Abstract
Background. Movement is described in terms of task-related end point characteristics in external space and movement quality (joint rotations in body space). Assessment of upper-limb (UL) movement quality can assist therapists in designing effective treatment approaches for retraining lost motor elements and provide more detailed measurements of UL motor improvements over time. Objective. To determine the extent to which current activity level outcome measures used in neurological practice assess UL movement quality. Methods. Outcome measures assessing arm/hand function at the International Classification of Function activity level recommended by neurological clinical practice guidelines were reviewed. Measures assessing the UL as part of a general mobility assessment, those strictly evaluating body function/structure or participation, and paediatric measures were excluded. Results. In all, 15 activity level outcome measures were identified; 9 measures assess how movement is performed by measuring either end point characteristics or movement quality. However, except for the Reaching Performance Scale for Stroke and the Motor Evaluation Scale for Upper Extremity in Stroke Patients, these measures only account for deficits indirectly by giving a partial score if movements are slower or if the person experiences difficulties. Six outcome measures neither assess any parameters related to movement quality, nor distinguish between improvements resulting from motor compensation or recovery of desired movement strategies. Conclusion. Current activity measures may not distinguish recovery from compensation and adequately track changes in movement quality over time. Movement quality may be incorporated into clinical assessment using observational kinematics with or without low-cost motion tracking technology.
Keywords
Introduction
Neurological disorders, a leading cause of disability worldwide 1 can lead to physical, psychological, and cognitive impairments affecting activity and social participation. 2 One of the most common impairments in individuals with neurological disorders is arm paresis, characterized by muscle weakness, changed muscle tone, decreased sensation, and impaired voluntary movement control, 3 resulting in slow, imprecise, and uncoordinated movement.3-5 The use of compensatory movement patterns, especially excessive trunk displacement and shoulder elevation and abduction are also common.4,6 Following stroke, the incidence of upper-limb (UL) impairment is estimated at ~80%,7,8 with residual impairments persisting into the chronic stage in more than 65% of cases despite intensive and prolonged rehabilitation. 9 Cervical spinal cord lesions, the most common (75%) site of spinal cord trauma, result in tetraplegia affecting arm/hand sensorimotor functions.10,11 UL paresis affects ~30% of individuals with traumatic brain injury 12 and 60% of individuals in the first year following the diagnosis of multiple sclerosis. 13 UL impairment can lead to limitations in activities of daily living (ADLs), such as eating, dressing, using the phone or computer, and handling medications or shopping as well as decreased social participation and quality of life.2,14,15
Recent reviews of the effectiveness of physical interventions for UL rehabilitation of individuals with stroke concur that current interventions may not be tapping into the full potential for motor recovery.16-18 The consensus view is that therapies aimed at remediating specific deficits may be more successful than more general approaches such as repetition, varied practice, and feedback only based on task success.16,19 These results may be attributed to the limited potential of current interventions to effect change and/or inadequacies of clinical outcome measures to distinguish between levels of recovery to identify improvement. For example, even if an intervention results in a significant increase in joint range, the improvement may not be noted if the outcome measure only quantifies task accomplishment not involving that range.
To identify true behavioral motor recovery (as opposed to neuronal recovery), measures should be able to distinguish between restitution of premorbid movement patterns and the use of alternative (compensatory) movement patterns during task accomplishment (Table 1). 20 Despite this important distinction, terminology describing how movement changes is inconsistent. Movement can be classified at 2 levels of description that distinguish between end point movement in external space in which variables such as trajectory speed, precision, and straightness can be quantified (ie, end point characteristics), and movements in body space, in which ranges of individual joints and segments (ie, trunk), spatial and temporal interjoint coordination, and muscle activation patterns can be measured (ie, movement quality variables). 20 End point characteristics may improve either by the use of compensations (eg, incorporation of trunk movement to assist reaching extent) or by improvement of movements in body space. Thus, only movements in body space (ie, movement quality variables) can distinguish whether behavioral recovery or compensation has occurred.
Definition of Behavioral Recovery and Compensation According to the International Classification of Functioning Model. a
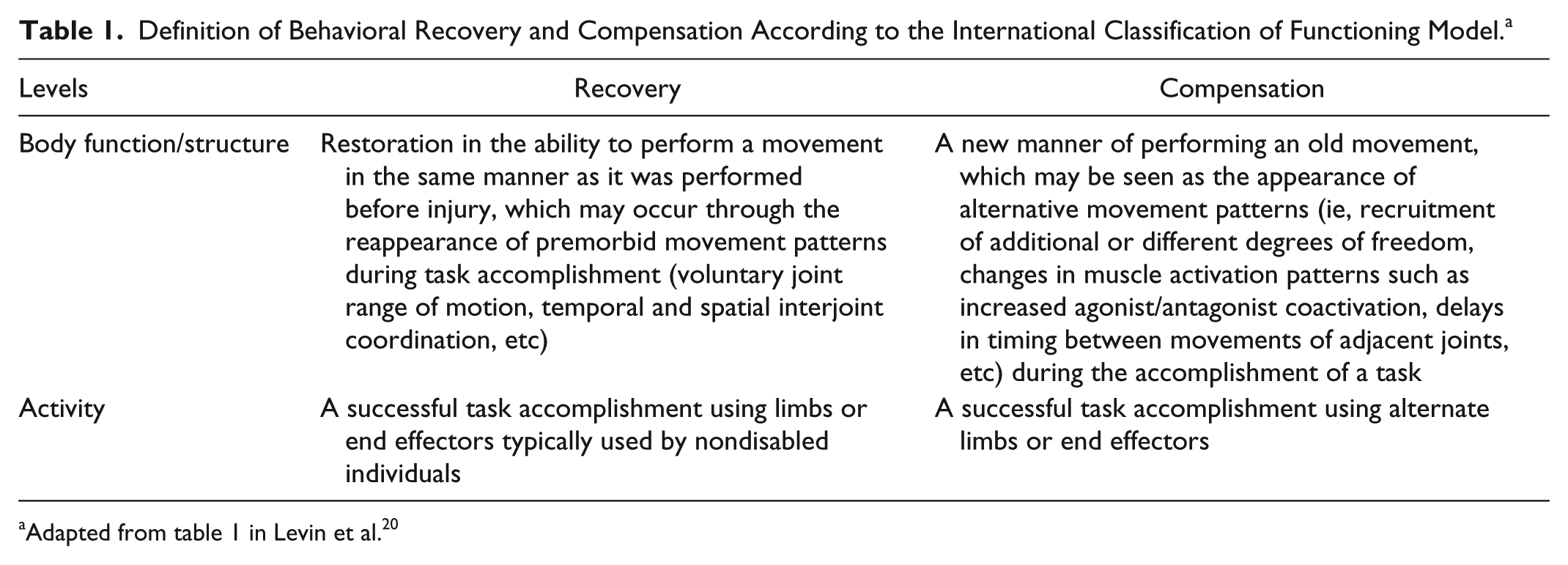
Outcome measures that assess movement quality may provide more information about how a person accomplishes a functional task, the compensatory strategies used, missing task elements, and other specific deficits. This information can be used to guide clinical decisions about personalizing treatment to optimize motor recovery, such as how to retrain lost motor skills that limit ADL accomplishment while focusing on decreasing maladaptive motor compensations. 21 In addition, movement quality assessment may help better capture small changes in movement over time, which can be useful in determining the effectiveness of a specific intervention. However, the ability of current outcome measures at the International Classification of Function (ICF) activity level to capture UL movement quality has been questioned.20-23
Most UL outcome measures used in neurological practice most often quantify the degree of task completion on an ordinal scale and/or by time to perform a task without considering movement quality. 24 Although movement quality may be assessed by recording movement kinematics, kinematic analysis is not widely used in clinical settings because of the high cost and complexity of recording equipment as well as lack of expertise and time.23,25 Thus, if outcome assessment is to be continued to be done using clinical scales, it is necessary to determine the extent to which UL movement quality is assessed by current measures at the ICF activity level. For the purpose of this article, the terms outcome measure, assessment, clinical scale, and measure are used interchangeably to indicate a method used to capture data in a standardized manner.
Selection of Outcome Measures Used in Neurological Practice
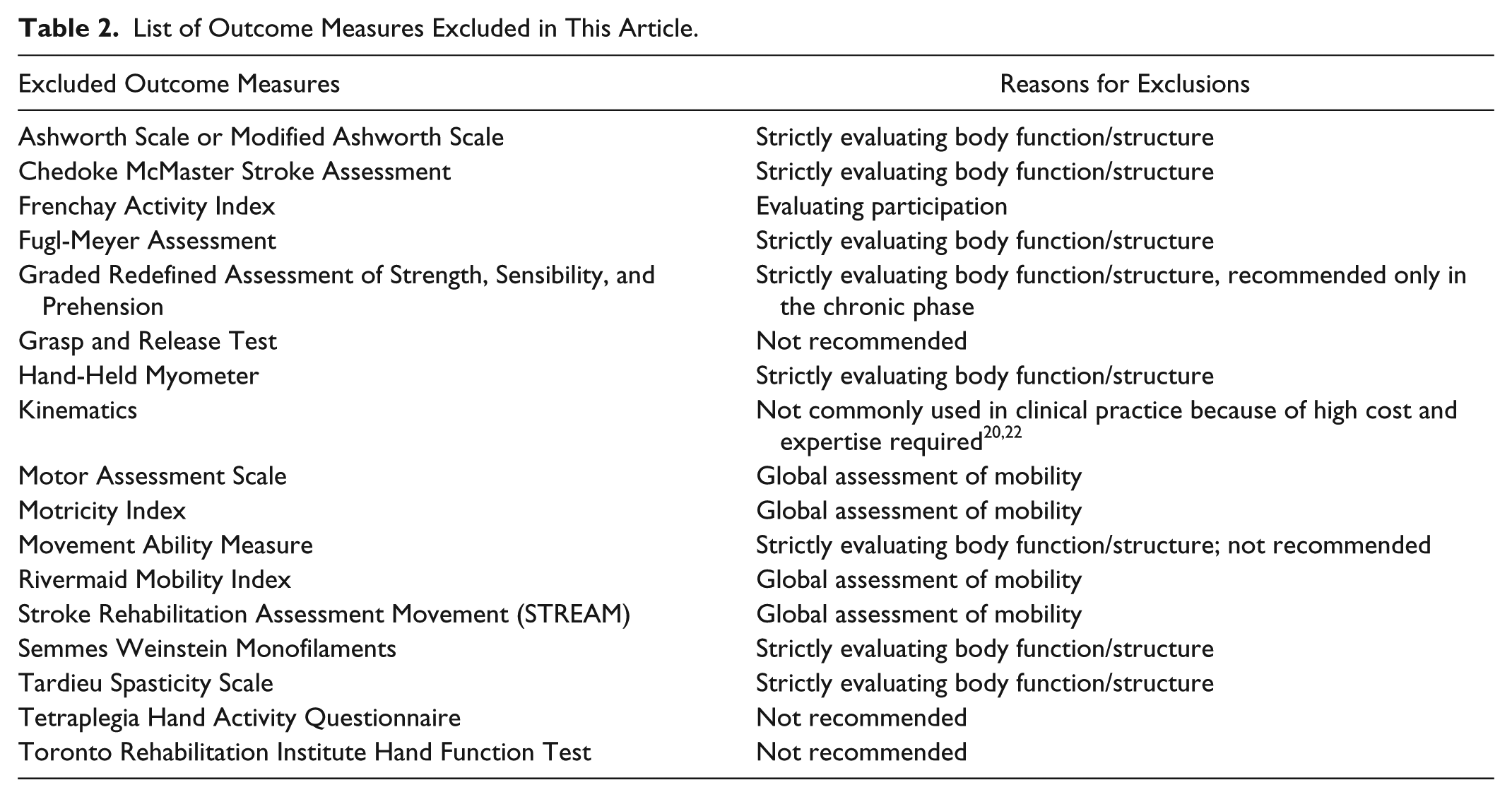
There is no consensus as to which outcome measures best assess UL movement quality. Therefore, measures recommended by the Evidence-based Review of Stroke Rehabilitation 26 and the Academy of Neurologic Physical Therapy Outcome Measures for multiple sclerosis, 27 spinal cord injury, 28 stroke, 29 and traumatic brain injury 30 were extracted. The Canadian Medical Association InfoBase for Clinical Practice Guidelines and the Agency for Healthcare Research Quality’s National Guideline Clearinghouse databases were also searched using the key words multiple sclerosis, spinal cord injury, stroke, and traumatic brain injury to identify additional clinical practice guidelines recommending UL outcome measures, but no additional guidelines were identified. Measures claiming to assess UL movement quality during activities and reported to be valid and reliable in a neurological population were also included. The inclusion criteria were the following: measures of arm or hand function at the ICF activity level and developed/used in adult neurological populations. Outcome measures strictly evaluating body function/structure or participation levels, those only used with pediatric populations or only assessing UL function as part of a global assessment of mobility were excluded (Table 2). Included measures are listed in Table 3. This review was limited to measures at the ICF activity level because they are often used to determine if rehabilitation interventions result in changes important in daily life. To assess the effectiveness of interventions, however, it is desirable to identify if changes occur as a result of compensation or recovery at this level. Thus, we included measures with items that assess the activity level, even though some items in the scale also assessed other levels.
List of Outcome Measures Excluded in This Article.
Outcome Measures Used to Assess UL Function at the Activity Level in Neurological Practice.
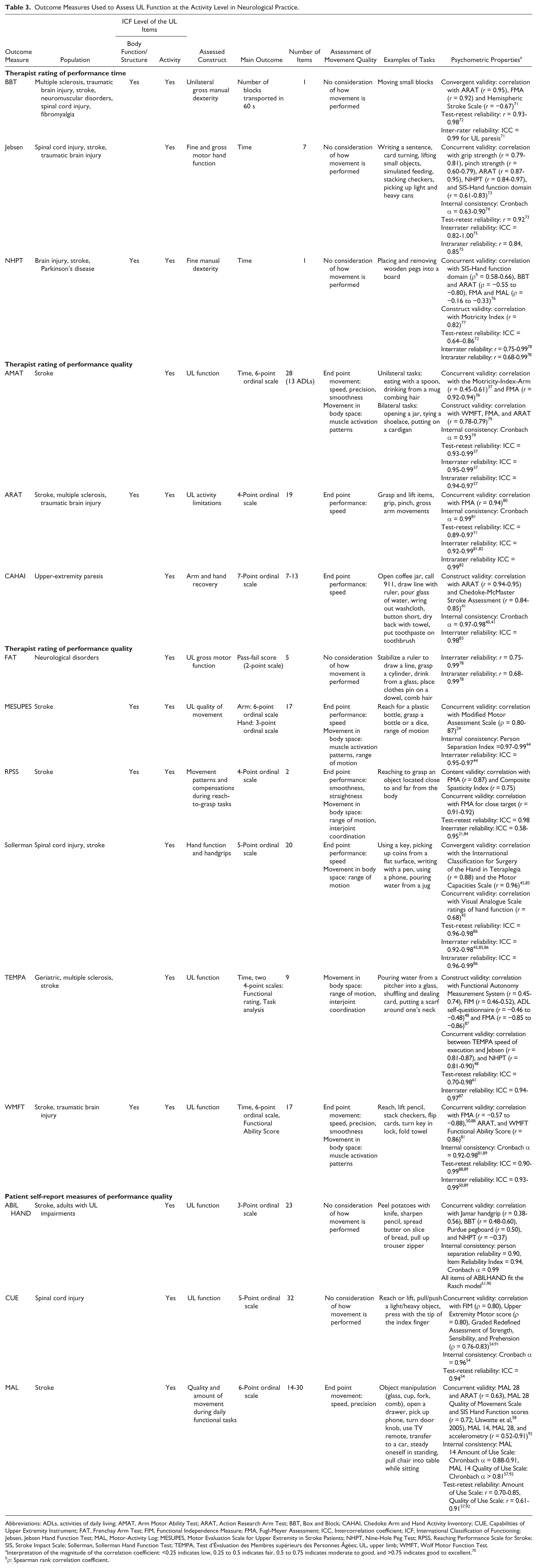
Abbreviations: ADLs, activities of daily living; AMAT, Arm Motor Ability Test; ARAT, Action Research Arm Test; BBT, Box and Block; CAHAI, Chedoke Arm and Hand Activity Inventory; CUE, Capabilities of Upper Extremity Instrument; FAT, Frenchay Arm Test; FIM, Functional Independence Measure; FMA, Fugl-Meyer Assessment; ICC, Intercorrelation coefficient; ICF, International Classification of Functioning; Jebsen, Jebsen Hand Function Test; MAL, Motor-Activity Log; MESUPES, Motor Evaluation Scale for Upper Extremity in Stroke Patients; NHPT, Nine-Hole Peg Test; RPSS, Reaching Performance Scale for Stroke; SIS, Stroke Impact Scale; Sollerman, Sollerman Hand Function Test; TEMPA, Test d’Évaluation des Membres supérieurs des Personnes Âgées; UL, upper limb; WMFT, Wolf Motor Function Test.
Interpretation of the magnitude of the correlation coefficient: <0.25 indicates low, 0.25 to 0.5 indicates fair, 0.5 to 0.75 indicates moderate to good, and >0.75 indicates good to excellent. 70
ρ: Spearman rank correlation coefficient.
Assessment of Activity Level Outcome Measures
Outcome measures were divided into 3 categories based on their metrics to facilitate comparisons between scales: therapist ratings based on time, therapist ratings based on a numerical scale, and patient self-report measures.
Therapist Ratings Based on Time
Box and Blocks (BBT)
The BBT 31 measures unilateral gross manual dexterity and is composed of a wooden box divided into 2 compartments and 150 wooden cubes. The participant grasps a cube, transports it over a partition, and releases it in the opposite compartment. The absolute number of cubes transported by the affected arm from one side of the box to the other in 1 minute or the number relative to the nonaffected arm is measured. The total score is compared with established norms. 31 The BBT has substantial floor effects in individuals without sufficient arm movement, strength, and grip function to transport blocks. 26
Jebsen Hand Function Test (Jebsen)
The Jebsen uses 7 timed, unimanual tasks to assess fine and gross motor hand function. 32 Each item is scored as the time for task completion. For each task, times are compared with age- and gender-based norms. 32
Nine-Hole Peg Test (NHPT)
The NHPT is 1 of 3 components of the Multiple Sclerosis Functional Composite score 33 that tests fine manual dexterity. 34 Participants pick up and place 9 wooden pegs into holes on a pegboard and remove them as quickly as possible. The total time required for the task is recorded and compared with age- and gender-specific norms,34,35 with faster times indicating better fine manual dexterity.
Therapist Ratings Based on a Numerical Scale
Arm Motor Ability Test (AMAT)
The AMAT was developed as a supplement to the Wolf Motor Function Test (WMFT; described below) to determine the efficacy of constraint-induced movement therapy on improving ADLs.36,37 The AMAT is designed for higher-functioning individuals with stroke and measures the quality and quantity of UL movement in everyday activities. The evaluation consists of 13 ADLs with 28 subtasks. Unimanual activities are performed with the affected arm. Bimanual tasks are performed using (or attempting to use) the dominant limb in the same role as before the stroke. The AMAT includes 2 scores for each task: time to complete the task and functional ability. Functional ability considers the actual motor performance of the affected arm and is subjectively rated on a 6-point ordinal scale from 0 = No use to 5 = Normal use. The instructions focus on quality rather than speed of movement, but no clear definition of movement quality is provided.
Action Research Arm Test (ARAT)
The ARAT assesses specific changes in UL activity limitations in patients with stroke. 38 It includes 19 items divided into 4 subscales (grasp, grip, pinch, and gross movement) grouped according to a hierarchical Guttman scale. Successful task completion implies that subsequent, easier tasks can also be successfully completed. 26 Similarly, failure on an easier item predicts failure on all items of greater difficulty in the subscale. Items within each subtest are scored on 4-point ordinal scales ranging from 0 to 3: 0 = No movement possible; 1 = Performs test partially; 2 = Completes the task, but takes abnormally long or has great difficulty; and 3 = Normal performance of the task. The overall score indicates task completion and is expressed as the sum of subscale scores ranging from 0 to 57. The ordinal scale does not allow tracking small changes in movement quality and does not identify or score compensations used during task completion. Based on a Rasch analysis, Chen et al 39 found a disordering of the ARAT threshold measure, indicating that the original 4-point scale inadequately differentiates stroke severity. They also suggested a redundancy in the 4 rating categories because a score of 1 was infrequently used compared with other categories. The authors suggested adopting a 3-point scale: 1 = Can perform no part of the test or partially perform the test within 60 s; 2 = Completes test but takes an abnormally long time (5-60 s) or has great difficulty; and 3 = Performs test normally within 5 s.
Chedoke Arm and Hand Activity Inventory (CAHAI)
The CAHAI is intended to complement the Chedoke-McMaster Stroke Assessment that classifies UL impairment into 7 recovery stages.40,41 It consists of 13 real-life functional tasks involving both ULs and incorporating a range of movements, pinches, and grasps reflecting poststroke recovery stages. Shorter versions of CAHAI are available with 7, 8, or 9 tasks. All tasks are scored using 7-point quantitative scales, similar to those used in the Functional Independence Measure. 42 The affected limb is scored according to its positioning and contribution to the task. For individuals who are able to complete the tasks without assistance (score of >5), the scoring scale discriminates between tasks completed normally, without apparent difficulty, and those made with compensations and are slower or with safety concerns.
Frenchay Arm Test (FAT)
The FAT is a measure of UL gross motor function during ADL performance in patients with neurological disorders. 43 The FAT includes 5 tasks completed with the affected arm. Each item is scored as either Pass = 1 or Fail = 0, with total scores ranging from 0 to 5. The FAT measures task accomplishment without accounting for movement quality.
Motor Evaluation Scale for Upper Extremity in Stroke Patients (MESUPES)
Unlike the other scales, the stated aim of MESUPES is to measure arm and hand movement quality in individuals with stroke.24,44 The MESUPES focuses on “normal” performance, defined as “movements that are painless, made without tremor, and executed with a normal range of motion using adequate muscle contraction and orientations of various body segments.” Thus, consistent with our definition, movement quality is assessed by identifying whether a normal range of motion of specific joints occurs. The scale includes 17 items divided into 2 subscales with 6 and 3 response categories for arm and hand function, respectively. For the arm subscale, movements are performed in 3 consecutive phases: (1) the therapist moves the arm passively to evaluate muscle tone (scores 0-1); (2) the therapist assists the patient during the movement and evaluates the active contribution through normal muscle contraction (score 2); and (3) the patient performs the task, and the therapist evaluates the range of active motion (score 3-5). For hand function (MESUPES hand test), patients are instructed to actively perform specific hand and finger movements. Range of motion and hand orientation are both scored on 3-point scales. For both subscales, compensations are partially taken into account by giving a score of 0 when there is inadequate tone, abnormal muscle contractions, or synergic (flexor/extensor) or mass movement patterns.
Reaching Performance Scale for Stroke (RPSS)
The RPSS was designed to address the gap in current outcome measures in assessment of movement quality. 21 This 6-item measure quantifies movement patterns and compensations on 4-point scales (0-3), used when reaching to grasp a cone located close to and far from the body. Movement components evaluated are the following: trunk displacement, end point movement smoothness, shoulder movements (eg, shoulder flexion and horizontal adduction with scapular elevation), elbow movements (eg, extension), quality of prehension, and global task performance. The first 5 items identify deficits in specific aspects of the movement. The global task performance item scores task accomplishment based on the presence or absence of compensatory strategies. The 6 task scores are summed for a total score of reaching performance ranging from 0 to 18 for each target distance.
Sollerman Hand Function Test (Sollerman)
The Sollerman was designed to assess hand function and handgrips needed for 20 ADL subtests. 45 Most subtests are unimanual tasks. Scoring accounts for the time taken, the level of difficulty, and the quality of performance, based on the observed use of a correct pinch or grip position. Each subtest is scored on a 5-point scale from 0 (task cannot be performed at all) to 4 (task is completed without any difficulty within the time frame and with the prescribed handgrip of normal quality). Examples of prescribed handgrips are provided for each task to facilitate scoring. The maximal score is 80, obtained by summing scores of the 20 subtests. Scoring the Sollerman is reportedly challenging because the assessor must be aware of multiple factors occurring simultaneously (eg, passage of time, difficulty, correct positioning, and quality of performance). 46
Test d’Évaluation des Membres supérieurs des Personnes Âgées (TEMPA)
The TEMPA evaluates 4 unimanual and 5 bimanual tasks related to routine daily activities in individuals older than 60 years. 47 Although not specifically designed for individuals with neurological disorders, it has been validated in patients with stroke and multiple sclerosis. 48 Normative values for execution time are available for people aged between 45 and 59 years old. 49 Three subscores evaluate each task: speed of execution, functional rating, and task analysis. Speed of execution measures the time taken to complete the task. The functional rating grades the level of autonomy on a 4-level ordinal scale: 0 = Task was performed successfully and easily without hesitation; −1 = Task completed with some difficulty; −2 = Significant difficulty in performing the task, trial-and-error or partial completion of the task; −3 = Unable to complete the task despite some assistance. Task analysis identifies and quantifies difficulties encountered during the task by rating 5 categories on 4-point scales: range of movement, strength, control of gross movement, prehension patterns, and fine movement.
Wolf Motor Function Test
The WMFT was originally developed to qualify the effects of constraint-induced movement therapy in individuals with mild to moderate stroke. 50 The WMFT consists of 2 strength-based and 15 function-based tasks, arranged in order of complexity, used to assess UL impairments and activity limitations. Task progression is based on proximal to distal joint involvement. 50 Performance is scored both on task completion time and on a 6-point ordinal scale (0 = Does not attempt with involved arm to 5 = Arm does participate and movement appears normal), rating the ability to perform the task as well as the presence of synergies influencing movement, required effort, movement speed, precision, fluidity, and fine coordination.
Patient Self-report Measures
ABILHAND
The ABILHAND is a semistructured questionnaire measuring manual ability according to an individual’s perceived difficulty to perform 23 bimanual tasks representing a wide range of complex daily activities.51,52 Patients rate their perceived level of difficulty on 3-point scales (0 = Impossible; 1 = Difficult; and 2 = Easy) when activities are done without help, irrespective of the limb(s) or strategies used. Scoring allows compensation, assistive equipment, or the use of the unaffected limb. The ABILHAND was developed using the Rasch measurement model. Task scores are entered into the WINSTEPS computer program, where the ordinal raw score is converted to a linear measure expressed in logits (a linear unit that expresses the patient’s odds of success on any given task). The center of the scale is set to 0 logit, whereby higher logit numbers indicate more perceived ability. 53
Capabilities of Upper Extremity Instrument (CUE)
CUE is a self-report measure assessing UL functional limitations in individuals with tetraplegia.54,55 The revised version consists of 15 items scored separately for each arm and 2 bimanual tasks. Tasks focus on the ability to reach or lift, pull/push a light/heavy object, move and position the arm and wrist, use the hands and fingers, and press with the index finger. Perceived difficulty is rated by patients on a 5-point scale from 0 (unable/complete difficulty) to 4 (no difficulty). The instructions ask respondents to think about the specific part of the arm or hand asked about in each question.
Motor-Activity Log (MAL)
The MAL is a semistructured interview assessing how much (Amount of Movement Scale) and how well (Quality of Movement Scale) individuals use their more-affected arm outside of the clinical setting. 56 Different versions include 14, 28, or 30 daily functional tasks.57,58 Tasks include object manipulation and arm use during gross motor activities (eg, transferring to a car or getting up from a chair). Each section is scored on 6-point scales ranging from 0 = Never use to 5 = Same as prestroke. Individuals may also select scores halfway between the anchors. The Quality of Movement Scale rates how well the most-affected arm contributes to the task and also captures if movements are slower, less accurate, or require more effort.
Analysis of the Degree to Which Clinical Scales Assess Movement Quality
Out of the 15 activity level outcome measures identified, 9 assess how movement is performed by measuring either end point characteristics or movement quality: AMAT, ARAT, CAHAI, MAL, MESUPES, RPSS, Sollerman, TEMPA and WMFT. Among those measures, the RPSS is the only one that assesses both end point characteristics and movement quality as well as the presence of compensatory movements. Unlike other measures, the RPSS quantifies how arm joints, trunk, and hand are used during reaching tasks and directly quantifies compensatory movements based on observational kinematics. The MESUPES also primarily assesses movement quality by considering how similar the movement is to premorbid movement patterns and the presence of compensatory movements but does not assess end point characteristics. The 7 other measures (AMAT, ARAT, CAHAI, MAL, Sollerman, TEMPA, and WMFT) account to some extent for how movements are performed and attempt to capture the consequences of diminished end point characteristics and movement quality, but deficits are quantified only indirectly by giving a partial score if movements are slower or if difficulties are experienced. Specifically, AMAT and WMFT score tasks according to task accomplishment (time) while accounting for movement strategies. By rating performance in 5 categories (range of movement, strength, control of gross movement, prehension patterns, and fine movement), TEMPA also identifies and quantifies difficulties encountered during the task. However, these measures use a summary task score or only rate global task performance, limiting their sensitivity to identify improvements in specific movement patterns. 59
Six outcome measures do not assess parameters related to movement quality and do not distinguish between improvements resulting from motor strategies or those related to the presence of compensatory strategies. These include measures based on time (BBT, Jebsen, and NHPT), 2 of the self-report measures (ABILHAND and CUE), and FAT. Outcome measures based on time assume that decreases in movement speed are a result of UL impairments, but this assumption has only been kinematically validated for interjoint coordination. 60 Although speed of execution is a good indicator of UL ability, it provides little information about movement quality, difficulties encountered, or why more or less time may have been needed. 61 For example, an improvement in score may be based on the increased use of compensatory strategies rather than a decrease in UL impairments.4,62 In particular, during a reaching movement, patients with stroke who used more trunk compensations made faster reaching movements than patients who used movement patterns more similar to those observed in healthy individuals.4,62 For self-report measures, an important barrier to movement quality assessment is the individual’s subjective perception, especially for patients with cognitive and/or communication difficulties that frequently occur after neurological injury.63,64 Actual performance can be overestimated or underestimated, depending on motivation and cognitive ability. 51 For example, Woodbury et al 65 noted some discrepancies between movement quality measured with kinematic analysis and patients’ self-perceptions. Patients who were asked to rate their performance did not perceive changes in movement quality despite significant objective improvements in arm movements, faster accomplishment of functional tasks, straighter hand trajectories, and decreased trunk displacement.
Limitations
This review focuses only on ICF activity level outcome measures specific to the UL. Therefore, common impairment level outcome measures that arguably measure some elements of movement quality were excluded. Furthermore, the selection of outcome measures was based on national clinical practice guidelines from Canada and the United States. There may have been relevant outcome measures recommended in guidelines from other countries that were not included. This review did not focus on the clinical utility of the outcome measures (eg, time of administration, availability), which may influence their selection in clinical practice.
Conclusion
We identified several UL outcome measures at the ICF activity level that distinguish between motor recovery and compensation as defined in the motor skill acquisition and motor control literature. 20 This is new information because most previous reviews on UL outcome measures have primarily focused on psychometric properties and/or clinical feasibility (see Alt Murphy and Häger 3 ). Whereas the systematic review by Lemmens et al 66 categorized outcome measures for individuals with stroke and cerebral palsy, the concept of movement quality was not defined and was rated subjectively on a yes/no basis, making the validity of the results questionable.
This review highlights an important problem with outcome measures used in neurological practice. Outcome measures that do not assess end point characteristics and movement quality are limited in their ability to track changes over time, distinguish compensation from recovery, and identify specific motor impairments. Those that do assess these 2 levels of behavior, such as the RPSS, can help clinicians by providing complementary information about functional performance measured on other UL activity scales. This review can help guide clinicians in the selection of outcome measures to better assess motor recovery and treatment effectiveness. As an alternative to using laboratory-based kinematic analysis, observational kinematics is an easy and low-cost approach to the assessment of UL movement in clinical practice. Observational kinematics consists of the assessment of body motions by an observer, in the absence of objective quantification using sophisticated technology (ie, motion analysis systems). The evaluator can assess kinematics by observing the individual perform a task and rating the performance (eg, movement speed, smoothness, hand path directness) and or movement quality (eg, joint/segment movement) with a rating system (eg, numerical or Visual Analogue Scale). Bernhardt et al 67 found that physical therapists could make moderately to highly accurate judgments about the characteristics of end point and joint movements for a given task. Observation of kinematics may also allow clinicians to make inferences about UL impairments, such as joint contractures or spasticity, that may interfere with movement. 21 Advances in low-cost markerless motion capture technology (Microsoft Kinect II, Washington, DC) also show promise for clinical assessment of UL movement quality during the accomplishment of everyday life tasks.68,69
Future research should focus on developing better outcome measures with strong psychometric properties to assess UL end point characteristics and movement quality during varied and meaningful ADLs, preferably done in real-life settings or in the patient’s own environment. It is anticipated that options for assessing UL movement quality will also expand with the development of new technology, making kinematic motion analysis more accessible to clinical practice. Incorporating more information about movement quality and use of compensatory strategies will allow clinicians to provide more individualized treatment by targeting key elements limiting motor recovery in individuals with neurological disorders.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: MD is supported by the Fonds de la Recherche du Québec en Santé (FRQS) and the David G. Guthrie Fellowships in Medicine from the Faculty of Medicine, McGill University. MFL holds a Tier 1 Canada Research Chair in Motor Recovery and Rehabilitation.
