Abstract
Background. Rhythmic auditory stimulation (RAS) can influence movement during straight line walking and direction transition in individuals with Parkinson disease (PD). Objective. The authors studied whether multidirectional step training with RAS would generalize to functional gait conditions used in daily activities and balance. Methods. In a matched-pairs design, 8 patients practiced externally paced (EP) stepping (RAS group), and 8 patients practiced internally paced (IP) stepping (no RAS group) for 6 weeks. Participants were evaluated on the first and last days of practice, and 1 week, 4 weeks, and 8 weeks after practice termination. Evaluations included a primary measurement—the Dynamic Gait Index (DGI)—and secondary measurements—the Unified Parkinson’s Disease Rating Scale (UPDRS), Tinetti-gait and balance tests, Timed-Up-and-Go (TUG), and Freezing of Gait Questionnaire (FOGQ). Results. The RAS group significantly improved performance on the DGI and several secondary measures, and they maintained improvements for the DGI, Tinetti, FOGQ, and balance and gait items of the UPDRS above pretraining values at least 4 weeks after practice termination. The no RAS group revealed several improvements with training but could not maintain these improvements for as long as the other group. Conclusions. Individuals with PD can generalize motor improvements achieved during multidirectional step training to contexts of functional gait and balance. Training with RAS is advantageous for enhancing functional gait improvements and the maintenance of functional gait and balance improvements over 8 weeks.
Keywords
Introduction
Difficulty in movement transition is a fundamental motor deficit for individuals with Parkinson disease (PD). 1,2 These individuals experience problems especially when adaptations to the environment become necessary, such as when changing directions or clearing obstacles. 3 This may explain why they have a 9 times greater risk of falling than age-matched controls. 4 They experience falls while turning 4 and freeze during gait initiation, 5 turning, 6 and performing dual tasks. 5
Although rates of motor learning in PD patients can be slower than those in age-matched controls, 7 these individuals can acquire motor skills and improve their motor performance with rehabilitation that supplements medical treatments, using motor learning principles 8 such as repetitive practice. 9-11 Adopting the use of external cues, such as rhythmic auditory stimulation (RAS), also appears to enhance motor performance in PD. 12 Such use assumes that those with PD are able to generate planned movements, 1,8 which can be released with an external cue 13 by circumventing the use of the defective internal trigger originating in the basal ganglia (BG). 14 Although some studies reveal no link between training improvements in PD and the modality of external cue used, 15,16 auditory cueing was preferred when presented with auditory, visual, and tactile cueing options. 15,16 Moreover, auditory cueing was considered more effective than visual cueing in terms of its translation to motor output 17 and its effect on motor performance during challenging tasks (ie, dual task walking). 18
Auditory stimulation in rhythmic forms is ideally suited for cyclical movements such as gait. RAS directly alters the gait kinematics 19-21 of individuals with PD. A single-session application of RAS leads to increased gait velocity, 20 step length, 20 and cadence 19 and decreased double support time. 19 Several weeks of practice with RAS also results in reduced variability of leg muscle activity patterns 22 and increased gait velocity, 21 stride length, 21 and cadence 23 during straight line walking but does not change the number of freezing episodes. 24
People with PD can alter and/or improve performances of noncued gait, 25 noncued gait and balance, 15 activities of daily living (ADL), 26 and the amount of time spent walking 16 after interventions using visual, tactile, and auditory external cueing paradigms. Corresponding improvements, using a multiple cueing paradigm, were maintained for 6 weeks after training for 6 weeks, 26 but only walking speed and step length of noncued gait were retained at 6 weeks after 3 weeks of cued training, which involved all 3 sensory modalities in the first week followed by 2 weeks of the preferred auditory or tactile cues. 25 Improvements in gait and balance items of the UPDRS 15 and walking time 16 were not maintained for 6 weeks, suggesting limits on the preservation of performance outcomes after training with various external cues.
One way of looking at successful training outcomes or motor learning ability is in terms of acquiring new skills. Another way of doing this, which is the emphasis of the present work, is in terms of the ability to transfer what is learned in one context to a different context. We reasoned that notable functional gains would result from challenging PD patients through bilateral movement alterations 27 and direction changes 28 during training, and these gains would transfer to notable improvements in untrained functional activities. Thus, the goals of this study were to determine if multidirectional step training with or without RAS for 6 weeks would improve functional gait and balance of people with PD and if any improvements beyond pretraining values would be maintained for 1, 4, and 8 weeks after training terminated.
Methods
Participants
A total of 20 individuals with PD were screened to take part in this study. Volunteers were recruited via local advertisement through a PD support group, retirement community, and neuromedical center. Of these, 4 patients did not meet criteria. Participants signed informed consent approved by the university’s internal review board. Inclusion criteria were diagnosis of idiopathic PD, Hoehn and Yahr (H&Y) stage 2 to 4, stable drug use, ability to stand independently and walk with or without an assistive device, and ability to differentiate auditory cues. The modified H&Y Scale 29 was administered for disease severity alongside the Unified Parkinson’s Disease Rating Scale (UPDRS) 30 for categorizing disability. Participants were excluded for a Mini-Mental State Examination Score (MMSE) < 24, other disorders that could potentially influence balance and walking, and off periods indicated by items 37 (score = 1) and 39 (score > 2) of the UPDRS. Participants satisfying enrollment criteria were also evaluated on the Dynamic Gait Index (DGI) and Timed-Up-and-Go (TUG) during the screening visit for test–retest reliability assessments of these measurements.
Study Design
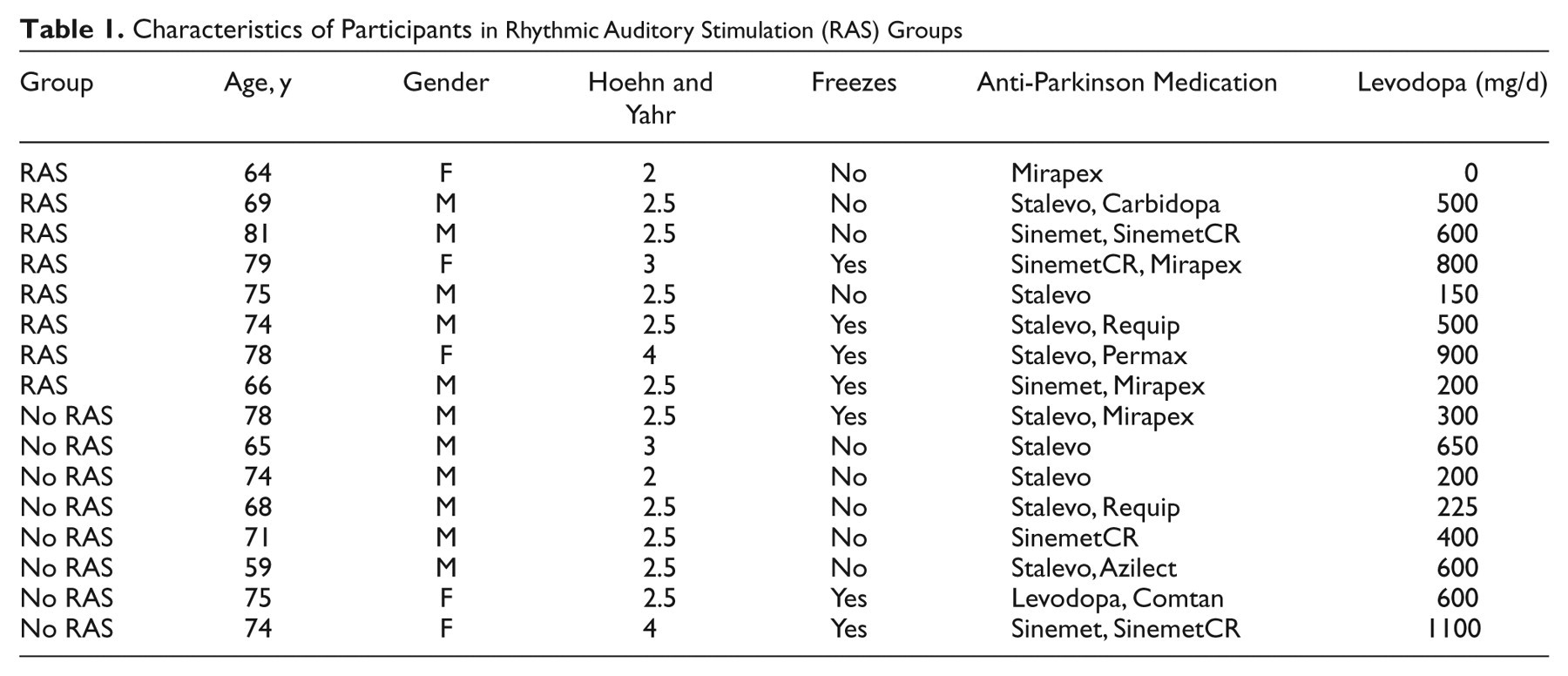
Participants were assigned to either a group that performed externally paced (EP) stepping to RAS (RAS group) or to a No RAS group that performed self-selected, internally paced (IP) stepping. Once disease stage was determined, the individual was randomly assigned to a group. The next participant with the same disease stage was assigned to the opposite group to maintain the same distribution of disease severity between groups. Pilot data from 5 people with PD not included in this trial were used in a within-subject analysis to indicate that 6 individuals per group were needed to achieve 80% power on our primary measure, the DGI. Participant and group information are included in Table 1.
Characteristics of Participants in Rhythmic Auditory Stimulation (RAS) Groups
Comfortable stepping cadence was also determined during the screening visit. Forward stepping cadence of 5 trials, each lasting 10 s, was averaged to establish normal step cadence and RAS frequencies.
Multidirectional Step Training
Participants trained 3 times per week (45-60 min/session) for 6 weeks. All tests were conducted in the same location and included a pretest on day 1 of training (PRE), a posttest on the last day of training (POST), and follow-up post tests, 1 week (POST+1), 4 weeks (POST+4), and 8 weeks (POST+8) after training terminated. Participants were encouraged to continue with normal daily activities from screening to POST+8 periods but received no step training after POST.
A physical therapist, certified in UPDRS administration by Movement Disorders Neurologists and experienced in working with PD patients, evaluated and trained all participants and thus was not blinded to the group assignment. Each participant attended training and testing sessions 1 hour after medication intake at the same time of day. Medication intake was confirmed before each session; 3 participants had to postpone and repeat 1 session each as a result of off periods or improper medication intake.
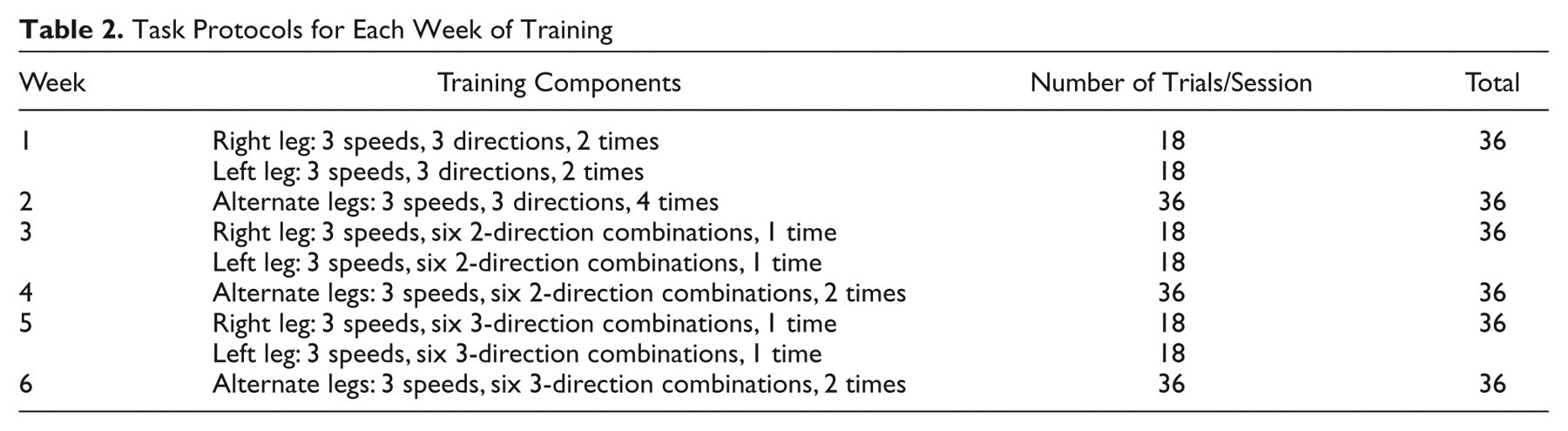
The participants in the RAS group started with their feet in the anatomic position at a self-selected distance apart. They stepped away and back in time with each beat of the RAS presented at 1 of 3 speeds: normal/comfortable cadence and 10% to 20% faster and slower than normal cadence according to individual preference/ability within the 10% to 20% range. Auditory cues used for the RAS group’s forward (F), side (S), and back (B) stepping included cluck, ding, and soft cork sounds recorded at a 22.5 kHz with durations of 88 ms, 915 ms, and 127 ms, respectively. Sound volumes were approximately 75 dB, well above average hearing loss range (25-40 dB) reported for older adults. 31 Participants acknowledged cues on presentation and could describe each. The total trial duration of 33.75 s was relatively short (about 30 s) to avoid fatigue in this population. 32 Participants were instructed to step in time with the beat and that the foot should completely leave the surface and then make complete contact with the floor for each step. Speed remained constant within trials. Demonstrations and/or instructions were repeated every session before practice/testing, after breaks, and when requested. Our training protocol was based on principles recommended to enhance motor learning in PD patients and involved the following: (1) repetitive task practice, 9,10 (2) simple to more difficult task progression, 33 (3) breaking a sequential task into its components, 34 and (4) long-term randomized practice. 9,11 Because of the challenges involved in bilateral alternating movements 27 and changing directions 28 for this population, training increased in complexity based on the weekly training protocol presented in Table 2. The table shows that 36 trials were performed in each training session. Each speed/direction combination was performed during weeks 1 to 2 for 33.75 s. Six 2-direction combinations (B→F, B→S, F→S, F→B, S→F, and S→B) were performed during weeks 3 to 4, allowing 11.25 s for the first direction and 22.5 s for the second direction. Six 3-direction combinations (F→S→B, F→B→S, B→S→F, B→F→S, S→F→B, and S→B→F) were performed during weeks 5 to 6, allowing 11.25 s for each direction. Participants stepped with each leg separately on weeks 1, 3, and 5 and alternated legs for weeks 2, 4, and 6. Direction combinations were randomized within each training day and for each leg separately on odd weeks. Four participants repeated 1 to 5 trials for not adhering to instructions. Note, the odd duration was because of limitations in software used to present tones for the RAS group. Sectioning a 45-s total time period allowed us to present different tones for the first 3 sections (the fourth was muted) needed for signaling direction switching while minimizing intratrial fatigue in PD patients (approximately 30 s 32 ). The smallest duration/direction of 11.25 s also allowed several steps in a given direction/trial even at the slow speed.
Task Protocols for Each Week of Training
Step training, number of trials, and setup for the no RAS group were similar to that for the RAS group with the following exceptions. Participants were instructed to perform the task for each direction at a “comfortable” pace or at a “relatively faster” or “relatively slower” pace than normal and to keep speed constant within a speed category. They were given a “last step” verbal cue approximately 1 s before a direction switch and a “stop” cue to end the trial to maintain the desired protocol and lessen the potential cognitive load. Three participants repeated 3 to 5 trials for not adhering to instructions.
Evaluation
Several functional measures were evaluated to offer a comprehensive evaluation of the gait and postural difficulties associated with PD not provided by a single measurement 35 and to gain insight into the improvements participants experienced after training. The DGI—a fall risk indicator 35 that challenges participants to perform simple-to-difficult gait patterns requiring straight line walking, obstacle avoidance, head movements, and stair use 36 —was selected as the primary measure. Secondary measures included the UPDRS-motor and UPDRS-ADL sections to evaluate overall motor symptoms of PD and ADL, respectively. Similar to previous work, 15 the UPDRS-composite score, involving UPDRS gait and balance items linked to PD disability (13-15, and 29-30), was also included. Evaluations of the Tinetti (gait and balance) also identified general impairment of gait and balance 4 and included some items that did not overlap with previous tests, such as sitting down, turning 360°, standing with eyes closed, and evaluation of trunk sway and left and right symmetry during gait. The TUG test evaluated the time to stand from a chair; walk 3 m; and turn, walk back, and sit down—that is, the temporal aspects of gait. 37 The Freezing of Gait Questionnaire (FOGQ) assessed perceived changes in gait and the quality and frequency of freezing episodes. 38 Measurement order was randomized within evaluation sessions.
Statistical Analyses
Intraclass correlations were used to determine test–retest reliability on the DGI, TUG, and UPDRS measurements collected during screening and PRE visits. Between-group differences on age, height, body mass, MMSE scores, disease duration, step cadence, and PRE functional measures were determined using t tests for independent samples.
Repeated-measures ANOVAs with between-subject factor of Group (RAS, No RAS) and repeated measures of Test-day (PRE, POST, POST+1, POST+4, and POST+8) were used to compare measurement differences between groups and among test days. A Kenward Rogers adjusted degrees of freedom was used because it accounts for the relatively small sample size and missing test values (Figure 1) while offering good control over a type I error risk. 39 Tukey post hoc tests were used when appropriate to answer the experimental questions via PRE/POST test comparisons within each group: PRE/POST comparisons for transfer abilities and PRE/POST+1, PRE/POST+4, and PRE/POST+8 comparisons for abilities to maintain improvements over baseline. RAS versus no RAS comparisons for each test day offered between-group comparisons for RAS effects on transfer abilities and abilities to maintain improvements over baseline. It is important to note that analyses account for the missing participants for POST+4 and POST+8 by omitting these values and using all available data points (PROC MIXED design, SAS Inc, Cary, North Carolina; see Figure 1 for number of participants on each test). Significance level was preset at P < .05 for all analyses. The DGI effect size (Cohen d) for PRE/POST comparisons for each group was also computed to estimate the effect of training.
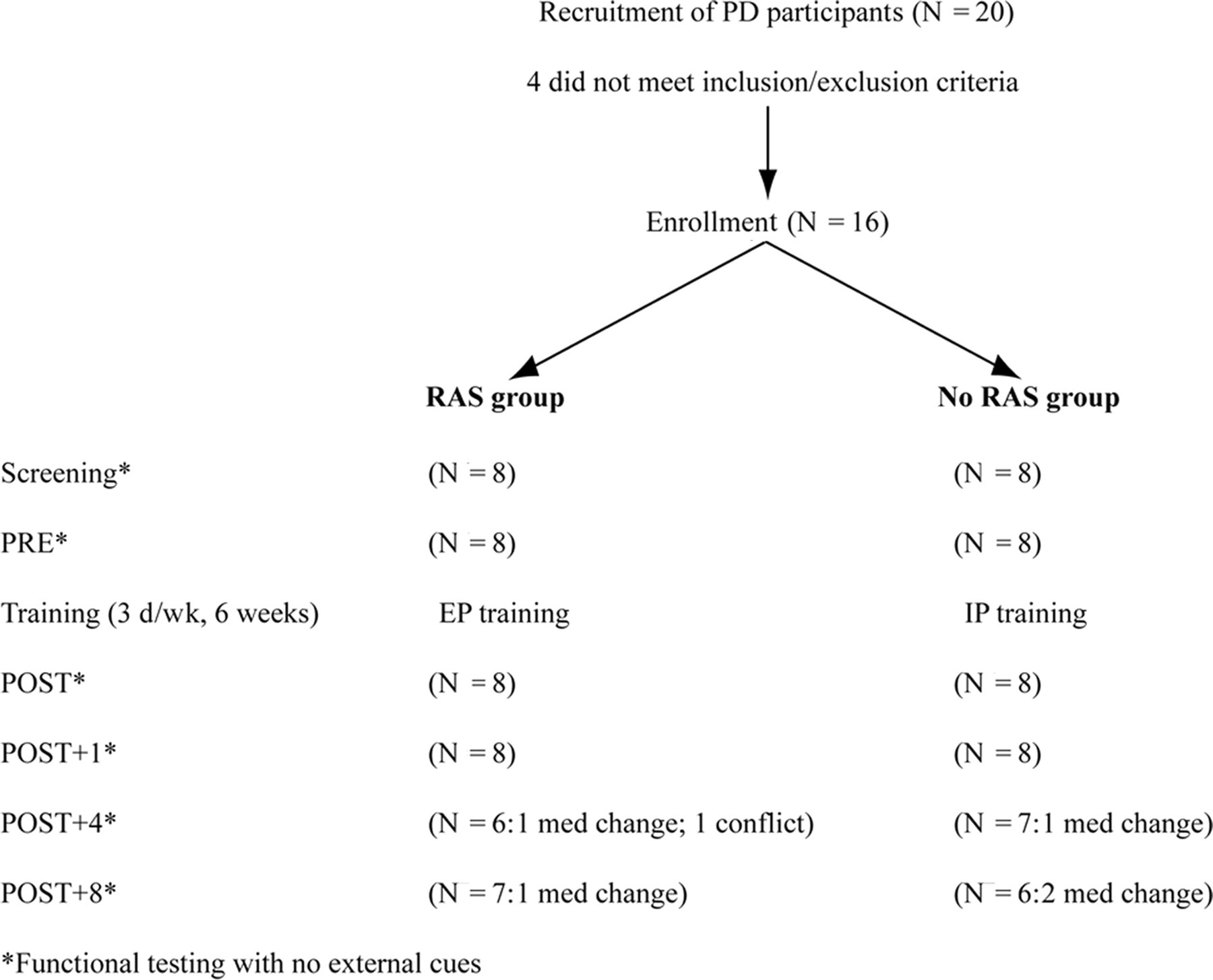
The schematic of the number of participants (N) at various stages of the study is shown. Testing was performed before training (PRE) and on the last day of training (POST) and 1 week (POST+1), 4 weeks (POST+4), and 8 weeks (POST+8) after training termination. Reasons for reduced numbers are provided: med change refers to change in medication, and conflict refers to scheduling conflict that was not made up. Abbreviations: RAS, rhythmic auditory stimulation; EP, externally paced; IP, internally paced.
Results
Similarities in the measurements identified between screening and PRE visits suggest good test–retest reliability on the DGI, TUG, and UPDRS measures (intraclass correlation coefficient [ICC] ≥ 0.99). Table 3 shows that the 2 groups did not differ in age, height, mass, disease duration, levodopa dosage, mental status, or step cadence and had similar functional scores at baseline. Note that comfortable step cadence, which involved stepping forward and back, thus, requiring continuous changes in direction, was much lower than comfortable walking cadence presented elsewhere. 15,40,41 The number of training trials (36 trials/d × 18 days = 648 trials) and the time spent training (33.75 s/trial × 648 trials/60 s = 364.5 minutes) in the present work was within the range of other repetitive practice studies for this population (30 trials, 5 minutes 10 and 3600-4600 trials, 400 minutes 9 ). A moderate disease stage (2.5 H&Y) was most common (Table 1). In all, 7 participants (4 RAS and 3 no RAS) experienced freezing at least once per week. 15 Figure 1 depicts the number of participants at each stage of the study and shows that all participants completed training; yet not all completed follow-up evaluations because of scheduling conflicts or a medication change.
Mean ± 1 Standard Error for Measures of the RAS and No RAS Groups Recorded on the First Day of Practice
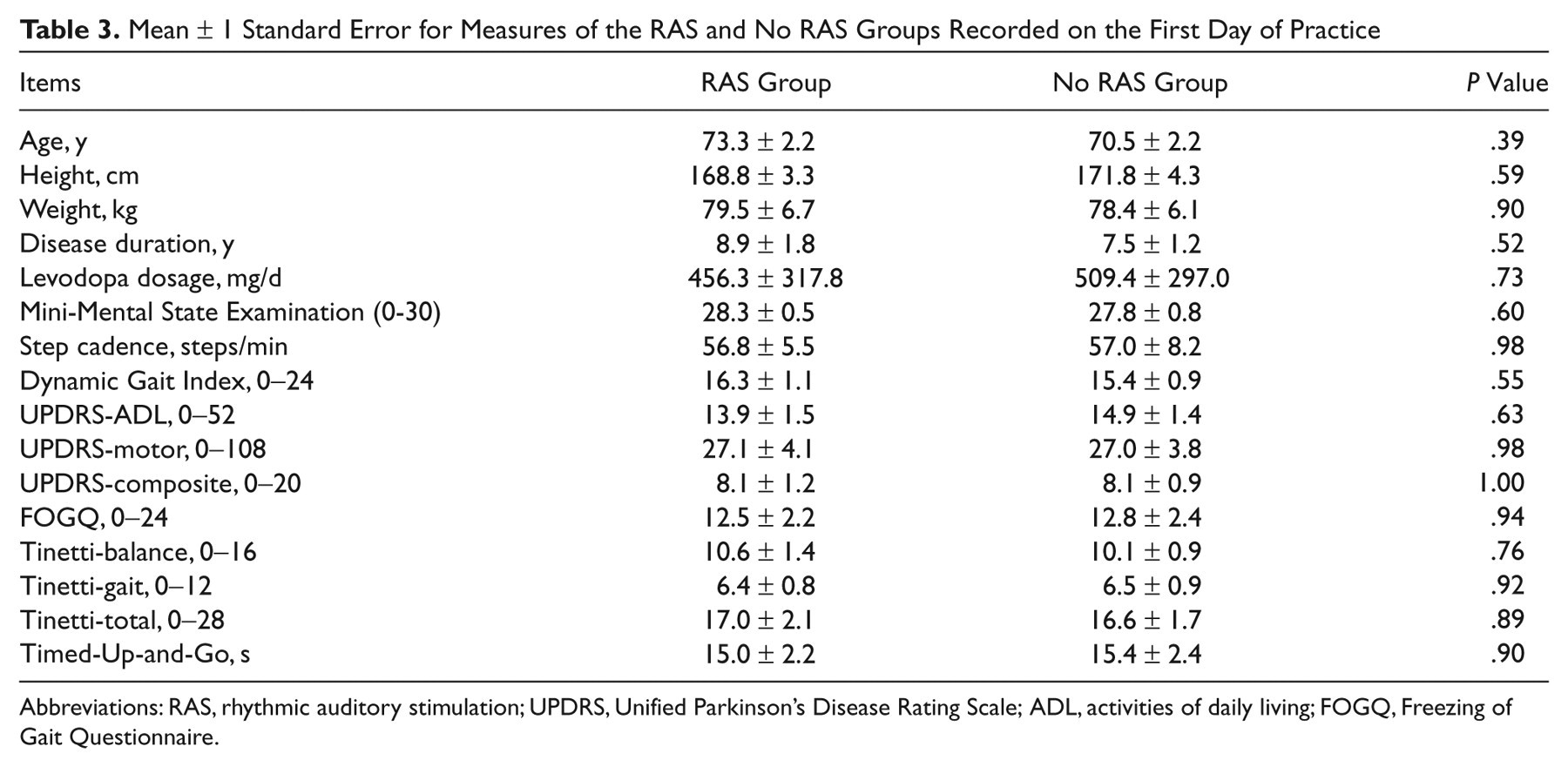
Abbreviations: RAS, rhythmic auditory stimulation; UPDRS, Unified Parkinson’s Disease Rating Scale; ADL, activities of daily living; FOGQ, Freezing of Gait Questionnaire.
The key results of the study indicated that for the primary measure—the DGI—there was a significant Group × Test-day interaction (F 4,10.8 = 13.99; P < .001) in addition to the main effects of Group (F 4,16 = 12.58; P < .01) and Test-day (F 4,10.8 = 27.20; P < .0001). Figure 2A presents the interaction and emphasizes the following findings. DGI scores for both groups improved on POST evaluation (see blue and red asterisks for the RAS and No RAS groups, respectively) and reveal high effect sizes. Improvements above PRE scores were maintained for the no RAS group through POST+1 and for the RAS group through POST+8. Significantly higher improvements were observed for the RAS group compared with the no RAS group on POST, POST+1, and POST+4 evaluations (see curly brackets with black asterisks). A review of individual scores of patients revealed that 4 participants in the RAS group recorded scores greater than the 22-point fall risk cutoff score 42 at POST.
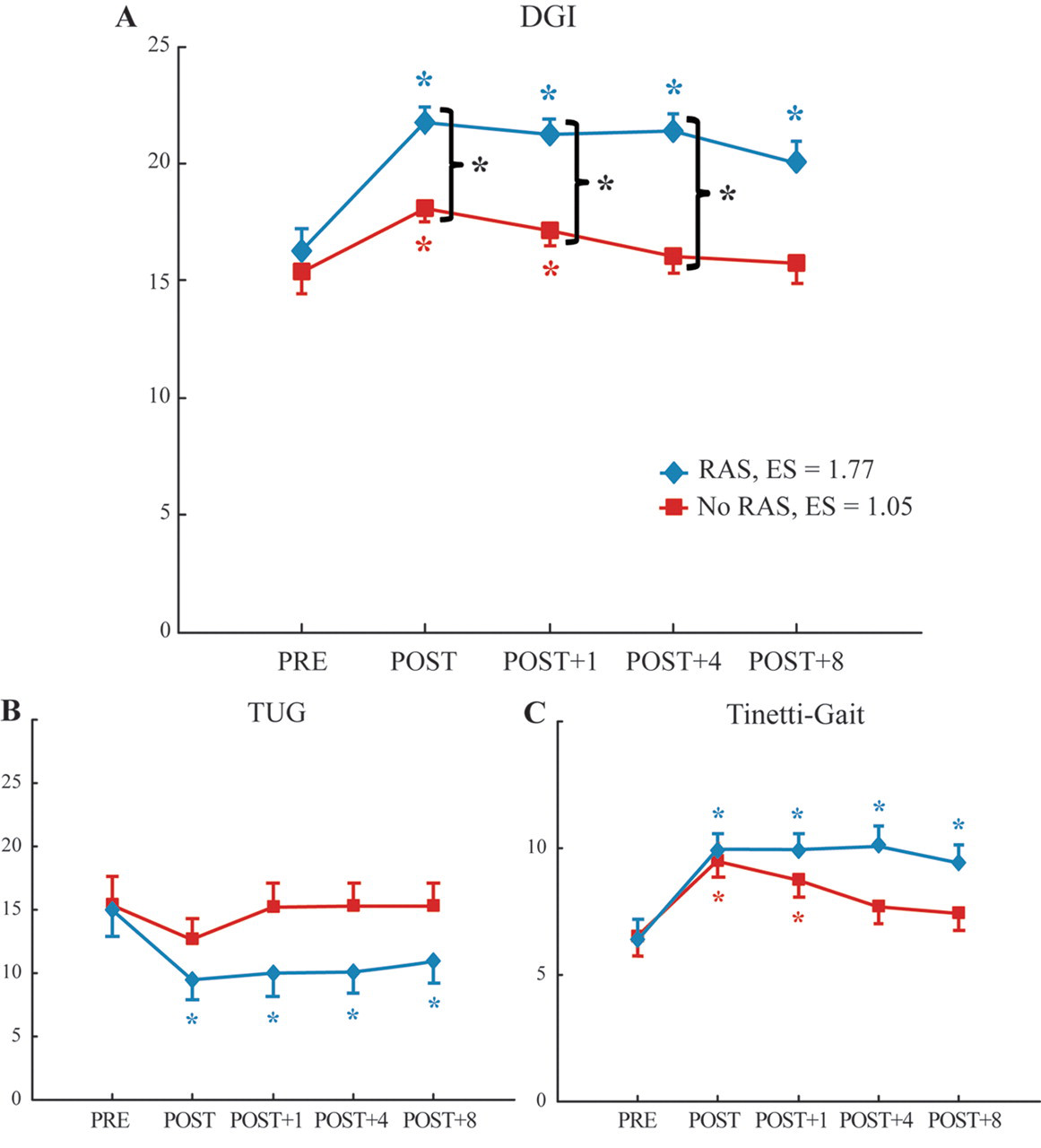
Mean scores of the (A) DGI, (B) TUG, and (C) Tinetti-gait for the RAS (blue) and no RAS (red) groups are shown. Scores are shown for pretest (PRE), posttest (POST), and 1 week (Post+1), 4 weeks (Post+4), and 8 weeks (Post+8) after training. Colored asterisks represent a significant difference from the PRE values for the corresponding group. Curly brackets with black asterisks indicate significant differences between RAS and no RAS groups. Error bars represent 1 standard error. Means and errors represent adjusted values where appropriate to account for missing data. Abbreviations: DGI, Dynamic Gait Index; TUG, Timed-Up-and-Go; RAS, rhythmic auditory stimulation; ES, effect size.
Outcomes from the secondary measures show some results that are similar to those reported for the DGI, excluding the main effect of Group (P > .05 for all secondary measures). Significant test-day effects and Group × Test-day interactions were identified for supporting functional gait measures (interaction for TUG, F 4,8.94 = 44.15, P < .0001; and Tinetti-gait, F 4,6.67 = 16.03, P < .01). The RAS group revealed improvements at POST with 4 participants from the RAS group and 1 participant from the no RAS group performing better than the 7.95-s fall risk cutoff on the TUG. 42 Participants were able to maintain improvements over PRE values through POST+8 for both measurements (see blue asterisks for TUG, Figure 2B, and Tinetti-gait, Figure 2C); however, the no RAS group only revealed significant POST training improvements on the Tinetti-gait (red asterisks, Figure 2C). Nonsignificant group differences on each test day for supporting gait measures suggest that the DGI was a more sensitive measure of functional gait in this study.
Although Test-day and interaction effects did not reach significance for the UPDRS-motor (.05 < P < .10), effects of Test-day were identified for the UPDRS-ADL (F 4,9.8 = 6.63; P < .01) and UPDRS-composite (F 4,10.2 = 11.46; P < .001) to indicate significant improvements over PRE values for both groups. Similar to results of cued therapy, 26 the improvements in the UPDRS-ADL also exceeded the 2-point minimum suggested for clinically significant findings. 43 A significant Group × Test-day interaction for the UPDRS-composite (F 4,10.2 = 3.87; P < .05) reveals that the RAS group maintained gait and balance improvements over baseline through POST+4, whereas the no RAS group only improved at POST for this measure (Figure 3A). A significant Test-day effect (F 4,11.6 = 8.03; P < .01) and Group × Test-day interaction (F 4,11.6 = 3.85; P < .05) observed for the Tinetti-balance evaluation reveals that significant balance improvements after training were also maintained at POST+8 only for the RAS group (Figure 3B). RAS clearly resulted in maintaining better functional gait and balance scores compared with training without it. Finally, a significant Group × Test-day interaction (F 4,8.94 = 5.99; P < .05) and main effect of Test-day (F 4,8.94 = 20.42; P < .001) were identified for the FOGQ. Figure 3C shows that FOGQ scores for the no RAS group did not change significantly despite the same levels of disease severity in those who experienced freezing in the 2 groups. Those who experienced freezing in the RAS group received FOGQ scores of 16 to 22 before training and 10 to 14 after training, whereas scores of their counterparts in the no RAS group only dropped from 15 to 20 before training, to 13 to 19 after training. Thus, unlike the RAS group, the no RAS group did not perceive meaningful changes in gait or freezing episodes with training.
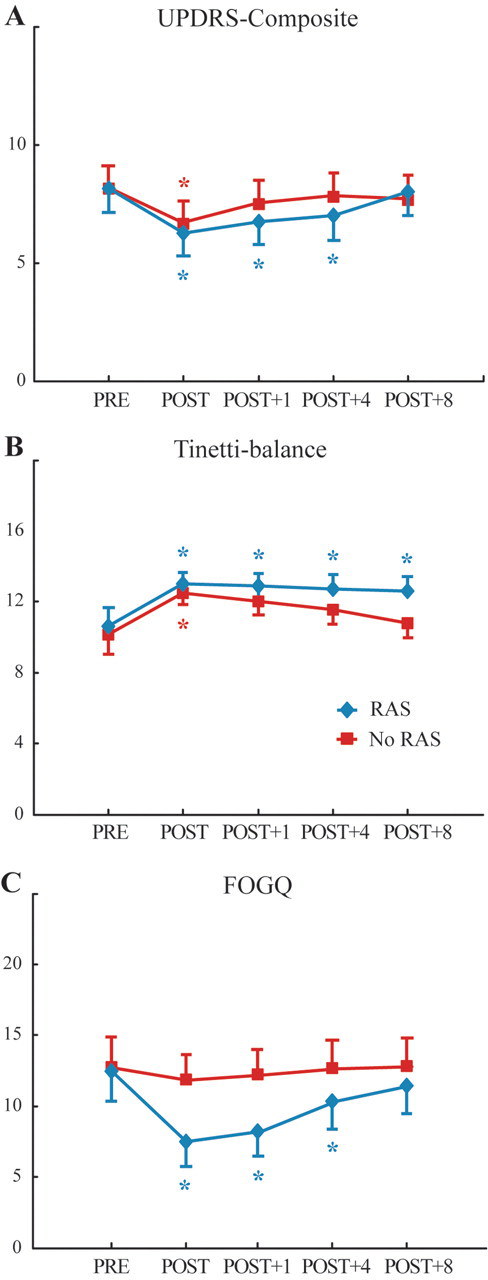
Mean scores of the (A) UPDRS-composite, (B) Tinetti-balance, and (C) FOGQ for the RAS (blue) and no RAS (red) groups. Scores are shown for the pre-test (PRE), posttest (POST), and 1 week (Post+1), 4 weeks (Post+4), and 8 weeks (Post+8) after training. Colored asterisks represent a significant difference from the PRE values for the corresponding group. Error bars represent 1 standard error. Means and errors represent adjusted values where appropriate to account for missing data. Abbreviations: UPDRS, Unified Parkinson’s Disease Rating Scale; FOGQ, Freezing of Gait Questionnaire.
Discussion
We found that the 18 sessions of multidirectional step training improved the DGI scores and other functional measures for individuals with PD, which demonstrates that step training transfers to a variety of functional gait and balance tasks. Performance improvements on the DGI noted immediately after 6 weeks of training (POST) were significantly greater for the RAS group compared with the no RAS group. Such performance differences between groups were not identified as significant on secondary measurements. However, participants in the RAS group were able to maintain various motor improvements involving gait and balance relative to baseline as indicated on the DGI and several secondary measurements. These findings support other studies that show that PD training programs improve function and highlight the additional benefits of RAS use to achieve or maintain improvements in gait and balance.
The present results support evidence that motor learning abilities are preserved in people with PD. 8-10,44 Functional gait and balance improvements after participation in a challenging repetitive step training protocol that did not involve walking indicate that participants transferred multidirectional step training effects to the different contexts of performing simple to complex gait patterns, temporal aspects of gait, and balance abilities related to steadiness during standing, sitting, and turning (compare PRE and POST tests in Figures 2, 3A, and 3B). The ability of PD patients to generalize 8 weeks of pedaling practice at rates 30% greater than self-selected rates to manual dexterity skills 45 also supports this view and suggests that forced exercise, which requires challenging different aspects of movement, can promote global functional improvements in PD. 45
Researchers recognize that immobility may be partially to blame for gait and balance deficits associated with PD. 46 Increased activity may help explain the improvements in function observed in the present study because 9/16 participants reported no commitments to regular physical activity before training. Although both groups revealed a large effect size for the DGI, the greater improvement in POST scores for the RAS group compared with the no RAS group (see Figure 2A) exemplify that step training with RAS offers additional benefits in functional gait performance. In addition, the RAS participants with lower disease severity were able to reduce their fall risk according to the DGI or TUG tests. 35
A major limitation of this study was that the evaluator was not blinded to group assignment. To reduce potential evaluator bias, scores were not tallied until all follow-up tests were complete. The sample size was relatively small and could not represent all stages of disease. Future studies should determine whether the protocol would benefit PD patients with greater disease severity or cognitive impairment.
We do show, however, that significant functional improvements relative to baseline were maintained longer for the RAS participants than the no RAS participants. Functional gait (Figure 2) and balance (Figure 3B) improvements over baseline were maintained for at least 8 weeks. These findings expand the limited research on the effects of external cueing on maintaining acquired skills. 15,16,25,26 In fact, the improvements in TUG scores for the RAS group that were still observed at POST+8 exceeded the pre–post improvements in median TUG scores after 3 weeks of cued gait and balance training. 15 Clearly, training duration and use of external cues, including RAS, can influence longevity of training effects on physical function and various gait parameters and, therefore, should be considered when long-term benefits are desired. RAS group participants identified perceptible differences in their freezing of gait, whereas the no RAS group did not (Figure 3C). These reductions exceed those of shorter past trials. 15,24
Based on the insights from this pilot trial, we conclude that further attempts to integrate RAS into PD training interventions may offer benefits over training without RAS to enhance functional outcomes in this population.
Footnotes
Acknowledgements
The authors would like to thank Drs Sue Leurgans and Lei Yu for statistical advice and the local Parkinson support group and all participants.
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: The research was partially funded by the Louisiana State University Graduate Dissertation Fellowship.
