Abstract
Introduction
Cognitive impairment is common after stroke 1 and is associated with worse recovery, 2 dementia, 3 worse functional outcome, 4 and poor survival. 5 It is also a risk factor for depression and reduced quality of life, 6 and hence interferes with the rehabilitation process. One of the most disabling cognitive disorders is aphasia, which occurs in 15% 7 to 38% 8 of stroke patients and results in a reduction of communicative ability.
Numerous stroke studies have investigated the prevalence, 9 course,2,10 and predictive value5,11 of cognitive impairment after stroke. However, information on cognitive impairment in aphasic patients is limited. Most studies have explicitly excluded all patients with aphasia9,10,12 or those with severe aphasia5,13 because communication problems interfere with standard cognitive testing. Two studies have reported on the cognitive performance of aphasic patients,14,15 but the number of patients was small and not all cognitive domains were examined.
The aim of this observational prospective follow-up study was to investigate the prevalence and course of nonlinguistic cognitive impairments in a large cohort of aphasic patients at 3 months and 1 year after stroke, and the association between aphasia, nonlinguistic cognition, and functional outcome.
Methods
Patients
Patients with aphasia due to a first-ever stroke were recruited from the stroke units of 17 hospitals in the Netherlands. They were screened by the local neurologist and speech-language therapist for the Sequential Prognostic Evaluation of Aphasia after stroKe (SPEAK) study from June 2007 to June 2009. Eligible patients were adult Dutch native/near-native speakers with an intracerebral hemorrhage or infarction who were testable with the ScreeLing16,17 within 14 days after stroke. From a clinical point of view, patients were considered testable when they were sufficiently alert and fit to tolerate at least 15 minutes of the ScreeLing assessment. The 3 linguistic components of the ScreeLing had to be completed within 2 consecutive days. Included were patients with aphasia as confirmed by the speech-language therapist and in addition a score below the cutoff point of the Token Test (ie, <29) 18 and/or the ScreeLing (ie, <68). 17 Patients with prestroke dementia (suspected or confirmed), psychiatric disorder, severe dysarthria, developmental dyslexia, severe perceptual disorders of vision and hearing, or illiteracy were excluded.
Patients and/or their close relatives gave written informed consent while the patient was admitted to hospital. The follow-up assessments were conducted in the subsequent setting, that is, nursing home, rehabilitation center, or at home.
This study was approved by the Medical Ethics Committees of Erasmus MC University Medical Center and of the participating centers.
Follow-up Assessments
Demographic data, collected at baseline, included age, sex, education level, and handedness. Neurologic data included stroke type, stroke localization, and stroke-related disability as measured by the Barthel Index,
19
which is a 20-point scale to assess the degree of independence in general daily life activities (eg, eating, getting dressed), and were also collected at baseline. At 3 months and at 1 year after stroke, nonlinguistic cognitive abilities were examined within 4 domains: (
The raw test scores were transformed into standardized scores (
Based on the number of impaired non-linguistic cognitive domains, patients were classified into 2 groups: severe cognitive impairment, that is, ≥2 impaired cognitive domains out of 4, and mild cognitive impairment, that is, <2 impaired cognitive domains.
Language was assessed at 3 months and 1 year after stroke by means of the following:
The ScreeLing, designed to detect aphasia in acute stroke patients, and to reveal deficits on the 3 core linguistic components, that is, semantics, phonology, and syntax.16,17,35 Each linguistic component is examined with 4 well-known tasks: semantic tasks include word–picture matching, identifying semantically anomalous sentences, verbal semantic association, and odd-word out; phonology comprises tasks for repetition, reading aloud, judging same/different spoken word pairs, and matching first phoneme of a spoken word with the grapheme; syntax comprises sentence–picture matching, wh-questions, identifying syntactic incorrect sentences, and sentence completion. 17 The ScreeLing correlates significantly with the Aphasia Severity Rating Scale (ASRS) of the Boston Diagnostic Aphasia Examination 36 and the Token Test, 18 indicating good concurrent validity. 17
Spontaneous speech, elicited in a 10-minute semistandardized interview on 4 topics, and evaluated with the ASRS of the Boston Diagnostic Aphasia Examination 36 as a measure of verbal communication.
The Token Test, 36-item raw scores version, to measure the presence and the severity of aphasia. 18 At baseline, this test was used to describe initial aphasia severity ranging from “very severe” to “no aphasia.” 18
Patients were classified into 2 groups: persisting aphasia, that is, Token Test score <29 and/or ASRS score <5, and recovered, that is, Token Test score ≥29 and ASRS score 5.
For functional outcome we used the modified Rankin scale (mRS), a measure of daily life functioning after stroke. 37 A previous study reported that this measure was used as a dichotomous scale in most stroke studies. 38 Poor functional outcome was defined as mRS 3 to 5 indicating functional dependence and good functional outcome as mRS 0 to 2 representing independence. 38 Information on depression was monitored during the complete follow-up period by reviewing medical records and interviewing patients and often also their close relatives. Patients treated with antidepressants were categorized as being depressed.
Statistical Analysis
First, we compared the nonlinguistic cognitive domain scores of our cohort at 3 months and 1 year after stroke with norm scores, that is,
Second, we compared patients with persisting aphasia with those who had recovered at both assessments. To compare the nonlinguistic cognitive domain scores between these 2 groups, we used independent
Third, we compared cognitive impairment with respect to functional outcome with the Mann–Whitney test. We performed univariable and multivariable logistic regression analysis to evaluate the relation between cognitive impairment and functional outcome at 1 year after stroke independently of aphasia severity. Finally, to evaluate the prevalence of depression in the different subgroups of patients we conducted frequency analyses.
A value of
Results
The baseline characteristics of the 147 included patients are shown in Table 1.
Baseline Characteristics of the 147 Included Patients With Aphasia.
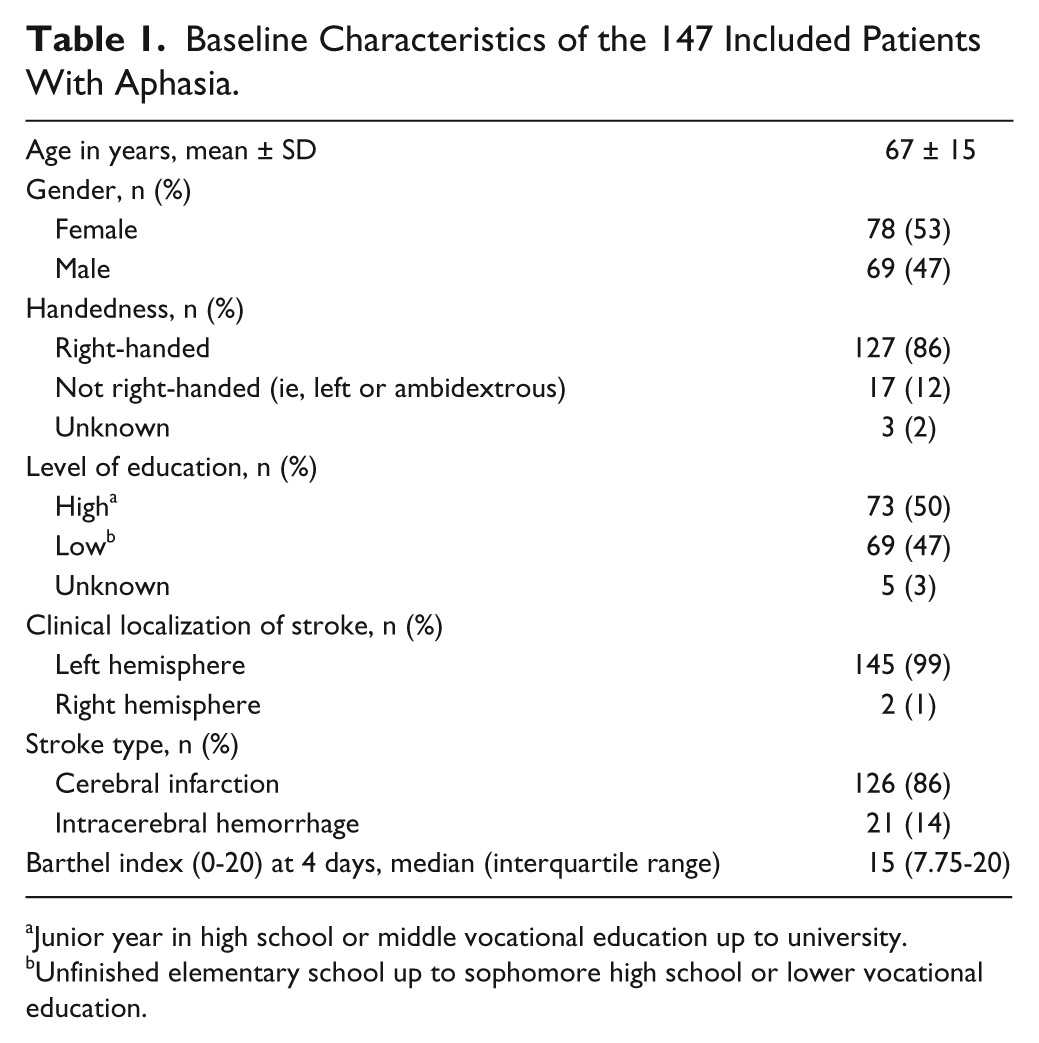
Junior year in high school or middle vocational education up to university.
Unfinished elementary school up to sophomore high school or lower vocational education.
As measured by the Token Test at baseline, 28% of the patients had very severe, 22% severe, 26% moderate, 15% mild, and 9% had no aphasia. Of all patients, 30% had isolated aphasia, 60% had a leg paresis and/or arm paresis, and 27% had hemianopia in addition to aphasia. We were able to assess 125 patients at 3 months (mean 92 ± 5.8 days) and 115 at 1 year (mean 372 ± 22 days; Figure 1). The patients who were lost to follow-up did not differ significantly from the rest of the cohort in terms of age, sex, handedness, education level, stroke type, and the baseline Barthel Index score. A total of 35 out of 125 patients failed to perform or complete the TMT A and B, and the WCST at 3 months, and 27 out of 115 at 1 year. These patients were significantly older, had lower baseline Barthel Index scores, and had a lower education level (
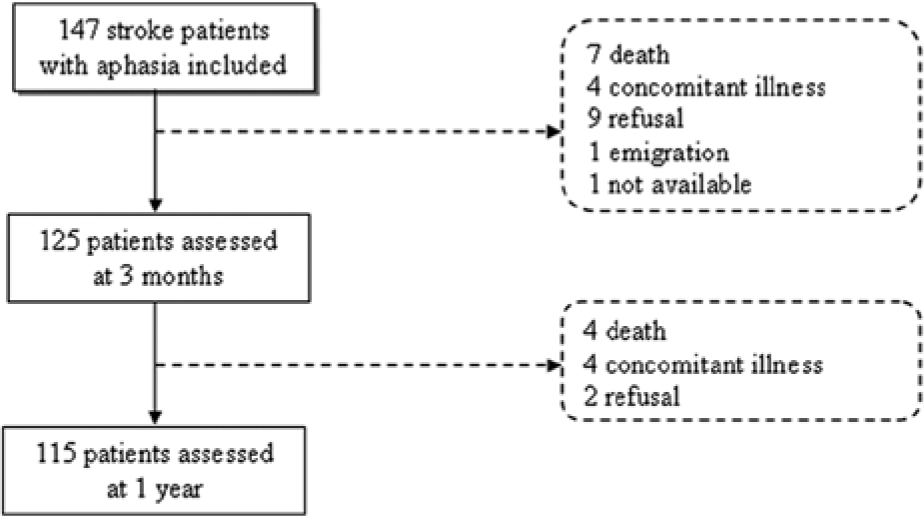
Flowchart of the included patients.
A total of 24 patients did not receive speech-language therapy because of early recovery (n = 21), or refusal (n = 3), and for 5 patients we could not ascertain whether or not they received therapy because of early refusal to further participate in the study. For 23 patients, it was reported that they received therapy, but information on the duration, amount, and content of therapy was not registered. The other 95 patients received a median of 27 hours (interquartile range = 3-83) of speech-language therapy during the 1-year follow-up period.
All Patients
Compared with the norm scores, our cohort was impaired (ie, differed significantly from
Difference in Cognitive Scores Between Cohort and the Norm (ie,
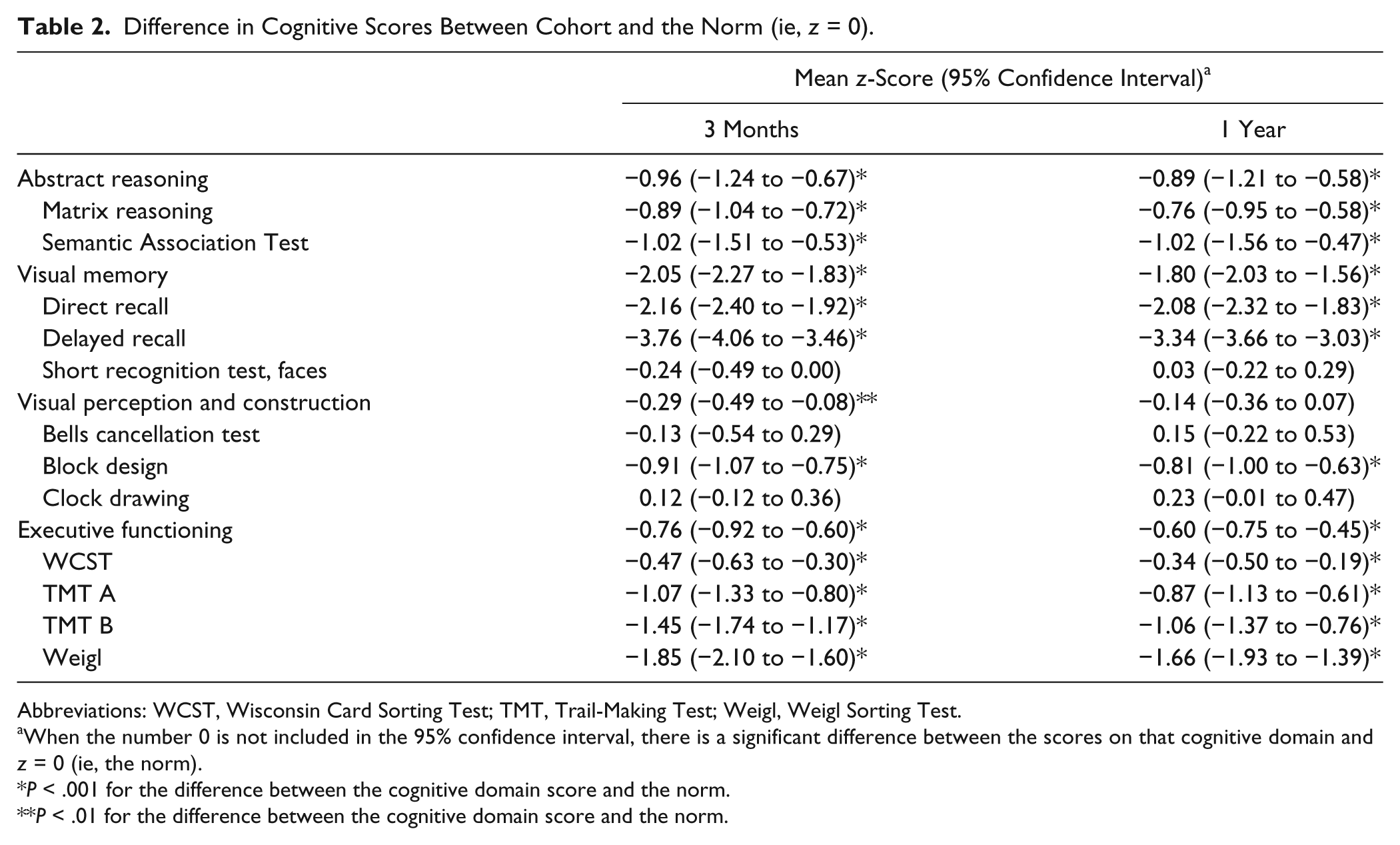
Abbreviations: WCST, Wisconsin Card Sorting Test; TMT, Trail-Making Test; Weigl, Weigl Sorting Test.
When the number 0 is not included in the 95% confidence interval, there is a significant difference between the scores on that cognitive domain and
Impairment of visual memory was most frequent, both at 3 months and 1 year. Impairment of visual perception and construction was least common. Only the prevalence of visual memory impairments decreased over time (Table 3). At 3 months, 107 (88%) patients were impaired in at least one nonlinguistic cognitive domain. At 1 year, this amounted to 91 patients (80%).
Frequency of Cognitive Impairment and Comparison Between the Assessments. a
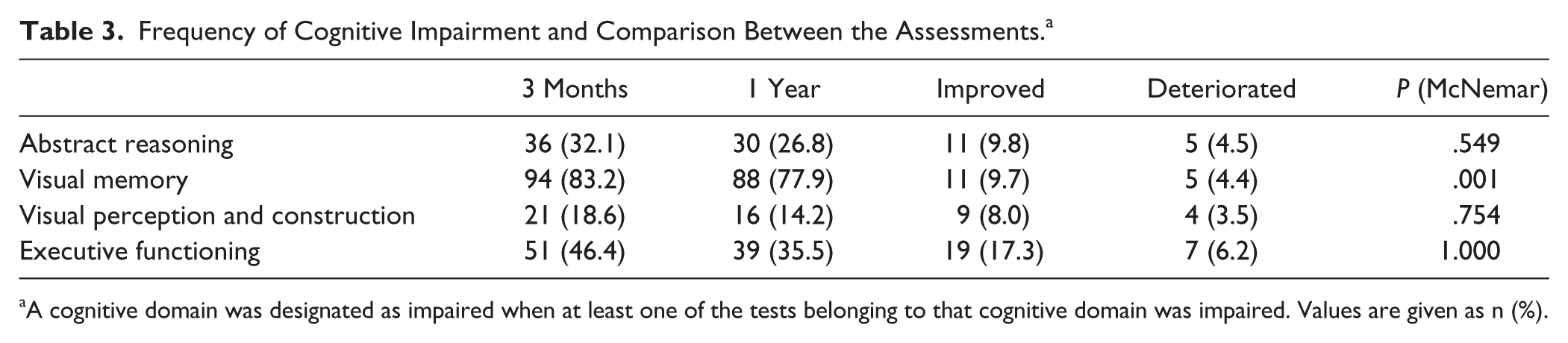
A cognitive domain was designated as impaired when at least one of the tests belonging to that cognitive domain was impaired. Values are given as n (%).
There was improvement between 3 months and 1 year on all nonlinguistic cognitive domains, except for abstract reasoning. Of the scores on the language tasks, that is ScreeLing, ASRS, and Token Test, only the ASRS rating showed improvement (
Comparison of Cognitive (
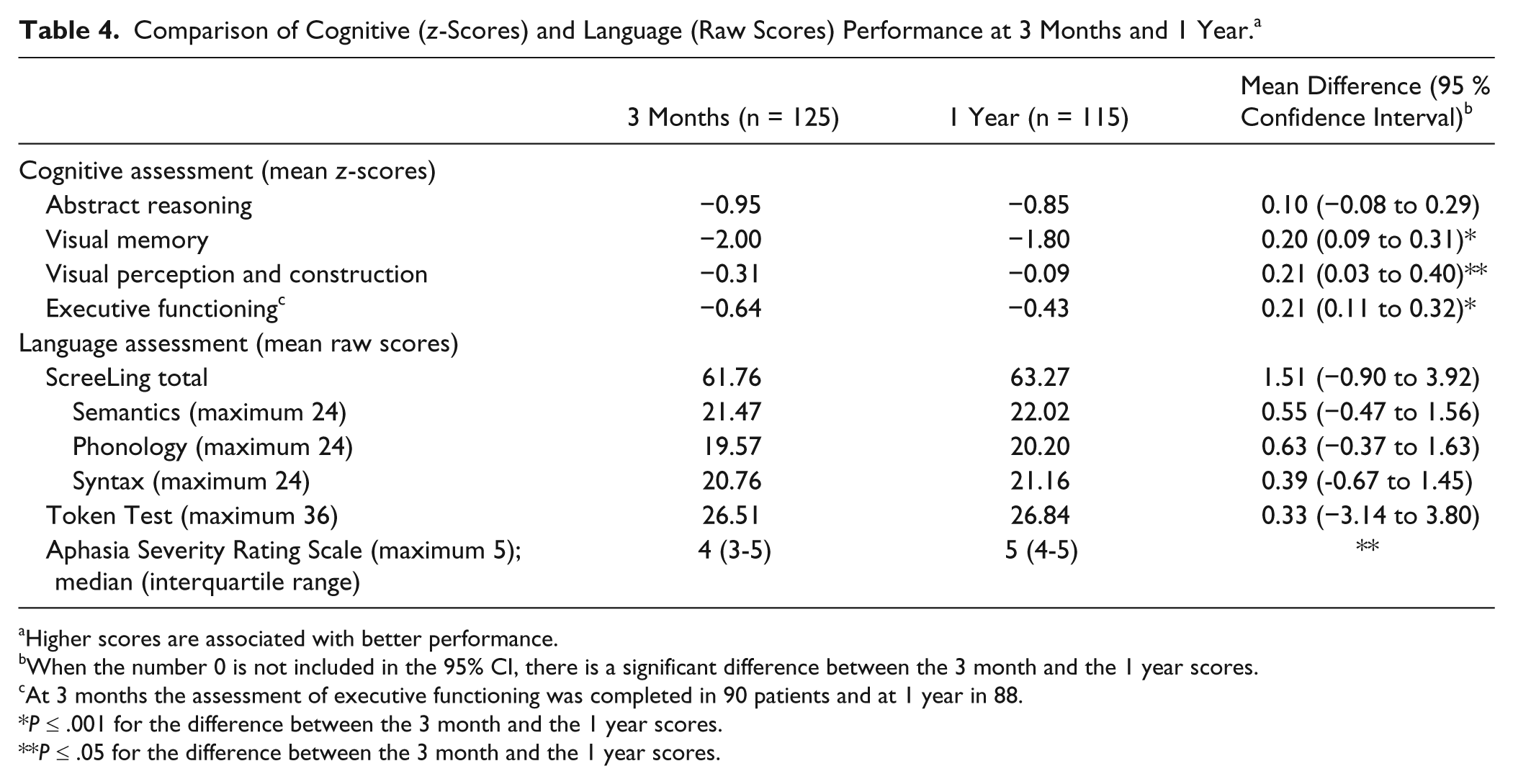
Higher scores are associated with better performance.
When the number 0 is not included in the 95% CI, there is a significant difference between the 3 month and the 1 year scores.
At 3 months the assessment of executive functioning was completed in 90 patients and at 1 year in 88.
Patients With Persisting Aphasia Versus Recovered Patients
At 3 months, 43 of the 125 patients assessed (34%) had recovered from aphasia, and at 1 year, 48 of the 115 patients (42%). At both assessments, the recovered patients had higher cognitive domain scores than those with persisting aphasia (Table 5). This finding was independent of age, sex, and education level. Also, patients with persisting aphasia showed more impairment (ie, impairments in ≥2 domains) than those who had recovered: at 3 months 63% versus 35% (
Comparison of Mean Cognitive
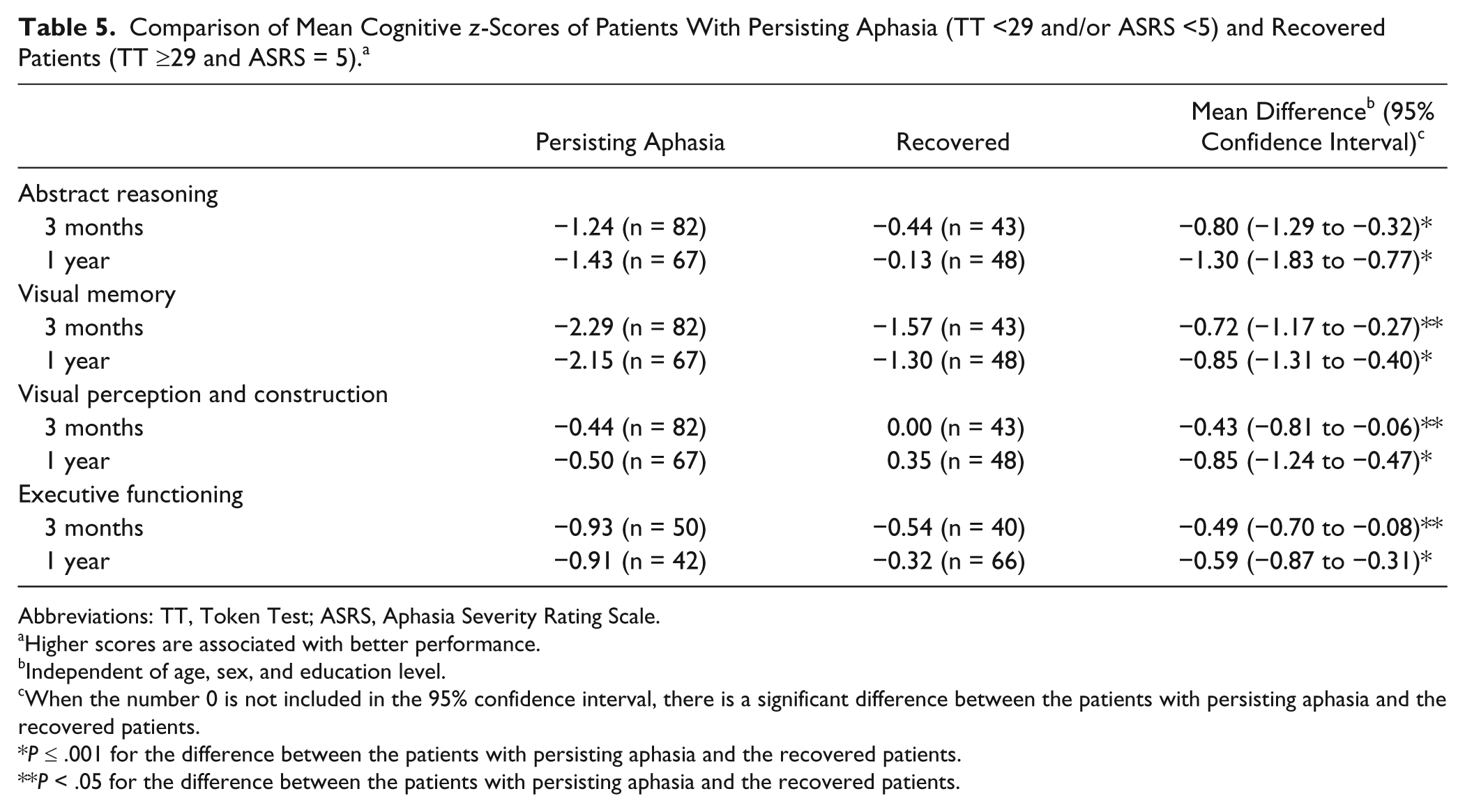
Abbreviations: TT, Token Test; ASRS, Aphasia Severity Rating Scale.
Higher scores are associated with better performance.
Independent of age, sex, and education level.
When the number 0 is not included in the 95% confidence interval, there is a significant difference between the patients with persisting aphasia and the recovered patients.
Patients With Nonlinguistic Cognitive Impairments and Depression
Patients with severe cognitive impairment more often had poor functional outcome than patients with mild cognitive impairment: 39% versus 9% (
Predictor of Functional Outcome at 1 Year After Stroke.
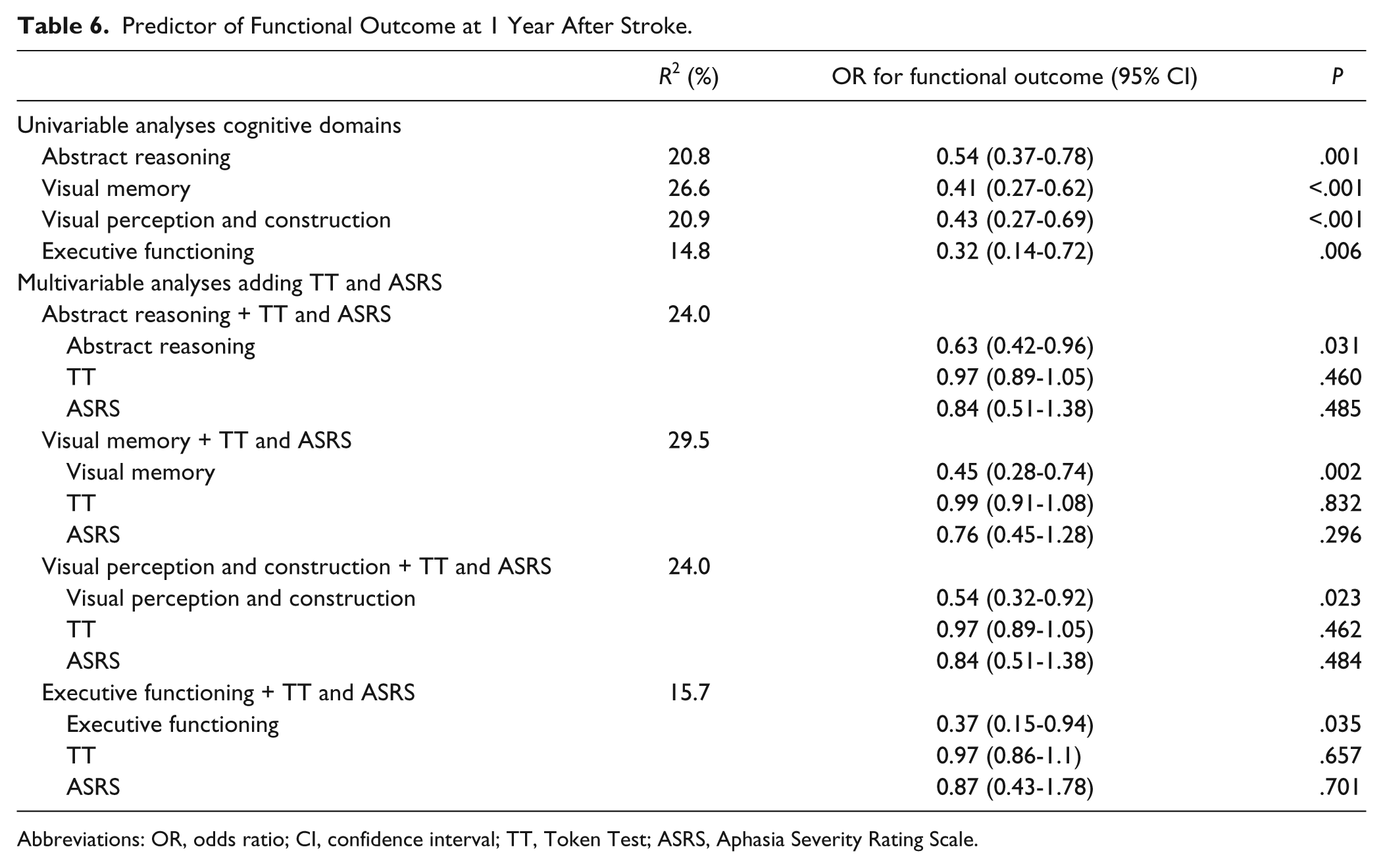
Abbreviations: OR, odds ratio; CI, confidence interval; TT, Token Test; ASRS, Aphasia Severity Rating Scale.
Finally, depression occurred in 11% of the entire cohort: at 1 year more often in patients with persisting aphasia (16%) than in recovered patients (6%), and more often in patients with severe cognitive impairment (20%) than in those with mild cognitive impairment (5%).
Discussion
Our study shows that nonlinguistic cognitive impairments are common in patients with either persisting or recovered aphasia in the first year after stroke. Both at 3 months and at 1 year, visual memory was most frequently affected, and impairment in visual perception and construction least frequently. We found improvement on visual memory, visual perception and construction, and executive functioning. Of the language assessments, only the ASRS results improved between 3 months and 1 year. At both assessments, patients with persisting aphasia had lower cognitive domain scores and more severe cognitive impairment than patients who had recovered from aphasia. Patients with persisting aphasia also more often had a poor functional outcome and depression. Furthermore, poor functional outcome and depression were more frequent in patients with severe cognitive impairment than in those with mild cognitive impairment. These findings stress the importance of assessing nonlinguistic cognition in patients with aphasia due to stroke in addition to language assessment as cognitive impairment may influence their rehabilitation. Moreover, aphasic patients may also benefit from psychological treatment and/or medication since the prevalence of depression was higher in the patients with persisting aphasia than in those who recovered.
Strength of our study is that it is the first to assess nonlinguistic cognitive impairments prospectively and thoroughly in a large cohort of patients with aphasia after stroke. Furthermore, this study is the first report on the difference in cognitive performance between patients with persisting aphasia and those who recovered. A limitation is that although we applied a nonlinguistic cognitive test battery of well-known and well-validated tests suitable for patients with aphasia, we cannot rule out that patients used covert language as a tool to process nonlinguistic tasks.
40
In addition, it has been reported that about 25% of the variance in nonlinguistic cognitive performance is accounted for by auditory comprehension indicating that impaired nonlinguistic functioning is not fully related to impaired language.
41
In patients with aphasia it is difficult to disentangle the relationship between performance on nonlinguistic cognitive tests and the language impairment: do aphasic patients have lower cognitive scores because of their language impairment or vice versa? Unfortunately, our study design and data do not justify any valid statements concerning the causality of this relationship. Another limitation is that we were unable to perform cognitive assessment in the acute stage, since it has been reported that acute cognitive impairments influence cognitive and functional outcome.
11
Half of our patients had severe aphasia in the acute stage, which hampered early and extensive cognitive examination. This is probably the reason why most previous studies on cognitive outcome after stroke have excluded patients with aphasia, even when conducted in the postacute stage.9,10,12 In this study, we were able to perform cognitive assessment in most patients using standard test instructions with examples, since the severity of aphasia had diminished substantially at 3 months and 1 year; the performance on comprehension measures (semantics component of the ScreeLing and Token Test) was quite well (Table 4). A test was not administered when a patient failed the example or, in case of no example, failed the first items because the instruction could not be comprehended. Hence, assembling a cognitive test battery that was suitable for patients with aphasia after stroke was one of the most difficult challenges in this study. A standard test battery used in clinical practice to assess cognition may not be appropriate for patients with aphasia because of their language deficits. Therefore, a cognitive test battery was needed that did not heavily depend on language skills. The selected tests had to be standardized, validated, and available in the Netherlands. As a result we selected some tests of which the results may be influenced by motor skills, for example during the clock drawing test. We expected that at 3 months after stroke, when the cognitive test battery would be used for the first time, the patients with a hemiparesis would have learned to deal with the weakness of the preferred hand during their rehabilitation and used to using the nonpreferred hand more often. Even though not ideal, we tried to minimize the number of tests in which patients are asked to make drawings. Another limitation is that we did not perform extensive neuropsychiatric assessments, particularly of mood disorders. We did, however, review medical records and performed interviews with all patients and often also with their relatives. Furthermore, our cognitive assessment included several aspects of a neuropsychiatric assessment (eg, overall questions on mood, behavior, etc) to ascertain that the cognitive scores were not influenced by possible neuropsychiatric disorders. All patients who had depression in our study were already treated for this disorder. It would have been better if we had been able to perform a thorough examination using the criteria of the
Our data show that 34% of the patients who were assessed at 3 months and 42% of those assessed at 1 year after stroke had recovered from aphasia. This is in line with a previous study that reported that 44% of their patients had fully recovered from aphasia already at the time of discharge from the hospital. 8
The reported prevalence of nonlinguistic cognitive impairment at 3 months after stroke ranges from 17% to 68%,5,10,12,42 and at 1 year from 27% to 73%,9,43 which is much lower than in our study (88% and 80%, respectively). This difference may be caused by the fact that most of these previous studies excluded patients with aphasia
12
or with severe aphasia.
5
An additional reason could be that some studies used only a cognitive screening tool and not an extensive cognitive assessment.
42
A final reason could be the manner in which cognitive impairment is defined. In our study, a cognitive domain was designated as impaired when the
Our finding that visual memory was most frequently impaired is consistent with an earlier study 5 using a similar task, that is, the visual reproduction test. 25 It has been reported that aphasia is related to memory impairments. 44
Impairments in visual perception and construction are reported to be associated primarily with right hemisphere lesions. 45 Therefore it was expected that impairments in this domain would be less frequent in our cohort since nearly all our patients had left hemisphere lesions. The prevalence of impairments in executive functioning and abstract reasoning are in line with an earlier study that examined the prevalence of impairments in various cognitive domains in the acute stage of stroke. 1
With respect to recovery, we found improvement in almost all nonlinguistic cognitive domains (except for abstract reasoning), which is in line with an earlier study that also reported improvement after 3 months. 2 In the language domain, we found only improvement on the ASRS, a measure of verbal communication. On the other language tests, that is, ScreeLing and Token Test, the recovery process had already reached a plateau within the first 3 months after stroke. 39 This is consistent with the general consensus that aphasia recovery occurs mainly within the first couple of months after stroke. 8 The prolonged recovery of verbal communication (ie, ASRS) suggests that nonlinguistic cognitive functions, for example, executive functioning, may be involved in verbal communication where the main language components (ie, semantics, phonology, and syntax) are integrated.
Our finding that patients with persisting aphasia had lower cognitive scores, more severe cognitive impairment, and more often a poor functional outcome, is consistent with a previous study in which a small group of severe aphasics was compared with a group of mild or moderate aphasics. 14 Our result that patients with persisting aphasia had a higher prevalence of depression than patients who had recovered, is in line with the report that aphasic patients are more often depressed compared with stroke patients without aphasia. 14
To summarize, nonlinguistic cognitive impairments are common in patients with acute aphasia after stroke, at 3 months as well as at 1 year. Although improvement occurred in most nonlinguistic cognitive domains and verbal communication, our cohort remained impaired. This implies that patients with aphasia may benefit from an extensive cognitive evaluation in addition to language assessments in order to optimize the rehabilitation process.
Footnotes
Acknowledgements
We thank the speech-language therapists of the following centers who included the patients and/or performed the language assessments:
Declaration of Conflicting Interests
The author(s) declared the following the following conflicts of interest with resoect to the research, authorship, and/or publication of this article: Dr. El Hachioui, Dr. Visch-Brink and Dr. van de Sandt-Koenderman receive royalties from the publication of the test ScreeLing. The publisher had no involvement in the study design, data collection, analysis and interpretation of data, in writing of the report, and in the decision to submit the paper for publication.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Netherlands Organization for Scientific Research (NWO; Grant No. 017.002.083).
