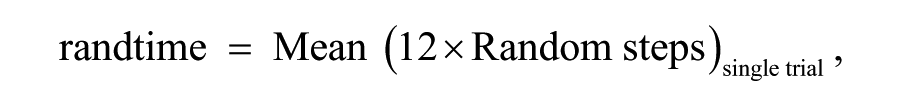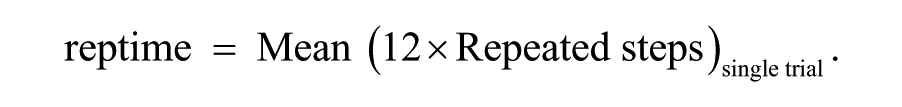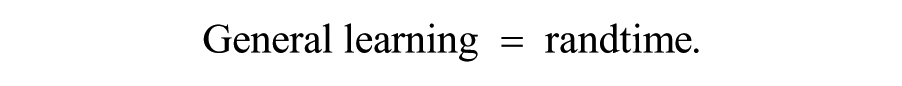Abstract
Introduction. Treatment of Parkinson’s disease (PD) with exogenous dopamine (ie, levodopa) may positively affect motor symptoms, but may negatively affect other functions such as the learning of motor skills necessary for rehabilitation. This study aimed to determine whether levodopa medication affects general and sequence-specific learning of a stepping task and the transfer of movement skill to untrained balance tasks in people with PD. Methods. Participants with PD were randomized to practice “on” (n = 14) or “off” (n = 13) levodopa medication. Participants practiced 6 blocks of 6 trials of 24 steps of a stepping task over an acquisition period of 3 consecutive days, followed by single retention blocks of 6 trials 2 and 9 days later. Participants were also assessed on untrained balance (ie, transfer) tasks “on” levodopa before practice and following late retention. Results. There were no between-group differences in general learning, sequence-specific learning, or transfer of skill to untrained balance tasks (P > .05). Both groups demonstrated general and sequence-specific learning (P < .001) and trends for improvement in untrained tasks (P < .001 to P = .26) following practice. Detailed analysis of early acquisition revealed no difference between medication groups. Conclusion. People with PD improved performance on the stepping task with practice. The between-group effect sizes were small, suggesting that levodopa medication status (“on” versus “off”) during practice did not significantly affect general or sequence-specific learning of the task or components of early acquisition. The practice dose required to optimally result in functional improvements in untrained balance tasks, including reductions in falls, remains to be determined.
Introduction
Idiopathic Parkinson’s disease (PD) produces postural motor deficits that lead to significant disability. 1 Because of postural instability, 2 falls are common and are more likely to cause injuries (eg, fractures) in people with PD compared with other older adults.3,4 Pharmacological management reduces the motor signs experienced and partially normalizes motor control 5 ; however, even with optimal pharmacological management, postural instability and falls may occur early in the disease6,7 and become more problematic as the disease worsens.5,8 Through relearning of anticipatory and reactive postural motor tasks, physical therapy seeks to improve mobility and fall risk. 9
Motor learning forms the basis for rehabilitation because it is the critical process for acquiring new skills or refining existing skills through practice. 10 The neural mechanisms of motor learning appear to involve both the associative and sensorimotor portions of the striatum. 11 Studies using a variety of motor learning paradigms document a retained, albeit slowed, motor learning ability in individuals with PD relative to those without neurological deficits,9,12 particularly in sequence-specific learning.10,13,14 People with PD also do not achieve the same extent of skill acquisition and retention as people without neurological deficits.12,13 Impaired motor learning in PD appears to be associated with disease severity,14,15 likely as a result of reduced endogenous dopamine and reduced dopamine binding associated with loss of dopaminergic receptors with disease progression. 16 Yet because exogenous dopamine is delivered systemically to treat the symptoms of PD, it may overdose and suppress activation of the associative striatum during the early acquisition stages of motor learning.15-17 Thus, impaired motor learning in PD may be the product of both dopamine deficiency organic to the disease and to overdosing of the basal ganglia with exogenous dopamine. Regardless of the cause, the clinical relevance of these findings is unclear, and the influence of exogenous dopamine on the acquisition, retention, and generalizability of clinically relevant anticipatory balance tasks such as stepping has not been examined.
Although motor learning can be examined in various ways, the use of tasks with embedded sequences allows researchers to segregate general skill learning from implicit motor sequence learning.18,19 Such a segregation provides insight into the general motor skills gained by participants through practice as well as their ability to acquire and retain movement sequences without explicit knowledge of the sequence. Understanding which factors optimize or impair learning in people with PD will allow rehabilitation clinicians to prescribe practice regimes that will successfully improve postural motor skills, which in turn may reduce falls. 9 We conducted a randomized clinical trial to examine the effects of exogenous dopamine on postural motor learning. The aims of this study were to determine if exogenous dopamine (ie, levodopa medication) affects (1) general and sequence-specific learning of a stepping task, including early acquisition, 17 and (2) transfer of skill to untrained balance tasks, in people with PD. We hypothesized that retention of the stepping task will be altered in people with PD who practiced “on” compared with “off” levodopa; this was our stated primary outcome of this trial. We also hypothesized that between-group differences in motor skill acquisition would be present in early acquisition (post hoc outcome), 17 and with motor practice, people with PD will improve performance in the untrained balance tasks, with greater change shown by those who practiced “off” levodopa (secondary outcome).
Methods
Design
This randomized controlled trial of an implicitly learned stepping task was prospectively registered on ClinicalTrials.gov (NCT02593812). The study adhered to the CONSORT guidelines 20 and was approved by the University of Utah Institutional Review Board. All participants gave written informed consent prior to study enrolment.
Outcome assessments were performed at pretest and at posttest at the University of Utah or in participants’ homes by a trained assessor blinded to group allocation. The posttest and late retention testing were both performed during each participant’s final study session, 13 to 15 days following pretest. Following pretest assessment, participants were randomly allocated by the project manager (SSP) to practice the stepping task “on” or “off” levodopa medication, using a computer-generated randomization schedule with randomly permuted blocks. The randomization schedule was generated prior to recruitment and was stratified according to each participant’s Hoehn and Yahr disease stage, with a threshold value of 2.
Participants
People with PD were recruited between July 2016 and May 2017 from the community of the greater Salt Lake City area in the United States. Participants were recruited from a Movement Disorders Center, local PD support groups, and a PD wellness exercise group. Volunteers were included if they had a neurologist diagnosis of idiopathic PD, were aged 50 to 80 years, were in Hoehn and Yahr stages 1 to 3, were able to walk ⩾10 m unaided when “off” levodopa medication, and had been on a stable medication regime for at least 1 month prior to pretest assessment and would maintain that same regime for the study duration. Individuals were excluded if they were not taking levodopa medication, had prior deep brain stimulation, had significant cognitive impairment (Montreal Cognitive Assessment score < 18), 21 had self-reported medication-resistant freezing of gait, or had any health conditions that would interfere with safely conducting the assessments or motor learning intervention.
To determine participants’ levodopa responsiveness, PD severity according to the motor section of the Movement Disorders Society revised version of the Unified Parkinson Disease Rating Scale (MDS-UPDRS) 22 was scored at pretest “off” levodopa following overnight withdrawal. Participants were subsequently scored “on” levodopa 30 to 60 minutes after taking their usual morning dose. Because participants were not withdrawn from all antiparkinsonian medication (eg, dopamine agonists), a difference that exceeded the standard error of measurement of the MDS-UPDRS motor section was taken as being responsive to levodopa. 23 All other demographic and outcome assessments were conducted “on” levodopa. Demographic information collected at pretest included medical history, current medications, time since diagnosis, and fall risk. 24
Motor Learning Intervention
Participants randomized to the “off” levodopa group practiced the stepping task in the morning following overnight withdrawal of levodopa medication, 25 although they were allowed to stay on their usual dosage of other antiparkinsonian medication, including dopamine agonists. Participants randomized to the “on” levodopa group practiced the stepping task while their antiparkinsonian medications were working optimally, usually 60 minutes after ingesting their morning dose.
To ensure that the practice dosage exceeded that provided in previous postural motor learning studies13,26,27 and that we tested the preservation of skill following a period of no practice, participants practiced 6 blocks of stepping task trials (Figure 1) over 3 consecutive days (acquisition period), followed by an initial retention test and a late retention test (2 and 9 days after the acquisition period ended, respectively). Each block consisted of 6 trials, with a 25-s rest between trials and a 4-minute seated rest between blocks. Each trial consisted of a 12-step random sequence and a 12-step repeated sequence presented in random order across trials. Participants were not told of the embedded repeating sequence. Practice sessions lasted approximately 1.5 h/d, and participants completed a total of 864 steps per day, for a total of 2592 steps over the 3 days of practice. Retention tests involved a single block of 6 trials (ie, 144 steps) and lasted approximately 15 minutes. To serve as feedback, during seated rest breaks, participants were told their median response time on their previous block of practice.

Stepping task.
The stepping task was delivered via visual cues and a custom-made step mat.28,29 The step mat had 4 arrows (right, left, and 2 in the front) that served as targets as well as 2 back arrows that served as the home position from which to initiate steps and return after each step. A cue appeared on a monitor as a bright green arrow. Participants were instructed to stand on the home position and step on the appropriate target arrow (right, left, right front, or left front, as indicated by the cue on the monitor) as fast as possible while maintaining balance independently. On registering a correct step, the arrow changed to dark green, at which point, the participants returned their stepping foot to the home position.
Outcome Measures
The primary outcomes were general learning and sequence-specific learning at initial retention, adjusted for baseline performance (ie, the first trial of the first block of the acquisition period, which occurred 1-3 days after pretest). Response time was calculated as the time for each step taken between cue appearance to stepping down on the correct arrow, using the mean of 12 steps within each random (
General learning was quantified as response time of the random sequences; thus, more negative values over time indicate greater learning:
Sequence-specific learning was quantified as the difference in response time between the random and repeated sequences; thus, more positive values over time indicate greater learning:
Examination of skills at retention provides a limited view of learning in that it does not account for the level of skill preserved from the end of acquisition, nor does it provide any insight into the generalizability of the skill to potentially related tasks. To provide a broad-spectrum view of learning, we also included secondary outcome measures of decrement (ie, how much learning was retained between the end of practice, defined as the last trial of the last block of the acquisition period, and retention testing) and transfer. The measures of skill transfer to untrained tasks were the Four-Square Step Test (4SST) 30 and the Mini Balance Evaluation Systems Test (MiniBESTest), 31 both tested “on” levodopa prior to the acquisition period (ie, pretest) and immediately following late retention (ie, posttest). The 4SST required participants to step as rapidly as possible while maintaining their balance across a grid of 4 squares and then back again for a total of 8 steps. 30 The 4SST was timed with a handheld stopwatch, and participants performed 2 trials, with the average time across both trials used for analysis; faster times indicate better performance. The MiniBESTest is a 14-item test covering multiple domains of balance, including anticipatory postural adjustments, reactive postural responses, sensory organization, and dynamic balance during gait. 31 Each item is scored from 0 (unable to perform) to 2 (normal performance), providing a sum score between 0 and 28, with higher scores indicating better balance.
Two post hoc outcome measures were obtained following analysis of primary and secondary outcomes because of their close resemblance to the primary outcomes: these were general learning and sequence-specific learning at late retention adjusted for baseline performance. The rates of learning during early acquisition 17 were also investigated as post hoc outcomes.
Sample Size and Data Analysis
No prior study in healthy or impaired populations, including PD, had examined a serial reaction time motor learning paradigm involving the lower extremities. Calculations of statistical power based on previously published skill acquisition by people with PD using a nonstepping postural tracking task 13 suggested that a sample size of 15 participants per group would detect a 66% between-group difference in sequence-specific learning (mean = 53 ms between-group difference; SD = 40 ms; power = 0.8; α = .05), allowing for 20% dropout.
Visual inspection of the data revealed some outlying data points (ie, response time) resulting from either participant inattention or technical difficulties. For each individual, the mean response time was calculated across all random and repeated steps, and extreme outliers exceeding the mean plus 3 SDs for each individual were excluded from analysis. This resulted in a loss of 1.4% of data across the entire sample. Data were analyzed as the average response time of each step from either the random or the repeated sequence within a single trial.
Repeated-measures analyses of variance (ANOVAs), with group (“on” versus “off” medication) modeled as the between-subject factor and time modeled as the within-subject factor, were used to assess between-group differences (ie, Group × Time interaction) for all outcomes. The time point in consideration varied for each outcome as follows. To assess general and sequence-specific learning at initial retention (primary outcomes) and late retention (post hoc outcomes), the time points analyzed were the first trial of the first block of the acquisition period versus the first trial of initial or late retention, respectively. To assess learning decrement between end of acquisition and retention tests (secondary outcomes), the time points analyzed were the last trial of the last block of the acquisition period versus the first trial of initial or late retention, respectively. To assess skill transfer following practice of the stepping task (secondary outcome), the time points analyzed were pretest versus posttest. Cohen’s d was used to quantify standardized between-group effect sizes following practice. Outcome data were analyzed by an investigator blinded to group allocation (SYS) using JMP Pro v13 (SAS Institute, Cary, NC).
To examine potential differences in early acquisition and provide comparison to studies that have suggested a dopamine overdose effect, 17 performance curves were analyzed for both the random sequences and the repeated sequence on day 1, blocks 1 and 2 (ie, the first one-third of practice on day 1). 17 We used multilevel modeling to fit a curvilinear slope to each individual’s mean response time over the 12 trials of blocks 1 and 2. These models included factors of medication status (“on” versus “off”), trial type (random vs repeated), and the interaction of these factors with time to measure the impact of medication and trial type on early skill acquisition. 17 Thus, the multilevel model allowed us to test the effects of medication on general learning and sequence-specific learning (through the medication status by trial type interactions). The models were implemented using mixed-effect regression (with a random effect for both slopes and intercepts) 32 using R v3.4.1. 33
Results
Participant Flow and Characteristics
Recruitment was ceased at 90% of the intended target because of slow recruitment. A total of 27 participants (Table 1) were randomized to practice the stepping task “on” (n = 14) or “off” (n = 13) levodopa medication. All enrolled participants completed the entire study (Figure 2) without variation in the number of practice trials completed.
Baseline Demographic Data for the Entire Sample and by Group. a
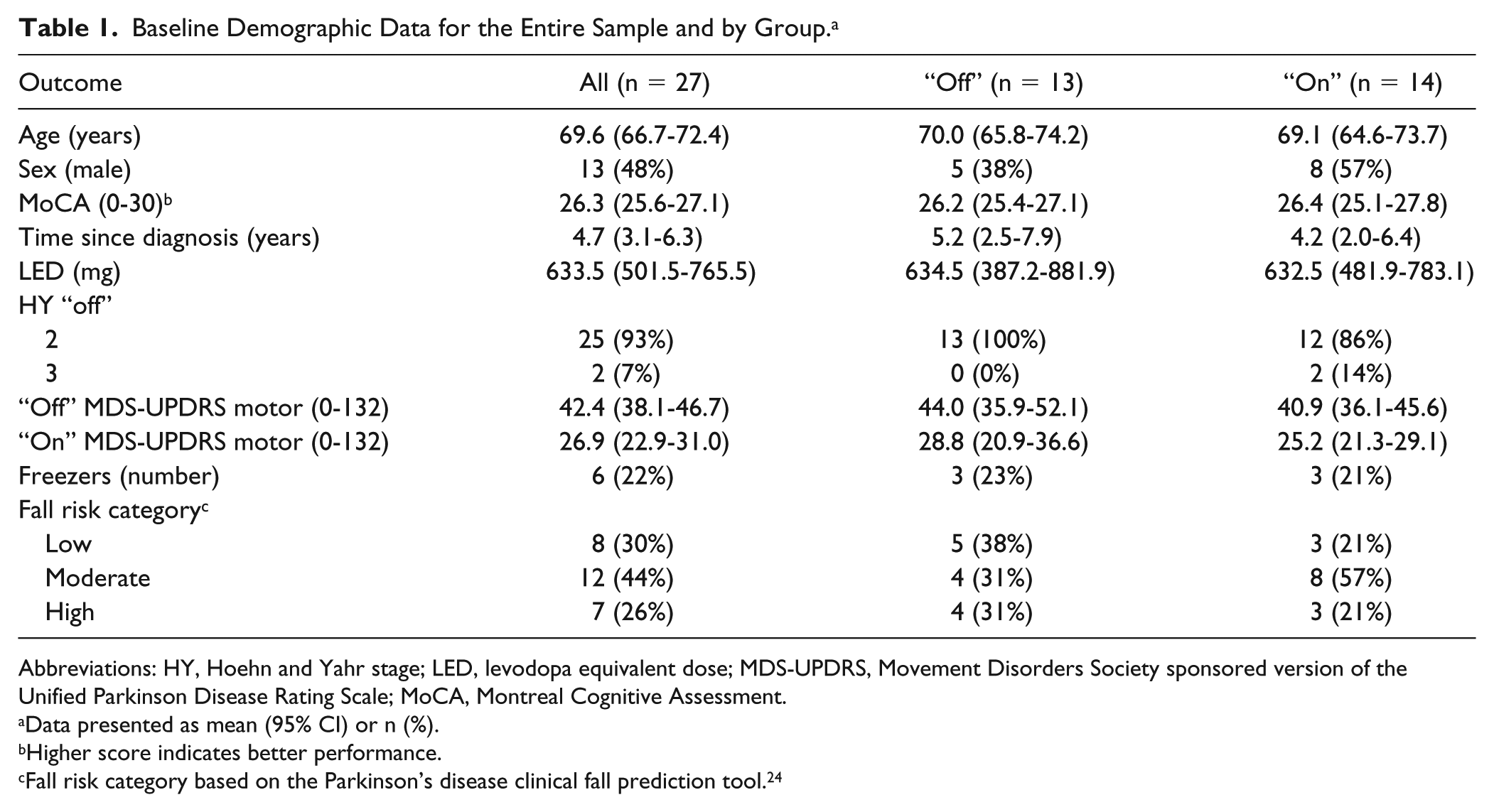
Abbreviations: HY, Hoehn and Yahr stage; LED, levodopa equivalent dose; MDS-UPDRS, Movement Disorders Society sponsored version of the Unified Parkinson Disease Rating Scale; MoCA, Montreal Cognitive Assessment.
Data presented as mean (95% CI) or n (%).
Higher score indicates better performance.
Fall risk category based on the Parkinson’s disease clinical fall prediction tool. 24
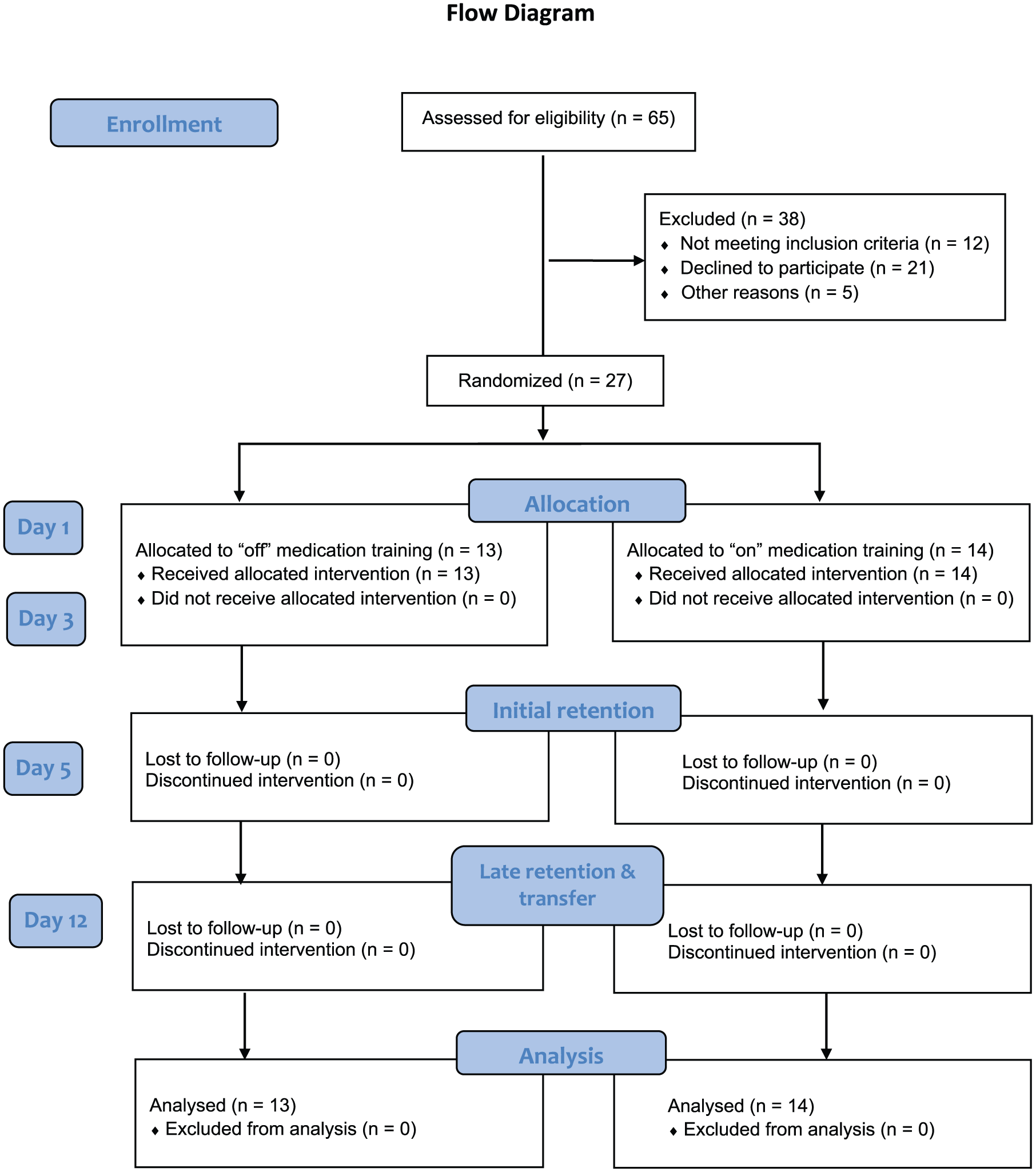
Participant flow through the pilot randomized controlled trial.
Impact of Levodopa Medication on Motor Learning
Performance at baseline (first trial of the first block of the acquisition period), end of acquisition (last trial of the last block of the acquisition period), and at both retention tests for both groups are presented in Table 2 and Figure 3. There were no significant between-group differences in general learning or sequence-specific learning at initial retention, which were the primary outcomes of this study, or at late retention (post hoc outcomes). This is evidenced by no significant interaction between group and time, regardless of whether the factor of time included initial or late retention, versus the first block of the acquisition period (Table 3). For the secondary outcomes measuring between-group differences in immediate and late-learning decrements from end of the acquisition period to retesting at retention, no significant interactions of group (“on” versus “off”) by time (retention vs end of acquisition) on step response time were found for general learning or sequence-specific learning (Table 3). The between-group effect sizes of all motor learning outcomes were small.
Mean (SD) of Raw Scores for (1) the Stepping Task for Each Group at Baseline (First Block of the Acquisition Period), End of Acquisition (Last Block of the Acquisition Period), Initial Retention, and Late Retention and (2) the Transfer Tasks for Each Group Prior to and Following Practice of the Stepping Task (Late Retention).
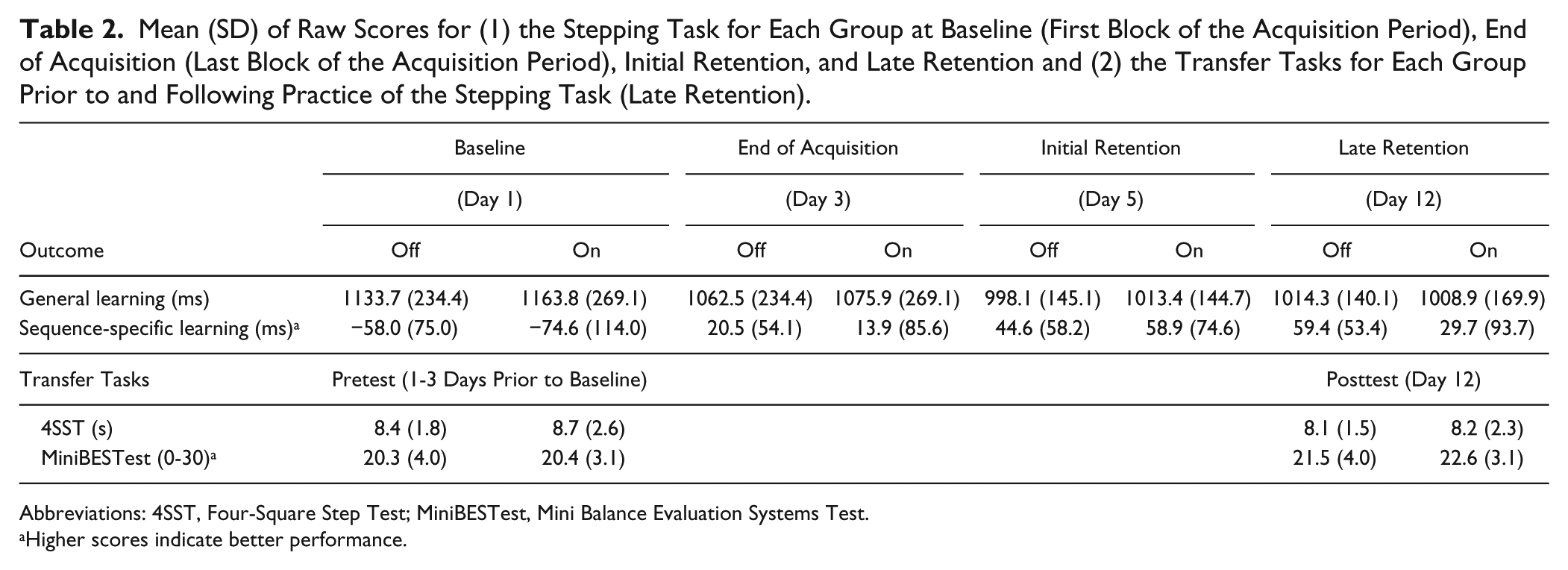
Abbreviations: 4SST, Four-Square Step Test; MiniBESTest, Mini Balance Evaluation Systems Test.
Higher scores indicate better performance.
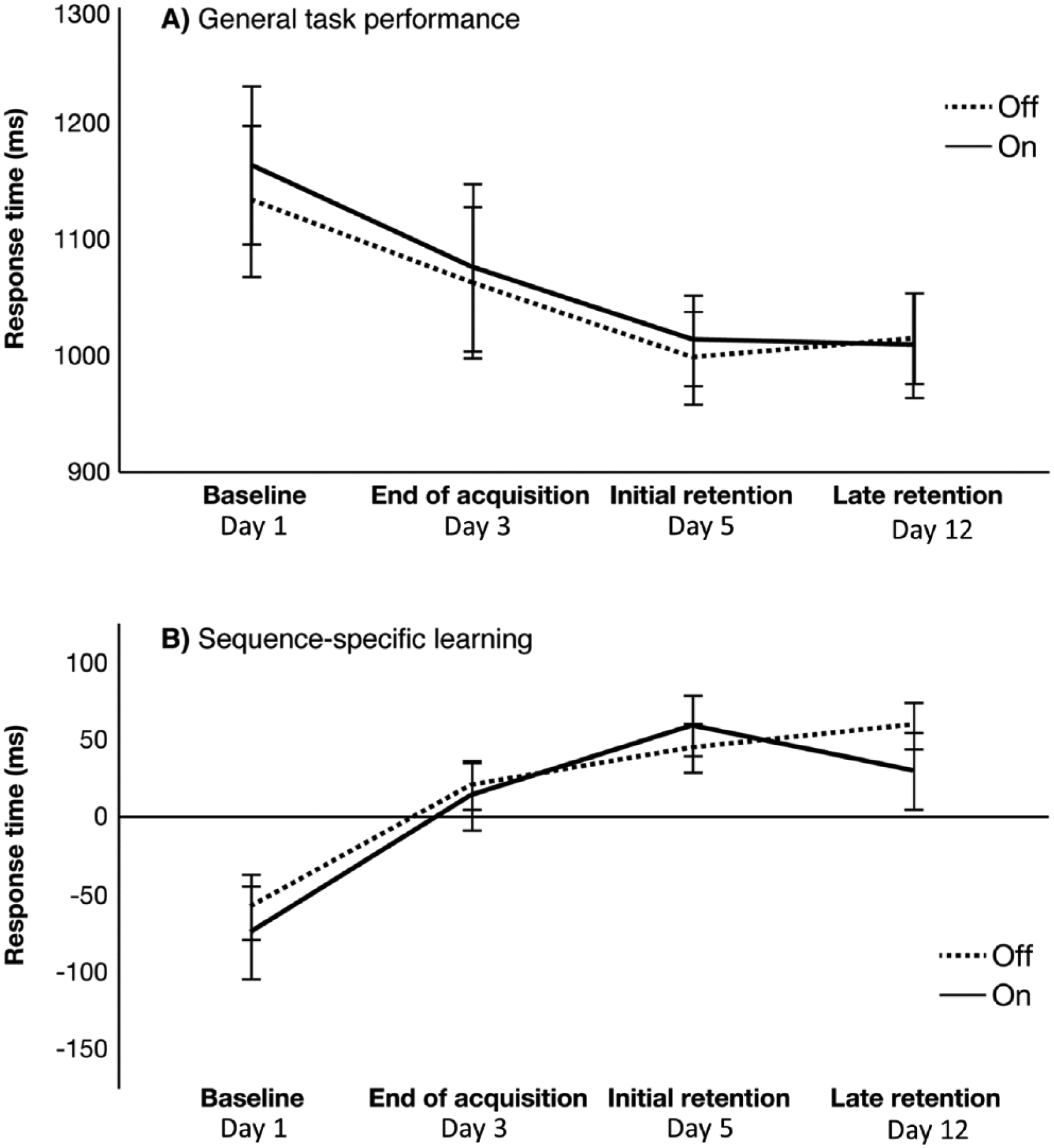
Mean and SD values for (A) general learning and (B) sequence-specific learning are shown for the “off” (dashed line) and “on” (solid line) practice groups. Smaller y-axis values in general learning (top panel, A) and larger y-axis values in sequence-specific learning (bottom panel, B) indicate greater improvement.
For Each Group, Mean (95% CI), Results From the ANOVAs and Between-Group Effect Sizes for (1) General and Sequence-Specific Learning at Initial Retention (Primary Outcomes), General and Sequence-Specific Learning at Late Retention (Post Hoc Outcomes), and Immediate and Late Decrements in General and Sequence-Specific Learning (Secondary Outcomes) and (2) the Untrained Transfer Tasks (Secondary Outcomes).
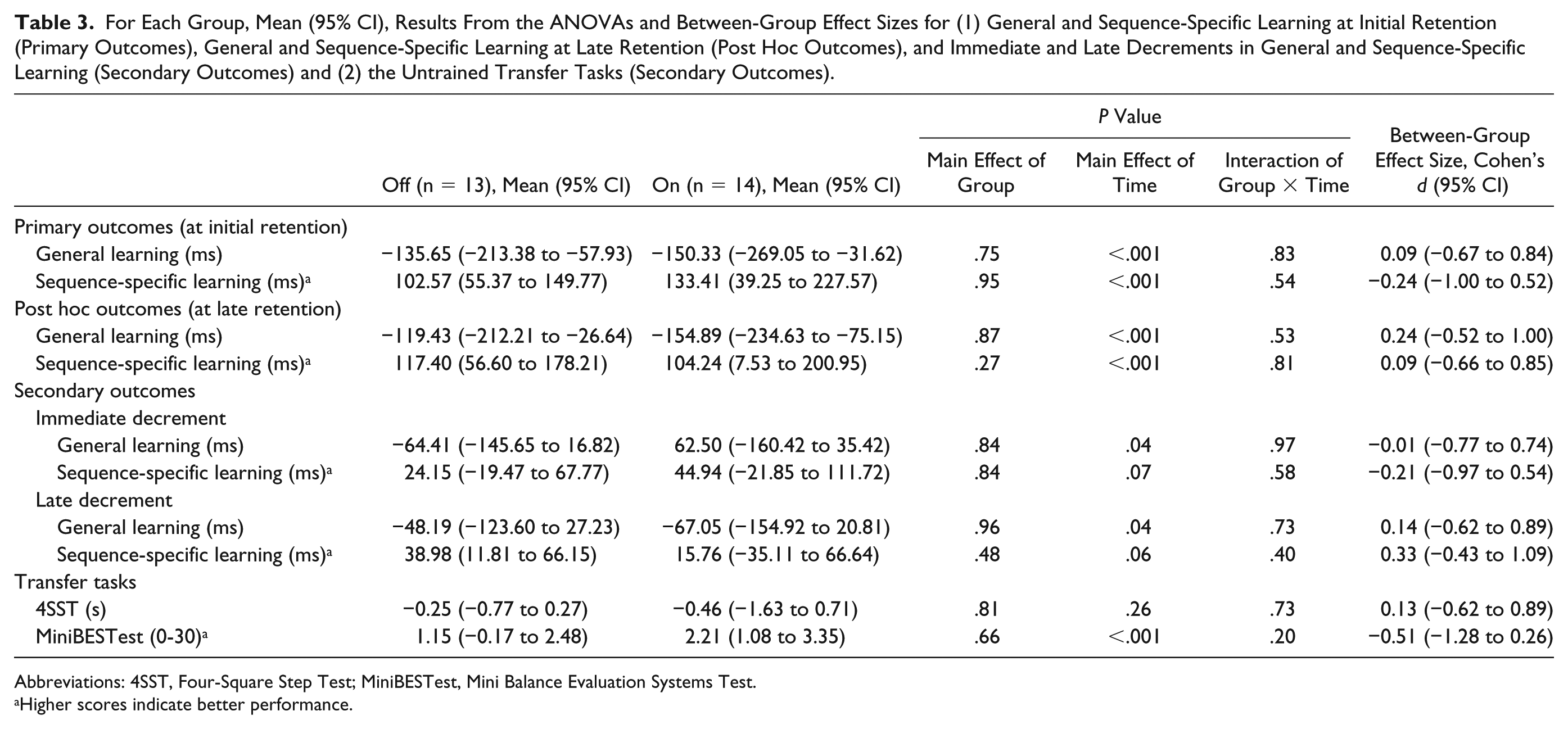
Abbreviations: 4SST, Four-Square Step Test; MiniBESTest, Mini Balance Evaluation Systems Test.
Higher scores indicate better performance.
Both groups showed similar amounts of general learning and sequence-specific learning of the stepping task with practice (main effect of time: P < .001 from baseline to both retention sessions), demonstrating that motor learning of both the general features of the postural task and the embedded sequence had occurred. Both groups also had stable sequence-specific learning (main effect of time: P ⩾ .06), indicating no learning decrement at either retention session, and had continued offline general learning following withdrawal of practice (main effect of time: P = .04).
Impact of Levodopa Medication on Transfer of Learning to Untrained Tasks
Both groups made small, clinically nonmeaningful improvements on the 4SST and MiniBESTest following practice of the stepping task (Table 2), without any significant Group × Time interaction on performance of either task (P ⩾ .66; Table 3). Improvements in the MiniBESTest following practice of the stepping task were statistically significant (effect of time: P < .001), but improvements in the 4SST were not (P = .26).
Learning Curves During Early Acquisition
There was no significant main effect of medication status on performance during early acquisition (ie, day 1, blocks 1 and 2; P = .76). Multilevel modeling revealed significant linear (P < .001) and quadratic (P < .001) effects of time, and the linear effect interacted with trial type (P < .001). As shown in Figure 4, this interaction revealed that response time improved more with practice for the repeated sequence (simple slope βtrial = −0.010) than for the random sequences (simple slope βtrial = −0.004). The rate of improvement for both trial types reduced over time (as indicated by the significant quadratic of time), but this quadratic effect was not different between the 2 trial types (P = .18). Furthermore, medication status did not significantly interact with either the linear (P = .68) or quadratic (P = .70) effects of time, indicating that participants improved at approximately the same rate in these early trials, regardless of their medication status.
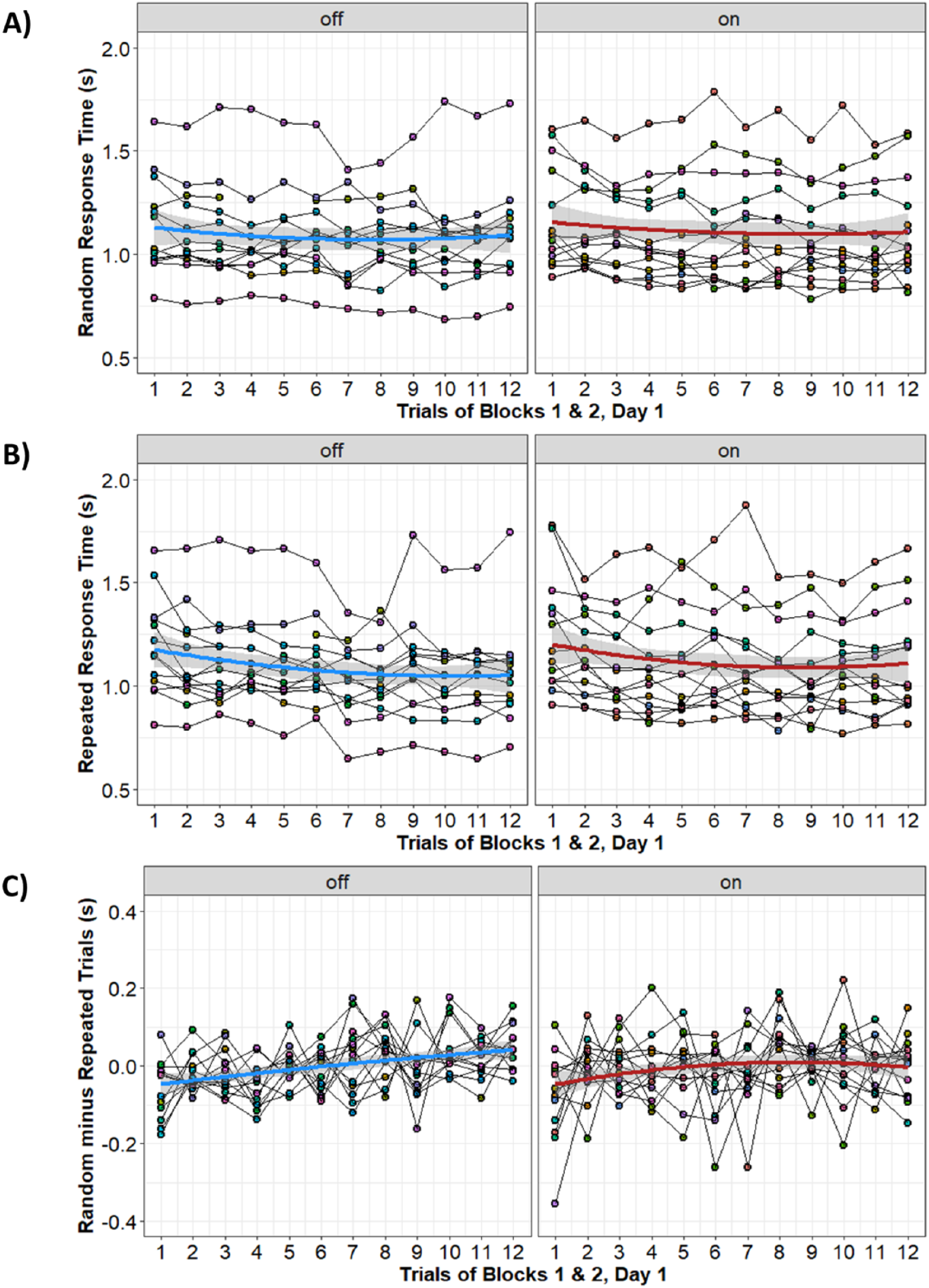
Individual learning curves during early acquisition for participants “on” and “off” dopamine replacement medication. (A) Response time for the random sequences; lower values on the y-axis denote better performance. (B) Response time for the repeated sequence; lower values on the y-axis denote better performance. (C) Random minus repeated response time (ie, sequence-specific learning); higher values on the y-axis denote better performance. Colored lines show the quadratic line of best fit in each group, and the shaded area represents the 95% CI for the best-fit line. Data points and thin lines show the response times for individual participants.
Discussion
Using a randomized clinical trial design, we examined the effects of practice and medication status on postural motor learning in people with mild to moderate PD. Exogenous dopamine did not appear to affect implicit learning because both groups (“on” and “off” levodopa medication) demonstrated similar gains in general and sequence-specific learning and retention of the postural task following practice. Our findings add to the evidence that people with PD are capable of learning motor skills10,12 despite potential dopamine replacement-mediated impairments. 16 However, they raise questions regarding the generalizability of the acquired skill.
A Close Look at Dopamine Replacement Effects
Systemic delivery of exogenous dopamine has been suggested to overdose and suppress activation of the associative striatum, impairing motor learning. 16 Prior evidence on the other hand suggested that lack of exogenous dopamine impairs learning of novel tasks performed with the upper limb 34 and impedes effective practice of tasks previously learned “on” levodopa medication.35,36 Such work motivated the design of this study using rigorous controls for threats to internal validity, examination of 2 types of learning, assessments of transfer, and examination of potential differences in early acquisition. Our results, however, show no consistent impact of levodopa on learning of a postural stepping task, transfer of performance to untrained balance tasks, or influence on learning curves in early acquisition. The small between-group effect sizes result in our findings being underpowered but are consistent with recent studies using a postural tracking task, 13 a reactive stepping task, 27 and a nonmotor task, 37 adding to evidence that suggests that any adverse effects of dopamine on learning interacts with a variety of factors, including but not limited to task type, the presence of explicit knowledge of what is being learned, the timing of measurements, and participant characteristics. Given the striatal changes associated with PD progression, it is likely that the negative impacts of exogenous dopamine on motor learning tend to occur only in very early stages of PD.15,16 People with early PD are most commonly treated with enzymatic inhibitors or dopamine agonist medications instead of dopamine replacement medications. Given that one of our inclusion criteria was that participants were taking dopamine replacement, our participants had mostly mild to moderate disease (mean 5 years postdiagnosis, r = 0.44 between disease duration and levodopa equivalent dose [LED]). In these individuals, it may be that any effects of exogenous dopamine overdosing the associative striatum were minimal. We also investigated implicit motor sequence learning, which appears less affected by levodopa 34 than explicit motor sequence learning. 38
Persistence of Learning and Clinical Implications
Similar to people with cognitive impairment who demonstrated retention following 1 month of no practice of a novel upper limb task 39 but unlike other short-13,40 and longer-term 41 motor learning paradigms in people with PD, we demonstrated that performance gains from practice were maintained up to 9 days following no practice in both groups. These finding are also similar to a recent upper limb motor learning paradigm of intensive writing training, which demonstrated that people with PD can retain skills up to 6 weeks posttraining. 42 Despite no observable between-group difference caused by levodopa medication, both groups demonstrated similar amounts of learning of the stepping task, with gains retained up to 9 days following withdrawal of practice. Although levodopa’s positive effects on tremor and bradykinesia are well understood, its impact on gait and postural stability are more variable, 5 which may partially explain the null between-group results of our outcomes. Interestingly, Peterson and Horak 27 demonstrated greater learning of a reactive postural task (which involves primarily nondopaminergic pathways) in people with PD who practiced “on” levodopa compared with those who practiced “off.” Such findings suggest that the motoric benefits of levodopa may augment postural motor learning, possibly by alleviating nonmotor symptoms 43 that can negatively influence learning (eg, fatigue or frustration).
The improvements in general and sequence-specific learning following practice demonstrated in our study are consistent with emerging evidence demonstrating that people with PD are capable of learning and retaining new upper limb and postural motor skills through practice.27,42 Such findings underscore the importance of delivering rehabilitation to people with PD to enable them to improve and maintain function, 44 particularly exercise interventions aimed at improving balance, which may potentially reduce falls.2,9
Limitations and Future Directions
Despite using rigorous methodological controls, the interpretation of our findings may have been affected by factors such as our sample size and the interaction of task characteristics with our participants. Our sample size and design resulted in variability that may have obscured between-group differences. It is important to note that our between-group effect size was substantially smaller than that observed in prior studies.17,27 One distinct difference between our study and those studies17,27 is our use of a between-subject design in contrast to within-subject designs where the same individual is tested both “on” and “off” medications. The extent to which these design features influence the results is not clear but our results provide a more conservative estimate of the effect of dopamine replacement on postural motor learning. Task difficulty may have influenced the extent of learning in our study. Unlike a non-stepping postural tracking task suggested by the authors to be inherently too difficult for people with PD to learn, 13 our postural stepping task may not have challenged some participants enough, causing a ceiling effect which could have limited further improvements with practice. This potential physiological ceiling effect has been noted previously in another study that used a similar stepping task in a different training context (mean step time 1090-1097 ms, 29 comparable to 1009-1014 ms in our study).
We also did not withdraw participants in the “off” medication group from all dopamine replacement medications. This was a pragmatic decision owing to the multiple consecutive days of practice and the long half-life of some dopamine agonists that would require participants to take no antiparkinsonian medication for multiple days. Five of the 13 participants in the “off” training group were taking other antiparkinsonian medication, with the LED from these medications accounting for a mean of 39% (SD = 14%) of their total LED. Yet participants’ change in “on” versus “off” medication MDS-UPDRS motor scores exceeded the standard error of measurement, 23 indicating that participants who practiced “off” medication were functionally “off.”
The theoretical rationale behind the dopamine overdose hypothesis also suggests that difficulties learning “on” compared with “off” levodopa may be more pronounced during early and/or mild stages of PD where there is a differential effect on the ventral versus dorsal striatum. 16 Our study included a mix of people in mild to moderate disease stages, which may have confounded the findings. Although falls are more prevalent with increasing PD severity, 45 emerging evidence reveals that de novo individuals may already experience postural instability 6 and falls 7 concomitant with greater disease severity. 6 Along with the longer term practice required for people with PD to benefit from rehabilitation programs targeting balance and falls prevention, 9 our results and those from other postural motor learning paradigms13,27,36 suggest that the potential impact of the dopamine overdose hypothesis on the early acquisition phase may not be very important for postural motor learning. Future studies should examine postural motor learning in varied PD subgroups (eg, de novo individuals, motor phenotype, freezing of gait status). 9
Conclusion
Levodopa medication did not appear to affect postural motor learning of a stepping task in people with mild to moderate PD. Both groups demonstrated similar gains in general and sequence-specific learning with practice, demonstrating that people with PD have the capacity to learn and retain a novel task with practice. The negative effects of practicing “off” levodopa medication—for example, increased motoric effort to overcome motor and nonmotor symptoms such as bradykinesia, hypokinesia, and fatigue—combined with the lack of a differential effect of levodopa medication on postural learning from this and other studies13,27,36 suggests that balance training should be delivered in the “on” medication state to people with PD. Further work is required to determine task difficulty and the practice doses required to derive functional improvements in balance performance and reductions in falls in this population.
Footnotes
Acknowledgements
The authors wish to thank Amy Ballard, Alicia Dibble, Kirsten Gorski, Jaclyn Hill, Anna Lundgren, Orin Ryan, Jane Saviers-Steiger, Dylan Wile, and Marianne Wilson for their assistance with data collection.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by an American Parkinson Disease Association Postdoctoral Fellowship Grant (2015-2017) and a University of Utah Office of Research seed grant (2015-2016).