Abstract
Cognitive impairment is now widely accepted as a fundamental aspect of Parkinson’s disease (PD). Given the prevalence of cognitive impairment and the associated impact on well-being, evidence-based interventions are needed. However, while research is continually accumulating in order to better understand the pathology and trajectory of cognitive changes, treatment options lag behind. Nonpharmacological approaches are of particular interest in this group, given the typical polypharmacy already present in PD patients. In this regard, cognitive training (CT) is a relatively new and prominent therapeutic option with accumulating scientific support and increasing public awareness. Research has now established benefits across many different populations, and trials investigating the use of CT specifically in PD are becoming more common. We offer a brief summary of CT and its efficacy in PD samples to date, as well as discuss areas requiring further exploration in this group. Crucially, we suggest that CT should be supported as a research priority in PD, given both proven and potential benefits as a noninvasive and well-tolerated behavioral intervention for cognitive impairment.
Introduction
Since its first identification as the “shaking palsy” in 1817, 1 Parkinson’s disease (PD) has been described, researched, and treated primarily as a movement disorder characterized by its classical motor features. However, over the past decade, in particular, there has been an exceptional and necessary expansion of interest into better understanding the nonmotor symptoms (NMS) of PD,2,3 revolutionizing how we now characterize and treat the condition. Central to the NMS syndrome are cognitive disturbances, whereby it is now recognized that PD with dementia (PDD) is a likely consequence of the disease. 4
Importantly, however, prior to the establishment of a frank dementia syndrome and/or even in those who do not progress to PDD, more subtle and specific cognitive impairment is often reported at earlier stages of the disease. Indeed, it has been shown that approximately 25% of newly diagnosed patients with PD have mild cognitive impairment (MCI). 5 The precise trajectory of these early cognitive impairments in PD remains unclear, possibly owing to the level of heterogeneity among patients that is known to affect MCI rates.6,7 For example, while most patients tend to present with a frontostriatal pattern of slowing and executive impairments, others may display a more posterior cortical pattern known to be associated with dominant Lewy-body pathology, typically more predictive of subsequent dementia.8-10 Changes to cognition in PD patients are significantly associated with reduced quality of life for both patient 11 and carer 12 and leads to increased health-related costs. 13 As such, it is vital that we explore options for interventions targeting cognitive deterioration in the early stages of PD, much like the roles of speech, occupational therapy, and physiotherapy prior to significant motor or functional deterioration. In this way, the ultimate aim must be to improve patient well-being and functioning as well as (ultimately) delaying further decline.
A Pressing Need for Cognitive Intervention in PD
At present, there is no established, successful pharmacological treatment for cognitive impairment in PD. 14 In PD patients with dementia, only modest improvements have been shown with medications, most prominently with cholinesterase inhibitors such as rivastigmine. 15 In patients with milder levels of cognitive impairment, there are even fewer treatment options available.14,16 In any case, current drug treatments targeting cognition are only modestly effective, are symptomatic rather than curative or slowing, and thus do not target underlying neuropathology. An additional problem with drug-based therapy for cognitive decline is the significant polypharmacy typical of this disease, with patients often taking multiple pharmacological agents already, relating to motor symptoms and other NMS such as sleep and/or mood disturbance. Additionally, high levels of pharmacological treatment are often accompanied by significant side effects. Given the current state of available treatment options for PD, ongoing investigation of disease-modifying therapies or behavioral interventions that may alter the trajectory of cognitive decline in PD are of the utmost importance. 17
Modifiable Risk Factors and the Need for Early Intervention
Unfortunately, like PD, other neurodegenerative syndromes such as Alzheimer’s disease remain in a similar state regarding treatment options. As such there has been a rapid shift in focus in the ageing literature more broadly to intervention strategies targeting cognitive impairment much earlier in the disease process.18,19 Recently, a large meta-analysis by Norton and colleagues 20 demonstrated critical findings with respect to the justification for early intervention, showing that half of dementia cases worldwide may be attributable to modifiable risk factors (eg, vascular risk factors, depression, and cognitive inactivity), or a third after adjusting for nonindependence between factors. Given this, there are a growing number of multifaceted studies now exploring the cognitive benefits of different nonpharmacological interventions. 21 Such findings can be linked back to PD patients, where benefits using this line of intervention may also be possible. For example, 2 recent studies have shown that physical exercise can be efficacious for cognition,22,23 although replication in randomized controlled trials (RCTs) is required to expand on this preliminary evidence. With regard to the modifiable risk factor of cognitive inactivity, another nonpharmacological intervention that is growing in popularity is cognitive training (CT).
A Framework for Cognitive Training in PD
In the following sections, we will provide a broad and inclusive review of the theory underlying CT in aging generally, leading into PD more specifically. We believe that a strong understanding of work in CT more broadly can help speed up the process of delivering high-quality trials in PD. This section is followed by a basic overview of CT in PD. We emphasize that this is not a systematic review of individual trials, nor simply a discussion of CT efficacy in PD, and thus a more detailed critique and summary of individual studies can be found elsewhere.24-27 The aim of the current review is to discuss important theoretical background and practical considerations gained from the broader ageing literature, which can inform future trial implementation in PD.
Cognitive Training as a Potential Therapeutic Technique
A Brief Introduction to CT
CT is an intervention that has long been utilized to target cognition in a range of psychiatric, developmental, and neurological disorders, most notably in schizophrenia, attention deficit disorder, and age-related cognitive decline. 28 In healthy older adults, the large (n = 2832) ACTIVE Trial reported positive and long-term effects of 3 types of CT programs over 2-, 5-, and even 10-year follow-up periods.29-31 Yet training effects were largely restricted to the cognitive domain targeted by each training program, and despite attenuation of Instrumental Activities of Daily Living compared to a no-contact control group, ACTIVE did not find a difference between trained and control participants in incidence of dementia at 5-year follow-up. 32 Nonetheless, the ACTIVE results provided the impetus for a number of studies seeking to utilize CT as a possible strategy for attenuating cognitive decline in ageing and neurodegenerative disease. 33
The central premise of CT is to provide theoretically driven skills and strategies, typically involving guided practice on tasks that reflect specific cognitive functions, such as processing speed, memory, attention, and executive functions (see description in Mowszowski et al 18 ). CT may be based on drill and practice computerized or pencil-and-paper exercises, may employ a strategy-based approach (eg, method of loci or instruction in the use of external memory aids), or may use a combination of both. Computerized CT is currently the most common approach in research settings and involves game-like exercises that target core cognitive abilities using engaging motivational cues and on-time feedback, thus functioning figuratively as a “brain gym.” Most programs use a staircase adaptive design, whereby task complexity and response time demands change frequently during and across sessions, in accordance with changes in individual performance in order to avoid over- or understimulation. In addition, several computerized CT programs adapt training content (ie, targeted domains) to individual needs, providing more training time in areas of relative weakness. This is of particular interest in conditions characterized by a pattern of specific cognitive changes.
Strategy-based CT involves teaching and practice of internal and external strategies and techniques to enhance cognitive performance in everyday life (eg, using visual imagery, categorization or face-name associations, as well as memory aids such as diaries, calendars, alarms, and lists). Both types of CT may be performed either at home or in laboratory, clinic or community settings, and in individual or group formats.
Evidence for CT Efficacy
A recent meta-analysis encompassing 51 RCTs of computerized CT in 4885 healthy older adults reported modest improvements in overall neuropsychological performance (g = 0.22, 95% confidence interval [CI] = 0.15-0.29) at immediate follow-up to training. 34 However, this meta-analysis found that effects differ across cognitive domains and intervention design. In particular, positive effects were found only in trials of supervised (group-based) training, provided for at least 30 minutes per session and not more than 3 times per week. It is not known whether such a pattern of efficacy transfers to PD patients. This is highly relevant given their significant mobility issues likely interfering with program involvement and adherence. In terms of memory strategy training, a separate meta-analysis in healthy older adults (35 studies, 3797 participants) reported a moderate effect size improvement in memory posttraining compared to controls (d = 0.31, 95% CI = 0.22-0.39), but this was not limited to RCTs. 35
In addition to healthy older adults, there is a growing literature showing cognitive and psychosocial improvements in older adults with MCI.36,37 A multifaceted group program in people with late-life depression that utilized both computerized and memory strategy training, along with psychoeducation for dementia risk factors, showed moderate to large effect size improvements in memory 38 as well as reduced disability. Using a similar methodology for older adults “at risk” of dementia (80% with MCI), our group showed medium effect size improvements in episodic memory, as well as independent effects in mood and sleep quality. 39 In addition, a small RCT of a combined intervention reported significant reduction in incident dementia in people with MCI. 40 However, the efficacy of CT in demented populations is less clear, with a paucity of adequate RCTs in the literature. We have discussed previously how the underlying neuropathological changes in patients who have progressed to established dementia may be too severe to benefit from this type of intervention, 18 and furthermore, their cognitive deficits may preclude them from the active participation required. A broader-focused program of cognitive stimulation rather than specific training may be more effective in such patients, and accordingly, the aim of therapy may be focused on other outcomes such as behavior, mood, or well-being.41,42
Despite evidence for efficacy on cognition in healthy older adults and those with MCI, there is neither consensus regarding whether CT can indeed prevent incident dementia nor what intervention approaches are most useful and for whom. The current body of evidence in the field lacks RCTs with sufficient sample size as well as intervention and follow-up periods to examine sustained benefits and the possible effects of CT on dementia prevention. Similarly, the great majority of trials to date have compared CT to no-contact or sham control groups. Future trials comparing 2 or more CT programs head-to-head (akin to active-controlled pharmacologic trials) are now warranted in order to determine which specific programs are most beneficial. In the meantime, as the field is still unregulated, many commercial “brain training” products are marketed directly to vulnerable consumers on the basis of flawed, irrelevant, or misinterpreted evidence bases.
Mechanisms of CT Efficacy
While cognitive inactivity is a key dementia risk factor, 20 evidence from epidemiological 43 and intervention studies 44 indicate that engagement in cognitively and socially stimulating activities can decrease neurodegeneration, cognitive decline, and dementia risk. The neural mechanisms underlying these effects are still unclear, as there is no systematic evidence as yet from postmortem studies linking such activities to reduction of key neuropathologies of Alzheimer’s disease or other dementias. 42 Rather, there is robust evidence for active lifestyle as a means to reduce risk for cognitive decline, and this is well described by the theory of cognitive reserve 45 as well as the scaffolding theory of ageing, 46 both of which link engagement over the lifespan to cognitive trajectories in later life.
The body of evidence for possible neurobiological benefits of CT is substantially smaller than that for cognitive effects, arguably because the complexity and cost of in vivo neuroimaging might have deterred researchers and funding bodies from such investigations until the basic questions of efficacy are elucidated. Nevertheless, at least a dozen published RCTs in healthy elderly and MCI confirm that the main neuroimaging modalities—structural, functional, and metabolic magnetic resonance imaging—are able to detect neurobiological changes with corresponding cognitive changes after several months of CT, and relate them to cognitive change.44,47,48
Neural changes in response to CT have been observed by way of 2 primary methods. Structural changes (ie, growth, or attenuated reduction in cortical thickness and/or white matter integrity) are potentially useful as a biomarker of CT-induced change. A number of studies have now shown altered brain structure in both white and gray matter in association with CT. For example, Engvig and colleagues have been able to show that in healthy older adults, 8 weeks of memory training led to regional increases in cortical thickness compared to controls. 49 Furthermore, these authors explored white matter changes in a second study, showing fractional anisotropy increases in those trained compared to control. 50 Additionally, functional changes (ie, altered patterns of cortical activity and resting state networks) are another possible indicator for improvement as illustrated in a study by Chapman and colleagues. 51 These authors found global and regional alterations to blood flow in resting state networks that were associated with cognitive changes following CT in older adults, as compared to a control group. Chapman and colleagues postulate that their results may reflect an aggregation of neurotransmitter receptors as a consequence of prior activation, that is, CT, which prepare the brain for better future performance. 51 In another intriguing result, Valenzuela et al 52 used metabolic imaging to uncover neurochemical changes in the brain as a direct result of memory training. These authors reported that training led to increases in creatine and phosphocreatine signals in the hippocampus compared to controls, which may have neuroprotective value. Interestingly, Backman and colleagues have illustrated increased striatal dopamine release following working memory training, 53 though this was in a young healthy sample. Nonetheless, such a finding may be of relevance in dopamine-depleted PD samples, and future research should look to investigate this further.
Nevertheless, reliable interpretation of such findings in the context of a restoration versus compensation framework are still under consideration and highlight the need for continued and thoughtful utility of neuroimaging in CT studies. As Park and Bischof 47 explain, both decreased activity in a particular region (by way of more efficient processing) and increased activity in additional regions (by way of compensatory processing) could act as mechanisms for improvement. In any case, though the specific processes at play need further investigation, and accurate interpretation remains unclear, it now appears that CT has the potential to produce neurobiological changes in older adults. Further investigations employing modern neuroimaging techniques are now required for better understanding and interpreting such observed changes while exploring the capacity for neuroplasticity in the context of neurodegeneration. 54
Is Cognitive Training Effective in Parkinson’s Disease?
Over the past decade, the substantial growth of interest in CT among researchers and the public alike has been particularly strong within the PD community. From our own clinical experience, PD patients and their families are attracted to advertising by commercial brain training companies, which typically promise a nonpharmacological, do-it-yourself, automatic, and at-home treatment option for cognitive symptoms. In light of this interest, it is essential for the clinical and research community to understand just how effective this intervention may be for patients with PD, in order to provide practical and appropriate advice to their patients.
Following the trend within the ageing literature more broadly, there has been a notable increase in published trials exploring CT in PD over the past 5 years. In 2011, our group was one of the first to commence a program combining strategy and computerized CT in patients with PD. 55 These novel results demonstrated that a 7-week multifaceted program combining psychoeducational, computerized CT, and memory strategy training was effective in improving performance on untrained, objective memory measures at follow-up. It was also associated with improved awareness of dementia risk factors, which is critical to foster lifestyle modifications that can be applied long after the intervention is ceased.
Many studies of CT in PD have now been published, of which 7 RCTs56-62 encompassing 272 patients with Hoehn & Yahr stages 1 to 3 were available for a 2015 meta-analysis. 26 CT was typically computerized rather than pen-and-pencil based, and more commonly facilitated in a group setting. Such a result is possibly surprising given the nature of the disease, though promising as these are seen as potentially more useful methods. 34 Interestingly, all but one of the studies included in the meta-analysis supplied multidomain training. It is currently unknown whether this is more or less effective than targeted domain-specific training in PD, and this could be an important avenue for further research.
While all of the studies included in this review were too small to detect statistically significant effects on overall cognition, the pooled overall effect size was significant (g = 0.23, 95% CI = 0.01-0.44). Perhaps more important given the specific pattern of impairment in PD, larger effects were found within individual cognitive domains, including measures of working memory (g = 0.74, 95% CI = 0.31-1.17), processing speed (g = 0.31, 95% CI = 0.01-0.61), and executive functions (g = 0.30, 95% CI = 0.01-0.58). 26 Again, many studies reported insignificant test results individually, and these findings therefore warrant larger RCTs. Such work will also help understand the optimal intervention strategy for mild to moderate PD, as well as to investigate the potential for CT to delay PDD onset.
The separation of patients from healthy, through MCI and dementia is common in CT studies more generally. In the studies included in the above meta-analysis, it was consistent that patients with cognitive impairment were removed from the trial at recruitment. While it has recently been shown that cognitively impaired PD patients have the capacity to improve after undertaking a motor-based rehabilitation program, 63 how this translates to cognitive interventions is unknown. Hindle and colleagues are currently in the process of addressing this very question with an exciting trial of cognitive rehabilitation for PDD underway. 64 As we have suggested above, these authors have determined that a more holistic program of rehabilitation, focusing on goal setting and real-life cognitive functioning as more appropriate in this cohort. These strategy-based cognitive remediation methods are known to be effective in older adults. 65
Another area requiring further research in PD relates to longitudinal outcomes and to the possible time-course effects during and after CT. 26 In this regard, a study by Petrelli and colleagues 66 was able to show, for the first time, that an RCT of CT may result in stable improvements that prevent further cognitive decline 12 months following training. These authors followed-up a subsample of patients from an earlier study 60 to investigate the probability of patients maintaining their cognitive abilities, dependent on which intervention they received. The authors found that participants who completed either structured or unstructured CT during the original trial 12 months previously were able to maintain similar levels of cognitive functioning, while those in the control group showed significant declines. Additionally, patients in the control group were 3 times more likely to develop MCI during this period than patients in either of the CT groups. Though hindered by a small sample and large number of dropouts, this trial provides hope that CT can be beneficial in the long term for patients with PD.
To illustrate the effect of CT interventions in PD over time, Figure 1 presents the potential pattern of changes using a simplified schematic. We propose that CT may either briefly stabilize cognitive decline—delaying the downward trajectory, or attenuate the rate of decline leading to a less dramatic rate of change. Importantly, this figure assumes patients undergo CT at the point of subjective changes to cognition. How these changes may be affected by the initiation of CT at earlier, or later, stages of the disease is important to uncover in future research. Finally, Figure 1 highlights the possible role of boosters, or additional CT training after completion. These may act to extend any observed changes for a longer period of time. Though clearly simplified, this diagram highlights the potential pathways to an ultimate goal of delayed dementia onset, given that the advancing neuropathological progression in PD makes sustained long-term improvement unlikely.
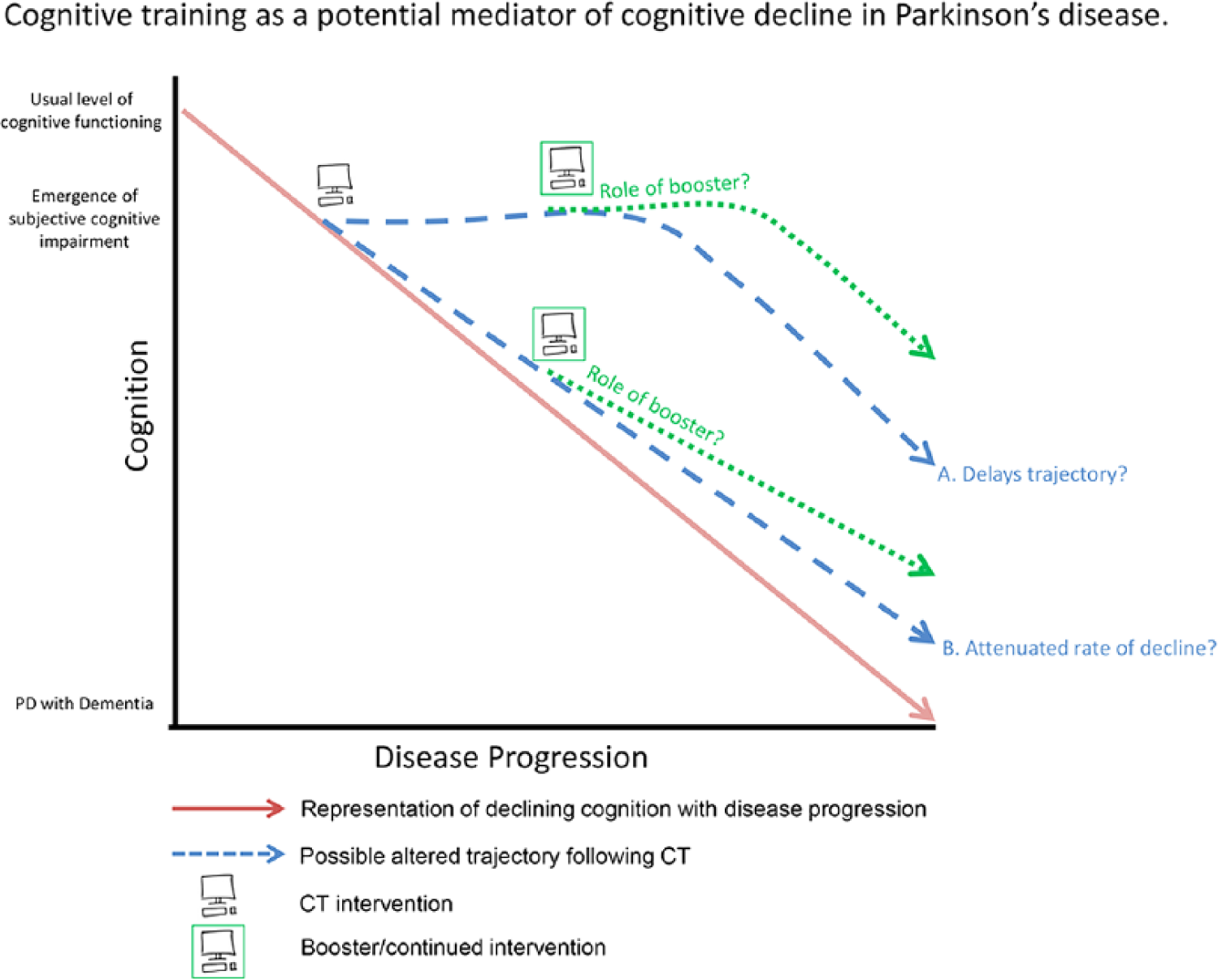
Schematic representation of potential outcomes from cognitive training (CT) in Parkinson’s disease (PD). It is currently unknown how a CT intervention affects long term on cognition in PD. We propose the following options: (1) Cognitive decline is delayed, possibly preventing dementia onset for an unknown period of time; (2) The trajectory of cognitive decline is slightly altered, again possibly delaying dementia onset. Finally, the role of boosters or long-term continued CT may lead to longer lasting or exaggerated effects on either of these outcomes.
Mechanisms of CT in PD
As previously discussed, one of the key mechanisms by which CT may take effect is through creating functional network changes in the brain to deal with neuropathology. While it has been known for some time that lifetime cognitive enrichment can allow older adults with neurodegenerative pathology to maintain normal clinical functioning,67,68 recent evidence suggests this is also true in PD. A meta-analysis conducted by Hindle et al showed that educational attainment is significantly associated with cognitive performance in PD. 69 However, as Lucero and colleagues rightly point out, to accurately determine the impact of cognitive reserve on the brain, the underlying neuropathology must be considered in addition to measures of cognitive performance, which may be biased toward more highly educated people. 70
To fill this gap in the literature, Lucero et al calculated mean cortical binding potentials for 155 patients as a marker of β-amyloid depositions and assessed the interactions between these and cognitive outcomes while taking into account educational achievement. Their results suggest that mean cortical binding potential is associated with poorer cognitive performance and Clinical Dementia Rating Scale scores in people with less than 16 years of education, while there was no such relationship for those who had more than 16 years of education, even though groups were matched on key variables. These results suggest than in PD, educational attainment (ie, a cognitively challenging lifestyle) may allow patients to clinically overcome underlying pathology in the brain. It is therefore plausible that cognitively stimulating activity as delivered through CT may also, under the right conditions, lead to functional neural changes in patients with PD (see Table 1).
Considerations for Future Cognitive Training Trials.
To the best of our knowledge, only 3 studies have investigated the neural underpinnings of CT effects in PD patients. An RCT by Angelucci and colleagues 71 investigated the effects of CT on brain-derived neurotrophic factor (BDNF), a neurotrophin that is highly involved in dopaminergic neuron protection, hippocampal neurogenesis, and which may affect cognitive functioning. In PD patients with MCI, BDNF is significantly associated with cognitive performance and has been suggested as a possible biomarker for evaluating cognitive change. 72 Increased levels of BDNF have previously been shown in patients with schizophrenia as a result of computerized CT. 73 Similarly, the aforementioned study by Angelucci and colleagues 71 showed that BDNF levels increase in response to CT in patients with PD. However, while cognition was improved at follow-up, BDNF and cognition were not correlated. This may relate to differential time-courses of neurobiological and behavioral changes following CT. 74 That is, it is likely that neurobiological changes may precede significant clinical outcomes.43,75
Two other studies have utilized functional magnetic resonance imaging and have linked CT-induced cognitive changes to altered BOLD response. Nombela et al 76 showed improvements in cognition after CT that were linked to altered neural activation in frontal areas and regions of the basal ganglia, while Cerasa et al 56 demonstrated cognitive benefits following CT, in addition to altered functional organization of key cognitive nodes in the dorsolateral prefrontal cortex and superior parietal lobule. While these results are promising with respect to uncovering the neurobiological mechanisms of CT specifically in PD, it is important to acknowledge that all 3 studies were significantly limited by small sample sizes, and furthermore, larger trials should employ neuroimaging markers where possible. As previously mentioned, research investigating dopaminergic responses to training in Parkinson’s patients should be investigated.
CT Targets More Than Cognition
Although cognition remains the central target for CT, there is a sufficient argument to be made that CT can induce improvements in other aspects of functioning. Given the substantial motor impairments and particularly gait abnormalities present in PD, these symptoms have more recently also become a focus of CT interventions. Previous studies have been able to show improvements in gait-related outcome measures in older adults as a result of computerized CT.77,78 This work stems from the growing understanding of how intimately cognitive functions such as attention and executive functioning are related to gait. 79 It is proposed that by improving the core cognitive features associated with gait, there may be accompanying improvements in motor functioning.
Indeed, an intriguing preliminary study conducted by Milman and colleagues 80 investigated this very question in PD patients using a 12-week CT program. Participants took part in attention and executive functioning–focused computerized CT, for three 30-minute sessions per week, over 3 months. The authors reported improvements in standardized “Up and Go” measurements, which were also associated with cognitive changes. Though this was a small, uncontrolled pilot study, it undoubtedly highlights the potential for such therapy in this field. Given the important role of cognition in freezing of gait in PD patients, we have also recently proposed that CT may work through a similar mechanism to relieve patients of this particularly debilitating motor symptom. 81 Indeed, we are currently undertaking a trial to investigate whether CT targeting attentional set-shifting, processing speed, and working memory may improve freezing of gait in PD (ANZCTR No. 12613000359730). Finally, it is plausible that computerized CT may also affect fine motor control and motor skills such as hand-eye coordination, psychomotor speed, and motor dexterity, making this area also worthy of further investigation.
In addition to these motor aspects of the disease, it is well known that patients with PD suffer a wide range of NMS such as sleep and mood disturbance. 82 Notably, in older adults “at risk” of dementia attending a specialist cognition clinic for concerns about cognitive decline, CT has been shown to induce improvements in these aspects of neuropsychiatric functioning. Diamond and colleagues from our group conducted an RCT implementing a 7-week CT program combining psychoeducation and CT, 39 using a similar protocol to that of our above-mentioned trial in PD. 55 This study showed that compared to a no-contact control group, the 36 patients who received the CT intervention showed not only improvements in memory but also in sleep and depressive symptoms. Indeed, a recent meta-analysis of computerized CT for major depressive disorder reported a moderate and statistically significant effect on depressive symptoms. 83 Yet pooling of depression outcomes from 5 trials of CT in PD did not reveal such effects, possibly because baseline depressive symptoms were subthreshold in all of the samples used. 26 CT in this context may be most effective for improving depressive symptoms in the 35% of PD patients with clinically meaningful symptomatology. 26 A particularly promising result was found by Pena and colleagues, 58 who showed a significant and large effect between treatment and control groups in reducing functional disability, as assessed with the World Health Organization Disability Assessment Scale. This is a considerable finding, though unique so far among studies in PD, and warrants replication. Overall, these encouraging findings emphasize the importance of including NMS such as sleep and depression in addition to measures of disability and quality of life as outcomes in future trials of CT. Indeed, a recent review has highlighted the transferable impact exercise can have on aspects including mood, cognition, and sleep in PD. 84
Future Considerations
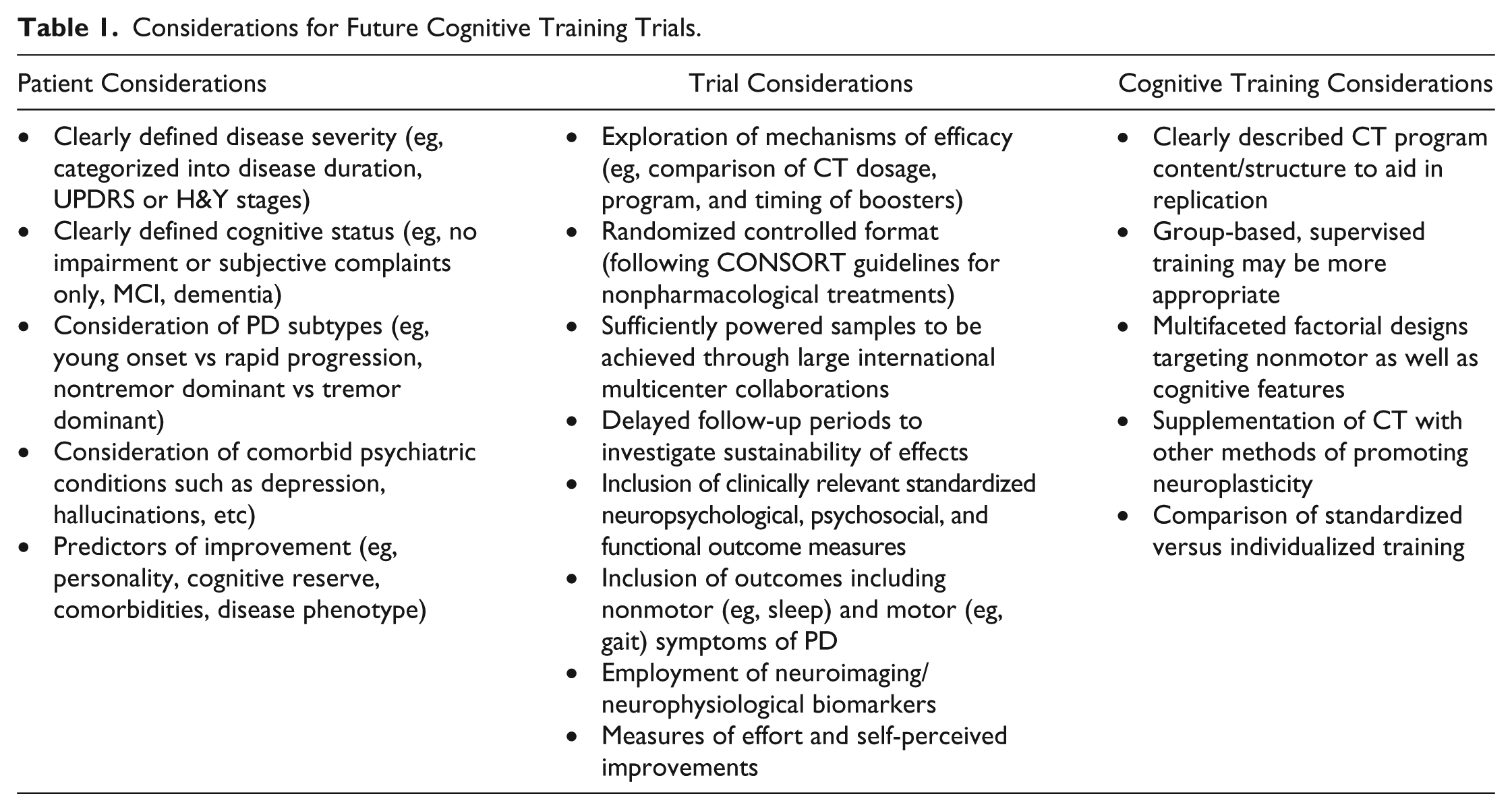
In introducing a recent special edition on the importance of cognition in Movement Disorders, Burn, Weintraub, and Robbins 85 draw attention to high priority areas for future research. Included is the need for large-scale clinical trials to determine the efficacy of various cognitive-enhancing treatments, including nonpharmacological interventions. Additionally, a brief correspondence86,87 between authors relating to the work of Petrelli and colleagues discussed above, 60 raises important questions regarding how to improve CT research in PD. Crucially, a new task force has been proposed by these authors, in order to establish recommendations for future studies, and we strongly agree that this would be a highly beneficial and necessary next step—something we have called for in the broader literature also. 33 Therefore, given its early demonstration of efficacy 26 and this recent call for action, we predict an upcoming surge of research into CT in PD. Thus, it is crucial that these trials be designed and planned in the most effective and rigorous manner, learning from the broader CT knowledge base where possible. In Table 1 we briefly summarize what we believe future trials should aim for.
At an introductory level, PD-CT trials should adhere to RCT guidelines at all times, minimize any potential methodological biases, and include adequately powered sample sizes. Our meta-analysis 26 suggested that, in general, samples in the field were relatively small (median N = 39), the majority did not include assessor blinding or intention-to-treat analyses, and in searching the literature it was clear that the proportion of RCTs to non-RCT designs was low. Large-scale international multicenter trials are now needed in order to address these pitfalls. Though there is clearly a need for broad inclusion in the early stages of work, future larger studies could be conducted to separate patients into mild, moderate, and severe classifications, in terms of both disease severity and cognitive impairment. Given findings in the ageing literature more broadly, it is highly likely, for example, that computerized CT may not be beneficial at the PDD stage, while it may be efficacious at the early phases of the disease, or where MCI is present. To a similar note, there is potential that particular subtypes within PD may respond to CT more strongly, given that some phenotypes are more predisposed to cognitive decline. 88 This has not yet been explored in the studies conducted so far, potentially due to the large sample sizes needed for such an analysis.
Conclusions
CT may well provide a unique and efficacious complimentary treatment for patients with PD who are either already faced with cognitive decline or in those who wish to prevent it. Though caution is warranted and expanding research is needed, this new focus of investigation should be embraced rather than resisted. As suggested by other authors, 87 a task force to further this cause may be highly valuable. In this regard, conducting large, long-term RCTs is key. Clinicians are advised to give positive, yet pragmatic and appropriate advice to patients wishing to take part in CT, particularly where substantial financial costs for commercial “brain training” programs are involved. Group-based community groups offering therapist-guided CT would be highly beneficial, most probably to a greater extent than commercial “brain training” for use at home.
Footnotes
Acknowledgements
We would like to thank the participants at the Parkinson’s Disease Research Clinic who have given their time to our cognitive training trials.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: CCW is supported by an Australian Postgraduate Award at the University of Sydney. SLN is supported by a National Health and Medical Research Council of Australia (NHMRC) Career Development Award (No. 1008117). AL is an NHMRC research fellow (ID 1108520). He receives in-kind research support in the form of no-cost software from BrainTrain Inc, HAPPYneuron Inc, and Synaptikon GmbH for projects unrelated to this work. LM is supported by an Alzheimer’s Australia Dementia Research Foundation Postdoctoral Research Fellowship at the University of Sydney. SJGL is supported by an NHMRC Practitioner Fellowship (No. 1003007) and funding from the Michael J Fox Foundation.
