Abstract
Background. The application of resistive forces has been used during body weight–supported treadmill training (BWSTT) to improve walking function after spinal cord injury (SCI). Whether this form of training actually augments the effects of BWSTT is not yet known. Objective. To determine if robotic-applied resistance augments the effects of BWSTT using a controlled experimental design in a rodent model of SCI. Methods. Spinally contused rats were treadmill trained using robotic resistance against horizontal (n = 9) or vertical (n = 8) hind limb movements. Hind limb stepping was tested before and after 6 weeks of training. Two control groups, one receiving standard training (ie, without resistance; n = 9) and one untrained (n = 8), were also tested. At the terminal experiment, the spinal cords were prepared for immunohistochemical analysis of synaptophysin. Results. Six weeks of training with horizontal resistance increased step length, whereas training with vertical resistance enhanced step height and movement velocity. None of these changes occurred in the group that received standard (ie, no resistance) training or in the untrained group. Only standard training increased the number of step cycles and shortened cycle period toward normal values. Synaptophysin expression in the ventral horn was highest in rats trained with horizontal resistance and in untrained rats and was positively correlated with step length. Conclusions. Adding robotic-applied resistance to BWSTT produced gains in locomotor function over BWSTT alone. The impact of resistive forces on spinal connections may depend on the nature of the resistive forces and the synaptic milieu that is present after SCI.
Introduction
Body weight–supported treadmill training (BWSTT) has been successfully used to improve locomotor recovery after spinal cord injury (SCI) and stroke. The effectiveness of BWSTT, however, has been questioned in light of evidence that it does not enhance recovery more than other forms of gait rehabilitation.1,2 Recent studies have explored applying resistive forces to augment the effects of BWSTT. Resistive forces applied at the ankle or the hip and knee during BWSTT have been successful in enhancing forward swing of the leg in patients with incomplete SCI.3,4 Importantly, these kinematic improvements translated to faster walking speed 3 and better performance of skilled locomotor tasks. 5
Although these results were promising, it is not yet clear whether training with resistive forces provided an advantage over BWSTT alone. There have been no studies that compared the effects of BWSTT with resistance versus BWSTT alone. Thus far, the effects of robotic resistance have been only compared to training with robotic assistance in which the robot fully assisted limb movements.3,5 Additional controlled studies were necessary to determine whether using robotic forces to resist leg movements could augment the effects of BWSTT. A second unanswered question was whether applying vertical resistive forces affected limb movements. BWSTT with resistive forces has been consistently shown to affect step length.3,4,6-9 Other characteristics such as step height or lift velocity may be affected with purely vertical resistive forces, but this has not yet been systemically studied.
One beneficial effect of BWSTT is to enhance synaptic connections within the lumbar spinal cord. We and others have shown in rodent models that BWSTT modulates synaptic inputs to motor neurons in the ventral horn.10-13 For example, the expression of synaptophysin, a marker for synapses, increased around ventral horn motor neurons in SCI rats that received BWSTT.10,11 The evidence to date suggested that this effect was activity dependent, meaning that synapses were strengthened by actively generating stepping movements.13-15 Because resistance training increases the effort and muscle activity needed to generate movements, 9 it follows that training with resistive forces would further drive synaptic plasticity within the spinal circuits. If true, then it would provide more rationale for incorporating resistive forces into BWSTT, but this has not yet been examined.
In the present study, we compared the effects of BWSTT with resistive forces versus BWSTT alone in a rodent model of SCI and robotic training.16,17 We also compared the effects of training with horizontal versus vertical resistive forces. We hypothesized that training with resistive forces would be superior to BWSTT administered without resistive forces. We also expected that horizontal and vertical resistance would induce different adaptations in step cycle characteristics. Furthermore, these changes would be associated with an increase in synaptophysin expression in the ventral horn compared with BWSTT alone. Given that the rodent model has been a successful test bed for robotic BWSTT algorithms,18,19 the findings have implications for the use of resistive forces during BWSTT in humans with SCI.
Methods
Experimental Design
A total of 40 female Sprague Dawley rats received a severe T9 spinal cord contusion injury. After 2 weeks of recovery, baseline locomotor ability was assessed using a robotic BWSTT system. The rats were distributed into 4 experimental groups. One group, referred to as the untrained group (UT), did not receive any training after SCI. A second group received standard BWSTT (ST) without resistive forces, as described previously. 20 A third group was trained with body weight support and velocity-dependent resistance applied against the horizontal movements of the ankle (HT). The fourth group was also trained with body weight support but received velocity-dependent resistance applied against the vertical movements (VT). Some rats were removed from the study for health reasons, resulting in an unequal number of rats in each group (UT, n = 8; ST, n = 9; HT, n = 9; VT, n = 8). A final test of stepping occurred after 6 weeks of training. A day later, all the rats were anesthetized with isofluorane and then underwent intracardiac perfusion, and the spinal cords were removed and processed. All procedures were carried out in accordance with NIH guidelines and protocols approved by the Institutional Animal Care and Use Committee at California State University, Los Angeles (Approval No. 15-3).
Procedures
The spinal cord contusion, robotic training, locomotor tests, and analyses were performed as previously described16,17,21-23 (also see supplementary material).
Results
Velocity-Dependent Forces Resisted Horizontal or Vertical Ankle Movement During Stepping
We used a robotic device to apply resistive forces to the ankles while the rats underwent BWSTT (Figure 1A). For rats trained with a horizontal resistance (HT group), the robot applied forces to oppose forward and backward ankle movements (see Forcex arrows in Figure 1B). For rats trained with a vertical resistance (VT group), the robot applied forces to oppose upward and downward ankle movement (see Forcey arrows in Figure 1C).
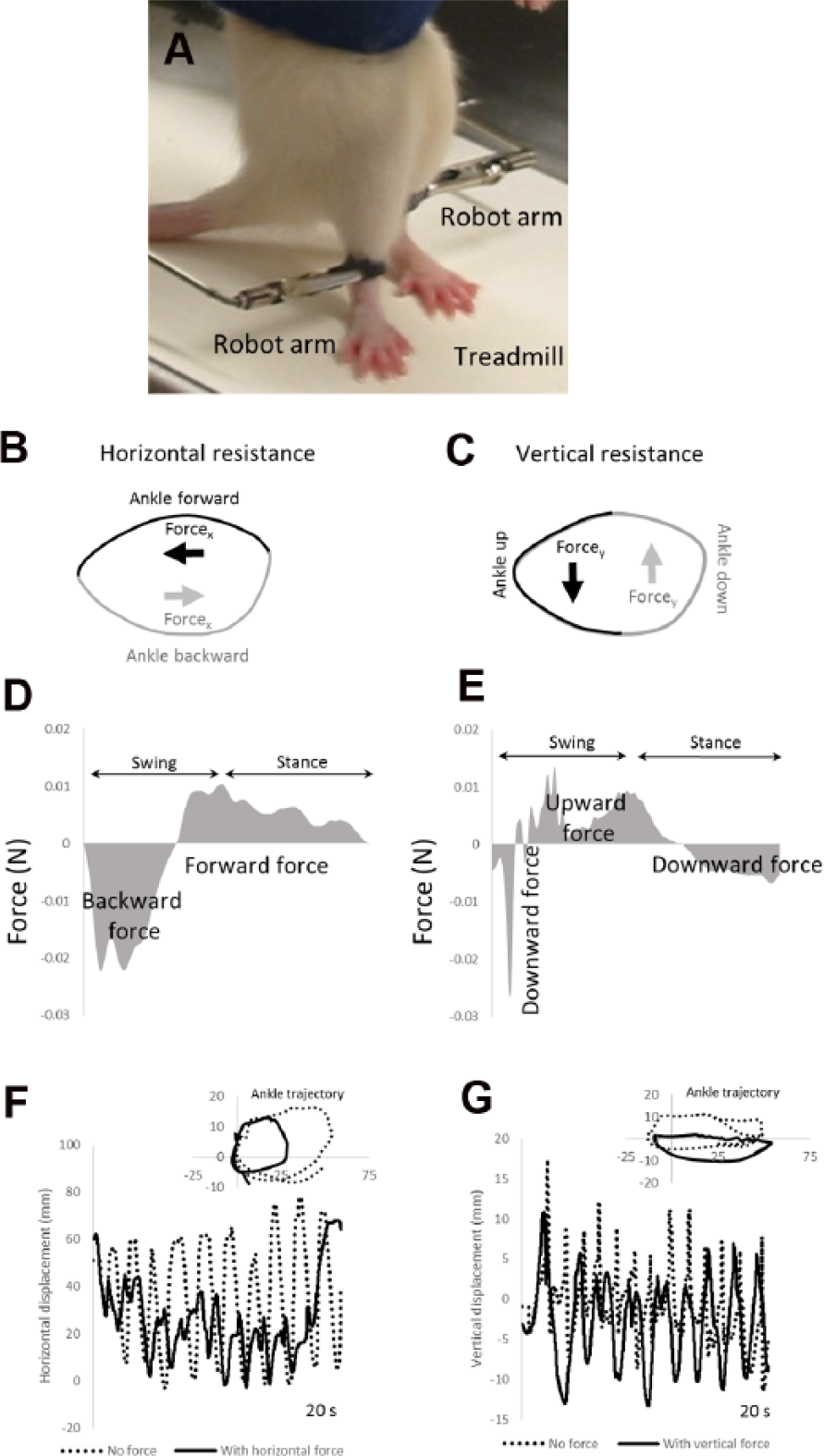
A robotic device applied resistive forces to oppose ankle movements during treadmill stepping. Picture of rat standing on a treadmill is shown in (A). Robot arms were attached to the ankles to apply forces while the rat stepped on a treadmill. Diagrams of the ankle trajectory during stepping with application of horizontal resistance (B) and vertical resistance (C). In (B), the ankle forward and backward movements (black and gray lines) are opposed by backward and forward forces, respectively (black and gray arrows). In (C), the ankle up and down movements (black and gray lines) are opposed by downward and upward forces, respectively (black and gray arrows). Applied force magnitudes are shown in (D) and (E), respectively, for 1 representative rat. In (D), backward force and forward force are show in gray. Robotic tracking of the ankle during stepping with and without the resistive forces is shown in (F) and (G). In (F), horizontal movements of the ankle during training with (black lines) and without (dashed lines) the horizontal resistance are shown in 1 representative rat over 20 s of stepping. In (G), vertical movements of the ankle with (black lines) and without (dashed lines) the vertical resistance are shown in another representative rat over 20 s of stepping. The insets in (F) and (G) show the ankle trajectory during 1 step cycle with (black lines) and without (dashed lines) resistance.
Figures 1D and 1E illustrate the relative magnitudes of the applied forces in representative rats over the course of a gait cycle. They illustrate how the applied forces resist the motion of the ankles. When the robot applied the resistance, the rats still generated step cycles, but the stepping amplitudes were smaller compared with stepping without resistance. The horizontal resistive force reduced the amplitudes of horizontal movement during training. This was mainly a result of a reduction in forward movement of the ankle during stepping (see “Ankle trajectory” and compare “No force” and “With horizontal force” in Figure 1F). In contrast, applying the vertical resistance primarily affected vertical movements of the ankle. The ankle trajectory was lowered with the vertical resistance compared with no resistance (see “Ankle trajectory” and compare “No force” and “With horizontal force” in Figure 1G).
Training With Resistance Did Not Restore a Normal Number of Step Cycles or Normal Cycle Period
We tested hind limb stepping ability before training began (week 0) and after 6 weeks of training (week 6) using a robotic device to record ankle movements. Figure 2 shows ankle movements recorded by the robotic device from representative rats in each group at week 6. All groups generated hind limb stepping (see “Horizontal” and “Vertical” movements and “Ankle trajectory” in Figure 2). However, there were occasional step failures that disrupted regular stepping movements (ie, the hind limbs dragged or withdrawal reflexes were elicited; see “d” and “w” in Figure 2). Compared with published data from uninjured rats walking in the robotic device, 20 none of the groups in the present study recovered a normal number of step cycles (29.3 ± 2.9 step cycles; see dashed lines in “Number of step cycles” in Figure 2) or a normal cycle period (1.47 ± 0.1 s; see dashed lines in “Cycle period” in Figure 2). In the ST group, the number of step cycles significantly increased, whereas cycle period significantly decreased from Week 0 to 6 and approached normal values (see “Step cycle” and “Cycle period,” week 0 vs week 6 in Figure 2B and for ST in Table 1). None of the other groups exhibited significant changes from Week 0 to 6. No significant between-group differences were found in the number of step cycles or cycle period performed at Week 0 or at Week 6 (see “Step cycles” and “Cycle period” in Table 1).
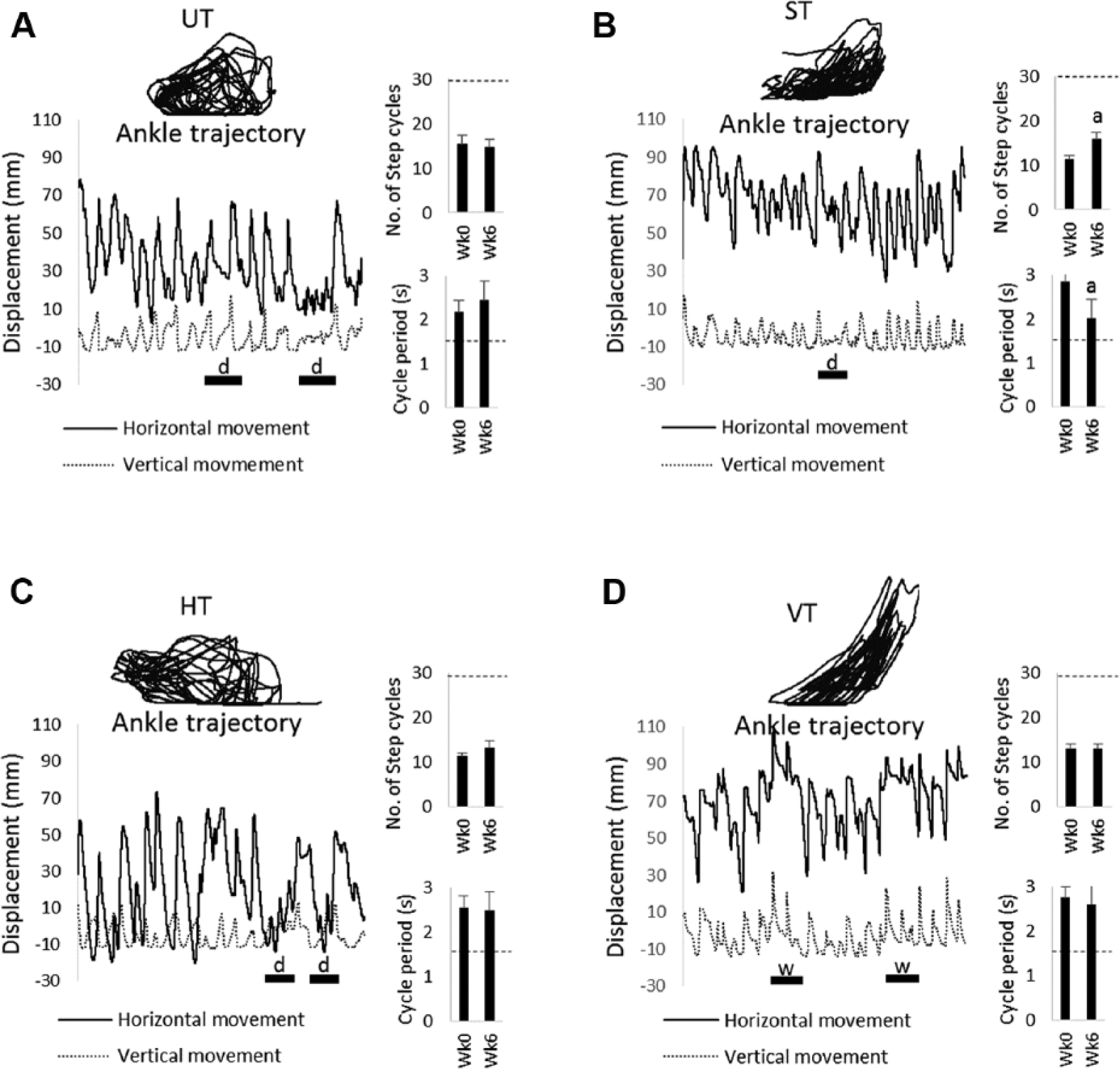
Examples of stepping movements performed by UT, ST, HT, and VT rats. Robotic data collected during stepping in representative rats from the UT (A), ST (B), HT (C), and VT (D) groups are shown. The ankle trajectory, horizontal (black line) and vertical (dashed line) movements of the ankle during a 30-s test of stepping (ie, no forces applied) are shown. All the data are from tests of stepping performed at week (Wk) 6. Stepping failures are denoted by the bars with “d” indicating a drag or a “w” indicating a withdrawal reflex. Bar plots of the step cycle (ie, number of steps detected during a 30-s test) and cycle period are shown next to the robotic data. An average ± SE is shown (UT, n = 8; ST, n = 9; HT, n = 9; VT, n = 8). The dashed line on the bar plots indicates the average value from normal (uninjured) rats.
Spatial, Temporal, and Velocity Measurements of the Step Cycle in UT, ST, HT, and VT Groups. a
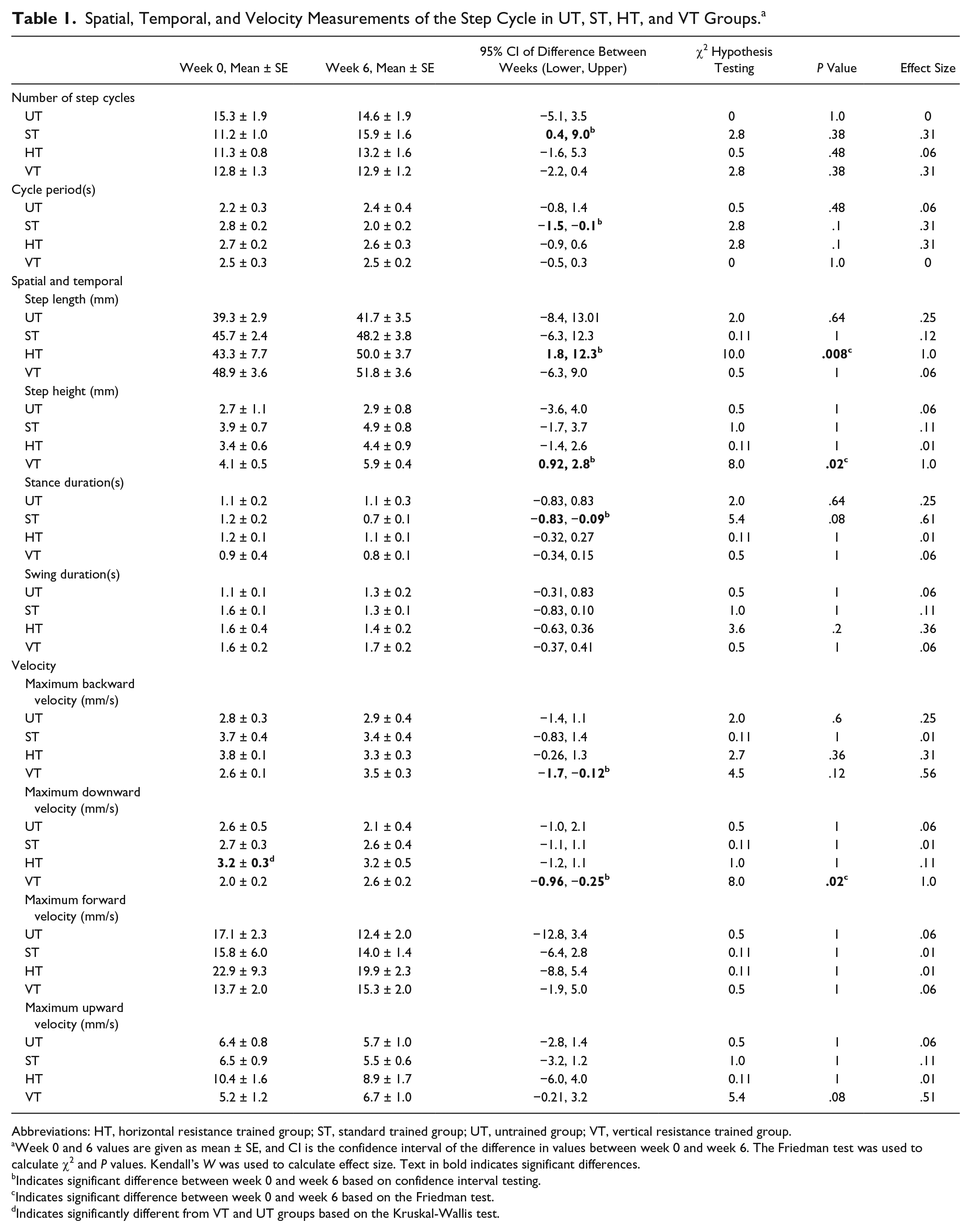
Abbreviations: HT, horizontal resistance trained group; ST, standard trained group; UT, untrained group; VT, vertical resistance trained group.
Week 0 and 6 values are given as mean ± SE, and CI is the confidence interval of the difference in values between week 0 and week 6. The Friedman test was used to calculate χ2 and P values. Kendall’s W was used to calculate effect size. Text in bold indicates significant differences.
Indicates significant difference between week 0 and week 6 based on confidence interval testing.
Indicates significant difference between week 0 and week 6 based on the Friedman test.
Indicates significantly different from VT and UT groups based on the Kruskal-Wallis test.
Training With Horizontal Resistance Increased Step Length, Whereas Training With Vertical Resistance Increased Step Height
We next analyzed the kinematic characteristics of the step cycles. Table 1 shows the results of the statistical analyses, and Figure 3A summarizes the significant changes that were found from week 0 to week 6. The average step length increased by 24% in the HT group (see “Step length” for HT in Figure 3A). This improvement was significant based on 2 different statistical analyses and was associated with a large effect size (see “Step Length” for HT, “Confidence interval of difference,” and P value in Table 1). None of the other groups exhibited significant changes in step length from week 0 to 6 (see “Step length” for UT, ST, and VT in Figure 3A). The effect of training with horizontal resistance was consistently observed in every rat in the HT group, but this was not the case in other groups (compare gray lines in Figures 4C to 4A, 4B, and 4D). Inspection of the average step cycle trajectory for the HT group shows that compared with week 0, there was greater forward swing of the ankle at week 6 (compare “Swing” in Figures 4E to 4F).
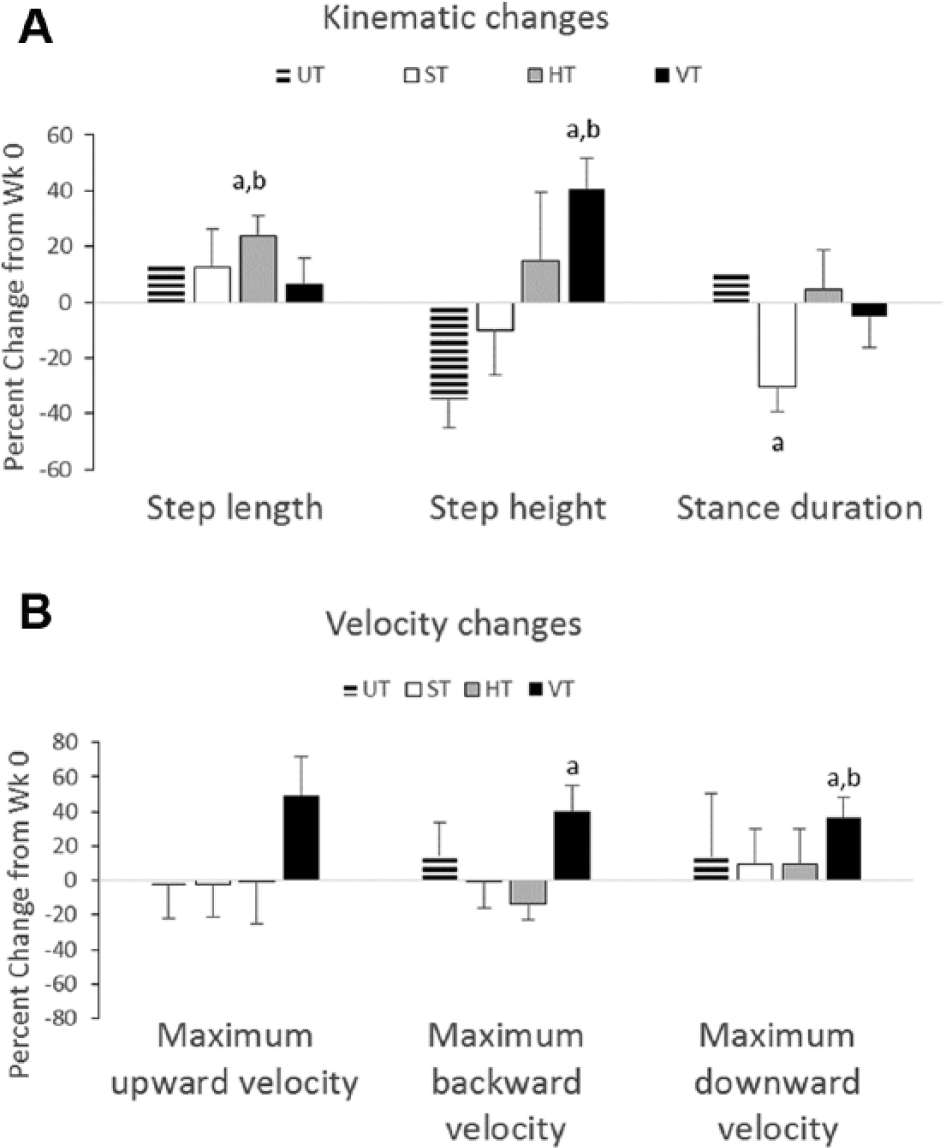
Changes in kinematic and velocity characteristics from week 0 to week 6. Bar plots showing the percentage change from week (Wk) 0 to Wk 6 for kinematic (A) and velocity (B) characteristics are shown for the UT, ST, HT, and VT groups. The data are average ± SE (UT, n = 8; ST, n = 9; HT, n = 9; VT, n = 8).
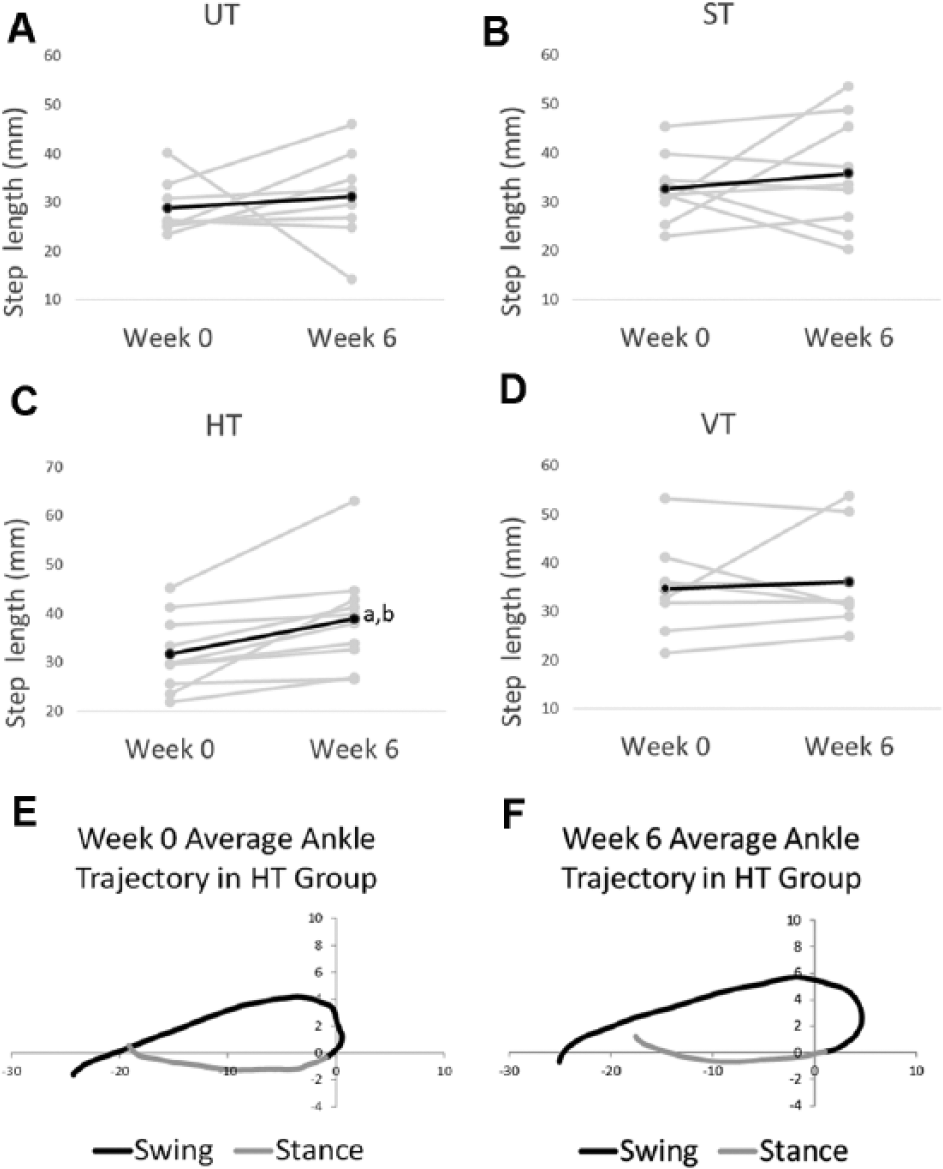
Step lengths in week 0 and week 6. In (A) to (D), step length is shown for the UT, ST, HT, and VT groups from week 0 to week 6. The dark line is the average, and the gray lines are data from individual rats. The average ankle trajectory from the HT group is shown for week (Wk) 0 (E) and Wk 6 (F). The black line indicates swing, and gray line indicates stance. The average was generated from all 9 HT rats from data collected during tests of stepping (ie, no forces were applied). Note, the scales are the same in (A) to (D) even though the absolute maximum and minimum values may be different.
A different effect on the step cycle characteristics was observed when training with vertical resistance. The height of the step increased by 41% in the VT group from week 0 to week 6 (see “Step height” for VT in Figure 3A). The increase was significant and was associated with a large effect size (see “Step height” for VT, “Confidence interval of difference,” and P value in Table 1). None of the other groups exhibited significant changes in step height (see “Step height” for UT, ST, or VT in Table 1). The increased step height occurred in every rat in the VT group, whereas greater variability was observed in the other groups (compare gray lines in Figures 5D to 5A, 5B, and 5C). Inspection of the average step cycle trajectory for the VT group shows that compared with week 0, there was greater lift of the ankle during swing at week 6 (compare “Swing” in Figures 5E and 5F).
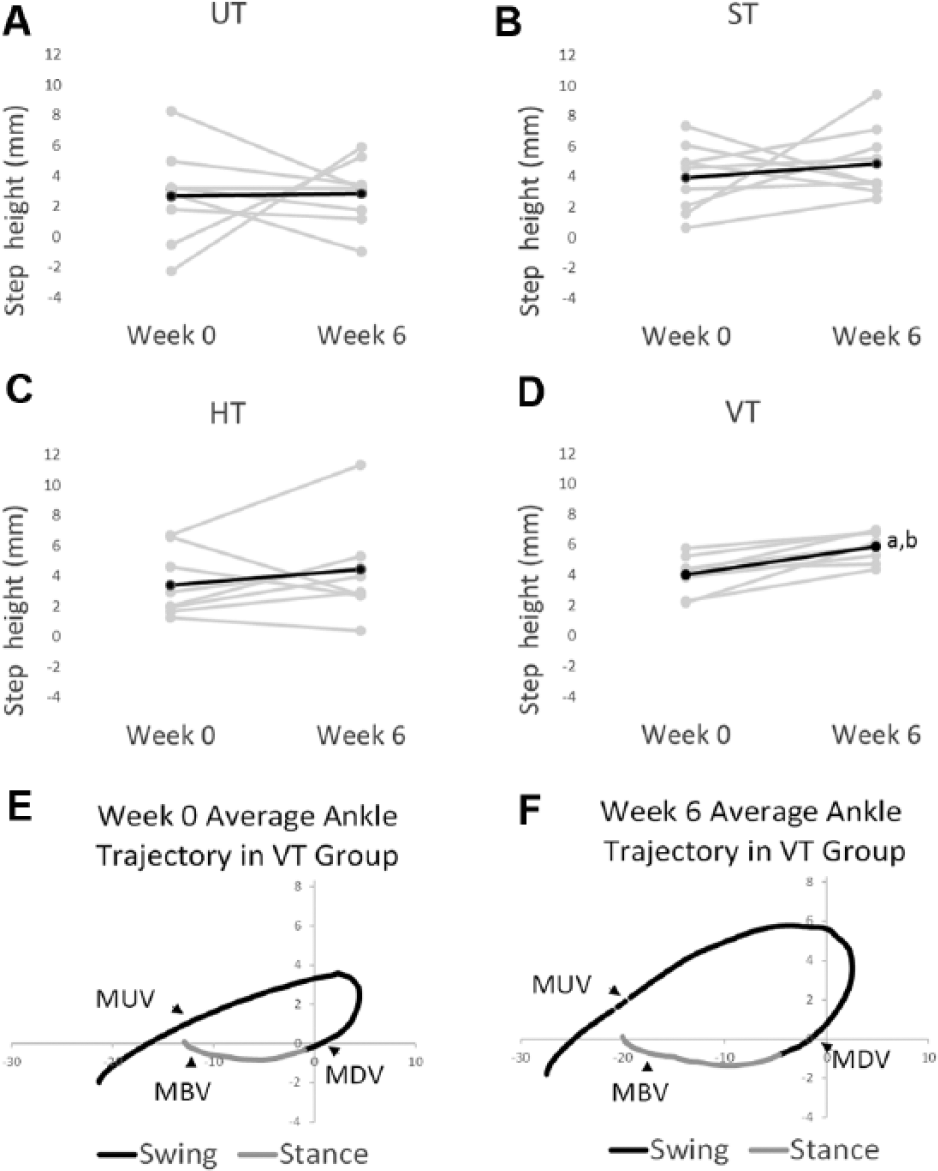
Step height at week 0 and week 6. In (A) to (D), step height is shown for the UT, ST, HT, and VT groups from week 0 to week 6. The dark line is the average, and the gray lines are data from individual rats. The average ankle trajectory from the VT group is shown for week (Wk) 0 (E) and Wk 6 (F). Black line indicates swing, and gray line indicates stance. The average was generated from all 8 VT rats from data collected during tests of stepping (ie, no forces were applied).
In the ST group, stance duration shortened by 30% from week 0 to week 6 (see “Stance duration” for ST in Figure 3A). No significant difference between the groups was observed at week 0 or week 6 (see “Step length,” “Step height,” “Stance duration,” and “Swing duration” at week 0 and week 6 in Table 1).
Only Training With Vertical Resistive Force Increased Movement Velocity
We also analyzed movement velocities, and the changes from week 0 to week 6 are summarized in Figure 3B. The largest changes (ie, 36%-50% increases) in the velocity were found in the VT group (see VT in Figure 3B). Maximum upward velocity occurred during early swing, whereas maximum downward velocity occurred just prior to paw contact; maximum backward velocity occurred in late stance (see MUV, MDV, and MBV in Figures 5E and 5F). The increases in maximum downward velocity and maximum backward velocity were significant, and large effect sizes were associated with these changes (see “Maximum backward velocity” and “Maximum downward velocity” for VT in Table 1). The increase in maximum upward velocity approached significance (see “Maximum upward velocity” for VT, P value in Table 1). None of the other groups exhibited significant changes in velocity (see “Velocity” for UT, ST, and HT in Table 1). No differences in velocity were found between groups with the exception of maximum downward velocity, which was significantly greater at week 0 in the HT group compared with the VT and UT groups (see “Maximum downward velocity” for HT in Table 1).
Synaptophysin Expression in the Ventral Horn Was High in the UT and HT Groups
Immunohistochemical experiments were performed to examine the synaptic marker, synaptophysin. Previous studies have shown that treadmill training in SCI rats increased synaptophysin expression in the lumbar spinal cord ventral horn and specifically around motor neurons.11,12 Intense synaptophysin labeling was observed in the ventral horn of the lumbar spinal cord in the HT and UT groups, whereas weaker labeling was observed in the ST and VT groups (Figures 6A-6D). A significant difference in synaptophysin labeling in the ventral horn was found between the groups (χ2 = 17.406; P = .001). Based on post hoc tests, synaptophysin labeling was significantly greater in the HT and UT groups compared with the ST and VT groups (compare UT, HT with ST, VT in Figure 6I).
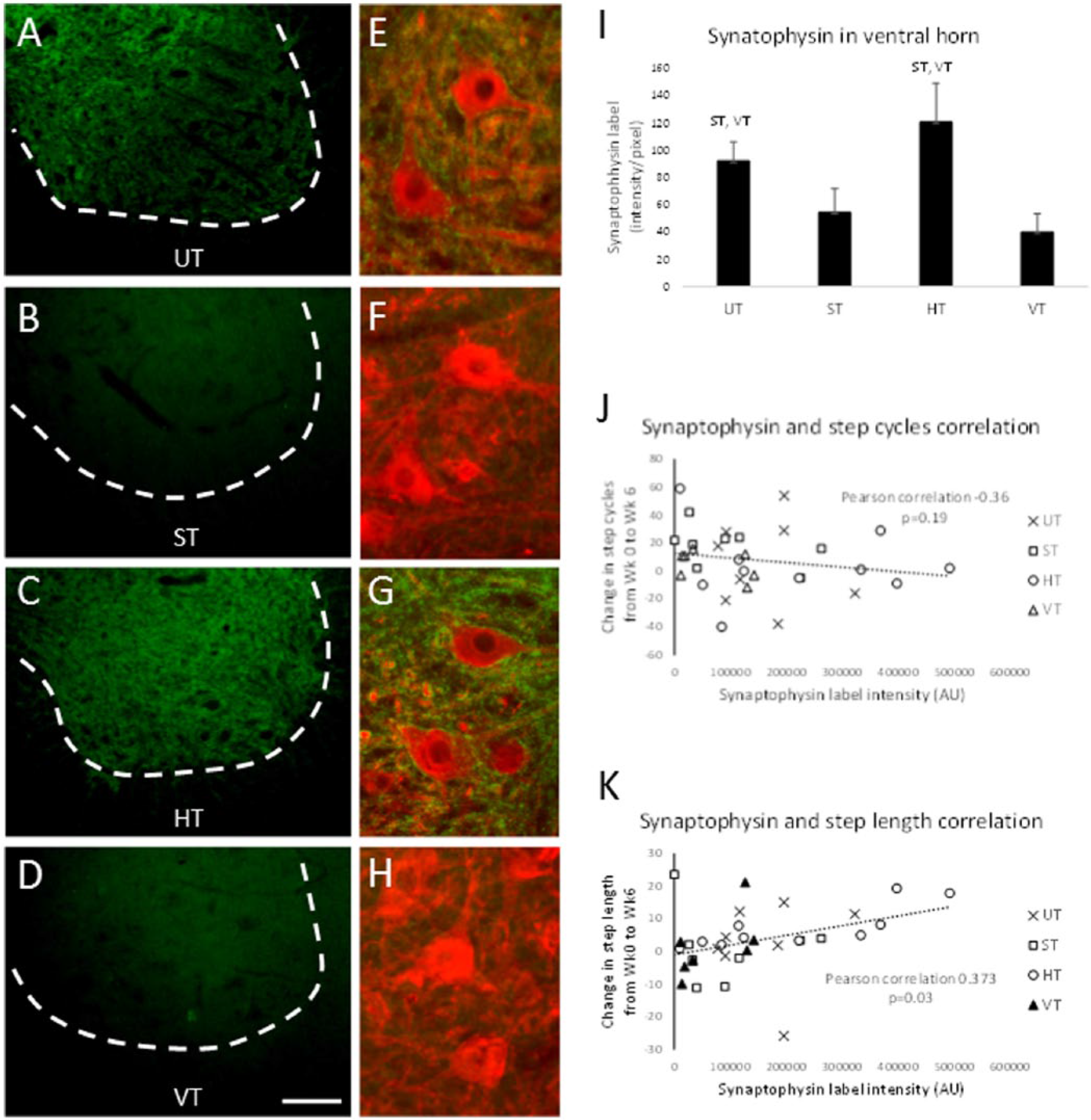
Synaptophysin expression in the spinal cord. Synaptophysin expression in the ventral horn of the lumbar spinal cord is shown for representative rats from the UT (A), ST (B), HT (C), and VT (D) groups. Higher magnification of synaptophysin labeling (green) and HSP27-labled motor neurons (red) is shown in (E) to (H) for the 4 groups. The calibration bar in (D) is 100 µm for (A) to (D) and 20 µm for (E) to (H). In (I), the intensity of synaptophysin expression in the ventral horn is compared between the UT, ST, HT, and VT groups. Mean ± SE is shown (UT, n = 8; ST, n = 9; HT n = 9; VT n = 8). ST, VT above the bars mean significantly different from the ST and VT groups respectively; P < .05. Scatterplot of synaptophysin intensity around motor neurons versus change in step cycle (J) and versus change in step length (K) is shown for all rats in each group (see legend). The dashed line shows the linear regression. The Pearson correlation and P value were based on data from all groups combined.
Synaptophysin Expression Around Motor Neurons Was Correlated With Step Length but Not With Step Cycle Number
In the HT and UT groups, intense synaptophysin labeling was observed around HSP27-labeled motor neurons, and in some cases, it formed an outline around the perimeter of the cell (see Figures 6E and 6G). This staining pattern was not observed in the ST and VT groups (see Figures 6F and 6H). The highest levels of synaptophysin expression around motor neurons were found in the HT and UT groups; however, the differences between groups did not reach statistical significance (χ2 = 6.117; P = .11).
We analyzed the relationship between synaptophysin expression and the stepping parameters step cycle number, cycle period, step length, and step height. Only step length was positively correlated with synaptophysin expression (Figure 6K). No significant correlations were found between synaptophysin expression and step cycle (Figure 6J), cycle period, or step height (data not shown).
White matter sparing was assessed in a subset of rats from each group (n = 3 per group). The mean percentages of white matter spared were 17.0% ± 4.2%, 19.4% ± 7.8%, 16.4% ± 2.0%, and 17.3% ± 5.2% in the UT, ST, HT, and VT groups, respectively.
Discussion
Training With Resistive Forces Augmented the Effects of BWSTT
Recent studies have explored using robotic resistance to augment BWSTT and have reported improvements in walking characteristics and performance of skilled locomotor tasks in those with SCI.3-5 Although these studies provided evidence for a beneficial effect of robotic resistance, in order to assess the value of adding robotic resistance to BWSTT, it was essential to determine whether the robotic resistance provided additional gains over BWSTT. In the present study, we used a rodent model to study the effects of BWSTT with robotic resistive forces. The design of the present study included 2 control groups—a BWSTT only group and an untrained group—in order to test the effects of training with resistive forces. Standard BWSTT alone changed the number of step cycles and cycle period values toward normal and shortened stance duration. These findings were consistent with previous results. 20 However, by adding horizontal resistance during training, there was an increase in step length, whereas adding vertical resistance increased step height and movement velocity. Importantly, these adaptations were not observed in rats that received standard training, suggesting that these changes were a result of the addition of the resistive forces.
Use of resistive forces to counter forward swing has consistently been shown to increase step length,3,4,9 and the observed changes with horizontal resistance were consistent with these findings. A unique aspect of the present study was that the effects of vertical resistive forces were studied. The effects of purely vertical resistive forces during BWSTT have not yet been examined in human patients with SCI or in animal models of SCI. We found that vertical resistance had a greater effect than horizontal resistance. Training with vertical resistance improved lift and movement velocity during swing. In addition, adaptations during stance (ie, faster backward velocity) were observed that have not yet been reported in other studies of resistive force training. It is not clear why training with the vertical resistance produced a greater number of adaptations than training with horizontal resistance. We attribute these adaptations to the rats actively generating movements against the vertical resistive forces. Recent findings in both human and animal studies suggested that locomotor training that encouraged active participation led to improved limb movements during stepping.24-26 The success of training with vertical resistance may be attributed to an increased level of activity within the nervous system. For example, the VT group was challenged to move against downward and upward forces that were applied throughout stance and swing (see Figure 1E). In contrast, the HT group primarily generated movements against a backward force during swing only (see Figure 1D). During stance, the hind limb was passively moved backward by the treadmill belt, and adaptations to horizontal forces during stance would not be expected. 4
Normal step cycle number and cycle period were determined from previous data collected from uninjured rats in the robotic device. Normal values of these parameters were not restored by training with resistance. Given a recent review of training studies in SCI animals, 1 a longer duration of training or shorter delay in the start of training may be necessary for optimal results, and these factors may have played a role in the rats not recovering normal stepping characteristics. However, we followed previous successful training protocols, making it less likely that the training parameters were insufficient. One reason for the lack of improvement may be that although the resistive forces enhanced step trajectory size, they may have disrupted the coordination between the 2 hind limbs, leading to frequent step failures. Unlike previous studies, we challenged the rats to step while resistive forces were simultaneously applied to both hind limbs. For example, the VT rats had to adapt to downward and upward forces in the swing limb while also adapting to an upward and downward force in the stance limb. Any adaptation in the swing limb (ie, faster upward and downward velocities) would have to be accompanied by a parallel adjustment in the contralateral limb. If not, intralimb coordination was altered and step failures were likely. Previous studies have applied resistive forces during swing only, and this made it easier for adjustments to occur in the nonperturbed limb. Interestingly, in the VT rats, there was an unexpected increase in maximum backward velocity observed during stance, and this may have been a necessary contralateral adjustment to the faster swing velocities. It is likely that the ability of the rats to regain normal step cycle numbers was hampered by having to make ipsilateral and contralateral adjustments to the complex perturbation. Another consideration is that a lack of trunk control contributed to our findings. Udoekwere et al 27 recently reported improved stepping in SCI rats following treadmill training that included the robotic application of elastic forces to the pelvis. 27 These forces help support and stabilize trunk movements, and with this training, even non–weight supporting SCI rats regained the ability to perform weight-supported quadrupedal stepping. We did not directly apply forces to the trunk, but given the recent evidence, integrating robotic forces on the trunk and hind limb may be beneficial in the recovery of normal stepping patterns.
Ultimately, the success of any gait training intervention will be determined by the impact on functional walking behaviors. In the present study, we did not assess whether treadmill training with resistive forces improved overground walking. There is evidence from human studies that treadmill walking following training with resistive forces does translate to better overground walking. The stride length and speed during overground walking improved in individuals with incomplete SCI following BWSTT with resistance. 7 Other recent findings indicate that resistive force training also improved the performance of skilled locomotor tasks that comprise the Spinal Cord Injury Functional Ambulation Profile (eg, stair walking, obstacle crossing, weighted walking). 5 In SCI rats, however, there is recent evidence that the beneficial effects of treadmill training were dependent on whether bipedal or quadrupedal walking occurred. Training SCI rats to perform quadrupedal treadmill locomotion improved overground walking, but bipedal treadmill locomotor training did not have the same beneficial effect.28,29 These data support the idea of a task-specific effect of locomotor training in SCI rats. 30 Given these recent findings, it seems unlikely that only training the hind limbs with resistive forces would improve quadrupedal locomotion, in part because of the lack of forelimb participation. It would be advantageous to apply resistive forces during quadrupedal overground walking if an impact is to be made on the animal’s natural form of locomotion. Application of resistive forces during quadrupedal locomotion is not feasible with the current configuration of our robotic system. This may be a goal for future studies, given the recent advances in devices for training weight-supported, quadrupedal overground walking in rats.31,32
Are Spinal Synapses Strengthened by Training With Resistive Forces?
We hypothesized that resistive force training would enhance synaptic connections within the lumbar spinal circuits. This hypothesis was based on current ideas about robotic training and activity-dependent plasticity. 19 Training stimulates activity within neural circuits that control locomotion, and with repeated training sessions, synaptic connections within the circuitry are reinforced. Resistive forces should increase activity and, therefore, further enhance plasticity. A significant correlation was found between synaptophysin expression and step length. Inspection of these data showed that the greatest expression was observed in the rats that received horizontal resistance training and, thus, performed the longest steps (see Figure 6K). This finding suggested that the effect of resistive training on step length may be associated with increased synaptic connections within the ventral horn.
However, the synaptophysin expression found in the rats trained with vertical resistance was not consistent with our hypothesis. These rats had the lowest levels of synaptophysin expression despite being trained with resistive forces that were comparable in magnitude to the forces imposed on the HT group (see Figures 1D and 1E). Given the more complex nature of the vertical forces, it is reasonable to expect that the impact on connections within the spinal cord would be different from the effect associated with horizontal resistance. Compared with horizontal resistance training, vertical resistance affected multidirectional movement properties throughout the step cycle. Generating stepping against the vertical resistance required modulating the activities across different extensor and flexor motor pools, likely exciting some and inhibiting others during each step cycle. The synaptophysin expression observed in the vertical trained rats may reflect these types of complex activity patterns. Other findings also indicate that a differential modulation of synapses occurs following training. In spinally transected rats, step training increased GABAergic terminals around ankle flexor motor pools but had the opposite effect on ankle extensor motor pools. 33 In addition, it appears that inhibitory and excitatory inputs to motor pools were differentially modulated, and importantly, whether synapses were increased or decreased depended on the type of activity patterns that are imposed during training.15,34,35 Such an effect could account for the differences between horizontal and vertical resistance training.
It is important to consider that the effects of resistive training may include other types of adaptations that were not examined here. Improvement in muscle properties were observed after just 1 week of treadmill training in spinally contused rats, 36 making it likely that several weeks of training with resistive forces would also produce gains in hind limb muscle that facilitated stepping. Likewise, treadmill training has been reported to improve pain measures in spinally contused rats, 37 and this may be a result of a reduction in afferent fibers entering the dorsal horn. 38 It is possible that applying resistive forces could initially heighten activity in afferent pathways. Whether long-term training with resistive forces would eventually reduce pain-related sensory activity remains to be determined.
The Effects of Training on Spinal Synapses Following a Contusion Injury
Several studies, including our own, have used immunohistochemical analyses of synaptophysin expression to show that treadmill training increased the amount of synaptic inputs onto motor neurons, and this was associated with improved ability to perform stepping.10-12,39 The present findings regarding synaptophysin expression were not consistent with these previous results. Specifically, untrained rats exhibited a higher level of synaptophysin expression than the standard trained rats. One important difference was that unlike previous studies that were performed in rats that had a complete spinal cord transection, a contusion injury was used in the present study. There is emerging evidence that plasticity induced by treadmill training may be different following a spinal cord contusion injury compared with a spinal cord transection injury. For example, Singh et al 38 recently showed in the spinally contused rat that treadmill training reduced primary afferent sprouting in the lumbar spinal cord. A unique effect of training in the contused rat may be to eliminate aberrant pathways that contribute to maladaptive plasticity and, thus, negatively affect the function of spinal circuits controlling walking. 40 Our findings of a reduced synaptophysin expression in the ST group is consistent with this idea.
In addition, after a contusion injury, spared descending axons contribute to locomotor recovery. 41 If enough axons are spared, it may not be necessary to add more synapses, but instead, treadmill training in the spinally contused rat may potentiate the activity in existing synapses. Recent findings are consistent with this idea. Training contused rats did not increase the presence of serotonergic terminals in the lumbar spinal cord despite an increase in synaptophysin expression. 42 Another recent study found that training did not change the amount of spared white matter at or near the lesion site of contused rats. 37 Our interpretation of these data is that a different synaptic environment occurs within the spinal circuitry after a contusion injury compared with a transection injury. The ramification is that the effect of training on spinal plasticity, that is, increasing synaptic density or enhancing existing connections, is dependent on the physiological state of the spinal circuitry after the injury. 43
An alternative explanation for the observed differences in synaptophysin expression was that the extent of spinal cord lesions differed between the groups. A less severe contusion injury could have resulted in greater sparing and, thus, higher synaptophysin expression in the UT and HT groups compared with the ST and VT groups. We found that spared white matter was not different between subsets of the groups. However, based on the locomotor performance at baseline testing, faster movement velocities were found in the HT group at week 0, which could reflect a greater sparing of descending fibers. Although it cannot be ruled out, this scenario seems unlikely given that the rest of the data (eg, step cycle number, cycle period, kinematic parameters) indicate that recovery was similar between the groups at baseline testing.
Conclusions/Implications
We used a rodent model of SCI to demonstrate how resistive forces augmented the effects of BWSTT. The rodent model facilitates systematic, controlled studies. Moreover, it has successfully been used as a small-scale testbed for robotic BWSTT algorithms. 19 The present findings, therefore, have implications for the use of robotic devices in locomotor rehabilitation, in particular, for devices that apply forces at a single point on the lower limb.6,44 Vertical resistance, in particular, may be useful for targeting specific deficits such as foot clearance and reduced movement velocity that are known to occur in high-functioning SCI individuals.45,46 Based on the present findings, the success of this strategy will require careful control not only of the magnitude, but also the direction (ie, horizontal vs vertical) and timing of the resistive forces.
Footnotes
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by NIH Grant R15NS082711.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
