Abstract
This systematic review and meta-analysis is to provide comprehensive evidence-based exercise recommendations targeting walking function for adults with Parkinson’s disease. Methods. Fixed- or random-effect meta-analyses estimated standardized effect sizes (Hedge’s g), comparing treatment effects from exercise with nonexercise and another form of exercise (non-EXE control and EXE control). Cuing and exercise duration were used as moderators for subanalyses. Results. The 40 included randomized controlled trials comprised 1656 patients. The exercise group showed significantly superior performance in timed up-and-go (g = −0.458; g = −0.390) compared with non-EXE control and EXE control; significantly greater improvement in comfortable walking speed (g = 0.449), fast walking speed (g = 0.430), and stride or step length (g = 0.379) compared with non-EXE control; and significantly greater cadence (g = 0.282) compared with EXE controls. No significant differences between intervention and control groups were observed for double-leg support time (DLST), dynamic gait index (DGI), 6-minute walk test, or freezing of gait questionnaire (FOG-Q). Notably, treatment effect from the exercise of interest compared with a standard exercise was greater than for nonexercise for cadence and FOG-Q. Moreover, EXE control was favored for DLST and DGI. Cuing had a significantly positive effect on stride length alone. Exercise duration significantly, but negatively, influenced the treatment effect on comfortable walking speed. Conclusion. Gait-specific training, rather than a general exercise program, should be emphasized if gait is the outcome of interest. Further investigation is needed on exercise dosage and its selective effect on more challenging walking tasks, endurance, and freezing of gait.
Introduction
Parkinson’s disease (PD) affects between 0.5% and 1.0% of adults aged 65 to 69 years, and 1% to 3% of those 80 years of age and older. 1 PD gait disturbance is produced by a combination of multiple factors, including bradykinesia, impaired postural reflexes, abnormal postures, rigidity, and tremor and characterized by a paucity of movement of the trunk, upper-body, and lower-body muscles, resulting in slow and shuffling steps that are short and rapid. Because of the shortened step length, the feet barely clear the ground, and the soles of the feet shuffle and scrape the floor. 2 Furthermore, freezing of gait in PD, one of the most disabling symptoms, is characterized by an inability to generate or sustain an effective stepping sequence, 3 which leads to sudden blocks in walking. 4 PD increases the risk of falling because of these gait disturbances.
Exercise has been widely used in allied health care for PD to address symptoms and improve motor functions,5,6 and the exercise mode includes balance and gait training, 7 resistance training, 8 treadmill training, 9 and complementary therapies. 10 However, the lack of comprehensive effective exercise guidelines, specifically focusing on gait function, makes it reasonable to evaluate the efficacy of a variety of exercise modalities and programs. The purpose of this systematic review and meta-analysis is to provide exercise recommendations (frequency, intensity, type, and time) for clinical program implementation by evaluating the training effect of exercise on gait function in adults with PD, thereby allowing a wider range of use and accessibility by patients and therapist.
Methods
Data Sources and Searches
The following databases were searched: MEDLINE (EBSCO), CINAHL Plus, PubMed, and SPORTDiscus to identify potential studies. Studies published from inception to June 2017 were included. The combinative keywords used for searching were the following: PD/Parkinsonism, exercise/physical therapy/physical activity/training, and gait/walking.
Inclusion Criteria
Inclusion criteria were a randomized control trial that included the following:
the effect of exercise interventions (include cuing strategies);
outcomes including gait/walking-related performance (gait speed, including comfortable and fast gait speeds; stride or step length; cadence; double support time [percentage of gait cycle]; timed up-and-go [TUG]; 6-minute walk test [6MWT]; freezing of gait questionnaire [FOG-Q]; and dynamic gait index [DGI]); and
articles available in English.
Exclusion Criteria
Exclusion criteria were as follows:
studies where the effects of a computer-based exercise intervention (such as computer games), walking-assist equipment (such as walkers or robotics), or exercise intervention with electrical/magnetic stimulation were evaluated and
studies where outcomes did not include gait assessments.
Methodological Quality
Information extracted from randomized controlled trials (RCTs) comprised a description of participants, details of the exercise, and details of outcome measures. The PEDro Scale was used to rate the methodological quality of the RCTs. Two independent investigators (MN and JBH) screened and scored RCTs. Interrater reliabilities for individual items of the PEDro Scale were calculated as Cohen’s κ. Differences of opinion on rating were resolved through discussion by the 2 investigators (MN and JBH). PEDro scores of 6 to 8 points were classified as “good quality,” 4 to 5 points as “fair quality,” and less than 4 points as “poor quality.” 11
Calculating Effect Size
Meta-analyses were conducted for 9 outcome measures. The primary outcome was comfortable walking speed. Other gait measurements included stride or step length, cadence, and double-leg support time (DLST; percentage of gait cycle) at comfortable walking speed, fast walking speed, TUG, DGI, 6MWT, and FOG-Q. The selected outcomes (gait speed, stride length, cadence, TUG, 6MWT, and FOG-Q) are the most frequently used measures in clinical settings and reported in published trails. The results of this article would be more clinically applicable. Meanwhile, double support time (percentage of gait cycle) and DGI provide more advanced gait analysis and comprehensive gait evaluation.
The standardized mean difference (Hedge’s g) was calculated for all meta-analyses. The effect size was calculated using the group mean difference of the change score between the pretest and posttest for each group and the pooled SD during patients’ “on” medication state. The group contrast was unbiased by the correction factor J = 1 − 3/(4 × [n1 + n2 − 2] − 1). Finally, the mean difference for the first assessment period was used for crossover studies.
Moderators
A univariate moderator model using categorical variables was conducted to assess the presence of cuing (yes/no or unclear) and estimates of the effect of cuing on gait-related performances. A univariate metaregression was conducted to assess the associations between the number of hours for the interventions in the trials. The metaregressions were only performed for those outcome measures where the studies included more than 10 trials to ensure sufficient statistical power.
Statistical Analysis
For all the outcome measures, the analysis was separated into 2 categories, exercise versus nonexercise control (non-EXE control), and exercise versus another form of exercise control (EXE control). Statistical heterogeneity was quantified using Q statistics. The overall fixed- or random-effect (restricted maximum likelihood) model was used based on the Q statistics. Publication bias for each outcome measure was assessed using Egger’s test. Post hoc analysis was used to examine the difference in the presence of cuing (yes/no or unclear) and the number of minutes of the interventions on the gait performance. The threshold for significance was set at P <.05. All data were analyzed using R Statistical Software 3.2.3.
Results
Identified Literature
Searching identified 669 RCTs, of which 47 were appropriate for inclusion and 40 were included in the meta-analysis (Supplement, PRISMA flow). A total of 1656 participants contributed to the studies reported in this meta-analysis. The minimal number of participants in a study was 14, and the maximum was 199. Among the included studies, 60.7% were male participants (1005/1656), and 2 studies did not report the sex.12,13 PD stage was described using Hoehn and Yahr’s disease stages (H&Y) in all trials except in 4 studies that used the UPDRS motor score. All studies included participants at H&Y stages from I to IV (Supplement Table 1).
Methodological Quality
PEDro scores ranged from 3 to 8 points (out of a maximum of 10 points), indicating low to high methodological quality (Supplement Table 2). The 2 reviewers who assessed the RCTs had 20 disagreements among the 470 ratings, yielding a Cohen’s κ of 0.914.The percentage of agreement was 91.4%, which suggested a “substantial” agreement between the 2 investigators. 14
Heterogeneity and Publication Bias
The test of heterogeneity indicated that the homogeneity of the population studied was not met for comfortable walking speed (non-EXE control: Q = 40.0, df = 20, P = .005; EXE control: Q = 38.0, df = 18, P = .004), stride or step length non-EXE control (Q = 38.1, df = 15, P < .001), and DGI non-EXE control (Q = 5.3, df = 1, P = .021). The homogeneity of the population was met for fast walking speed (non-EXE control: Q = 1.16, df = 3, P = .76; EXE control: Q = 3.3, df = 1, P = .07), stride or step length cadence EXE control (Q = 22.3, df = 15, P = .10), cadence (non-EXE control: Q = 5.9, df = 8, P = .663; EXE control: Q = 5.0, df = 11, P = .933), DLST (non-EXE control: Q = 1.1, df = 3, P = .770; EXE control: Q = 4.5, df = 7, P = .726), TUG (non-EXE control: Q = 10.0, df = 9, P = .354; EXE control: Q = 14.9, df = 13, P = .315), 6MWT (non-EXE control: Q = 2.1, df = 2, P = .354; EXE control: Q = 10.5, df = 8, P = .229), DGI EXE control (Q = 0.81, df = 1, P = .370), and FOG-Q (non-EXE control: Q = 2.7, df = 2, P = .259; EXE control: Q = 1.5, df = 1, P = .216). Egger’s test indicated that outcome measures were not significantly biased across all publications.
Outcomes
Comfortable Walking Speed
The effect size for exercise compared with non-EXE control was 0.449 with a standard error (SE) of 0.11 and was statistically significant (95% CI = 0.23 to 0.66, P < .001; Figure 1). The effect size for exercise compared to EXE control was 0.241 with a SE of 0.13 but not statistically significant (95% CI = −0.052 to 0.50, P = .065; Figure 2).
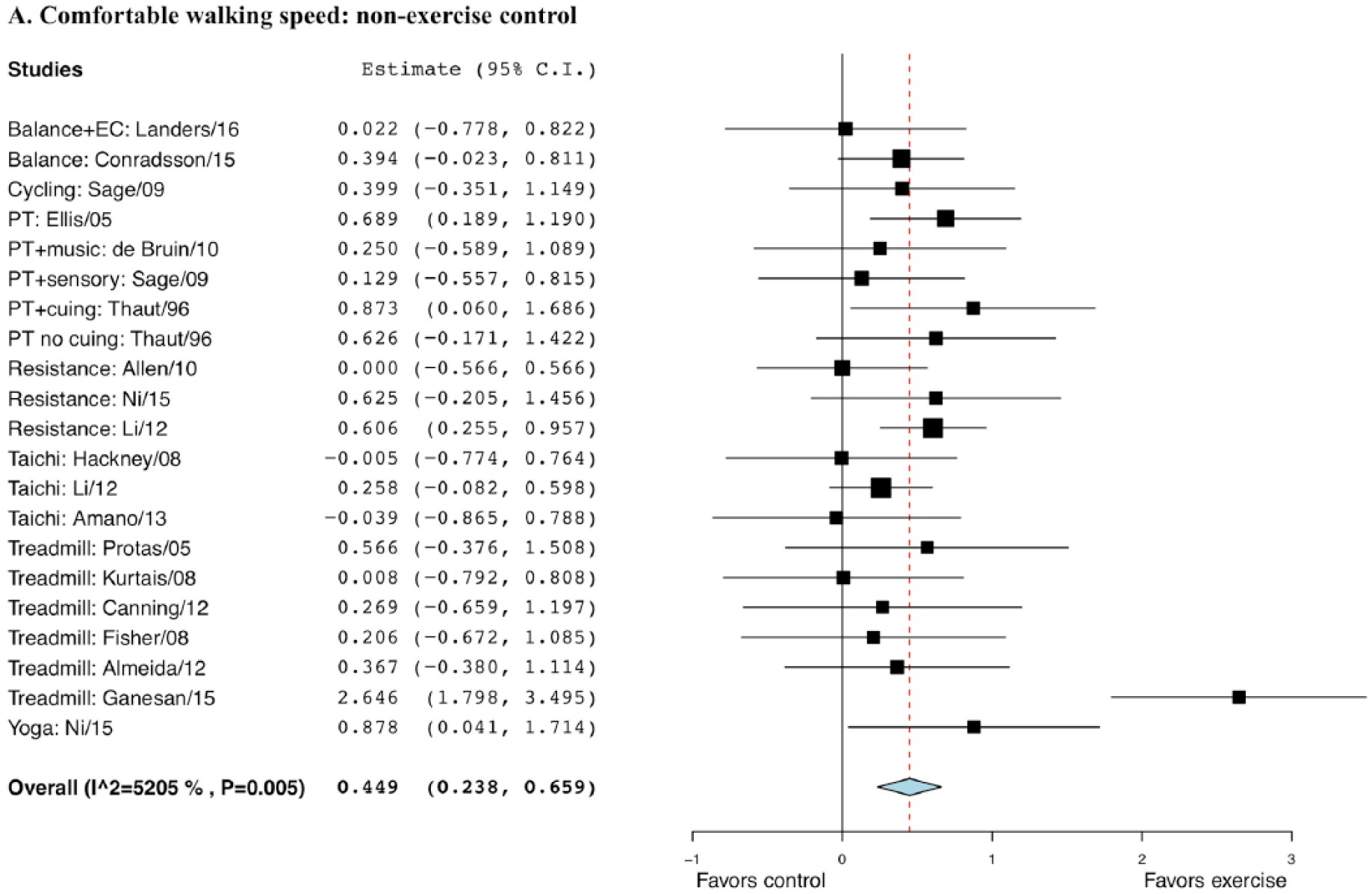
Forest plot of exercise effect on comfortable walking speed compared with nonexercise controls.
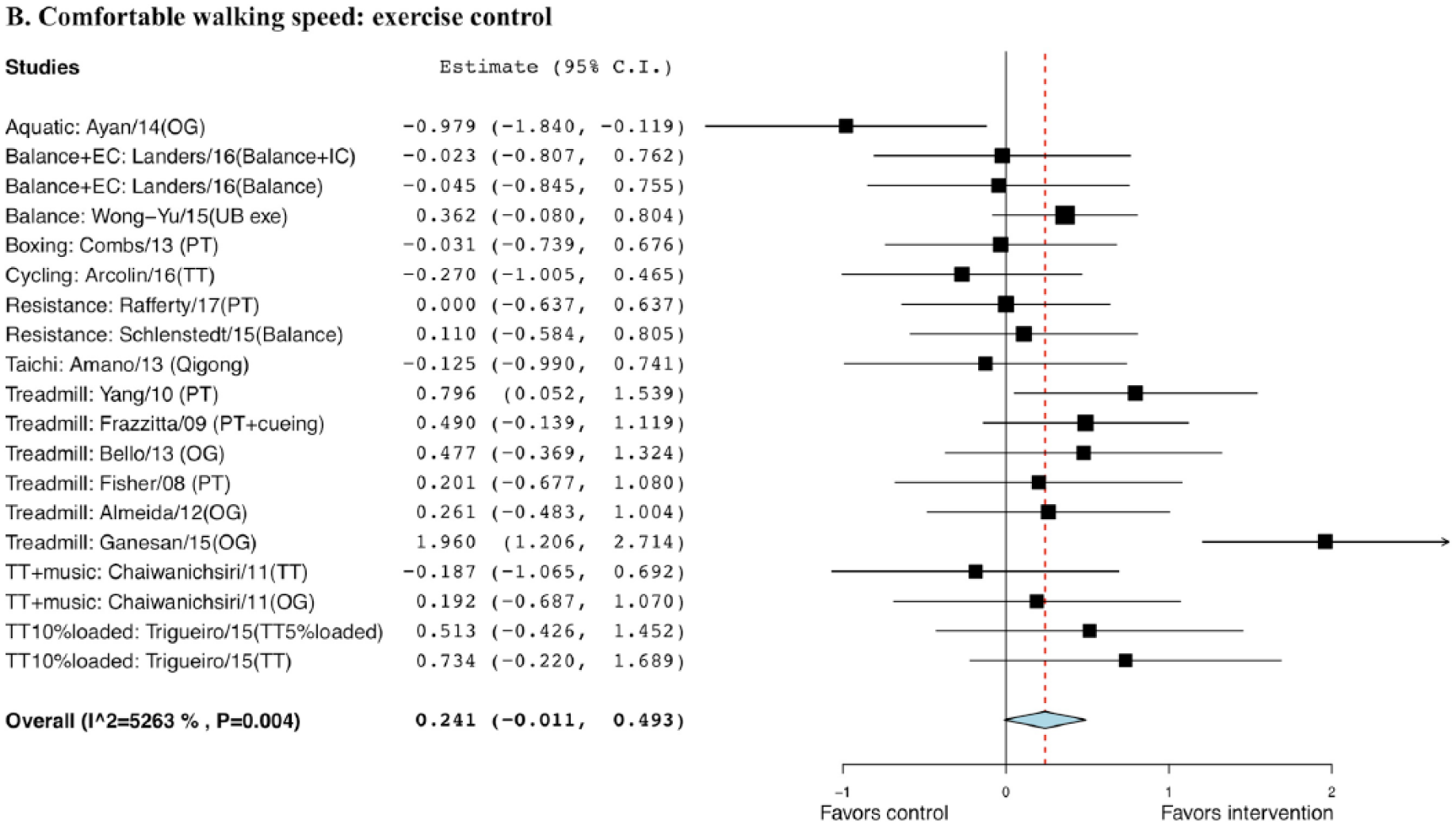
Forest plot of exercise effect on comfortable walking speed comparing to exercise controls.a
Fast Walking Speed
The Hedge’s g for fast walking speed when comparing exercise with non-EXE control was 0.430, with a SE of 0.19 and statistical significance (95% CI = 0.06 to 0.80; P = .023); the g when comparing exercise with EXE control was 0.324, with a SE of 0.23, but not statistically significant (95% CI = −0.12 to 0.77, P = .152; Figure 3).
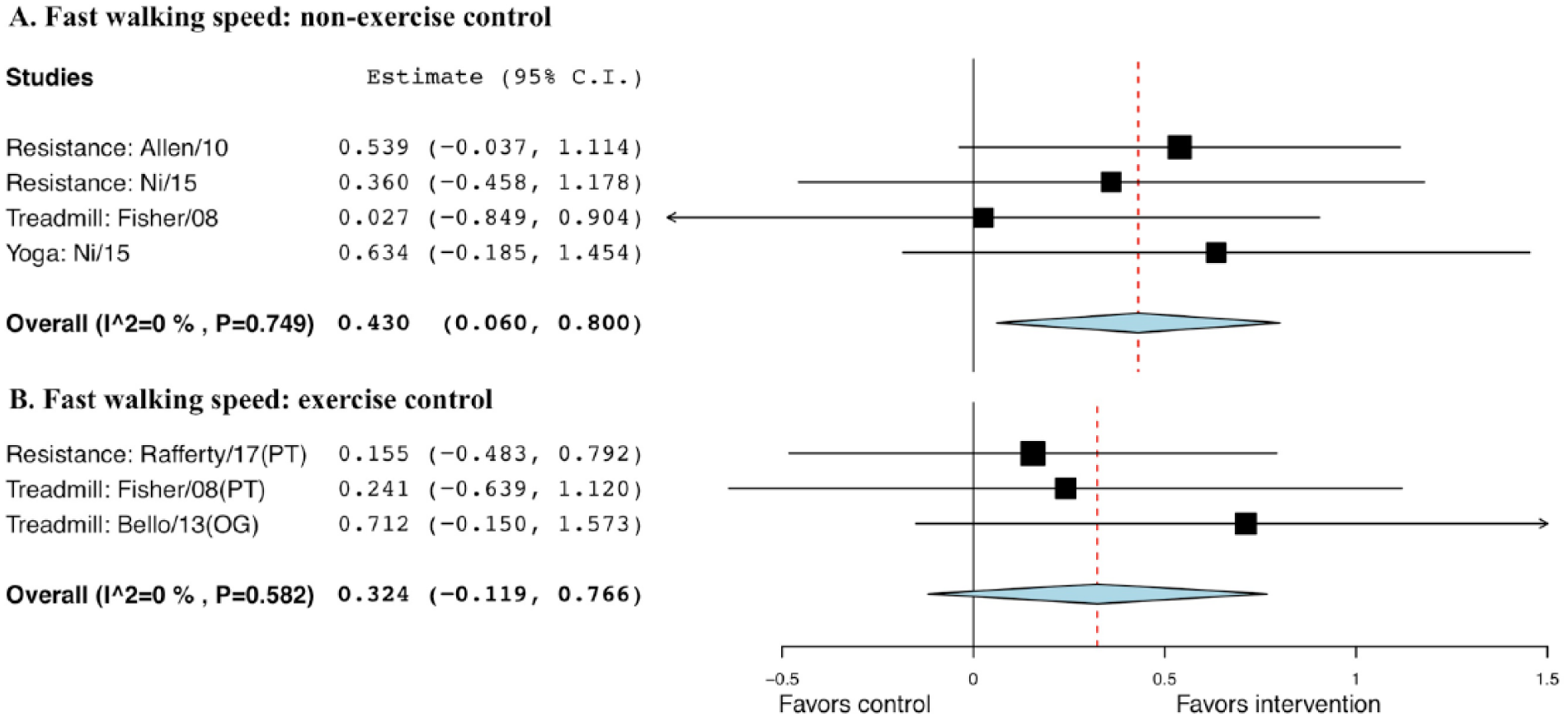
Forest plot of exercise effect on fast walking speed. A. Comparison of exercise interventions with nonexercise controls. B. Comparison of exercise interventions with exercise controls.a
Stride or Step Length
The effect size comparing exercise with non-EXE control was statistically significant (0.379), with a SE of 0.14 (95% CI = 0.11 to 0.65, P = .006; Figure 4). But it was not significant when comparing exercise with EXE control, with a g of 0.198 and a SE of 0.13 (95% CI = −0.05 to 0.45, P = .118; Figure 5).
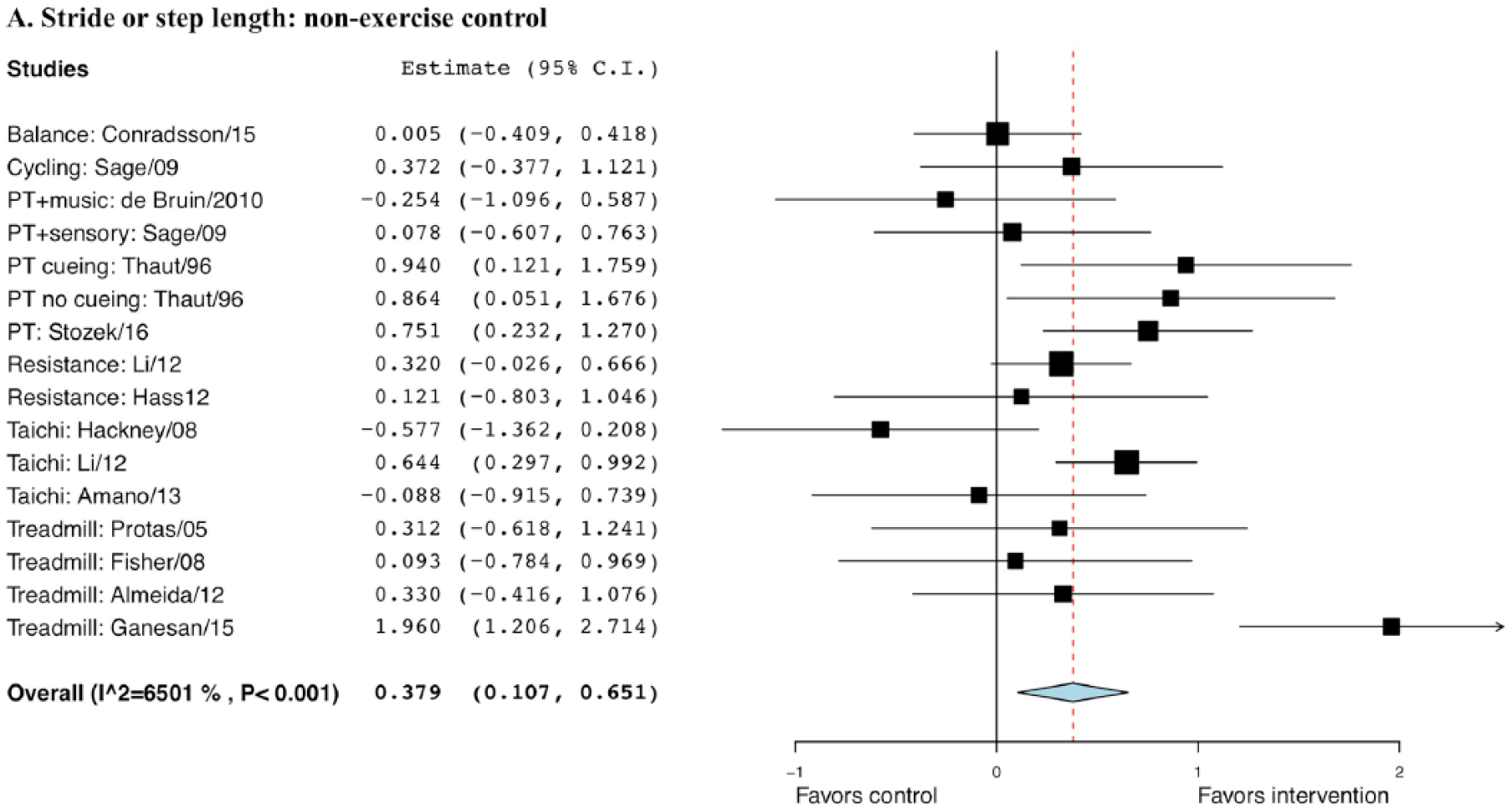
Forest plot of exercise effect on stride or step length compared with nonexercise controls.
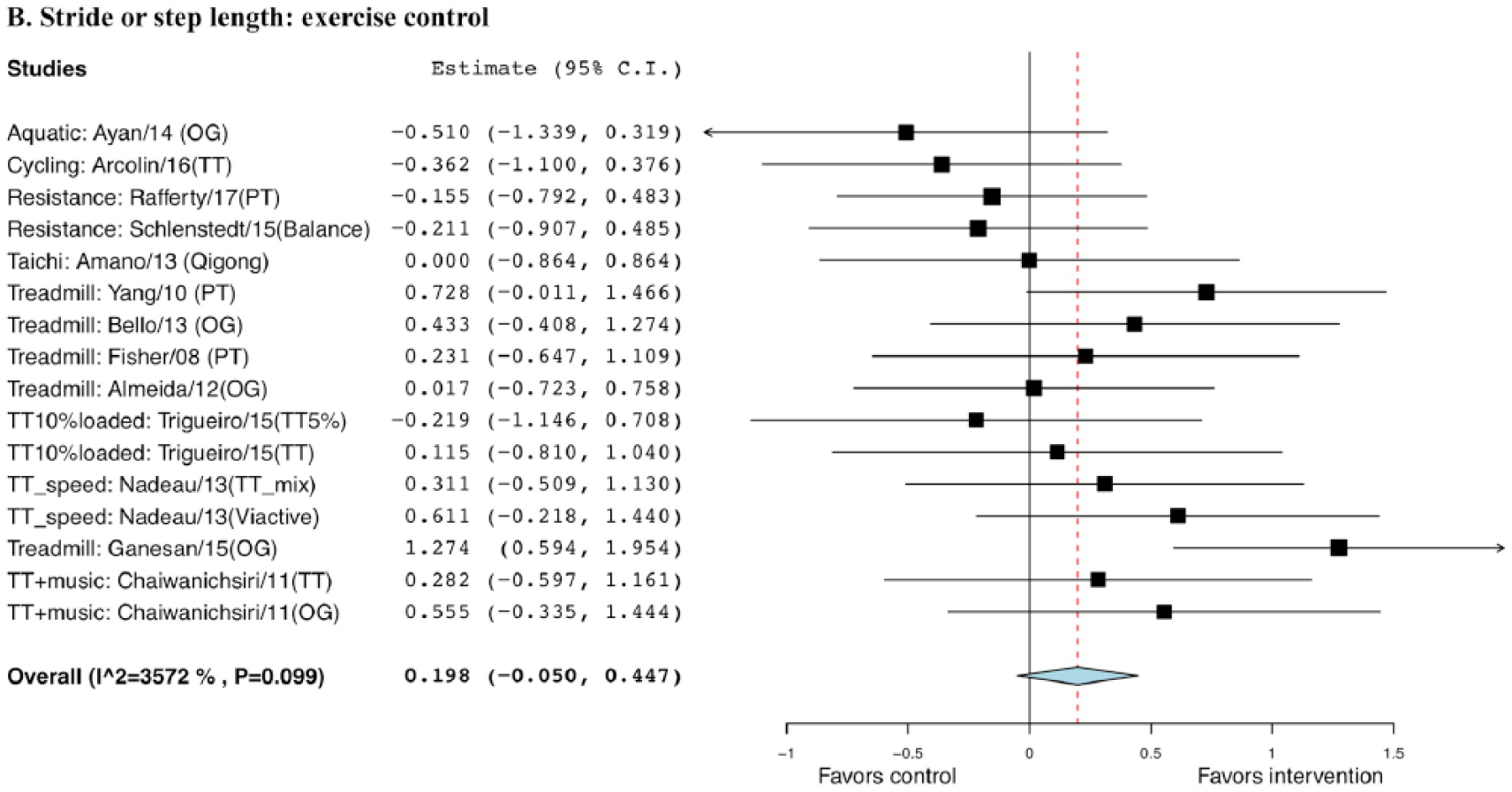
Forest plot of exercise effect on or step length compared with exercise controls.a
Cadence
The standard difference between exercise intervention and non-EXE control (g = 0.223; SE = 0.12; 95% CI = −0.01 to 0.46; P = .062) was not statistically significant; however, for EXE control (g = 0.282; SE = 0.11; 95% CI = 0.06 to 0.51; P = .014), it was statistically significant (Figure 6).
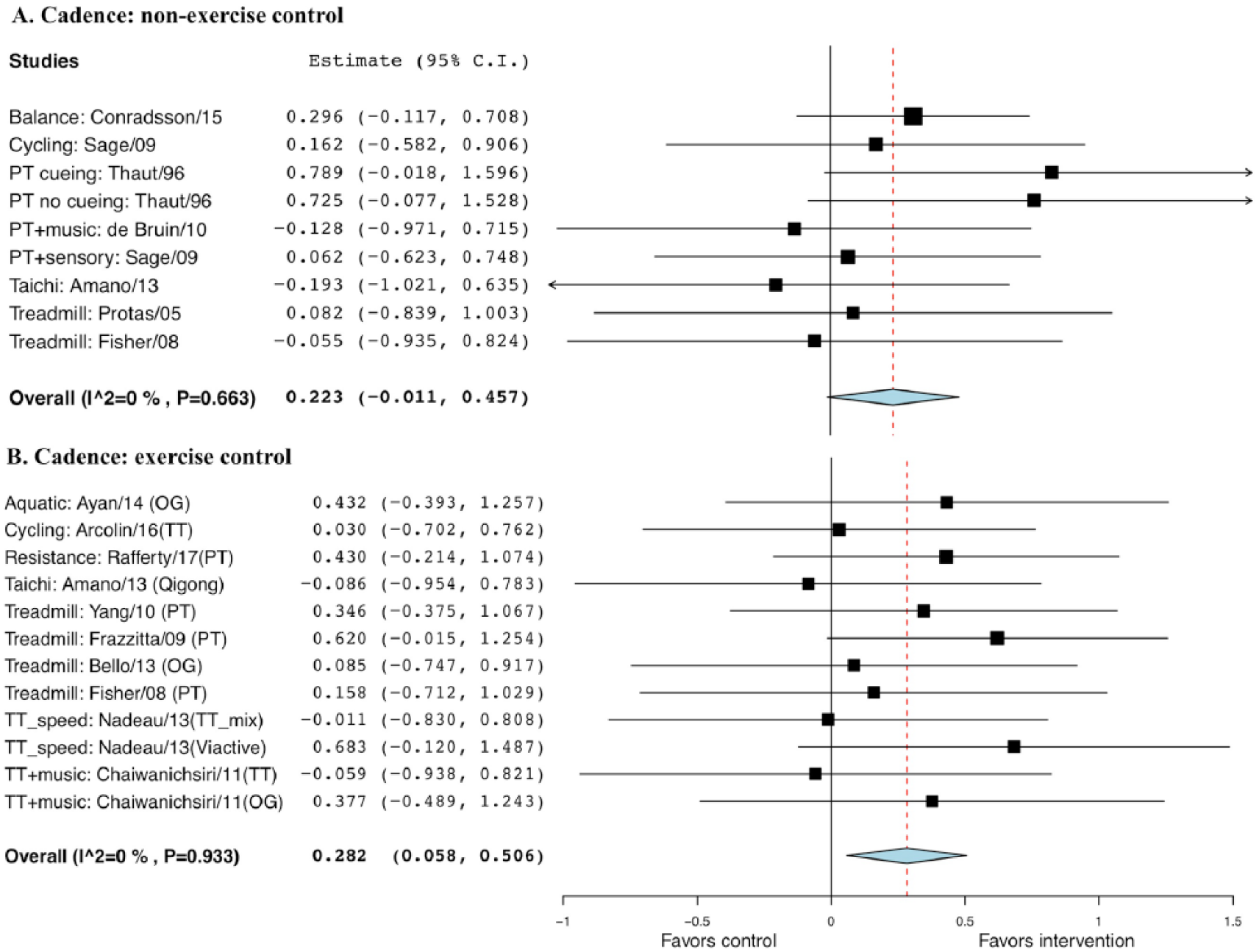
Forest plot of exercise effect on cadence. A. Comparison of exercise interventions with nonexercise controls. B. Comparison of exercise interventions with exercise controls.a
Double-Leg Support Time
No significant difference was observed neither when comparing exercise with non-EXE control, with a g of −0.135 and SE of 0.20 (95% CI = −0.52 to 0.25; P = .495), nor with EXE control, with a g of 0.095 and SE of 0.15 (95% CI = −0.19 to 0.38, P = .515; Figure 7).
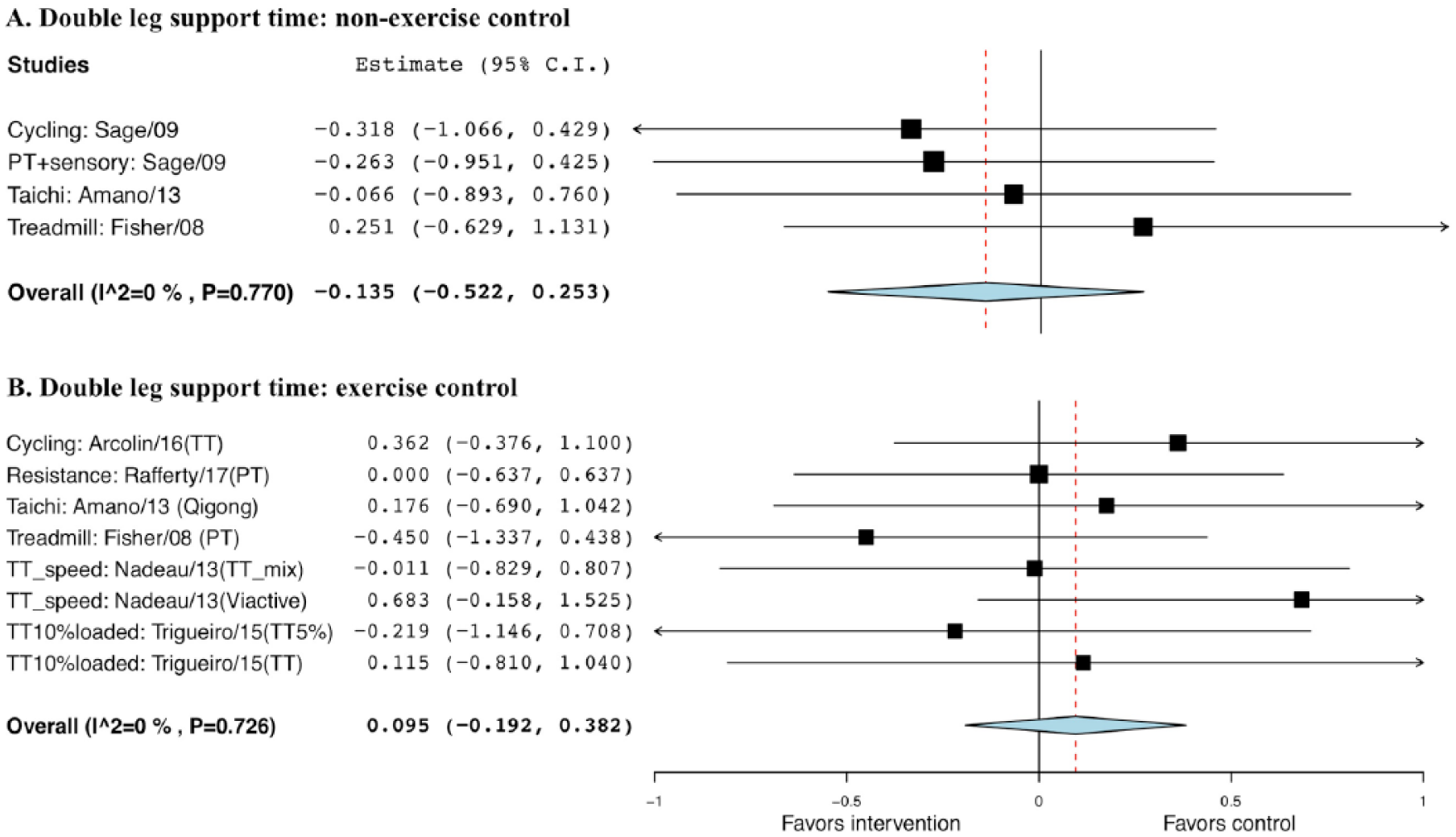
Forest plot of exercise effect on double-leg support time. A. Comparison of exercise interventions with nonexercise controls. B. Comparison of exercise interventions with exercise controls.a
Timed Up-and-Go
A significant difference was observed when comparing exercise with non-EXE control, with a g of −0.458 and SE of 0.13 (95% CI = −0.71 to −0.21, P < .001), and EXE control, with a g of −0.390 and SE of 0.08 (95% CI = −0.55 to −0.23, P < .001; Figure 8).
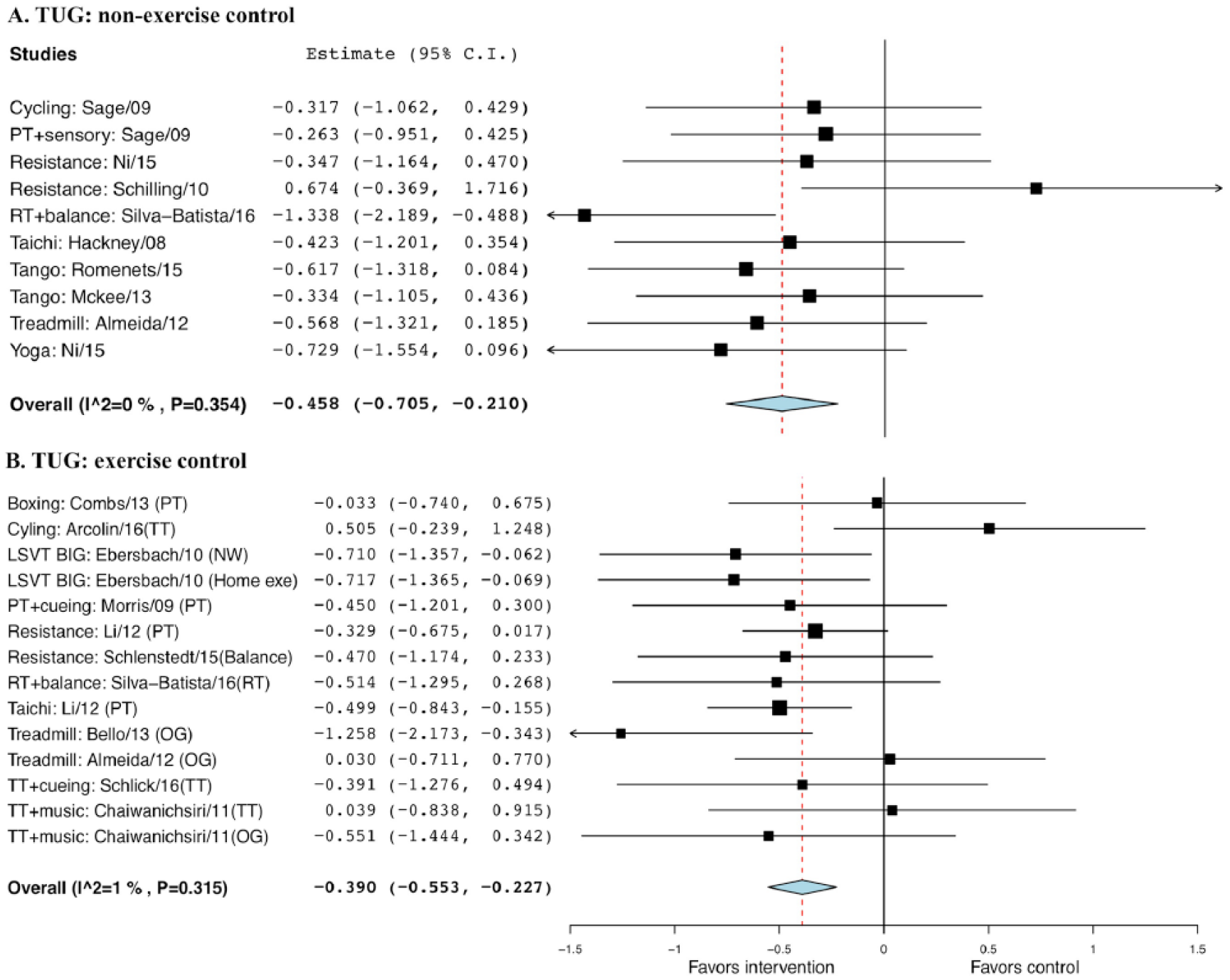
Forest plot of exercise effect on timed up-and-go (TUG) test. A. Comparison of exercise interventions with nonexercise controls. B. Comparison of exercise interventions with exercise controls.a
Dynamic Gait Index
No statistical significance was observed when comparing exercise with non-EXE control, with a g of 0.443 and SE of 0.24 (95% CI = −0.02 to 0.91; P = .063), and EXE control, with a g of −0.063 and SE of 0.29 (95% CI = −0.63 to 0.50, P = .827; Figure 9).
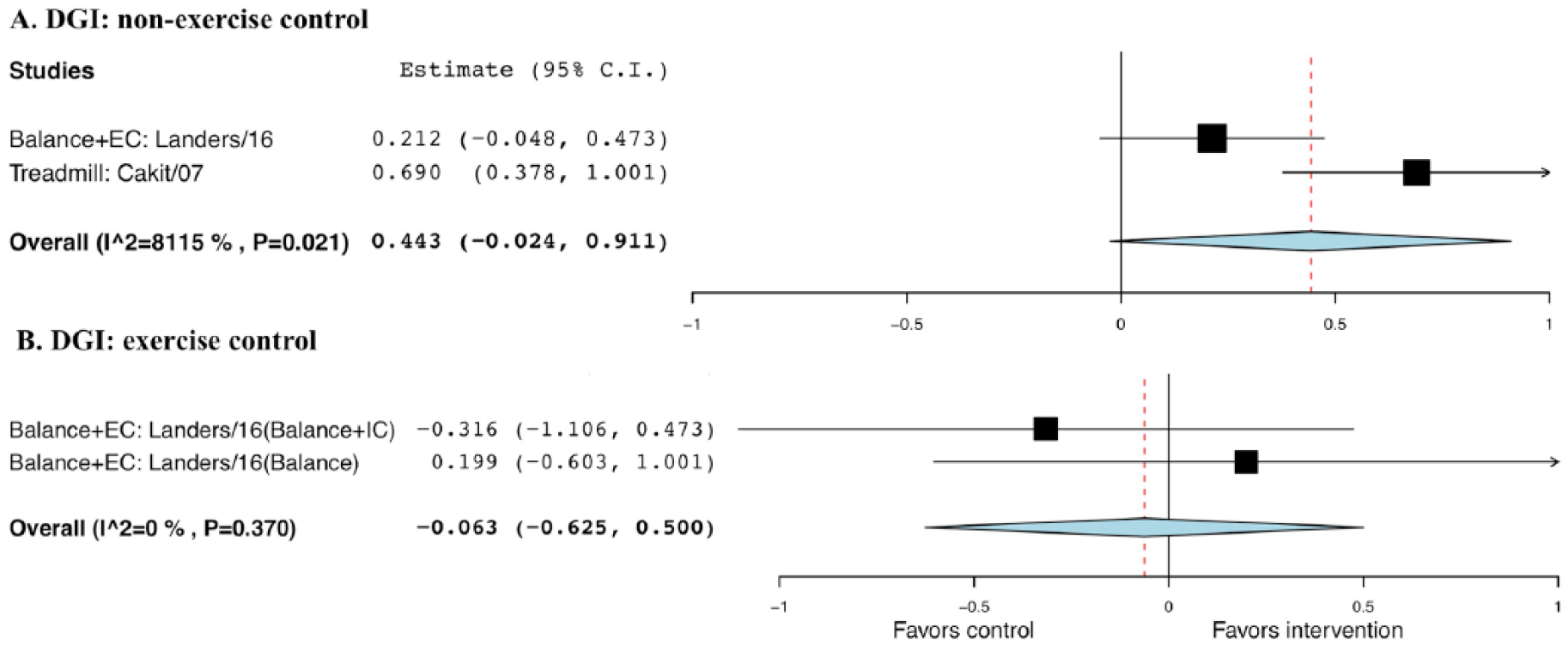
Forest plot of exercise effect on Dynamic Gait Index (DGI). A. Comparison of exercise interventions with nonexercise controls. B. Comparison of exercise interventions with exercise controls.a
6-Minute Walk Test
No statistical significance was seen comparing exercise with non-EXE controls (g = 0.341; SE = 0.28; 95% CI = −0.21 to 0.90; P = .23) or EXE controls (g = 0.190, SE = 0.15, 95% CI = −0.10 to 0.48, P = .20; Figure 10).
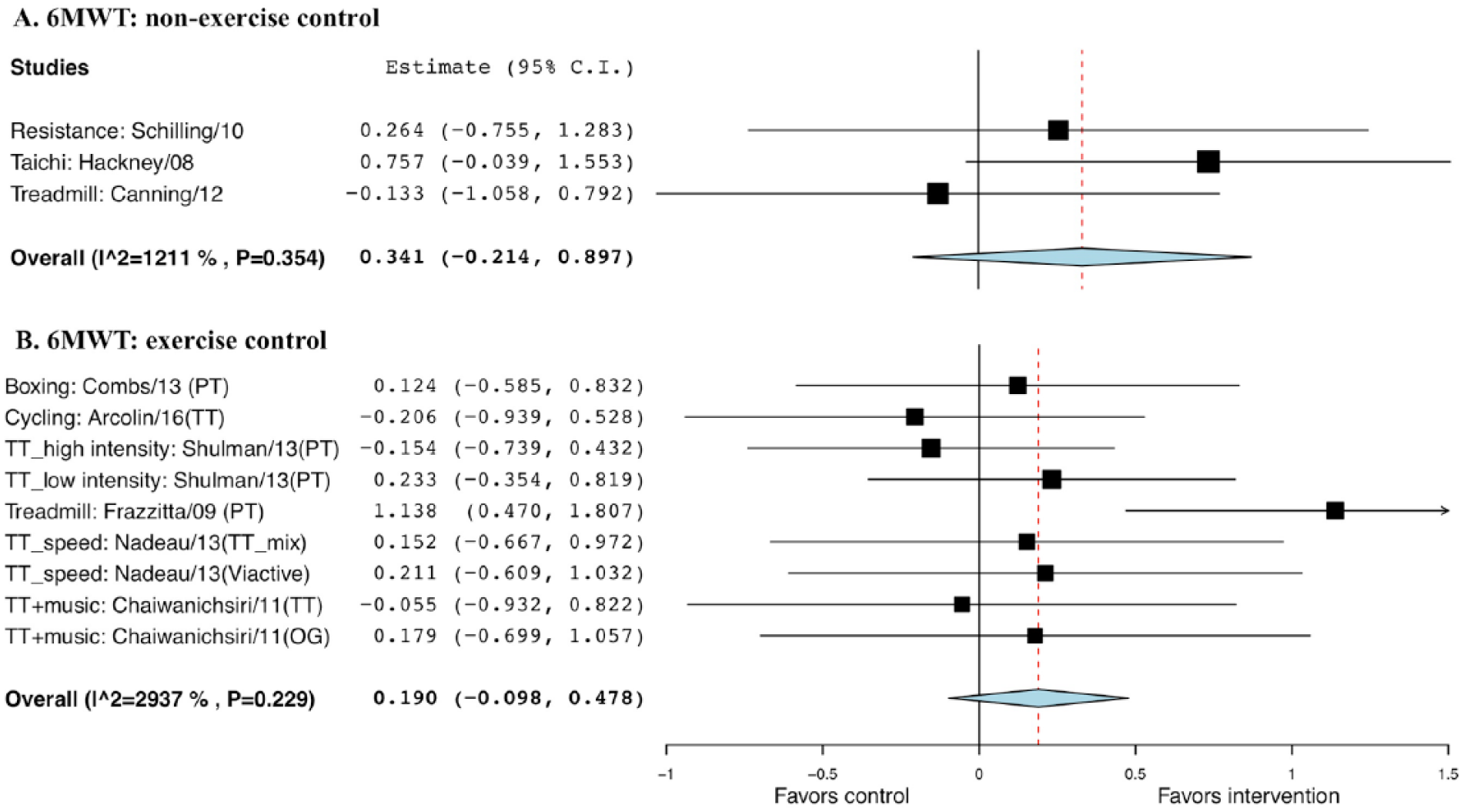
Forest plot of exercise effect on 6-minute walk test (6MWT). A. Comparison of exercise interventions with nonexercise controls. B. Comparison of exercise interventions with exercise controls.a
Freezing of Gait Questionnaire
The random-effect model was not statistically significant comparing exercise with non-EXE controls (g = −0.057; SE = 0.24; 95% CI = −0.52 to 0.41; P = .808) or EXE controls (g = −0.236, SE = 0.33, 95% CI = −0.89 to 0.42, P = .480; Figure 11).
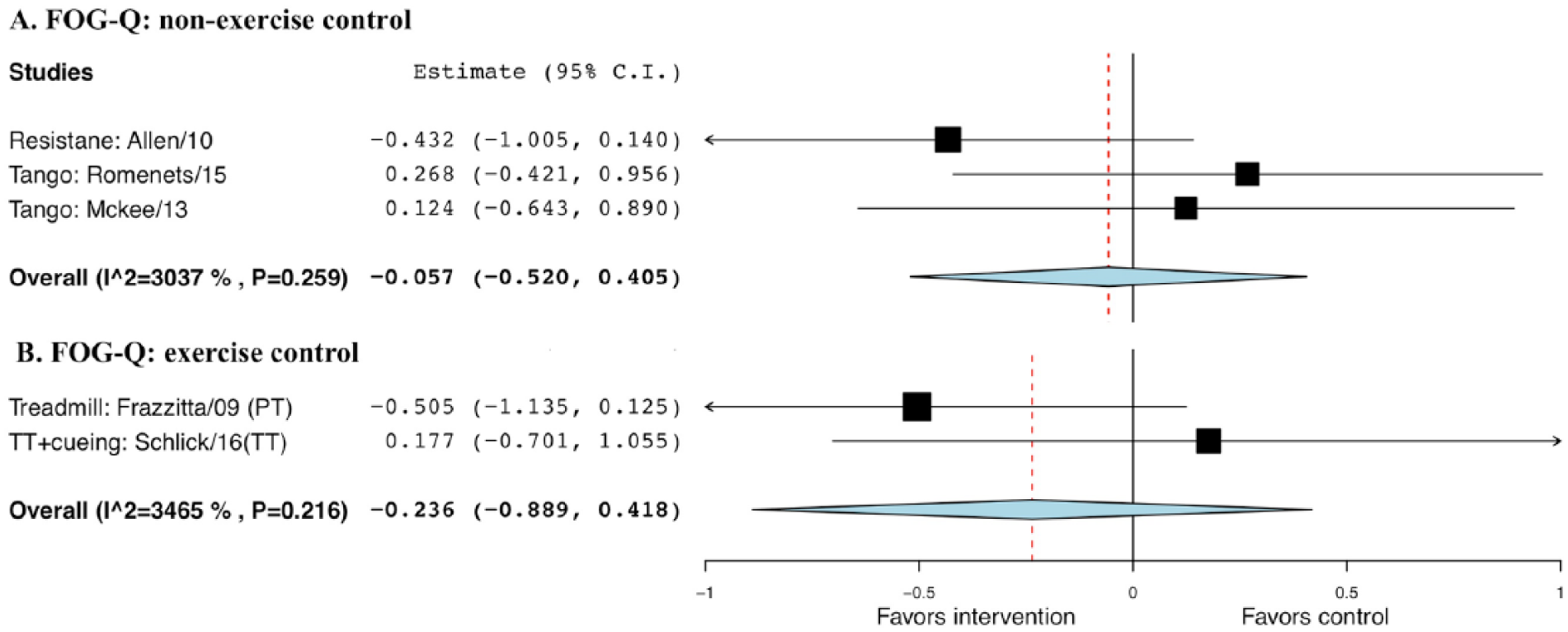
Forest plot of exercise effect on freezing of gait questionnaire (FOG-Q), compared with nonexercise or exercise controls.a
Effect of Moderators
Cuing
A significant effect of cuing (yes/no or unclear) was only observed for the stride or step length during exercise comparing with EXE controls, where a larger effect for exercise with cuing (g = 0.543; SE = 0.24; 95% CI = 0.06 to 1.0; P = .026) than exercise without cuing or with unclear cuing (g = 0.357; SE = 0.21; 95% CI = −0.59 to 0.21) was observed.
Exercise Duration
When the total length of intervention (6 to 45 hours or 360 to 4320 minutes) in the trials was used as a moderator for gait performance, a significant effect of exercise duration was only observed for the comfortable walking speed during exercise comparing with EXE controls. The estimated intercept and slope were 0.66 (SE = 0.22; 95% CI = 0.22 to 1.09; P = .003) and −0.023 (SE = 0.01; 95% CI = −0.03 to −0.003; P = .024). The estimated mean effect size was 0.66 when the intervention of interest was 6 hours; for each additional increase in hour of intervention, the effect size decreased by 0.023, comparing with another form of exercise.
Discussion
Exercise recommendation for improving gait functions are shown in Table 1.
Evidence-Based Exercise Recommendations for Improving Gait Function for Parkinson’s Disease (H&Y I-III).
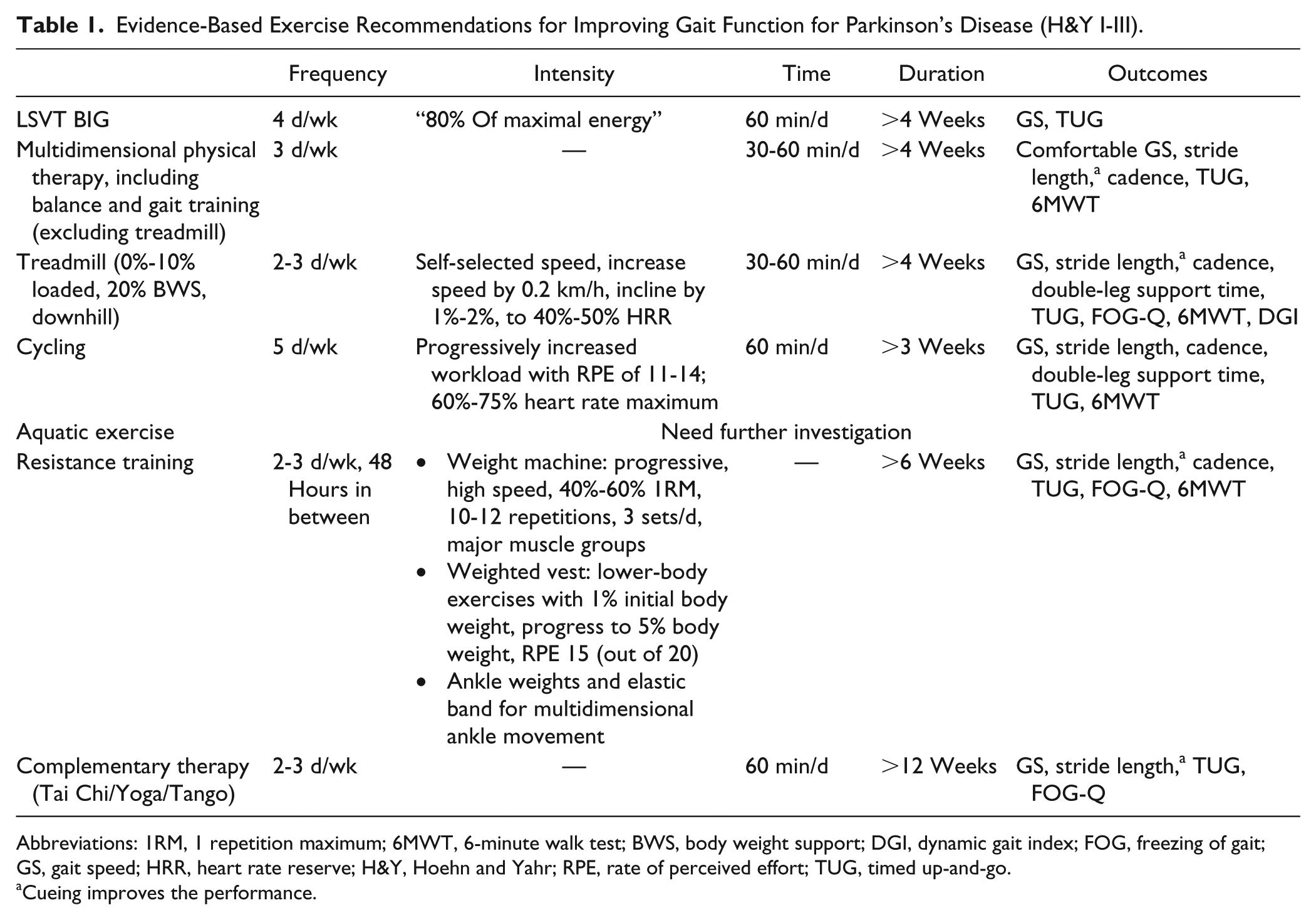
Abbreviations: 1RM, 1 repetition maximum; 6MWT, 6-minute walk test; BWS, body weight support; DGI, dynamic gait index; FOG, freezing of gait; GS, gait speed; HRR, heart rate reserve; H&Y, Hoehn and Yahr; RPE, rate of perceived effort; TUG, timed up-and-go.
Cueing improves the performance.
Exercise Mode
Our results indicate that variations in the magnitude of gait improvement may depend on the types of training. Walking function appears to be improved by (1) LSVT BIG; (2) multidimensional physical training, including balance and gait training (excluding treadmill); and aquatic exercise; (3) treadmill and cycling training; (4) resistance training focusing on lower-body musculatures; and (5) Tai Chi, yoga, dance, and boxing.
LSVT BIG Program
The LSVT BIG group 15 showed a significant improvement in TUG compared with nonsupervised home exercise, which also included high-amplitude movements. This suggests that patient-therapist face-to-face training is recommended for achieving the greatest improvement in motor function.
Multidimensional Physical Training
Researchers reported results of function-based physical therapy with external cues (music, 16 auditory, and visual12,17) and physical therapy combined with medication. 18 Sensory-enhanced physical therapy produces significantly greater improvement in gait parameters, including comfortable walking speed,17,18 stride length,16,17,19 cadence,16,17,19 and DLST. 19 The improvements in gait parameters may be attributable to enhanced motor learning and retention. 20
Multidimensional physical intervention, emphasizing balance and gait components, have been gaining prominence, where training includes flexibility and strengthening, balance, step training with trunk rotation, functional training, and postural re-education in one training session. Training adopting highly challenging tasks, such as obstacle negotiation, utilizing unstable surfaces, and dual task seems effective to improve general motor function21-23 as well as cognitive status.21,22
Another type of physical training is water-based exercise. Three trials24-26 included in the current study (1 study 27 included in the meta-analysis) compared the effects of aquatic physiotherapy with conventional land therapy. Neither group nor time differences were seen in overground gait-related performance after these interventions. Therefore, the capacity of aquatic training effects to transfer into overground walking function needs further investigation.
Treadmill and Cycling Training
These training methods have been presented as a potentially effective approach to reduce freezing of gait and promote a comfortable stepping pattern. 28 Treadmill endurance training could be classified into 4 types: regular treadmill walking (unloaded), body weight support (BWS), loaded, and downhill walking. Treadmill training combined with auditory and visual cues yielded better performances in gait speed and stride cycle than conventional physical therapy with external cues. 29 Three studies30-32 compared treadmill training with overground gait training and reported that both training modalities improve gait performances, including speed,30,32 stride or step length, 32 cadence, 30 and TUG. 31 However, the persistence of treatment effect between treadmill and overground training seems to be different. In the work of Bello et al, 33 treadmill training produced significant increases in comfortable walking speed, cadence, stride length, and fast walking stride length after a 5-week training, and these improvements were maintained for 1 month. In contrast, overground training only improved preferred walking speed and cadence, and these improvements were not maintained across the 1-month follow-up period. For the treadmill with BWS or external load, Miyai et al 34 showed that a 20% BWS was the most comfortable percentage of weight reduction compared with 0%, 10%, and 30% (least comfortable) BWS, whereas Toole et al 35 reported that the 5% loaded and 25% BWS treadmill training produced similar performances in stride length and cadence; but Trigueiro et al 13 suggested that 10% loaded, 5% loaded, and unloaded treadmill training yielded similar improvements in gait speed, stride length, and DLST. This might suggest that regular treadmill training would be sufficient to induce improvement in gait function and that loads from 20% BWS to 10% loaded treadmill training could also be utilized as alternatives. Downhill treadmill walking has also been shown to be effective for generating higher walking speed and stride length, which may be attributed to the imposed trunk extension in an effort to pull the center of mass backward and decrease the forward momentum produced by gravity during downhill walking, which results in reduced trunk flexion and improved posture. 36 Also, it may be associated with the increased knee extensor strength resulting from eccentric contraction but lower oxygen demand during downhill walk.37,38 Finally, it seems that the training session should be supervised in order to induce significant improvements in walking parameters because it was shown that a semisupervised home-based treadmill training program was not sufficient to generate improvement. 39
Two trials evaluated the effect of cycling on gait performance and compared the treatment effect with no intervention 19 and treadmill training. 40 Sage and Almeida 19 only observed improved step length, but Arcolin et al 40 showed improved gait speed, step length, cadence, single-leg support time and DLST, TUG, 6MWT, Mini-BESTest, and UPDRS motor score, even though in this analysis, the treatment effect of cycling is not as high as that of treadmill (see Figures 1-6, 8). The difference in the findings might result from the exercise dosage (Sage and Almeida: 30 min/d, 3 d/wk; Arcolin et al: 2 sessions of 30 min/d, 5 d/wk). However, the smaller treatment effect from cycling compared with treadmill may be a result of biomechanical specificity because treadmill training is similar to the task of walking, with sensory input provided by the treadmill. 30 Moreover, it should be noted that in the study by Arcolin et al, 40 cycling and full body exercises (strengthening, balance, and stretching) were combined in the training program; this suggests that intervention programs should be designed to be multidimensional while maintaining a level of specificity in order to maximize the treatment effect.
Resistance Training
Progressive resistance training, using resistance machines41-43 or lower-body strengthening exercise with a weighted vest25,44 or ankle weights 44 or elastic bands, 41 has been shown to significantly increase gait speed,41,43,44 stride length,41,44 cadence,41,43,44 and FOG-Q. 25 Power-based resistance training,45,46 using high-speed and moderate intensity, was shown to reduce bradykinesia, increase movement speed, 46 and improve balance, 46 gait performance, 46 6-MWT time, 45 and leg press strength45,46; however, whether this intervention is superior to traditional resistance training in PD is unknown. Two trials25,47 combined resistance training with a balance component, but only the work of Silva-Batista et al 47 showed improved balance and strength after the resistance and stability training compared with resistance training alone. The nature of resistance may lead to improved postural control and walking ability through enhanced ankle and hip strategies 48 and neuromuscular relearning. 44
Tai Chi, Yoga, Dance, and Boxing
Three RCTs examined the impact of Tai Chi on gait.44,49,50 They used Yang Short or 8-form styles, and 2 of them produced significant improvement in the gait speed and stride length compared with no intervention or stretching.44,50 Li et al 44 showed a significantly greater increase in stride length through Tai Chi compared with resistance training. The improvement in gait seen may be attributable to the characteristics of Tai Chi, which uses large and alternating steps to effectively reduce rigidity and enhance the ability to control the center of gravity, and sustains knee bending during squatting and stepping. It is highly likely that Tai Chi could improve muscle strength in the lower extremity and subsequently enhance the gait performance, as evidenced by increased muscle strength of bilateral knee extensors and flexors after Tai Chi training. 44 Another complementary therapy that continues to gain attention is yoga. A specially designed power yoga program using Vinyasa yoga style, provided by Ni et al, 46 significantly improved mobility, including comfortable and fast walking speed, balance, and agility (TUG). The fast transition from one posture to another may have contributed to improved postural control and increased movement speed, whereas the repetitive and multidirectional step training may have facilitated the motor learning process. Finally, Argentinian tango, a type of dance therapy, has been increasingly utilized because of its potential to address freezing of gait 51 and enhance internal rhythm by imposing external rhythm from the music. 52 Three RCTs evaluated the effect of tango, but the findings varied. Romenets et al 53 reported improvement in TUG performance but no significant change in FOG-Q, whereas Duncan and Earhart 54 reported improved FOG-Q, and McKee et al 55 detected no significant improvement in TUG or FOG-Q.
The combination of resistance, gait training, and boxing specific activities was effective in producing increases in walking speed and aerobic capacity compared with traditional physical therapy. 56 In the study, participants were trained as intensely as was tolerated, and repetitions were increased during each 3-minute training interval. This high-intensity interval training may be an alternative approach to a task-specific training with a focus on gait.
Importance of Task-Specific Training
Current evidence suggests that for training designed to enhance motor skill learning, both repetition and task-specific practice is required to maximize training effect,57,58 and repeated practice of challenging movement tasks results in larger brain representations of the practiced movement.59,60 Based on the calculated effect sizes, the treatment effect from a task-specific exercise compared with a standard exercise control, such as physical therapy or stretching, is even greater than that compared with a nonexercise control. It is, therefore, logical that exercise emphasizing task specificity would contribute to a higher quality of gait, especially during an activity that requires greater physical and cognitive demands. This is reflected by the successful outcomes in variables included in our analysis, such as cadence and FOG-Q. Finally, in addition to functional improvements, task-specific practice has been associated with neuroplastic changes within the cortical and subcortical areas of the brain.61,62 These results provide strong support for the use of gait-specific training when improvements in Parkinsonian gait are the major goal.
Exercise Frequency/Intensity/Duration
Overall, it seems that 12 to 14 training sessions spreading out over 2 to 4 weeks is the minimum exercise dosage for a short-term improvement. Even though the metaregression yielded a significant but negative relationship between total exercise duration and exercise interventions on the comfortable walking speed (the treatment effect would decrease with an increase in the duration of exercise), it should be noted that the variations in the training effect resulting from differences in population sample, exercise modality, and training volume may have contributed to this finding or the lack of significance.
LSVT BIG and Multidimensional Physical Training
For a general physical therapy program, the required intensity has not been determined because of the variations in therapeutic goal. A notable exception was the LSVT BIG program, where intensity was specified as “80% of subject’s maximal energy” for each repetition, 15 with H&Y stage I to III. This provides a guideline for implementing the LSVT BIG program for PD patients with mild to moderate mobility limitations; however, the LSVT program only yielded a trend toward increased walking speed. This may be a result of the low dosage of training, where the total training time was 960 minutes with a relatively short training duration (4 weeks) and insufficient training frequency (60 min/d, 4 d/wk). In contrast, a strategy-based physical therapy program used with inpatients (45 min/d, 7 d/wk, 2 weeks) induced significant increases in walking speed and 2-minute walking endurance at discharge. 12 However, it should be noted that only the improvement in 2-minute walking endurance was maintained during the 3-month follow-up. Consequently, long-term effects need to be verified in future investigations using higher exercise doses. For other programs,16,19,21-23,63,64 the exercise frequency varied among studies (2-7 times/wk), but the majority of the trials used a frequency of 3 times/wk, 30 to 60 minutes per session, for 4 to 12 weeks, which induced significantly improved gait function.
Treadmill and Cycling Training
For treadmill training, training with low intensity and longer duration produced the most consistent improvements in gait speed. It appears unnecessary to utilize high-intensity walking to achieve these benefits. This is fortunate because low-intensity exercise is more accessible and safer for a larger group of patients. Because the treadmill imposes external pace and enhances the focus on gait, starting training at a comfortable walking speed and gradually increasing to 40% to 50% heart rate reserve is sufficient to make meaningful improvements. 65 Other trials used the approach of starting with patients’ self-selected comfortable pace and increasing the speed by 0.2 km/h, 66 increasing the incline by 1% to 2%,35,36,65 or a combination of both,65,66 thereby reaching 70% to 80% heart rate maximum.13,67 This was accomplished without any step difficulty or gait deviation. Training sessions ranging from 20 to 60 minutes, 2 to 3 times/wk appear sufficient to increase gait function.13,31,32,34-36,65-72
For cycling training, moderate-intensity training (60%-75% heart rate maximum, 19 rate of perceived effort of 11-14 out of 20 40 ), 30 to 60 minutes per day, can induce significant improvement in stride length. However, other components, including strengthening and balance, should be included in the cycling regimen to maximize the treatment effect on gait outcomes.
Resistance Training
Resistance training, 2 to 3 times per week, is also recommended for significantly improving gait function, lower-body strength, and power, which are critical factors for predicting walking and balance functions. 73 For progressive resistance training,41-43 starting with 30% to 40% of 1 repetition maximum (1RM) for upper-body exercises and 50% to 60% of 1RM for lower-body exercises and increasing with 5% loads, 12 to 20 repetitions to volitional fatigue, 2 to 3 sets per day, is recommended. For 2 strength training programs using weighted vests, divergent results were seen.25,44 Allen et al 25 reported no significant increase in gait function using gradually increasing weight to a maximum rate of perceived exertion of 15 out of 20. In contrast, Li et al 44 reported a significant increase in gait velocity using an initial load of 1% of body weight that gradually increased by 1% to 2% every 5 weeks until reaching the maximum weight of 5% body weight. The difference in the training responses between these 2 studies was likely a result of the variation in loading and training duration (Allen et al: 6 weeks; Li et al: 24 weeks). Furthermore, the intensity of 1 power-based resistance training study used each individual’s load that generated maximal power across 7 intensities, and these loads were adjusted with each power plateau with 8 to 12 repetitions for each exercise and 3 sets per training session over a 12-week course of training. 46 However, the training devices, pneumatic resistance machines, have been used nearly exclusively in the research setting and have limited access in treatment facilities. Another power-based resistance training program adopted 5 to 8 repetitions to volitional fatigue, 2 to 3 sets per training session, and progressed with adding 5% to 10% additional weights over 8 weeks. 45 This program only showed significant improvement in walking endurance (6MWT) but not in mobility (TUG).
Tai Chi, Yoga, Dance, and Boxing
For the 3 studies using Tai Chi,44,49,50 the total training duration was 1200 to 2880 minutes with 1 hour training per day, 1 to 3 times per week. For yoga 46 and tango,53-55 it is likely that training 1 hour per session, 2 times per week, for 12 weeks would be sufficient to induce changes in gait function.
Effect of Cuing
External cuing, including visual and auditory rhythmic cuing, has been widely used for gait training because it helps normalize temporal and spatial gait parameters. 74 Previous systematic reviews75,76 and a meta-analysis 77 have suggested that auditory cuing could significantly improve walking speed, 75 and visual cuing significantly improved stride length. 77 The effect sizes resulting from auditory cuing reported in that meta-analysis were moderate for walking speed (g = 0.544), stride length (g = 0.497), and cadence (g = 0.556). 77 In our analysis, the effect size of cuing was significant for the stride length (g = 0.543), but not significant for speed (g = 0.501) or cadence (0.079). This indicates that using auditory or visual feedback is an effective strategy if stride length is the target gait parameter. The difference in the effect size could be a result of the difference in the analysis strategy, where the control groups were separated into non-EXE controls or EXE controls, and the types of cuing were not specified in our analysis.
Limitations
Given that PD is a progressive disease, long-term preservation of physical function is of marked importance to delay disease-related and aging-related declines. Most published studies confirm that exercise could have benefits on the completion of an intervention program; however, the prolonged persistence of the treatment effect from exercise is less known and should be investigated. In addition, most existing studies evaluated the benefits of exercise for those with mild to moderate PD (H&Y stage I-III) who were capable of ambulation. Future research is needed to establish the feasibility and dose–response effect of exercise in patients with a severe loss of gait function.
Conclusion
Exercise can improve gait performance, including walking speed, stride length, cadence, and dynamic balance (double-leg support, TUG, and DGI) in adults with PD. However, the effect on walking endurance and freezing of gait needs further investigation. The development of effective and sustainable task-specific programs, which yield a short-term positive effect as well as a persistent long-term positive effect, is of great importance. This study offers evidence-based exercise recommendations to help ensure optimal exercise dosage to enhance gait function in patients with PD.
Supplemental Material
PRISMA_Flow – Supplemental material for Exercise Guidelines for Gait Function in Parkinson’s Disease: A Systematic Review and Meta-analysis
Supplemental material, PRISMA_Flow for Exercise Guidelines for Gait Function in Parkinson’s Disease: A Systematic Review and Meta-analysis by Meng Ni, Joseph B. Hazzard, Joseph F. Signorile and Corneliu Luca in Neurorehabilitation and Neural Repair
Supplemental Material
Supplement_Table_1 – Supplemental material for Exercise Guidelines for Gait Function in Parkinson’s Disease: A Systematic Review and Meta-analysis
Supplemental material, Supplement_Table_1 for Exercise Guidelines for Gait Function in Parkinson’s Disease: A Systematic Review and Meta-analysis by Meng Ni, Joseph B. Hazzard, Joseph F. Signorile and Corneliu Luca in Neurorehabilitation and Neural Repair
Supplemental Material
Supplement_Table_2 – Supplemental material for Exercise Guidelines for Gait Function in Parkinson’s Disease: A Systematic Review and Meta-analysis
Supplemental material, Supplement_Table_2 for Exercise Guidelines for Gait Function in Parkinson’s Disease: A Systematic Review and Meta-analysis by Meng Ni, Joseph B. Hazzard, Joseph F. Signorile and Corneliu Luca in Neurorehabilitation and Neural Repair
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
