Abstract
Background. Somatosensory input to the motor cortex may play a critical role in motor relearning after hemiparetic stroke. Objective. The authors tested the hypothesis that motor recovery after hemiparetic stroke relates to changes in responsiveness of the sensorimotor cortex (SMC) to somatosensory input. Methods. A total of 10 hemiparetic stroke patients underwent serial functional magnetic resonance imaging (fMRI) during tactile stimulation and testing of sensorimotor function over 1 year—at early subacute, late subacute, and chronic poststroke time points. Results. Over the subacute poststroke period, increased responsiveness of the ipsilesional SMC to tactile stimulation of a stroke-affected digit correlated strongly with concurrent gains in motor function. Increased responsiveness of the ipsilesional and contralesional SMC over the subacute period also correlated strongly with motor recovery experienced over the first year poststroke. Conclusions. These findings suggest that increased responsiveness of the SMC to somatosensory stimulation over the subacute poststroke period may contribute to motor recovery.
Introduction
The mechanisms underlying recovery of motor function after hemiparetic stroke are incompletely understood. Functional neuroimaging studies suggest that a change in processing of somatosensory information in the sensorimotor cortex (SMC) may play an important role in motor recovery.1-6 Neurophysiological studies have provided evidence that somatosensory input to the motor cortex, via corticocortical connections with the somatosensory cortex, is important for learning new motor skills.7-9 Somatosensory input to the SMC may similarly be critical to relearning motor skills in hemiparetic stroke patients.
We hypothesized that motor recovery after hemiparetic stroke involves increased responsiveness of the SMC to somatosensory input. Patients underwent functional magnetic resonance imaging (fMRI) and sensorimotor function testing at 3 sessions over the first year after hemiparetic stroke—at early subacute, late subacute, and chronic poststroke time points. We tested whether change in SMC responsiveness to somatosensory stimulation over study session intervals related to motor recovery.
Methods
Participants
A total of 10 patients fulfilling the following inclusion criteria were enrolled: (1) first-ever unilateral ischemic stroke sparing precentral and postcentral cortices based on MRI; (2) at study enrollment, stroke symptoms involved weakness of contralesional hand grip, defined by scoring ≤4 on the Medical Research Council Scale (score 0-5; 5 = normal strength) 10 or reduced individuated finger movement evaluated clinically. Exclusion criteria were the following: (1) inability to detect with ≥80% accuracy the tactile stimulus to be used during fMRI; (2) hemisensory neglect or inattention; (3) significant aphasia or altered mental status that would preclude competent participation in study procedures or informed consent; (4) prestroke orthopedic or neurological injury causing somatosensory or motor impairment of the contralesional upper limb that could interfere with sensorimotor testing or confound motor recovery; and (5) MRI risk factor. Patient characteristics are provided in Table 1 and Figure 1. All patients received standard physical rehabilitation during the study period. No patient had a surgical intervention (e.g., carotid endarterectomy) during the study period.
Patient Characteristics
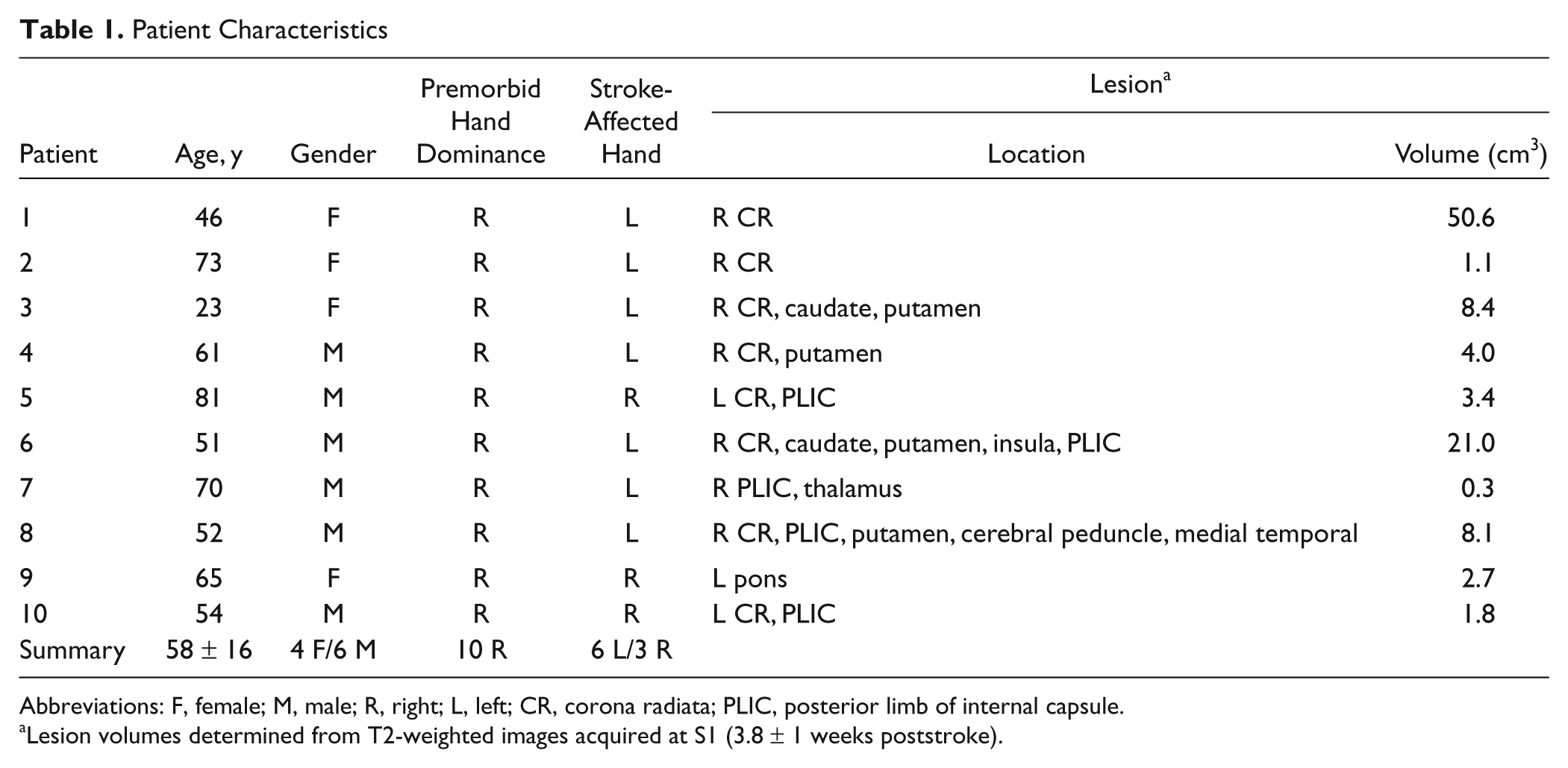
Abbreviations: F, female; M, male; R, right; L, left; CR, corona radiata; PLIC, posterior limb of internal capsule.
Lesion volumes determined from T2-weighted images acquired at S1 (3.8 ± 1 weeks poststroke).
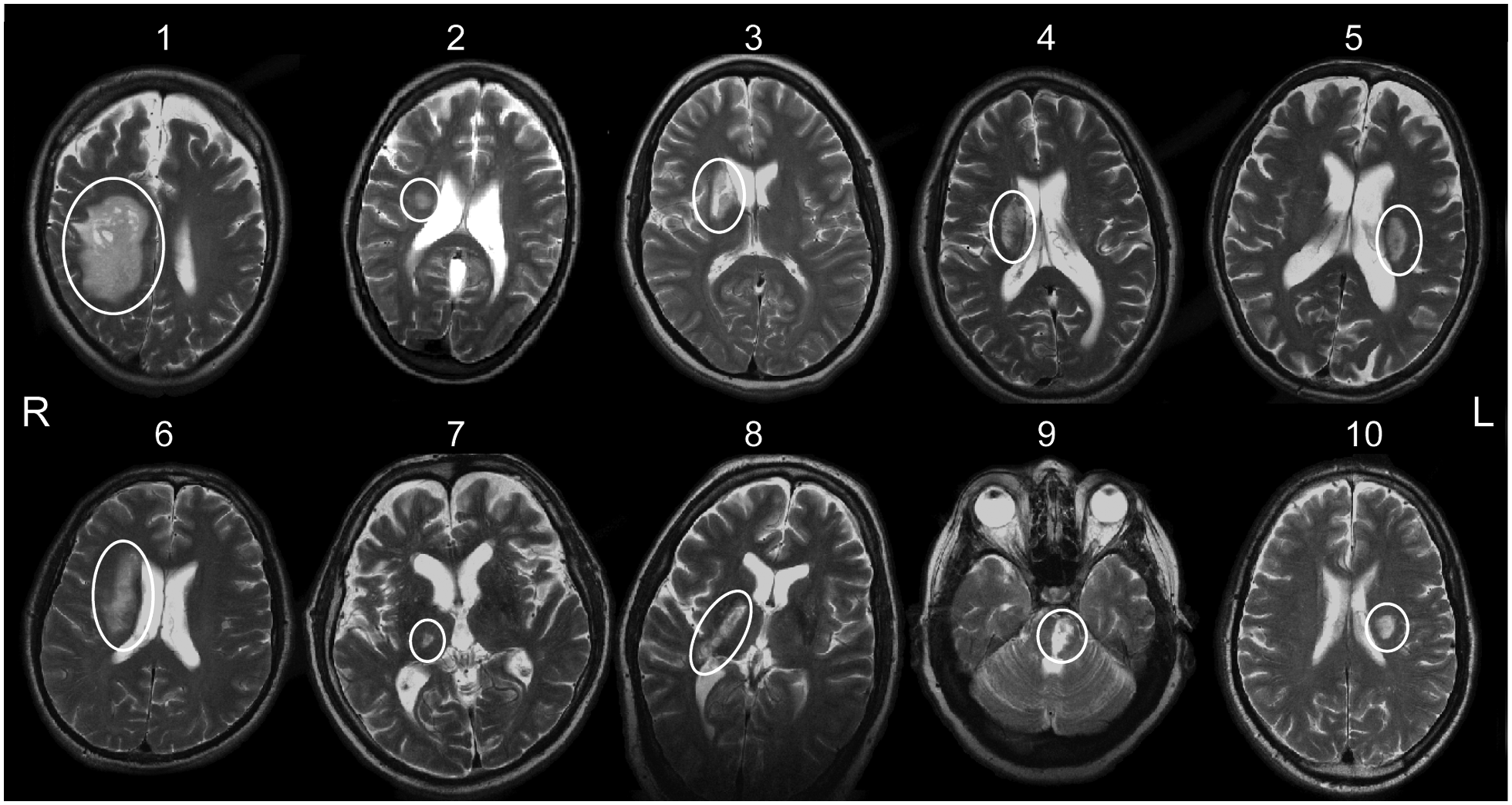
Axial T2-weighted image from each patient at the level where the infarct (circled) was at its maximal cross-sectional extent; patient numbers correspond with those in Table 1.
A total of 7 control participants with no history of stroke and normal neurological examination were also enrolled. These participants were well matched to the enrolled patients with regard to age (mean, 58 years; standard deviation [SD], 16; range, 27-72 years) and gender (3 female and 4 male participants). All controls and patients (prestroke) were right-hand dominant based on the Edinburgh Handedness Inventory (laterality quotient ≥ 62.5; scale: 100 to −100, 0 = ambidexterous). 11
All participants provided written informed consent in accordance with the Human Subjects Committee of the Partners Institutional Review Board.
Study Design
Participants were evaluated at 3 time points. For patients, these evaluations were targeted for 2 to 4 weeks (early subacute), 2.5 to 3 months (late subacute), and 12 months (chronic) poststroke. Actual study visits were 3.8 ± 1 weeks (session 1, S1), 2.7 ± 0.2 months (S2), and 11.8 ± 1.3 months (S3) poststroke; 2 patients (#3 and #6, Table 1) failed to return for the 12-month evaluation. For controls, S2 and S3 were targeted for 3 and 12 months after S1, respectively. Actual intervals were 3.0 ± 0.4 months (S2) and 12.2 ± 0.7 months (S3) relative to S1.
Testing of Sensorimotor Function
Motor function of the stroke-affected upper limb was tested at each study session. The Fugl-Meyer Stroke Scale measured overall motor impairment (score 0-66; 66 = normal). 12 The score was converted to percentage of maximum. A computerized dynamometer measured grip strength (mean peak force, 2 × 5-s trials). 13 Grip strength of the stroke-affected hand was normalized, in percentage, to that of the opposite hand. The Jebson Hand Function Test measured hand function in everyday activities, with 5 of the original 7 items applied (turning cards, moving small objects, stacking checkers, moving empty can, and moving full cans) based on prior recommendations. 14 The summed time for the stroke-affected hand to accomplish the tasks was normalized, in percentage, to that taken by the opposite hand, with 0% recorded if the stroke-affected hand was not able to perform the Jebson Hand Function Test items. While these tests measure different aspects of motor function, they were highly correlated among the patients (r = 0.86-0.88). Accordingly, to provide a reasonable univariate measure of motor function while minimizing multiple statistical comparisons, a composite motor function score was computed for each study session for each patient by averaging the 3 normalized scores. Previous serial studies have used similar data reduction strategies to evaluate recovery-related changes in brain function.15,16 Motor recovery was computed as the composite motor function score at the later session minus the earlier session.
Also, at each study session, tactile sensitivity at the glabrous surface of the middle phalanx of both third digits (D3) was tested using Semmes–Weinstein monofilaments (0.008 to 300 g, North Coast Medical, Inc, San Jose, California). This was the same skin surface to which tactile stimulation was applied during fMRI (see below). Testing was conducted using a forced-choice, adaptive psychophysical algorithm 17 over 40 trials, with the tactile sensitivity determined as the force perceived with 80% accuracy.
Image Acquisition
A 3T Siemens Allegra MRI scanner was used for image acquisition. A thermoplastic (WFR/Aquaplast Corporation, Avondale, Pennsylvania) head restraint was custom fit at S1 and reused at subsequent sessions. This system has been shown to reduce head motion during scanning and enable reproducible intrasubject head positioning at serial imaging sessions. 18 The upper limbs of each participant were positioned using an apparatus we described previously, 6 allowing easy access to the volar surface of D3 for receiving tactile stimulation during fMRI and minimizing stimulation-induced limb movement.
At S2, high-resolution, T1-weighted, magnetization-prepared rapid gradient echo (MP-RAGE) images (repetition time [TR] = 7 ms; echo time [TE] = 3 ms; flip angle [α] = 7°; field of view (FOV) = 256 × 256 mm2; slice thickness = 1.33 mm; voxel size = 1 × 1 × 1.33 mm3) were collected for cortical surface modeling and spatial normalization. At S1 and S3, lower resolution MP-RAGE images were collected with the same parameters except for voxel size (1 × 1 × 2.98 mm3) for spatial normalization of BOLD data acquired at these sessions. T2-weighted turbo spin-echo images (TR = 10 s; TE = 65 ms; α = 120°; FOV = 210 × 210 mm; slice thickness = 5 mm; interslice gap = 1 mm; voxel size = 0.45 × 0.45 × 6 mm) were collected at S1 for lesion volume determination.
For fMRI, a blood oxygenation level–dependent (BOLD), T2*-weighted, gradient-echo, echo planar imaging sequence (TR = 2 s; TE = 30 ms; α = 90°; FOV = 200 × 200 mm2; matrix size = 64 × 64; slice thickness = 5 mm; interslice gap = 1 mm; voxel size = 3.125 × 3.125 × 6 mm3; number of slices = 20; number of acquisitions/slice = 130) equipped with real-time correction for head motion 19 was used. During BOLD imaging, periods of suprathreshold, punctate tactile stimulation (6 × 20 s) alternated with periods of no stimulation (7 × 20 s). Tactile stimulation to the glabrous surface of the D3 middle phalanx using a Semmes–Weinstein monofilament (60 g) was delivered by an investigator and was paced by a 3-Hz computer-generated metronome (MacStim, version 2.6) via pneumatic headphones. We chose to apply this intensity of tactile stimulation because our previous studies found that it elicited robust SMC activation in stroke patients and normal participants without discomfort.6,20 In all, 4 functional runs were collected during unilateral D3 stimulation (2 right D3 and 2 left D3) to each participant. The side of stimulation during the first functional run was randomized across participants and alternated during subsequent functional runs for each individual. Participants were instructed to close their eyes. (See Authors’ Note for additional information).
Image Analysis
MP-RAGE and BOLD images from patients with left-sided stroke (Patients #5, 9, 10) were flipped midsagittally, thereby lateralizing the lesioned hemisphere to the right in all patients. Accordingly, hereafter, the right hemisphere will be considered the ipsilesional hemisphere contralateral to the left, stroke-affected hand.
FreeSurfer software 21 was used to compute a model of each individual’s cortical surface based on the high-resolution MP-RAGE images.22,23 Each cortical surface model was spatially normalized to a spherical surface template using an automated procedure that optimally aligns major cortical gyri and sulci. 24 Visual inspection of each normalized cortical surface model to the spherical surface template in the region of the precentral and postcentral cortices—our region of interest—showed good alignment, as would be expected given that the stroke spared these cortices in all patients. Each normalized cortical surface was transformed to a node-based cortical surface using cortical surface–based tools of SUMA 25 within AFNI software. 26
BOLD images were preprocessed using AFNI/SUMA software. They were motion-corrected, intensity-normalized, and coregistered with the high-resolution structural volume. The BOLD data were mapped to the node-based cortical surface, then spatially smoothed using a 2-dimensional Gaussian filter 27 with a full width at half maximum (FWHM) of 6 mm.
To test our main hypothesis, we sought to first empirically identify regions of the SMC that normally respond to D3 tactile stimulation. Therefore, using BOLD data from controls, a general linear model was fit nodewise that included a stimulus input function defined as a boxcar convolved with a canonical hemodynamic response and baseline and linear drift terms. The resultant regression coefficient reflecting the stimulation-induced BOLD response was entered into a nodewise mixed-model regression analysis (3dLME program) to test for a possible effect of time interval from S1, with subject as a random effect. Output statistical maps were corrected for multiple comparisons to a clusterwise significance level of .01. The parameters for this correction were determined by Monte Carlo simulations (10 000 iterations) performed on synthesized, white Gaussian noise data that were resampled onto the cortical surface and smoothed. These simulations indicated that with the node probability set to 0.05, a cluster having a surface area of ≥250 mm2 controlled the false-positive rate to 0.01. Mixed-model regression analyses revealed no significant effect of time on the BOLD response to right or left D3 stimulation in controls. Accordingly, the group-average BOLD response to unilateral tactile stimulation was determined by averaging regression coefficients across the 3 imaging sessions of each control, then performing a 1-sample t test nodewise. Resultant statistical maps were corrected for multiple comparisons to a clusterwise significance level of .01 as described above. Significant clusters in the SMC, considered to include the precentral and postcentral cortices superior to the inferior frontal and intraparietal sulci, were deemed the SMC functional regions of interest. These SMC functional regions of interest were applied in analysis of BOLD data acquired from patients during tactile stimulation.
For each functional run acquired from patients, the BOLD signal time series was averaged over all nodes of each SMC functional region of interest; the linear trend was removed, and the detrended time series of replicate functional runs was averaged. Within a SMC functional region of interest, the mean BOLD response over the 6 epochs of stimulation relative to the no-stimulation baseline was calculated in percentage signal change. Finally, the average BOLD response over the 2- to 22-s interval from stimulus onset was calculated, with the 2-s offset applied to account for the hemodynamic delay; these values were entered into secondary analyses. For the purpose of comparing the BOLD response in the SMC functional regions of interest in patients relative to controls, the same computations were done on data from controls.
Statistical Analysis
Statistical analyses were conducted using JMP software (version 8.01; SAS Institute Inc). Mixed-model regression analyses were used to test for an effect of time poststroke on (1) motor function, (2) tactile sensitivity, and (3) BOLD response to tactile stimulation in SMC regions of interest, with subject as a random effect for each analysis. Post hoc paired t tests were applied when appropriate. Unpaired Student t tests were used to test for between-group differences in the average BOLD response in SMC regions of interest. Correlation analyses were used to evaluate relationships between (1) motor recovery over 2 study session intervals, (2) change in BOLD response and change in motor function over study sessions, and (3) change in BOLD response over sessions and sensorimotor function at a single study session.
The .05 2-tailed significance level was applied to test hypotheses. Results are presented as the mean ± SD unless otherwise noted.
Results
Sensorimotor Function
At the early subacute study session (S1), patients showed a range in motor function of the stroke-affected upper limb (composite motor function, 2.5%-91.6%; Figure 2A; see Supplementary Figure online for measures on each test). As a group, patients showed significant motor recovery over the year (P < .001, mixed-model regression analysis), with a significant gain over both study session intervals (S1 to S2: P < .05; S2 to S3: P = .01; paired t tests; Figure 2B). Motor recovery over the subacute period (S1 to S2) was not significantly correlated with motor recovery over the later period (S2 to S3: r = 0.49, P = .22), but was highly correlated with motor recovery over the year (S1 to S3: r = 0.91, P < .005).
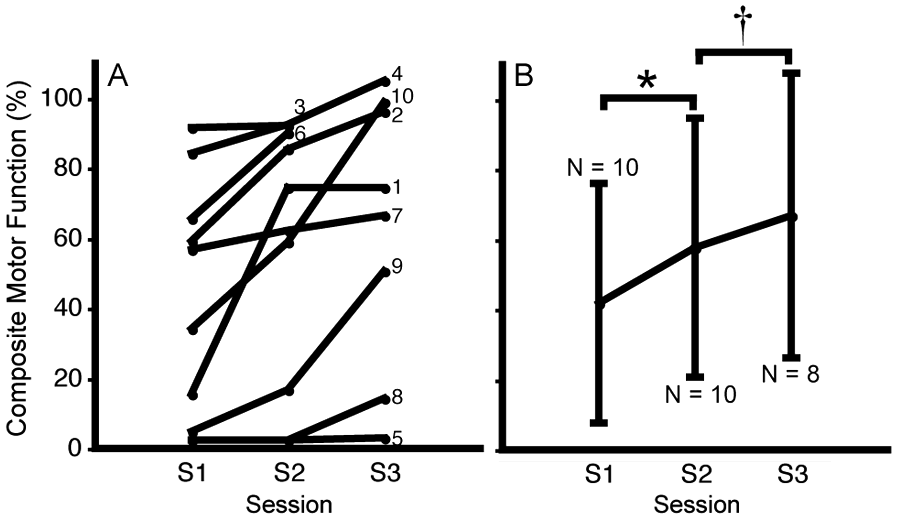
Composite motor function scores of the stroke-affected upper limb over the 3 study sessions: S1 to S3 at about 1, 3, and 12 months poststroke, respectively: A. Trajectory of composite scores for each patient. B. Mean ± standard deviation composite scores at each session. Number (N) of patients included in calculations is noted. *P < .05 (N = 10), †P < .01 (N = 8), paired t test.
At S1, patients exhibited a range in tactile sensitivity of the stroke-affected D3 (0.008-4 g; see Supplementary Table). Tactile sensitivity of only 1 patient (#7) was outside the range of the opposite-sided D3 (0.008-1 g). Notably, even with consideration given to this patient, tactile sensitivity of all patients was more than an order of magnitude greater than the strength of tactile stimulation delivered during fMRI (60 g). Accordingly, the stimulus applied during fMRI to the stroke-affected D3 at S1 was detectable in all patients, as per enrollment criterion. Tactile sensitivity of the stroke-affected D3 did not change significantly over the study period (P = .22; mixed-model regression analysis).
BOLD Response to Tactile Stimulation
Figure 3 shows regions of the SMC with a significant BOLD response to D3 tactile stimulation in controls. Left D3 stimulation elicited a significant positive BOLD response in the right, contralateral hand knob region 28 that included the postcentral gyrus and precentral gyrus (Figure 3A1), with the BOLD signal increasing relative to baseline (Figure 3A2). Left D3 stimulation also elicited a significant negative BOLD response in the left, ipsilateral postcentral and precentral gyri that extended both superiorly and inferiorly from the hand knob (Figure 3B1), with the BOLD signal decreasing relative to baseline (Figure 3B2). Right D3 stimulation yielded a significant positive BOLD response in the left, contralateral hand knob region of the postcentral and precentral gyri (Figure 3C1 and 3C2). Right D3 stimulation also yielded a negative BOLD response in the right, ipsilateral SMC, but it did not reach statistical significance (data not shown). The former 3 SMC regions were thereafter regarded as the functional regions of interest.
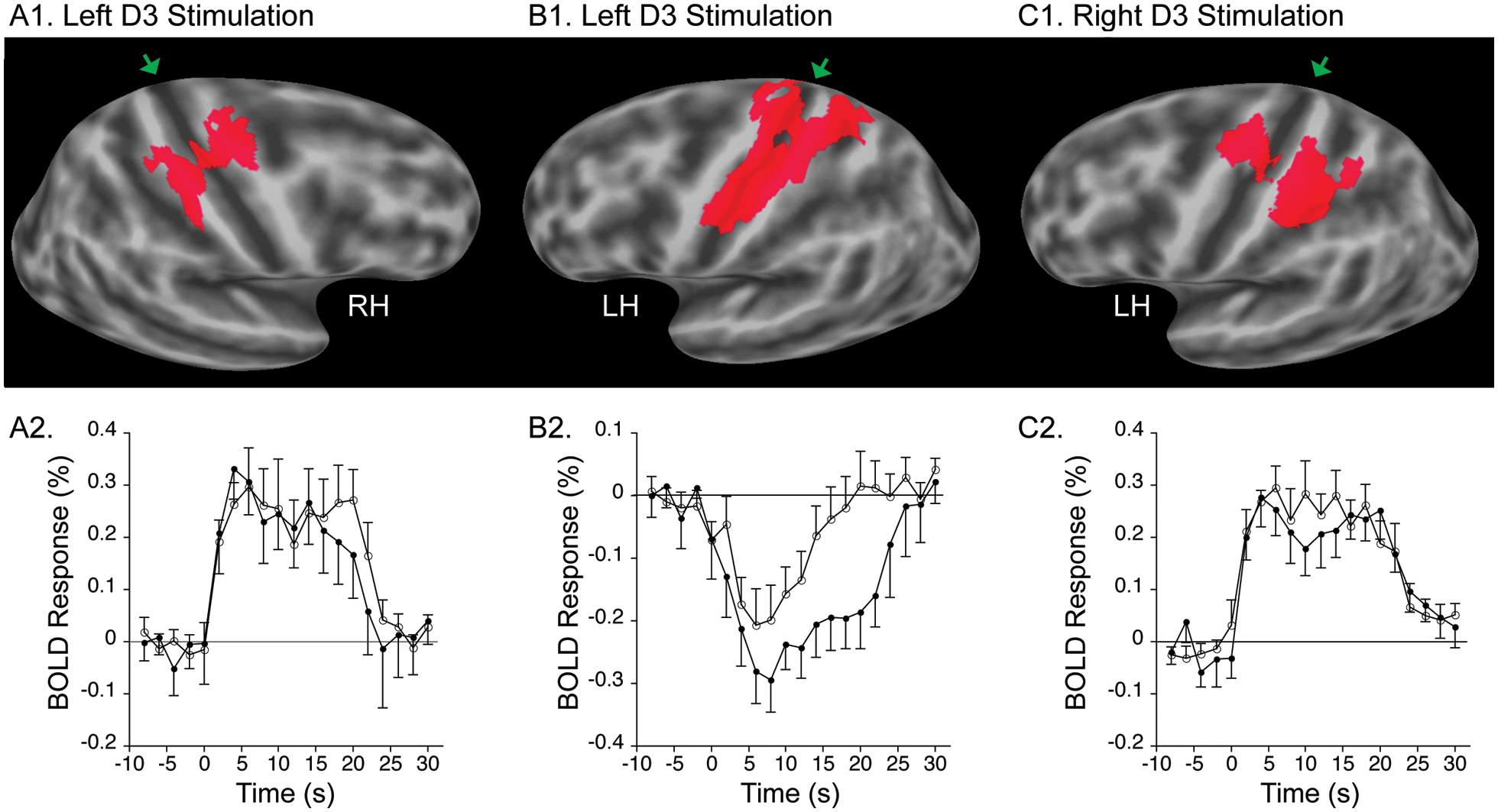
BOLD response to unilateral tactile stimulation in SMC functional regions of interest. Top row: Regions of the SMC with a significant (P < .01, corrected) BOLD response to unilateral tactile stimulation in controls. A1. Region in right hemisphere (RH) of SMC with positive BOLD response to left D3 stimulation. B1. Region in left hemisphere (LH) of SMC with negative BOLD response to left D3 stimulation. C1. Region in LH of SMC with positive BOLD response to right D3 stimulation. Maps are overlaid on a model of the group-average, inflated cortical surface. Dark gray region is the fundus of a sulcus; light gray region is the crown of a gyrus. Green arrowheads point to central sulcus. Bottom row: Mean BOLD response (±SEM) to unilateral tactile stimulation in controls and patients in the respective SMC functional regions of interest shown in the top row. Filled circles correspond to controls and open circles to patients. Abbreviations: BOLD, blood oxygenation level–dependent; SMC, sensorimotor cortex; SEM, standard error of the mean.
The BOLD response in each SMC functional region of interest in controls was compared with that in patients, with the response of each participant averaged over study sessions. The negative BOLD response in the left (contralesional) SMC to left (stroke-affected) D3 stimulation was diminished in patients to a level that neared statistical significance (patients: −0.11% ± 0.13%; controls: −0.21% ± 0.08%; P = .053, unpaired t test; Figure 3B2). There was no significant between-group difference in the positive BOLD response in the right (ipsilesional) SMC to left (stroke-affected) D3 stimulation (Figure 3A2) nor in the positive BOLD response in the left SMC to right (stroke-unaffected) D3 stimulation (Figure 3C2).
Change in patients’ BOLD response in each SMC functional region of interest over the year was evaluated. The positive BOLD response in the ipsilesional SMC to stroke-affected D3 stimulation showed a significant change over time (P < .05, mixed-model regression analysis), with post hoc tests revealing a significant increase in the BOLD response over the year (S1: 0.16% ± 0.37%, S3: 0.42% ± 0.31%; P < .05, paired t test; Figure 4). There was no significant effect of time on the negative BOLD response in the contralesional SMC to stroke-affected D3 stimulation nor on the positive BOLD response in the contralesional SMC to stroke-unaffected D3 stimulation.
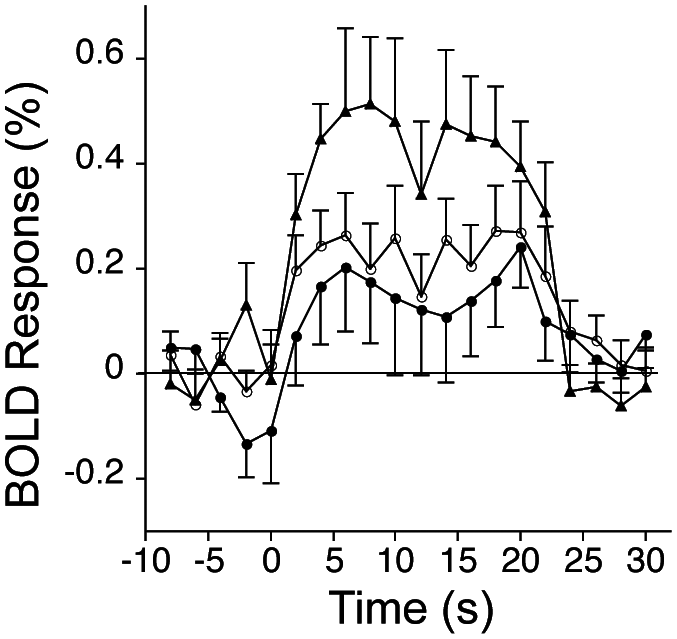
BOLD response (mean ± SEM) to left, stroke-affected D3 stimulation in the right, ipsilesional SMC at each of the 3 study sessions in patients: the average BOLD response during the 2- to 22-s poststimulus onset interval was significantly greater at S3 relative to S1 (P < .05, paired t test). S1 = filled circles; S2 = open circles; S3 = filled triangles. Abbreviations: BOLD, blood oxygenation level–dependent; SEM, standard error of the mean; SMC, sensorimotor cortex.
Correlations Between Change in BOLD Response and Sensorimotor Function
Relationships between change in the BOLD response to stroke-affected D3 stimulation and motor recovery of the stroke-affected upper limb over study session intervals were evaluated. Over the subacute period (S1 to S2), there was a significant positive correlation between change in the BOLD response in the ipsilesional SMC and motor recovery (r = 0.81; P < .005; Figure 5A). This strong correlation was not simply an epiphenomenon of patients’ initial level of sensorimotor function because change in ipsilesional SMC responsiveness was not significantly correlated with S1 motor function (r = −0.20; P = .58) or S1 tactile sensitivity (r = −0.27; P = .46). Over the subacute period, there was also a trend toward a significant correlation between change in the BOLD response in the contralesional SMC and motor recovery (r = 0.59; P = .07).
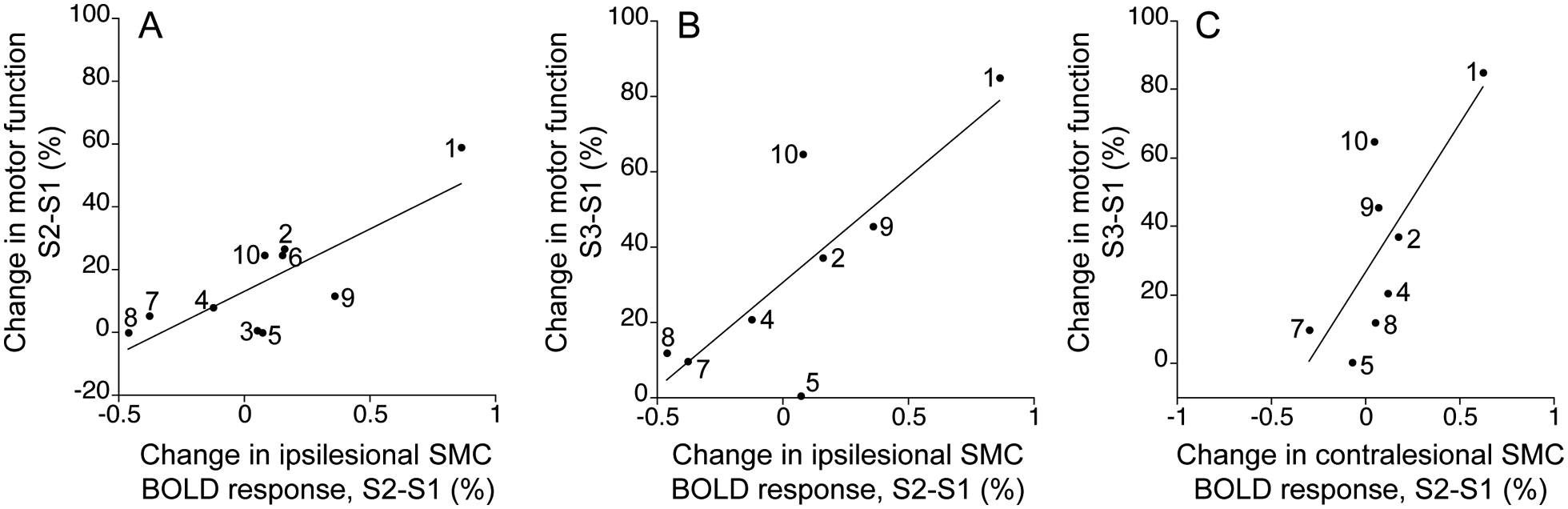
Plots of relationship between change in BOLD response to stroke-affected D3 stimulation and motor recovery of the stroke-affected upper limb over study session intervals: numbers identify patients as listed in Table 1. A. Correlation between change in BOLD response in ipsilesional SMC and motor recovery over subacute period (S1 to S2; r = 0.81; P < .005). B. Correlation between subacute (S1 to S2) change in BOLD response in ipsilesional SMC and motor recovery over full study period (S1 to S3; r = 0.80; P < .05). C. Correlation between subacute (S1 to S2) change in BOLD response in contralesional SMC and motor recovery over full study period (S1 to S3; r = 0.77; P < .05). Abbreviations: BOLD, blood oxygenation level–dependent; SMC, sensorimotor cortex.
Motor recovery over the year (S1 to S3) significantly and positively correlated with change in the stimulation-induced BOLD response over the subacute period (S1 to S2) in the ipsilesional SMC (r = 0.80; P < .05; Figure 5B) and contralesional SMC (r = 0.77; P < .05; Figure 5C). These results indicate that long-term motor recovery is related to change in the SMC BOLD response over the subacute period. Partial correlation analyses revealed that the correlations between long-term motor recovery (S1 to S3) with subacute change (S1 to S2) in the BOLD response in the ipsilesional and contralesional SMC were no longer significant after accounting for each variable’s correlation with subacute motor recovery. Collectively, these results suggest that subacute change in SMC responsiveness correlates with subacute motor recovery, and by virtue of a strong correlation between subacute and long-term motor recovery, the subacute change in SMC responsiveness also predicts long-term motor recovery.
In notable contrast to the strong correlation between change in the BOLD response to stroke-affected D3 stimulation and motor recovery over the subacute period, the correlation between change in the stimulation-induced BOLD response and motor recovery from the late subacute session to chronic session (S2 to S3) was weak (ipsilesional SMC: r = −0.20; contralesional SMC: r = −0.20). There was also no significant correlation between change in the BOLD response in the contralesional SMC resulting from stroke-unaffected D3 stimulation and motor recovery of the stroke-affected upper limb over any study session interval. Together, these findings indicate that change in SMC responsiveness to stroke-affected D3 stimulation is linked most strongly to motor recovery during the subacute poststroke period.
Discussion
Prior studies suggest that somatosensory input to the motor cortex is normally important for learning new motor skills7-9 and may play a critical role in motor relearning after hemiparetic stroke.29-31 This serial fMRI study probed the relationship between motor recovery and change in SMC responsiveness to tactile stimulation over the first year poststroke in hemiparetic patients. The most important finding was that increased responsiveness of the ipsilesional SMC over the subacute period correlated with concurrent motor recovery and predicted motor recovery experienced over the year. This finding suggests a strong link between change in processing of somatosensory information in the SMC during the early poststroke period and motor recovery in hemiparetic patients.
BOLD Response to Tactile Stimulation
Tactile stimulation to D3 of controls elicited a positive BOLD response in the hand knob region of the contralateral SMC involving the postcentral and precentral gyri. The sign and location of this BOLD response is consistent with findings of our prior fMRI studies6,20 and studies by other groups that delivered tactile stimulation to a digit. 32 D3 tactile stimulation in controls also resulted in a negative BOLD response in the ipsilateral SMC, reaching statistical significance with left- but not right-sided stimulation. A negative BOLD response in the SMC ipsilateral to digit tactile stimulation in normal individuals has been described previously 33 and may be secondary to the positive BOLD response in the contralateral SMC. Neuroanatomical studies have observed transcallosal connections between digit representations in area 2 of the somatosensory cortex 34 and between forelimb representations in the primary motor cortex.35,36 Intrahemispheric SMC connections 37 may have contributed to spatial spreading of the negative BOLD response in the ipsilateral SMC. The neurovascular mechanism underlying the stimulus-induced negative BOLD response is an area of active research, though it is generally believed to represent decreased local blood flow caused by reduced neuronal activity.38,39 The nearly significant reduction in the negative BOLD response in the contralesional SMC may reflect impaired intracortical inhibition, a phenomenon observed by others in stroke patients.40,41
BOLD Response Related to Motor Recovery
From about 1 month to 1 year poststroke, the BOLD response in the ipsilesional SMC to tactile stimulation of the stroke-affected D3 increased significantly. This increase in SMC responsiveness was not paralleled by a significant change in tactile sensitivity of the stroke-affected digit. A previous serial study in hemiparetic stroke patients with minimal somatosensory deficit showed that the BOLD response in the ipsilesional SMC during tactile exploration increased from 2 to 4 weeks to >1 month poststroke. 42 Together, these studies suggest that the ipsilesional SMC increases its response to a relatively fixed level of somatosensory input from the stroke-affected hand over the initial months following stroke, perhaps reflecting a loss of intracortical inhibition in the ipsilesional SMC early after stroke. 41
The functional relevance of increased SMC responsiveness to somatosensory stimulation is supported by the observed relationships to motor recovery. Increased responsiveness of the ipsilesional SMC to tactile input over the subacute poststroke period correlated with motor recovery of the upper limb over the same period. Although this correlation does not necessarily mean that increased SMC responsiveness causes motor recovery, the result suggests that the 2 variables are linked. Prior studies showed that somatosensory stimulation to hemiparetic stroke patients results in improved motor function30,43-46 and motor cortex plasticity.46,47 Together, these studies raise the possibility that an increase in ipsilesional SMC responsiveness to somatosensory stimulation over the subacute period is an important factor in motor recovery.
In contrast to the strong correlation between change in ipsilesional SMC responsiveness and motor recovery over the subacute poststroke period, this correlation was weak over the subacute to chronic poststroke period. Collectively, these findings suggest 2 phases of motor recovery related to changes in ipsilesional SMC responsiveness. The first phase may be within the first few months after stroke when a gain in SMC responsiveness to somatosensory input is linked directly to motor recovery. The second phase may be over the remaining months of the first year poststroke when restorative mechanisms other than enhanced SMC responsiveness to somatosensory input play a more significant role in motor recovery. Previous animal and clinical studies have similarly suggested that functional recovery proceeds by successive phases during which different brain restorative mechanisms dominate.16,48
The patients in our study exhibited a range in change in SMC responsiveness in relation to motor recovery over the subacute period. This range suggests differences in spontaneous plasticity of the SMC among subacute stroke patients, perhaps related to genetic (e.g., brain-derived neurotrophic factor polymorphism 49 ) and other inherent variations. As others are exploring the use of supplemental somatosensory stimulation to promote motor recovery in stroke patients,30,43-46 the variability in SMC change we observed may translate into differential effectiveness of this approach because of individual differences in the linkage of somatosensory stimulation to favorable SMC plasticity.
Conclusions
Findings of the current study suggest that over the subacute poststroke period, the increase in SMC responsiveness to somatosensory input is linked to motor recovery in hemiparetic patients. These findings provide new insight into plastic changes in the SMC that may play a role in motor recovery.
Footnotes
For patients with sufficient motor ability and all controls, separate functional runs were conducted while a hand motor task was performed. Results of these data will be presented elsewhere.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
This study was supported by grants from the American Heart Association–New England Affiliate (to JDS and RMD), the Royal Netherlands Academy of Arts and Sciences (to RMD), the Netherlands Heart Foundation (2003B196), the National Institutes of Health K23-HD044425 (to JDS), NCRR (P41-RR14075), and the MIND Institute.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
