Abstract
Purpose
Anticholinergic medication use measured via the Anticholinergic Cognitive Burden (ACB) scale has been associated with an increased dementia incidence in older adults but has not been explored specifically for Parkinson disease dementia (PDD). We used adjusted Cox models to estimate the risk of incident PDD associated with demographic factors, clinical characteristics, and time-varying total ACB in a longitudinal, deeply-phenotyped prospective PD cohort.
Major findings
56.5% of study participants were taking ACB-scale drugs at enrollment. Increasing age, motor symptom burden and psychosis were associated with PDD risk. Female sex and educational achievement were protective against PDD. ACB categories were not associated with PDD overall, but depression and impulse control disorder were strongly associated with PDD in a subsample with high baseline ACB.
Conclusions
Patient and clinical factors modify PDD risk. PD drug safety and drug-disease interaction studies may require considering multiple mechanisms and including dose-based, prospectively acquired medication exposure measures.
Introduction
Individuals diagnosed with Parkinson disease (PD) are at high risk of developing cognitive impairment, with longitudinal data suggesting 75% will have measurable cognitive impairment by the 10th year of living with PD.1,2 PD dementia (PDD) is the most dreaded long-term outcome of PD. There is a critical need to identify potentially modifiable risk factors for PDD. Exposure to medications with anticholinergic properties has been associated with an increased risk of all-cause dementia in older adult populations.3-9 These medications are frequently used among persons with PD,10,11 usually for the treatment of nonmotor symptoms of PD (eg, overactive bladder, insomnia, depression, or psychosis), and occasionally for motor symptoms.10,12,13 A few studies have suggested an increase in dementia risk associated with anticholinergic use in persons living with PD,14-17 but these studies frequently used retrospective or cross-sectional designs, increasing the potential influence of confounding and bias. The objective of this study was to evaluate risk factors for developing PDD, including sociodemographic and clinical characteristics, with a focus on the association of the Anticholinergic Cognitive Burden (ACB) scale as a potential modifiable factor, using existing data from a deeply-phenotyped longitudinal cohort study of persons with PD.
Methods
Ethical and Institutional Approval
The University of Pennsylvania’s Institutional Review Board approved this research (protocol #827738). The National Institutes of Health had no input on the study’s conduct or interpretation.
Data Source and Study Population
The National Institute on Aging Penn U19 (NIA U19) Center PD clinical cohort consists of 350+ individuals with PD, recruited from the University of Pennsylvania Parkinson Disease and Movement Disorders Center (PD&MDC) starting in 2007. The PD&MDC has a steady state census of approximately 3000 PD patients with approximately 1200 new patient evaluations annually by eight fellowship-trained academic movement disorders neurologists. NIA U19 Center PD participants receive extensive annual (years 1-5) and biennial (year 6 and beyond) motor, cognitive, and functional assessments. 18 Detailed information and assessments were collected on this cohort, including (1) demographic information, (2) family medical and neurodegenerative disease history, plus assessments of (3) motor signs and PD severity (original Unified Parkinson’s Disease Rating Scale [UPDRS], 19 then Movement Disorder Society UPDRS Part III [MDS-UPDRS-III]), 20 (4) cognitive function (Dementia Rating Scale-2 [DRS-2]) 1 , 2 (5) psychiatric symptoms (Cognition/Behavior Questionnaire, 21 and Geriatric Depression Scale [GDS-15]). 22 Over-the-counter and prescription medications were ascertained via participants’ self-reports and recorded by study personnel during study visits. The data for this study was queried and extracted on June 6, 2023, and all data were deemed current as of that date. Only participants with a normal DRS-2-measured cognitive function (scores >130) at study enrollment were included for analyses.
Exposure Definition
Because PD is associated with cholinergic deficit, we focused our analyses on anticholinergic medication exposure. There is no gold standard for measuring anticholinergic drug exposure in observational studies. Retrospective measurements commonly use the Anticholinergic Cognitive Burden (ACB) scale,4,23 which can be derived using the medication list recorded and updated at each study visit by study coordinators for study participants. Medications for each participant were classified using the ACB scale as having a low/possible (ACB = 1), medium (ACB = 2), or high (ACB = 3) ACB (Table S1). A time-varying total ACB was calculated as the sum of ACBs for individual medications at each visit for the duration that the participant was in the study. Considering the necessity of anticholinergic medication use for certain PD symptoms, 23 we created an alternative time-varying total ACB measure that excluded amantadine (ACB = 2), quetiapine (ACB = 3), and trihexyphenidyl (ACB = 3) from the scale.
Outcome Definition
The primary outcome of this study was PDD. The DRS-2, recommended by the MDS for assessing global cognition in PD, was used to diagnose dementia.24-26 The DRS-2 score consists of 5 subscales in which points are earned for attention, initiation/perseveration, construction, conceptualization, and memory (score range 0-144, with lower scores indicating impairment). 25 Study participants were considered to have PDD if they had dementia based on a DRS score of ≤130.27-31 Participants were censored upon the earliest occurrence of (1) the study outcome, (2) death, (3) loss to follow-up, or (4) end of dataset.
Covariates
Variable selection for our analytical model was based on literature review, expert opinion, and completeness/availability of data and included the following: age at enrollment, sex, highest education level completed, family history of PD, neuropsychiatric disorders/symptoms (psychosis, anxiety, impulse control disorder [ICD], 21 and depression 22 ), motor symptom burden and treatment (UPDRS-III score and total levodopa equivalent daily dose [LEDD] 32 estimated from PD medications), and use of other central nervous system (CNS)-acting drugs, that, according to the American Geriatrics Society (AGS) Beers Criteria®33 (Table S2), may potentially worsen cognitive function. Neuropsychiatric disorders/symptoms, LEDD, and UPDRS-III scores were treated as time-varying, whereas the remaining variables were measured at study enrollment. Missing values (<2% of selected variables) were imputed based on the last observation carried forward; 34 remaining missing values were excluded during time-varying analyses. Data on the Vascular Risk Factor Questionnaire, Rapid Eye Movement (REM) Sleep Behavior Disorder Questionnaire, magnetic resonance imaging (MRI), and cerebrospinal fluid (CSF) collection via lumbar puncture were not collected for all participants and were excluded.
Statistical Analysis
All analyses were performed using SAS v9.4 and R v4.3.1. We evaluated total ACB categories as a time-varying exposure over follow-up visits for the primary analyses. We first organized the data in a counting process style (i.e., reshaping the data so that each patient had multiple rows containing covariate values across different time intervals), then used Cox proportional hazards models with time-varying covariates 35 to examine the association between demographic and clinical characteristics, anticholinergic medication exposure (based on total ACB categories) and the risk of PDD, adjusting for the remaining covariates. We conducted a similar analysis using the alternative aforementioned time-varying ACB (with medications used to treat PD symptoms removed) as the exposure. We also conducted a post-hoc sensitivity analysis to examine whether there would be an association between time-varying ACB and PDD risk among people with a total ACB of 2+ (indicating at least moderate exposure) at study enrollment.
Results
Characteristics of Participants at Baseline Enrollment Visit (n = 384).

aMissing 6/384 (1.56%).
bMissing 2/384 (0.52%).
cMissing 1/384 (0.26%).
dScore excluded 3 drugs: amantadine, quetiapine, and trihexyphenidyl.
eMedications included in 2019 Beers Criteria® but not listed in ACB scale.
ACB: anticholinergic cognitive burden; IQR: interquartile range; LEDD: levodopa equivalent daily dose; PD: Parkinson disease; UPDRS-III: Unified Parkinson’s Disease Rating Scale Part III.
Associations Between Participants’ Characteristics and Risk of Developing PDD (n = 287).

aAdjusted for remaining covariates.
ACB: anticholinergic cognitive burden; HR: hazard ratio; CI: confidence interval; LEDD: levodopa equivalent daily dose; PD: Parkinson disease, PDD: Parkinson disease dementia; UPDRS-III: Unified Parkinson’s Disease Rating Scale Part III. Bolded values indicate statistical significance.
Associations Between Participants’ Characteristics and Development of PDD Using Alternative a ACB Categories (n = 287).
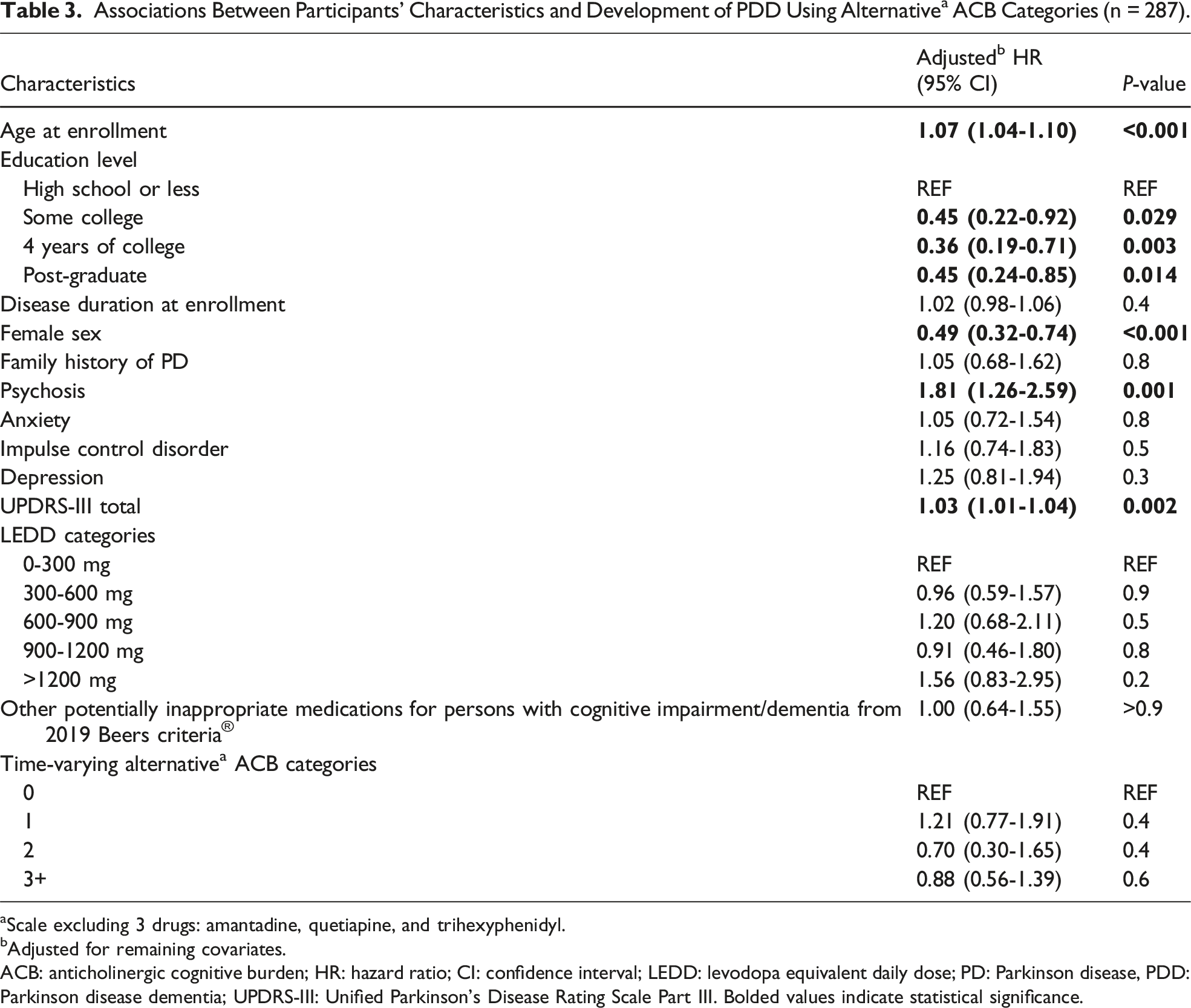
aScale excluding 3 drugs: amantadine, quetiapine, and trihexyphenidyl.
bAdjusted for remaining covariates.
ACB: anticholinergic cognitive burden; HR: hazard ratio; CI: confidence interval; LEDD: levodopa equivalent daily dose; PD: Parkinson disease, PDD: Parkinson disease dementia; UPDRS-III: Unified Parkinson’s Disease Rating Scale Part III. Bolded values indicate statistical significance.
Discussion
Preventing or delaying dementia is a priority for persons living with PD. In this study of a single-center sample of individuals enrolled in a natural history study of PDD, we examined whether anticholinergic medication use was associated with PDD. Our findings confirmed known associations between age, sex, motor symptom severity, psychiatric symptom burden, and PDD risk, but we did not detect a significant drug-disease interaction when using a standard anticholinergic drug exposure tool.
We found that age was associated with an increased risk of PDD. Age is an independent risk factor for all forms of dementia, including PDD. 36 In our cohort, female sex appeared to be protective against PDD development. This may be attributed to the selective nature of the cohort, which we believe consisted of female participants with above-average access to subspecialist health care and better management of risk factors for other types of dementia. Prior studies have found that males living with PD tend to have worse cognitive abilities37,38 and have a higher rate of PDD. 39 Although some studies found no association between sex and PDD after adjustment for potential confounding factors such as age, history of dementia, smoking, and number of siblings,40,41 a meta-analysis reported that male sex is a risk factor for PDD before confounding adjustment. 42 These findings collectively support the hypothesis that the relationship between biological sex and dementia may differ across dementia types. While Alzheimer’s disease is consistently more prevalent in women,43-45 the development of PDD appears to follow a different pattern, suggesting that much more remains to be understood about the role of biological sex in PDD. Our finding that higher levels of education were protective against dementia was also consistent with previous literature, not only among PD patients 42 but also in the general population,46-50 with a more consistent association occurring when the years of education reflected cognitive capacity. 49
Higher UPDRS-III scores were associated with an increased risk of developing PDD in our cohort, consistent with prior studies of PDD risk.1,42,51 We did not observe that increasing LEDD is associated with PDD, which may reflect the clinical practice of reducing levodopa as PD progresses and drug response declines. Antiparkinsonian medications can have harmful effects on cognition depending on striatal basal dopamine levels and tend to cause more intolerable adverse effects without sustained motor benefit at advanced stages.52,53
Our deeply-phenotyped cohort allowed us to examine a number of psychiatric nonmotor symptoms. Psychosis was associated with an increased risk for PDD in our cohort. Visual hallucinations1,42,54,55 or psychosis56-58 have been previously described as risk factors for developing PDD, and psychosis and dementia often co-exist.59,60 Depression61-63 and anxiety 64 have been consistently linked to dementia risk in the general adult population. The data on mood or ICD and cognitive decline in the PD population, however, have been mixed. One natural history study reported that persons with PD and depression were nearly twice as likely to be diagnosed with mild cognitive impairment within four years.65,66 Using a more precise cognitive instrument, we found depression (measured using the GDS-15) was associated with PDD in the subsample of NIA U19 Center’s participants with a higher ACB at enrollment. Anxiety has been reported as a risk factor for cognitive impairment, verbal memory impairment, 67 and longitudinal decline in verbal and visual learning performance among PD patients. 68 However, we did not observe an association between anxiety (measured using the Cognition/Behavior Questionnaire) and PDD risk. Some studies have found that PD patients with ICD have deficiencies in cognitive tasks localizing to frontal regions and visual-spatial planning; 69 others have reported no between-group differences in working memory tasks,69,70 but prior studies have not found a relationship between ICD and PDD. 71 As with depression, we found an association between ICD and PDD risk only in persons with greater baseline ACB. Taken together, these data highlight the complex roles psychiatric symptoms and disorders play in the propagation, recognition, and measurement of cognitive decline in persons living with PD.
Several studies have reported an association of the use of anticholinergic medications with the risk of all-cause dementia,3-9,14-17 We hypothesize that a greater risk of cognitive impairment from central cholinergic blockade is possible in the PD population (as compared with the general older adult population) due to PD related loss of cholinergic neurons. Cholinergic degeneration begins in the prodromal phase of PD, contributing to cognitive and gait dysfunction and eventually to freezing and dementia. 72 We did not observe an association between higher total ACB categories and incident PDD. Exposure misclassification is a potentially strong reason for our findings. There are no standard pharmacologic assays to detect anticholinergic activity in the CNS, despite the availability of serum anticholinergic assays.73-76 Imaging-based biomarkers for anticholinergic activity have not been developed, even though anticholinergic use was associated with cortical and ventricular abnormalities (as well as cognitive decline) in the Alzheimer’s Disease Neuroimaging Initiative (ADNI) and the Indiana Memory and Aging Study (IMAS) cohorts. 77 Without standardized biological tools, multiple scales to assess anticholinergic burden via application to existing data have been developed, including the widely used ACB scale. However, the scales have low to moderate concordance between them, and medications are often inconsistently included in one scale but not another. 23 The available anticholinergic exposure measures were also not developed in populations with underlying cholinergic deficits, as found in PD. Ceiling effects in the PD population may be observed, with toxicity occurring at a lower anticholinergic level than captured by current scales. Of note, the anticholinergic effect on cognition (AEC) score 78 also accounts for the central anticholinergic effects of anticholinergic medications based on literature reviews of their ability to cross the blood-brain barrier, which could make it more relevant for future research in this population. However, there remains limited in vivo data on these effects, particularly concerning drug-drug interactions and the evolving pharmacokinetic and pharmacodynamic properties in older adults. Our findings underscore the need for biological tools to assess central anticholinergic activity or for the development and validation of pharmacoepidemiology tools for PD research that consider critical factors such as dosing, CNS penetration, and drug-drug interactions, allowing characterization of the pharmacological exposome for use in randomized controlled trials and observational cohort studies.
Strengths of our study include a well-phenotyped cohort that had longitudinal assessments. Several weaknesses should be considered in the interpretation of these findings. Data on genes and health behaviors (eg, APOE-ε4, exercise) associated with dementia risk, as well as comorbidities (e.g., cardiovascular, cerebrovascular, or metabolic diseases), were not readily available or routinely collected as aforementioned (partly due to the COVID-19 pandemic). Moreover, medication data were collected by study personnel based on participants’ self-reports, and we did not have information on the duration of exposure to anticholinergic or other potentially inappropriate medications prior to study enrollment, nor on medication doses. As a result, we were unable to assess cumulative anticholinergic doses, which may introduce potential residual confounding. Our small sample only included people from 1 clinical center, where most patients cared for were White, highly educated, and had above-average income; thus, our findings are not generalizable to the average older adult population with PD. We used DRS-2 scores as a proxy for dementia diagnosis; our score cut-off was conservative as compared with other studies,27-31 resulting in potential outcome misclassification.
Conclusions
In this single-center sample of individuals with PD, we found that older age, psychosis, and greater motor symptom burden were positively associated with the risk of PDD development, aligning with previous studies that have identified these factors as predictors for dementia. Alternatively, we observe that female sex and higher education level were protective against PDD. While we did not see an association between ACB and PDD risk, this finding should be interpreted cautiously. Careful weighing of known anticholinergic medication risks vs. benefits should still be applied in this vulnerable patient population, which is usually also elderly, and deprescribing opportunities should be implemented whenever possible. New pharmacological exposome research tools are needed to understand the potential CNS effects of anticholinergics on PD cognitive function and clinical trajectory.
Supplemental Material
Supplemental Material - Impact of Anticholinergic Burden and Clinical-Demographic Characteristics on Incident Dementia in Parkinson Disease
Supplemental Material for Impact of Anticholinergic Burden and Clinical-Demographic Characteristics on Incident Dementia in Parkinson Disease by Thanh Phuong Pham Nguyen, Dylan Thibault, Shelly L. Gray, Daniel Weintraub, and Allison W. Willis in Journal of Geriatric Psychiatry and Neurology.
Footnotes
Acknowledgements
The authors thank Ms. Eugenia Mamikonyan and Ms. Julia Gallagher for their support in creating the study datasets.
Declaration of Conflicting Interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Pham Nguyen was a member of the Junior Investigator Intensive Program of the 2022-2023 U.S. Deprescribing Research Network, which was funded by the National Institute on Aging (Grant #R24AG064025) and receives support from the National Institute of Health (Grants #1RF1NS132673-01, #R01NS099129, #R01AG02515215, #R01AG06458903) and Acadia Pharmaceuticals Inc, unrelated to this project. Dr Weintraub receives support from the National Institutes of Health (Grant #R01NS099129) and has also received research funding or support from Michael J. Fox Foundation for Parkinson’s Research, Alzheimer’s Therapeutic Research Initiative (ATRI), Alzheimer’s Disease Cooperative Study (ADCS), the International Parkinson and Movement Disorder Society (IPMDS); honoraria for consultancy from Acadia Pharmaceuticals Inc, Aptinyx, Biogen, Bracket, CHDI Foundation, Clintrex LLC, Enterin, F. Hoffmann-La Roche Ltd, Ferring, Promentis, Sunovion, and Takeda; and license fee payments from the University of Pennsylvania for the QUIP and QUIP-RS. Dr Willis receives financial support from the National Institutes of Health (Grants #1RF1NS132673-01, #R01NS099129, #K24AG075234, #RF1NS132673), the Parkinson’s Foundation, Acadia Pharmaceuticals Inc, and the University of Pennsylvania. All other authors declared no competing interests in this work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is funded by a grant from the National Institute of Neurological Disorders and Stroke (#R01NS099129).
Ethical Statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
