Abstract
Background. Brain-machine interfaces (BMIs) have been recently proposed as a new tool to induce functional recovery in stroke patients. Objective. Here we evaluated long-term effects of BMI training and physiotherapy in motor function of severely paralyzed chronic stroke patients 6 months after intervention. Methods. A total of 30 chronic stroke patients with severe hand paresis from our previous study were invited, and 28 underwent follow-up assessments. BMI training included voluntary desynchronization of ipsilesional EEG-sensorimotor rhythms triggering paretic upper-limb movements via robotic orthoses (experimental group, n = 16) or random orthoses movements (sham group, n = 12). Both groups received identical physiotherapy following BMI sessions and a home-based training program after intervention. Upper-limb motor assessment scores, electromyography (EMG), and functional magnetic resonance imaging (fMRI) were assessed before (Pre), immediately after (Post1), and 6 months after intervention (Post2). Results. The experimental group presented with upper-limb Fugl-Meyer assessment (cFMA) scores significantly higher in Post2 (13.44 ± 1.96) as compared with the Pre session (11.16 ± 1.73; P = .015) and no significant changes between Post1 and Post2 sessions. The Sham group showed no significant changes on cFMA scores. Ashworth scores and EMG activity in both groups increased from Post1 to Post2. Moreover, fMRI-BOLD laterality index showed no significant difference from Pre or Post1 to Post2 sessions. Conclusions. BMI-based rehabilitation promotes long-lasting improvements in motor function of chronic stroke patients with severe paresis and represents a promising strategy in severe stroke neurorehabilitation.
Keywords
Introduction
Motor impairment after stroke is the leading cause of long-term disability in the adult population. 1 Approximately 30% of stroke survivors experience severe motor impairment and need assistance for daily living activities.1,2 It was reported that current rehabilitation strategies or bilateral arm training improve motor function in chronic stroke patients with limited paresis,2-5 but patients with severe upper-limb paresis are not eligible for these techniques because they are unable to perform the requested movements.
For those patients with severe paresis, we demonstrated for the first time in a controlled randomized double-blind study 6 the clinical efficacy of brain-machine interface (BMI) coupled with physiotherapy training to promote upper-limb motor recovery. Subsequently, these findings have been confirmed by other studies.7-9 We used patients’ voluntary modulation of oscillatory brain activity associated with motor intention (sensorimotor rhythm [SMR]10,11) to trigger a BMI-controlled orthotic device to move the paretic limb. This procedure created a contingency between the neural correlate of motor intention with sensory (visual and proprioceptive) feedback of the intended movement. We compared improvements in motor function between an experimental group receiving contingent BMI training and a control group receiving sham BMI training over 20 training days. In both groups, BMI sessions were followed by identical behaviorally oriented physiotherapy. Our previous results suggest that in chronic stroke patients with severe paresis, a close associative connection between the neural correlate of motor intention and the feedback of the intended movement (established via a BMI) elicits (1) superior associative learning of voluntary modulation of SMR, as demonstrated before in healthy participants, 12 and (2) motor impairment reduction after 4 weeks of intervention, demonstrating previous theoretical predictions13-15 and indicating that BMI has a beneficial effect on motor rehabilitation of severely paretic chronic stroke patients. Our results indicate that learning to control oscillatory brain activity through a BMI approach and the repetitive contingency between motor intention and sensory feedback constitute necessary therapeutic ingredients for motor recovery and that behavioral physiotherapy allows generalization of relearned motor skills to meaningful real-life activities. 6
Previous studies evaluating long-term effects of rehabilitative interventions for stroke motor recovery reported contradictory findings regarding maintenance of improvements in motor outcomes. For example, whereas some studies indicated long-lasting improvements in motor function after distinct rehabilitative interventions,16,17 other studies reported improvements in motor function that were not preserved 6 months after intervention.18,19 Here, we analyzed the long-term effects (6 months after intervention) of the BMI-based rehabilitative intervention. 6 In analogy to previous stroke motor rehabilitation studies,16,17 we hypothesized that if (4 weeks) BMI-based training promotes significant and stable motor functional gain, it should be maintained 6 months after intervention. The data presented in this manuscript were acquired between 2007 and 2011.
Methods
This is a follow-up study of the clinical and neurophysiological results obtained in the previous interventional study. 6 Because of personnel, organizational, and time constraints, the publication of these data and results has been delayed.
Patients
All patients who completed the previous interventional double-blinded controlled study were invited for the follow-up session, which ended in December 2011. Inclusion criteria for acceptance in the interventional study included the following: (1) paralysis of one hand, with no active finger extension; (2) time since stroke of at least 10 months; (3) age between 18 and 80 years; (4) no psychiatric or neurological condition other than stroke; (5) no cerebellar lesion or bilateral motor deficit; (6) no pregnancy; (7) no claustrophobia; (8) no epilepsy or medication for epilepsy during the past 6 months; (9) eligibility to undergo magnetic resonance imaging (MRI); and (10) ability to understand and follow instructions.
Patients were recruited via public information. From 504 potentially eligible patients, 32 were allocated to the intervention (see CONSORT chart in the supplementary material). Patients were matched for age, gender, paretic side, and motor impairment scores at the time of inclusion and were randomly assigned to experimental or control groups. Group assignment was blinded for all participants and for the scientific-clinical personnel; none of the patients or therapists was able to identify group assignment reflected in the results of placebo and motor function scales below. Two of the patients from the control group did not receive correct allocated intervention, and 2 of the 30 patients who received intervention had to be excluded from the follow-up analysis because of (1) incidence of a second stroke and (2) economic and health-related issues (see Figure 1). Both patients were from the control group and presented with mixed lesions including cortical and subcortical structures (experimental group: n = 16 [7 female], age at study admittance = 49.3 ± 12.5 years; sham group: n = 12 [5 female], age at study admittance = 50.3 ± 12.2 years).
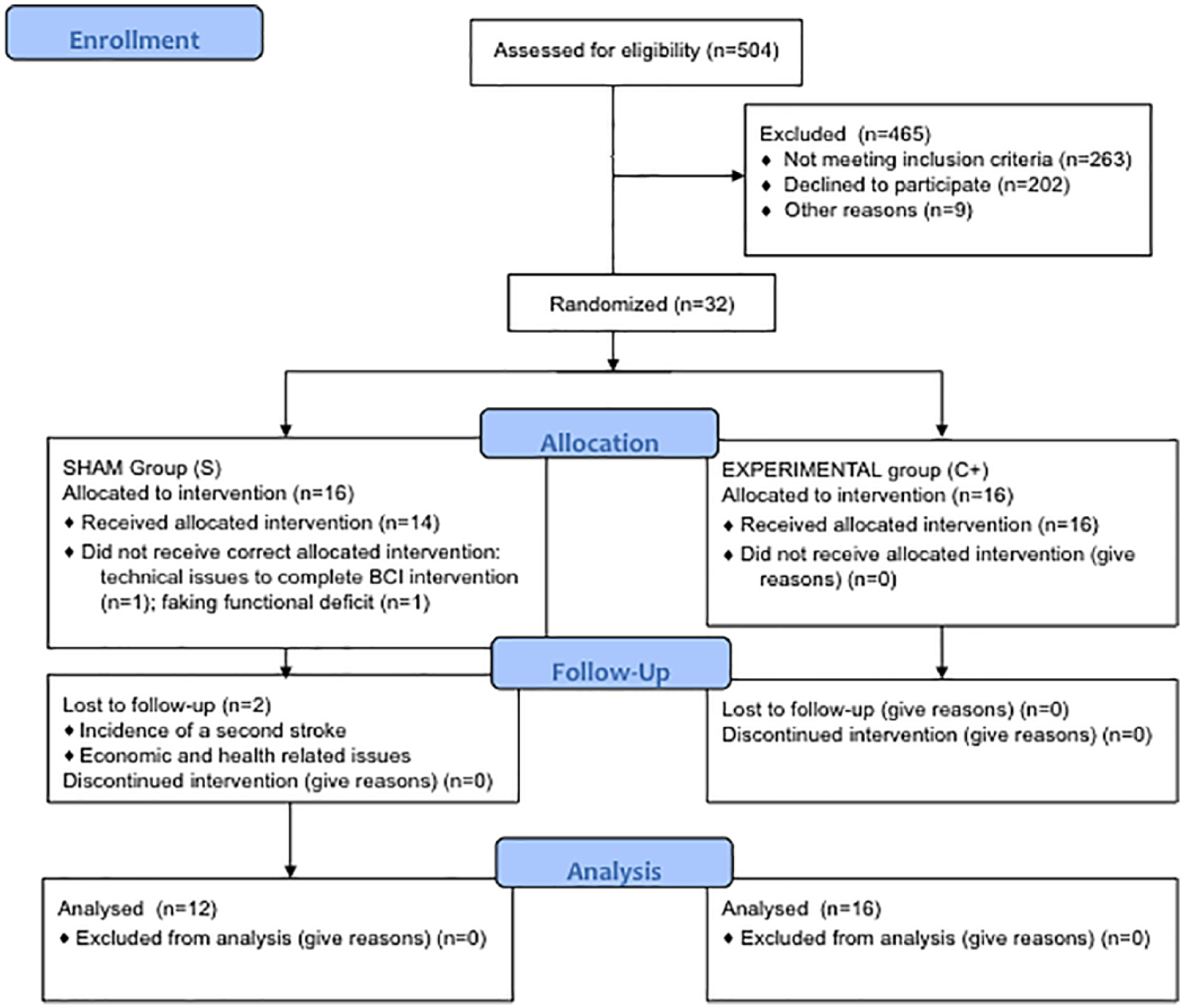
CONSORT flow diagram: study enrollment diagram. In all, 504 patients were screened to be eligible for the study, and 465 were excluded; 32 underwent intervention and were randomly assigned to 2 groups depending on the brain-machine interface feedback received: (1) experimental group (brain activity was contingently and concurrently linked with orthoses movements) and (2) control or sham feedback group (orthoses movements were random and not associated with brain activity). In the sham group, 2 patients did not receive correct allocated intervention. Patients were invited for follow-up measurements 6 months after the intervention. Two patients from the control group dropped out and did not come back for the final measurements.
Study Design
Intervention involved daily training for 4 weeks (excluding weekends), including 1 hour of BMI training followed by 1 hour of behavioral physiotherapy. There was no difference in time of training (BMI + Physiotherapy) between groups (unpaired t-test for BMI runs between experimental and Sham groups resulted in no significant differences, P = .42; experimental (mean ± SE), 281 ± 10.03; sham, 290.29 ± 5.13 runs).
In the experimental group, patients’ successful control of ipsilesional SMR desynchronization was translated concurrently into movement of the orthosis attached to the paretic limb, whereas in the Sham group, movements of the orthosis occurred randomly—that is, unrelated to patients’ SMR desynchronization (for methodological details, please see Ramos-Murguialday et al 6 ). During BMI training, patients were instructed to try to move their paretic upper limb. Two movements were practiced, each of them associated with a specific robotic orthosis: (1) open and close the fingers or (2) move the paretic upper limb forward and backward. Only 1 type of movement was practiced on any given day. The level of paresis determined the kind of movement to be performed during BMI training, but all patients performed the movement of opening and closing the fingers. The number of sessions with hand or arm movements was balanced between groups (more details in Ramos-Murguialday et al 6 ).
After each intervention session, physiotherapists taught and demonstrated to the patients several exercises comprising functional movements of their affected limb acquired during BMI training and mainly daily live activities for home exercise (eg, to use a toothpaste tube 20 ). These exercises were also provided to the patients in a handbook. 21 The primary outcome measure used was the combined hand and arm scores (motor part) from the modified upper-limb Fugl-Meyer Assessment (cFMA; mean ± SD, maximal score is 54 points [more details in Ramos-Murguialday et al 6 ]), which was 14.75 ± 2.71 for all patients who underwent BMI and behavioral physiotherapy intervention.
The study was conducted at the University of Tübingen, Germany. Informed consent was obtained from all patients involved. The study was approved by the ethics committee of the Faculty of Medicine of the University of Tübingen, Germany.
Assessments
To evaluate the long-term BMI-based rehabilitation effects on motor function, the identical comprehensive battery of assessment instruments given twice before (8 weeks and 1 day before the first training session) and once immediately after treatment (Post1), described in our previous study 6 and reported below, was repeated 6 months after the 4-week intervention period (Post2). Scores from the 2 preintervention assessments were averaged (Pre) to reduce variability of the data 22 and increase reliability of preintervention assessment.
Primary Behavioral Outcome Measure: Combined Hand and Arm Scores (Motor Part) From the Modified cFMA
We used the combined hand and arm scores (motor part) from the modified cFMA of our previous study (with a maximum score of 54) as the primary behavioral outcome measure 23 because they are related to the 2 body parts trained during the BMI (hand and arm), reflect motor recovery, and measure motor impairment that may limit but is unrelated to task accomplishment (eg, joint motion). We excluded cFMA scores related to (1) coordination and speed and (2) reflexes because (1) before intervention, patients in this study could not touch their noses with the index finger fully extended and had no remaining finger extension (no active finger extension was an inclusion criteria) and (2) reflex scores add uncertainty to the measurement. 24
Secondary Outcome Measures: GAS, MAL, and Ashworth Scores
Goal Attainment Scale (GAS), 25 Motor Activity Log (MAL), 26 and Ashworth Scale were used as secondary behavioral measures. It should be mentioned that during the treatment phase, placebo questionnaires demonstrated no difference between the 2 groups in placebo responding. 6
Assessments Associated With the Primary Behavioral Outcome Measure
We used electromyography (EMG) to document muscle activity and innervation 27 and blood-oxygen level–dependent (BOLD) signal from functional MRI (fMRI) 28 to identify changes in brain function associated with changes in motor outcomes.
Electromyography
EMG activity was recorded from 16 bipolar electrodes placed at 8 different locations obtaining data from muscles in both paretic and healthy upper limbs: (1) extensor carpi ulnaris; (2) extensor digitorum; (3) flexor carpi radialis, palmaris longus, flexor carpi; (4) biceps; (5) triceps; (6) anterior deltoid; (7) deltoid; and (8) posterior deltoid. Patients were asked to perform 6 concurrent bilateral movements from the upper-limb arm and hand FMA: (1) shoulder flexion, (2) shoulder abduction, (3) elbow extension, (4) supination, (5) wrist extension, and (6) finger extension. In another task, patients were asked to perform continuous opening and closing of the paralyzed hand at a comfortable speed and pace (finger extension and flexion).
Acquired changes in EMG amplitude during muscle contraction provided indicators for EMG signal amplitude and frequency 6 and could be used to decode motor intention. 27
Functional Magnetic Resonance Imaging
Inside the scanner, patients were asked to perform 3 different tasks: (1) to perform (try to perform) hand closing and opening, (2) to imagine hand closing and opening, and (3) to remain motionless. Conditions and movement pace (every 1.5 s) were cued by auditory-visual signals. A lateralization index (LI) was calculated to assess changes in cortical lateralization.29,30 The LI, computed as the normalized difference between the number of all active voxels in the ipsilesional and contralesional areas (anatomically defined regions of interest according to MNI space) was assessed separately for motor and premotor cortices, and for the somatosensory cortex of the paretic and healthy hands. 31 All patients underwent fMRI, but only those who underwent the follow-up measurement and presented with subcortical lesions only (not directly affecting sensorimotor and premotor areas) were considered for LI assessment (more information about fMRI data acquisition and processing can be found in our previous study 6 ).
Postintervention
Patients were allowed to be involved in any other rehabilitation intervention. A battery of exercises comprising functional movements performed during intervention physiotherapy sessions were explained, trained, and summarized in a handbook, which was given to the patients for home training. Exercises for home training were individually adapted based on each patient’s goals and residual or regained motor capacity, but the expected frequency of home training was the same for all patients comprising 2 sessions of 30 to 45 minutes of exercises per day. These movements were designed to maintain and improve motor function. In the follow-up measurement, patients were required to fill out questionnaires regarding the use and practice of the aforementioned home training. A value ranging from 3 to 1 was calculated for each person to evaluate his/her frequency of exercising at home, on which 3 = Exercises were regularly done (maximal score) and 1 = Exercises were not done (minimal score).
Results
Correlation of Motor Assessment Scores With Frequency of Home and BMI Training
Self-reported frequency of home training presented no significant difference between C+ and Sham groups (C+: 2.6 ± 0.09; Sham: 2.27 ± 0.17; P = .08). We found no significant correlation between frequency/intensity of home training and changes in motor function or impairment (as evaluated by cFMA scores, EMG activity, MAL, GAS, and Ashworth scores) between Post1 and Post2 (Supplementary Table 1). Correlation between number of BMI runs (ie, BMI training frequency) and changes in motor function or impairment indicated a significant correlation with changes in cFMA scores from Pre to Post2 (P = .001; r = 0.761) and near significance correlation with changes in cFMA scores from Pre to Post1 (Post1 − Pre: P = .03, r = 0.543; significant values were Bonferroni corrected for multiple [12] comparisons [P = .004]; see Supplementary Table 2). Only 7 patients did not visit a therapist, and 12 and 11 patients from the experimental sham group, respectively, visited a therapist between Post1 and the follow-up. All but 1 patient from the sham group (who trained the home exercises with the therapist) did the physiotherapy that they used to do before the intervention, and none of them was involved in any other therapy that was not the standard physiotherapy treatment they got before the intervention.
Primary Behavioral Outcome Measure: Combined Hand and Arm Scores (Motor Part) From the Modified cFMA
In our previous study, analyses of cFMA scores (2-way mixed-model ANOVA) indicated a significant Group (C+ and Sham) × Session (Pre and Post1) interaction, and post hoc tests indicated that cFMA changed significantly over time in the experimental group only. 6 Here, we assessed the maintenance of this effect after 6 months comparing Pre with Post2 and Post1 with Post2 measurements. A paired-samples t-test revealed a significant increase in cFMA scores in the C+ group when comparing Pre (11.16 ± 1.73) and Post2 sessions (13.44 ± 1.96, P = .015) and no significant changes between Post1 (14.56 ± 1.95) and Post2 sessions (P = .2). Paired-samples t-tests showed the same long-term effects in the arm scores but not in the hand FMA scores (see Figure 2 and Table 1). Analyses of cFMA scores in the Sham group indicated no significant changes between Pre (13.29 ± 2.86) and Post2 (14.75 ± 2.71; P = .07) sessions nor between Post1 (13.64 ± 2.91) and Post2 (P = .52) sessions.
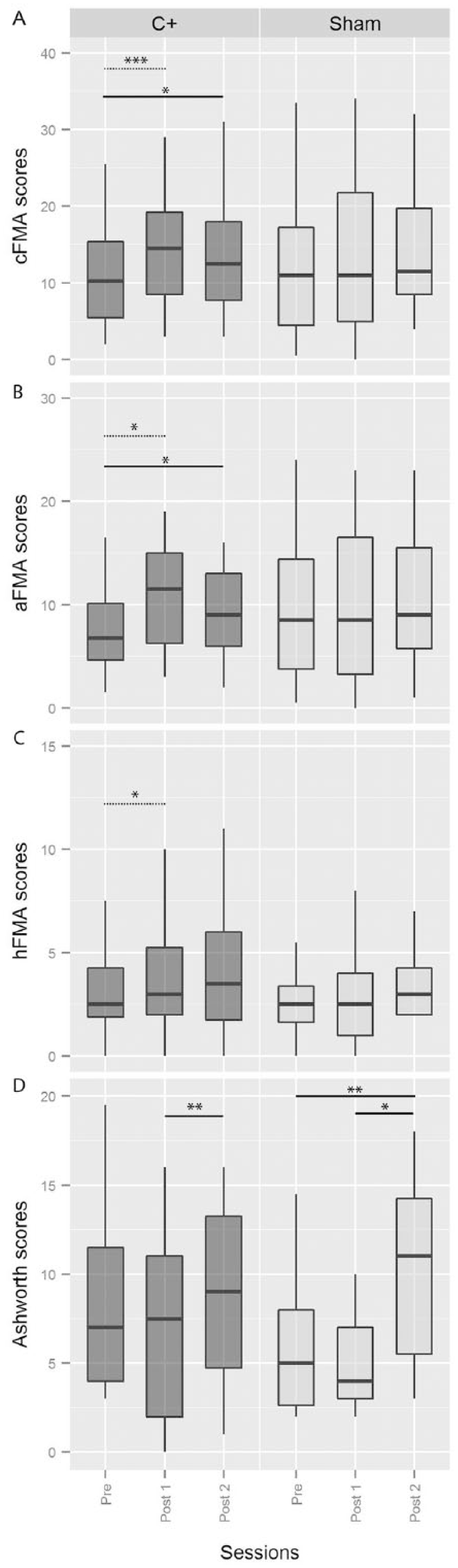
Motor impairment and spasticity. FMA and Ashworth scores before, immediately after, and 6 months after BMI intervention. Values are presented as medians and quartiles.a
Fugl-Meyer Assessment Scores Changes in Hand, Arm, and Upper Limb. a
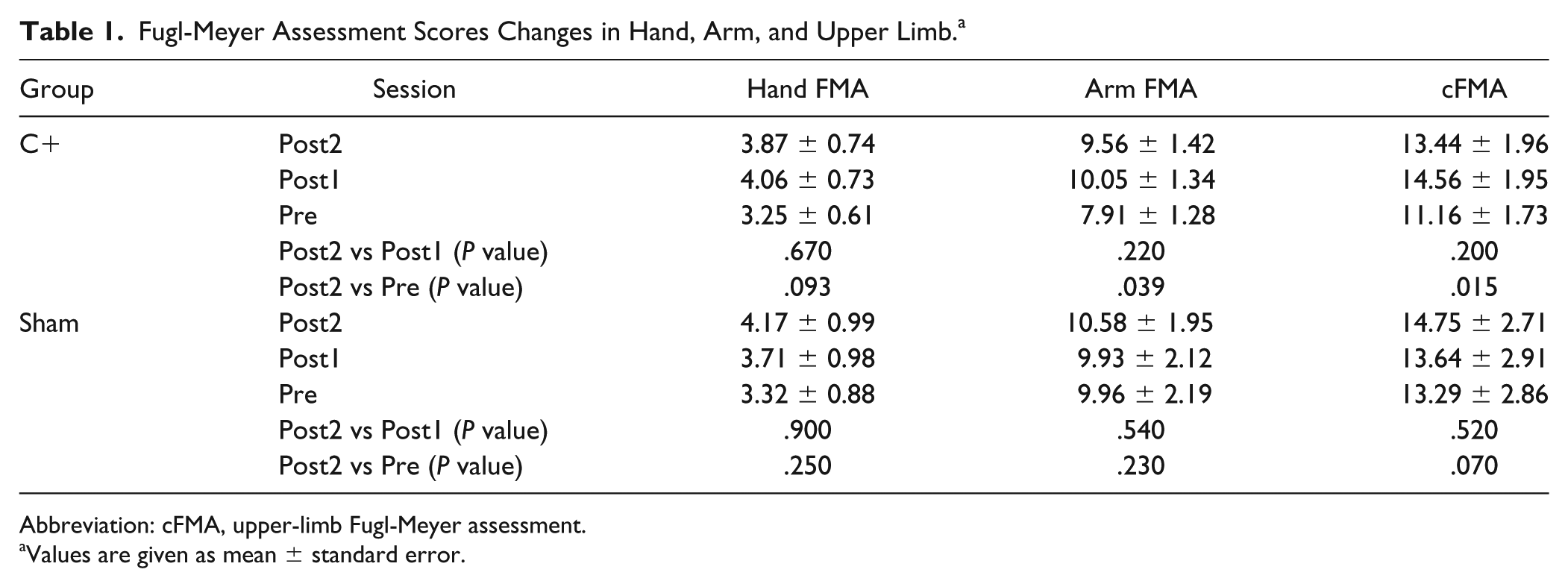
Abbreviation: cFMA, upper-limb Fugl-Meyer assessment.
Values are given as mean ± standard error.
Primary Behavioral Outcome Measure: Analyses of Motor Scores From the Modified cFMA in Patients With Mixed or Subcortical Lesions
Although we found a significant increase in cFMA scores only in the experimental group immediately after intervention and 6 months after intervention, it could be argued that there are slightly more patients with mixed lesions (ie, affecting cortical and subcortical structures) in the control group (n = 9) than in the experimental group (n = 6), and this bias could influence changes in motor outcomes after BMI-based rehabilitation. To investigate the influence of preserved cortex on motor recovery after BMI-based rehabilitation, we analyzed changes in cFMA scores between C+ and Sham groups after intervention in patients with either subcortical lesions only—that is, when excluding patients with mixed lesions—or with mixed lesions only—that is, when excluding patients with subcortical lesions.
A 2-way mixed model ANOVA with Time (Pre, Post1, and Post2) as within-subjects factor (repeated measures) and Group (C+ and Sham) as between-subjects factor indicated that in patients with subcortical lesions, there was only a significant Time × Group interaction (F1,13 = 6.143; P = .015), but no significant effect of Time (F1,13 = 1.271; P = .32) or Group (F1,13 = 1.57; P = .23). Post hoc t-tests indicated a significant increase in cFMA scores in the C+ group (n = 10) only from Pre (10.4 ± 2.33) to Post1 (13.7 ± 2.74; P = .002) sessions, but there were no significant changes between Pre and Post2 (12.1 ± 2.82; P = .17) or between Post1 and Post2 (P = .24) sessions. No significant changes in cFMA scores were found in the Sham group (n = 5).
Results in mixed lesion patients indicated no significant Time × Group interaction (F1,13 = 0.258; P = .77) and no significant main effect of group (F1,13 = 1.312; P = .28) but a significant main effect of time (F1,13 = 7.524, P = .003). Post hoc t tests indicated that patients with mixed lesions (regardless of feedback modality) significantly increased cFMA scores from Pre (11 ± 1.88) to Post1 (13.33 ± 2.17; P = .034) and from Pre to Post2 (13.23 ± 1.7; P = .005) sessions.
Furthermore, we separated the patients in 2 groups depending on lesion location (independent on feedback group) and performed a 2-way ANOVA (delta cFMA, Lesion) to analyze the difference in recovery based on lesion location. We found no significant difference in delta cFMA scores (delta Pre-Post1 F1,28 = 2.197, P = .149; and delta Pre-Post2 F1,26 = 0.017, P = .896) depending on lesion location. These results suggest that in mixed lesion patients, the BMI effect might not be key in the overall patients’ minimal but significant motor improvement effect, as it is in the subcortical lesion only patients. However, lesion location was not the main factor regarding recovery because when feedback contingency was ignored, no significant result was found. Nevertheless, because of the low number of patients, these results should be carefully interpreted.
Secondary Outcome Measures: GAS, MAL, and Ashworth Scores
In the previous published analyses between Pre and Post1 data, we observed a significant improvement of MAL and GAS scores in both groups and no significant improvement of Ashworth scores. 6 In the follow-up analyses, we found a significant increase in MAL and GAS scores from Pre to Post2 sessions and no significant change in MAL or GAS scores between Post1 and Post2 sessions in both the C+ and Sham groups (Table 2). We found a significant increase in Ashworth scores (ie, increase in spasticity) from Pre to Post2 sessions in the Sham group (z = −2.63; P = .009) but not in the C+ group (z = −1.269; P = .2). Moreover, we found a significant increase in Ashworth scores from Post1 to Post2 sessions in both experimental (z = −2.764; P = .006) and Sham groups (z = −2.271, P = .023; Table 2). To investigate the influence of the increase of spasticity in the cFMA, MAL, and GAS scores during the follow-up measurement, we correlated delta (score difference between 2 sessions) of GAS and MAL with delta of Ashworth scores (Post2 − Pre and Post2 − Post1). We found no significant correlations between delta Ashworth and delta GAS, delta MAL scores, or delta cFMA scores in any group (Supplementary Table 3). Furthermore, a Mann-Whitney U test on delta Ashworth scores (ie, changes in Ashworth scores between Post1 and Post2 or between Pre and Post2 sessions) did not show significant differences between the experimental and control groups (Delta Ashworth scores from Post1 to Post2 session: C+ = 2.75 ± 0.79, Sham = 3.92 ± 1.36, P = .47; from Pre to Post2 session: C+ = 1.53 ± 1.09, Sham = 4.58 ± 1.28, P = .094).
Experimental and Sham Group MAL, GAS, and Ashworth Scores in Assessment Pre, Post1, and Post2. a
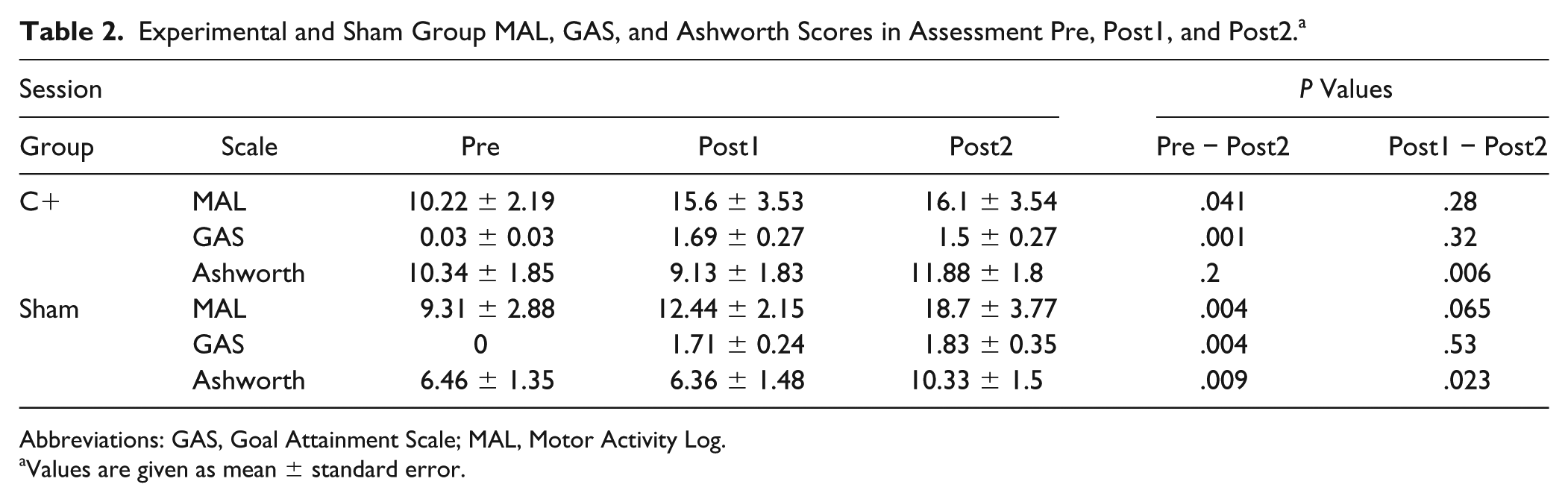
Abbreviations: GAS, Goal Attainment Scale; MAL, Motor Activity Log.
Values are given as mean ± standard error.
EMG Waveform Length Delta (After − Before) Values of Arm and Hand Muscles During Distinct Upper-Limb Movements. a
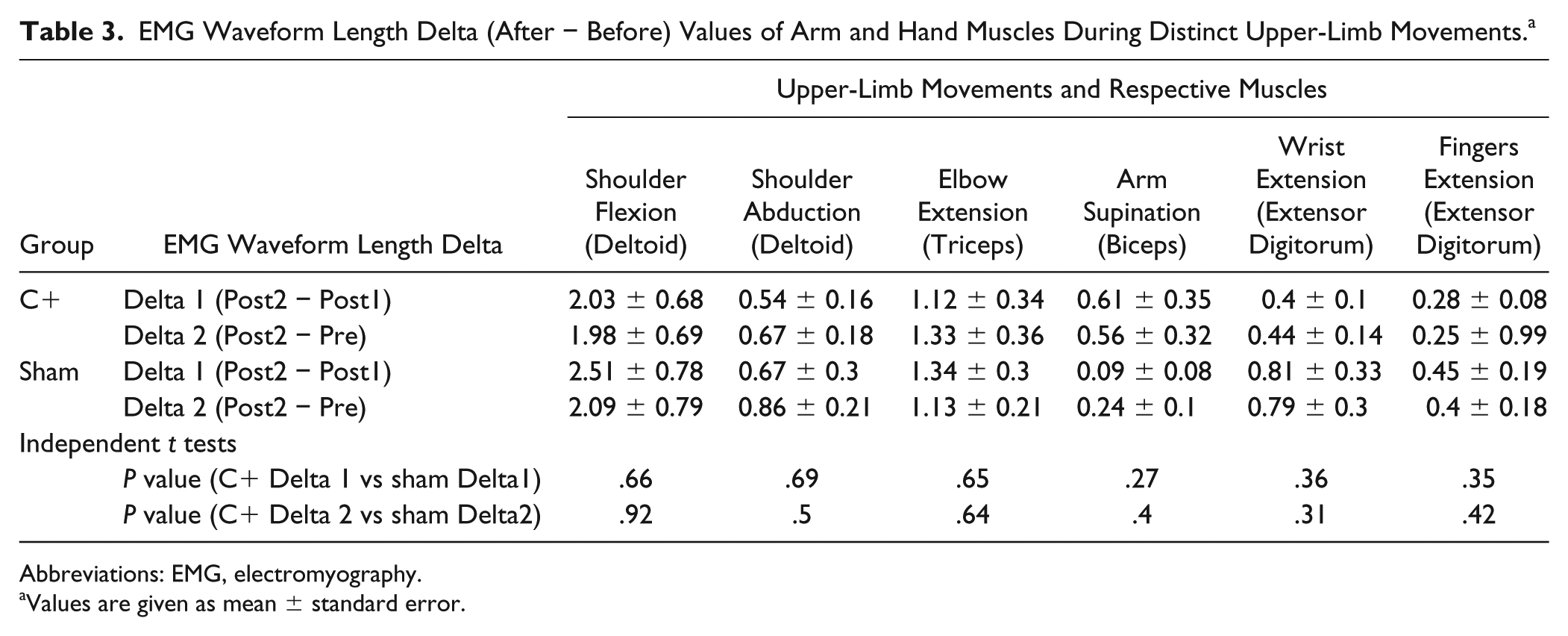
Abbreviations: EMG, electromyography.
Values are given as mean ± standard error.
Electromyography Waveform Length
Analyses of forearm EMG activity during continuous attempt to perform opening and closing of the hand (extend and flex the fingers) with Wilcoxon signed rank test (data were not normally distributed) indicated no significant changes between Pre and Post2 sessions on the C+ (Pre = 2.5 ± 0.49; Post2 = 2.28 ± 0.54; z = −0.943; P = .35) or Sham groups (Pre = 2.33 ± 0.48; Post2 = 3.74 ± 1.11; z = −0.628; P = .53), confirming the results we observed previously when comparing Pre and Post1 data. 6 Similarly, we found no significant difference between Post1 and Post2 sessions in the C+ (Post1 = 3.66 ± 0.74; z = −0.114; P = .91) or Sham groups (Post1 = 3.58 ± 0.97; z = −0.941; P = .35). Mann-Whitney U tests indicated no significant difference between experimental and Sham groups in EMG waveform length delta (after-before difference) from Post2 and Pre (C+ = −0.57 ± 0.66; Sham = 1.41 ± 1.07; U = 60; P = .33) or Post2 and Post1 (C+ = −1.41 ± 1.04; Sham = −0.29 ± 1.25;U = 75; P = .89) sessions.
EMG waveform length was also assessed during performance of upper-limb movements and posture holding. Whereas in the previous analyses comparing Pre with Post1 data we observed significant improvement of EMG activity in the experimental group only during elbow and upper-arm extension at the frontal and lateral parts of the deltoid and at the triceps, paired t tests on EMG waveform length of the different muscles evaluated during distinct upper-limb movements revealed a statistically significant increase of muscle activity in both groups from Pre to Post2 and from Post1 to Post2 sessions (Supplementary Table 4). Independent t tests indicated no significant difference between experimental and Sham groups’ EMG waveform length changes from sessions Pre to Post2 or Post1 to Post2 in any of the upper-limb muscles, indicating that both groups presented no significant difference on the amount of increase in upper-limb EMG activity (Table 3).
Because spasticity can be seen as an agonist/antagonist EMG ratio (ratEMG) conflict, we calculated and analyzed ratEMG changes and their relationship with recovery and spasticity, and we found no significant interactions or main effects (see supplementary information).
fMRI Analyses
Whereas in the previous analysis between Pre1 and Post1 we found significant differences in LI in the experimental group only toward normalized contralateral activation during paretic hand movements, no significant changes between Pre1 and Post2 sessions in the C+ (Pre1 = −0.044 ± 0.097; Post2 = −0.157 ± 0.124; P = .17) or Sham (Pre1 = −0.119 ± 0.149; Post2 = −0.066 ± 0.245; P = .77) groups were observed. We also found no significant changes between Post1 and Post2 sessions in the C+ (Post1 = −0.271 ± 0.13; P = .41) or Sham (Post1 = 0.271 ± 0.159; P = .33) groups, and this time no correlation was found between changes in cFMA and LI (Supplementary Table 5).
Discussion
In a previous study, we demonstrated that BMI training associated with behavioral physiotherapy is an efficient strategy to promote arm and hand motor recovery in severely paretic chronic stroke patients. 6 Our current findings complement this previous study and indicate that significant improvements in upper-limb motor function (FMA, GAS, MAL) are partly preserved 6 months after BMI-based rehabilitation. Specifically, the experimental group only showed a sustained significant improvement in cFMA motor scores 6 months after intervention as compared with baseline assessments. When analyzing hand and arm FMA scores separately, we found that long-term effects are preserved for the arm FMA scores only.
The exact instructions for the home training were designed to maintain the residual movement the patients presented at Post1. Not having a day-by-day control on the training might be a confounding factor because some patients may train more than others, and the ones with more residual movement capacities could train more and might be more motivated to do so. However, if one looks at the mean cFMA scores, the sham group patients had higher scores than the control group and, therefore, could have had a larger gain at Post2, which was not the case. Furthermore, no significant difference related to frequency of home training after the intervention between experimental and Sham groups was found, and along the same line, no correlation between frequency of home training and any of the outcome measure changes was found. These findings indicate that significantly higher cFMA scores 6 months after intervention as compared with baseline measurements found only in the experimental group cannot be attributed to differences in home training between groups and were not a result of behavioral physiotherapy only. These results strengthen the importance of the contingent visual and proprioceptive feedback attempting to “associatively bridge” the lesion via BMI and may overcome the learned nonuse effect produced by paralysis.32,33
The significant increase in spasticity in the Post2 session on both did not correlate with any of the motor assessment (cFMA, EMG, GAS, MAL) scores. This strengthens the functional motor improvement result from our interventional study as independent from changes in spasticity, indicating no long-term effect of the BMI intervention in the reduction of spasticity seen after intervention in the experimental group only.
Moreover, besides increased spasticity, both groups also presented significant increase in voluntary EMG activity during upper-arm and hand movements, suggesting an overall increase in patients’ capacity for muscular contraction—either voluntarily (as indicated by EMG) or associated with spasticity. Because both groups significantly increased spasticity and upper-limb EMG activity between Post1 and Post2 and both groups increased EMG activity from Post1 to Post2 by a similar degree (no statistical difference was found between increase in voluntary EMG activity in both groups), these results may reflect an effect of home training and increase of muscle use instead of BMI training. However, there was no correlation between reported frequency of home training and increase of EMG activity from Post1 to Post2. Alternatively, increased spasticity and EMG activity in the Post2 session in both groups could be an effect of the physiotherapy, provided in equal extent to both groups during training. Physiotherapy may affect muscle strength in the long run more than BMI because BMI is focused on the association between intention and movement, whereas physiotherapy is focused on visible muscle activation. Therefore, the importance of the BMI to guide correct, or at least well-timed, muscle activation seems to be confirmed. Furthermore, because no significant correlation was found between frequency of home training and increased Ashworth scores or EMG activity during upper-limb movements, we hypothesize that this effect is associated with the quality (ie, type) of exercises instead of frequency of exercises during home training. For example, without proper therapeutic guidance, patients may have performed more strenuous movements during home training plausibly inducing increased spasticity and increasing nonspecific EMG activation during attempts to perform the requested task as a consequence. This would likely be associated with less-efficient movements (ie, more “noisy” movements). This assessment of movement quality may not be identifiable by EMG but could explain why increased EMG activity between Pre and Post1 in the experimental group 6 was not preserved in the follow-up assessment (Post2). Alternatively, participation in this highly motivating treatment (for most patients, this may be the last resource of medical hope because no other treatment was proposed for these patients by their medical consultants) increased EMG activity and Ashworth as an unspecific side effect.
We want to emphasize that the patients in the control group received contingent correct feedback by chance sometimes during the intervention. The random movements of the orthoses coincided sometimes with desynchronization of the SMR rhythm. This difference was computed to be on average around 20% of the training time, implying that even random feedback can sometimes result in correct feedback and may induce some limited neuroplastic changes. These results emphasize the importance of the link between brain motor activity and the relevance of improving decoding of brain activity to allow instrumental learning to stimulate functional neuroplasticity. The consequences of neuroplastic mechanisms during the chronic phase have been demonstrated to be very limited compared with the time windows during the acute phase. 34 BMI might allow access to some of the neuroplastic mechanisms to allow significant motor functional recovery even in the chronic phase. How movement intention decoding accuracy, intensity of training (intervention schedule: how many days, hours and how often), movement controlled by BMI, and physiotherapy would affect the functioning and stability of neuroplastic mechanisms needs to be further investigated.
Our fMRI results suggest a trend toward significance when comparing Pre and Post2 in the experimental group only. This might mean that if the associative link between brain and behavior is no longer there, the brain activity change toward normalized ipsilesional activity, which might be considered as functional plasticity, is slowly being reversed, because residual movement–based training might not suffice to regain impaired movement specific ipsilesional activity. Furthermore, our results demonstrating that LI changes did not correlate with cFMA changes between Post1 and Post2 nor between Pre and Post2 suggest that BOLD activity changes observed during the intervention might represent motor learning (BMI motor control learning) and not motor recovery, as we hypothesized in our previous work. 6 On the other hand, the recovery might have consolidated in spinal neural networks, and therefore, their appearance might last longer than the brain effect and are not captured by the LI analysis.
We demonstrated that altering a brain signal (increase in SMR desynchronization) time-contingent with visual and proprioceptive sensory feedback associated with orthosis and, thus, limb movement leads to significant motor improvement and functional neural reorganization, promoting motor function improvement and partial maintenance of the found effect. However, most other findings (secondary outcomes, hand cFMA, imaging, EMG) do not point in the same direction. The sustained significant improvement in cFMA scores 6 months after intervention might reflect recovery from motor impairment. For example, a clinically meaningful change from no activity to some activity in lifting and stretching the arm, turning the forearm, and extending the wrist and/or fingers was preserved 6 months after intervention. It is conceivable that BMI training engaging a crucial network of brain regions related to intent (visuomotor task) could stimulate activation of neuroplastic mechanisms that allow physiotherapy to promote functional motor recovery in chronic severe stroke (evidenced in cFMA scores and EMG activity immediately after the intervention 6 and in the sustained cFMA scores 6 months after the intervention). However, the experimental design used here does not allow firm conclusions to be drawn relating the relevance of behavioral physiotherapy to the improvements in motor function.
Despite the small samples we used in the analyses regarding lesion location relevance in motor recovery when using BMI coupled with physiotherapy as intervention, our results suggest that lesion location might not be a limiting factor. However, these results should be considered with caution. Further studies, including more patients or including only patients with specific lesion locations (eg, mixed or subcortical lesions) in the inclusion criteria are necessary to better evaluate BMI training effects in motor recovery according to the patient’s lesion location. Moreover, it may be considered that motor recovery after BMI training and physiotherapy has not reached a level of motor improvement that has a significant clinical impact on chronic stroke patients. Still, we believe that as a proof of concept, this study demonstrates the potential of BMI to promote long-term motor recovery in chronic stroke patients with severe paresis, a population that is currently unable to undergo any rehabilitation. In any case, to promote stronger clinical impacts, further development of BMI training may include implementation of (1) functional electrical stimulation to induce paretic limb movements, because it also activates muscle contractions 35 ; (2) invasive SMR recordings (invasive BMI), which record SMR with superior signal quality (eg, less noise and better resolution); (3) broader range of practiced movements; and (4) longer rehabilitation interventions.
Conclusions
In summary, our proof of concept study demonstrated that BMI-based rehabilitation in chronic stroke patients with severe paresis successfully promotes motor recovery and cortical reorganization associated with recovery of function and induces long-lasting improvements in motor function.
Supplemental Material
Supplementary_Material – Supplemental material for Brain-Machine Interface in Chronic Stroke: Randomized Trial Long-Term Follow-up
Supplemental material, Supplementary_Material for Brain-Machine Interface in Chronic Stroke: Randomized Trial Long-Term Follow-up by Ander Ramos-Murguialday, Marco R. Curado, Doris Broetz, Özge Yilmaz, Fabricio L. Brasil, Giulia Liberati, Eliana Garcia-Cossio, Woosang Cho, Andrea Caria, Leonardo G. Cohen and Niels Birbaumer in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We would like to thank Dr Massimiliano Rea, Dr Leonhard Laeer, and Dr Manuel Agostini for their assistance during data acquisition.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
Authors’ Notes
1. Ander Ramos-Murguialday and Marco R. Curado contributed equally. Study concept/design: AR-M, NB, and LGC. Data acquisition: AR-M, MRC, DB, OY, FLB, GL, EG-C, WC, AC. Statistical analysis and figures: MRC and AR-M. Manuscript drafting: AR-M and MRC. Manuscript revision and final approval: all authors. Andrea Caria is now at University of Trento, Trento, Italy.
2. Özge Yilmaz is now affiliated with Department of Psychology at Altinbas University, Istanbul, Turkey.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Bundesministerium für Bildung und Forschung (BMBF: MOTORBIC, FKZ 13GW0053) and AMORSA (FZ 16SV7754) as well as the Deutsche Forschungsgemeinschaft (DFG, Koselleck); Baden-Württemberg Stiftung (GRUENS ROB1); Diputacion Foral de Gipuzkoa (INKRATEK), the fortüne-Program of the University of Tübingen (2422-0-1 and 2452-0-0); the Basque Government Science Program (EXOTEK: KK 2016/00083); the Intramural Research Program (IRP) of the National Institute of Neurological Disorders and Stroke (NINDS), Bethesda, Maryland, USA; the Center for Neuroscience and Regenerative Medicine (CNRM); Uniformed Services University of Health Sciences, Bethesda, Maryland, USA. Several authors were supported by the DAAD (Deutscher Akademischer Austauschdienst) to EGC and WC; CNPq (Brazilian National Counsel of Technological and Scientific Development) to FB; CAPES (Coordination for the Improvement of Higher Level -or Education-Personnel, Brazil) to MRC; and Humboldt Award to LC. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
