Abstract
Background. Ten percent of stroke survivors develop dementia, which increases to more than a third after recurrent stroke. Other survivors develop less severe vascular cognitive impairment. In the general population, depression, and diabetes interact in predicting dementia risk, and they are both prevalent in stroke. Objective. To assess the cumulative association of comorbid depressive symptoms and type 2 diabetes with cognitive outcomes among stroke survivors. Methods. Multicenter observational cohort study of people within 6 months of stroke. Depression and cognitive status were screened using the Center for Epidemiological Studies Depression (CES-D) scale and the Montreal Cognitive Assessment (MoCA), respectively. Processing speed, executive function and memory were assessed using the Trail Making Test parts A and B, and the 5 Word Delayed Free Recall task. Results. Among 342 participants (age 67.0 ± 13.5 years, 43.3% female, 46 ± 35 days poststroke), the prevalence of type 2 diabetes was 32.2% and depressive symptoms (CES-D ≥16) were found in 40.6%. Diabetes and depressive symptoms increased the risk of severe cognitive impairment (MoCA <20) with adjusted odds ratio (OR) 2.12 (95% confidence interval [CI] 1.20-3.74, P = .010) for 1 comorbidity and OR 3.18 (95% CI 1.26-8.02, P = .014) for both comorbidities. Associated cognitive deficits included executive function (F1, 168 = 3.43, P = .035) but not processing speed (F1, 168 = 1.86, P = .16) or memory (F1, 168 = 0.82, P = .44). Conclusions. Diabetes and depressive symptoms were associated cumulatively with poorer cognitive screening outcomes poststroke, particularly deficits in executive function. Having 1 comorbidity doubled the odds of screening for severe cognitive impairment, having both tripled the odds.
Introduction
With an increasing prevalence of individuals living with the effects of stroke, 1 impediments to recovery and independence are increasingly important public health concerns. Ten percent of stroke survivors develop dementia within a year after their first stroke, and more than a third will have dementia after recurrent stroke. 2 Depression and diabetes are risk factors for both stroke3,4 and dementia5,6 in the general population, suggesting that these comorbidities may also be related to cognitive status poststroke. While some develop vascular and mixed dementias, other stroke survivors develop milder forms of vascular cognitive impairment (VCI) that do not impact instrumental activities of daily living and therefore do not meet dementia criteria; however, they may portend further cognitive and functional decline. 7
It is estimated that between 25% and 50% of stroke survivors will experience depressive symptoms poststroke, 8 which predict functional decline and increased risks of mortality and recurrent stroke. 3 Poststroke depressive symptoms have been associated with deficits in memory, executive function, and attention/psychomotor processing speed.8-10 Diabetes has also been associated with mortality and poorer functional outcomes in stroke,11-16 with poststroke dementia, 2 and with deficits in multiple cognitive domains in populations not selected for stroke 17 ; however, the relationship between type 2 diabetes and cognitive screening in the recovery period following stroke has not been assessed.
In a Danish national cohort study, depression and diabetes interacted to increase the risk of dementia, 18 suggesting that the risk of screening for significant cognitive impairment in those living with stroke may also be additively associated with these comorbidities. Therefore, the present study examines depressive symptoms and type 2 diabetes cumulatively, summarized using a parsimonious comorbidity index. It is hypothesized that this index will be associated with stroke severity and cognitive status, screened concurrently within 6 months of stroke. Measures of psychomotor processing speed, executive function and verbal memory, each of which have been associated with depression, diabetes, and cerebrovascular disease, 9 are explored. Given that VCI has been most consistently associated with deficits in executive function, 7 it is hypothesized the comorbidity index will be associated particularly with poorer performance in this domain.
Methods
Participants and Study Details
Participants were part of the Canadian Partnership for Stroke Recovery Rehabilitation Affiliates Study, a multicenter investigation designed to characterize physical and cognitive recovery after stroke in Ontario, Canada. 19 The present analysis reports on a cross-sectional cohort of these participants who presented within 180 days of acute stroke. The criteria for inclusion in this study were: a primary diagnosis of ischemic or hemorrhagic stroke, ability to speak and understand English, eligibility for stroke rehabilitation, screening for depression and cognitive status, and assessment for stroke severity as part of the Rehabilitation Affiliates Study. Participants with type 1 diabetes and participants beyond 180 days poststroke were excluded. This study was approved by local research ethics boards, and participants provided informed consent.
Depressive Symptoms
Depression was screened using the Center for Epidemiological Studies scale for Depression (CES-D), a self-report instrument that assesses the presence and severity of symptoms over the past week. A CES-D score ≥16 is a sensitive and reliable screen for poststroke depression, 20 and it is not susceptible to confounding due to the presence of other medical comorbidities in the screening for depressive episodes. 21
Cognitive Assessments
The Montreal Cognitive Assessment (MoCA) was selected as a validated screen for cognitive impairment after stroke. 22 The 30-point MoCA test score was adjusted for education, and a cutoff below 20 was chosen to reflect severe impairment, as per standard practices.23,24 Cognitive subdomains of attention and psychomotor processing speed, executive function, and verbal memory were assessed in a cursory sense using the Trail Making Test parts A (TMT-A) and B (TMT-B),25,26 and a 5 Word Delayed Free Recall task (5WDFR), 27 respectively. The National Institutes of Health Stroke Scale (NIHSS) was used to assess stroke severity. 28
Statistical Analyses
Categorical variables were summarized using counts and percentages, while continuous variables were summarized using means and standard deviations or medians and interquartile ranges, as appropriate.
The burden of depressive symptoms and diabetes was summarized as a comorbidity index. A score of 1 was given to each participant reaching a CES-D threshold of ≥16, as appropriate to screen for poststroke depression, 20 and a separate score of 1 was given to each participant with a confirmed history of type 2 diabetes. The comorbidity index was derived from the sum of these variables (range: 0-2).
Univariate analyses of covariance (ANCOVA) were used to assess differences in continuous outcome measures associated with the comorbidity index. The comorbidity index was also used in logistic regression models to predict the risk of screening for severe cognitive impairment. These models were adjusted for age, gender, days since stroke, use of antidepressant and hypoglycemic medications, hypertension, dyslipidemia, and NIHSS score, which were chosen as covariates a priori. The MoCA was prespecified as the primary outcome measure, with Bonferroni correction for 2 comparisons (ANCOVA and logistic regression). Relationships between the comorbidity index and domain-specific cognitive tests were explored in multivariate ANCOVA models.
All analyses were carried out in SPSS Statistics (version 22.0.0.0, IBM Corporation). Preplanned subgroup analyses were conducted in men and women and in participants older than and younger than 65 years, since diabetes and depression may differentially affect stroke risk and stroke outcomes across these strata.4,29,30 To address the possibility of additional confounding, analyses post hoc included demographics, stroke characteristics, vascular risk factors, and concomitant medications found to be associated with diabetes, depressive symptoms, or cognitive outcomes (P < .05) in chi-square or t tests, as appropriate.
Results
Cohort Characteristics
Of the 453 participants, 425 were assessed for stroke severity within 180 days poststroke, and of those, 342 were screened with both the CES-D and the MoCA and included in this study (January 2009 to December 2013); their demographics, stroke characteristics, vascular risk factors, and concomitant medications at the time of assessment are summarized in Table 1.
Participant Characteristics (n = 342).

Abbreviations: BMI, body mass index; NIHSS, National Institute of Health Stroke Scale.
Depressive Symptoms and Type 2 Diabetes
The mean CES-D score was 15.0 ± 10.7 (n = 342), with 40.6% screening for significant depressive symptoms. Of those who screened positive, 35.3% were using an antidepressant medication. Participants with significant depressive symptoms tended to be younger (F1, 341 = 5.28, P = .022) and less likely to be married (χ2 = 4.43,P = .035).
Of those with a history of type 2 diabetes mellitus, 34.8% were using an insulin preparation, 65.2% were using a biguanide (metformin) and 23.2% were using a sulfonylurea. Participants with diabetes were more likely to have hypertension (χ2 = 8.70, P = .003) and to be using a calcium channel blocker (χ2 = 5.14, P = .023), an angiotensin receptor blocker or an angiotensin converting enzyme inhibitor (χ2 = 16.85, P < .001), more likely to have hyperlipidemia (χ2 = 19.05, P < .001) and to be using a statin (χ2 = 4.04, P = .044), less likely to be Caucasian (χ2 = 7.61, P = .006), and tended to have higher body mass index (BMI) (F1, 317 = 11.71, P = .001).
There were 42 participants (12.3% of this cohort) who screened for depression and had a history of type 2 diabetes, 167 participants (48.8%) who had 1 comorbidity alone, and 133 participants (38.9%) who had neither comorbidity. Diabetes and having a CES-D ≥16 were not more or less likely to co-occur than might be expected by chance (χ2 = 0.68, P = .409).
Stroke Severity
In an ANCOVA model, the diabetes and depressive symptoms comorbidity index was associated with higher NIHSS scores (F2, 341 = 3.71, P = .025). The use of metformin was associated with lower NIHSS scores (F1, 341 = 4.82, P = .029), while hypertension (F1, 341 = 3.96, P = .047) and antidepressant use (F1, 341 = 4.22, P = .041) were associated with higher NIHSS scores.
Comorbidity and Screening for Cognitive Status
The mean MoCA score was 20.6 ± 5.3, with 39.8% screening for significant cognitive impairment. In an ANCOVA model (adjusted R2 = .168), the comorbidity index was associated with poorer MoCA scores (F2, 341 = 4.60, P = .011; Figure 1A), as was NIHSS score (F1, 341 = 30.63, P < .001) and age (F1, 341 = 26.51, P < .001).
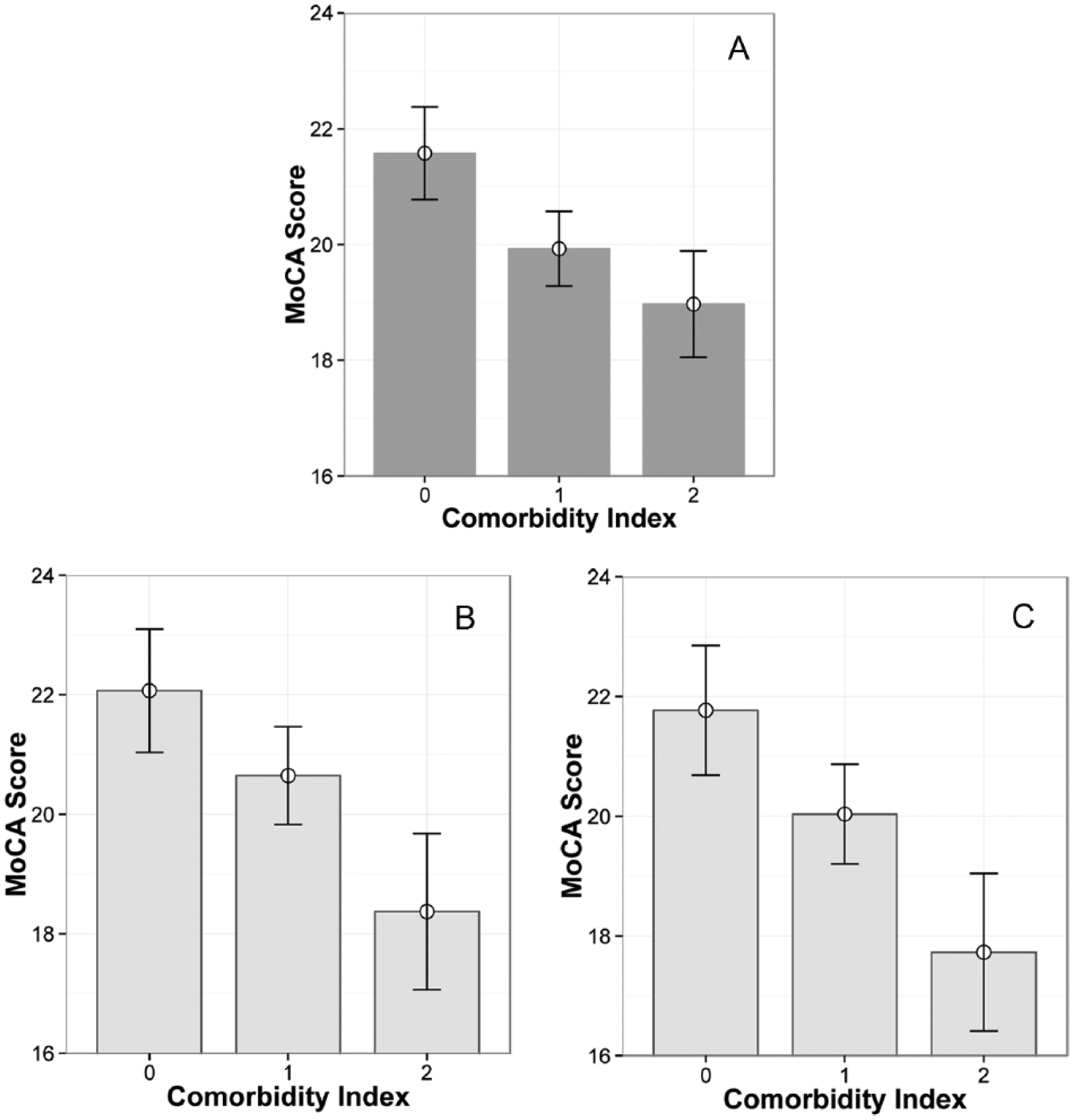
Relationship between type 2 diabetes and depression comorbidity and cognitive status screened poststroke. (A) Montreal Cognitive Assessment (MoCA) scores in the entire cohort (n = 342). (B) MoCA scores in men only (n = 194). (C) MoCA scores in participants older than 65 years only (n = 185). MoCA scores are presented as marginal means ± standard error estimated in ANCOVA models. Subgroups are shown in light grey boxes. Comorbidity is indexed as 0 (neither depressive symptoms nor type 2 diabetes), 1 (either depressive symptoms or type 2 diabetes), or 2 (depressive symptoms and type 2 diabetes).
In a logistic regression model (Nagelkerke R2 = .223) predicting MoCA <20, the comorbidity index was associated with adjusted odds ratios (ORs) of 2.12 (95% confidence interval [CI] 1.20-3.74, P = .010) for 1 comorbidity and 3.18 (95% CI 1.26-8.02, P = .014) for both comorbidities. Older age (OR 1.04, 95% CI 1.02-1.06, P < .001), NIHSS score (OR 1.16, 95% CI 1.08-1.24, P < .001), dyslipidemia (OR 1.74, 95% CI 1.01-2.97, P = .044) and female gender (OR 1.72, 95% CI 1.05-2.80, P = .030) also predicted increased risk of screening cognitively impaired.
Comorbidity and Cognitive Domains
Of the participants who were administered the TMT-A, TMT-B, and 5WDFR tasks (n = 217), 2 could not complete the TMT-A task and 33 could not complete the TMT-B task. Among completers, the mean TMT-A time was 72 ± 42 seconds and the mean TMT-B test time was 214 ± 144 seconds. Participants remembered 1.82 ± 1.65 of 5 words in the 5WDFR task.
In an adjusted multivariate model, the diabetes and depressive symptoms comorbidity index was associated with poorer performance in the TMT-B (F1, 168 = 3.43,P = .035) but not in the TMT-A (F1, 168 = 1.86, P = .160) or 5WDFR (F1, 168 = 0.82, P = .440). In this model, stroke severity and less time since stroke also predicted poorer TMT-A and TMT-B scores. Only older age predicted poorer memory performance.
Sensitivity Analysis by Gender
The estimated risk of screening MoCA <20 associated with diabetes and depressive symptoms comorbidity was larger in men (OR 2.77, 95% CI 1.26-6.13, P = .012 for 1 comorbidity and OR 6.69, 95% CI 1.79-24.99, P = .005 for both comorbidities) when compared with women (OR 1.24 95% CI 0.67-2.23, P = .492). The comorbidity index was associated with lower MoCA scores in men (F2, 193 = 3.59, P = .030; Figure 1B) but not in women (F2, 147 = 1.82, P = .166); however, it was associated with greater stroke severity in women (F2, 147 = 3.78, P = .025) but not in men (F2, 193 = 0.699, P = .498). Without stroke severity in the models, the comorbidity index predicted MoCA scores in both men (F2, 193 = 3.97,P = .020) and women (F2, 147 = 3.34, P = .039).
Sensitivity Analysis by Age
The comorbidity index was associated with MoCA scores in older participants (F2, 184 = 3.970, P = .021; Figure 1C) but not in younger participants (F2, 156 = 1.65, P = .196), with estimated ORs for MoCA <20 of 1.86 (95% CI 0.875-3.96, P = .107) for 1 comorbidity and 4.48 (95% CI 1.03-19.48,P = .046) for both comorbidities among participants older than 65 years. The comorbidity index was associated with stroke severity in older participants (F2, 184 = 3.56, P = .031) but not in younger participants (F2, 156 = 1.33, P = .268).
Post Hoc Models
In models fully adjusted for all potential confounders identified post hoc, the comorbidity index remained associated with poorer MoCA scores (F2, 317 = 3.90, P = .021) and with increased risk of screening cognitively impaired (adjusted OR 2.16, 95% CI 1.16-4.00, P = .015 for 1 comorbidity, and OR 2.81, 95% CI 1.01-7.82, P = .049 for both comorbidities). The use of an angiotensin receptor blocker or an angiotensin converting enzyme inhibitor was associated with less risk of screening cognitively impaired (OR 0.457, 95% CI 0.25-0.85, P = .013).
Discussion
Here we show that diabetes and depressive symptoms are associated with poorer cognitive performance among people living with stroke (Figure 1A). These 2 comorbidities were not especially likely to co-occur in this poststroke cohort, even though they have been shown to be reciprocal risk factors in the general population.31,32 The relationship between cognition and these comorbidities in the current cohort of stroke survivors appeared to be cumulative, with 1 comorbidity approximately doubling the odds of a cognitive screen suggesting severe impairment, and both comorbidities approximately tripling the odds. The cumulative nature of the relationship between these comorbidities and cognitive symptoms was particularly prominent in men (Figure 1B) and in older participants (Figure 1C).
Previously, the impact of type 2 diabetes on VCI severity after stroke has not been explored extensively. Providing context, however, 1 stroke study found that patients with diabetes had poorer neurological outcomes and suffered more stroke-related deaths than those without, even though initial stroke severity, lesion size, and lesion location were comparable between the 2 groups, and diabetes was a risk factor for post-stroke dementia in a large meta-analysis. 11 Those findings suggested the need to explore relationships between diabetes and cognitive status among those entering rehabilitation in the recovery period poststroke. On the other hand, depression is a well-established risk factor for poststroke cognitive impairment, including problems with memory, executive function, and attention/psychomotor processing speed. 9 In the current study, the cumulative burden of depressive symptoms and diabetes was most evident using the TMT-B, a test of executive function (complex attention and set-shifting ability that relies on frontal-subcortical communication), reflecting a commonly salient and somewhat specific feature of VCI. 7 In contrast, performance on the TMT-A, a measure of psychomotor processing speed, as well as the 5WDFR memory task, were not significantly related to the cumulative burden of these comorbidities. The findings suggest that the comorbidities may worsen the presentation of VCI poststroke, and their clinical importance is underscored by a previously identified relationship between TMT-B scores and poststroke mortality. 33
In this study, 40.6% presented with CES-D ≥16, which is high, but consistent with previous studies that used psychiatric diagnoses, in which the overall prevalence of major or minor depression was 41.2%. 8 Among prospective, consecutive recruitment studies, the pooled prevalence of depression was similarly elevated (28% to 35%). 34 In the present study, depressive symptoms were frequently untreated. In previous studies, the resolution of depressive symptoms was associated with cognitive improvement during stroke recovery, 10 justifying our consideration of concurrent depressive symptoms as opposed to relying on a history of premorbid psychiatric illness to estimate the current burden of depression; however, including a psychiatric history and the timing of antidepressant use might have been informative.
Type 2 diabetes and depressive symptoms were associated with increased stroke severity; however, this relationship was seen only in women, which would be consistent with gender-specific effects of diabetes on the risk of stroke and on stroke outcomes previously observed. 4 Because of this association, stroke severity was entered as a covariate in the analyses of cognitive outcomes. In women, the relationship between the comorbidity index and cognition appeared to depend on the presence of greater stroke severity. In men, the comorbidity index was associated with poorer poststroke cognitive status independent of stroke severity. In addition, the cumulative nature of the burden of depression and diabetes was more pronounced in men, and in participants older than 65 years. Although causation cannot be inferred, it is possible that male sex and older age may confer neural vulnerability to these comorbidities, especially when they occur together.
In the current analysis, the use of metformin was associated with lower stroke severity when controlling for the use of other antidiabetic medications. This observation is consistent with that of a previous prospective study in which metformin use prior to stroke was associated with less severe stroke and a trend toward better outcomes at 90 days, as compared with the use of no antidiabetic medication. 35 Although in some previous studies not selected for stroke, metformin use has been associated with reduced risk of cognitive decline,36,37 we found no cross-sectional associations between medication use and cognitive screening results, except for antihypertensive agents acting on the angiotensin system, which were associated with better cognitive performance.
Diabetes and depressive symptoms were cumulatively associated with cognitive outcomes, particularly in subgroups of older and male participants; however, larger sample sizes would be needed to formally test a statistical interaction. 18 As a potential limitation, poststroke depression and poststroke cognitive status were screened rather than formally diagnosed; however, the prevalence of depression screened was consistent with studies that used formal diagnoses. 8 As a limitation, premorbid depression diagnoses and cognitive screening were not available, precluding comment on the relative importance of premorbid vs. comorbid depressive symptoms and effects of stroke 38 ; however, in some studies, the resolution of poststroke depressive symptoms was associated with cognitive improvement 10 suggesting that symptoms present at the time of cognitive screening are likely to be important in the context of recovery. Diabetes was ascertained through medical history and patient interview; duration and complications of diabetes, and prediabetes among nondiabetic subjects, were not ascertained and therefore the burden of this comorbidity may have been underestimated. Psychomotor processing speed, executive function, and memory assessments were limited to single tests or screens that do not comprehensively assess these domains, although the Trail Making Tests are sensitive, specific and clinically meaningful. Although all included participants had adequate acuity to perform testing, interference with cognitive testing due to diabetic complications, or interference with depression screening due to cognitive impairment, cannot be strictly ruled out. While some medications were associated with stroke severity, functional capability and cognitive status, cross-sectional associations do not imply causality, and therefore such inferences are beyond the scope of this study.
Future studies probing physiological39-41 and behavioral 42 factors that mediate the observed relationships may inform strategies to facilitate cognitive recovery. For instance, further study of how these comorbidities are related to inflammation, oxidative stress and revascularization in peri-infarct cortex may yield further insights into neural recovery and repair processes. 43 If their effects are truly additive, studies might seek to identify mechanistic overlap in the postischemic brain. As a corollary, investigating effects of specific treatments for these comorbidities may inform optimization of pharmacotherapies for these comorbidities that also promote cognitive recovery.44-46
In conclusion, 61.1% of this cohort entering rehabilitation within 6 months of mild to moderate stroke were affected by type 2 diabetes and/or depressive symptoms, which were associated cumulatively with poorer cognitive outcomes. The burden of these comorbidities was related to cognition differently in men and women; the relationship appeared to be mediated by stroke severity in women, but not in men. Of the cognitive domains assessed, executive function was particularly involved. In a utilitarian way, the findings emphasize the importance of considering the potential contributions of these comorbidities to cognitive outcomes when planning neurorehabilitation. The comorbidity index appears to be clinically relevant, particularly in men and in older survivors of stroke.
Footnotes
Acknowledgements
The authors would like to thank the staff, participants and investigators of the Rehab Affiliates Study, Dr William E. McIlroy for providing data, Cynthia Danells and Ellen Cohen for help retrieving and interpreting the data, and Drs Peter Karagiannis and Benjamin I. Goldstein for editing the manuscript.
Declaration of Conflicting Interests
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Canadian Partnership for Stroke Recovery funded this study.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding support was provided by the Canadian Partnership for Stroke Recovery. Dr Swardfager gratefully acknowledges support from the Hurvitz Brain Sciences Program, Sunnybrook Research Institute, and the Department of Psychiatry, Sunnybrook Health Sciences Centre.
