Abstract
Background. Constraint-Induced Movement therapy (CIMT) has controlled evidence of efficacy for improving real-world paretic limb use in non-progressive physically disabling disorders (stroke, cerebral palsy). Objective. This study sought to determine whether this therapy can produce comparable results with a progressive disorder such as multiple sclerosis (MS). We conducted a preliminary phase II randomized controlled trial of CIMT versus a program of complementary and alternative medicine (CAM) treatments for persons with MS, to evaluate their effect on real-world disability. Methods. Twenty adults with hemiparetic MS underwent 35 hours of either CIMT or CAM over 10 consecutive weekdays. The primary clinical outcome was change from pretreatment on the Motor Activity Log (MAL). Results. The CIMT group improved more on the MAL (2.7 points, 95% confidence interval 2.2-3.2) than did the CAM group (0.5 points, 95% confidence interval −0.1 to 1.1; P < .001). These results did not change at 1-year follow-up, indicating long-term retention of functional benefit for CIMT. The treatments were well tolerated and without adverse events. Conclusion. These results suggest that CIMT can increase real-world use of the more-affected arm in patients with MS for at least 1 year. Clinical Trial Registration. ClinicalTrials.gov NCT01081275.
Keywords
Introduction
Multiple sclerosis (MS) is considered the leading cause of nontraumatic neurological disability for young adults and is increasing in incidence worldwide. 1 Its disability progression is commonly considered irreversible.2-5 Standard care, which rests on disease-modifying pharmacological treatments, at best slows central nervous system (CNS) atrophy progression but does not improve or arrest clinical deterioration in the progressive forms of disease.2,6
However, certain nonpharmacological treatments can substantially improve specific activity limitations in MS. In particular, about 200 clinical trials of various physical training regimens, including exercise and rehabilitation, have demonstrated at least short-term gains on diverse measures of within-laboratory measures, including maximal motor capability, as well as, in some instances, real-life physical function.7,8 The studies are limited, however, by both their lack of follow-up past 6 months 9 and their lack of assessment of whether the treatment effects transfer to everyday life situations in MS. It is therefore important to develop and assess novel physical training regimens for persons disabled by MS to improve their real-life activity as well as to establish whether such improvements can extend past 6 months.
Constraint-Induced Movement therapy (CIMT) 10 is a rehabilitation intervention that has demonstrated efficacy in clinical trials for improving real-life motor disability and increasing cortical gray matter in hemiparetic presentations of nonprogressive neurological diseases (stroke, cerebral palsy [CP]).11-14 In contrast, the benefits of CIMT for progressive neurological disorders such as MS have been little studied. A pilot study of CIMT for hemiparetic MS demonstrated safety, tolerance, and real-world functional efficacy. 15 Accordingly, the following small phase II randomized controlled trial was formulated to test whether CIMT could increase real-life physical function in persons with MS who had reduced use primarily of one arm. Part 1 of this study reports the clinical responses of the contrasting training approaches that were used in this randomized controlled trial (RCT). The effects on white matter cerebral structure in this RCT are reported in Part 2.
Methods
Participants
We enrolled adults who were diagnosed with MS by their MS specialists according to the 2011 revised McDonald criteria, 16 who reported reduced use primarily of one arm. Subjects were referred by their local MS specialists or self-referred from the study’s registration with ClinicalTrials.gov. Potential candidates were screened by telephone and then on-site by a project occupational therapist and a neurologist.
Inclusion criteria were: average item score <3/5 concerning the more-affected arm on the Motor Activity Log (MAL, briefly described below and in greater detail in the Supplementary Material, available in the online version of the article), 10 indicating substantially reduced real-world use of the more affected arm; mild-moderate motor deficit on active hand movement (at least 20° extension of the wrist and 10° at each of the fingers against gravity); able to stand without another person’s assistance at least 2 minutes; no more than mild-moderate pain during arm movement; relapse-free at least 3 months; absence of other neurological disorder associated with altered CNS structure; absence of other disorder that could substantially limit hand use (eg, arthritis); no prior exposure to CIMT; and agreement not to participate in other rehabilitation during the training period. The study did not alter the patients’ medications, and subjects were not paid for study participation.
The institutional review board at the University of Alabama at Birmingham approved the study procedures. All participants gave written informed consent.
Randomization and Masking
Participants were randomly assigned either to CIMT or a program of complementary and alternative medicine (CAM) treatments. Both treatment groups received 3.5 hours per day of direct therapist contact over 10 consecutive weekdays. The study coordinator entered the identity code of participants into a computer application after participants gave informed consent; participants were assigned to treatment group by a computer-generated random numbers table. Clinical testers were not masked.
Procedures
CIMT 10 originated from basic neuroscience research involving monkeys with a surgically deafferented limb 17 to treat learned nonuse, which is the sustained suppression of spontaneous use of a neurologically impaired limb in the life situation despite the capability to move it adequately, along with the simultaneous compensation by other limbs that have better movement ability.17,18 Upper extremity CIMT for patients with learned nonuse involves (1) intensive training with the more-affected arm on movements and activities of functional significance 3 hours per day with rest periods as needed; (2) training by behavioral shaping, which involves approaching motor goals in small increments, with encouragement to improve on personal best performance and positive feedback at each step; (3) restraint of the less-affected hand with a padded mitt for a target 90% of waking hours; and (4) a set of behavioral procedures, termed the Transfer Package, to facilitate transfer of training benefits from the clinic to the real world. 19 Transfer Package procedures include (1) behavioral contracting, (2) home practice assignments to be conducted during and after the end of formal treatment, (3) keeping an activity diary, and (4) the therapist conducting 30-minute daily interviews with the patient that include administering the MAL, reviewing the diary, and problem-solving discussions to overcome perceived barriers to arm use. Pilot results from our laboratory suggest that CIMT is safe and can functionally benefit MS in the real world for years after treatment.15,20,21
A CAM treatment program (see online Supplementary Material) was the comparison intervention. This was selected to control for patient expectancy to benefit from treatment and amount of therapist-supervised training, while providing a holistic approach that would markedly contrast with CIMT. The CAM program included (1) aquatic therapy, (2) massage, (3) gentle yoga, and (4) relaxation techniques (meditation with attention to breathing).
Study Endpoints
Study measures were administered in the week pretreatment and immediately posttreatment. The primary clinical endpoint was pre- to posttreatment change on the average item Arm Use score from the MAL. 10 The MAL is a structured scripted interview with the patient concerning the spontaneous real-world use of the more-affected arm relative to its pre-illness use across 30 common, primarily unimanual activities (detailed in the online Supplementary Material). Each item is rated by the patient separately for quality of movement and amount of use of the more-affected arm on a scale of 0 to 5, where 0 means no use by the more-affected hand and 5 means its use before the illness. Because the 2 scales have been found to be highly correlated, a convention in the CIMT literature, which we follow here, is to report only the scale for quality of movement, which is hereafter referred to as the Arm Use scale.22,23 The MAL has been validated against objective real-world wrist accelerometry in hemiparetic stroke 24 and has high test-retest reliability in persons with MS who report reduced use primarily of one arm. 25
Secondary measures included the Wolf Motor Function Test (WFMT) 10 and a participant opinion survey (POS) on which patients rated their agreement with a statement concerning anticipated (at pretreatment) or perceived (at posttreatment) benefit on a scale of 1 to 7, with 7 indicating maximal agreement. The Wolf Motor Function Test (WMFT) has been found to be reliable and valid for assessing motor capacity of the more-affected arm (see online supplementary material for details). This laboratory test contains 15 timed upper extremity tasks that range from items that require simple movements, such as reaching, to complex movements, such as stacking checkers, to be performed as quickly as possible. The mean of the Performance Rate score for each item is reported here. This score is calculated by dividing 60 seconds by the time required to complete the item, except when an item requires more than 120 seconds, in which case a score of 0 is assigned. 26 We evaluated the WMFT for both arms to evaluate whether asymmetric performance occurred in our participants, which would be consistent with a clinical impression of hemiparesis.
Patients were asked to return at 1-year posttreatment for follow-up clinical testing.
Statistical Analysis
Analysis of covariance was used to compare posttreatment and follow-up clinical changes of CIMT patients to CAM patients; pretreatment scores were entered as the covariate in these models. The f statistic 27 was used to quantify the between-groups effect size (ES). For this statistic, values ≥0.4 are considered large. Within-group ESs were assessed with the d′ statistic, for which values ≥0.57 are considered large.
Results
Following screening, 20 patients met inclusion criteria and were enrolled, randomized, and treated, 10 per group (Figure 1). There were no dropouts at posttreatment. Clinical 1-year follow-up results were available for 8 from each group. One CIMT patient could only be contacted at follow-up by telephone, which permitted administration of the MAL but not the WMFT. Two patients, one from each group, had experienced additional medical problems and were therefore excluded from follow-up. The other two patients could not be reached for follow-up.
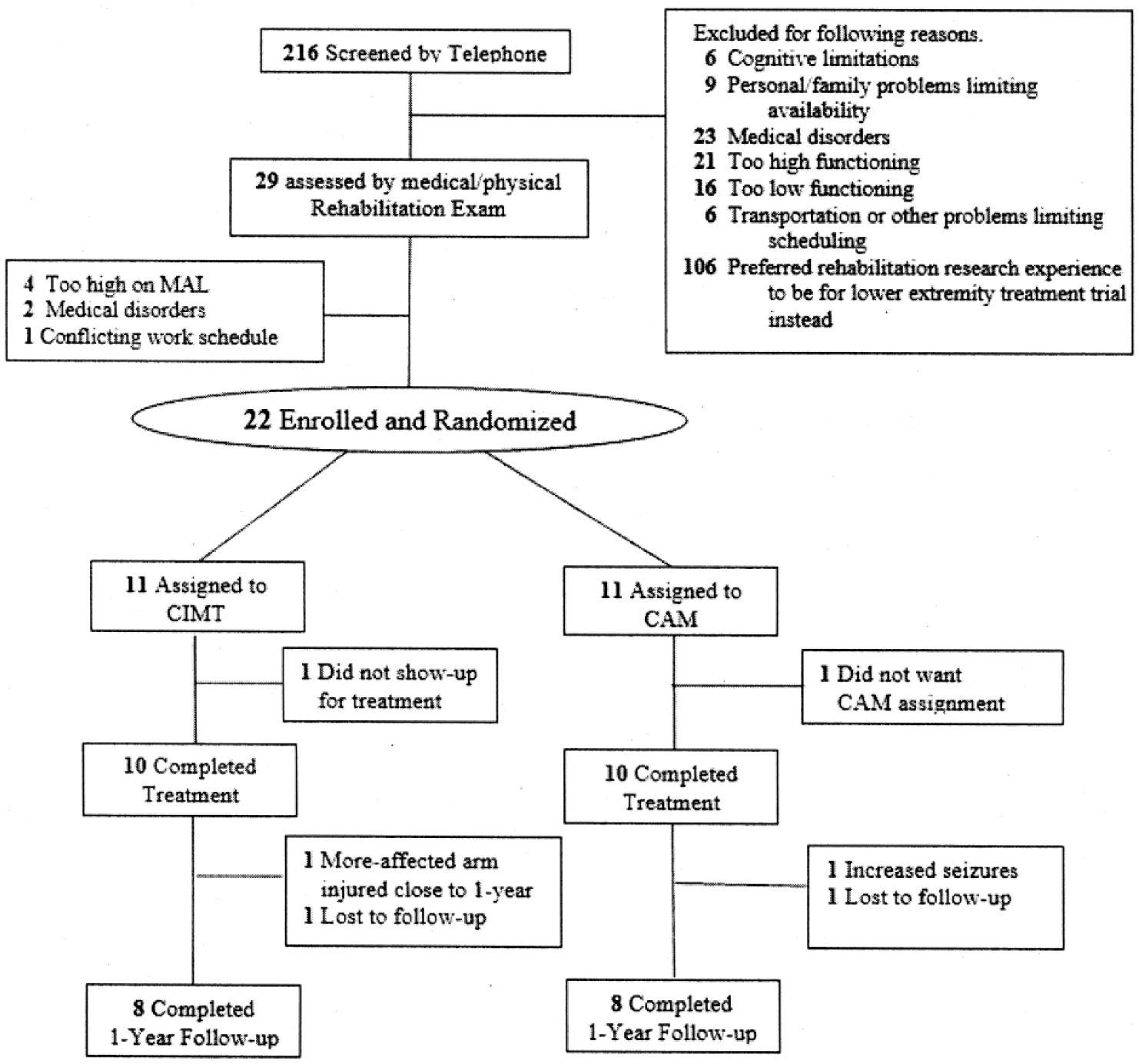
Trial profile. CAM, complementary and alternative medicine; CIMT, Constraint-Induced Movement therapy; MAL, Motor Activity Log.
Table 1 provides the baseline characteristics of the participants. There were no significant differences between treatment groups with respect to age, disease chronicity, time since last relapse (if there had been one), and scores on the Expanded Disability Status Scale for MS, 28 POS, MAL, and WMFT. As indicated by Table 1, the WMFT results reflected significant asymmetric performance speeds, F(1, 19) = 11.6, P = .003. The individual WMFT results (not shown) indicated that 3 of the patients (1 in the CIMT group, 2 in the CAM group) had bilateral upper extremity motor deficits, based on normative data. 29
Baseline Demographics and Clinical Characteristics of the Study Participants. a
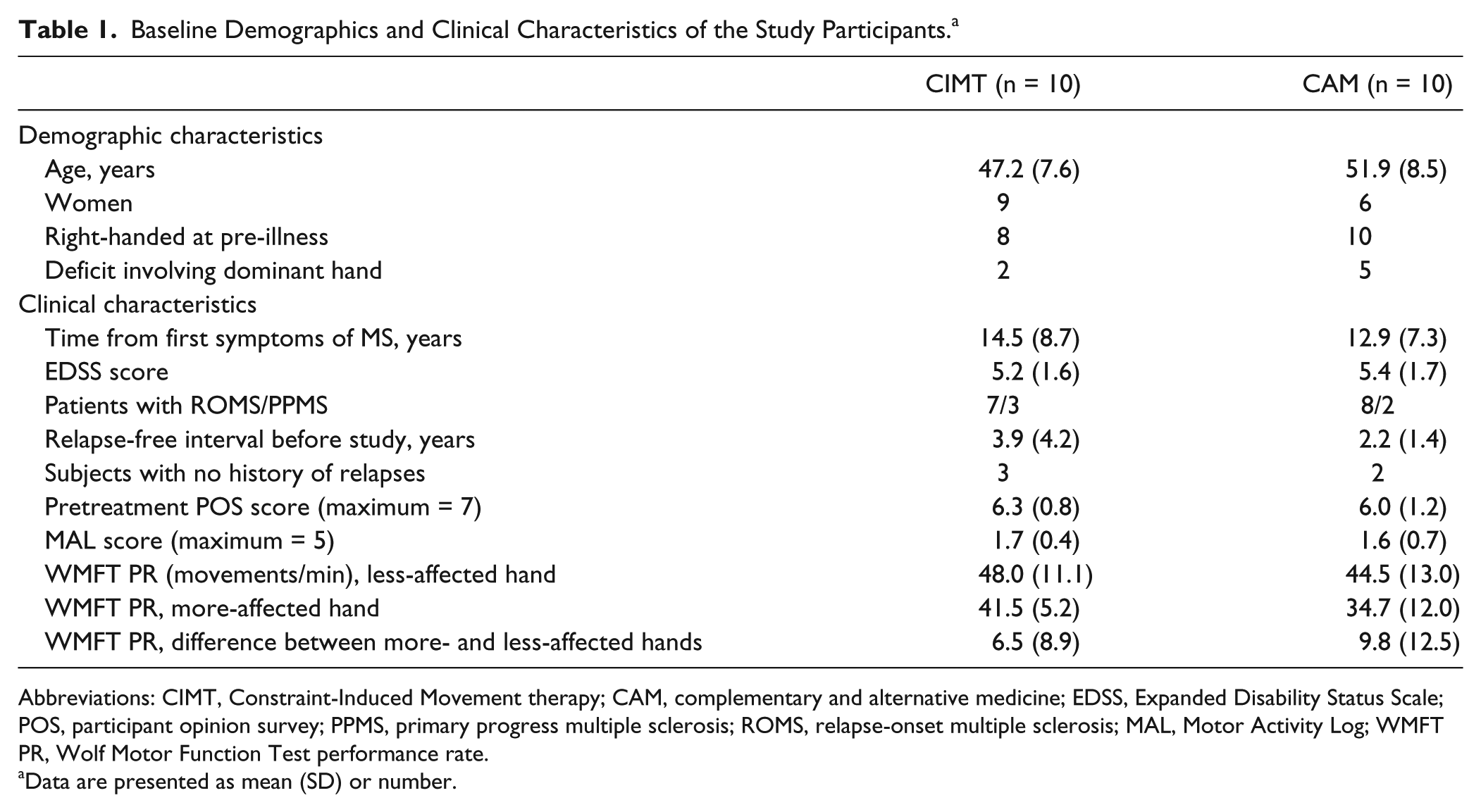
Abbreviations: CIMT, Constraint-Induced Movement therapy; CAM, complementary and alternative medicine; EDSS, Expanded Disability Status Scale; POS, participant opinion survey; PPMS, primary progress multiple sclerosis; ROMS, relapse-onset multiple sclerosis; MAL, Motor Activity Log; WMFT PR, Wolf Motor Function Test performance rate.
Data are presented as mean (SD) or number.
Table 2 summarizes the pre- to posttreatment changes on the MAL, more-affected hand WMFT, and POS, along with the pre- to 1-year follow-up changes on the MAL and the WMFT. At posttreatment, there was a very large treatment effect on the MAL in favor of CIMT, F(1, 17) = 31.3, P < .001, covariate-adjusted between-group ES = 1.6. The CIMT group improved on average on the MAL by 2.7 points (95% confidence interval [CI] = 2.2-3.1, within-group ES = 3.7). Thus, at posttreatment after CIMT, functional real-world arm use was approximately 85% of normal; before treatment it had been approximately 25% of normal. In contrast, the CAM patients improved only by 0.5 points (95% CI = 0.1-1.0, within-group ES = 0.7).
Clinical Outcomes. a
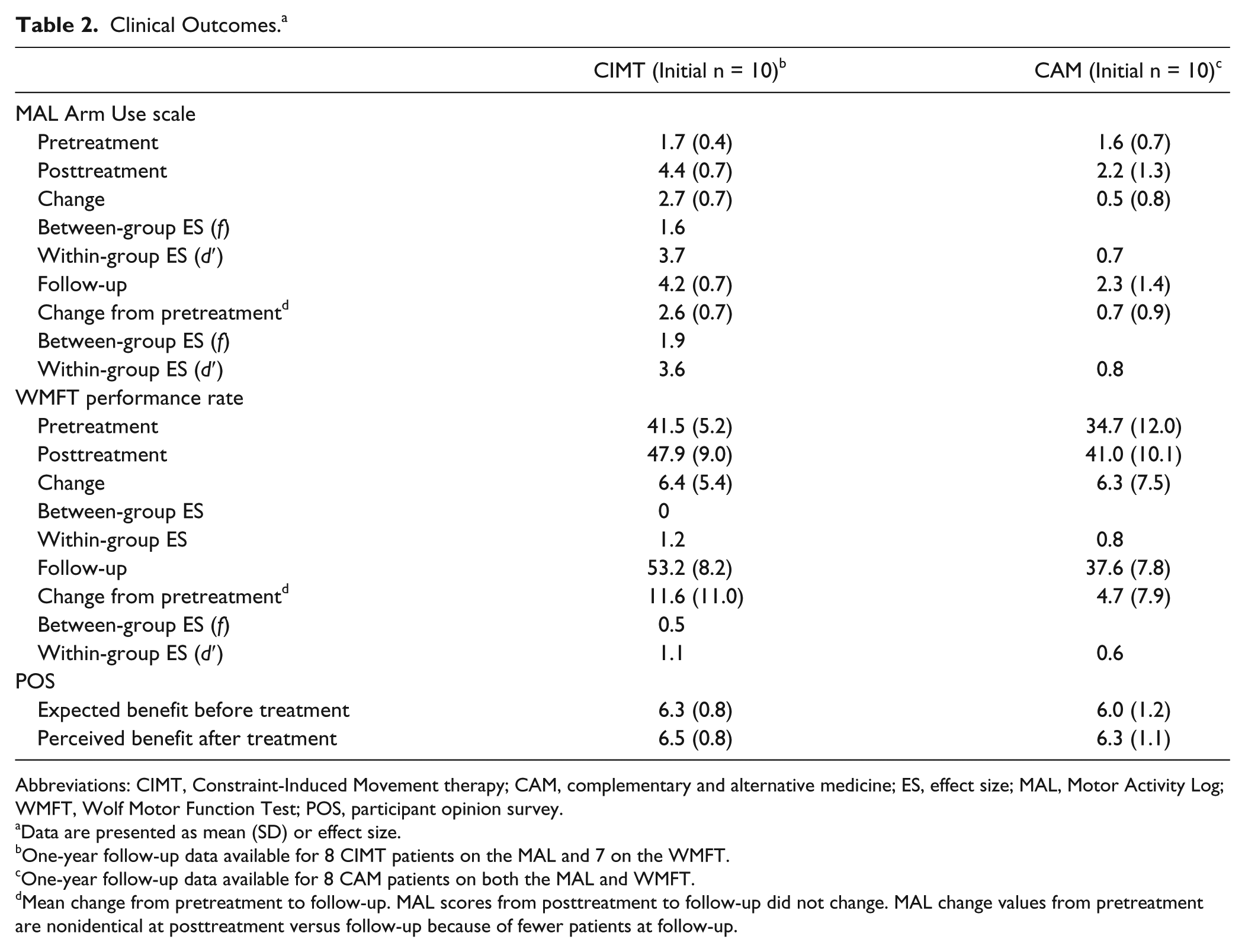
Abbreviations: CIMT, Constraint-Induced Movement therapy; CAM, complementary and alternative medicine; ES, effect size; MAL, Motor Activity Log; WMFT, Wolf Motor Function Test; POS, participant opinion survey.
Data are presented as mean (SD) or effect size.
One-year follow-up data available for 8 CIMT patients on the MAL and 7 on the WMFT.
One-year follow-up data available for 8 CAM patients on both the MAL and WMFT.
Mean change from pretreatment to follow-up. MAL scores from posttreatment to follow-up did not change. MAL change values from pretreatment are nonidentical at posttreatment versus follow-up because of fewer patients at follow-up.
Among the patients whose 1-year follow-up MAL results were available, their scores were unchanged from post-treatment in both groups. Thus, at 1-year follow-up there remained a very large treatment effect in favor of CIMT, F(1, 15) = 31.9, P < 0.001, covariate-adjusted between-group ES = 1.9.
Individual outcomes on the MAL are shown in Figure 2. The participants in the CIMT group had large and relatively uniform improvement. In contrast, the individual outcomes of the CAM group were, on average, much smaller and varied widely, despite starting from the same mean pre-treatment MAL level as the CIMT group. Using a change of 1.0 units as a cutoff, which Lang et al 30 have estimated as a minimal clinically important difference on the MAL in acute stroke patients, all of the CIMT patients but only three of the CAM patients had clinically meaningful motor improvement at both post-treatment and one-year follow-up (see Table 4 in the supplemental material).
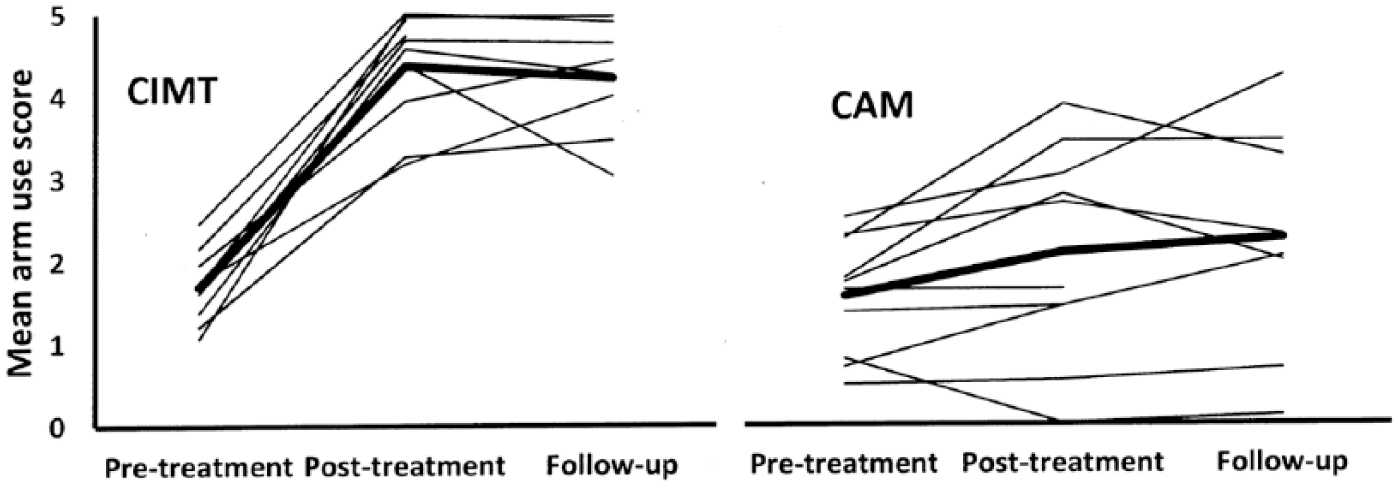
Individual (thin lines) and mean (thick lines) MAL Arm Use changes in the CIMT and CAM groups. CAM, complementary and alternative medicine; CIMT, Constraint-Induced Movement therapy; MAL, Motor Activity Log.
There was a large advantage on the WMFT in favor of CIMT at 1-year follow-up, F(1, 12) = 8, P = .015, covariate-adjusted between-group ES = 0.5, but not at posttreatment, F(1, 17) = 0.25, P = 0.6. Both groups had similar, large, pre- to posttreatment improvements on the WMFT rate score: CIMT group = 6.4 (5.4), 95% confidence interval = 3.0-9.6, within-group ES = 1.2; CAM = 6.3 (7.5), 95% confidence interval = 1.7-11.0, within group ES = 0.8. At 1-year follow-up, however, the groups diverged considerably. The 7 CIMT patients for whom WMFT data were available showed an additional mean gain of 3.6 from posttreatment, increasing the improvement from pretreatment to 11.6 repetitions per minute (95% CI = 3.4-19.7, within-group ES = 1.1). In contrast, the 8 CAM patients showed a mean loss in rate score of 2.1, reducing the improvement from pretreatment to 4.7 repetitions per minute at 1-year follow-up (95% CI = −1.9 to 11.4, within-group ES of the loss = 0.6.
On the POS, both groups had a similar high expectancy of benefit at pretreatment and a similar high satisfaction with the therapy posttreatment (Table 2). There were no adverse events.
Discussion
This preliminary phase II RCT found significantly greater improvement in spontaneous real-world use of the more-affected arm among adults with MS after CIMT than a dose-matched holistic training program, with treatment groups matched for pretreatment disability and expectancy to benefit. The pre- to posttreatment gain of 2.7 points on the MAL for the CIMT group was large, similar to the MAL gains for chronic stroke in prior RCTs of CIMT from our laboratory. The mean MAL gains for CIMT in our prior RCTs were: 2.0 (SD = 0.4; n = 9), 10 1.9 (SD = 0.6; n = 41), 13 and 1.9 (SD = 0.8; n = 40). 19 As noted, a clinically important change on the MAL has been found to be 1.0 points for stroke patients in the acute phase. 30
CIMT was developed specifically to overcome learned nonuse, which as described under the Methods section is the inhibited daily use of the more-affected upper limb after hemiparetic illness despite the limb’s ability to perform activities of daily living on request.17,18 Although the term “learned nonuse” itself has not appeared in the MS literature, this condition likely commonly follows chronic motor disability in this disease. In contrast, the term is widely used in both the stroke clinical31-33 as well as the experimental neuroscientific literature.34-36 Multiple reports describe persons with MS who have chronically self-restricted limb use and concurrent behavioral compensation, who nonetheless can move substantially more when urged,37-40 thus suggesting learned nonuse. The incidence of unilateral upper extremity motor deficit in MS has been found to range from 14% to 49%, depending on the particular assessment. 41 Although upper limb deficits in MS have more often been found to be bilateral than unilateral on these assessments, it is still possible that inter-limb performance differences may occur, which could induce unilateral learned nonuse in the same way that could follow a unilateral upper limb motor deficit. As a result, the incidence of the motor conditions that could induce unilateral learned nonuse could be considerably greater than would be suggested only by the reported incidence of unilateral upper extremity motor deficits in MS. For nonprogressive functional deficits resulting from damage to the CNS that are event-related, such as stroke and traumatic brain injury, the development of learned nonuse is somewhat different, but the outcomes are the same.17,18
Physical training trials in MS for upper extremity motor deficit, apart from those that used CIMT, have concentrated on improving within-laboratory measures of maximal motor capacity, rather than real-world use of a paretic limb,8,42 where the presence of learned nonuse would have its greatest effect.18,19 It would appear important, therefore, to expand rehabilitation trials for MS to develop and test methods to improve paretic limb use outside the treatment setting.
Because a primary goal of CIMT is to improve learned nonuse, the MAL is the preferred instrument in RCTs of CIMT to measure changes of use by the more-affected upper extremity. 43 Studies of CIMT in hemiparetic stroke have shown that posttreatment increases on the MAL were associated with increased posttreatment real-world spontaneous paretic arm movement that had been assessed by objective community-based wrist-worn accelerometry over 3 consecutive days.44,45 Consequently, we used the MAL for the present RCT as its primary clinical outcome measure.
The MAL changes in our patients support the importance of the Transfer Package for clinical outcomes. The CAM group, which did not receive a Transfer Package, improved on the WMFT, that is, in motor capacity of the more-affected arm, to about the same extent as the CIMT group, yet the CAM group showed negligible improvement on the MAL. Although the activities carried out by participants in the CAM group appear to have improved their motor capacity, the limited improvement in the real-world use of the more-affected extremity by the CAM patients could have resulted either from the content of the physical training exercises that distinguished the CAM program from the CIMT program, the absence of a Transfer Package in the CAM program that would otherwise have facilitated the transfer of the improved movement achieved in the treatment setting to everyday activities in the life situation, or both features.
Few physical rehabilitation studies have reported on the important factor of retention of physical rehabilitation treatment effects over a substantial period after the end of treatment. A number of studies from this laboratory10,13,19,46,47 and elsewhere 48 using the Transfer Package 19 have evaluated changes in the MAL out to at least 1 year posttreatment in adults with hemiparetic neurological disorders (stroke, traumatic brain injury). Figure 3 (upper) demonstrates the long-term course of the MAL in the earlier studies (thin lines) relative to the CIMT MS patients in the present study (thick line). As can be seen, the present MS patients demonstrated the greatest improvement on the MAL scale from baseline. The lower part of Figure 3 indicates the retention of MAL changes from posttreatment, as a percentage of the mean posttreatment MAL score for each clinical sample. When expressed in these terms, the MS patients of our study had essentially a complete retention of their posttreatment MAL values at one-year follow-up. The high, close-to-normal MAL scores of 6 of the 10 CIMT patients at post-treatment suggest that there is a strong possibility that a ceiling effect was operating for assessing changes long after treatment. This is further suggested by the fact that 3 of the 4 patients who had room to improve in real-world use of the more-affected arm after the end of treatment did so. All decline in the group follow-up data was because of the performance of the remaining relatively lower performing patient, whose MAL score showed a substantial decline at 1-year follow-up. The MAL scale may not be sufficiently sensitive at its high end to capture possible further real-world improvements at 1-year posttreatment for the 6 MS patients who were very high performing immediately after treatment. The findings therefore suggest that the neuropathology of MS does not preclude long-term real-world functional improvement that is comparable to the responses of other, nonprogressive neurologically disabling illnesses (stroke, traumatic brain injury) in response to the same form of efficacious motor rehabilitation.
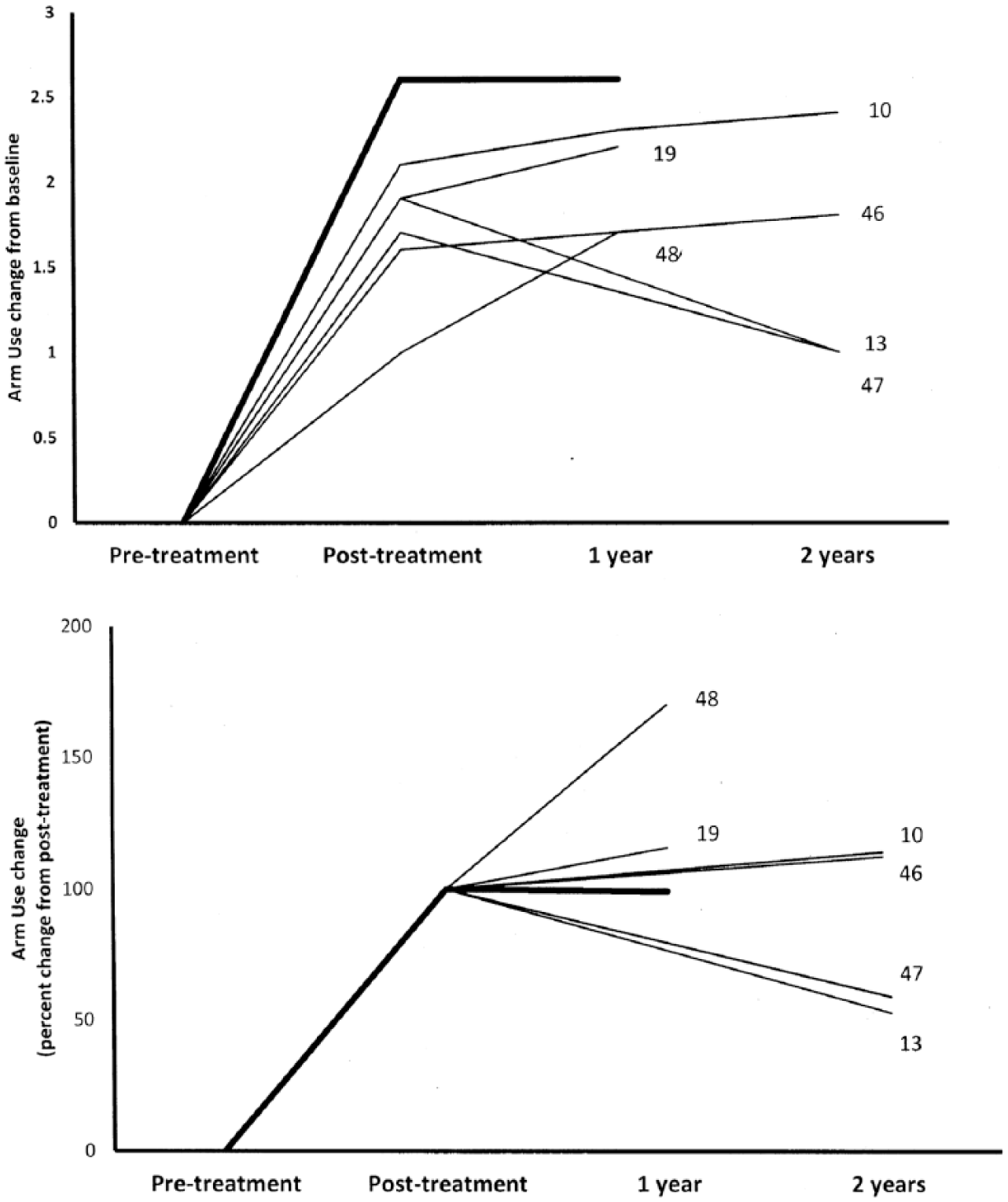
The graphs depict the changes in CIMT studies in average MAL values from pretreatment to posttreatment and follow-up, showing 2 different presentations of the same data. The studies included are only those in which the complete CIMT protocol was employed. The thick lines represent the present MS results. The thin lines represent the other published studies of clinical CIMT trials (stroke, traumatic brain injury); each thin line is associated with the specific numbered study in the references list. Upper: longitudinal course of average MAL values. Lower: relative retention (percentage) of posttreatment MAL values at follow-up. CIMT, Constraint-Induced Movement therapy; MAL, Motor Activity Log; MS, multiple sclerosis.
The small sample size was a weakness of the study design. The cell size of 10 limited the confidence for concluding that that the WMFT performance rates did not differ between the CIMT and CAM groups at posttreatment, given that the 95% CI around the difference of the covariate-adjusted means between the groups at posttreatment ranged from −5 to 8 repetitions per minute (mean difference between the groups = 1.5). In contrast, because the confidence interval around the difference between the CIMT and CAM groups in the performance rate at 1 year ranged from 3 to 22 (difference of 12.6 repetitions, favoring CIMT), this suggested that a study with a larger sample would still have found an advantage in favor of CIMT. A similar relationship concerning the confidence interval applies also to the findings on the MAL, both at posttreatment and follow-up. The confidence intervals around the difference between the covariate-adjusted means of the 2 groups on this scale at posttreatment and 1-year follow-up were 1.3 to 2.8 and 0.9 to 2.8 points, respectively (difference 1.8 points favoring CIMT).
The Transfer Package procedure requiring conducting the MAL interview on each treatment day raises the possibility that the advantage found for CIMT here was from the inflation of the participants’ ratings of use of their more-affected arm due to repeated questioning. This is unlikely for a few reasons. First, in an RCT of CIMT for adults with chronic stroke with impairment of their more-affected arm similar to the participants here, a placebo control group (n = 20) was administered the MAL on each treatment day, as was the case here, but showed virtually no change in MAL score. 13 Second, changes on the MAL from pre- to post-CIMT have been found to be strongly correlated with corresponding changes in wrist-worn accelerometer recordings obtained in the community from adults with chronic stroke with motor impairment similar to the participants in the present study: r range = 0.74 to 0.91; Ps < .01; n range = 20 to 27.22,44 The strong correlations between changes in MAL scores and changes in physical measures of arm movements make it unlikely that the advantage found for CIMT over CAM here is because of alterations in perceptions that are divorced from actual changes in use of the more-affected arm.
Another limitation of this study is that the CAM group had more men and more individuals whose more-involved hand had been dominant premorbidly. In addition, because of funding limitations, clinical assessors were not masked to treatment condition. However, the POS results indicate that CIMT and CAM patients had similar expectations at the outset and similar degrees of general perceived benefit afterward.
Although our findings do not allow us at this stage to indicate the generalization of benefit to persons with MS who have either less motor ability or greater real-life upper extremity activity, they support designing later expanded trials to evaluate the potential benefit of this form of training to persons with MS with more diverse motor deficits. As noted, the MAL may not have sufficient sensitivity at the high end to capture the full extent of improvements in the CIMT group. To address this shortcoming, future studies might consider the ABILHAND49,50 or Manual Ability Measure–36, 51 which assess difficulty in use of the hands in the community regardless of which arm is used, as well as for bimanual tasks, and tasks that require a wider range of dexterity than those on the MAL.
Conclusion
This article suggests a method to improve real-world motor function in MS patients in the part of the body that is trained, with little diminution over a year. MS is a grim diagnosis because it is considered to be a promise of serious long-term decline. We feel that further study is warranted.
Supplemental Material
Online_supplementary_material_clean_11212017 – Supplemental material for Phase II Randomized Controlled Trial of Constraint-Induced Movement Therapy in Multiple Sclerosis. Part 1: Effects on Real-World Function
Supplemental material, Online_supplementary_material_clean_11212017 for Phase II Randomized Controlled Trial of Constraint-Induced Movement Therapy in Multiple Sclerosis. Part 1: Effects on Real-World Function by Victor W. Mark, Edward Taub, Gitendra Uswatte, David M. Morris, Gary R. Cutter, Terrie L. Adams, Mary H. Bowman and Staci McKay in Neurorehabilitation and Neural Repair
Footnotes
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Gary R. Cutter serves on the Data and Safety Monitoring boards of AMO Pharmaecuticals, Apotek, Gilead Pharmaceuticals, Horizon Pharmaceuticals, Modigenetech/Prolor, Merck/Opko Biologics, Merck, Neuren, Sanofi-Aventis, Reata Pharmaceuticals, Receptos/Celgene,Teva Pharmaceuticals, NHLBI (Protocol Review Committee), NICHD (OPRU oversight committee), and on the consultation or advisory boards of Atara Biotherapeutics, Bioeq GmBH, Cerespir, Inc, Consortium of MS Centers (grant), Genzyme, Genetech, Innate Therapeutics, Jamsen Pharmaceticals, Klein-Buendel Incorporated, Medimmune, Medday, Nivalis, Novartis, Opexa Therapeutics, Roche, Savara, Inc., Somahlution, Teva pharmaceuticals, Transparency Life Sciences, and TG Therapeutics. Dr Cutter also is the President of Pythagoras, Inc, a private consulting company located in Birmingham, Alabama.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the National Institutes of Health (HD061767) and the National Multiple Sclerosis Society (RG 4221).
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
