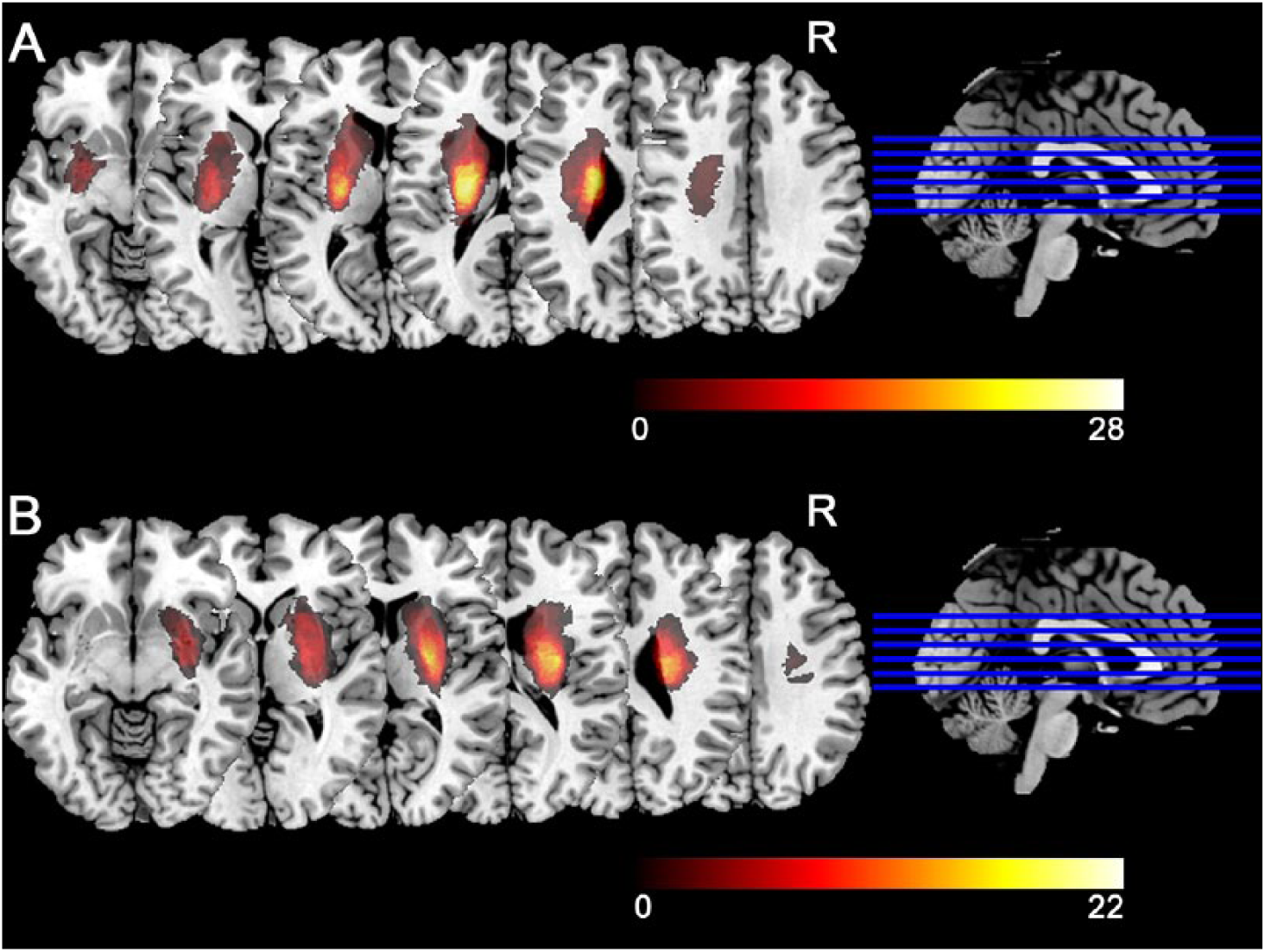
Lesion maps in patients. Panels A and B, respectively, denote the lesion maps from patients with left- and right-hemisphere stroke. Colors represent the numbers of patients with lesions at the given location.
Participants’ Demographic Information, Clinical Characteristics, and Behavioral Test Scores. a
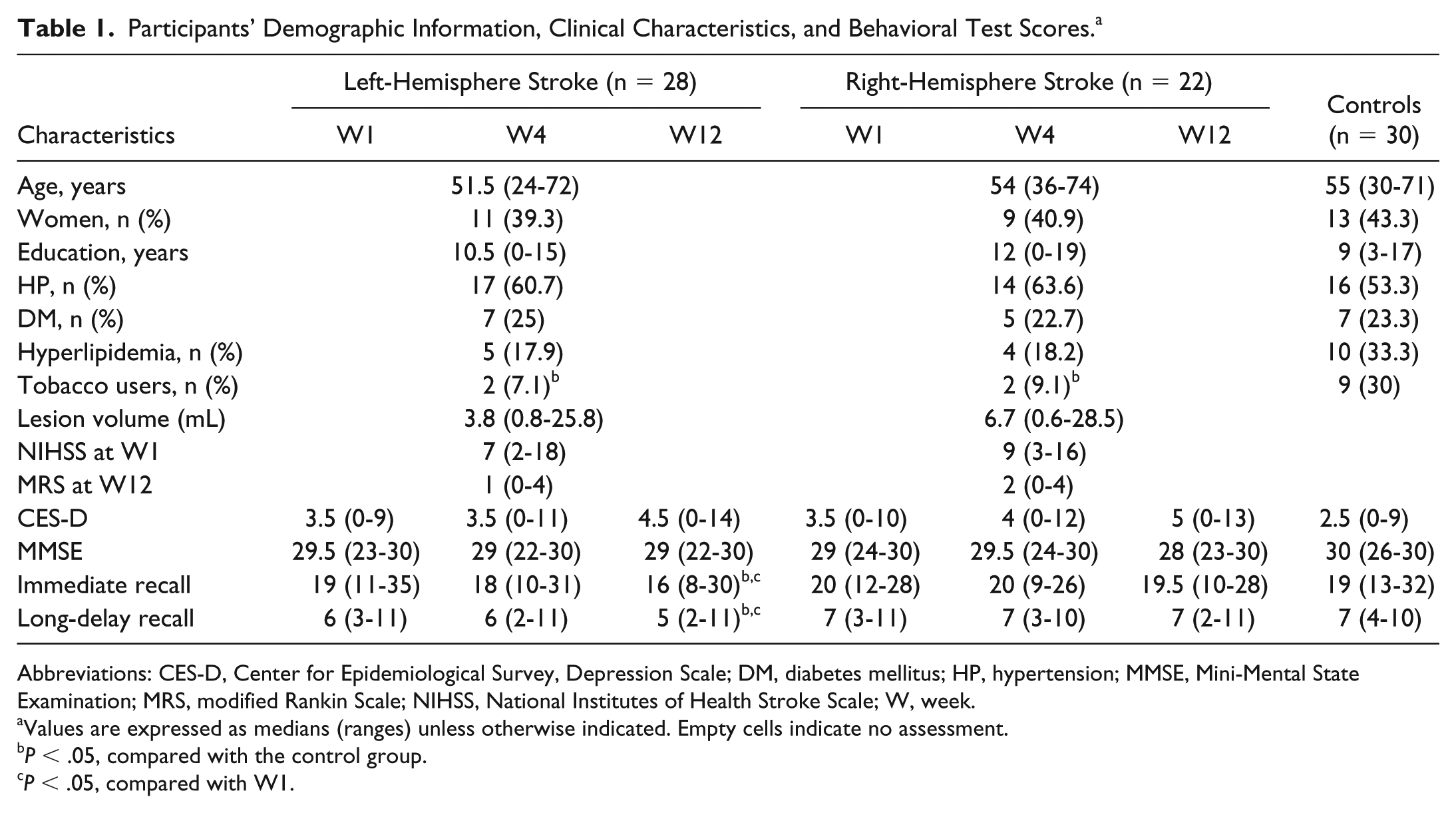
Abbreviations: CES-D, Center for Epidemiological Survey, Depression Scale; DM, diabetes mellitus; HP, hypertension; MMSE, Mini-Mental State Examination; MRS, modified Rankin Scale; NIHSS, National Institutes of Health Stroke Scale; W, week.
Values are expressed as medians (ranges) unless otherwise indicated. Empty cells indicate no assessment.
P < .05, compared with the control group.
P < .05, compared with W1.
SVR-LSM Analysis
The SVR-LSM analysis revealed no significant associations between lesion location and the change rates of scores in immediate verbal recall or long-delay recall in the left- and right-hemisphere stroke patients when age, sex, education, and lesion volume were included as covariates.
Whole-Brain Analysis
As shown in Supplemental Table C, in our whole-brain segmentation based volumetric analyses, we did not find any significant group difference for any of the 54 structures of interest, between any 2 sequential time points (W1 vs W4 and W4 vs W12), after FDR correction. Between W1 and W12, 3 left-hemisphere structures (the basal ganglia, the inferior white matter, and the thalamus) survived FDR correction in the left-hemisphere stroke group. Among those 3 structures, only the rate of change in thalamus volume was significantly positively correlated with the rate of change in immediate recall, but not long-delay recall, after another FDR correction (Supplemental Table D). For the right-hemisphere stroke group, only the basal ganglia in the right hemisphere survived the W1 versus W12 group comparison, but its change rate was not significantly correlated with the verbal memory change rate (Supplemental Table D). From our whole-brain volumetric analyses, we have identified the thalamus as the key structure for more refined investigation.
Thalamus Analysis
As shown in Table 2, the volume of the ipsilesional thalamus in each stroke group decreased gradually from W1 to W4 and finally to W12, with the group differences between W4 and W12 as well as between W1 and W12 reaching statistical significance. There were no significant group differences among the 3 time points in the contralesional thalamus volumes. The change rate of the left-thalamus volume was positively correlated with that of immediate recall, but not long-delay recall, in the left-hemisphere stroke group (PCC = 0.4497; P = .0164). The correlation for the ipsilesional thalamus in the right-hemisphere stroke group did not reach statistical significance. In addition, we found that a larger stroke lesion volume was associated with a larger volume reduction of the ipsilesional thalamus from W1 to W12 in all patients (PCC = −0.394; P = .005).
Longitudinal Group Comparisons Result in Terms of the Thalamus Volumes (mm3). a
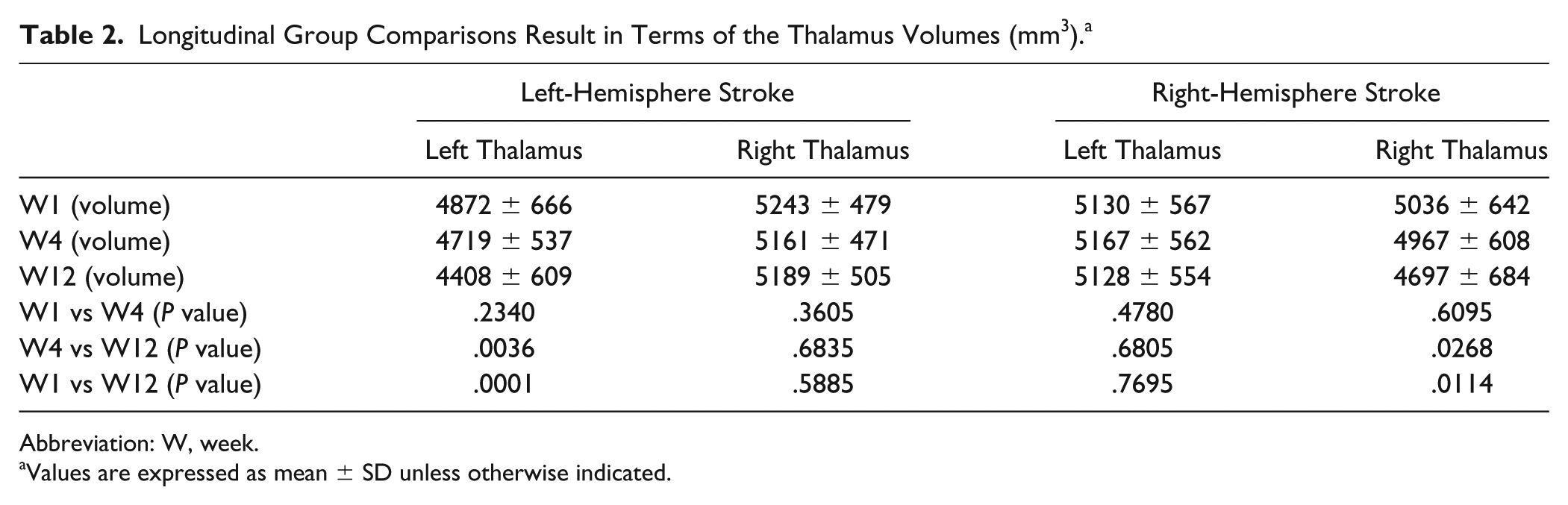
Abbreviation: W, week.
Values are expressed as mean ± SD unless otherwise indicated.
As shown in Figure 2, we observed significant region-specific group differences between W1 and W12 in the ipsilesional thalamus shape in both stroke groups. As revealed by the previous subdivision results (Figure 2C), 16 the W1 versus W12 shape differences mainly occurred at vertices connecting to the prefrontal, temporal, and premotor cortices (Figure 2A). From the vertex-wise correlation analysis (Figure 2B), we also detected region-specific significant positive correlations in the left-thalamus shape of the left-hemisphere stroke group. The location of vertices involved in the shape correlation results agreed with the group comparison results. No vertex of the right-thalamic shape survived in the correlation analysis, after FDR correction, in the right-hemisphere stroke group.
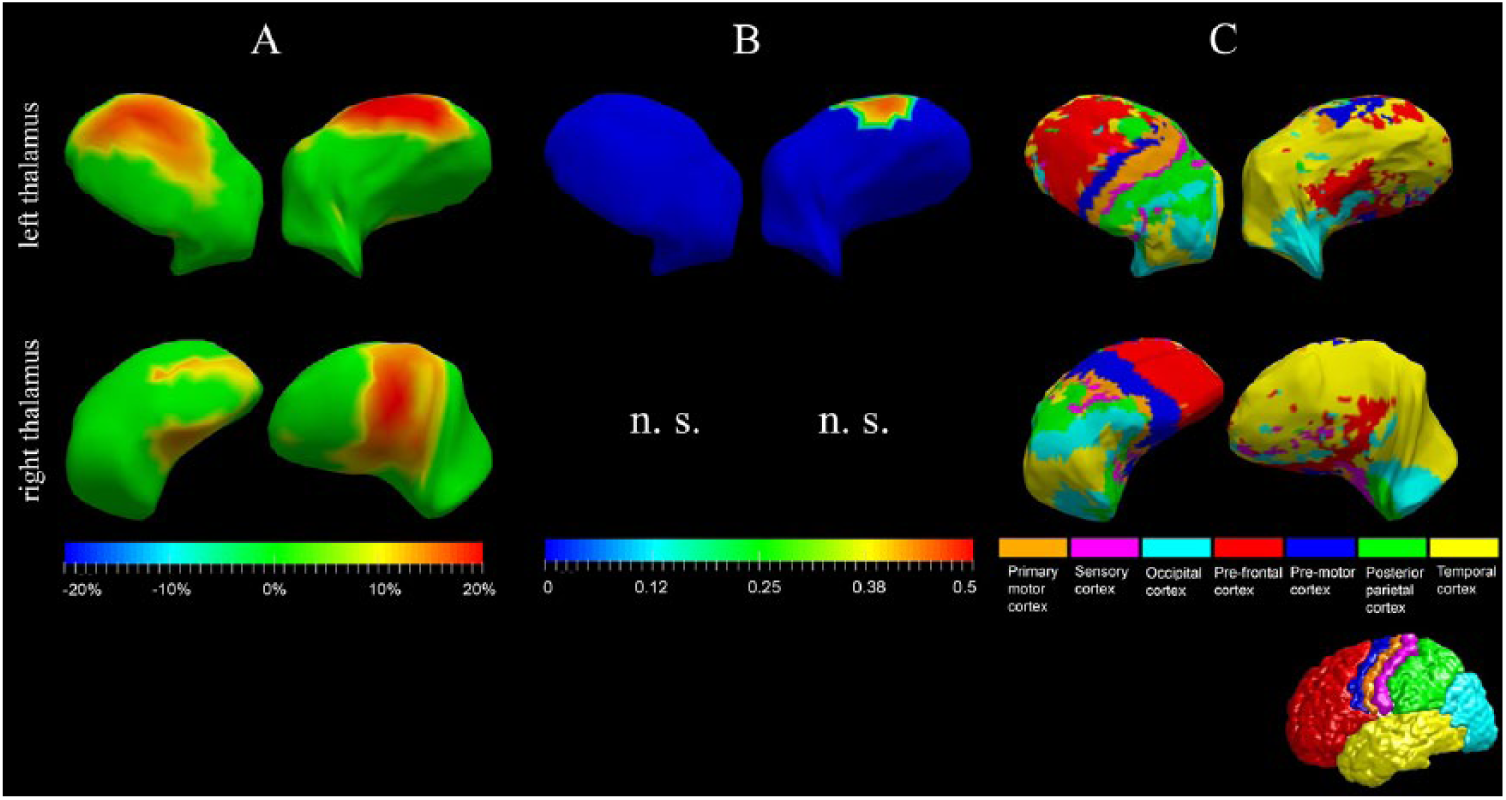
Thalamic shape analysis. Panel A denotes the W1 versus W12 group differences in terms of the ipsilesional thalamus shape in both stroke groups (left: top panel; right: bottom panel). Only vertices on which the group differences are significant after FDR correction are highlighted. The color bar represents the percentage of compression (positive values) or expansion (negative values) in W12 relative to W1 in terms of the vertex-wise surface areas. Panel B denotes the significant correlation results between the vertex-wise shape changing rates and the rate of change in immediate recall, and the color bar represents the PCC values. Again, only vertices for which the correlations are significant after FDR correction are highlighted. Panel C denotes the 7-subregion division results of the left- and right-thalamus shapes according to their connection to different cortical regions.
Discussion
Consistent with previous studies,6,8 in this study, we found that patients with subcortical infarction, particularly those with left-hemisphere stroke, had a poor performance on verbal memory subtests. This complies with the classic notion of verbal memory deficits following lesions of the left (“dominant”) hemisphere and nonverbal memory deficits following lesions of the right (“nondominant”) hemisphere. 32 These findings are in line with those reported previously, 33 wherein the left-hemispheric lesion was a better predictor of poorer performance on the RAVLT subtests of immediate recall and delayed recall at 3 months after onset than the right-hemispheric lesion. Our results nevertheless seem to contradict findings from previous studies in which verbal memory deficits were also observed in the chronic phase of right-hemispheric stroke.34,35 The relatively small sample size, short observation period, and practice effects over the short course of the present study may explain this difference. Longitudinal studies with a larger sample size and a longer observation period should be conducted to refine our findings.
The blood to the thalamus is not directly supplied by the MCA. Thus, rather than being the direct consequence of a vascular impairment to the thalamus, there must be a differential secondary mechanism of thalamic damage following MCA infarcts. Microstructural damage to the ipsilesional thalamus, as characterized by mean diffusivity increases, after MCA infarcts have been demonstrated by previous DTI studies,36,37 which was considered a consequence of a secondary degeneration of cortico-thalamic or thalamo-cortical pathways.36,37 In this study, we also found volumetric reductions of the ipsilesional thalamus at an early stage after subcortical infarction within the territory of the MCA. We adopted a longitudinal design, and our data demonstrated a significant association between stroke and temporal loss of the ipsilesional thalamic volume, given that we have observed gradually reduced volumes of the ipsilesional thalamus but not the contralesional one, within 3 months after stroke onset. Additional support for the association between stroke and ipsilesional thalamic volume comes from our finding that a larger stroke lesion volume was associated with a larger change in the ipsilesional thalamic volumes between W1 and W12 in all patients. Collectively, our findings suggest that it is highly unlikely that these patients already had smaller ipsilesional thalamic volumes before stroke onset.
Studies have shown that strategically located thalamic infarcts, especially damage to the left-anterior thalamic nucleus, can impair short-term and long-term verbal memory, particularly encoding and recalling. 13 It is assumed that a strategically located lesion in the thalamus may disrupt an entire neuronal circuit and its memory processing. Findings from this study revealed that nonstrategically positioned subcortical infarcts could also induce immediate recall deficits. Our SVR-LSM analysis did not reveal any significant associations between lesion location and verbal memory impairment, which is consistent with previous findings. 38 These results suggest that the lesion itself may not be the critical neural mechanism for verbal memory impairment in the early phase of subcortical stroke in the area of the MCA. We found a significant positive correlation between the change rate of the left-thalamus volume and that of immediate recall, but not that of long-delay recall, in the left-hemisphere stroke patients within 12 weeks after onset. Shape analysis further revealed that atrophy of thalamic subregions connected to the left-temporal, premotor, and prefrontal cortices was significantly correlated with immediate recall deficits in an early stage of stroke. A robust correlation between verbal memory task performance and the volume of the thalamic region connected to the left-temporal lobe has been recently reported in 121 healthy individuals. 11 In addition, functional imaging studies indicated that the left-premotor and prefrontal cortices are important components of the core areas involved in immediate verbal memory processing in healthy individuals.39,40 Together, these findings suggest that the possible neural mechanism underlying verbal memory deficits at an early stage of subcortical stroke may be the result of axonal degeneration secondary to stroke lesions in the thalamo-cortical pathways and that thalamic atrophy is seen. Somewhat surprisingly, our correlation analyses yielded no significant association between the change rate of the left-thalamus volume and that of long-delay recall in patients with left-hemisphere stroke, whereas such a relationship has been detected in patients with Alzheimer’s disease. 41 In general, patients with Alzheimer’s disease have relatively large reductions in thalamic volume. It is plausible that once the thalamic volume loss in stroke becomes sufficiently large (similar to that observed in Alzheimer’s disease), the relationship between thalamic volume and long-delay verbal memory may become more evident. 42
It has been shown that the morphology of brain regions that are functionally relevant to task performance is important for rehabilitation outcomes. 43 As such, our findings are helpful for identifying efficient biomarkers for evaluating poststroke verbal memory rehabilitation outcomes from memory rehabilitation programs using targeted, cognitive-behavioral or brain stimulation techniques. Specifically, greater degrees of atrophy in left-thalamic subregions connected to the temporal, premotor, and prefrontal cortices following subcortical stroke may predict less favorable outcomes from specific verbal memory rehabilitation programs. If an accurate prediction can be achieved, structural neuroimaging could eventually become a tool to help individuals with subcortical stroke and their caregivers more objectively select among candidate rehabilitation programs based on poststroke structural scans. In addition, electrophysiological evidence has shown increased accuracy of subsequent recall when applying electrical stimulation to the left thalamus during encoding of verbal tasks. 44 Currently, most studies have primarily focused on cognitive areas of the cortex, including the fronto-parietal and cingulo-opercular networks, as target sites during brain stimulation therapies in cognitive rehabilitation for stroke patients. 45 The results of the present study raise the possibility that the left thalamus, or more specifically certain left-thalamic subregions, could theoretically serve as an alternative locus of stimulation for verbal memory rehabilitation in these patients.
Some limitations of the current study should be considered. First, the patterns of changes in the ipsilesional thalamo-cortical pathways and their roles in thalamic atrophy and verbal memory impairment after subcortical infarction cannot be fully elucidated from this study, because verbal memory impairment following subcortical stroke may be the result of pathway disruption and that thalamic atrophy is seen. Additionally, a practice effect may occur in the verbal memory testing after stroke because practice effects are particularly prominent during early phases of high-frequency repetitive cognitive testing. This phenomenon may partially explain why verbal memory deficits were not apparent at 4 weeks poststroke but emerged at 12 weeks. Only 1 session of MRI acquisition as well as cognitive testing for the control group is another limitation. Having data at similar intervals from controls would be ideal. In addition, considering that the thalamus has multiple neuronal connections with cortical areas involved in various cognitive functions and mental activities, the relatively small sample size as well as the lack of a complete measurement in neuropsychological function, including visuospatial memory, language, perception, and emotion, are limitations of the present study. Finally, inclusion of controls whose education level did not directly match that of the patients is also a limitation of this study.
Conclusions
In summary, we found significant volumetric reductions in the ipsilesional thalamus within 3 months of stroke onset in patients with subcortical infarction in the area of MCA. Moreover, atrophy in the left-thalamic subregions connected to the temporal, premotor, and prefrontal cortices in patients with left-hemisphere stroke was significantly associated with early verbal memory impairment. To better understand the neural mechanisms underlying verbal memory dysfunction after subcortical infarction, it is recommended that future studies use larger sample sizes and other neuroimaging techniques to focus on changes in thalamo-cortical pathways and functional connectivity between the thalamus and cortical areas in patients with verbal memory deficits.
Supplemental Material
Supplementary_materials – Supplemental material for Regional Shape Abnormalities in Thalamus and Verbal Memory Impairment After Subcortical Infarction
Supplemental material, Supplementary_materials for Regional Shape Abnormalities in Thalamus and Verbal Memory Impairment After Subcortical Infarction by Gang Liu, Xiaoqing Tan, Chao Dang, Shuangquan Tan, Shihui Xing, Nianwei Huang, Kangqiang Peng, Chuanmiao Xie, Xiaoying Tang and Jinsheng Zeng in Neurorehabilitation and Neural Repair
Footnotes
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of the article.
Authors’ Note
Gang Liu and Xiaoqing Tan contributed equally to this work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Key R&D Program of China (2017YFC1307500 and 2017YFC0112404), the National Natural Science Foundation of China (81371277, 81571107, 81501546, 81500994, 81600998, and 81771137), the Natural Science Foundation of Guangdong Province (2016A030310132 and 2016A030310165), the Special Funds of Public Interest Research and Capacity Building of Guangdong Province (2014B020212003), grants from the SYSU-CMU Shunde International Joint Research Institute Start-up Grant (20150306), the National Key Clinical Department, National Key Discipline, the Sun Yat-sen University Clinical Research 5010 Program (2018001), the Guangdong Provincial Key Laboratory for Diagnosis and Treatment of Major Neurological Diseases (2017B030314103), the Southern China International Cooperation Base for Early Intervention and Functional Rehabilitation of Neurological Diseases (2015B050501003), the Guangdong Provincial Engineering Center For Major Neurological Disease Treatment, and the Guangdong Provincial Translational Medicine Innovation Platform for Diagnosis and Treatment of Major Neurological Disease.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.