Abstract
Background. Paired exercise and cognitive training have the potential to enhance cognition by “priming” the brain and upregulating neurotrophins. Methods. Two-site randomized controlled trial. Fifty-two patients >6 months poststroke with concerns about cognitive impairment trained 50 to 70 minutes, 3× week for 10 weeks with 12-week follow-up. Participants were randomized to 1 of 2 physical interventions: Aerobic (>60% VO2peak using <10% body weight–supported treadmill) or Activity (range of movement and functional tasks). Exercise was paired with 1 of 2 cognitive interventions (computerized dual working memory training [COG] or control computer games [Games]). The primary outcome for the 4 groups (Aerobic + COG, Aerobic + Games, Activity + COG, and Activity + Games) was fluid intelligence measured using Raven’s Progressive Matrices Test administered at baseline, posttraining, and 3-month follow-up. Serum neurotrophins collected at one site (N = 30) included brain-derived neurotrophic factor (BDNF) at rest (BDNFresting) and after a graded exercise test (BDNFresponse) and insulin-like growth factor–1 at the same timepoints (IGF-1rest, IGF-1response). Results. At follow-up, fluid intelligence scores significantly improved compared to baseline in the Aerobic + COG and Activity + COG groups; however, only the Aerobic + COG group was significantly different (+47.8%) from control (Activity + Games −8.5%). Greater IGF-1response at baseline predicted 40% of the variance in cognitive improvement. There was no effect of the interventions on BDNFresting or BDNFresponse; nor was BDNF predictive of the outcome. Conclusions. Aerobic exercise combined with cognitive training improved fluid intelligence by almost 50% in patients >6 months poststroke. Participants with more robust improvements in cognition were able to upregulate higher levels of serum IGF-1 suggesting that this neurotrophin may be involved in behaviorally induced plasticity.
Keywords
Introduction
There is a limited time window of recovery poststroke that corresponds to biological events involving the upregulation of growth promoting factors, such as brain-derived neurotrophic factor (BDNF) and insulin-like growth factor–1 (IGF-1), and subsequent blocking of postischemic plasticity by growth-inhibiting factors. 1 Large cohort studies examining poststroke impairment confirm that motor recovery plateaus at about 12 weeks.2-4 The plateau of recovery following this critical period could also be related to the low intensity interventions provided. Several groups worldwide have reported the low physical demand of stroke rehabilitation.5-7
Research groups are examining whether pharmacological treatments or high levels of practice can “reopen” the window to extend stroke recovery.8,9 Aerobic exercise is one intervention that is underutilized during stroke rehabilitation despite the fact that patients have low fitness levels; often too low to comfortably carry out activities of daily living without excessive fatigue.10-12 Aerobic exercise has global direct effects on the brain by increasing levels of BDNF, IGF-1, and other neurotrophins,13-15 which could serve to make the brain more amenable to change, particularly when combined with other forms of rehabilitation.16-18 However, the majority of studies examining aerobic exercise and neurotrophin upregulation have employed animal models. 13 Recent research in humans casts doubt on whether exercise-induced circulating levels of neurotrophins BDNF and IGF-1 are linked to fitness, cognition, or hippocampal volume.19-21 Whether an aerobic intervention could enhance cognition among people with stroke is inconclusive.22,23
Despite the fact that the majority of people with stroke have cognitive impairment, 24 cognition is rarely a target of rehabilitation interventions. 25 Furthermore, patients poststroke with cognitive loss, particularly those with impaired attention and executive function, are unable to fully engage in sensorimotor training 26 and likely do not receive the full benefits of physical rehabilitation. 27 Impairments related to higher order thinking, especially fluid intelligence, are important predictors of longer lengths of stay and requirement for ongoing services after stroke. 28 Furthermore, in healthy individuals, higher fluid intelligence scores, measured using Raven’s Progressive Matrices Test (RPMT), are associated with an efficiently organized and clustered resting functional brain network. 29 Following stroke, such efficiency of activation suggests better recovery. 30 When patients are contacted 2 to 3 years after achieving “successful functional outcomes” at rehabilitation discharge, they continue to have problems with cognition and reintegration. 31 The fact that cognition and motor recovery are linked is typically disregarded in rehabilitation and interventions such as cognitive rehabilitation, speech therapy, and walking exercise are provided singly and temporally spaced from one another. 32
It is conceivable that aerobic exercise could serve to “prime” the brain to enhance relearning of subsequent tasks. 18 However, evidence of a priming effect is lacking in stroke. Quaney et al 33 demonstrated enhanced learning of a hand grip task on the nonhemiparetic side in patients poststroke who engaged in exercise. In contrast, Ploughman et al 34 reported that people in the chronic poststroke period failed to improve their performance on a working memory task after acute treadmill exercise. Liu-Ambrose and Eng 35 showed that among chronic stroke patients with mild cognitive impairment, mixed aerobic, resistance and balance training combined with enriching recreational and social activities for 6 months (n = 12) improved cognitive scores compared with a waitlist control (n = 16). Bo et al 36 showed that combining mixed physical exercise and cognitive training enhanced cognition more than the interventions provided on their own among people less than 6 months after stroke. In terms of nonstroke populations, a recent meta-analysis confirmed that among older adults with cognitive impairments, combining physical and cognitive training improved executive function; however, the benefits seemed to be less robust when compared with an active control group. 37 The temporal pairing of exercise and cognitive training to reopen the “window of recovery” beyond 6 months poststroke has yet to be examined.
We aimed to determine whether aerobic exercise combined with cognitive (dual-n-back) training would improve fluid intelligence compared with the interventions alone and when compared with an active control group. We also examined the relationships between cognitive improvement and change in serum levels of neurotrophins BDNF and IGF-1. We targeted people who were greater than 6 months poststroke; beyond the period when major recovery would occur. Furthermore, by instituting a 12-week follow-up, we aimed to determine whether combined intervention would result in sustained improvement in cognition. Since exercise, cognition, and depressive symptoms are linked, 38 we included measurement of depressive symptoms.
Methods
Design
The study was a block-randomized, single-blinded pilot trial conducted at 2 sites (NC1674790). The study was approved by the site ethics boards and all participants provided written informed consent. The second site began enrolling participants 6 months after the first and due to resources available, the RPMT was the only prespecified cognitive outcome that was measured at both sites. Group assignment to 1 of 4 intervention groups was conducted via opaque envelope randomization using randomly permuted blocks of 5 to 8 participants. Outcomes were assessed by an evaluator blinded to group allocation. With power set at 0.8 and 5% type 1 error rate, sample size was calculated using data from Mattay et al 39 who examined the effects of the stimulant amphetamine on RPMT scores. Our study was testing the principle that aerobic exercise could also stimulate the brain and provide synergistic benefit using the same outcome measure (RPMT score). 39 Mattay et al 39 reported mean RPMT scores of 78.5% in controls and 87.4% in drug-treated with 8.7 combined standard deviation. In a 2-group design, sample size was estimated at n = 20. To account for attrition and use of four groups with multiple comparisons, we tripled the estimate to n = 60.
Participants
Participants were recruited from a list of patients discharged from two rehabilitation hospitals between January 2014 and June 2016. Inclusion criteria: (a) age ≥18 years, (b) ischemic or hemorrhagic stroke >6 months, (c) self-reported cognitive problems related to stroke interfering with daily functioning, (d) ability to perform 2-step instruction, (e) ambulation with/without aid ≥10 m, (f) negative high-risk screening, 40 (g) agreement to refrain from aerobic exercise outside of trial interventions, and (h) time commitment to participate in the study. Exclusion criteria: (a) moderate/severe receptive aphasia, (b) life-threatening comorbidity or concomitant neurological or psychiatric illness, and (c) participation in other studies that could confound the outcomes of this trial. Given their active role throughout the trial, we were unable to blind the participants to their individual interventions. However, they were all informed that they were participating in a study to improve “thinking” with no information provided on the superiority of particular groups. Participants were not provided any specific instructions about ongoing physical or cognitive training following the 10-week intervention and activity was not monitored.
Intervention Protocols
Laboratory-supervised sessions were scheduled for 50 to 70 minutes per session, 3 sessions per week for 10 weeks. The intervention dosage was chosen based on consolidated evidence that aerobic training of sufficient intensity and duration (3 times per week for >8 weeks at moderate to vigorous intensity) could increase neurotrophins, improve aerobic fitness, and enhance cognition. 12 Each intervention group participated in one 20- to 30-minute physical intervention (Aerobic or Activity) and one 20- to 30-minute cognitive intervention (cognitive training [COG] or Games). Of the 4 groups (Aerobic + COG, Aerobic + Games, Activity + COG, and Activity + Games), the Activity + Games group was considered the active control. Rating of perceived exertion (RPE) and heart rate (HR) was monitored in each session to document physical and mental effort.
Aerobic Exercise (Aerobic)
Aerobic exercise was performed on a treadmill that had body weight support (BWS) and speed/incline was adjusted to maintain the target heart rate zone corresponding to 60% to 80% of peak oxygen uptake (VO2peak), which was determined by the symptom-limited graded exercise text (GXT) performed at baseline. Verbal instructions were provided to promote stepping symmetry and proper stepping kinematics, and BWS was reduced such that the harness provided safety rather than support.
Physical Activity (Activity)
Participants engaged in therapeutic activity including interventions designed to improve range of motion and comfort of the affected side (massage and active and passive range of motion exercises) and to relearn routine mobility tasks such as lying to sitting, rolling, sit to stand, and standing balance (functional task training). 41
Cognitive Training (COG)
Computerized dual-n-back training was performed with level of difficulty adapted to the individual’s performance. The working memory task involved monitoring a series of 2 concurrent stimuli (ie, visual locations on a screen and auditory single letters) and indicating whether the current stimuli match those presented n items back in the series. Value of n started at 1 and gradually increased, dependent on participant performance. 42
Cognitive Games (Games)
Participants sat at a workstation and played a non-adaptive computer-based game that involved strategically placing descending puzzle pieces.
Assessment Protocol
Participant characteristics (age, sex, education level, and comorbid conditions), stroke history (date of onset, type and location of lesion, severity using National Institutes of Health Stroke Scale [NIHSS] 43 ), cognitive status (Montreal Cognitive Assessment [MoCA] 44 ), physical function (self-selected walking speed, Chedoke-McMaster Stages of Recovery of Leg and Foot 45 ) were collected prior to randomization. Primary and secondary outcome measures were assessed at baseline, posttraining, and 3-month follow-up by the same independent assessor. All assessments for each participant were scheduled at a time corresponding to the intervention time slot for that individual. On testing day, participants were asked to eat at their regular times and take their medications as usual.
Primary Outcome
The primary outcome was fluid intelligence (also known as abstract reasoning) measured using the RPMT; a nonverbal and nonmathematical measure which tests the ability to solve new visual puzzles independently of previously acquired knowledge. 46 Participants were presented with geometric figures of progressing design complexity and asked to indicate the design that completed a missing piece. Repeated testing alternated between “odd” and “even” items to prevent familiarity with the geometric patterns and testing was preceded by 2 standard practice blocks to limit any learning effects. RPMT was scored out of 60 with higher score indicating greater fluid intelligence.
Secondary Outcomes
Depression score
The Depression subscale of the Hospital Anxiety and Depression Scale (HADS-D) was used, which consists of 7 items with higher scores indicating more severe symptoms. 47
Aerobic fitness
Symptom-limited maximal GXT’s were performed on either the treadmill (with 10%-15% BWS) or total body recumbent stepper (TBRS; NuStep LLC, Ann Arbor, MI, USA) according to best practice guidelines. 48 Expired gas was analyzed via open-circuit spirometry (SensorMedics 2900, Yorba Linda, CA, USA or Moxus, AEI Technologies, Pittsburgh, PA, USA) to measure oxygen consumption. Briefly, the treadmill protocol involved walking at a self-selected speed and 0% incline for 2 minutes, followed by a 2.5% increase in grade every 2 minutes until an incline of 10% was reached and, thereafter, a 0.05 m/s increase in speed every 2 minutes, until test termination. 49 The TBRS protocol was adapted from previous work in this population 50 and involved increments in workload (~20 W) every 2 minutes. Cardiovascular and ventilatory responses were monitored throughout the GXT and RPE was taken at the end of each 2-minute stage. All testing was terminated according to American College of Sports Medicine criteria. 51 Volitional exhaustion was considered the point at which the participant indicated that they could no longer proceed. The GXT was conducted by a trained physiotherapist or exercise physiologist and electrocardiogram activity was monitored by a physician when indicated, for example, among participants with atrial fibrillation.
Walking speed
Self-selected walking speed data were extracted from an instrumented gait analysis walkway (Protokinetics, Havertown, PA, USA). Walking speed at post and follow-up was measured only at one site.
Serum levels of neurotrophins
Other than resting BDNF, levels of BDNF and IGF-1 were measured from peripheral blood samples collected at one site. Since both resting and exercise-induced levels of neurotrophins could be important, samples were taken before the GXT (BDNFresting, IGF-1resting) and directly following the GXT (BDNFresponse and IGF-1response change scores) at pre, post and follow-up. A 10 mL blood sample was obtained from the antecubital vein using (no additive) Vacutainer tubes. The blood was left to clot at room temperature for 30 minutes, spun at 2200 rpm for 10 minutes, aliquoted into microcentrifuge tubes and placed at −80°C. Assays were completed in triplicate according to manufacturer’s instructions (Human/Mouse BDNF and Human IGF-1 DuoSet enzyme-linked immunosorbent assay, R&D systems).
Data Analysis
Primary and secondary behavioral outcomes and BDNF/IGF-1 levels were analyzed using repeated-measures 4 (groups) × 3 (pre, post, follow-up) analysis of variance (ANOVA) or log transformations/nonparametric equivalent if assumptions of normality were violated. Normality of data was tested using Shapiro-Wilk test. When data were not normally distributed, we log-transformed the data. Analysis was completed on the primary outcome with site, baseline cognition (Montreal Cognitive Assessment [MoCA]), and fitness (VO2peak) as covariates. We employed Greenhouse, Geisser, and Bonferroni corrections for multiple comparisons (significance set at P < .05). Baseline differences were evaluated using ANOVA. Linear regression was used where appropriate to examine relationships among variables. Effect sizes were expressed as partial eta squared (η 2 ) where η 2 of 0.01 was considered a small effect, 0.06 a moderate effect, and 0.14 a large effect. 52
Results
Participants
Ninety-three potential participants were screened of whom 60 were randomized with 15 participants per group (see Figure 1). Eight participants withdrew in the first 4 weeks. Their data were excluded from the analysis. Two participants were unable to be contacted at follow-up but were included in an intention-to-treat analysis with their post score brought forward using last-observation-carried-forward 53 ; leaving 34 participants in one site and 18 at the second. As part of a pilot test to examine feasibility, 4 participants at one site (2 from the Aerobic + COG group and 2 from the Aerobic + Games group) received the aerobic training following the cognitive training (randomly assigned). Their data were included in the analysis as per their assigned group. The remainder of the participants received the physical activity intervention directly before the cognitive. There were no adverse events such as falls or other injuries, chest pain or other cardiac related symptoms, or intolerance to the treatments. Participants were on average, 63 years of age and 3.4 years poststroke; 69% were male and 77% were diagnosed with ischemic stroke. Demographic data are shown in Table 1.
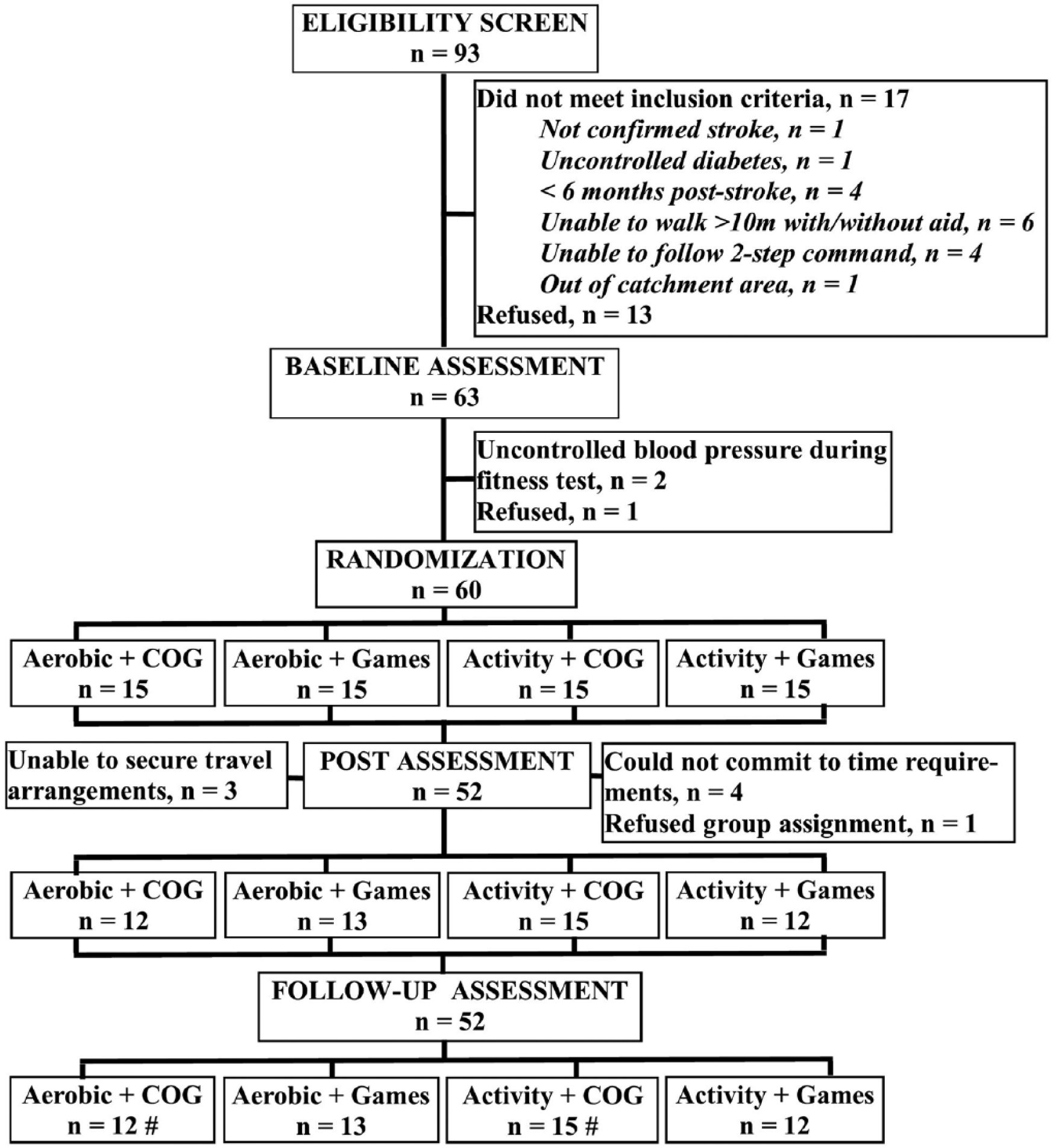
Consort diagram. #The last measure of one participant within the group was carried forward as an intention to treat analysis.
Participant Characteristics.
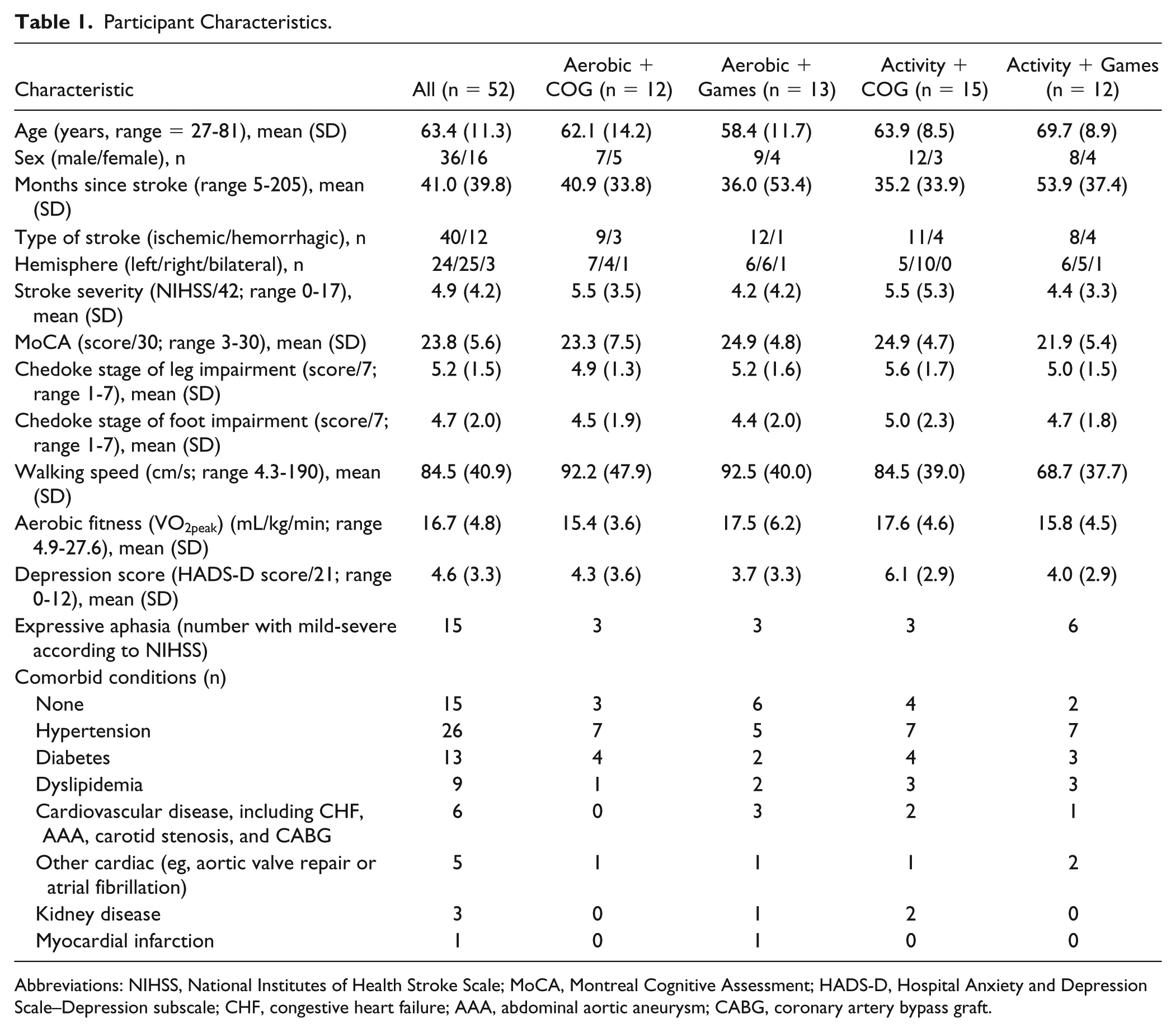
Abbreviations: NIHSS, National Institutes of Health Stroke Scale; MoCA, Montreal Cognitive Assessment; HADS-D, Hospital Anxiety and Depression Scale–Depression subscale; CHF, congestive heart failure; AAA, abdominal aortic aneurysm; CABG, coronary artery bypass graft.
Fifteen participants had no comorbid conditions, 18 participants had 1 comorbid condition, 8 had 2 conditions, 7 had 3 conditions, 3 participants had 4 conditions, and 1 participant had 7 comorbid conditions. Because of language difficulties, 33 of 52 participants were able to complete the MoCA. Fifteen of those (45%) scored below the cutoff for normal cognition (26/30). In terms of depressive symptoms, 3 participants reported moderate depression and 8 had mild depression based on the HADS cutoff values. 47 Medications are described in Table 2. On average, participants were prescribed 4.1 medications (SD = 4.3). At the extreme ranges, 4 participants took no medications while 1 person was prescribed 13 medications. There were no significant baseline differences between the groups in terms of age, sex, type of stroke, side of stroke, stroke severity, MoCA, Chedoke stage of leg and foot impairment, walking speed, aerobic fitness, depression score, presence of aphasia, or number of comorbid conditions (P values ranged from .089 [age] to .864 [Chedoke stage of foot impairment], data not shown).
Medications.
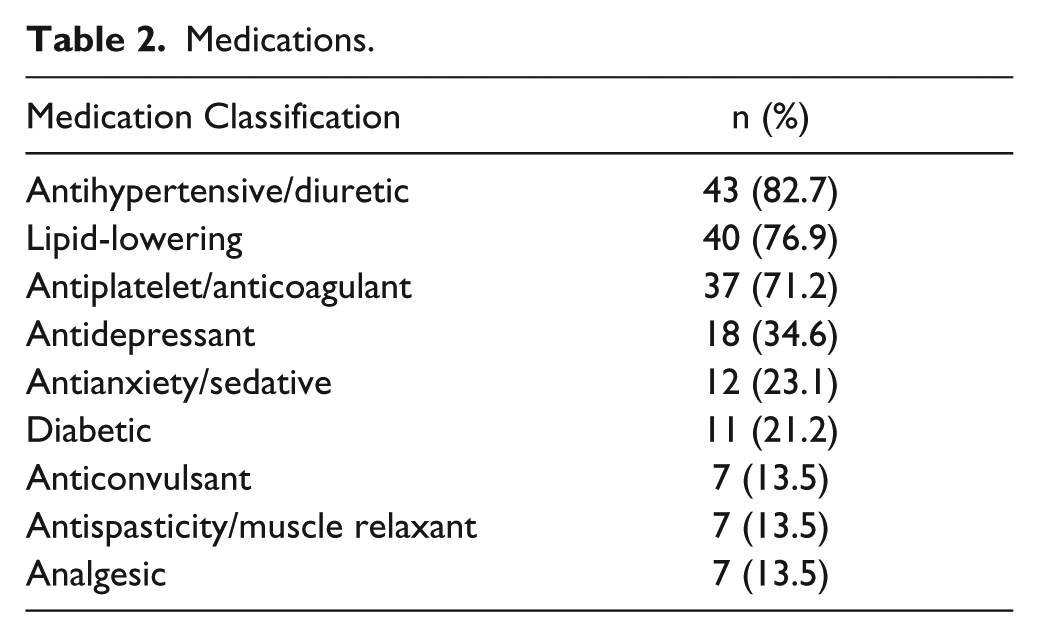
All participants reached volitional exhaustion during GXT’s performed on the treadmill (n = 35) and TBRS (n = 17). In terms of relative indicators of maximal exercise, maximal HRs achieved during the GXT were within 10% of age-predicted maximum (91.8%, SD = 12.6) and the average respiratory expiratory exchange ratio was more than 1.0 (1.10, SD = 0.14). As expected, significantly higher relative workloads were maintained during the aerobic exercise interventions compared with the activity groups, F(1, 141) = 21.12, P < .0001, and participants were able to achieve the target HR range during the aerobic exercise interventions measured at the first, fifth, and tenth weeks (77%, SD= 12; 80%, SD = 13; and 82%, SD = 12, of age-predicted maximal HR, respectively). Although not statistically different, relative HRs were higher in the Aerobic + COG compared with the Aerobic + Games group at week 1 (11%, SD= 7.07; P = .110), week 5 (13%, SD = 7.04; P = .151), and week 10 (11%, SD = 6.45; P = .063). Participants attended 28.8 (SD = 1.61) sessions with a range between 25 and 30. There were no significant differences between the groups in terms of attendance (P = .750).
Effects of Training on Fluid Intelligence
When controlling for site, baseline cognition, and baseline fitness, there was no significant effect of Time, F(2,88) = 0.83, P = .440, η2 = 0.020, but there was a significant interaction of Group × Time, F(6, 88) = 2.84, P = .015, η2 = 0.162, indicating a large effect; see Figure 2A). Three of the groups demonstrated nonsignificant trends of improvement in RPMT between pre- and postassessments (see Table 3 and Figure 2A). At 12-week follow-up, both the Aerobic + COG and Activity + COG groups made significant improvements from baseline. For the raw change in fluid intelligence from baseline to follow-up, only the performance by the Aerobic + COG group was significantly greater than that by the active control group: Activity + Games, F(3, 48) = 4.03, P = .012 (see Figure 2B). Significant differences between Aerobic + COG/Aerobic + Games (P = .045) and between Activity + COG/Activity + Games (P = .010) were lost with Bonferroni corrections (see Figure 2B). In terms of percentage change in performance, the Aerobic + COG group improved 47.8% (SD = 81.10), Activity + COG 20.7% (SD = 29.97), Aerobic + Games 7.0% (SD = 34.19) while the Activity + Games group declined (−8.5%, SD = 21.49); ANOVA, F(2, 48) = 3.18, P = .032. Data were normally distributed and there were no significant differences between groups at baseline (P = .801). Since 4 participants at 1 site received the aerobic training following the cognitive training, we recalculated the main results for RPMT with those 4 participants removed. The results were not substantially changed; no significant effect of Time, F(2, 80) = 0.52, P = .567, η2 = 0.014, but significant Group × Time interaction, F(6, 80) = 2.61, P = .025, η2 = 0.164.
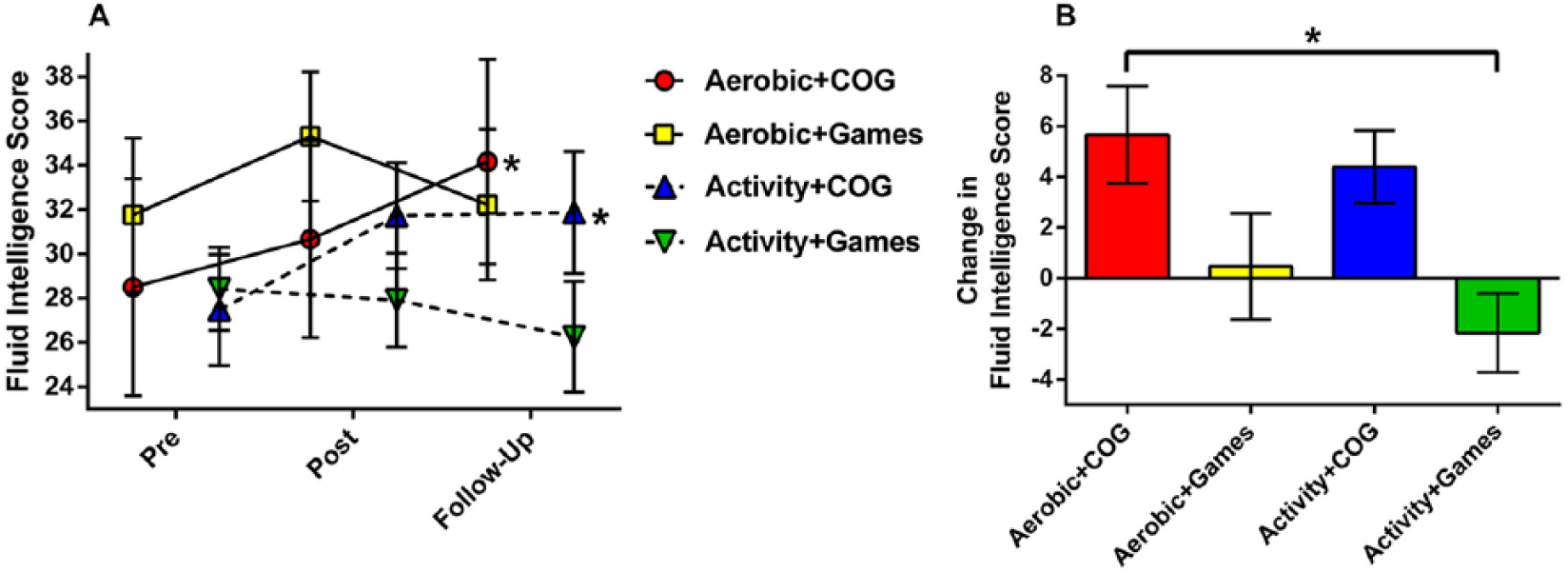
(A) Effects of interventions on Raven’s Progressive Matrices Test performance at Pre, Post (after 10 weeks), and Follow-up (12 weeks). *Significant difference from pre, P < .05. (B) Change in fluid intelligence score between Pre and Follow-up for each group. *P < .05. Error bars are standard error of the mean (SEM).
Fluid Intelligence, Depression Score, Aerobic Fitness, and Walking Speed.
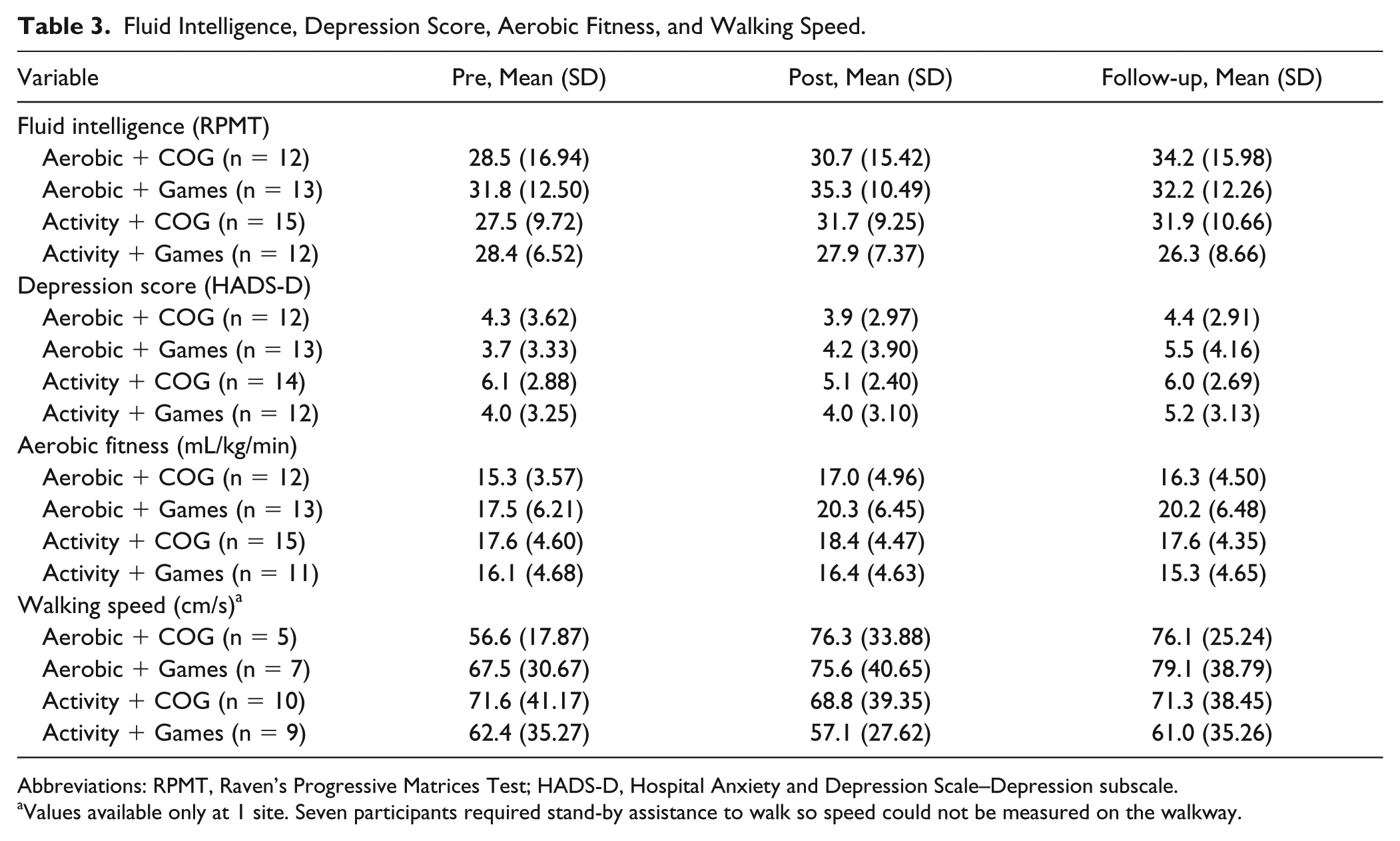
Abbreviations: RPMT, Raven’s Progressive Matrices Test; HADS-D, Hospital Anxiety and Depression Scale–Depression subscale.
Values available only at 1 site. Seven participants required stand-by assistance to walk so speed could not be measured on the walkway.
Effects of Training on Depression Score
Overall, the depression score increased over time with no significant differences between groups (Table 3) There was a significant effect of Time, F(2, 94) = 3.44, P = .036, η2 = 0.068, but no interaction of Group × Time, F(6, 94) = 0.79, P = .570, η2 = 0.048, indicating small to moderate effects. Data were normally distributed and there were no significant differences between groups at baseline (P = .195).
Effects of Training on Aerobic Fitness and Walking
In terms of aerobic fitness (Table 3), there was a significant effect of Time, F(2, 94) = 8.04, P = .001, η2 = 0.146, and significant Group × Time interaction, F(6, 94) = 2.51, P = .032, η2 = 0.138, indicating large effects. The Aerobic + Games group was the only group to make significant improvement in VO2peak, which was maintained at follow-up compared with pre (from 17.46 to 20.21 mL/kg/min at follow-up; see Figure 3A). The Aerobic + COG made improvements from pre to post (P = .023) but significance was reduced to P = .068 with Bonferroni correction. Notably, both Activity groups demonstrated a trend toward declines in fitness below baseline values. Data were normally distributed and there were no significant differences between groups at baseline (P = .455).
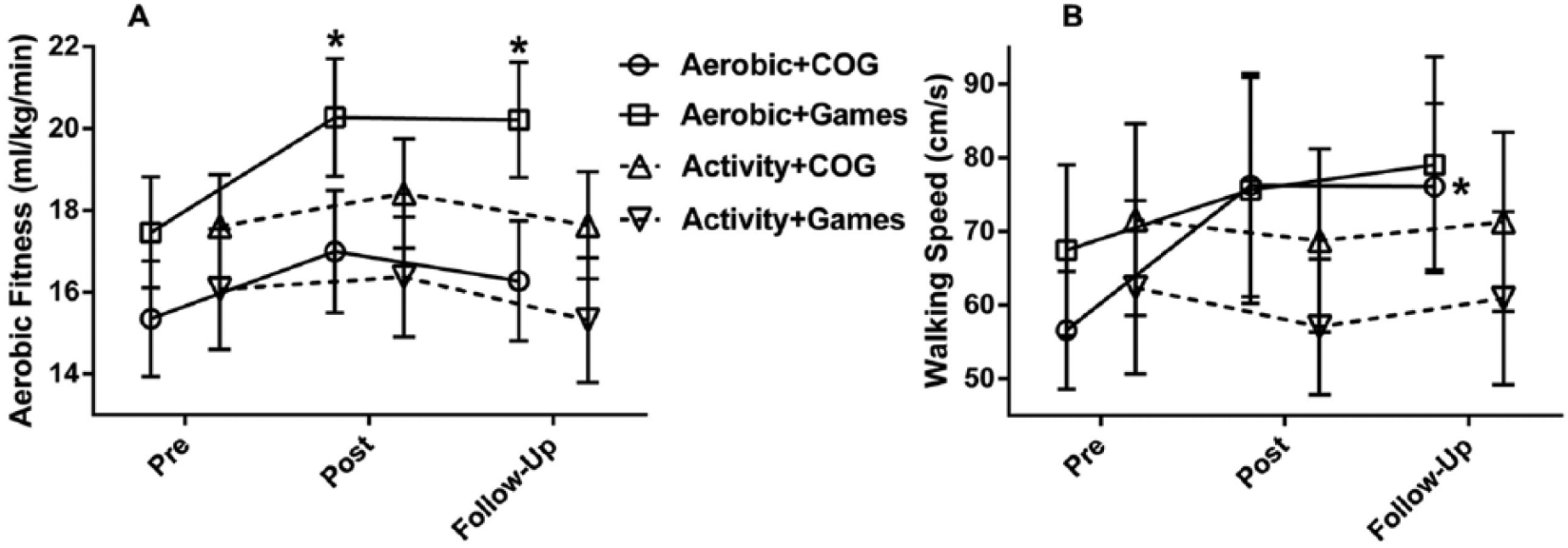
Effects of training on aerobic fitness and walking. (A) Both Aerobic groups improved from Pre values but only the Aerobic + Games significantly so. *P < .05 compared with Pre. (B) The Aerobic + COG and Aerobic + Games groups improved self-selected walking speed but only the Aerobic + COG group significantly so before Bonferroni correction. *P < .05 compared with Pre. Error bars are standard error of the mean (SEM).
For walking speed (Table 3), there was a significant effect of Time, F(2, 54) = 4.42, P = .033, η2 = 0.141, and significant Group × Time interaction, F(6, 54) = 2.80, P = .042, η2 = 0.237, indicating large effects. Although both Aerobic + COG and Aerobic + Games groups increased self-selected walking speed, the improvement was significant before Bonferroni correction only for the Aerobic + COG group (Post compared with Pre P = .074; Follow-up compared with Pre P = .023; see Figure 3B). Data were normally distributed and there were no significant differences between groups at baseline (P = .557).
The Effects of Training on Circulating IGF-1 and BDNF Levels
IGF-1resting changed during the trial (Table 4) but was not significantly different across groups—significant effect of Time, F(2, 46) = 35.11, P < .00001, η2 = 0.604; no Group × Time interaction F(6, 46) = 1.29, P = .279, η2 = 0.144, indicating large effects. Data were normally distributed and there were no significant differences between groups at baseline for IGF-1resting (P = .101). Combining the groups, serum IGF-1resting at pre (3.52, SD = 2.52 ng/mL) was significantly lower compared with post levels (7.05 SD = 4.24 ng/mL; P <.001) and dropped to levels lower than pre at follow-up (1.52 SD = 2.58 ng/mL; P < .00001; Figure 4A). IGF-1response was the only variable not normally distributed and therefore was log transformed. There were no significant differences between groups at baseline (P = .245). IGF-1response increased over the trial (see Table 4) but was not significantly different across groups: significant effect of Time, F(2, 38) = 6.22, P = .008, η2 = 0.246; no Group × Time interaction, F(6, 38) = 1.41, P = .249, η2 = 0.181, indicating large effects. Combining groups, IGF-1response, expressed as a change in IGF-1 following the GXT, did not change significantly from pre to post (pre −0.98, SD = 1.76 ng/mL; post 0.72, SD = 4.48 ng/mL; P = 1.00; Figure 4B). Levels at follow-up (3.39, SD = 3.88 ng/mL) were significantly higher than those at pre (P = .002) and post (P = .024).
Serum Levels of Brain-Derived Neurotrophic Factor (BDNF) and Insulin-Like Growth Factor–1 (IGF-1).
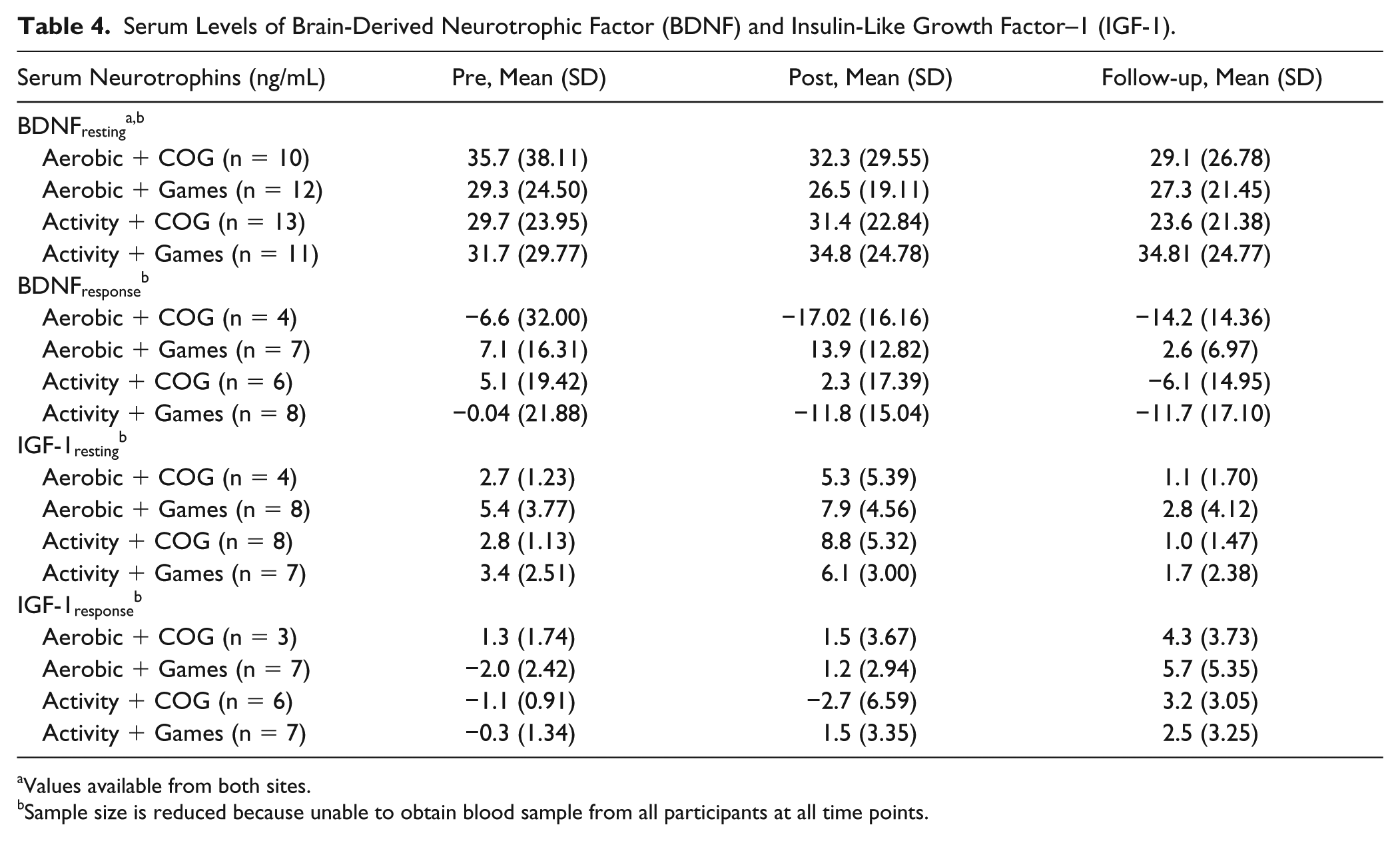
Values available from both sites.
Sample size is reduced because unable to obtain blood sample from all participants at all time points.
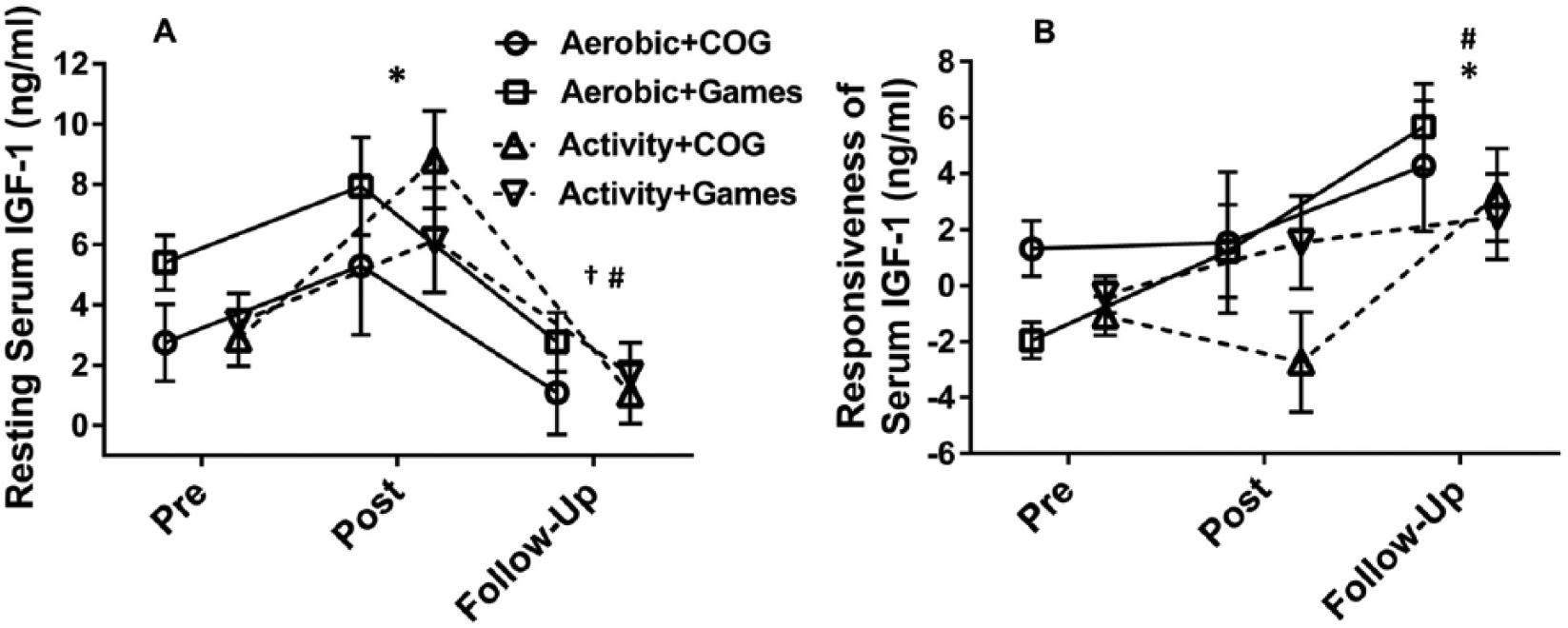
Effects of interventions on serum insulin-like growth factor–1 (IGF-1). (A) Resting IGF-1. *Significantly different from Pre P < .001, #significantly different from Post P < .0001, †significantly different from Pre P < .0001; groups combined. (B) IGF-1 acute response to exercise significantly different from PRE. *P = .002, #significantly different from Post P = .024; groups combined. Error bars are standard error of the mean (SEM).
BDNFresting did not change over the trial (Table 4). Data were normally distributed and there were no significant differences between groups at baseline (P = .922). There was no effect of Time, F(2, 46) = 0.64, P = .514, η2 = 0.015 or Group × Time interaction, F(6, 46) = 0.36, P = .786, η 2 = 0.035; indicating small effects. BDNFresponse was also normally distributed and not altered, Time, F(2, 42) = 1.00, P = .371, η2 = 0.045; Group × Time, F(6, 42) = 0.36, P = .884, η2 = 0.049; indicating small effects. There were no significant differences in BDNFresponse between groups at baseline (P = .548).
Factors Predicting Intervention Response
Six baseline variables—aerobic fitness (VO2peak), cognition (RPMT), and serum levels of BDNF (BDNFresting, BDNFresponse) and IGF-1(IGF-1resting, IGF-1response)—were examined to determine their relationships with improvement in fluid intelligence (change from pre to follow-up). We first examined correlations between the 6 baseline variables and the change in fluid intelligence. Two variables, higher baseline aerobic fitness (VO2peak) and greater acute increase in IGF-1 with exercise (IGF-1response), were correlated with improvement in fluid intelligence (R = 0.40, P = .038 and R = 0.63, P = .016, respectively). To build a predictive model these significant variables were entered into a stepwise regression model with change in fluid intelligence as the outcome (see Figure 5A). Only IGF-1response was retained in the model (see Figure 5A). The model explained 40% of the variance in change in cognitive scores in the COG training groups (Aerobic + COG and Activity + COG; F = 7.94, R2 = 0.40, P = .016) but little variance in the scores of groups receiving cognitive games (F = 2.26, R2 = 0.08, P = .155). Since others have reported that greater increases in aerobic fitness were related to improvements in cognition, 54 we examined the correlation between these 2 variables (Post-Pre) in the 4 groups combined. There was no relationship between change in fitness and improvement in cognition (R = 0.07, P = .623; see Figure 5B).
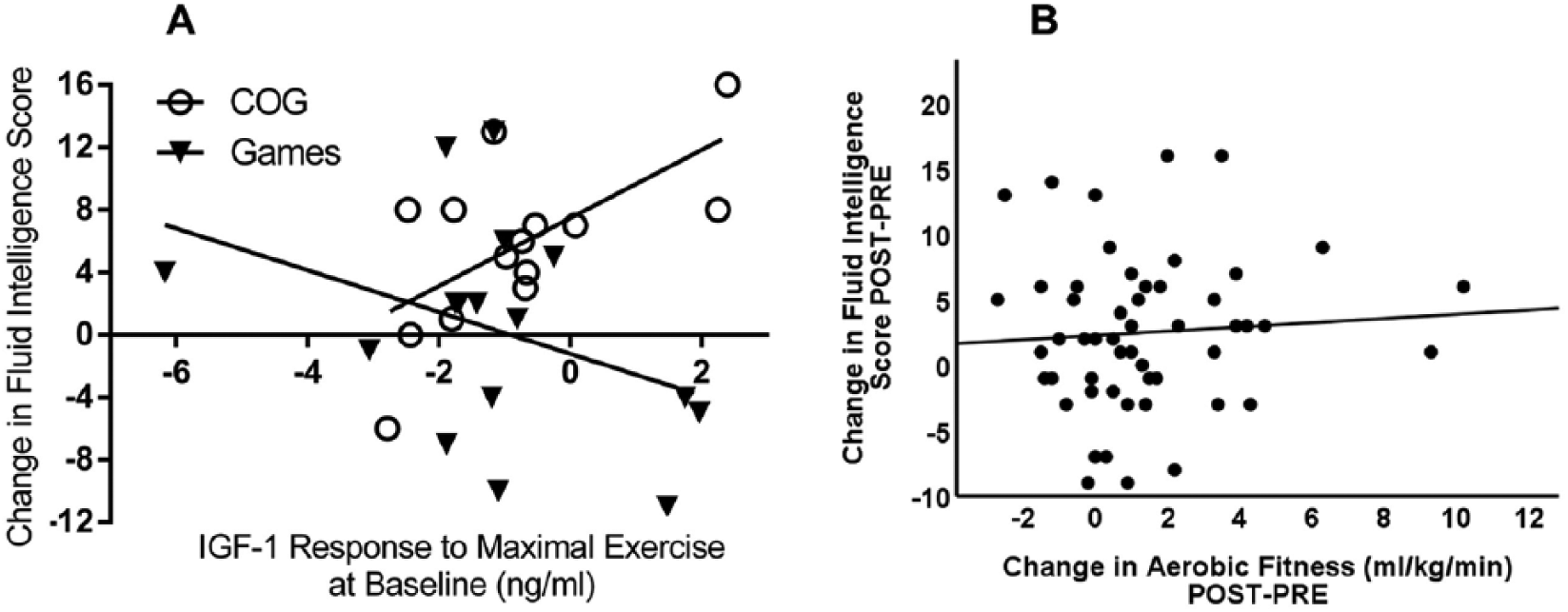
Predictors of cognitive improvement. (A) IGF-1response at baseline strongly and significantly predicted improvement in cognition in the cognitive training groups (Aerobic + COG and Activity + COG; open circles). IGF-1response was not associated with change in fluid intelligence in the groups receiving cognitive games (black inverted triangles). (B) There was no relationship between change in fitness and improvement in cognition. IGF-1, insulin-like growth factor–1.
Discussion
The prevalence of cognitive impairment measured using sensitive assessments has been reported to be as high as 83% in acute stroke units and 71% at 3-month follow-up. 24 We undertook this study to determine whether 10 weeks of combined aerobic exercise and cognitive training would improve fluid intelligence among people with chronic stroke who had cognitive impairment. We compared the combined training to the interventions provided on their own (with other control activities in order to match for treatment duration) and an active control group (Activity + Games). We found that Aerobic + COG and Activity + COG made significant improvements in fluid intelligence although the Aerobic + COG group was the only group in which the improvement was significantly greater than the active control group (Activity + Games). Furthermore, participants in the Aerobic + COG group also experienced gains in aerobic fitness and walking (although some significant effects were lost after Bonferroni corrections), suggesting that combined interventions have the potential to improve multiple outcomes at the same time. 12 Aerobic exercise without being combined with cognitive training (Aerobic + Games) did not improve fluid intelligence. Finally, levels of serum IGF-1 in response to acute exercise at baseline (IGF-1response) significantly predicted improvements in fluid intelligence in the groups receiving cognitive training. The dual-n-back training program we employed was an adaptive working memory task requiring attentional maintenance and updating and has been shown to predict performance on RPMT 55 as well as involve a widespread bilateral frontal-parietal network. 56 Our results support that dual-n-back training aimed at fluid intelligence likely promoted neuroplasticity and could be an important cognitive training technique in stroke.
Our findings as well as those of Tang et al 22 and Bo et al 36 demonstrated that aerobic/physical exercise did not significantly enhance fluid intelligence poststroke when not temporally linked with cognitive training (ie, Aerobic + Games group in our study). Others, however, have shown that 8 weeks of aerobic exercise training during the chronic period poststroke improved other cognitive functions, such as attention, 57 visual spatial ability, 57 and learning. 33 Marzolini et al 54 reported that 6 months of combined aerobic and resistance training resulted in improvement in MoCA score, specifically in the visuospatial/executive and attention/concentration domains. Furthermore, Moore et al, 58 in a randomized controlled trial involving 40 individuals greater than 6 months poststroke, demonstrated that 19 weeks of fitness and mobility training improved executive function. Quaney et al 33 also showed that aerobic exercise improved procedural motor learning. It is important to appreciate that the participants in the aforementioned studies were more mildly affected (ie, walking speeds of about 120 cm/s, 58 normal MoCA scores 33 ) than our cohort. Moore et al 58 also reported a significant improvement in mood, which could confound cognitive testing. 59 Furthermore, most studies examining the effects of exercise on cognitive function did not include follow-up, precluding determination of the sustainability of effects.22,57,58,60,61 In fact, Quaney et al 33 reported that benefits declined by 8-week follow-up. We show that with cognitive training (Activity + COG), and even more robustly with combined aerobic and cognitive training (Aerobic + COG), people poststroke showed large and significant gains in fluid intelligence measured 12 weeks after the intervention ceased. This finding suggests that the combined interventions are potent and are able to overcome the recovery plateau even when there was no benefit in depressive mood.
Neurotrophins are considered to be potential biomarkers in stroke recovery. 13 BDNF and IGF-1 promote recovery and repair processes such as angiogenesis, neurogenesis, synaptogenesis, and long-term potentiation. 13 In humans, serum or plasma levels of BDNF and IGF-1 are used as proxies of brain levels of these growth factors. 23 We found that resting BDNF and exercise-induced BDNF did not change significantly during the trial, nor did BDNF predict improvements in cognition; however, we did not test for BDNF genotype nor did we require fasting before blood collection. As well, the serum BDNF values were highly variable making interpretation difficult.
IGF-1 is primarily produced in the liver and crosses the blood-brain barrier following exercise. 62 We showed that higher exercise-induced levels of serum IGF-1 at baseline predicted 40% of the variability in improvement of fluid intelligence. It is noteworthy that exercise-induced levels, but not resting levels of IGF-1, were predictive of this response. Furthermore, the ability to upregulate circulating levels of IGF-1 with exercise increased over time in all the groups. Serum IGF-1 concentrations have been shown to correlate with enhanced brain vascular activation 63 and with better functional outcomes after stroke. 64 The transport of circulating IGF-1 into the brain is enhanced by an activity-driven process triggered by the release of glutamate in active brain regions that promotes vasodilation and increases movement of this growth factor across the blood-brain barrier. 65 Neuronal release of glutamate leads to calcium fluxes in astrocytes that promote the release of diffusible mediators such as nitric oxide and arachidonic acid derivatives, which promote vasodilation. 66 These messengers also stimulate matrix metalloproteinase-9 activity resulting in cleavage of IGF binding protein-3 and release of IGF-1. 65 The combined actions of these 2 events results in the increased local availability of free serum IGF-1. 67 The elevation of serum IGF-1 concentrations by exercise coupled with the stimulation of activity-dependent entrance of IGF-1 from the circulation into the brain by cognitive training may contribute to the benefits of combining aerobic and cognitive interventions on fluid intelligence.
Several groups have confirmed that participants poststroke with the greatest gains in cognition also had the largest improvements in fitness54,68 and that the 2 outcomes are linked. Marzolini et al 54 showed that improvements in attention and concentration after 6 months of aerobic and resistance training were correlated with improvement in fitness. Kluding et al 68 also reported a relationship between improved aerobic fitness with 12 weeks of aerobic and resistance exercise and better performance on the Flanker task. 68 We found no such relationship between fitness and cognitive gains. Our participants were similar in terms of severity and time since stroke; however, our intervention included cognitive training whereas the aforementioned studies did not, potentially lessening the impact of fitness on outcome.54,68
Intensity and structure of cognitive training may be important in order to maximize cognitive gains. Stroke best practices recommend taking either a compensatory or restorative approach to cognitive rehabilitation depending on patient-specific goals. 69 Within the guidelines, therapist-supervised computerized cognitive training was advised but with no specific intensity or duration recommended. Computerized cognitive training is an attractive method to enhance cognition considering the pleasurable gaming format and ability to log on at home with no requirement for health care personnel. Whether these types of interventions actually improve cognition is a topic under scrutiny. In this study, we showed that 15 hours of computerized dual-n-back working memory training (with control activity, Activity + COG) translated into a 20.7% (SD = 29.97) improvement in fluid intelligence. Although instruction and coaching were provided for the first 2 weeks, participants completed the training in a laboratory environment with minimal degree of monitoring thereafter. van de Ven et al 70 examined the effects of 30 hours of home-based computerized training and found that people poststroke showed a trend in improving cognitive outcomes, albeit not significant, compared with a waitlist control group. Bo et al 36 showed that 12 weeks of computer-based cognitive training improved some but not all cognitive domains among people less than 6 months poststroke. In another trial, people in the chronic stage of stroke with self-perceived cognitive impairment who participated in 8 weeks of cognitive training with a commercial Lumosity showed short-term improvements in reaction time but no changes in objective and subjective measures of cognition. 61 Rather than targeting working memory specifically, Lumosity provided practice in a broad range of cognitive domains such that the working memory aspects of the intervention were diluted compared with our focused laboratory intervention. Furthermore, only half of the participants successfully completed the computerized cognitive training at the recommended dose. 61 As with other forms of exercise, the intensity and type of cognitive training are likely factors that mediate outcome so monitoring would be required to ensure compliance and performance. 71
Even though we showed robust and clinically meaningful gains in cognition, there were some limitations to the study. First of all, the study took place at 2 sites but data on serum neurotrophins and walking speed were only available at 1 site, reducing an already limited sample size. Second, we did not assess presence of the BDNF val66met polymorphism, which limited interpretation of serum BDNF values. 72 Carriers (at least 1 Met allele) of the BDNF polymorphism have shown decreased brain activation during movement of the hemiparetic side 73 and seem to benefit less from brain stimulation techniques aimed at improving limb recovery. 74 Future research should include genetic analysis. Furthermore, the relatively high levels of neurological impairment observed among participants in the current study may have precluded assessment of true physiological maximum during the GXT, which could have influenced our ability to detect an association between change in aerobic fitness and fluid intelligence. Although we randomly assigned participants to groups, there were (nonsignificant) baseline differences between them (such as age and walking speed), which may have accounted for some aerobic exercise response differences between the aerobic groups. Last, variability was introduced in control group activities because activity selection was based on the participant’s level of impairment.
Conclusion
Cognitive impairment is common after stroke 26 and interferes with successful sensorimotor rehabilitation. 27 In our trial, aerobic exercise combined with cognitive training improved fluid intelligence by almost 50% in patients >6 months poststroke who were presumed to have reached their recovery plateau. Participants in the Aerobic + COG group also experienced gains in aerobic fitness and walking, suggesting that combined interventions have the potential to address several recovery domains. 12 Aerobic exercise without being combined with cognitive training (Aerobic + Games) did not improve fluid intelligence. Levels of serum IGF-1 in response to acute exercise at baseline (IGF-1response) significantly predicted improvements in fluid intelligence among the groups receiving cognitive training, supporting its usefulness as a recovery biomarker in stroke.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by grants from the Heart and Stroke Foundation Canadian Partnership for Stroke Recovery (MP, JM), the Canada Research Chairs program (MP), the Canada Foundation for Innovation (MP), The Newfoundland and Labrador Research and Development Corporation (MP), the Nova Scotia Health Research Foundation (MM-L), Capital Health Research Foundation, Halifax Nova Scotia (MM-L) and the Dalhousie University Faculty of Health Professions Innovation Fund (MM-L).
