Abstract
Introduction
With over 80 million global stroke survivors as of 2016, 1 of which 40% suffer long-term deficits, 2 there is need to enhance long-term motor recovery. Humans often show chronic impairment in distal forelimb control,3,4 including digit individuation and muscle synergies, affecting the ability to precisely apply force. Controlled application of force is separate from strength in both functional effect and recovery time-course.5,6 Cortical stroke in rodents affects similar aspects of control.7,8 but it has yet to be determined whether precision is captured outside of kinematic measurements for rodents.
To address this, we modified an automated reach-and-grasp task (requiring synergies but not digit individuation), 9 which may have previously assessed mixed impairment of the generation of force (“weakness”) and conditional application and coordination of force (“precision”). We trained mice to pull between 8 and 11 g instead of over 20 g, allowing errors via force overexertion, to determine if this assay measures precision deficits rather than weakness alone.
Materials and Methods
Subjects
Six female and 3 male adult C57-Bl6 wild-type mice from Jackson Laboratories were tested during the dark period of a 12-hour reverse light cycle. All procedures were approved by the UT Southwestern Medical Center Institutional Animal Care and Use Committee.
Behavior
Procedures were nearly identical to those described in Becker et al. 9 Briefly, mice gripped a handle through an opening in an enclosure wall. A trial began at paw extension and ended after 1 second of withdrawal. An isometric transducer recorded force applied when the handle was pulled toward the mouse. As a modification, we required a peak force between 8 and 11 g. Baseline continued until 3 consecutive success rates varied by ≤15%.
Stroke
Photothrombotic stroke was induced as previously described. 10 Mice were anesthetized with 30% O2, 70% N2O, and 1% to 4% isoflurane. The scalp was injected with lidocaine and incised. One minute after intraperitoneal injection of 0.3 cm3 Rose Bengal (1.5 mg), a laser (Sapphire 561) 5 inches above the head delivered 65 mW above the forepaw motor cortex, 1.7 mm to the left of bregma, for 15 minutes. Animals then received 0.2 cm3 saline and 0.1 cm3 buprenorphine.
Poststroke
Animals underwent reach sessions on the third day, seventh day, then weekly until 6 weeks poststroke, then were sacrificed with an overdose of isoflurane. Coronal sections were collected as previously described. 9 The section closest to 1.7 mm anterior to bregma and 5 slices every 720 μm posterior were stained with cresyl violet and visualized. Ipsilateral healthy tissue was subtracted from contralateral and the sum multiplied by 720 μm to estimate stroke volume.
Analysis of Behavioral Data and Statistical Analysis
The first 50 trials of each session were analyzed and success rate noted (successful trials/50). Dependent variables were closest force (the average across trials of the peak that was closest to 9.5 g), maximum force (the average across trials of the highest peak), all pulls (the average of all peaks in all trials), closest force error (the root mean square of the difference between the closest force and 9.5 g), and pull latency (initiation to first pull).
Animals were considered impaired if their success rate on day 7 was <70% of baseline. Further analysis used only impaired animals. A one-way analysis of variance assessed changes in success rate over time (
Results
Impairment (success <70% of baseline) occurred in 4 females and 2 males and was sustained through 6 weeks (Figure 1A). Four mice increased all pulls over 11g (Figure 1C). Neither the highest force per trial (
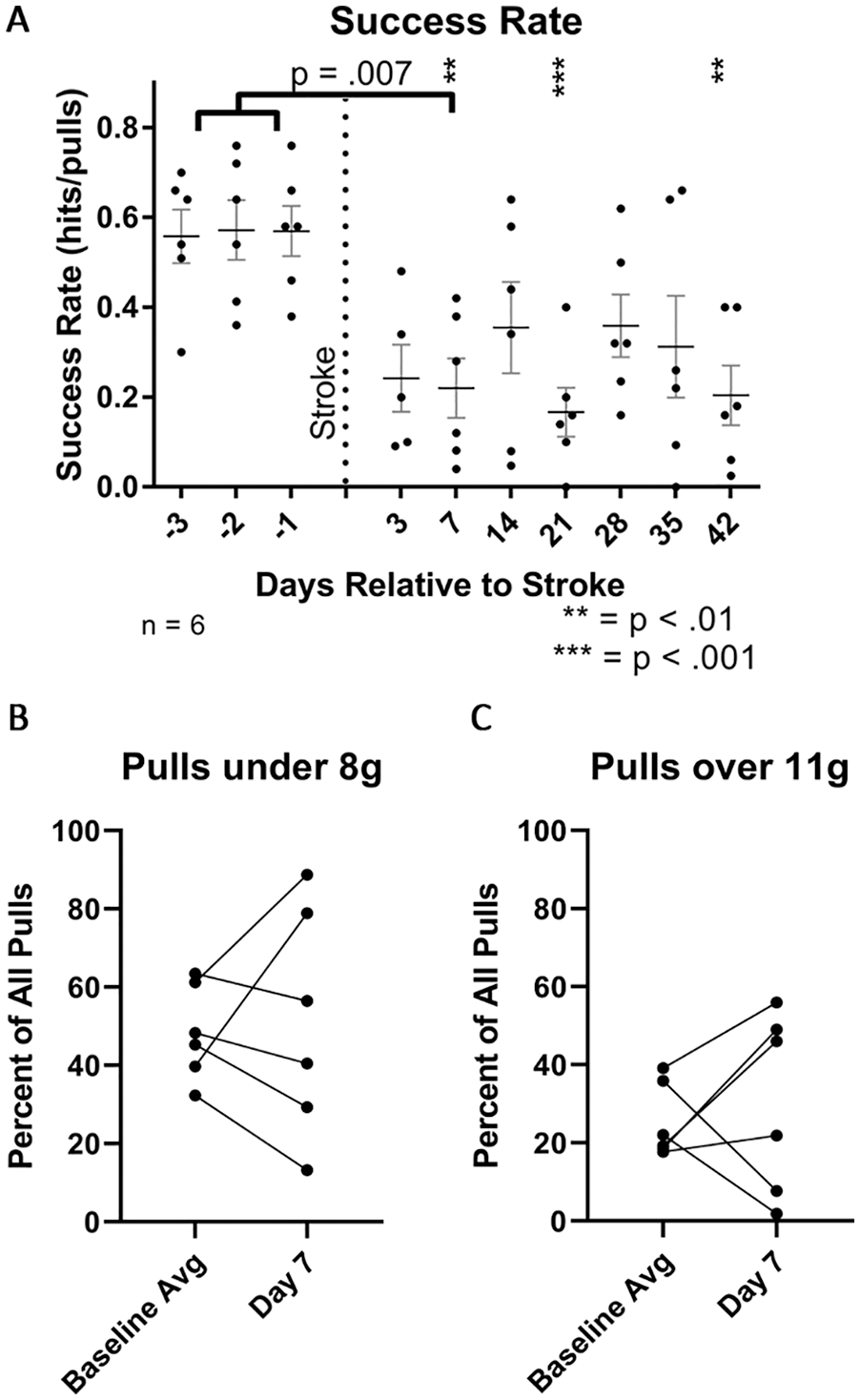
Individual impairments show increases and decreases in errors on both sides of the target. (A) Reduction in success rate on day 7 (analysis of variance
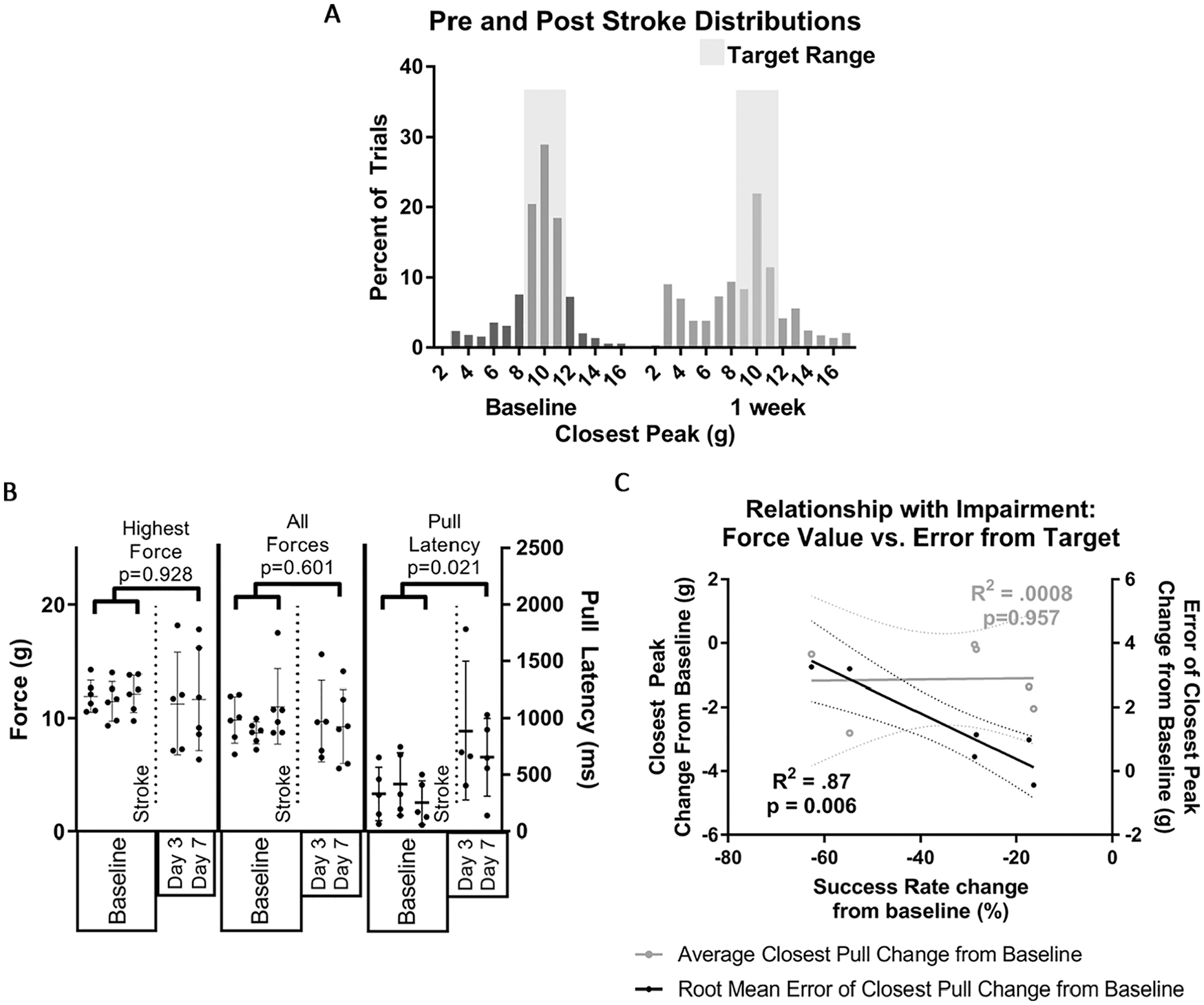
Behavioral impairment following stroke in animals with <70% baseline success rate. (A) Errors in the closest force to the target were redistributed poststroke. (B) Neither highest force nor average of all pulls were affected; latencies trended toward an increase. (C) Changes in the error of the closest force was correlated to change in success rate, but the change in the force values themselves were not, indicating that deficits are not due only to downward shifts in force exertion.
Discussion
We sought to determine if this assay measures stroke-induced precision impairment. Weakness alone should shift the distribution of pull forces downward; this simple shift did not occur. Neither the average force of all pulls nor the maximum force was affected by stroke. The distribution of pulls closest to the target shifted; pulls over or under the target force range (12-17 g and 6-7 g), and just above the threshold required to register a pull (2-4 g) increased after stroke (Figure 1A). This may represent distinct errors: overshoot, undershoot, or failure to properly grip the handle (which could account for trending pull latencies). Impairments were smaller than with the 20 g criterion, 9 potentially because the latter requires both strength and precision or because strokes were smaller in this group (5.94-9.11 mm3).
We conclude that this assay can measure motor cortical impairments in precision. Since we did not assess strength directly, future work is needed to determine whether similar approaches may be able to measure strength and precision separately to better model the separation demonstrated in the human literature.3-6
Footnotes
Acknowledgements
We are grateful to Ann Stowe, Robert Rennaker, Michael Kilgard, Hanan Muzemil, Eric Meyers, and Erik Plautz for support.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by NIH training grants T32 NS069562 (AMB) and T32 GM113896 (DMB), NIH U24 NS107176 (MPG), the Patrick and Beatrice Haggerty Foundation (MPG), and the Texas Institute for Brain Injury and Repair (MPG).
