Abstract
There is a proliferation of research examining the effects of exercise on mobility and cognition in the general population and those with neurological disorders as well as focal research examining possible neural mechanisms of such effects. However, there is seemingly a lack of focus on what it is about exercise, in particular, that drives adaptive central nervous system neuroplasticity. We propose a novel conceptual framework (ie, PRIMERS) that describes such adaptations as occurring via activity-dependent neuroplasticity based on the integrative processing of multisensory input and associated complex motor output that is required for the regulation of physiological systems during exercise behavior. This conceptual framework sets the stage for the systematic examination of the effects of exercise on brain connectivity, brain structure, and molecular/cellular mechanisms that explain improvements in mobility and cognition in the general population and persons with multiple sclerosis (MS). We argue that exercise can be viewed as an integrative, systems-wide stimulus for neurorehabilitation because impaired mobility and cognition are common and co-occurring in MS.
Introduction
Multiple sclerosis (MS) is a common neurological disease that is initially characterized by inflammatory processes and demyelination of axons in the central nervous system (CNS). The disease is further neurodegenerative, resulting in irreversible damage of gray and white matter. 1 This CNS damage accumulates over time and manifests as impairments in mobility and cognition, among other deficits.2,3 Of note, mobility and cognitive impairments are highly prevalent and often co-occurring, disabling, and poorly managed with disease-modifying pharmacotherapies in persons with MS.4,5 This highlights the importance of considering other approaches for managing mobility and cognitive disability in this population. One such approach is exercise training.
The effects of exercise (ie, planned, structured, repetitive physical activity performed with the goal of improving or maintaining one or more aspects of physical fitness) on mobility and cognitive outcomes are of interest in the MS population because there is a substantial body of evidence describing exercise-related improvements in mobility and cognition in healthy older adults. 6 This interest further is based on evidence that exercise training is efficacious for improving mobility and cognition in other neurological populations (ie, Parkinson’s disease, stroke, schizophrenia).7-9 Meta-analyses indicate that exercise training improves walking speed and endurance in MS.10-12 In comparison, one comprehensive systematic review reported overall conflicting evidence of exercise, physical activity, and physical fitness on cognitive outcomes in MS. 13 However, that review, coupled with evidence from other populations, spurred several emerging randomized controlled trials that support beneficial effects of exercise, physical activity, and physical fitness on cognitive performance in MS.14-16
There is substantial interest in studying underlying mechanisms of action for the promising, therapeutic effects of exercise training on behavior in general and particularly in persons with MS. As a starting point, we previously17,18 highlighted the roles of central (ie, brain and spinal cord) mechanisms for explaining the effects of exercise on mobility and cognition in MS, 17 given that cognition is a centrally mediated process and the regulation of walking requires higher-level brain function (ie, executive control) as well as spinal cord processes (ie, central pattern generation).19,20 Indeed, there is evidence of positive associations among cardiorespiratory fitness (as a cross-sectional surrogate for aerobic exercise training), physical activity levels, and volumes of subcortical brain structures that are important for both mobility and cognitive performance (eg, hippocampus, basal ganglia, thalamus) in MS.21-24 Another study reported associations among higher cardiorespiratory fitness, greater activation of the prefrontal cortex during a neurocognitive test, and better cognitive performance among persons with MS. 25 Furthermore, there is preliminary evidence of improved cognitive processing speed, walking endurance, hippocampal viscoelasticity (ie, shear stiffness), and thalamocortical resting-state functional connectivity based on functional magnetic resonance imaging (fMRI) with 12 weeks of aerobic treadmill walking exercise training in persons with MS.14,15,26 These observations are consistent with research in aging,27-29 Parkinson’s disease, 7 schizophrenia, 8 and stroke, 9 whereby exercise-related changes at the brain systems level co-occur with changes in mobility and cognition. This too is consistent with the substantial body of animal research that describes molecular/cellular brain changes (ie, upregulation of neurotrophic and vascular trophic factors) with exercise that accompany improvements in mobility and cognition.30,31 Collectively, this supports the likelihood that exercise-related improvements in mobility and cognitive functioning in MS are driven primarily by central processes.
The observation of putative exercise effects on central processes for explaining gains in mobility and cognition in MS and other populations begs the question, what is it about exercise, in particular, that might induce brain changes and secondary behavioral/performance improvements? By itself, the granular observations of central molecular and/or cellular changes (eg, hippocampal neurogenesis, synaptogenesis, angiogenesis) after exercise based on animal work do not provide an adequate explanation for the beneficial effects of exercise on brain systems and behavioral outcomes. Such mechanistic research is critical for providing a better understanding of what occurs in the brain with exercise training; however, this reductionist approach largely ignores integrative, systems-wide exercise-related neurophysiological processes that may drive such CNS adaptations. Exercise is a highly complex behavior that involves coordinated activation of nearly every physiological system in the human body that is regulated through an integrative effort of brain regions in the CNS. This behavior stimulates a multitude of sensory receptors and pathways and involves numerous interconnected neurophysiological processes that are regulated by the CNS. 32 For example, the successful execution of acute walking exercise involves ongoing locomotor control, visual processing, proprioception, spatial navigation, cardiopulmonary regulation, thermoregulation, mood regulation, and other processes.33,34 As physiological systems adapt to the increasing demands of chronic exercise training (ie, progressive, repeated bouts of acute exercise over time), the CNS must also adapt by more efficiently integrating the plethora of neurophysiological processes required for the successful regulation of exercise behavior over time. Those processes must be weaved together into the fabric of a successful bout of exercise, and the continual and repetitive integration of those processes over time with chronic exercise training might be the stimulus or reason why exercise training results in long-term adaptations within the CNS. Indeed, such CNS adaptations are likely to result in co-occurring adaptations in mobility and cognition, given that the regulation of exercise, mobility, and cognitive performance involve common neural circuitry.
We propose that the key to exercise-related brain adaptation over time might involve enhanced efficiency of communication within and across brain networks that are required for the regulation of the various interconnected neurophysiological processes involved in exercise behavior. This is consistent with an activity-dependent neuroplasticity framework wherein exercise-related brain adaptations occur based on improved connectivity between subcortical gray matter structures and neocortical structures because subcortical/cortical communication is a driver of several important neurophysiological processes associated with exercise (eg, locomotion, proprioception, thermoregulation, cardiorespiratory regulation, visuospatial navigation).32,34 In other words, the neural processing and integration of multisensory inputs and associated complex motor outputs that are required for the regulation of physiological systems during acute exercise behavior become more efficient over time with ongoing, repeated bouts of exercise (ie, chronic exercise training), and this results in improved subcortical/cortical connectivity and ultimately mobility and cognition (ie, the PRocessing and Integration of Multisensory Exercise-Related Stimuli [PRIMERS] conceptual framework).
The provision of a conceptual framework for elucidating a locus of the molecular, cellular, and brain system changes associated with exercise could provide a springboard for the systematic design, implementation, and optimization of exercise training approaches for improving mobility and cognition in MS and beyond. This is particularly relevant for the application of targeted exercise training for rehabilitating mobility and cognitive function among persons with MS and perhaps other neurological disorders. 17 This article initially proposes and discusses a novel, systems-wide conceptual framework (ie, PRIMERS) for characterizing integrative CNS plasticity with exercise by incorporating 3 distinct components from different bodies of literature for explaining what it is about exercise, in particular, that induces neural and behavioral changes over time. The first component for characterizing integrative CNS plasticity focuses on the processing and integration of multisensory input and associated complex motor output required for the regulation of physiological systems during exercise behavior in the healthy brain. The second and third components address how multisensory input and motor output associated with repeated (ie, chronic) exercise behavior lead to improved efficiency of communication within and across major neural networks and potential downstream improvements in mobility and cognition over time, respectively. Within this framework, molecular and cellular (ie, increased upregulation of neurotrophic and vascular trophic factors) cascades support such brain adaptations with exercise, rather than explain why such adaptations occur. A summary of this framework is presented in Figure 1. This article then proposes and discusses the PRIMERS conceptual framework that encapsulates observations in animal and human exercise research in general and neurological populations (including MS) describing exercise-related changes in brain structure and function. Finally, this article discusses the importance of the application and systematic testing of this conceptual framework in persons with MS and other neurological disease/disorders.
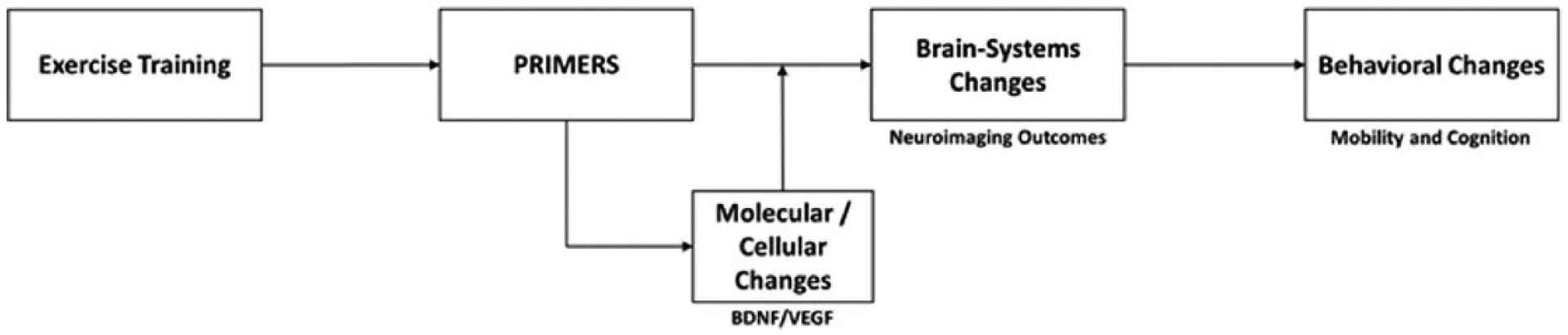
Summary of PRIMERS Conceptual Framework. Exercise training involves the continual processing and integration of multisensory input and associated complex motor output in neural networks that are important for regulating physiological systems during exercise behavior, mobility, and cognition, leading to changes in brain-systems (supported by molecular/cellular changes), which manifests as changes in mobility and cognition.
Integrative CNS Plasticity With Exercise
Exercise training is associated with beneficial behavioral changes (ie, improved mobility and cognitive performance)6,35 that are associated with adaptations in brain systems (ie, improved brain structure, function, and connectivity)27-29,36-40 and molecular/cellular (ie, increased upregulation of brain-derived neurotrophic factor [BDNF], neurogenesis, angiogenesis)30,31,41-50 processes. Of note, these adaptations are commonly observed in hippocampal and prefrontal cortical regions. However, those observations tend to be studied in isolation, as opposed to a systems-wide approach that provides an integrative explanation for CNS plasticity with exercise. Furthermore, molecular/cellular changes are often conceptualized as a primary driver of exercise-related CNS adaptations. 31 We propose a multicomponent conceptual framework that posits that the aforementioned adaptations with exercise training support an underlying process of increased efficiency of communication within and across selective neural networks that are critical for the regulation of physiological systems during exercise behavior by the CNS (Figure 1). Such increased efficiency of communication with exercise likely represents improved connectivity based on strengthened existing adaptive connections, formation of new adaptive connections, and/or removal of extraneous, maladaptive connections between brain regions. 51 The multisensory demands and integrated regulation of the human physiology with single bouts of exercise (Figure 2A) provide an ongoing challenge within the CNS that requires long-term adaptation within brain networks supporting mobility and cognition (Figure 2B). We first discuss this framework in the healthy brain below and then discuss its application in the MS brain in the following section.
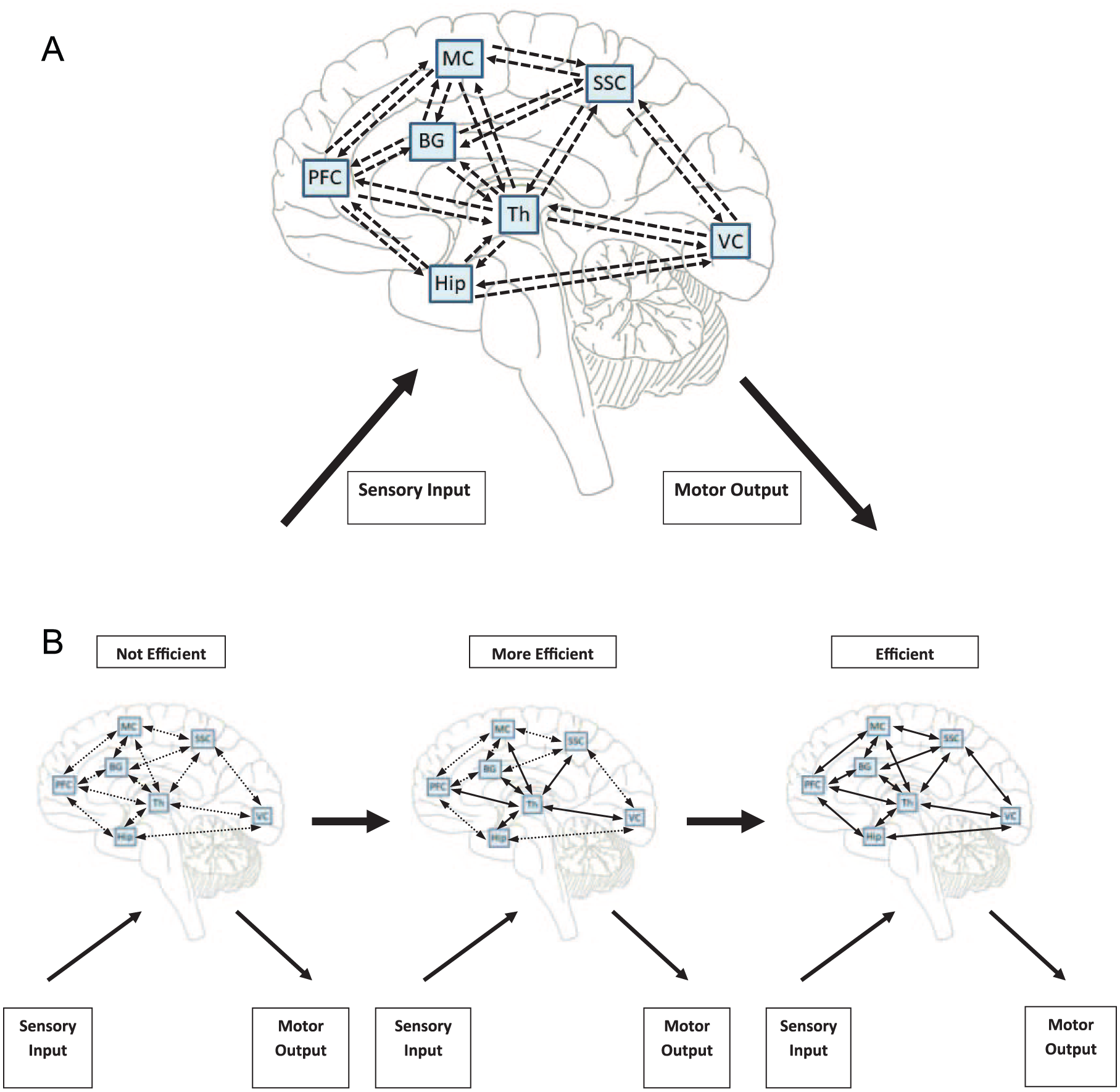
A. Summary of processing and integration of multisensory input and associated complex motor output within major neural networks required for the regulation of physiological systems during acute exercise behavior. The figure depicts simplified bidirectional communication among brain regions involved in visual processing, locomotor control, proprioception, and spatial navigation during acute exercise. For example, when processing visual stimuli, afferent pathways from the optic nerve project to the primary visual cortex via the lateral geniculate nucleus of the thalamus. Visual information is then integrated in the extrastriate cortex and projected to the prefrontal cortex via the dorsal (ie, posterior parietal cortex) and/or ventral (ie, medial temporal lobe) streams for further integration. Information in the prefrontal cortex is then projected to the motor cortex and basal ganglia (via the thalamus) for preparing a locomotor response via descending pathways (ie, corticospinal tract) to the spinal cord. Furthermore, somatosensory input is processed and integrated in the somatosensory cortex, which relays such sensory information to the thalamus for preparing an appropriate physiological response. This information is then relayed from the thalamus to the prefrontal and motor cortices for higher-level, context-dependent integration and preparation of an eventual motor response via the pyramidal tract, respectively. The hippocampus further interfaces with the thalamus and prefrontal cortex for spatial navigation processes associated with exercise behavior. Regarding locomotor control, the basal ganglia receives input from the thalamus and motor and somatosensory cortices. This information is then projected back to the thalamus, motor, and somatosensory cortices, which further project directly to the spinal cord for generating a motor response. B. Increasing efficiency of communication within and across neural networks that are important for the regulation of physiological systems during acute exercise behavior with chronic exercise over time. Dashed lines represent possible inefficient baseline connectivity (ie, weak existing connections, slowness of communication between regions, or indirect [maladaptive] connections between regions); solid lines represent more efficient baseline connectivity.
Multisensory Inputs and Complex Motor Outputs
Exercise is a highly complex and regulated behavior. The regulation of exercise behavior by the CNS involves the rapid processing of sensory input from multiple sources, the integration and processing of that information within the brain, and the associated efferent output for the maintenance of multiple body systems supporting the behavior. Importantly, this follows a feedback/feed-forward system. Using aerobic walking exercise as an example, this behavior involves the processing of afferent or sensory feedback from visual, vestibular, locomotor, proprioceptive, cardiopulmonary, thermoregulatory, and endocrine sources. This information is then relayed, processed, and integrated across selective brain regions (eg, motor cortex, basal ganglia for regulating locomotion, hippocampus, and/or posterior parietal cortex for spatial navigation) for generation of an appropriate output for the ongoing, moment-by-moment regulation of aerobic walking behavior given specific environmental demands. Such sensory feedback further is integrated in higher-level brain regions (eg, prefrontal cortex) because the successful regulation of walking requires executive control processes19,20,52 even in MS. 53 Of note, the integration of such multisensory input involves interconnected, often bidirectional (reciprocal), projections across brain structures (eg, thalamus, basal ganglia, prefrontal cortex, motor cortex). 34 Efferent projections from the brain then relay/transmit this information through the spinal cord pathways and networks (ie, via pyramidal, corticospinal tracts) and eventually motor effectors (ie, central pattern generation) for the continuation and maintenance of aerobic walking exercise. This is summarized in Figure 2A. We note that cardiorespiratory regulation during exercise involves different afferent/efferent pathways (ie, via projections involving the medulla oblongata) than those described above; this is beyond the scope of the current article and can be reviewed elsewhere. 54 We further acknowledge the roles of the cerebellum and spinal cord in the maintenance of balance and adaptation/control of locomotion, respectively55,56; this too is beyond the scope of the current article. Importantly, at the center of this traffic jam is the thalamus—the brain’s relay station—which as a collection of nuclei, is a critical node for numerous sensory afferent and motor efferent pathways. Importantly, these feedback and feed-forward loops are constantly operating and adjusting to ever-changing environmental demands for regulating behavior throughout a single bout of exercise. We propose that the continually changing environmental demands of exercise might be the stimulus that requires the CNS to continually integrate multiple neurophysiological processes in a coordinated manner and causes adaptations at the molecular/cellular, brain systems, and behavioral levels that result in chronic changes with exercise training. Given equivocal evidence regarding exercise-related changes in cerebral blood flow and its relationship with changes in behavior, it is unlikely that such adaptations are a direct product of increased cerebral oxygenation. 57
Communication Efficiency Is Key
During exercise, maximizing the efficiency of the feedback/feed-forward systems for processing and integrating multisensory input and associated complex motor output is critical for maintaining homeostasis, maximizing safety and stability, and optimizing physical performance. 58 This places a premium on the efficiency of brain connectivity (ie, maximizing strength of adaptive connections while minimizing communication time and metabolic expenditure), 59 given the neurophysiological demands that are required for the coordinated regulation of exercise. Indeed, the organization of the human CNS is likely based on maximizing efficiency of communication (ie, improved connectivity) for optimizing cognitive functioning.60,61 In particular, the brain is considered to be organized based on a small-world architecture, with a high level of local connection, along with several long-distance connections to maximize efficiency of both local and global communication between regions.60-62 Such a perspective further might extend to the CNS’s adaptations to exercise training. 58
Exercise behavior, in particular, involves numerous processes that require efficient, often bidirectional communication in networks with far-apart brain hubs. This includes, among others, the dorsal and ventral visual streams, corticospinal tract, motor pathways, and somatosensory pathways. Exercise behavior requires a high degree of communication between subcortical and cortical structures. The degree of communication between subcortical and cortical structures during exercise further might depend on the demand characteristics of the exercise bout itself. For example, exercise modalities that involve greater environmental demands (eg, walking on a treadmill versus riding a stationary cycling) likely require additional sensory input, integration, and motor outflow. The more complex the exercise behavior (ie, the more diverse array of sensory receptors that are stimulated), the greater the demand for efficient communication (ie, better connectivity) between subcortical and cortical brain hubs.63,64
Activity-Dependent Neuroplasticity
In exercise physiology, the acute effects of a single bout of aerobic exercise on the cardiorespiratory system (ie, the network involving oxygen delivery and use, rather than isolated organs) are additive and cumulative when delivered in a chronic, longitudinal intervention. 65 It further is well established that those adaptations in oxygen delivery and use translate into secondary physiological benefits. 66 Perhaps this is similar for brain connectivity across a neural network, wherein the continual processing and integration of multisensory input and associated complex motor output requisite for acute exercise induces small, beneficial adaptations (ie, improvements) in baseline subcortical-cortical brain connectivity. These small beneficial changes may accumulate over time with chronic exercise training and result in meaningful adaptations. This is similar to the concept of motor learning 67 and is consistent with notions of activity-dependent neuroplasticity 68 and Hebbian learning (ie, repeated stimulation of synapses increases their strength). 69 We note that it is not known whether Hebbian learning is the actual process required for the hypothesized exercise-related improvements in connectivity. Nevertheless, improved subcortical/cortical connectivity across neural networks over time likely is supported by molecular/cellular changes (ie, increased upregulation of neurotrophic and vascular trophic factors, dendritic arborization) with exercise. In comparison, it is unlikely that exercise-related changes in the upregulation of neurotrophic and vascular trophic factors are dependent on changes in brain connectivity. 70 As such, the systematic examination of molecular/cellular mechanisms is not a primary component of the current framework because exercise-related molecular/cellular changes are secondary and supportive within the context of an integrative, systems-wide approach for characterizing CNS plasticity with exercise.
Although the relationship between behavioral change and neuroplasticity is not 1:1, under certain circumstances, behaviors can induce changes in brain circuitry, which may in turn, lead to adaptations at the behavioral level. 68 That is, repetitive, specific practice might induce specific neuronal adaptations that may result in improved behavioral performance specific to the domain of practice (ie, activity-dependent neuroplasticity). We note that activity-dependent neuroplasticity is not always adaptive because some behaviors can induce maladaptive neuronal changes that may result in worsened behavioral outcomes. 71 Activity-dependent neuroplasticity has been highlighted as a model describing the neural underpinnings of the development of new motor skills, 72 musical expertise, 73 and pain hypersensitivity 74 and has been used as a framework for rehabilitation interventions (ie, constraint-induced movement therapy in stroke). 75 Applications of activity-dependent neuroplasticity are based, in part, on the concept of Hebbian learning wherein the repeated stimulation of a synapse eventually improves its strength. 69 In other words, “neurons that fire together, wire together.” Perhaps the best-characterized example of Hebbian learning is long-term potentiation in the hippocampus. 76
Within the context of activity-dependent neuroplasticity, a single bout of exercise presumably involves the repetitive stimulation (ie, activation) of synapses involved in neural networks that are critical for the regulation of physiological systems during exercise behavior over the duration of the exercise bout. This might result in slightly improved baseline connectivity (ie, adaptive activity-dependent neuroplasticity) in brain regions that are important for mobility and cognitive performance; this idea is consistent with observations of transient, beneficial effects of acute aerobic exercise on those outcomes.77,78 With chronic exercise training, perhaps improvements in mobility and cognition are a product of improved baseline connectivity within neural circuits that are repetitively and continually stimulated with bouts of acute exercise (ie, Hebbian learning; Figure 2B). This is analogous to an orchestra, wherein individual musicians must be repetitively and specifically trained over time to play together to create a symphony, rather than a cacophony. Such transfer effects with chronic exercise training are plausible considering that baseline connectivity has been proposed to reflect intrinsic brain activity as opposed to situational, task-related brain activation. 62 Of note, it is possible that exercise-related improvements in connectivity might reflect pruning of extraneous, maladaptive connections between brain regions in addition to strengthened existing adaptive connections or formation of new adaptive connections between regions; those processes are likely supported by the increased upregulation of neurotrophic and vascular trophic factors. 31 Given the importance of efficient communication within and across neural networks for the regulation of exercise, improved connectivity with chronic exercise training provides an explanation for observations of selective improvements in speed-related mobility and cognitive outcomes in adults. 79
One challenge of adopting an activity-dependent neuroplasticity framework within the context of explaining integrative CNS plasticity with exercise involves the interplay between specificity and generalizability of transfer effects. For example, the neural circuits that are continually activated by exercise would be most likely to adapt (ie, perhaps based on improved connectivity). As such, functions that are closely associated with those neural networks are likely to improve with exercise training. However, consistent with the notion of activity-dependent neuroplasticity, 68 the degree of improvement will be larger for functions that are more proximal to exercise behavior (ie, cardiorespiratory capacitance) and smaller for functions that rely on the activated neural networks, but are more distal from exercise behavior (ie, cognitive performance). Furthermore, it would not be expected that all functions associated with the activated neural networks would improve with behavioral intervention. This is consistent with research demonstrating larger exercise-related improvements in cardiorespiratory fitness than the magnitude of changes in cognitive performance.27,29 Based on this framework, there also exists the possibility of cross-modality transfer, wherein non–exercise behaviors might repetitively activate a particular neural network that is involved in the regulation of exercise behavior, resulting in adaptations at the behavioral level. For instance, there is evidence in older adults wherein cognitive rehabilitation (ie, computerized training of attention and executive functions) improves mobility, 80 though cognitive rehabilitation is likely to result in larger improvements in cognitive performance than mobility. 81
The PRIMERS Conceptual Framework: Enhanced Connectivity as a Basis for Brain Adaptations With Exercise Training
The examination of mechanisms for brain adaptations with exercise training tends to focus on molecular and cellular mediators (ie, BDNF, vascular endothelial growth factor [VEGF]) and processes (ie, neurogenesis, angiogenesis), but there is seemingly a lack of focal inquiry on integrative CNS plasticity with exercise, wherein the aforementioned phenomena are merely scaffolding of a larger, systems-wide picture. We propose that observations of exercise-related upregulation of BDNF and VEGF and the subsequent processes of neurogenesis and angiogenesis, respectively,30,31,41-50 might represent adaptations that support communication within and across neural networks that become more efficient with chronic exercise training. That is, within the context of the PRIMERS conceptual framework, the molecular/cellular adaptations related to exercise training may be a consequence of the repeated, feedback/feed-forward processing and integration of multisensory input and associated complex motor output necessary for the regulation of physiological systems during exercise behavior (Figure 1). With chronic exercise training, the aforementioned improvements in subcortical/cortical baseline connectivity within and across select brain networks (Figure 2B) would be supported by molecular/cellular processes (ie, increased upregulation of neurotrophic and vascular trophic factors). We apply this integrative conceptual framework for explaining brain adaptations with exercise based on exemplar animal and human research focusing on the hippocampus, prefrontal cortex, and thalamus, respectively, below.
Voluntary wheel running is robustly and selectively associated with the increased upregulation of BDNF, neurogenesis, synaptogenesis, long-term potentiation, and angiogenesis in the dentate gyrus of the hippocampus and concomitant improvements in motor and cognitive performance among animals.31,41,42 By extension, human studies support exercise training for increasing hippocampal volume and hippocampal blood volume using neuroimaging paradigms as mechanisms for exercise-related improvements in learning and memory.29,48 Importantly, for the regulation of exercise behavior, rapid and efficient communication is required in several neural networks that involve the hippocampus. For example, aerobic walking exercise presumably involves processing and integration of complex visual information (ie, involving both the dorsal and ventral visual pathways). The ventral visual stream involves projections from the visual cortex through the medial temporal lobe and eventually to the ventromedial prefrontal cortex, with the hippocampus possibly playing a role in pattern separation of perceived objects. 82 Aerobic walking exercise involves some degree of spatial navigation, another hippocampally mediated process, that involves occipital, mediolateral parietal, and posterior cingulate regions. 83 Of note, the degree of spatial navigation associated with walking exercise is likely reduced if performed on a standard treadmill compared with overground walking exercise 63 or possibly exercise with virtual reality. 84 The regulation of acute walking exercise behavior places a premium on efficient subcortical/cortical communication (ie, efficient connectivity) within these neural circuits. With chronic aerobic walking exercise training, perhaps the repetitive stimulation of specific neural networks involving the hippocampus results in increased efficiency of subcortical/cortical communication within those networks (ie, improved connectivity). Hypothetically, this improved connectivity would require the upregulation of neurotrophic and vascular trophic factors for subsequent hippocampal neurogenesis/angiogenesis that support the demands of more efficient baseline communication within neural networks involving the hippocampus. Indeed, aerobic walking exercise training is associated with increased resting-state functional connectivity between the hippocampus and anterior cingulate cortex, in addition to increased hippocampal cerebral blood flow in older adults. 85 Because the hippocampus is a structure that is highly plastic, 86 the PRIMERS conceptual framework can provide an integrative, systems-wide characterization of exercise-related increases in hippocampal volume and downstream improvements in learning and memory.8,29 Within the context of this conceptual framework, observations of increased functional connectivity between the hippocampus and anterior cingulate cortex with exercise 85 could be attributed to enhanced involvement of the salience network (ie, an intrinsic neural network involved in self-awareness via integration of somatosensory, emotional, and cognitive stimuli) 87 in the regulation of exercise behavior, leading to downstream hippocampal and behavioral adaptations.
The prefrontal cortex is a brain structure that is critical for several neurophysiological processes that are required for the regulation of exercise behavior. Indeed, recent functional near-infrared spectroscopy (fNIRS) evidence indicates that this structure is particularly important for the regulation of walking exercise by the CNS in the general population19,20,52,88 and in persons with MS. 53 The prefrontal cortex further is a critical functional hub for 2 major behavioral networks (ie, default-mode and executive control networks). 87 Interestingly, chronic exercise training in humans is associated with increased prefrontal cortical volume 30 and activation 27 as well as increased resting-state functional connectivity within this region 39 and concomitant improvements in mobility and executive functioning (ie, a cognitive domain that is largely tied to the prefrontal cortex). 89 There is a large body of evidence suggesting small, transient improvements in speed-related executive functions with an acute bout of aerobic exercise.77,90 Accordingly, the PRIMERS conceptual framework can provide a systems-wide, integrative characterization of prefrontal cortical adaptations with exercise that encapsulates focal observations of improved prefrontal cortical structure and function. Returning to the example of aerobic walking exercise, this behavior requires the processing and integration of complex visual information (involving bidirectional, reciprocal communication between subcortical structures and dorsolateral and ventromedial prefrontal cortices), 32 somatosensory and proprioceptive input (involving bidirectional communication between prefrontal regions and other cortical [ie, motor, somatosensory, posterior parietal cortices] and subcortical [ie, thalamus, cerebellum] regions), 32 and regulation of locomotion (involving bidirectional communication between the prefrontal and motor cortices with subcortical regions (ie, basal ganglia, thalamus)), among other neurophysiological processes. Within the context of the PRIMERS conceptual framework, the repetitive activation of those selective neural networks with chronic aerobic walking exercise training results in improved baseline connectivity over time, as has been observed in older adults. 39 This improved connectivity further might require more efficient activation of the prefrontal cortex to successfully complete motor and cognitive tasks, or even at rest.27,39 In turn, such prefrontal brain adaptations likely provide a neural substrate for exercise-related improvements in mobility and cognitive performance.
One central theme of the myriad neurophysiological processes involved in the regulation of exercise behavior is the role of the thalamus as the brain’s relay station (in addition to other roles in information processing) for efficiently communicating between subcortical and cortical brain regions.91,92 Highlighting the thalamic demands of exercise behavior, one interesting study reported increased thalamic firing rates with increased locomotor speed and visual stimulation during normal locomotion. 93 Based on the PRIMERS conceptual framework, we propose that efficient communication within thalamocortical networks, in particular, might be among the most important processes for the regulation of exercise behavior and thus a potentially robust neural substrate for explaining exercise-related improvements in mobility and cognition. Indeed, motor training has resulted in increased thalamic synaptogenesis in rodent models. 94 However, despite preliminary evidence describing exercise-related improvements in cognitive processing speed in MS, 14 we are unaware of any studies reporting increases in thalamic volume or activation during cognitive tasks with chronic exercise training in humans. Interestingly, there are very few studies that have even examined baseline or task-related connectivity within thalamocortical circuits as a neuroimaging outcome in exercise training studies in any population.36,95 The lack of focus on thalamocortical connectivity in exercise research, furthermore, is somewhat puzzling, considering that communication within various thalamocortical networks is improved with other behavioral training interventions that involve processing and integration of multisensory input (ie, cognitive rehabilitation).96,97
Summary
Considering the growing body of evidence describing exercise-related improvements in mobility and cognition, as well as focal interest on specific brain adaptations with exercise, we propose a novel conceptual framework that adopts a systems-wide approach for characterizing integrative CNS plasticity with exercise. Critical components of this framework include the multisensory experience with exercise, importance of efficient communication within and across neural networks for the regulation of physiological systems during exercise behavior, and activity-dependent neuroplasticity. The PRIMERS conceptual framework itself (Figure 1) proposes that the repeated processing and integration of multisensory input and associated complex motor output with chronic exercise training results in improved subcortical/cortical baseline connectivity within and across neural networks that is likely supported by a molecular/cellular milieu (ie, molecular/cellular mechanisms are secondary to the multisensory exercise experience). We further apply the framework for explaining previous observations of brain adaptations with exercise training (ie, resulting in changes in mobility and cognition) in the healthy brain with a particular focus on the hippocampus, prefrontal cortex, and thalamus. Although the majority of such research involves healthy samples, this framework is particularly relevant for neurorehabilitation research in persons with MS and other neurological disorders.
Implications for Rehabilitation in Persons With MS and Other Neurological Disorders
As cognitive and mobility impairments are hallmark consequences of MS,4,5 exercise training represents a highly promising approach for managing these interrelated functions. 17 MS pathophysiology is associated with subcortical (ie, hippocampal, thalamic) and cortical (ie, neocortex) atrophy, reduced white matter integrity (eg, reduced fractional anisotropy), and disruptions in connectivity in major brain networks that are important in the regulation of exercise, mobility, and cognitive performance.98-104 Based on such a pattern of neural damage, MS might represent a particularly promising platform for systematically examining tenets of the PRIMERS conceptual framework. Indeed, this integrative framework can provide a basis for elucidating the neural underpinnings for observations of beneficial effects of exercise on mobility10-12 and cognition 13 in MS.
The most common and disabling cognitive impairment in persons with MS is slowed cognitive processing speed. 5 This presumably would have deleterious effects on the regulation of exercise behavior in persons with MS-related cognitive processing speed impairment, given the premium on efficient communication within and across major neural networks during acute exercise. MS-related cognitive processing speed impairment is associated with extraneous, maladaptive connections as well as fewer and weaker adaptive connections in neural networks that are important for the regulation of exercise behavior and other functions.102,103 In the case of poor baseline connectivity in networks associated with cognitive processing speed impairment in MS, there may be an even stronger link between exercise, cognition, and mobility than in the healthy brain, given evidence of cognitive-motor coupling being stronger in MS than in healthy controls. 3 That is, within the context of the PRIMERS conceptual framework, the neurophysiological sensorimotor demands of exercise may have a particular affinity for improving mobility and cognition (ie, cognitive processing speed) in those with MS-related cognitive processing speed impairment by strengthening existing adaptive connections, forming new adaptive connections (possibly forming connections with homologous regions of the contralateral hemisphere, in addition to new subcortical/cortical connections), restoring reversed connections, and pruning extraneous, maladaptive connections over time. This is consistent with data indicating stronger associations between cardiorespiratory fitness (as a cross-sectional surrogate for aerobic exercise training) and cognitive processing speed in persons with MS-related processing speed impairment than in non–processing speed impaired persons with MS. 105 However, there exists the scenario wherein the MS disease process is so severe that it overwhelms the capacity for exercise training to induce adaptive neuroplastic changes in connectivity and downstream improvements in mobility and cognitive functioning. In this case, communication in networks important for the regulation of physiological systems during exercise behavior may be so inefficient that the repeated processing and integration of multisensory input and associated complex motor output fails to improve baseline connectivity. Indeed, this is consistent with data indicating selective associations between measures of cardiorespiratory fitness, physical activity, and cognitive processing speed in persons with mild, but not severe, MS disability.106,107 We further note that it is not known whether or not the presentation of MS-related fatigue would independently affect the effects of exercise training on mobility and cognition. Given that fatigue is a complex phenomenon that affects brain function, mobility, and cognition in MS,108,109 future research efforts might consider the role of severe MS-related fatigue when testing the PRIMERS conceptual framework.
Understanding putative systems-wide and focal neural mechanisms of exercise effects on mobility and cognition in MS is important from both a research and clinical perspective. Exploring the selective effects of exercise behavior on neural networks that lead to beneficial changes in behavior can serve to inform the development of optimized rehabilitative interventions, wherein a single intervention may improve multiple domains of functioning. 17 Within the context of the PRIMERS conceptual framework, exercise training intervention approaches that involve processing and integration of certain sensory information (eg, treadmill walking exercise training) would particularly improve baseline connectivity within associated neural networks leading to specific behavioral (ie, mobility and/or cognition) adaptations. By extension, this framework posits that exercise training interventions that involve a greater degree of neurophysiological sensorimotor demands should result in more robust brain and behavioral adaptations because the CNS would need to coordinate and integrate more neurophysiological processes to successfully regulate and maintain the exercise behavior. Such behavioral adaptations from exercise training may be particularly robust given that improved connectivity within neural networks that are critical for the regulation and maintenance of exercise behavior are also critical for regulating aspects of mobility and cognitive performance.32,62
The adoption of the PRIMERS conceptual framework within MS exercise research presents a promising integrative and network-based approach for rehabilitating specific deficits that are common and debilitating among persons with MS using targeted and optimized exercise training interventions. This further has clinical relevance because testing the aforementioned framework might provide a biological basis for clinicians prescribing exercise for managing cognitive and mobility deficits in MS.110,111 This might strengthen the patient-provider interaction, which has been recognized as an important component for persons with MS to realize the benefits of exercise behavior. 111 Indeed, the integrative, systems-wide examination of the effects of exercise behavior on CNS plasticity might better position exercise as a possible countermeasure for CNS decline in MS and/or an adjunct to approved disease-modifying therapies. 112 Ultimately, the PRIMERS conceptual framework can provide guidance for clinicians for potentially modifying existing exercise rehabilitation approaches to optimize mobility and cognitive outcomes among persons with MS. Possible approaches for manipulating exercise stimuli for maximizing mobility and cognitive benefits are discussed in the section below.
The PRIMERS conceptual framework of integrative CNS plasticity with exercise further is important for positioning exercise training as a rehabilitative approach for improving mobility and cognition in other neurological disorders that present with alterations in brain structure and function (eg, stroke, Parkinson’s disease). There is evidence that exercise is associated with increased resting-state functional connectivity and concomitant improvements in mobility and cognition in rodent models of Parkinson’s disease 113 as well as other work describing exercise benefits on brain integrity in rat models of ischemic stroke.114,115 Testing the PRIMERS conceptual framework in neurological populations beyond MS further is important, considering that the effects of exercise on mobility and cognition at the molecular/cellular, brain-systems, and behavioral levels may vary as a function of differing pathophysiological mechanisms. Providing disease-specific, empirical evidence on brain adaptations with exercise training from an integrative, systems-wide perspective would be incredibly useful for the creation of stepped-care models and decision trees for managing mobility and cognitive dysfunctions associated with those neurological disorders.
Future Directions: Testing the PRIMERS Conceptual Framework
The complete testing of the PRIMERS conceptual framework will likely involve the implementation of both acute and chronic exercise paradigms in the general population as well as in persons with MS and other neurological disorders. Using both acute and chronic exercise paradigms allows for systematic research efforts to strategically address certain aspects of the PRIMERS conceptual framework in order to provide a complete test. We now describe several experimental approaches and outcome measures for guiding the comprehensive testing of the conceptual framework.
Multisensory Inputs and Complex Motor Outputs
At the behavioral level, an interesting approach for examining the importance of multisensory input and complex motor output with exercise behavior involves manipulating the sensory input associated with exercise. As different modalities of exercise clearly involve different levels of sensory input, 63 one research endeavor that warrants consideration is examining the acute effects of different modalities of exercise on cognitive and mobility outcomes. Within the context of the aforementioned framework, more “complex” exercise modalities (eg, treadmill walking vs seated cycle ergometry) might involve a greater degree of processing and integration of multisensory input and associated complex motor output, leading to larger acute improvements in mobility and cognition. Interestingly, this is consistent with data in MS that indicate larger acute improvements in inhibitory control for walking compared with cycling exercise. 116 Relatedly, the manipulation of multisensory input using an acute exercise paradigm could also occur via adding visual and/or auditory stimuli (ie, music) and comparing the effects on neuroimaging or behavioral measures with exercise “alone.” Such a manipulation of sensory input further can include comparisons of group exercise (ie, environmental enrichment) versus exercise in isolation, comparisons of free-living physical activity (ie, in the real-world) versus supervised exercised training (ie, in a gym setting), comparisons of sport-specific training versus “general” exercise training, and/or comparisons of differential rates of exercise progression. As the addition of new stimuli dramatically increases the complexity of neural networks involved in behavior, we hypothesize that additional sensory stimuli will require additional communication within and across neural networks involved in both mobility and cognition during exercise and result in larger acute improvements in mobility and cognition compared with “standard” exercise alone.
The impact of multisensory input and complex motor output associated with different exercise modalities on mobility and/or cognition can be addressed with chronic exercise training research. For example, future clinical trials can compare the effects of different modalities of exercise training (yoking the conditions on exercise volume) or different levels of “complexity” of the same exercise modality on behavioral outcomes over time. Alternatively, an approach for testing the PRIMERS conceptual framework might involve examining the comparative and combined effects of exercise training and cognitive rehabilitation on mobility and cognitive outcomes, 17 because a combined intervention approach involves additional processing and input of multisensory input compared with each intervention in isolation. If the PRIMERS conceptual framework is correct, such a combined intervention approach (ie, based on additional multisensory inputs and complex motor outputs) should be optimal for improving both mobility and cognition. Relatedly, another test of the PRIMERS conceptual framework might involve comparisons between exercise training and other behavioral rehabilitation outcomes that involve continually changing environmental demands (eg, art, music therapy) on brain connectivity, mobility, and cognitive outcomes. Indeed, as the multisensorial experience of exercise presumably involves the coordinated activation of a larger number of neural circuits than other less physiologically demanding rehabilitative therapies, it is hypothesized that exercise training would result in greater improvements in brain connectivity in circuits that are highly involved in mobility and cognition as well as greater improvements in those behaviors.
Acute and chronic exercise approaches for examining the effects of differential multisensory inputs and complex motor outputs (ie, the first component of the PRIMERS conceptual framework) can also focus on network-level outcomes via the inclusion of neuroimaging end points. One particularly promising technique for addressing this aspect of the framework involves the use of fNIRS. fNIRS is advantageous for measuring cortical activation (ie, based on oxygenated hemoglobin) during exercise because it is less sensitive to motion artifact than other neuroimaging modalities. 117 Future research efforts might consider examining activation in key neural regions (eg, prefrontal, motor, and somatosensory cortices) during single (ie, acute) bouts of exercise that involve differing levels of multisensory input in persons with MS or other neurological disorders. Indeed, there is evidence of greater activation of the prefrontal cortex during acute walking exercise in healthy and neurological samples using fNIRS.20,52,53,118 One limitation of fNIRS is that it can only penetrate 2 to 3 cm into the cortex, 119 so it is not possible to examine activation in portions of networks that might fully reflect subcortical/cortical communication (ie, thalamus) during exercise. Nevertheless, measuring cortical activation during exercise using fNIRS might provide initial, albeit incomplete, evidence supporting selective activation of certain neural regions that are presumably involved in the regulation of exercise behavior as well as mobility and cognitive performance.
Communication Efficiency Is Key
A critical aspect of the PRIMERS conceptual framework is the importance of enhanced efficiency of communication within and across neural networks required for the regulation of physiological systems during exercise behavior. In neuroscience research, the concept of efficiency is often interpreted incorrectly. 59 To establish that communication (connectivity) truly becomes more efficient, the strength of connectivity between 2 regions must increase and the number of pathways between regions and metabolic expenditure of connectivity must decrease relative to baseline.51,59,62 Given that this aspect of the framework occurs at the network level and is an intermediary of the effects of exercise on behavior, future high-quality experimental tests of enhanced connectivity will presumably rely on neuroimaging techniques as primary outcome measures. Testing this component of the framework further is critical, given that the exact mechanism of the proposed exercise-related improvements in baseline connectivity is unknown.
One possible marker of this communication includes resting-state functional connectivity neuroimaging measures (ie, correlations in spontaneous brain activation [BOLD signal] between remote brain regions in the absence of a task) based on fMRI. 62 A focus on resting-state functional connectivity might be particularly advantageous because it presumably represents core neuroplastic changes, rather than situational, task-specific adaptations. 120 We further speculate that the increased efficiency of subcortical/cortical communication (based on improved resting-state functional connectivity) with exercise training leads to structural brain changes over time (ie, increased subcortical deep gray matter volume, increased prefrontal cortical volume). This has relevance for persons with MS, considering that there is evidence that suggests that changes in hippocampal function (ie, activation and connectivity) predate MS-related memory impairment and hippocampal atrophy.121,122 Importantly, we note that it is not known if functional connectivity changes precede structural brain changes in response to any behavior/intervention. However, this notion is consistent with observations in the general population, whereby acute exercise is associated with changes in blood flow perfusion, task-related brain activation, and resting-state functional connectivity, but not changes in brain volume or integrity.49,78 In comparison, human studies of chronic exercise training have reported changes in brain structure, integrity, function, and connectivity.27,39,87 Within the context of the PRIMERS conceptual framework, it is possible that exercise might influence functional brain changes, but not volumetric changes of certain structures (eg, basal ganglia) 29 within networks that are important for the regulation of exercise behavior, mobility, and cognition.
A related neuroimaging technique that might be particularly useful for examining the possible enhancement of efficiency of communication in response to exercise involves effective connectivity based on fMRI. Effective connectivity is advantageous over functional connectivity measures in that effective connectivity can provide information on the directionality of connections between regions either at rest or during execution of a task. Indeed, examining effective connectivity before and after an exercise training intervention can provide information on the mechanisms of improved connectivity (eg, reversal of maladaptive connections) and how brain connectivity might change during the execution of cognitive tasks. Within the context of the PRIMERS conceptual framework, the application of such paradigms could provide a neural substrate for quantifying the degree of improvement of efficiency of communication that is needed for meaningful behavioral adaptations to occur. Effective connectivity measures further can be included in acute exercise paradigms.
Another important issue for consideration is that the PRIMERS conceptual framework highlights the importance of subcortical/cortical connectivity as part of integrative CNS plasticity with exercise. Within this context, thalamocortical resting-state functional connectivity or effective connectivity might be particularly sensitive neuroimaging measures to exercise training, given the large role of the thalamus in efficiently processing and integrating multisensory input and the associated complex motor responses in the regulation of exercise behavior. Curiously, in human studies, there has been a general lack of focus on thalamocortical resting-state functional connectivity or effective connectivity as neuroimaging outcomes in exercise studies, as has been done in cognitive rehabilitation research.96,97,123 Although some research has described no significant increases in thalamic volume with exercise training, 29 perhaps exercise-related thalamic changes are more related to connectivity, rather than volume, given a role of the thalamus as the brain’s relay/integration station in addition to other roles in information processing across multiple neural networks. More focal research in this area is needed, especially in neurological disorders such as MS that involve thalamic atrophy 124 and disrupted thalamic connectivity.102,125
Using such neuroimaging techniques, one exemplar experimental protocol for testing this aspect of the framework involves performing fMRI immediately before and after a single bout of exercise or a control comparison condition for measuring exercise-related changes in resting-state functional connectivity, cerebral blood flow perfusion (using pseudo-continuous arterial spin labeling techniques), or effective connectivity in select neural networks, particularly those that involve both subcortical and cortical communication. In particular, resting-state functional connectivity measures are highly sensitive to experience 120 and represent intriguing candidates for inclusion as primary outcome measures in an acute exercise paradigm. Although it is not feasible to adopt such an approach during actual exercise because of the setup of MRI and sensitivity of MRI to motion artifact, those MRI techniques (ie, including both pre-exercise and postexercise MRI) might be advantageous for measuring exercise-related adaptations in the efficiency of communication at the network level. Such a study could provide information as to which neural networks are activated in response to a single bout of exercise relative to a control comparison condition. This could then inform the development of other research examining secondary exercise training–related adaptations at the molecular/cellular, brain-system, and behavioral levels over time, lending support for the final aspect of the PRIMERS framework (ie, activity-dependent neuroplasticity).
Activity-Dependent Neuroplasticity
Within the context of integrative CNS plasticity with exercise training, the efficiency of communication within select neural networks for the regulation of physiological systems during exercise behavior must improve in order for behavioral adaptations to occur. That is, the final aspect of the PRIMERS conceptual framework involves small, acute improvements in efficiency of communication (ie, connectivity) within and across neural networks during a single bout of exercise, which presumably accumulate and lead to larger, more meaningful adaptations in connectivity and secondary adaptations in behavior with chronic exercise training. This can be tested via a longitudinal study that measures changes in resting-state functional connectivity or effective connectivity in neural networks that are important for the regulation of exercise behavior, mobility, and cognitive performance—for example, at several intervals throughout an exercise training intervention. This unique study design could examine the trajectory of change in the efficiency of subcortical/cortical communication (ie, improved resting-state functional/effective connectivity) within and across those networks and whether or not such changes are accompanied by secondary changes in brain structure and/or behavior. This could provide an exciting snapshot of adaptive and integrative CNS plasticity with chronic exercise training and provide preliminary evidence of a possible dose-response effect of exercise on brain connectivity or if a certain volume of exercise training is required (ie, threshold) to induce neuroplasticity. However, given the reliance on repeated neuroimaging, such a study would be highly expensive. Such a design further could be disadvantageous for examining the periodic changes in cognitive performance with exercise training, given the influence of practice (ie, repeated cognitive testing) on cognitive performance, whereas structural and resting-state functional/effective connectivity neuroimaging measures can be collected in the absence of a cognitive task, minimizing the effects of practice. Such a research question further can be addressed within the context of animal research, particularly in animal models of neurological disorders, wherein researchers can examine the potential influence of exercise-related changes in functional or effective connectivity on molecular/cellular changes that are commonly observed with chronic exercise training. This could provide the first empirical evidence characterizing molecular/cellular changes associated with exercise training as a supportive mechanism of brain and behavioral changes, rather than as a primary driver of such adaptations.
Summary
Collectively, there is a proliferation of research examining the effects of exercise on mobility and cognition in the general population and those with neurological disorders as well as focal research examining possible neural mechanisms of such effects. However, there is a limited understanding of what it is about exercise, in particular, that causes such adaptive CNS neuroplasticity. We proposed that this involves a systems-wide, multidisciplinary view that integrates the neurophysiology of exercise behavior, network neuroscience, and neurorehabilitation for characterizing exercise-related CNS plasticity. This involves the proposition of a novel conceptual framework (ie, PRIMERS) that describes such adaptations occurring via activity-dependent neuroplasticity based on the processing and integration of multisensory input and associated complex motor output that is required for the regulation of physiological systems during exercise behavior. This conceptual framework sets the stage for the systematic examination of the effects of exercise on brain connectivity, brain structure, and molecular/cellular markers that may explain improvements in mobility and cognition. This further sets the stage for positioning exercise as an integrative, network-based approach for rehabilitation in persons with MS and other neurological disorders, considering that mobility and cognitive impairment, respectively, are highly prevalent, co-occurring, and disabling in these populations.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This article was supported in part by a grant from the National Multiple Sclerosis Society (CA 1069-A-7).
