Abstract
Introduction
Most people who prefer to use their right hand during unimanual tasks are more dexterous or deft when using their right rather than left hand. This deftness includes the ability to make more precise and independent but coordinated finger–hand movements. 1 Tests for assessing arm–hand–finger deftness include coin rotation and the pegboard test. Studies of nonhuman primates and humans have suggested that deftness is primarily mediated by the primary motor cortex and its connection to the neurons in the spinal cord as well as premotor areas. 2-4 Injury to the left hemisphere’s motor areas that are contralateral to the dominant (right) hand reduce the deftness of the ipsilesional (left) hand more than damage to the right motor areas reduces ipsilesional (right) hand deftness. 1 One explanation for this finding is that, among right-handed people, the left hemisphere’s motor cortex might have a greater influence on the right hemisphere’s motor cortex than the right hemisphere’s motor cortex has on the left. A similar explanation postulates that the left motor cortex may have more ipsilateral projections to the motor neurons that control left hand muscles. However, both hypotheses are speculative and other explanations may exist.
After the loss of a limb, numerous physiological changes may occur in the organizational map of cortical neurons. One example is a shift of neighboring representational zones in both the primary somatosensory cortex (S1) and motor cortex (M1) into the areas that were formerly represented by the amputated extremity, 5-7 thereby inducing a degradation of the amputated limb’s cortical representation. 8 Studies have shown that damage to 1 side of the cortex can induce neurological changes that increase both the attention allocated to the contralateral limb and the dexterity of the contralateral limb. 5,6 Although cortical changes in an area representing an amputated limb and cortical damage caused by stroke are different phenomena, there are some similarities. One study focusing on attentional changes in individuals who had suffered a hemispheric lesion caused by stroke found that homologous regions in the other hemisphere that previously received inhibitory input from the lesioned side were released from inhibition. This caused a hyperorientation of attention to the ipsilesional side. 5 According to these findings, it is possible that after an amputation, the area representing that limb undergoes cortical degradation, causing more attention to be focused on the opposite intact limb. If a person’s brain is attending more strongly to a limb than it was prior to the amputation, it is possible that this increased awareness may enhance that limb’s deftness. Studies using repetitive transcranial magnetic stimulation of the motor cortex with recording of motor-evoked potentials in the upper limb have revealed that there is predominantly interhemispheric inhibition, and these inhibitor effects appear to be greatest for the neuronal assemblies that control the most distal muscles. 9,10
A study using transient local anesthesia of 1 hand has shown that during this anesthesia, the sensibility in the other hand increases. 6 To deftly manipulate objects, a person must have the capacity to feel how small precise movements of the limb influence the objects with which this limb is interacting. Because anesthesia temporarily “removes” the upper limb from the brain’s sensory systems and increases the sensibility of the other hand, it is possible that a permanent removal by amputation will elicit similar changes.
Studies of both nonhuman primates and humans have also revealed that the cerebral cortex of adults is capable of functional and structural plasticity. During the learning of new skills, cortical regions associated with sensorimotor function of the body parts that are most used for the skilled task enlarge. 11 After a person loses use of their dominant right hand, if they practice using their previously nondominant hand, this practice may induce changes in the motor and premotor cortex that mediate deftness of the nondominant left hand. Thus, following amputation of the dominant hand the nondominant hand might be able to develop a level of deftness that is greater than that of the nondominant hand of people who have not undergone amputation.
The influence of amputation on deftness has not been systematically studied. Therefore, the purpose of this study was to learn if amputation of the dominant right hand was associated with an increase or a decrease in deftness of the left-upper limb and if amputation of the nondominant left-upper limb was associated with an increase or decrease in the deftness of the right-upper limb. In addition, because with amputation there might be a shift of neighboring representational zones in both the primary somatosensory cortex (S1) and motor cortex (M1), we also wanted to learn if amputation of a lower extremity would improve deftness of an upper extremity or the contralateral lower extremity.
Methods
Participants
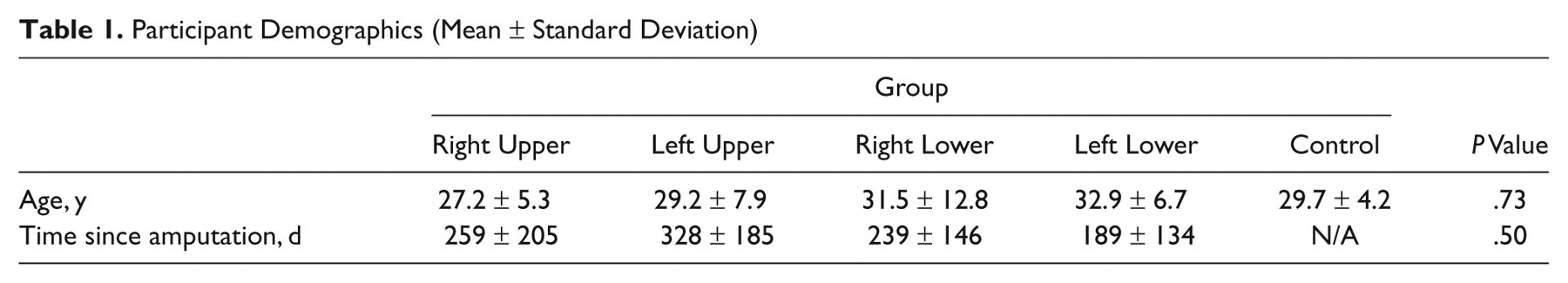
There were 37 right-handed (as determined using the Edinburgh Handedness Inventory) male participants, 31 who had either unilateral upper- or lower-limb amputations, and 6 healthy control participants. All participants were recruited from the Walter Reed Army Medical Center Amputee Clinic. This project received approval from the institutional review board, and all participants provided informed consent. The participants with amputations included the following: 6 right-upper-extremity (3 below elbow, 3 above elbow), 4 left-upper-extremity (2 below elbow and 2 above elbow), 10 right-lower-extremity (5 below knee and 5 above knee), and 11 left-lower-extremity amputees (6 below knee and 5 above knee). Further characteristics of the participants are described in Table 1. Excluding limb amputation, all participants had a normal neurological examination. All amputee participants were tested at least 2 months after their limb amputation.
Participant Demographics (Mean ± Standard Deviation)
Apparatus and Procedures
Although most studies of deftness–dexterity have focused on hand–finger functions during the performance of tasks, there are also requirements for precision of the more proximal portions of the upper limb. Thus, upper-limb deftness can be anatomically divided into 2 types: proximal and distal. We assessed distal (finger) deftness–dexterity in the intact upper limb using the coin rotation task and both proximal and distal deftness using the grooved pegboard. In the grooved pegboard task, picking up pegs and rotating them so they fit into a hole requires hand deftness, but lifting each peg from a well and carrying this peg to the target hole in another region as well as targeting the arm so it accurately reaches the correct hole also requires proximal arm deftness. Thus, we considered the grooved pegboard task (Lafayette Instruments, Lafayette, Indiana), 12 in which participants attempt to place grooved metal pegs into a 5 × 5 grid of key-shaped holes as fast as possible, as an indication of proximal arm and finger deftness. In the modified coin-rotation task, participants attempted to rotate a quarter as rapidly as possible for 30 s, and their score was the number of successful rotations. 13 To test lower-extremity deftness, we asked participants to hold a pen between the first and second toes and use their foot to draw independent circles (requiring lifting of the foot off the paper and moving it to a different location). The score for this task was the number of circles drawn in a 30 s.
Analyses
Demographic data comparing the participant groups were analyzed using a 1-way analysis of variance (ANOVA). The performance of participants with amputations was compared with that of controls by using Levene test to determine equal variances, followed by repeated-measures ANOVA. When ANOVA indicated statistical significance, we performed post hoc Tukey tests to determine among which pairs the significant differences lay. A Bonferroni correction was applied for multiple comparisons, with
Results
Levene test revealed equal variances among all participant groups (
Coin Rotation and Pegboard Performance by Left and/or Right Hand by Amputee Groupa
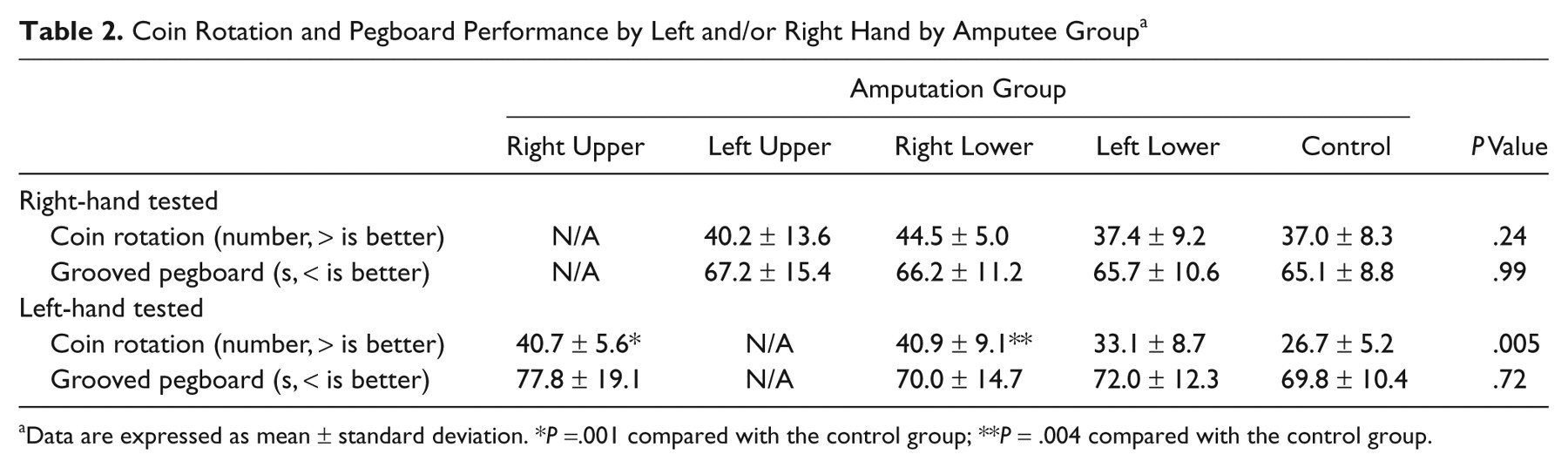
Data are expressed as mean ± standard deviation. *
When right-lower-limb amputees were compared with controls, they were also statistically significantly superior to the normal control participants in coin rotation performance when using their left hand (
Circle Drawing by Left and/or Right Foot by Amputee Group (Mean ± Standard Deviation)
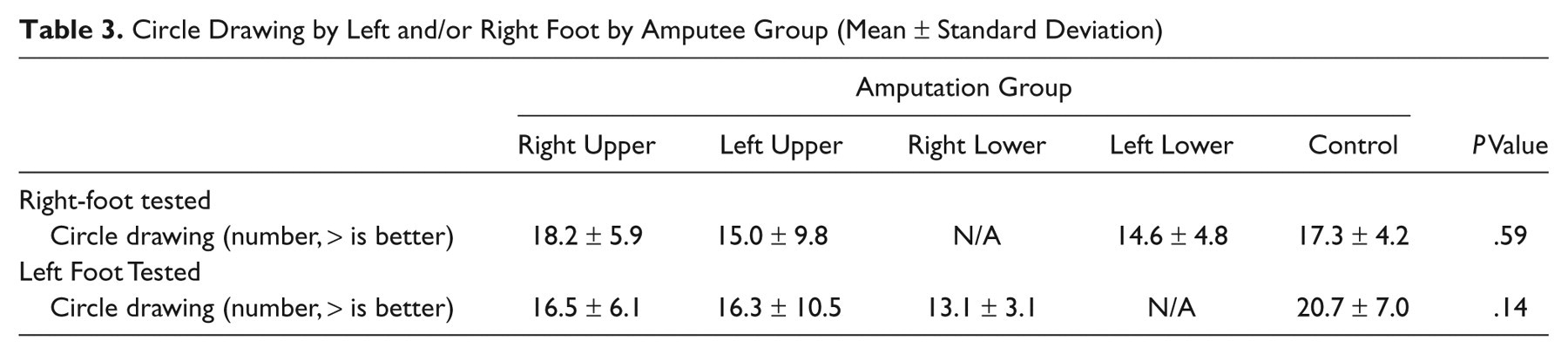
Discussion
This study demonstrates that amputation of the dominant right upper extremity leads to enhanced deftness of the remaining nondominant left hand. In contrast to improvement in the left hand seen with right hand amputation, removal of the left nondominant hand did not influence the deftness of the dominant right hand. The lack of effect on the right hand following left-upper-extremity amputation could be a result of a ceiling effect, given that all participants were right handed.
The reason amputation of the dominant right hand was associated with greater deftness of the left hand is not known, but there are several possible explanations. As mentioned above, amputation of the dominant right hand might have forced these participants to use their left hand to perform unimanual acts that were previously performed with their right upper limb. An increase in the frequency of tasks that required deftness of the left hand might have altered the hand representation in these patients’ right motor cortex. It is also possible, as mentioned above, that the amputation of the right upper limb improved these amputees’ attention or sensory feedback from their left hand.
This increased use, attentional, and sensory hypothesis cannot, however, fully account for the observation that amputation of the right lower extremity was also associated with superior deftness of the left hand’s fingers. Motor-evoked potential studies in healthy participants suggest that in right-handed people, the left motor cortex inhibits the right more than the right motor cortex inhibits the left, 14 and hence, it is possible that the physiological changes induced by right-sided amputation of either the upper or lower limb reduced this inhibition. Although the counterargument can be put forward that asymmetries in interhemispheric inhibition are challenged by the fact that right-lower-extremity amputees showed even higher right-hand coin rotation scores than left-hand coin rotation scores, the evidence in support of this disinhibition hypothesis mounts when we directly compare the performances of right-lower- and left-lower-extremity amputees. That is, the result that the right-lower, but not left-lower amputees’ left-hand coin rotation performance was statistically significantly superior to that of the controls cannot be fully explained by a deftness asymmetry in the controls. This result supports the postulate that the results are related to a reduction in the left hemisphere’s inhibition of the right hemisphere’s motor system.
The improvement in deftness of the left upper extremity was primarily noted on the coin rotation task, suggesting that this enhanced deftness is primarily for distal (finger) movements. The finding that there were no differences in the pegboard task suggest that there were no alterations in the deftness of the more proximal arm movements of the left upper limb. Brinkman and Kuypers 15 demonstrated in nonhuman primates that whereas distal movements are almost entirely controlled by the contralateral motor cortex, more proximal movements can be controlled by both the contralateral and ipsilateral motor cortices. Thus, if there is asymmetrical cortical motor representation of the limb following unilateral limb amputation, these differences would be more likely to influence distal than proximal movements, 16,17 which is what our data appear to demonstrate. However, further studies are needed to replicate these findings, and methods such as functional imaging may be used to test both the interhemispheric inhibition and distal (versus) proximal motor control hypotheses.
Footnotes
Acknowledgements
We wish to thank Ms Lindsay Hussey-Andersen and Ms Katie Hughes for assistance with subject testing and Ms Robin Howard for assistance with data analysis. KMS and JWT had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. The opinions or assertions contained herein are the private views of the authors and are not to be construed as official or as reflecting the views of the United States Department of the Navy, the Department of the Army, the Department of Defense, or the Department of Veterans Affairs.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
