Abstract
Objective. Conduct a systematic review of nonpharmacological interventions applied during sleep to enhance physical rehabilitation outcomes of individuals with a neurological diagnosis. Data Sources. Three online databases were searched for original research. Study Selection. Intervention studies were included that used outcome measures of impairment, activity, and/or participation. Data Extraction. Two reviewers independently screened 2287 titles and abstracts, reviewed 101 full texts, extracted data, and assessed study quality and risk of bias for 9 included studies. Data Synthesis. All included studies were randomized controlled trials involving continuous positive airway pressure (CPAP) with inpatient individuals with stroke and sleep apnea. Several studies also included long-term outpatient follow-ups. Results in terms of outcomes based on impairment, activity, and participation were mixed. However, several studies found that the use of CPAP following stroke and sleep apnea during early stroke recovery had benefits relative to no CPAP. Conclusions. The only nonpharmacological intervention to be administered during sleep in a neurological population to improve physical rehabilitation outcomes was found to be CPAP. This review was complicated by the variety of outcome measures used, lack of physical rehabilitation description, and CPAP compliance. In general, participants who had acceptable to good CPAP compliance saw the largest improvements in physical rehabilitation outcomes. Several other promising methods of brain stimulation during sleep are discussed.
Keywords
Introduction
Physical rehabilitation tends to focus on repetitive task practice to promote skill acquisition, but there is an additional element to learning that has received little attention. This stage is called sensorimotor memory consolidation. Following initial skill acquisition, the neural activation pattern associated with a given learned sensorimotor task evolves toward consolidation over several hours, days, and subsequent weeks.1-3 These changes result in long-term stabilization and/or enhancement of performance, 4 allowing performance with greater efficiency and automaticity and decreased cognitive burden.5-7 Sleep is an important driver of consolidation 8 and remains important following neurological injury such as stroke.9-11 However, people with neurological conditions or neurological injury often experience sleep disruptions.12,13 Therefore, evidence that sleep-related processes enhance sensorimotor consolidation provide a promising mechanism by which rehabilitation outcomes may be improved.
Recently, interest in the potential to enhance sensorimotor consolidation by augmenting the effects of sleep has increased. Such techniques include continuous positive airway pressure (CPAP) to prevent disruption of sleep as well as various methods of brain stimulation to enhance aspects of sleep, such as transcranial magnetic stimulation (TMS) and transcranial direct current stimulation (tDCS). Given that various methods of external stimulation are being used during the awake period as a means to enhance skill acquisition during physical rehabilitation (eg, TMS, tDCS), this systematic review aimed to determine if any such methods have been investigated during sleep to enhance physical rehabilitation outcomes in neurological populations.
Objective
To conduct a systematic review of nonpharmacological interventions applied during sleep to enhance physical rehabilitation outcomes of individuals with a neurological diagnosis.
Methods
Criteria for Considering Studies for This Review
Studies
The search included all study designs. Studies of interest that did not meet all criteria are reported in the future directions section of the discussion.
Participants
Those who participated were individuals with a neurological diagnosis involving the sensorimotor system.
Interventions
Interventions included any nonpharmacological intervention provided during sleep.
Outcomes
Changes in commonly used and accepted rehabilitation measures were the outcomes.
Search Methods for Identification of Studies
On September 15, 2017, PubMed (1809 to present; Appendix 1, available online), EMBASE (embase.com, 1974 to present; Appendix 2), and Cochrane Central Register of Controlled Trials (Appendix 3) were searched, and results were uploaded into systematic review software (http://www.Covidence.org). Results had at least 1 term from each of 3 concepts: sleep, functional capacity and rehabilitation, and neurological disorders. Terms were modified for each database, and strategies incorporated both keywords and subject headings. No date restriction or other limits were applied. Only studies in English were considered. Bibliographies of included studies were searched for additional studies.
Data Collection and Analysis
Two authors reviewed titles, keywords, and abstracts of reports identified from search results, and extracted data from included reports were placed into a data extraction template. Extracted data were used to assess the risk of bias using the Cochrane Risk of Bias Assessment Tool for each included study. 14 Cases of conflicting decisions between reviewers were resolved through further discussion.
Results
Study Descriptions
The study selection process is outlined in the Preferred Reporting Items for Systematic Reviews (PRISMA) diagram (Figure 1). The searches yielded 2757 references (PubMed: 624; EMBASE: 1863; Cochrane: 270), with 470 duplicates. Of 2287 references, 2168 were irrelevant, and 99 were reviewed in full. Full-text review excluded 92, and 2 additional studies were identified from bibliographies of the included studies and subsequently passed all previous steps. Therefore, 9 studies were included (Table 1). All 9 studies were randomized controlled trials involving CPAP poststroke.
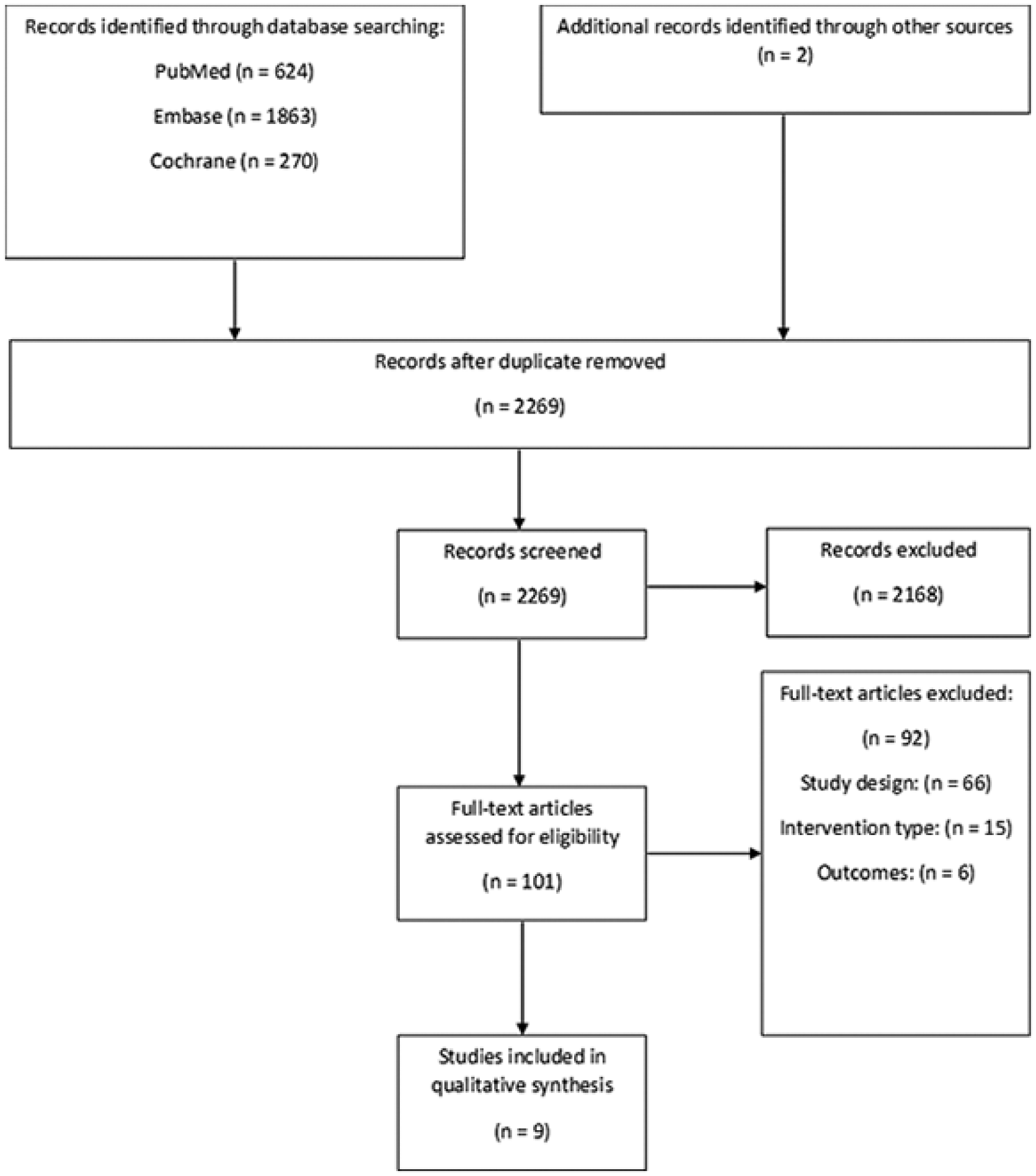
Study flow diagram.
Overview of Included Studies.
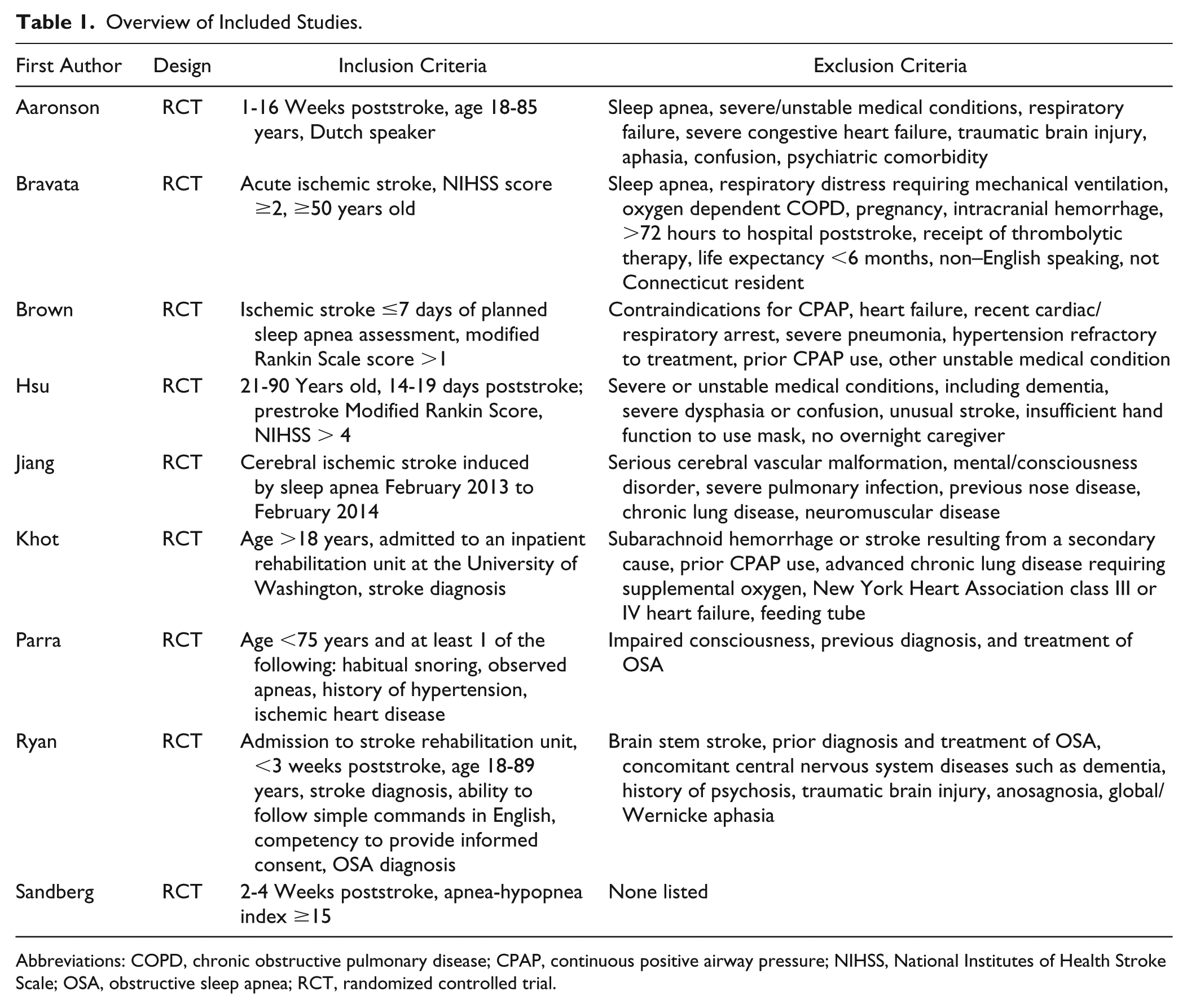
Abbreviations: COPD, chronic obstructive pulmonary disease; CPAP, continuous positive airway pressure; NIHSS, National Institutes of Health Stroke Scale; OSA, obstructive sleep apnea; RCT, randomized controlled trial.
Included Studies
Aaronson et al 15 compared nightly CPAP use with usual treatment for 4 weeks on an inpatient rehabilitation unit in individuals who were between 1 and 16 weeks poststroke. Patients who were discharged prior to 4 weeks were followed as outpatients. Groups were balanced by age, severity of sleep apnea, stroke subtype (ischemic or hemorrhagic), and severity of cognitive impairment. All participants in the CPAP group presented with sleep apnea (n = 20), whereas participants in the control group who did not receive CPAP treatment either presented with (n = 16) or without (n = 44) sleep apnea.
Bravata et al 16 assessed the use of autotitrating CPAP (auto-CPAP) for 30 days compared with no CPAP on an inpatient rehabilitation unit among people with ischemic stroke <72 hours. Randomization to CPAP and control groups was balanced by stroke severity (ie, National Institutes of Health Stroke Scale Score, NIHSS <10 or ≥10). Only those individuals randomized to the CPAP group with evidence of sleep apnea after 2 days continued in the intervention group for the complete 30 days (n = 16). Individuals randomized to the no-CPAP control group were diagnosed with or without sleep apnea (n = 24).
Brown et al 17 randomized individuals admitted to an inpatient neurology unit with ischemic stroke of <7 days and sleep apnea to receive 3 months of either CPAP (n = 15) or sham CPAP (n = 17).
Hsu et al 18 enrolled individuals with ischemic stroke and severe sleep apnea, who were between 21 and 25 days poststroke, to either a group with nasal CPAP (n = 15) or conservative (n = 15) treatment for severe sleep apnea. To ensure CPAP compliance, an experienced research nurse provided input on days 1, 3, and 5 and weeks 1, 2, 4, and 6 of treatment. Rehabilitation programs were maintained as usual. In addition to the 8-week assessment, follow-up testing was conducted for all participants at 6 months poststroke.
Jiang et al 19 investigated individuals diagnosed with ischemic stroke induced by obstructive sleep apnea hypopnea syndrome (OSAHS). In this study, nasotracheal suction of mechanical ventilation (n = 29) was the treatment of interest, and CPAP (n = 24) was the control intervention. Both interventions took place during the 7 days after hospital admission. The authors state that nasotracheal suction of mechanical ventilation therapy can help individuals with moderate to severe OSAHS get sufficient sputum drainage and maintain airway patency. All patients underwent conventional therapy for stroke.
Khot et al 20 investigated the use of auto-CPAP (n = 20) compared with sham CPAP (n = 20) in individuals with ischemic or hemorrhagic stroke (median days poststroke = 10) throughout their stay on an inpatient rehabilitation unit or for a maximum of 28 days. The authors noted that, given the high prevalence of sleep apnea in individuals with strokes and inconsistencies in cutoff points, a screening diagnostic test was not conducted. A sleep technologist met with patients at least twice weekly to reinforce CPAP adherence.
Parra et al 21 assessed the use of nasal-CPAP (n = 71) compared with conventional no-CPAP (n = 69) therapy in individuals admitted to a neurology unit with an ischemic stroke 3 to 6 days prior to study initiation and moderate to severe sleep apnea. Participants were followed for 2 years. Assessments were conducted at baseline and at 1, 3, 12, and 24 months. Physical rehabilitation was conducted prior to discharge from the hospital (mean ± SD length of hospital stay: 8.7 ± 4.3 days in the CPAP group and 9.3 ± 4.6 days in the control group).
Ryan et al 22 compared 4 weeks of CPAP use (n = 22) to no CPAP use (n = 22) in participants who were admitted to an acute stroke rehabilitation unit within 3 weeks of ischemic or hemorrhagic stroke onset, with a diagnosis of obstructive sleep apnea. Time spent and level of participation in physical therapy was recorded throughout the trial and found to be similar between the 2 groups (approximately 45 min/d, 5 d/wk).
Sandberg et al 23 randomized individuals with stroke 2 to 4 weeks prior and with central or obstructive sleep apnea to receive nasal CPAP (n = 33) or no CPAP (n = 30) for 4 weeks while on an inpatient stroke rehabilitation unit. Assessments were conducted at baseline, 7 days, and 28 days.
Excluded Studies
Studies that were excluded after full-text review did not meet the inclusion criteria relating to study design (ie, not an intervention study; 66 excluded), intervention type (15 excluded), outcomes (6 excluded), language other than English (3 excluded), or population (2 excluded).
Risk of Bias in Included Studies
Judgments regarding each risk-of-bias item for the included studies are presented in Table 2.
Risk-of-Bias Summary. a
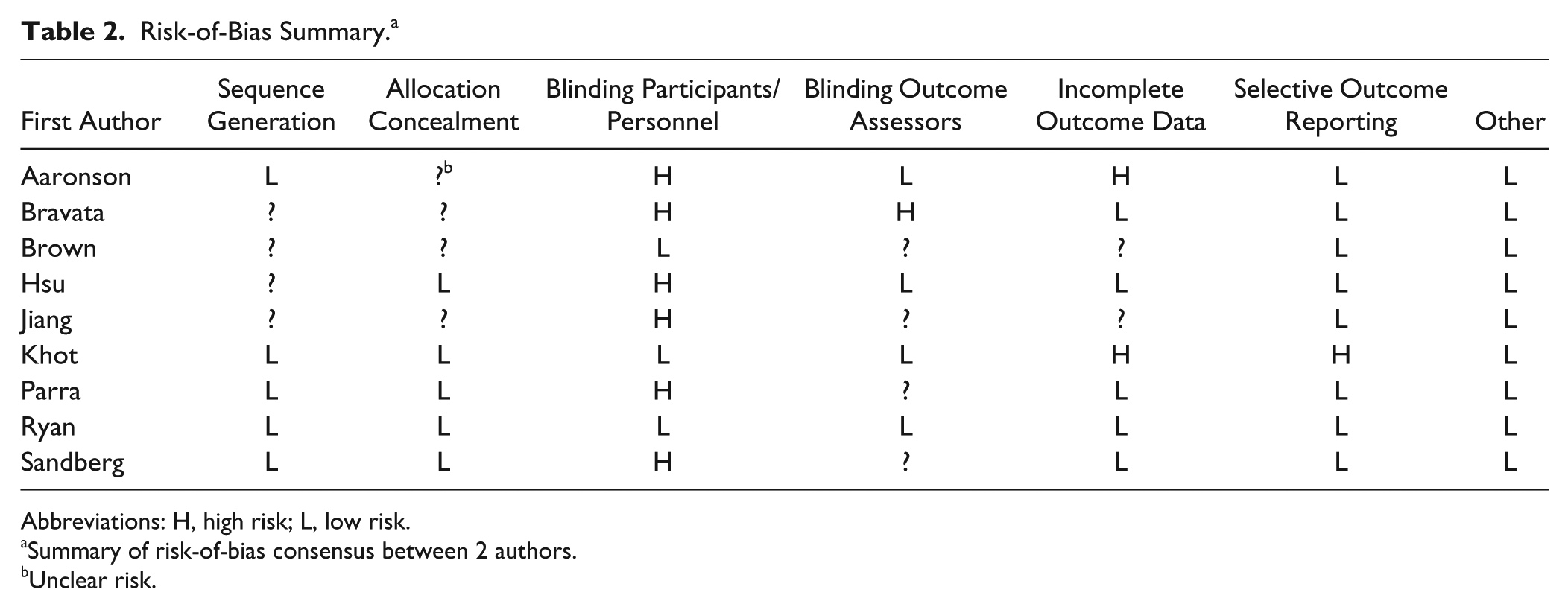
Abbreviations: H, high risk; L, low risk.
Summary of risk-of-bias consensus between 2 authors.
Unclear risk.
Effects of Intervention
Because various rehabilitation outcome measures were used, results are grouped by outcome measure type as defined by the International Classification of Functioning, Disability, and Health 24 : impairment, activity, and participation. Briefly, impairments involve problems in body structure or function, whereas activity involves executing a task or action, and participation involves a life situation or role. 24
Impairment
Aaronson et al 15 assessed neurological status by transforming NIHSS and Canadian Neurological Scale scores into z-scores and then averaging the 2 scores together. Using this method, no significant difference in change scores were found between the CPAP and no-CPAP groups (P = .08).
Bravata et al 16 found a greater median NIHSS improvement in the CPAP group compared with the no-CPAP control group from baseline to the end of the 30-day period (auto-CPAP: −3.0; no-CPAP: −1.0; P = .03). A trend was also noted toward greater NIHSS improvement linked to earlier use of auto-CPAP following stroke (median NIHSS change: −3.0 for <24 hours; −3.0 between 24 and 48 hours; and −0 for ≥48 hours) and greater CPAP use (median NIHSS change: −3.0 for acceptable adherence [n = 10], −2.5 for some use [n = 6], and −1.0 for no use [n = 13]).
Brown et al 17 assessed active CPAP compared with sham CPAP based on the NIHSS at the 3-month follow-up. Of the participants who returned for follow-up (active CPAP, n = 7; sham CPAP, n = 10), no significant differences were found (NIHSS 1 versus 2, respectively).
Hsu et al 18 found no statistically significant difference in NIHSS scores between the CPAP and no-CPAP groups at the 3-month assessment period (NIHSS 2 versus 3, P = .622). NIHSS scores were not obtained at the 6-month follow-up.
Jiang et al 19 assessed between-group differences in nasotracheal suction mechanical ventilation compared with CPAP, with subgroups defined by severity of obstructive sleep apnea. After 7 days of treatment, there were no differences between groups in terms of NIHSS score in the mild obstructive sleep apnea group (suction: 8.54; CPAP: 9.03; P = .114). However, an evaluation of severity subgroups revealed that the NIHSS was significantly lower (ie, more improved) among individuals in the nasotracheal suction group with moderate (suction: 9.32; CPAP: 11.77; P = .033) and severe (suction: 11.45; CPAP: 14.85; P = .023) obstructive sleep apnea.
Parra et al 21 compared the percentage of individuals with improvements on the Canadian Neurological Stroke Scale (ie, increases of ≥5 points or score 10 from the beginning) and found that the percentage of participants who improved at 1 month was significantly higher in the CPAP group than in the no-CPAP group (88.2% vs 72.7%; P = .038). However, there were no statistically significant differences found between the groups in terms of raw scores at 3, 12, and 24 months (3, 12, 24 months: CPAP, 9.3, 9.4, 9.3; no CPAP, 9.3, 9.4, 9.5). The authors did not state whether this analysis was corrected for multiple comparisons.
Ryan et al 22 observed significant improvement in the total score on the Canadian Neurological Scale from baseline to 1 month in the CPAP group (7.3 to 9.6, P = .001) but not in the no-CPAP (7.7 to 8.4, P = .16) group, with the difference in change also being significantly different between groups (P = .001). The ratio of hand grip strength between the affected and nonaffected hands did not change significantly within or between groups (CPAP: 0.39 to 0.45, P = .09; no CPAP: 0.48 to 0.61, P = .36).
Activity
Brown et al 17 found that of the participants who returned for 3-month follow-up, no significant differences were identified between CPAP and sham CPAP groups in terms of the Barthel Index (CPAP [n = 8]: 95; sham CPAP [n = 11]: 100) or modified Rankin Scale scores (CPAP [n = 8]: 2; sham CPAP [n = 10]: 2).
Hsu et al 18 found no statistically significant difference in Nottingham Extended Activities of Daily Living (EADL) total (CPAP: 18; no-CPAP: 30; P = .229) or Barthel Index (CPAP: 17; no-CPAP: 19; P = .166) scores between the CPAP and no-CPAP groups at the 3-month assessment period. However, a retrospective exploratory analysis revealed that individuals in the CPAP group had significantly lower EADL mobility subscale scores than the no-CPAP group at 3 months (CPAP: 3; no-CPAP: 6; P = .048). The authors point out that the CPAP group tended to have lower Barthel Index scores at baseline and that when controlling for baseline scores, the difference between groups in the EADL mobility subscore was no longer significant (P = .077). At the 6-month follow-up assessment, there were no statistically significant differences between groups in EADL total (CPAP: 23; no CPAP: 28; P = .498) and subscale scores nor in the Barthel Index scores (CPAP: 19; no CPAP: 18; P = .640).
Jiang et al 19 found no differences between nasotracheal suction and CPAP groups in terms of the Barthel index score in the mild obstructive sleep apnea group (suction: 60.53; CPAP: 59.79; P = .955). However, an evaluation of the obstructive sleep apnea severity subgroupings revealed significantly more improved scores resulting from nasotracheal suction compared with CPAP treatment in the moderate (suction: 55.12; CPAP: 53.97; P = .028) and severe (suction: 54.20; CPAP: 49.13; P = .021) groups.
Khot et al 20 found that patients with active CPAP had a significantly greater change in the cognitive component, but not physical component or total score on the Functional Independence Measure (FIM) compared with those who received sham CPAP. This result occurred in both intention-to-treat (FIM Total, Cognitive, Physical Change: active CPAP, 34, 6, 26; sham CPAP, 26, 2.5, 23; P = .25; P = .04; P = .42) and on-treatment analyses (FIM Total, Cognitive, Physical Change: active CPAP, 32, 6, 29; sham CPAP, 26, 2, 23; P = .11; P = .06; P = .17).
Parra et al 21 compared the percentage of individuals with improvements on the Rankin Scale (ie, reduction of ≥1 point or score 0 from the beginning) and Barthel Index (ie, improvement in the level of disability or no disability from the beginning). The percentage of patients with improvements in the Rankin Scale 1 month after stroke was significantly higher in the CPAP group than in the no-CPAP group (90.9% vs 56.3%; P = .002). However, there were no differences between groups at this time point on the Barthel index (82.7% vs 74.3%; P = .567). The raw scores of these measures did not reveal statistically significant differences between the groups at the reported time points of 3, 12, and 24 months (Rankin Scale at 3, 12, 24 months: CPAP, 1.6, 1.6, 1.8; no CPAP, 2.0, 2.1, 2.2; Barthel Index at 3, 12, 24 months: CPAP, 95.0, 95.3, 94.3; no CPAP, 92.8, 91.4, 93.1).
Ryan et al 22 found that although within-group improvements were generally noted in both groups, the CPAP group improved to a larger extent on measures involving the lower-extremity motor function and gait. In particular, although significant within-group improvements were observed in the total FIM score of both the CPAP group (78.3 to 105.6, P = .001) and no-CPAP group (85.8 to 105.8, P = .001), no statistically significant between-group difference was found (P = .07). However, the CPAP group demonstrated a significantly greater improvement (P = .05) in the motor FIM subscale (54.8 to 78.2) compared with the no-CPAP group (62.5 to 79.2). Similarly, the arm, hand, leg, and foot scores of the Chedoke-McMaster Stroke Assessment were significantly improved in both CPAP (Chedoke-McMaster Stroke Assessment arm, hand, leg, and foot score: 3.8 to 4.9, P = .001; 4.0 to 4.7, P = .001; 4.2 to 5.0, P = .001; 3.6 to 4.3, P = .001) and no-CPAP (Chedoke-McMaster Stroke Assessment arm, hand, leg, and foot score: 4.3 to 4.8, P = .020; 4.0 to 4.7, P = .001; 4.1 to 4.5, P = .027; 3.8 to 4.3, P = .007) groups, with significantly greater improvement in the leg score in the CPAP than in the no-CPAP group (P = .001). In addition, the CPAP group demonstrated a significant increase in the 6-minute walk distance compared with baseline (113 m, P = .02), whereas no difference was observed in the no-CPAP group (46 m, P = .29), although the between-group difference was not significant (P = .75). There were also significant improvements in the Berg Balance Scale within both the CPAP (29.5 to 43.7, P = .01) and no-CPAP (28.2 to 44.3, P = .001) groups, but these scores did not differ between groups (P = .64). No within- or between-group differences were found on the Purdue Pegboard score (CPAP group, affected hand: 28 to 32, P = .62; no-CPAP group, affected hand: 30 to 37, P = .35).
Sandberg et al 23 found that the CPAP treatment and control groups did not significantly differ in Barthel Index change scores from day 1 to night 7 (treatment: 1.5; control: 1.0) or from day 1 to night 28 (treatment: 1.1; control: 1.1).
Participation
Aaronson et al 15 found no group difference between those with and without CPAP treatment in terms of total score change on Utrecht Scale for Evaluation of Rehabilitation (CPAP: 11.85; no CPAP: 9.00; P = .11).
Hsu et al 18 found no statistically significant difference in Short Form (SF)-36 total scores between the CPAP and no-CPAP groups at the 3-month and 6-month assessments. However, a retrospective exploratory analysis revealed that individuals in the CPAP group had significantly improved SF-36 physical summary scores than the no-CPAP group at the 3-month point (CPAP: 23.7; no-CPAP: 30.8; P = .022), which remained even after adjusting for differences in Barthel Index scores at baseline (P = .0008). By the 6-month follow-up, however, the difference on the SF-36 physical subscale was no longer noted (CPAP: 19.8; no-CPAP: 28.4; P = .248).
Parra et al 21 found no significant differences between the CPAP and no-CPAP groups in terms of the physical or mental components of the SF-36 at any of the assessment time points, although progressively better mean scores were noted from 1, 3, 12, to 24 months (Physical Component at 1, 3, 12, 24 months: CPAP, 42.6, 44.9, 46.7, 45.8; no CPAP, 42.3, 44.8, 46.5, 46.0; Mental Component at 1, 3, 12, 24 months: CPAP, 43.3, 46.9, 49.1, 47.6; no CPAP: 43.7, 46.3, 44.6, 47.8).
CPAP Compliance
Participant compliance with CPAP was a common limitation (Table 3).
CPAP Compliance.
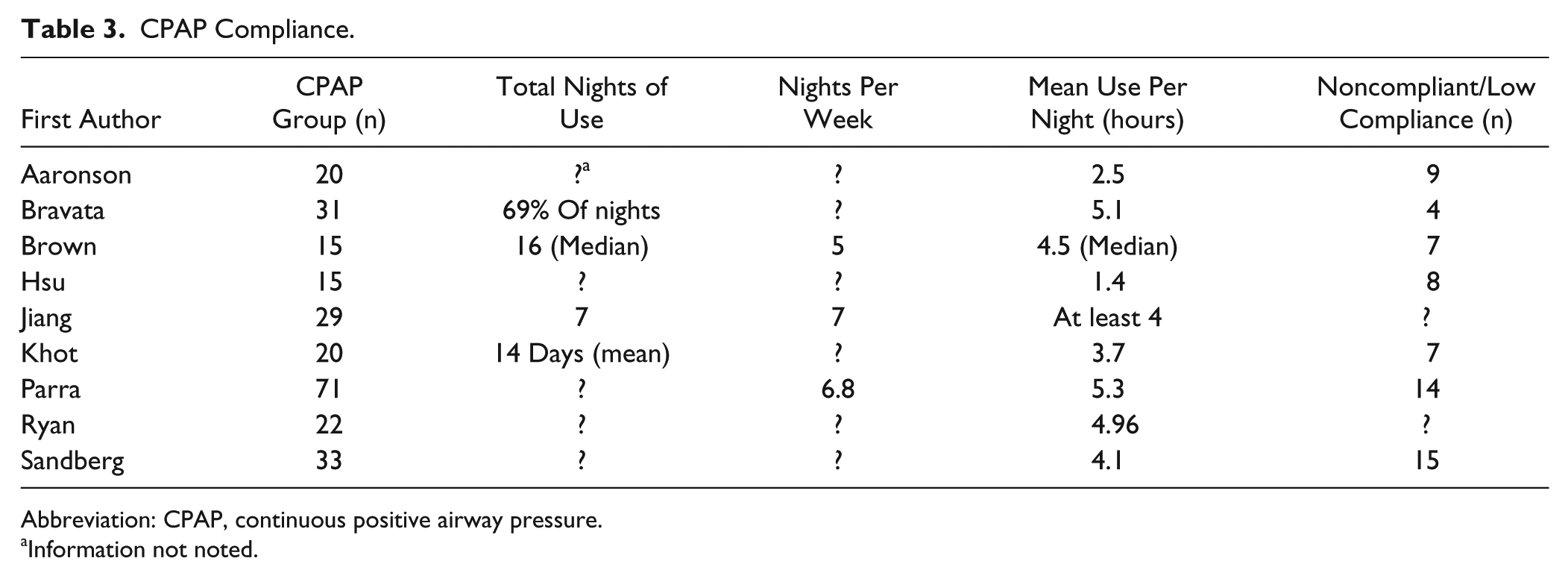
Abbreviation: CPAP, continuous positive airway pressure.
Information not noted.
Aaronson et al 15 found compliance with CPAP to be associated with larger improvements in cognitive functioning. Bravata et al 16 observed that greater CPAP use related to increased improvements in NIHSS at 30 days (median NIHSS change: −3.0 for acceptable adherence, −2.5 for some use, and −1.0 for no use [ie, no-CPAP control group]; no P value). Hsu et al 18 found that CPAP use positively correlated with better Barthel Index scores at baseline (P = .035). Khot et al 20 reported that neither baseline NIHSS (P = .55) nor FIM (no P value) score predicted CPAP compliance.
Discussion
This study aimed to investigate nonpharmacological interventions applied during sleep to enhance physical rehabilitation outcomes of individuals with neurological diagnoses. The only published nonpharmacological intervention to be provided during sleep to enhance physical rehabilitation outcomes in neurological populations has been CPAP in individuals with stroke and sleep apnea. All the included studies were conducted during the acute and subacute phase of stroke recovery, with few including long-term follow-up. When considering the results of participants with good CPAP compliance, an increased benefit was seen in Aaronson et al, 15 Bravata et al, 16 and Hsu et al. 18 Overall, it appears that CPAP use during inpatient rehabilitation following stroke in patients with sleep apnea may accelerate recovery and rehabilitation in the short term, but long-term benefits remain unclear.16,20-22 It should also be noted that the inpatient period tended to have higher CPAP compliance compared with outpatient use, though CPAP compliance was quite variable in the included studies (Table 3) and in other studies including individuals with stroke.25-27 Therefore, more research with high CPAP compliance in the longer term is needed to better understand the benefits of CPAP use in individuals with stroke undergoing physical rehabilitation.
The studies had fair-good methodological quality. However, this review was complicated by 3 factors of heterogeneity: physical rehabilitation regimen, poststroke function, and CPAP compliance. Few descriptions of physical rehabilitation were provided in the included studies. Time in physical rehabilitation and the interventions chosen are known to vary greatly. Given that the rehabilitation effects of CPAP were, in general, better during the inpatient stays, it may be that active participation in daily intensive rehabilitation is a critical factor to pair with nightly CPAP use.16,18,20-22 Moreover, evaluations of subgroup differences revealed that individuals with moderate to severe sleep apnea demonstrated better outcomes than those with milder sleep apnea, suggesting a greater benefit from CPAP use. 19 Furthermore, response to physical rehabilitation following stroke is complicated by many factors, including stroke location, stroke severity, baseline status, age, time since stroke, prior history of stroke, and secondary diagnoses, among others.28-33 In addition, as documented here and elsewhere, a common secondary diagnosis following stroke is sleep apnea, at least during the early (<3 months poststroke) phase of recovery. 12 Given the complicated nature of response to physical rehabilitation following stroke as well as the seemingly ubiquitous difficulty with CPAP compliance across populations, the mixed results of the studies included in this review were not unexpected.
Future Directions
Although interventions applied during sleep following stroke are limited to CPAP, additional interventions have been used with healthy populations that were aimed at enhancing outcomes related to sleep physiology and, in some cases, subsequent learning. These investigations open up new possible directions for research in neurological populations as a means of enhancing rehabilitation outcomes. Such interventions include the following: tDCS, TMS, and auditory stimulation.
Transcranial Direct Current Stimulation
tDCS is a noninvasive neuromodulatory technique to provide weak direct current to the brain and subsequent cortical excitability changes. 34 Application of anodal tDCS oscillating at 0.75 Hz (slow oscillation) bilaterally over frontolateral locations during non–rapid eye movement sleep (NonREM) sleep has been used to enhance slow-wave sleep activity and declarative (but not procedural) learning in healthy young adults,35,36 healthy older adults, 37 and adults with paranoid schizophrenia. 38 Alternately, tDCS applied at a higher frequency of 5 Hz (theta) during NonREM in healthy young adults depressed slow-wave sleep activity and local sleep spindle power, with no resultant change, 35 and even impairment of, 39 subsequent declarative and procedural learning. These contrasting results suggest the importance of tDCS frequency settings, with lower frequencies resulting in enhanced sleep physiology related to motor learning.
Transcranial Magnetic Stimulation
TMS is a noninvasive technique that causes electrical stimulation of neural tissue via rapidly changing magnetic pulses. 40 This technique has been used to trigger slow-wave activity when applied across the scalp during light NonREM sleep, which subsequently entrained thalamic spindles and spread over the scalp. 41 Evidence that the entrainment of thalamic spindles can enhance the sleep-related consolidation process suggests a novel potential role of TMS in motor learning.
Auditory Stimulation
Rhythmic auditory stimulation
Use of rhythmic auditory stimulation has recently been used to entrain and enhance cortical firing during sleep. Several studies have applied rhythmic auditory stimulation phase locked to slow-wave up-states in healthy young adults. This has led to increased slow-wave electroencephalographic power density, peak frequency, and amplitude42-44; enhanced spindle activity phase-locked to slow oscillation up-states43-45; prolonged slow oscillation trains 43 ; and enhanced performance of a previously learned declarative task.43-45
Targeted memory reactivation
Targeted memory reactivation (TMR) is a noninvasive technique used to covertly cue neuroreplay via sensory cueing, typically auditory or olfactory. 46 Several studies have applied TMR during NonREM sleep in healthy young adults. Doing so has temporarily enhanced performance of a previously learned explicit simple motor sequence task,47-49 declarative task,50,51 and a complex non–dominant arm throwing task,52,53 but not a simple hippocampus-independent procedural memory task. 51 Repeated TMR during NonREM sleep while undergoing functional magnetic resonance imaging following performance of a declarative task led to enhanced performance and reactivated the hippocampus.50,51 Conversely, delivery of TMR while awake has led to decrements or no changes in performance of explicit simple motor sequence, 49 declarative,50,51 hippocampus-independent procedural memory 51 and complex non–dominant arm throwing tasks, 52 with mainly prefrontal cortical areas activated following declarative task practice. 50 Similarly, applying TMR during rapid eye movement did not affect performance of explicit simple motor sequence, 48 declarative, 51 or hippocampus-independent procedural memory tasks. 51
Limitations
Although a methodologically rigorous review process was used for this systematic review, a few limitations must be noted. First, it is possible that all relevant studies were not captured in the search. Second, most studies had relatively small sample sizes by study end, which was generally a result of poor CPAP compliance. Therefore, it is possible that many of the studies reported in this review were underpowered. Third, it was not possible to perform a meta-analysis of the included studies because of the small number of studies and the heterogeneity of stroke severity, treatment, and outcomes. Finally, despite the broad search criteria, the currently available evidence base meant that only studies with participants with stroke were included in this review.
Conclusion
This systematic review found that the only published nonpharmacological intervention to be provided during sleep to enhance physical rehabilitation outcomes in neurological populations has been CPAP in individuals with stroke and sleep apnea. The use of CPAP, when initiated during the acute and subacute period of recovery following stroke, tended to show short-term benefits to physical rehabilitation outcome measures relative to no CPAP use. However, little long-term benefit was shown in this regard and was often complicated by low CPAP compliance, and possibly a reduction in intensive rehabilitation, once patients transitioned to an outpatient setting. Studies that analyzed CPAP compliance related to physical rehabilitation outcome measures found a positive relationship between the two. Therefore, more evidence with higher CPAP compliance in inpatient rehabilitation settings is needed. Other future directions of research involve methods of noninvasive brain stimulation via the external application of electrical currents, magnetic pulses, and sensory cues during sleep to enhance motor performance during subsequent awake periods.
Supplemental Material
Sleep_systematic_review_Appendix_for_Stroke-formatted – Supplemental material for Systematic Review Investigating the Effects of Nonpharmacological Interventions During Sleep to Enhance Physical Rehabilitation Outcomes in People With Neurological Diagnoses
Supplemental material, Sleep_systematic_review_Appendix_for_Stroke-formatted for Systematic Review Investigating the Effects of Nonpharmacological Interventions During Sleep to Enhance Physical Rehabilitation Outcomes in People With Neurological Diagnoses by Brian P. Johnson, Andrea G. Shipper and Kelly P. Westlake in Neurorehabilitation and Neural Repair
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
