Abstract
Background. In the chronic stages of moderate-severe traumatic brain injury (TBI), progressive hippocampal volume loss—continuing well after acute neurological insults have resolved—has now been well documented. Previous research in other populations suggests that elevated anxiety symptoms are associated with compromise to the medial temporal lobes. Objective. To examine whether higher anxiety symptoms predict greater hippocampal volume loss in moderate-severe TBI. Methods. We conducted an analysis of prospectively collected, longitudinal behavioral and magnetic resonance imaging (MRI) data from 5 to 12 to 30 months post-injury. Eighty participants were included in the study, with anxiety symptom and MRI data collected at a minimum of 2 time points. Correlational and bivariate latent difference score (with imputation) analyses were used to examine the relationship of Beck Anxiety Inventory scores with hippocampal volume loss, while controlling for depressive symptoms and total brain volume. Results. Analyses revealed that higher anxiety symptoms at 5 and at 12 months following moderate-severe TBI predicted significant later volume loss in the right hippocampal complex and the right hippocampal head. Right hippocampal volume and volume change did not predict subsequent anxiety scores or anxiety change scores. Conclusions. These novel findings implicate anxiety symptoms as a possible predictor of progressive hippocampal volume loss in the chronic stages of moderate-severe TBI.
Introduction
Moderate-severe traumatic brain injury (TBI) has long been viewed as a discrete, non-progressive neurological insult, whereby the brain remains stable once acute damage (eg, edema, gliosis) has resolved.1,2 This view has recently been questioned, and a novel conceptualization of moderate-severe TBI as a progressive and neurodegenerative disorder has emerged, with growing findings of ongoing losses to brain volume and white matter integrity in the chronic stages of injury.1,3-8 To date, little is known about the mechanisms of these progressive changes. 1
Recent research in amnestic mild cognitive impairment (aMCI) and Alzheimer’s disease (AD) has shown a significant positive relationship between higher anxiety symptoms and compromise to the medial temporal lobes. 9 Mah et al 9 reported that aMCI patients with anxiety symptoms were significantly more likely than aMCI patients without anxiety symptoms to experience subsequent atrophy of the medial temporal lobes over a median of 36 months. As well, the level of anxiety symptoms was associated with higher conversion from aMCI to AD during this time period. In another study of participants with MCI and AD, Sturm et al 10 found that elevated emotional contagion—a phenomenon that is associated with anxiety11-14 and involves synchronizing one’s own emotions with those of others 13 —was significantly related to degeneration of the temporal lobes, with the strongest effects in the right temporal pole and right anterior hippocampus (HPC). We therefore examined the relationship between self-reported anxiety symptoms and HPC atrophy in moderate-severe TBI.
In this prospective, longitudinal study of adults with moderate-severe TBI, we examined the temporal relationship between anxiety symptoms and volume change in terms of total HPC volume and subdivisions along the longitudinal axis of the HPC (ie, head, body, and tail). We predicted that higher symptoms of anxiety at 5 and 12 months post-TBI would predict greater HPC volume loss from, respectively, 5 to 12 and 12 to 30 months post-injury, particularly in the head region (ie, anterior HPC). These hypotheses were formed based on the findings of Mah et al 9 and Sturm et al 10 as well as the vulnerability of the HPC head to damage after TBI, 15 and its known role in anxiety (as demonstrated in animal 16 and human 17 studies).
Methods
Participants
Participant data were drawn from a larger, prospective, longitudinal study of recovery from TBI (the Toronto TBI Recovery Study) 18 for which collection of the relevant data for the current study (ie, Beck Anxiety Inventory [BAI] and MRI data) commenced in 2004. Primary inclusion criteria for the larger study were as follows: (1) a clinically confirmed moderate-severe TBI as indexed by a Glasgow Coma Scale (GCS) 19 score of 12 or less and/or post-traumatic amnesia (PTA) 20 lasting 24 hours or more; (2) resolution of PTA by 3 months post-injury; (3) age between 17 and 80 years; (4) functional use of at least one upper extremity; and, (5) functional command of English. Exclusion criteria comprised the following: (1) a diagnosis of a disease affecting the central nervous system (e.g., AD, multiple sclerosis, lupus); (2) history of psychotic disorder; and (3) a prior TBI or stroke. Participants in the Toronto TBI Recovery Study underwent a comprehensive neuropsychological assessment and magnetic resonance imaging (MRI) at 3 time points, averaging 5.1 months (SD = 1.0; range = 3.7-10.4; N = 80), 12.7 months (SD = 1.7; range = 6.4-17.6; N = 61), and 30.3 months (SD = 6.9; range = 23.0-56.1; N = 50) post-injury. For the current analyses, additional inclusion criteria were the completion of the BAI and MRI for at least 2 of the 3 assessments. Incomplete data were due to participants either (1) deciding not to participate further in the study, (2) missing their 12-month assessment, but returning at 30 months or (3) not yet having reached the final (30-month post-TBI) assessment at the time of this study. Eighty of 159 participants from the larger study met this extra inclusion criterion and were therefore included in the study (see Supplementary Table 1).
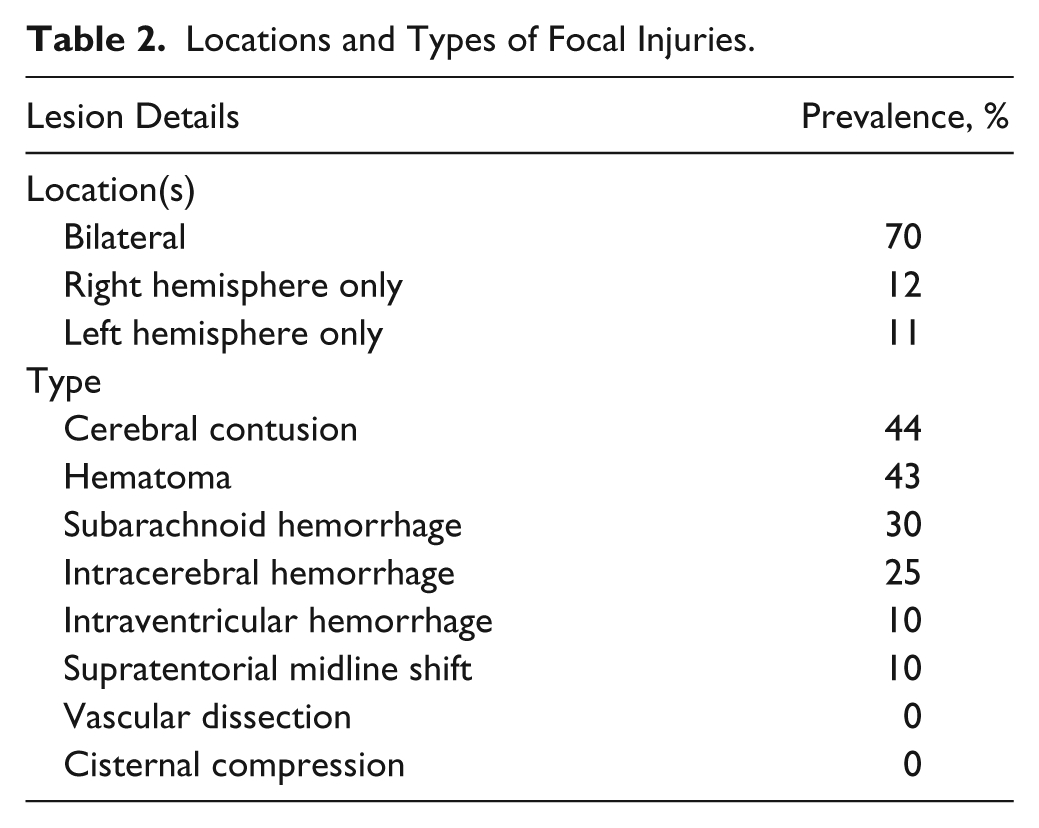
Participants’ demographic information and injury severity information are presented in Table 1. The types and locations of injuries observed are shown in Table 2. For a comparison of baseline demographics, injury severity, anxiety and depressive symptoms, and HPC volume data for participants with and without complete MRI and BAI data, see Supplementary Table 2.
Demographic and Injury Severity Data (N = 80).

Locations and Types of Focal Injuries.
MRI Data Acquisition and Processing
MRI was performed on a General Electric (GE) Signa-Echospeed 1.5 Tesla HD scanner (SIGNA EXCITE, GE Healthcare, Milwaukee, WI) with an 8-channel head coil. Several types of scans were acquired within a 1-hour session. Of relevance to this study, high-resolution isotropic T1 weighted, 3-dimensional IR prepped radiofrequency spoiled-gradient recalled-echo (3DIRSPGR) images (inversion time/repetition time/echo time [TI/TR/TE] = 12/300/5 ms, flip angle [FA] = 20°, slice thickness of 1 mm with no gap [256 × 256 acquisition matrix]) were acquired in the axial plane using a 25-cm field of view (FOV).
The HPC was traced as a whole and also into 3 subsections, including the head, body, and tail as in prior studies of TBI 15 and in reference to the Duvernoy atlas, 21 using gray matter segmented high-resolution T1 mapping. Outcome measures included left and right HPC total volume, as well as volumes for the left and right HPC head, body, and tail. Manual tracing of the right and left HPC was completed with Analyze 8.1 (Brain Imaging Resource, Mayo Clinic, MN) by a single expert tracer, Dr Jerome Maller (Dr Maller has more than 10 years of MRI tracing experience and has developed a handbook for structural MRI analysis),22-24 from coronally oriented MR images in the anterior-posterior direction, as detailed by Maller et al 22 and Watson et al. 25
Here, a strict measurement protocol 22 including the assessment of intrarater reliability for 10 randomly selected cases was employed. For the HPC head and body, intraclass correlation (IC) was 0.99 for the right and >0.99 for the left, and for the HPC tail IC was 0.98 for the right and 0.99 for the left, indicating high intrarater reliability. 22 The HPC in this sample ranged from ~30-50 slices (ie, 3-5 cm) and each took ~30-40 minutes to be manually outlined. All raw HPC volumes are expressed in mm3. Volumes were automatically computed by multiplying the number of voxels traced in each slice by their depth (ie, slice thickness). Subsections of the HPC were measured per the Watson protocol.25,26 Total volumes for each slice were then summed by the software to calculate HPC subsection and total volumes. (See Maller et al 22 for a fuller description of the HPC tracing methods used in this study.) HPC total and substructure volume change scores used in our correlational analyses were calculated using the following formula: ([V2 – V1] / [V1+V2]) × 100, where V1 = volume of a given structure at time 1 and V2 = volume of a structure at time 2. Total brain volume (TBV) was calculated as gray plus white matter. Average total right and total left HPC volumes at 5 months post-TBI were 4031 (SD = 497) and 3769 (SD = 633) mm3, respectively.
Beck Anxiety Inventory
The BAI is a 21-item, self-report measure of anxiety symptoms. 27 It has high demonstrated internal consistency and test-retest reliability 27 and is frequently used to measure anxiety symptoms in TBI.28-31 The BAI has also been shown to predict emotional, behavioral and cognitive outcomes of acquired brain injury, including TBI. 31 As noted by Julian, 32 a potential limitation of the BAI is that to distinguish anxiety from depression,33,34 it measures predominantly somatic aspects of anxiety; therefore, there may be some trans-diagnostic items on the BAI for medical conditions such as TBI. 33 To address this issue, we undertook supplementary analyses to examine the validity of the BAI for individuals with a moderate-severe TBI at 2, 5, 12, and 24 months post-injury, with sample sizes of 157, 137, 108, and 69, respectively. We found that at each of these time points, the BAI demonstrated divergent validity from two indices of TBI severity, including GCS score 19 and length of PTA. 34
Covariates
The Beck Depression Inventory (BDI) 35 was included as a covariate in our analyses as the relationship of depression with smaller HPC volume is well-documented. 36 The BDI has shown high internal consistency and convergent validity with other depression scales as well as divergent validity from measures of anxiety. 37 For the analyses in this study, we took the average of participants’ BDI scores across the 3 time points and used that average as a covariate. Note that we used the average score from across time points because repeated-measures analysis of variance revealed no significant differences in these values across time at the group level, F(2, 90) = 1.33, P = .27.
We tested the relationship of other potential control variables with the primary anxiety and HPC variables. Because of their known associations with HPC volume, the candidate covariates were the GCS (the most widely used measure of injury severity in TBI), 19 anxiety/antidepressant medication use, 38 TBV at 5 months post-injury, 22 age, years of education (YOE), and sex.39,40
Design and Procedures
For the current study, a secondary analysis of prospectively collected longitudinal data was employed. Demographic and injury severity data were collected from medical records and confirmed, where possible, through clinical interview. The BAI and MRI were completed within 48 hours of one another for each participant. The BAI was administered during a testing session at the Toronto Rehabilitation Institute, University Health Network (UHN) lasting approximately 6 hours that included a clinical interview, and performance-based and self-report neuropsychological measures. All MRI scans were conducted at a separate nearby site (Toronto General Hospital, UHN). All participants provided informed consent, except where participants were clinically judged to be unable to provide informed consent, in which case a substitute decision maker consented on his or her behalf. Some participants were unable to provide their fully informed consent at the time of recruitment for the larger study (ie, 1.5-2 months post-injury or less); as such, their ability to validly complete the BAI at that time might be called into question. By the first time point of the current study (5-months post-injury) all participants included in this study were judged by the assessor to be able to validly and independently complete the BAI. The study was approved by the Research Ethics Board of the Toronto Rehabilitation Institute, UHN.
Data Analysis
Primary correlational and bivariate latent difference score (BLDS) analysis procedures are reported below. See Supplementary Methods for additional data analysis details.
Correlations
Participants with MRI and BAI data available at all three time-points of assessment (n=39) were included in partial correlation analyses. Here, averaged 5 and 12 as well as 12 and 30 month anxiety scores were correlated with right and left HPC volume change from 5 to 12 and 12 to 30 months post-TBI, respectively, while controlling for depressive symptoms. Our aim was to examine these relationships irrespective of directionality (assessed using BLDS modeling described below) and without imputation procedures (see Supplementary Methods). If either the left or the right HPC volume change scores were significantly correlated with anxiety symptoms, it would be carried forward to the BLDS analyses.
We also carried out bivariate correlations to determine which covariates to include in the BLDS models. Potential covariates for the BLDS models were GCS, TBV at 5 months post-injury, anxiety/antidepressant medication use, age, YOE, and sex in addition to depression. Candidate covariates were included in the BLDS models if they correlated significantly (P < .05 on a 2-tailed test) with a BAI or HPC variable.
Bivariate Latent Difference Score Modeling
BLDS modeling was used to explore the temporal relationship of anxiety symptoms with HPC volume. The BLDS method simultaneously models two univariate LDS (ULDS) models (eg, models of anxiety and right HPC total complex volume; see Supplementary Methods and Supplementary Figure 1) with cross-lag regression paths from a static latent variable score to the opposite variable’s subsequent latent change score (depicted graphically in Supplementary Figure 2). Using this approach, a previous anxiety score can be used to predict subsequent change in HPC volume, and a previous HPC volume score can be used to predict subsequent change in anxiety scores. BLDS modeling has previously been valuable in exploring brain-behavior relationships over time 41 and has been shown to be a robust and consistent method for modeling bivariate longitudinal data. 42
Results
Correlations
Correlational analyses for participants with HPC volume data collected at all 3 time points (presented in Table 3; n = 39) demonstrated that BAI scores, averaged across 5 and 12 month assessments, correlated significantly with change in total right HPC volume from 5 to 12 months post-TBI (controlling for depressive symptoms; r = −0.36, P = .01). Similarly, BAI scores averaged across 12- and 30-month assessments correlated significantly with total right HPC volume change from 12 to 30 months after injury (r = −.37, P = .01). There were no statistically significant correlations of left HPC total volume change scores with average anxiety scores observed for either set of correlations, and effect sizes were small. Further, static right HPC total and head volumes did not concurrently correlate with anxiety scores at 5, 12, or 30 months post-TBI.
Partial Correlations of BAI With HPC Total Volume Change. a
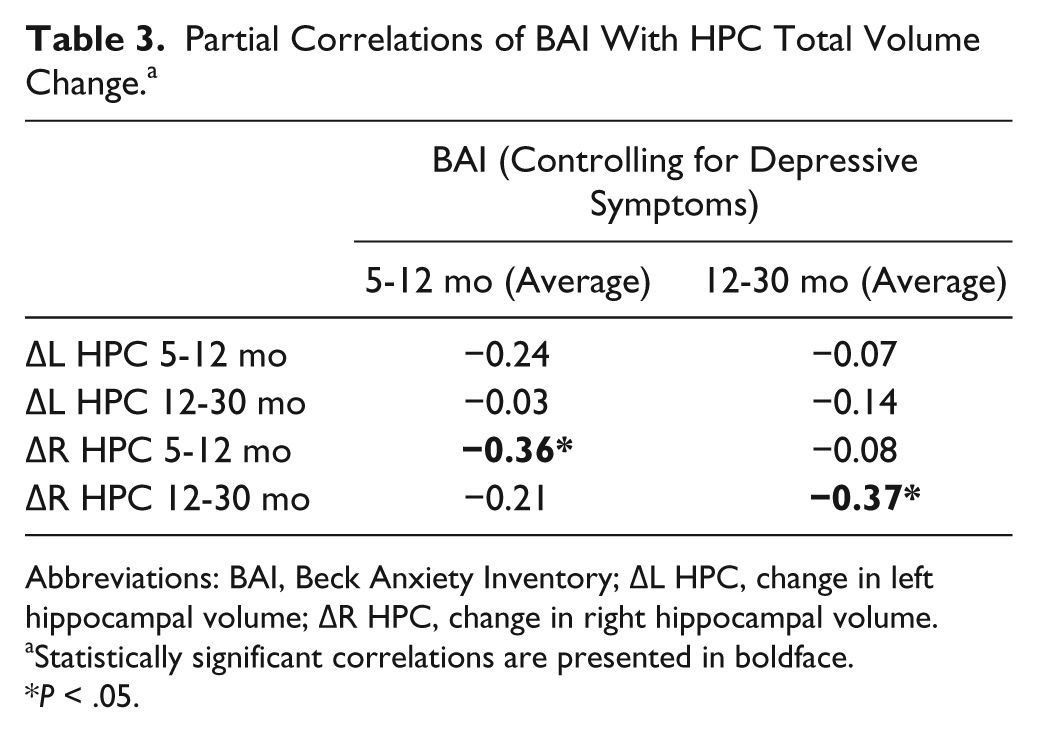
Abbreviations: BAI, Beck Anxiety Inventory; ∆L HPC, change in left hippocampal volume; ∆R HPC, change in right hippocampal volume.
Statistically significant correlations are presented in boldface.
P < .05.
Of the candidate covariates included in the correlational analyses with the BAI and the HPC variables, only TBV showed a statistically significant relationship (Supplementary Table 3), and it was therefore incorporated into the BLDS models. Notably, the correlations of anxiety symptoms with right HPC volume change remained significant when we controlled for TBV in addition to depressive symptoms (see Supplementary Results).
Bivariate Latent Difference Score Modeling
BLDS models were used to examine the dynamic, temporal relations of anxiety symptoms with right HPC total and head, body, and tail volumes from 5 to 12 to 30 months post-TBI (ULDS model selection results for anxiety symptoms and right HPC total, head, body, and tail volumes are presented in Supplementary Results and Supplementary Table 4). Results showed that higher anxiety scores at 5 and 12 months post-TBI significantly predicted subsequent reductions in right HPC total and head volumes from 5 to 12 months (CR = −2.06 for HPC total; CR = −1.86 for HPC head) and from 12 to 30 months (CR = −2.22 for HPC total; CR = −2.24 for HPC head) post-TBI (see cross-lag coefficients γa and γb in the “Anxiety” columns of Table 4).
Results of the Bivariate Latent Difference Score Models Examining the Dynamic Bidirectional Relations of Anxiety Symptoms with Right Hippocampus Total and Head Volumes (N = 80). a
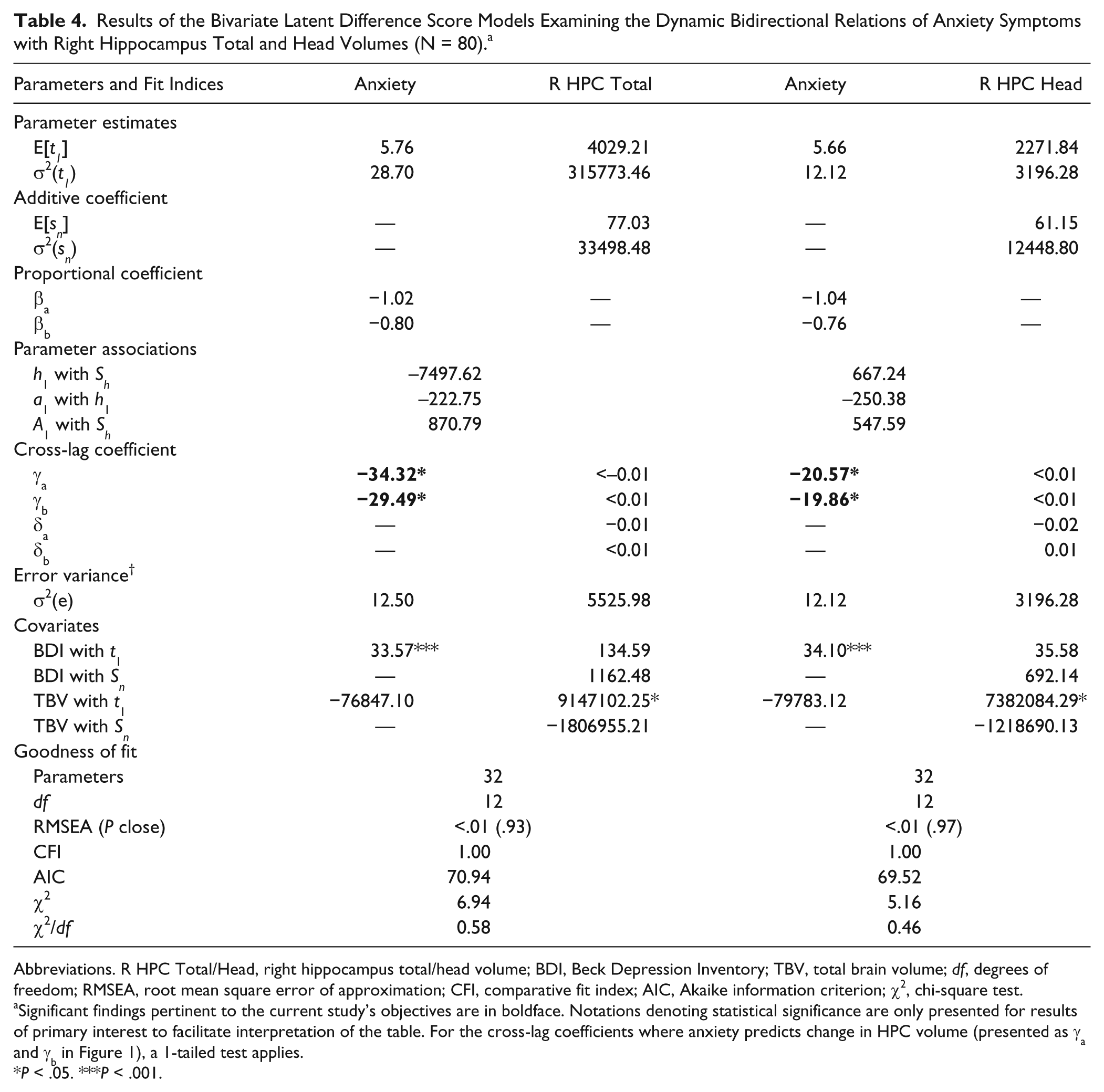
Abbreviations. R HPC Total/Head, right hippocampus total/head volume; BDI, Beck Depression Inventory; TBV, total brain volume; df, degrees of freedom; RMSEA, root mean square error of approximation; CFI, comparative fit index; AIC, Akaike information criterion; χ2, chi-square test.
Significant findings pertinent to the current study’s objectives are in boldface. Notations denoting statistical significance are only presented for results of primary interest to facilitate interpretation of the table. For the cross-lag coefficients where anxiety predicts change in HPC volume (presented as γa and γb in Figure 1), a 1-tailed test applies.
P < .05. ***P < .001.
More specifically, a 1-point increase on the BAI at 5 months post-TBI significantly predicted a reduction in right HPC total volume of 34.32 mm3 and a reduction in right HPC head volume of 20.57 mm3 from 5 to 12 months post-TBI. Similarly, a 1-point increase on the BAI at 12 months post-TBI significantly predicted a reduction in right HPC total volume of 29.49 mm3 and a reduction in right HPC head volume of 19.86 mm3 from 12 to 30 months post-TBI.
Right HPC total and head volumes at 5 and 12 months did not predict change in anxiety scores from 5 to 12 months (CR = −1.19 for HPC total; CR = 0.28 for HPC head) or from 12 to 30 months (CR = 0.41 for HPC total; CR = 0.48 for HPC head) post-TBI, respectively (see cross-lag coefficients γa and γb in the “R HPC Total” and “R HPC Head” columns of Table 4). Furthermore, right HPC total and head volume change from 5 to 12 and from 12 to 30 months post-TBI did not predict anxiety scores at 12 months (CR = −1.60 for HPC total; CR = −1.49 for HPC head) or 30 months (CR = 0.07 for HPC total; CR = 0.46 for HPC head), respectively (see cross-lag coefficients δa and δb in the “R HPC Total” and “R HPC Head” columns of Table 4).
Relationships between anxiety symptoms and HPC volume change were also examined for the right HPC body and tail; however, these relationships were not statistically significant (see Supplementary Results and Supplementary Table 5). Therefore, a unidirectional effect of greater anxiety symptoms predicting right HPC total and head volume loss (but not body or tail) was observed. Stated differently, the results of these models show that anxiety scores at 5 months predicted HPC total and head atrophy from 5 to 12 months post-TBI, and anxiety scores at 12 months predicted HPC total and head atrophy from 12 to 30 months post-TBI. Significant relationships found for the BLDS models of anxiety symptoms with right HPC total and head volumes are presented graphically in Figure 1.
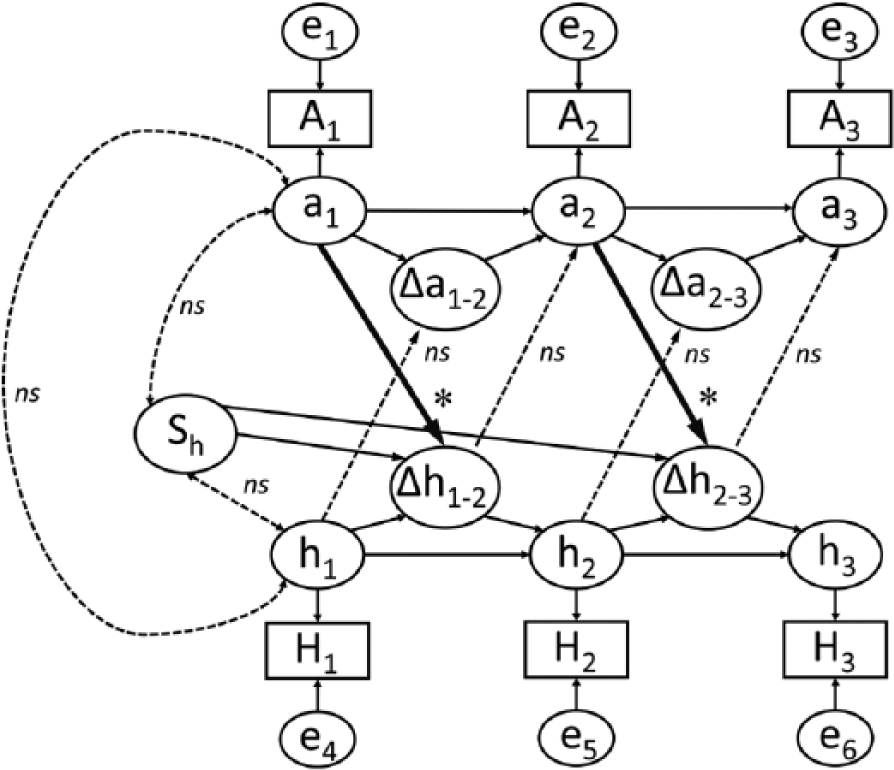
Path diagram of bivariate latent difference score modeling results for anxiety scores and right hippocampus (HPC) total complex and head volumes from 5 to 12 to 30 months post–traumatic brain injury. H1, H2, and H3 represent observed HPC volume at 5, 12, and 30 months postinjury, respectively; A1, A2, and A3 represent participants’ total Beck Anxiety inventory scores at 5, 12, and 30 months post-injury, respectively; and h1-3 and a1-3 represent latent scores for these variables (which take into account measurement error, labeled e1-6). Changes in HPC volume and anxiety symptoms across time points are denoted by Δhi-i and Δai-i, respectively. Arrows representing statistically significant regression weights and covariances are boldfaced, where *P < .05 on a 1-tailed test. Dotted line arrows indicate covariance and regression weight values that did not reach statistical significance. Solid thin lines represent regression weights constrained to 1. Depressive symptoms and total brain volume covariates are omitted to simplify presentation.
Note that these analyses accounted for the effects of depressive symptoms from 5 to 30 months and TBV at 5 months post-TBI; therefore, our results indicate that anxiety symptoms were related to right HPC total and head volume loss independent of these variables.
Discussion
HPC volume loss has been widely observed in the chronic stages of moderate-severe TBI, but contributing factors involved in this loss are unclear. Understanding what factors play a role in HPC volume loss is important for the identification of treatment targets. We undertook to examine the role of self-reported anxiety symptoms in HPC volume loss in chronic TBI. We predicted that anxiety scores at 5 and 12 months would significantly predict reductions in HPC volume from 5 to 12 and from 12 to 30 months post-TBI, respectively, and we expected to see these relationships most strongly for the HPC head bilaterally. In support of our hypotheses, analyses showed that these relationships were significant for the right HPC total, and right HPC head volume, when controlling for depressive symptoms and TBV. Notably, right HPC total and substructure volumes from 5 to 12 and from 12 to 30 months post-TBI did not significantly predict anxiety change scores, suggesting a unidirectional effect of anxiety symptoms on change in volumes of the right HPC total and head in our sample. These findings converge with previous studies documenting an association of anxiety with atrophy of medial temporal lobe structures in a MCI and AD 9 as well as social anxiety disorder. 43
Although HPC volume loss across participants in this study was observed bilaterally, anxiety symptoms predicted volume loss in the right HPC, but not the left, a finding which was not consistent with our hypothesis that the relationship would be observed bilaterally. Further, the relationship between anxiety and the HPC was observed with the right HPC head, but not the body and tail, raising the question whether the right HPC head has a unique vulnerability to anxiety symptoms in TBI. Our findings coincide with those of Sturm et al, 10 who found that emotional contagion (related to anxiety, particularly in social situations)11 -14 was significantly associated with atrophy in the right anterior HPC (ie, the HPC head), but not the posterior right HPC or any region of the left HPC. Also, consistent with the localization of our findings to the right HPC, previous studies of HPC morphometry in social anxiety disorder have shown significantly smaller right, but not left, HPC volumes relative to controls. 43
Though the findings of Sturm et al 10 were correlational, the authors speculated that higher emotional contagion was likely a consequence, rather than a predictor, of the observed volume loss in right temporal lobe structures. Their interpretation that damage to the right anterior HPC may precipitate emotional dysfunction is supported by previous findings that the ventral HPC in rodents (which is functionally analogous to the anterior HPC/HPC head in humans) plays a functional role in anxiety. 44 In the context of human TBI, it is important to consider that a dynamic relationship may exist between anxiety and the HPC. While we did not find evidence that HPC volume loss predicted increased anxiety across the timelines studied, it is possible that HPC atrophy caused by the acute injuries may have resulted in elevated anxiety. For example, previous research into the relationship between anxiety and the HPC suggests that compromise to the HPC (particularly the dentate gyrus) could give rise to elevated anxiety. 45 Our data did not provide support for a dynamic relationship between anxiety and HPC atrophy, whereby anxiety predicts HPC volume loss, and in turn HPC volume loss predicts anxiety. However, further research is needed to investigate this question, employing more frequent assessments and a larger sample size.
The relationship between anxiety and atrophy (particularly of the right HPC head) suggests the involvement of glucocorticoids. Glucocorticoids have been shown to induce both negative HPC remodeling46,47 as well as anxiety-related behavior48,49; thus, the mechanism for the relationship between anxiety symptoms and right HPC volume loss deserves further inquiry. It may be worth noting that anxiety has been associated with prolonged activity of the HPA axis.50,51 One possible interpretation of our findings is that anxiety promotes sustained glucocorticoid release resulting in greater HPC atrophy in regions rich with glucocorticoid receptors (eg, the HPC head).38,47,52,53 Thus, the current findings offer intriguing clinical implications that warrant further investigation.
Limitations
The HPC was of particular interest for this study because it is especially vulnerable in TBI and mediates important cognitive functions known to be impaired following moderate-severe TBI. 54 However, investigation of other relevant structures that are compromised in TBI and have demonstrated associations with anxiety symptoms—for example, the entorhinal cortex, 9 medial prefrontal cortex55,56 and amygdala57,58—would help us to better understand the role of anxiety in the injured brain.
It was shown that GCS scores and HPC total volumes were significantly lower for participants who participated in all three study visits compared with those who only participated in two of the three visits, suggesting that there may have been a systematic loss of participants with less severe injuries, as well as larger HPC volumes, at the second and third time points. As the findings of this study were relevant to HPC volume change rather than static HPC volume, these differences may not have biased our conclusions, however. Critically, right HPC (the region of interest in this study) total volume change scores did not differ significantly between participants with complete versus incomplete data. Nonetheless, the conclusions of this study may be most relevant to individuals with more severe TBIs.
Given that different imaging parameters and quantification techniques are likely to yield different HPC volumes, generalization of our volumetric findings to other studies or samples should be made cautiously. 54 However, a manual for HPC segmentation protocol recently published by the EADC-ADNI group (www.hippocampal-protocol.net)59-61 suggests that the segmentation process used for the current study is likely to be consistent with what has become the recommended protocol for best practice in HPC segmentation.
Another potential limitation of this study is that the level of anxiety observed in our sample is lower than that found in other studies of moderate-severe TBI.28,31 Therefore, the volume loss observed as a function of self-reported anxiety symptoms may not be representative of the moderate-severe TBI population as a whole. Indeed, the findings may underestimate HPC volume loss associated with more severe anxiety in chronic moderate-severe TBI.
Also of note is that our analyses do not account for the relationship of post-traumatic stress disorder (PTSD) with HPC volume loss. Many previous studies have shown that PTSD is associated with reduced HPC volume,62-65 including in the right HPC 64 and HPC head. 65 It should be noted that, while PTSD is indeed an important consideration for mild TBI, 66 PTSD is much less prevalent in moderate-severe TBI because of lack of memory for the event due to PTA and/or loss of consciousness. Where there is PTSD following a TBI, this is found to occur predominantly in those with a PTA of less than 12 hours. 67 All but one participant in our study had a length of PTA greater than 24 hours, suggesting that PTSD likely does not account for significant outcome variance in our study, though in the absence of a measure of PTSD on the participants, this cannot be confirmed.
Conclusions
The current study provides the first evidence that anxiety symptoms predict right HPC volume loss in the chronic stages of moderate-severe TBI, particularly in the right HPC head. It is also the first study to demonstrate a predictor of neurodegeneration in chronic moderate-severe TBI. What is particularly clinically important about the findings is that the relationships are “post-injury,” offering the possibility that treating anxiety symptoms after TBI may reduce the observed HPC atrophy. Future research is needed to replicate the current findings and assess whether treatment of clinically elevated anxiety could mitigate the HPC atrophy that is observed in the chronic stages of TBI.
Footnotes
Acknowledgements
The authors wish to thank Drs David Mikulis and Jerome Maller for their roles in MRI image analysis and HPC tracing, respectively. We would also like to thank Drs Naomi Koerner and Sandra Black for helpful discussion and feedback on this project. Finally, we wish to acknowledge the participants of this study for their time and effort.
Authors’ Note
A previous version of this article appears in a master’s thesis available online through the University of Toronto’s research repository, TSpace.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a Canada Research Chair in Traumatic Brain Injury grant from the Canadian Institutes of Health Research, a Toronto Rehabilitation Institute Student Scholarship, and a Margaret and Howard Gamble Research Grant through the University of Toronto.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
