Abstract
Background. Environmental enrichment (EE) confers benefits after traumatic brain injury (TBI) when provided daily for > 6 hours, but not 2 or 4 hours, which more accurately reflects the daily amount of clinical rehabilitation. The lack of benefit with sub-therapeutic EE suggests that augmentation with galantamine (GAL), which enhances cognition after TBI, may be indicated to confer benefits. Objective. To test the hypothesis that 2 and 4 hours of EE paired with GAL will provide benefits comparable to 24 hours of EE alone. Moreover, all EE groups will perform better than the standard (STD)-housed GAL group. Methods. Anesthetized rats received a TBI or sham injury and then were randomized to receive intraperitoneal injections of GAL (2 mg/kg) or saline vehicle (VEH; 1 mL/kg) beginning 24 hours after surgery and once daily while receiving EE for 2, 4, or 24 hours. Motor and cognitive assessments were conducted on postoperative days 1-5 and 14-19, respectively. Results. Motor function was significantly improved in the TBI + 24-hour EE group versus the TBI + STD + VEH and TBI + STD + GAL groups (P < .05). Cognitive performance was enhanced in all EE groups as well as in the TBI + STD + GAL versus TBI + STD + VEH (P < .05). Moreover, the 2- and 4-hour EE groups receiving GAL did not differ from the 24-hour EE group (P > .05) and performed better than GAL alone (P < .05). Conclusions. The findings support the hypothesis and have clinical relevance because, often, only brief rehabilitation may be available in the clinic and, thus, augmenting with a pharmacotherapy such as GAL may lead to outcomes that are significantly better than either therapy alone.
Keywords
Introduction
An estimated 10 million people worldwide incur a traumatic brain injury (TBI) each year. 1 Although mild TBI may not consistently produce salient motor and cognitive disability, moderate-to-severe brain trauma results in substantial and persistent neurological impairments.2-4 The costs associated with acute medical care, long-term rehabilitation, and the loss of productivity resulting from the inability to regain preinjury employment significantly taxes the health care system, which is estimated in the billions of dollars each year.5-7 To combat this problem, numerous preclinical studies evaluating a plethora of potential therapies have been conducted in the pursuit of elucidating effective strategies to promote cognitive and/or motor recovery after TBI.8-10
Although many pharmacological agents have been found to confer significant improvement in the laboratory,8-11 few have translated to the clinic,12-14 suggesting that single therapeutic approaches may not be sufficient to promote recovery from an injury that is multifaceted.8,15 Hence, the inclusion of additional therapeutic manipulations, such as exercise,16-18 behavioral combinations,19,20 and environmental enrichment (EE)11,21, which can mimic clinical rehabilitation are warranted.
EE is an experimental paradigm designed to mimic the novelty and complexity of rehabilitation used with humans in a rat model where the environment and the range of activities are more limited. The exploratory (i.e., exposure to multiple objects of differing shape and size that can be manipulated), physical (ie, vast living environment with ample room for walking, running, and climbing), and social (group housing) elements provided by EE are considered enriching.11,21 This unique milieu is markedly different from the standard (STD) housing paradigm where paired subjects live in traditional-sized laboratory cages and receive only basic sustenance (ie, food and water). Numerous studies have demonstrated that EE, provided continuously after experimental TBI, confers motor, cognitive, and histological benefits relative to STD-housed controls.21-27 It has also been shown that even relatively brief periods of EE after TBI produce effects that are comparable to the continuous EE paradigm in both male 28 and female 29 rats. However, when EE is reduced further and limited to just 2 or 4 h/d the paradigm is rendered insufficient to promote benefits.28,29 Given that rehabilitation session times reported in the clinical literature range from 2 to 8 h/d,30-32 and perhaps are more likely to fall within the lower range because of costs and resources, it is necessary to design preclinical studies such that subtherapeutic EE can promote benefits. Demonstrating that significant benefits can be achieved with an EE paradigm that parallels the clinic in terms of time allotments would further validate EE as a preclinical model of neurorehabilitation.
One viable and clinically relevant approach is to augment EE with pharmacotherapies particularly because in the clinical rehabilitation setting, many TBI patients have cognitive impairments that impede their ability to comprehend and function effectively and, therefore, necessitate pharmacological intervention. Because galantamine (GAL) has recently been shown to facilitate cognitive function after experimental TBI 33 and has also been reported in clinical studies to enhance vigilance and attention, leading to better general function, 34 significantly improve episodic memory, 35 and enhance cognition for up to 12 months in Alzheimer’s disease, 36 it is a logical choice as a treatment that could be combined with subtherapeutic EE to determine whether synergism would lead to better outcomes relative to GAL treatment alone.
Hence, the specific aim of this study was to test the hypothesis that subtherapeutic EE (ie, 2 and 4 hours) combined with GAL will confer benefits beyond that of GAL alone and will be comparable to continuous EE. If the hypothesis is supported, the findings will refine, enhance, and further support the EE paradigm as a viable preclinical model of neurorehabilitation, particularly because it would mimic clinical rehabilitation, which is often limited in duration and, in many instances, augmented by pharmacotherapies.
Materials and Methods
Subjects and Presurgical Procedures
A total of 63 adult male Sprague-Dawley rats (Harlan, Indianapolis, IN) weighing 300 to 325 g on the day of surgery were housed in standard steel-wire mesh cages and maintained in a temperature (21°C ± 1°C) and light (on 7:00
Surgery and Acute Neurological Evaluation
Surgical anesthesia was induced and maintained with inspired concentrations of 4% and 2% isoflurane (IsoFlo, Abbott Laboratories, North Chicago, IL), respectively, in 2:1 N2O:O2 in a vented anesthesia chamber. After endotracheal intubation, the rats were secured in a stereotaxic frame and ventilated mechanically during surgery. Utilizing aseptic techniques, a midline scalp incision was made, the skin and fascia were reflected to expose the skull, and a craniectomy (6 mm in diameter) was made in the right hemisphere (encompassing bregma and lambda and between the sagittal suture and the coronal ridge) with a Michele trephine. The resulting bone flap was removed and the craniectomy was enlarged further with rongeurs. Controlled cortical impact (CCI) injury was produced in rats weighing 300 to 325 g, as previously described.37-44 Briefly, the impacting shaft was extended, and the impact tip was centered and lowered over the craniectomy until it contacted the dura mater; then, the rod was retracted, and the impact tip was advanced 2.8 mm farther to produce a brain injury of moderate severity (2.8-mm tissue deformation at 4 m/s). Immediately after the CCI, anesthesia was discontinued, and the incision was promptly sutured. The rats were subsequently extubated and assessed for acute neurological outcome before being placed in a temporary cage until the effects of anesthesia waned (as indicated by spontaneous movement). Sham rats underwent similar surgical procedures but were not subjected to the impact.
Acute Neurological Evaluation
Hind limb reflexive ability was assessed immediately following the termination of anesthesia by gently squeezing the rat’s paw every 5 s and recording the time to elicit a withdrawal response. Return of the righting reflex was determined by quantifying the time required to turn from the supine to prone position on 3 consecutive trials. These neurological indices are used to determine the level of injury severity.42-44
Housing Conditions: Standard and Environmental Manipulation
After the effects of anesthesia waned (as evidenced by spontaneous movement in the holding cage), the rats were returned to the colony where those designated for continuous enrichment (ie, 24-hour group) were immediately placed in an EE, which consists of a stainless steel–wire cage (91 × 76 × 50 cm3) with 3 levels and ladders to ambulate from one level to another and contains various toys (eg, balls, blocks, and tubes), nesting materials (eg, paper towels), and food and water ad libitum.42,45 To maintain novelty, the objects were rearranged every day and changed each time the cage was cleaned, which was approximately every 3 days. Ten to 12 rats, which included both TBI and sham controls, as well as VEH and GAL-treated rats, were housed in the EE at any given time. Rats in the STD conditions were placed in standard steel-wire mesh cages (37 × 25 × 18 cm3, 2 rats per cage) with only food and water. The rats receiving the abbreviated enrichment sessions were removed from the STD cages and placed in the EE cages for 2 or 4 hours and then returned to the STD conditions. These rehabilitative manipulations occurred each day for 19 days.
Drug Administration
GAL (Sigma-Aldrich, St Louis, MO) was prepared daily by dissolving in sterile saline, which also served as the VEH. GAL (2 mg/kg) or a comparable volume of VEH (1 mL/kg) was administered intraperitoneally beginning 24 hours after cortical impact or sham injury and once daily for 19 days. On the days of behavioral testing, the injections were administered 1 hour prior to testing. The dose of GAL was selected based on published data from our laboratory showing a beneficial effect. 33 The route of administration is standard protocol in our laboratory.38-41
Motor Performance (Beam Balance and Beam Walk)
Established beam-balance and beam-walk tasks were utilized to assess motor function. Beam balancing consists of placing the rat on an elevated (90 cm) narrow wooden beam (1.5 cm wide) and recording the time it remains on for a maximum of 60 s. Beam walking, originally devised by Feeney et al 46 and used extensively in our laboratory,38-44 consists of training/assessing rats using a negative-reinforcement paradigm to escape ambient light and white noise by traversing an elevated narrow wooden beam (2.5 cm × 100 cm) and entering a darkened goal box situated at the opposite end. The aversive stimuli (light and noise) were terminated when the rat entered the goal box, thus serving as reinforcement (reward) for completing the task. Performance on the beam walk consisted of recording the time to traverse the beam. Rats were tested on these motor tasks 1 hour prior to surgery to establish baseline performance and then again on postoperative days 1 to 5. Three trials (60 s allotted time) were provided per day on each task, and the average daily scores for each subject were used in the statistical analyses.
Cognitive Function: Acquisition of Spatial Learning
Spatial learning was assessed in a Morris water maze task 47 that has been shown to be sensitive to cognitive function/dysfunction after TBI.38-44 Briefly, the maze consisted of a plastic pool (180 cm diameter; 60 cm high) filled with tap water (26°C ± 1°C) to a depth of 28 cm and was situated in a room with salient visual cues that remained constant throughout the study. The platform was a clear Plexiglas stand (10 cm diameter, 26 cm high) positioned 26 cm from the maze wall in the southwest quadrant. Spatial learning began on postoperative day 14 and consisted of providing a block of 4 daily trials (4-minute intertrial interval) for 5 consecutive days (14-18) to locate the platform when it was submerged 2 cm below the water surface (ie, invisible to the rat). On day 19, the platform was raised 2 cm above the water line and marked with white tape, which made it visible to the rats and served as a control procedure to determine the contributions of nonspatial factors (eg, sensory-motor performance, motivation, and visual acuity) on cognitive performance. For each daily block of trials, the rats were placed in the pool facing the wall at each of the 4 possible start locations (north, east, south, and west) in a randomized manner. Each trial lasted until the rat climbed onto the platform or until 120 s had elapsed, whichever occurred first. Rats that failed to locate the escape platform within the allotted time were manually guided to it. All rats remained on the platform for 30 s before being placed in a heated incubator between trials. The times of the 4 daily trials for each rat were averaged and used in the statistical analyses.
Cognitive Function: Probe Trial (Memory Retention)
One day after the final acquisition training session (day 19), all rats were given a probe trial to measure retention. Briefly, the platform was removed from the pool, and the rats were placed in the maze from the location point most distal to the quadrant where the platform was previously situated (ie, target quadrant) and allowed to freely explore the pool for 30 s. Typically, rats that have learned the specific location of the escape platform exhibit a spatial bias and spend significantly more time in the target quadrant. The time spent in the target quadrant was recorded using a spontaneous motor activity recording and tracking system. Swim speed was also recorded during this testing period. The percentage time spent in the target quadrant was used in the statistical analysis.
All experimental procedures were preapproved by the University of Pittsburgh’s Institutional Animal Care and Use Committee. All efforts were made to limit the number of rats used as well as to minimize their discomfort.
Data Analysis
Statistical analyses were performed on data collected by observers blinded to drug treatment conditions using Statview 5.0.1 software. The motor and cognitive data were analyzed by repeated-measures analysis of variance (ANOVA). Acute neurological assessments, swim speed, and visible platform performance were analyzed by 1-factor ANOVAs. When the overall ANOVAs revealed a significant effect, the Newman-Keuls post hoc test was utilized to determine specific group differences. The data are presented as the mean ± standard error of the mean and are considered significant when the corresponding P values are ≤.05.
Results
There were no significant differences in any assessment among the sham control groups, regardless of housing condition or treatment, and thus, the data were pooled into a single SHAM group. Additionally, there were no variables that precluded accurate assessments, and thus, the statistical analyses are based on all 63 rats.
Acute Neurological Evaluation
No significant differences were observed among the TBI groups for return of hind limb reflex ability after a brief paw pinch (range for right = 166.9 ± 18.8 to 194.1 ± 9.2 s; left range = 174.0 ± 18.6 to 200.5 ± 9.6 s) or righting reflex latency (range = 333.3 ± 36.1 to 397.5 ± 36.0 s) following the termination of anesthesia (P values > .05). The lack of neurological differences among the TBI groups indicates that all rats received similar levels of anesthesia and injury severity.
Motor Performance: Beam Balance
No baseline differences were observed among the groups as all rats balanced on the beam for the allotted 60 s (Figure 1). Post-TBI, the repeated-measures ANOVA revealed significant Group (F5,57 = 2.759; P = .027) and Day (F5,285 = 23.564; P < .0001) differences as well as a significant Group × Day interaction (F25,285 = 2.412; P = .0003). The post hoc analysis revealed that the SHAM controls, which were able to balance for the full 60 s, were significantly better than the TBI + STD + VEH, TBI + STD + GAL, and TBI + 4-hour EE + GAL groups (P values < .05) but not the TBI + EE + VEH and TBI + 2-hour EE + GAL groups (P values > .05). Over the course of the 5 days of testing, all TBI groups improved; however, the TBI + EE + VEH group reached baseline performance quicker than the TBI + STD + VEH and TBI + STD + GAL groups (P values < .05) and did not differ from either the TBI + 2-hour EE + GAL or TBI + 4-hour EE + GAL groups (P values > .05).
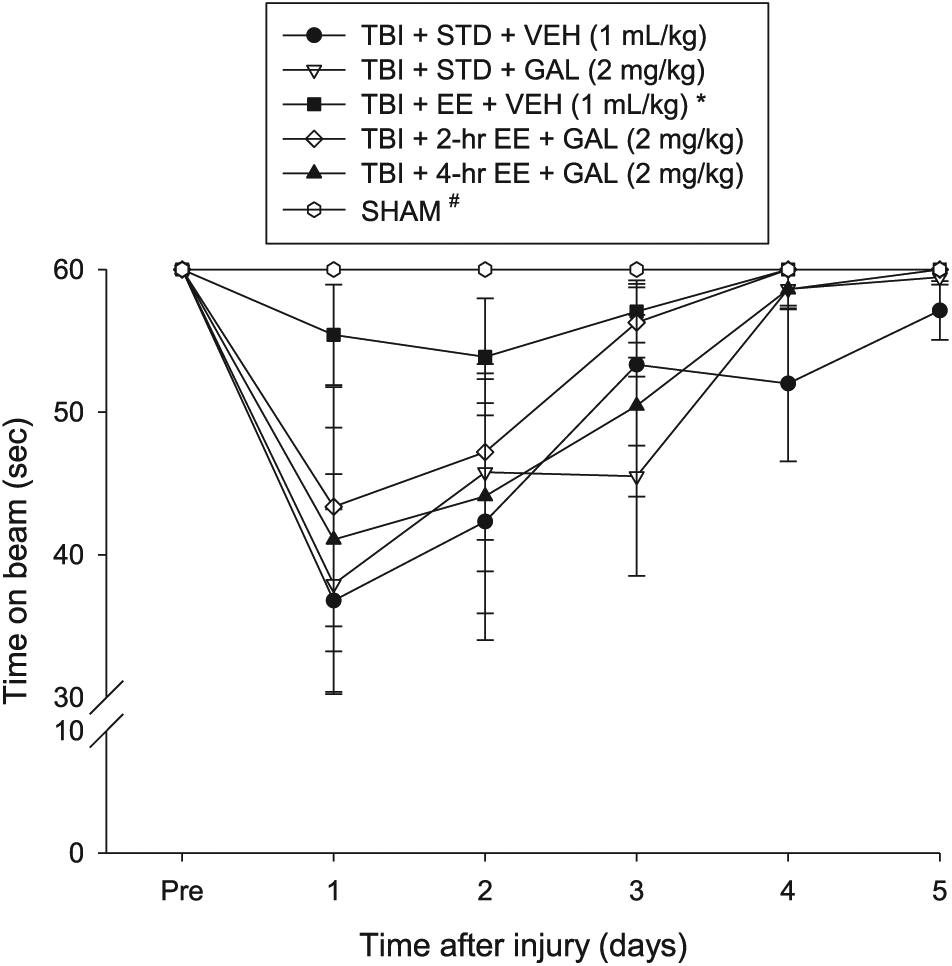
Mean (±SEM) time (s) balancing on an elevated narrow beam prior to and after TBI or sham injury. There were no significant differences among the sham groups and, therefore, the data were pooled. No difference was revealed between the TBI + EE + VEH and the TBI + 2-h EE + GAL or TBI + 4-h EE + GAL groups (P values > .05).
Motor Performance: Beam Walk
No significant differences were observed among any of the groups in the time to traverse the beam prior to surgery (Figure 2). However, after surgery, the repeated-measures ANOVA revealed significant Group (F5,57 = 5.393; P = .0004) and Day (F5,285 = 65.276; P < .0001) differences as well as a significant Group × Day interaction (F25,285 = 4.128; P < .0001). The post hoc analysis indicated that all TBI groups were significantly impaired compared with the SHAM controls (P values < .05). Like the beam-balance findings, the TBI + EE + VEH group recovered better than the TBI + STD + VEH and TBI + STD + GAL groups (P values < .05) and did not differ from either the TBI + 2-hour EE + GAL or TBI + 4-hour EE + GAL groups (P values > .05).
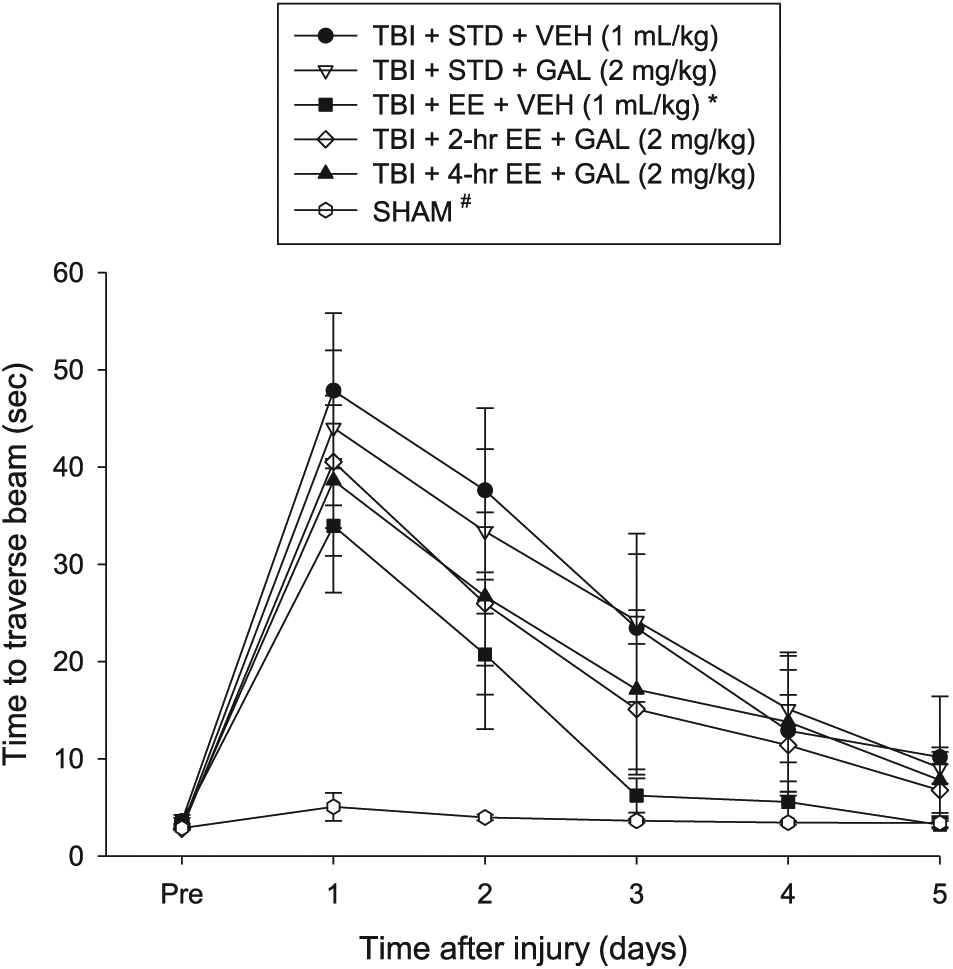
Mean (±SEM) time (s) to traverse an elevated narrow beam prior to and after TBI or sham injury. There were no significant differences among the sham groups and, thus, the data were pooled. No difference was revealed between the TBI + EE + VEH versus the TBI + 2-h EE + GAL or TBI + 4-h EE + GAL groups (P values > .05).
Cognitive Function: Acquisition of Spatial Learning
Analysis of the spatial learning data revealed significant Group (F5,57 = 3.119; P = .0148) and Day (F4,228 = 37.157; P < .0001) differences. The post hoc analysis revealed that the SHAM group was better than the TBI + STD + VEH and TBI + STD + GAL groups (P values < .05) but did not differ from the TBI + EE + VEH, TBI + 2-hour EE + GAL, and TBI + 4-hour EE + GAL groups (P values > .05). The TBI + EE + VEH, TBI + 2-hour EE + GAL, and TBI + 4-hour EE + GAL groups were markedly better at learning the location of the escape platform relative to both the TBI + STD + VEH and TBI + STD + GAL groups (P values < .05) but did not differ from one another (P > .05). Additionally, the TBI + STD + GAL group was better than the TBI + STD + VEH group (P < .05; Figure 3). No significant differences in swim speed (range = 28.5 ± 1.7 to 31.4 ± 1.4 cm/s) or time to reach the visible platform were observed among the TBI or SHAM groups (P values > .05).
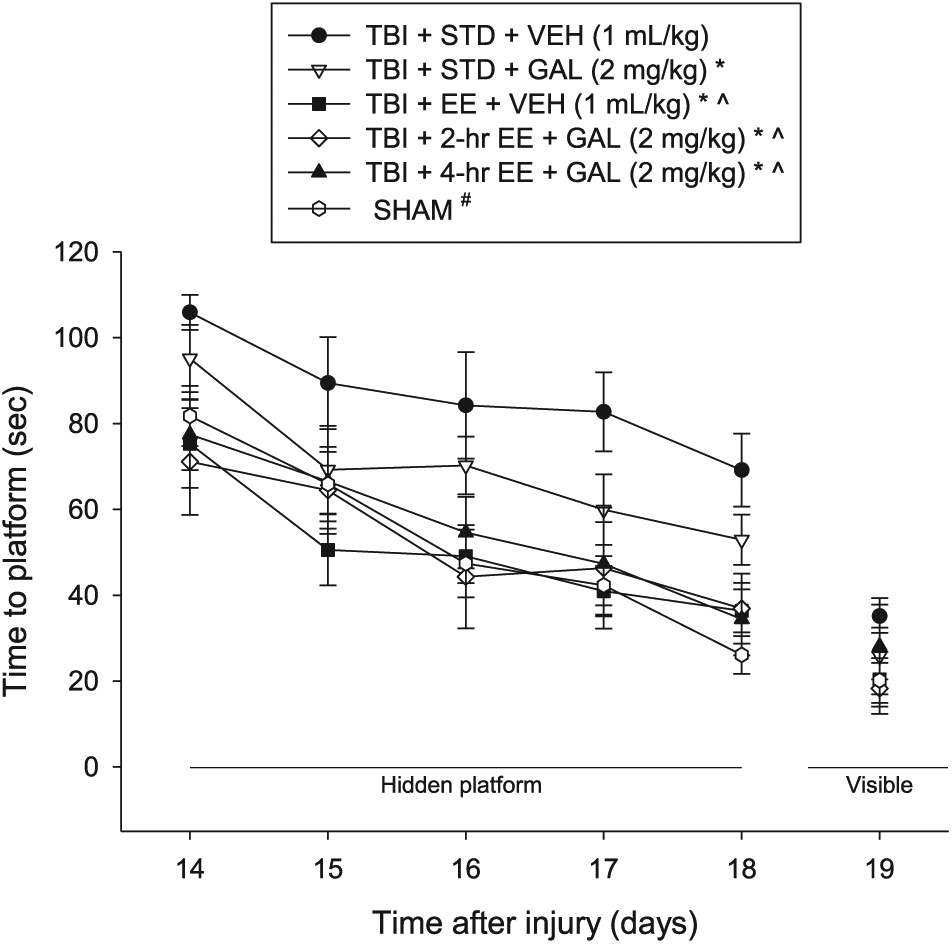
Mean (±SEM) time (s) to locate a hidden and visible platform in the water maze. There were no significant differences among the sham groups, and therefore, the data were pooled. No difference was revealed between the TBI + EE + VEH versus the TBI + 2-h EE + GAL or TBI + 4-h EE + GAL groups (P values > .05). No significant differences were revealed among the groups in the visible platform assessment.
Cognitive Function: Probe Trial
Analysis of the probe trial (ie, memory retention) data revealed a significant Group effect (F5,57 = 4.717; P = .0011). The post hoc analysis revealed that the SHAM control, TBI + EE + VEH, TBI + 2-hour EE + GAL, TBI + 4-hour EE + GAL, and TBI + STD + GAL groups spent a greater percentage of the allotted time in the target quadrant (42.3% ± 1.9%, 39.9% ± 2.1%, 36.4% ± 3.7%, and 38.1% ± 2.7%, respectively) than the TBI + STD (26.2% ± 3.3%) group (P values < .05) but did not differ from one another (P values > .05; Figure 4).
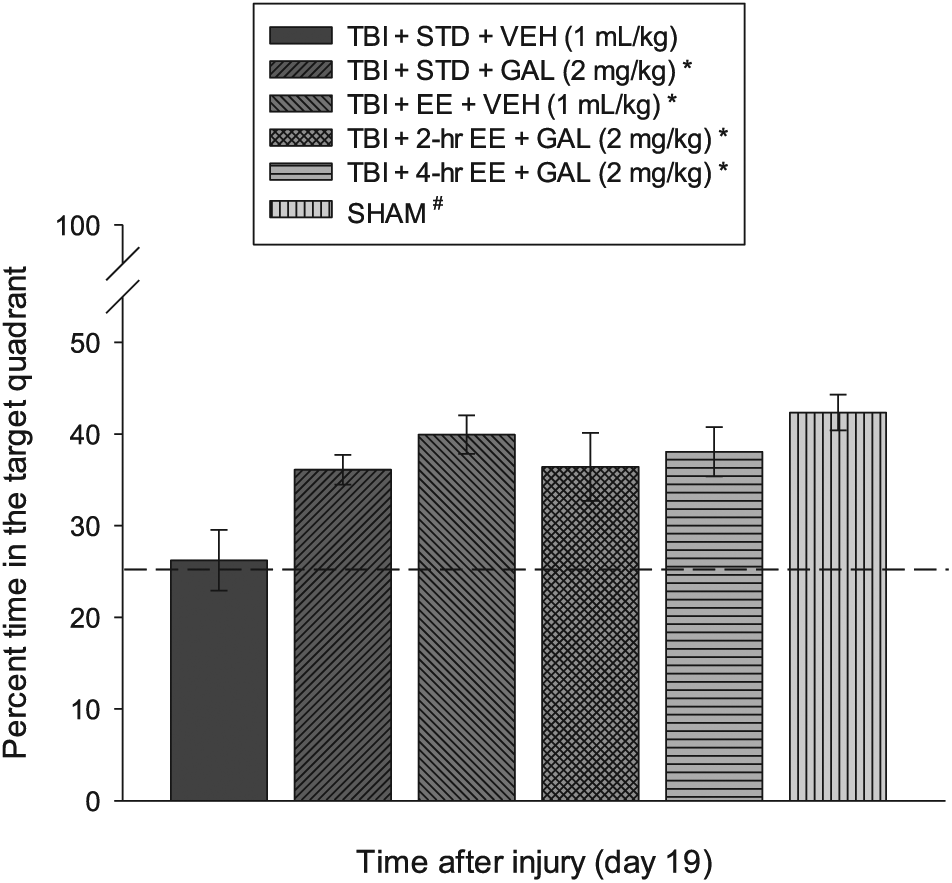
Mean (±SEM) percentage time spent in the target quadrant (ie, where the escape platform was previously located) following a single probe trial 19 days after TBI or sham injury. No other comparisons were significant (P > .05). The dotted line represents performance at the chance level (25%).
Discussion
The aim of the study was to test the hypothesis that EE abridged to the point where it becomes subtherapeutic can still be transformed into an effective rehabilitative paradigm by combining it with a clinically relevant pharmacotherapy like GAL. In support of this hypothesis, the data showed that 2 or 4 hours of EE combined with GAL was as effective as continuous EE in promoting cognitive benefits after a CCI injury of moderate severity in adult male rats. The ameliorative effects were not solely a result of GAL because the abbreviated EE groups combined with GAL performed better relative to the GAL-only group, suggesting that the combination led to a synergistic effect, resulting in increased performance. This additive effect was observed even after minimal enrichment (ie, 2 hours) because there were no differences between the GAL-treated 2-hour EE and 4-hour EE groups.
The augmented benefits seen with the combination of GAL and abbreviated EE therapy is contrary to previous reports where various pharmacotherapies provided in conjunction with continuous EE in adult male rats did not produce benefits beyond those of the individual therapies.33,40-42,44 The lack of additional benefit when combining effective drug therapies with the typical EE paradigm is theorized to be a result of the robustness of continuous EE affording little room for additional recovery. To this end, the results from the current study indicate that the rehabilitation-relevant abbreviated EE paradigm may provide an experimental model with which to test combination treatments that include limited or subtherapeutic EE and various potentially efficacious pharmacotherapies. Moreover, the paradigm may have clinical relevance because rehabilitation is not provided continuously in the clinic, but rather may range from 2 to 8 h/d.30-32 Indeed, a recent study required patients to engage for a minimum of 3 hours of physical therapy, occupational therapy, or speech-language pathology each day for 5 d/wk. 48
GAL is a dual-functioning drug that increases acetylcholine (ACh) levels and stimulates nicotinic ACh receptors (nAChRs) on neurons and glia 49 in brain regions such as the frontal cortex and hippocampus 50 that are subject to injury after clinical or experimental TBI.51,52 Stimulation of microglial nAChRs may be involved in GAL’s inhibitory effects on inflammation and oxidative stress53,54 because the nicotinic antagonist mecamylamine blocks the GAL-mediated reduction of pro inflammatory cytokines and subsequent cognitive recovery after brain injury. 55 GAL increases synaptic plasticity, as evidenced by enhanced long-term potentiation. 56 Moreover, pharmacological enhancement of α7nAChR activation has been shown to produce potent neuroprotective and anti-inflammatory effects by reducing blood-brain barrier permeability, TUNEL-positive apoptotic cells, and reactive gliosis after TBI. 57 Additionally, GAL increases firing activity of dopaminergic cells in the ventral tegmental area and dopamine output from the prefrontal cortex. 58 These GAL-induced influences on dopamine-mediated function are intriguing because pharmacotherapies with D2 receptor agonist properties have been shown in preclinical and clinical studies to enhance neurobehavior after TBI.38,39,43,44,59
Potential mechanisms of EE are plentiful11,21 and consist of, but are not limited to, protection of the cholinergic system by reducing TBI-induced ChAT-positive cell loss, 40 a reduction of CA1 and CA3 cell loss after TBI,11,21 and induction of hippocampal and cortical plasticity, which includes long-term potentiation. 60 EE also produces higher levels of nerve growth factor mRNA in the hippocampus, increases brain-derived neurotrophic factor gene upregulation and progenitor cell survival, enhances dendritic branching, and reduces markers of oxidative stress and inflammation (for comprehensive reviews, see Bondi et al11,21).
Apart from the potential mechanisms attributed to GAL or EE alone or in combination that may have led to the robust cognitive benefits observed,11,21 GAL could also be increasing the arousal of the TBI rats during their time in EE, thus making shorter periods of EE (ie, rehabilitation) as effective as longer periods through increased interaction with the multiple and various cage accoutrements (ie, novel stimuli) and, thus, providing additional cognitive therapy. As shown in previous studies,28,29 there appears to be a threshold amount of EE necessary to confer benefit, with 6 hours being sufficient, but 2 and 4 hours being ineffective. Thus, increased arousal during the time spent in EE may be successful in lowering this threshold. Another possibility is that the increased dopaminergic or cholinergic activity induced with GAL may be promoting neural remodeling and subsequently enhancing the effect of EE outside of any increase in arousal.
In conclusion, abbreviated EE and GAL produce benefits that are comparable to the robust, albeit not clinically relevant, continuous EE approach. Although we acknowledge that EE does not simulate the skill-based training elements of rehabilitation in humans, EE does provide a unique opportunity to examine the effects of novelty and complexity on functional recovery over time.
Footnotes
Authors’ Note
Elizabeth R. Skidmore is a member of ASNR.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported, in part, by NIH grants HD069620, HD069620-S1, NS060005, NS084967 (AEK), NS094950, NS099683 (COB), the University of Pittsburgh Physicians/UPMC Academic Foundation, and the UMPC Rehabilitation Institute (COB).
