Abstract
Background. The neural basis underlying somatosensory impairment and recovery poststroke is virtually unexplored. Objective. To investigate the relationship between touch discrimination impairment and task-related brain activation in stroke survivors with somatosensory impairment following subcortical or cortical lesions. Methods. A total of 19 stroke survivors with touch impairment were investigated using fMRI and a touch discrimination paradigm 1-month poststroke; 11 had subcortical and 8 cortical sensory lesions; 12 age-matched healthy controls were also studied. Mean task-related contrast images were regressed with sensory impairment using random effects analysis for each subgroup and the total group. Results. There was no significant difference in touch impairment between stroke subgroups. Touch discrimination of the affected hand correlated negatively with task-related activation in the ipsilesional primary somatosensory cortex (SI; adjacent to the SI hand area activated in healthy controls); ipsilesional secondary somatosensory cortex (SII); contralesional thalamus; and attention-related frontal and occipital regions in the subcortical group. In contrast, the cortical group did not show significant correlated activity. Yet there was no significant between-group difference in a priori somatosensory regions: only in the superior medial frontal gyrus. A negative correlation was observed in the contralesional thalamus for the total group, irrespective of lesion type. Conclusion. The findings provide novel evidence of neural correlates of poststroke touch impairment involving a distributed network of ipsilesional SI and SII, the contralesional thalamus, and frontal attention regions, particularly following subcortical lesions. Further systematic investigation of a modulatory role for ipsilesional SI, the thalamus, and frontal attention regions in sensory processing and recovery is warranted, particularly given implications for rehabilitation.
Keywords
Introduction
Neural correlates of poststroke impairment severity can provide important insights into specific brain targets for therapy. 1,2 Changes in perilesional activation, laterality of brain activity, and involvement of distributed cortical networks have been characterized with varying recovery poststroke and over time. 1-4 In motor recovery, different activation patterns have been reported following cortical or subcortical lesions, 5 and models of poststroke motor and sensory recovery provide theoretical foundations for these differences. 1,4 Furthermore, persistent activation in distributed and secondary motor areas is correlated with partial motor recovery, whereas return to a “normal” pattern of focal activation in the primary sensorimotor cortex (SIMI) is correlated with good recovery. 6,7 Changes in cortical excitability and balance of activity between hemispheres also occur with sensorimotor recovery following cortical and subcortical lesions and with near complete versus partial recovery. 3 However, while motor recovery has been well studied, neural mechanisms underlying somatosensory impairment and recovery remain virtually unexplored.
In daily life, intact somatosensation is crucial for perception and action. Loss of body sensations, such as touch discrimination, occurs in 50% to 85% of stroke survivors, hampering the ability to explore the immediate environment and tactually recognize and manipulate objects, as well as negatively affecting personal safety, quality of life, and length of hospital stay. 8,9 Tactile input and activity in somatosensory brain regions have also been linked to motor recovery poststroke. 10,11
Neuroimaging studies of somatosensory recovery poststroke are limited to a few case reports and small group studies (n ≤ 7), mostly of patients with thalamic lesions. Return of activation in the ipsilesional primary somatosensory cortex (SI) was reported with good recovery, 3,12,13 or this region was relatively spared with mild impairment. 13,14 With more severe impairment, reduced activation of ipsilesional SI with preserved responsiveness of the secondary somatosensory cortex (SII), 15 bilateral SI and distributed activation, 16 or return of activation in SII at 6 months poststroke 12 were reported. Patients with ventroposterior thalamic lesions also showed selective hypoperfusion in ipsilesional SIMI at rest relative to the contralesional hemisphere. 14 Investigation of patterns of activation with impairment following lesions to cortical somatosensory structures is currently lacking.
Here, we systematically investigated the relationship between touch discrimination impairment and task-related brain activation in stroke survivors with a wide range of impairment severity in the subacute phase of recovery (approximately 1-month poststroke) using functional magnetic resonance imaging (fMRI). Separate analyses were conducted for subcortical and cortical somatosensory lesion groups given: evidence of different patterns of activation associated with motor recovery following cortical and subcortical lesions 5 ; the current lack of investigation following lesion to cortical sensory structures; and the prediction, based on a theoretical model of sensory recovery, that patterns of task-related activity would differ following lesions to cortical and subcortical sensory structures. 1 We predicted correlated activity in ipsilesional SI and bilateral SII for subcortical lesions. In both groups, we also expected correlated activity in distributed brain regions, especially involving the contralesional hemisphere and thalamus, with partial recovery. For these reasons, we investigated changes in bilateral somatosensory regions and the whole brain. We also investigated the group as a whole to identify the presence of correlated activity irrespective of lesion location. Positive and negative blood oxygenated level–dependent (BOLD) signal were identified given poststroke changes in resting activity 3,14 and evidence of an association between sustained negative BOLD response and sensory thresholds in healthy participants. 17,18
Methods
Participants
We studied 19 stroke patients with impaired touch discrimination of the upper limb. Inclusion criteria were as follows: first episode infarct, medical stability, ability to give informed consent and comprehend simple instructions, and right-hand dominance. Exclusion criteria included the following: brainstem infarct or hemorrhagic stroke, previous neurological dysfunction, medical history impairing hand function or precluding MRI, or evidence of neglect based on standard neuropsychological tests. We also studied 12 age-matched, right-hand dominant controls (7 men, mean age 61.75 years; range, 42-79 years) without any history of neurological or sensory impairment. The protocol was approved by university and hospital human ethics committees, and written informed consent was obtained from all participants.
Demographic and Clinical Profile
Background information included age, gender, and premorbid hand dominance. 19 A clinical profile obtained within 48 hours of the MRI study included the following: severity of neurological impairment, using the National Institute of Health Stroke Scale 20 ; severity of global disability, using the Barthel Index 21 ; and hand function, using the Action Research Arm Test. 22 Severity of somatosensory impairment was quantified across several modalities, including: touch (see below); limb position sense, using the Wrist Position Sense Test 23 ; tactile object recognition, using the functional Tactile Object Recognition Test 9 ; and temperature discrimination, using the Roylan hot and cold discrimination kit.
Quantification of Touch Discrimination and Impairment
The primary outcome was a psychophysical measure of touch discrimination of the fingertips, the Tactile Discrimination Test (TDT). 24 Participants discriminate differences in finely graded plastic texture surfaces using the method of constant stimuli and a 3-alternative forced-choice design. Five surface sets, which span the Weber function of texture differences, are each presented 10 times. The test score is the probability of correct discrimination response across all stimuli presented (n = 50) and represents the area that subtends the psychometric function after accounting for chance. The TDT has high test–retest reliability, age-appropriate normative standards, and excellent discriminative properties. 24 Touch detection of fingertips was assessed using the Weinstein Enhanced Sensory Test hand monofilaments and the rapid threshold procedure. 25
Sensory Stimulation Paradigm for fMRI
A custom-made touch stimulation device presented a texture grid to the fingertips in a controlled manner, as previously described, 12 while the patient lay supine in the scanner. A plastic texture grating was repeatedly moved from side to side under the second, third, and fourth digits at alternating rates of 1.5 Hz and 3 Hz within each 30-s stimulus block. A metronome, played through headphones to the stimulus presenter, set the stimulation rate. The relative pressure of the fingertips on the surface was regulated through a weighted pulley system. The dynamic setup ensured relative motion between the skin and texture surface, a critical feature of texture discrimination, while at the same time not requiring active movement that would confound the brain activation study. Participants were told to keep their eyes closed, that “sometimes there would be a stimulus under their fingertips and sometimes not,” and that they would be asked what they felt at the end of the run. They were instructed to “feel the stimulus and keep feeling it as it moves under your fingertips,” thus, encouraging attentive perception of the touch stimulus. In the control condition, the surface was moved at the same speed but did not contact the fingertips. A brief period of familiarization with the task was provided prior to each run. Immediately after the session, participants reported on their degree of concentration during the task and perception of stimulus intensity, using visual analogue scales. We have previously used this paradigm in healthy controls with robust findings. 26,27
Magnetic Resonance Imaging
Functional imaging sequences
Whole brain fMRI studies were conducted on a 3-T GE Horizon LX Signa MRI scanner with quadrature head coil (GE Medical Systems, WN) during the controlled touch stimulation paradigm. Alternating touch and rest conditions were presented in a block design, comprising 6 × 30 s blocks of each condition for each hand, providing a total of 60 volumes per condition per hand. Separate runs were conducted for each hand, with the affected done first. Images were acquired in 25 axial slices spanning cerebellum to the apex of the cerebrum using a gradient-echo, echoplanar (EPI) sequence (Repetition Time [TR] = 3000 ms; Echo Time [TE] = 40 ms; flip angle = 75°; Field of View [FOV] = 240 mm; 128 × 128 matrix; slice thickness = 4 mm; interslice gap = 1 mm; in-plane voxel size = 1.95 × 1.95; bandwidth = 100).
Structural imaging sequences
Whole brain anatomical and angiographic images were acquired at the same session and included the following: a high-resolution 3D anatomical image, 2D T1-weighted and axial 2D T2-weighted images in same plane as EPI, and 2D angiographic images.
Data Analysis
First-level imaging analysis
Preprocessing for each participant’s data included image conversion, slice timing correction, determination of optimum realignment target (median center-of-within-brain intensity), motion detection and realignment (rigid body with 6 degrees of freedom), normalization to a customized EPI brain template (see below), Gaussian smoothing (8-mm full width at half maximum), and automated creation of within-brain mask of normalized images, using Statistical Parametric Mapping, SPM2 (www.fil.ion.ucl.ac.uk), and iBrain software. 28 Task-related changes in BOLD signal intensity were mapped for activated (increased) and deactivated (decreased) voxels relative to the rest control condition for each individual. Motion correction parameters were included as covariates of no interest. Data from each imaging run were scaled to a grand mean of 100.
For group analyses, fMRI data were brought into standard space. The spatial normalization target used was a symmetrical version of a custom template, approximating the EPI template in Montreal Neurological Institute (MNI) space supplied with SPM2. The custom template was created in an iterative fashion from an independent group of 30 healthy participants scanned on our 3-T MRI scanner. 29 Images of patients with right hemisphere lesions were flipped such that all infarcts were in the left hemisphere.
Second-level imaging analysis
Mean contrast images of task-related changes in BOLD signals (one for each hand of each individual) were linearly regressed with TDT scores using random effects analysis. A region of interest (ROI) mask of SI, SII, and the thalamus of both hemispheres was defined using the Wake Forest University PickAtlas v2.4 30 and automated anatomical labeling. 31 An independent template was used to avoid possible interference from using the same data set as part of the selection criteria for hypothesis testing. 32 A dilation factor of 1 voxel was included a priori to allow for variation across participants. We first examined correlation results for each subject group. We then determined the between-group differences in regression slopes by entering data for cortical and subcortical groups into a single design matrix and testing the difference between β weights of the zero-mean-corrected TDT score columns. Group correlation maps were first thresholded at a voxel height of P = .001 (uncorrected), and we report clusters significantly correlated at P < .05, small-volume corrected for the somatosensory ROI, and at P < .05 corrected for multiple comparisons across the whole brain.
We performed a post hoc analysis to investigate the nature of the relationship between task-related activation (task minus rest parameter estimate) and TDT score in clusters of significant correlation within our identified regions of interest. For each significant cluster, we calculated the average signal change (task minus control) in the first-level single-subject analyses of each individual, then plotted these against the TDT score. To facilitate interpretation of location, we inspected whether correlated sites intersected with a mask of activated regions of healthy controls and stroke survivors (random effects analysis; P < .05 cluster corrected). The infarct was outlined on axial slices of a 3D MRI sequence, plotted into stereotactic space 33 as previously described, 34 and displayed on a template for subcortical and cortical groups.
Results
In all, 19 stroke survivors (12 men, mean age 59.53 years; range, 18-79 years) were studied (Table 1) at 4.5-weeks poststroke (±1.5 weeks). All were right-hand dominant with a median laterality quotient of 95. 19 The left hemisphere was infarcted in 14 patients; 11 had subcortical lesions involving the thalamus (n = 8) and/or internal capsule (n = 4), and 8 had cortical lesions involving SI and/or SII (Table 1; Figure 1). Patients presented with wide variation in severity of touch discrimination, ranging from −11.58 (very severe impairment, ie, discrimination less than that expected by chance) to 79.31 (just within the normal range) on the TDT. Mean affected-hand score for the subcortical group was 35.36 ± 24.01 standard deviation (SD; median = 31.03) and for the cortical group was 20.17 ± 30.73 SD (median = 14.29), compared with 79.42 ± 7.42 SD (median 80.54) for healthy controls. There was no significant difference in impairment between patient groups (Mann-Whitney U = −1.07, P = .31). The interquartile range for touch detection was 1.1 g (reduced tactile sensation) to 200 g (profoundly insensate). 25 Self-reported intensity of touch perception and concentration during scanning and clinical test scores are presented in Table 1.
Background and Clinical Data
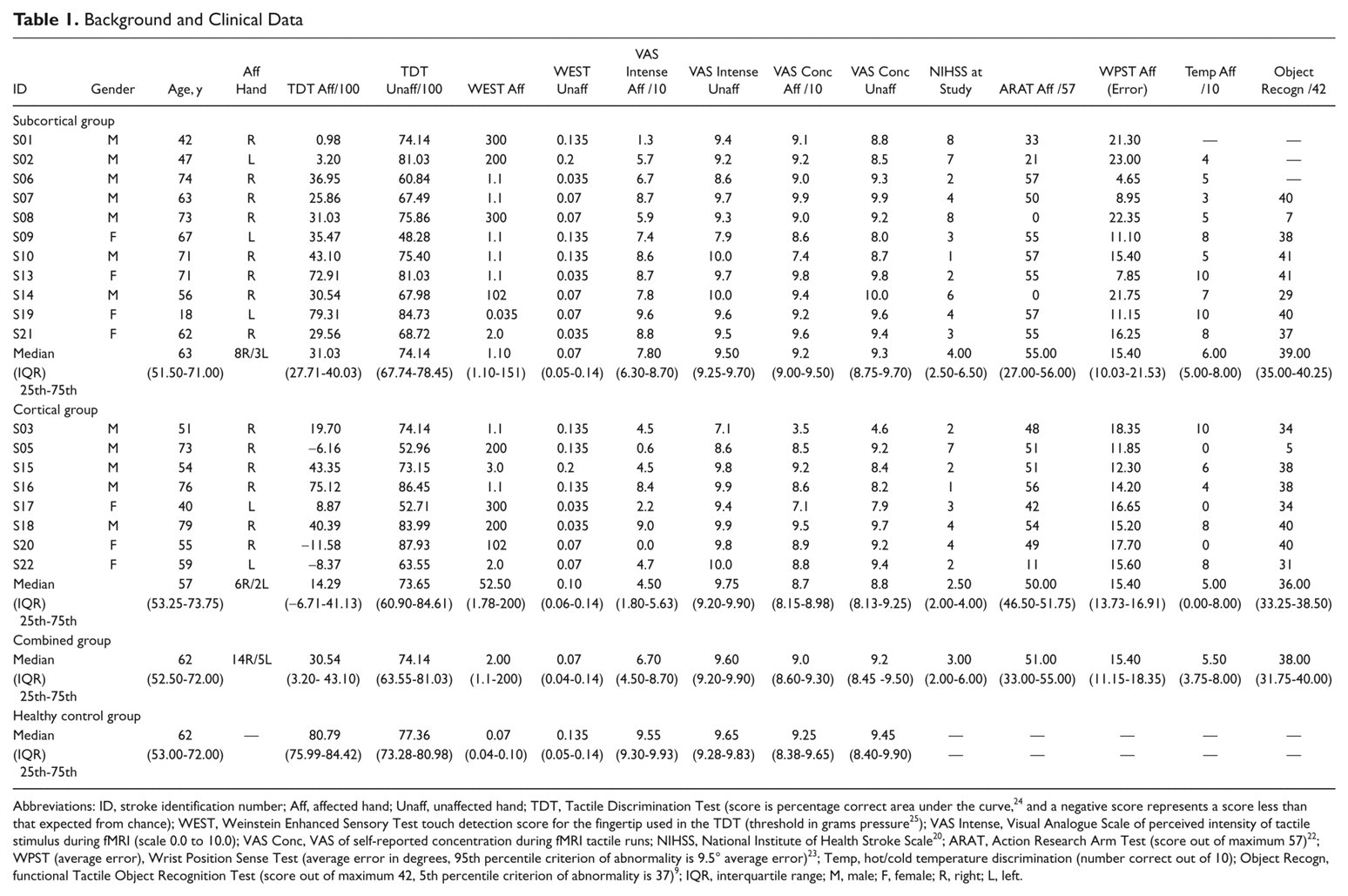
Abbreviations: ID, stroke identification number; Aff, affected hand; Unaff, unaffected hand; TDT, Tactile Discrimination Test (score is percentage correct area under the curve, 24 and a negative score represents a score less than that expected from chance); WEST, Weinstein Enhanced Sensory Test touch detection score for the fingertip used in the TDT (threshold in grams pressure 25 ); VAS Intense, Visual Analogue Scale of perceived intensity of tactile stimulus during fMRI (scale 0.0 to 10.0); VAS Conc, VAS of self-reported concentration during fMRI tactile runs; NIHSS, National Institute of Health Stroke Scale 20 ; ARAT, Action Research Arm Test (score out of maximum 57) 22 ; WPST (average error), Wrist Position Sense Test (average error in degrees, 95th percentile criterion of abnormality is 9.5° average error) 23 ; Temp, hot/cold temperature discrimination (number correct out of 10); Object Recogn, functional Tactile Object Recognition Test (score out of maximum 42, 5th percentile criterion of abnormality is 37) 9 ; IQR, interquartile range; M, male; F, female; R, right; L, left.
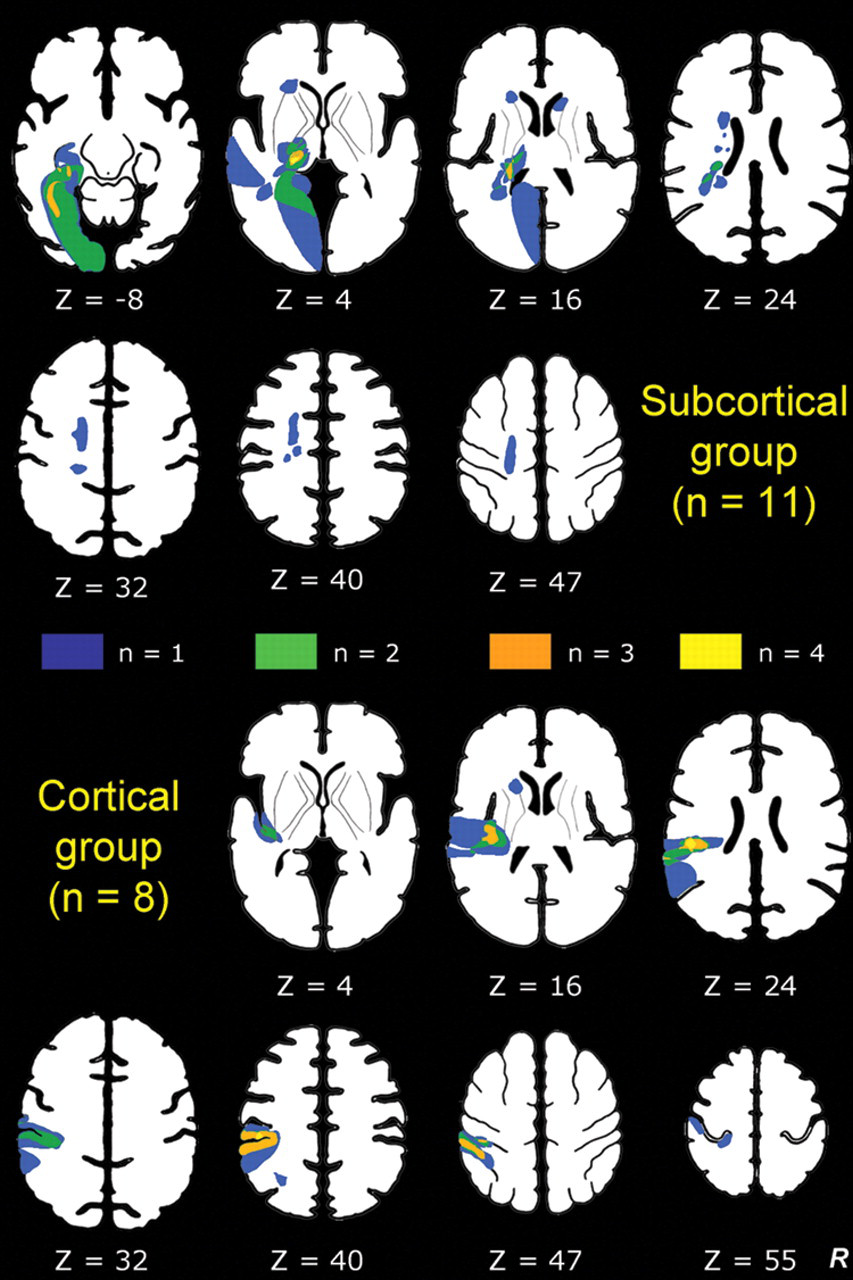
Infarct locations for subcortical and cortical lesion groups: Infarct locations for each individual are plotted in stereotactic space, with color coding representing locations where one or more patients had an infarct: for example, orange represents overlap of infarct location for 3 patients. Lesions in the right hemisphere were flipped so that all lesions are represented in the left hemisphere. Z values represent axial slice position in Talairach space. 33
Correlation of Touch Discrimination and Sites of Brain Activation
The subcortical group showed significant negative correlations between BOLD activation during affected-hand tactile stimulation and TDT affected-hand scores. The task-related BOLD signal change was negatively correlated in somatosensory regions—that is, ipsilesional SI and SII and the contralesional thalamus (Table 2; Figure 2). Correlated SI activity was adjacent to (but not within) the SI hand region activated by the group of healthy controls (see Figure 2, inserts top right). Correlated activity was also observed in distributed regions, including the ipsilesional middle occipital gyrus, superior and middle frontal gyri and contralesional putamen, calcarine and lingual gyri, and cuneus.
Brain Regions Showing Correlated Activity With Touch Outcome for the Subcortical Group During Stimulation of the Affected and Unaffected Hands a
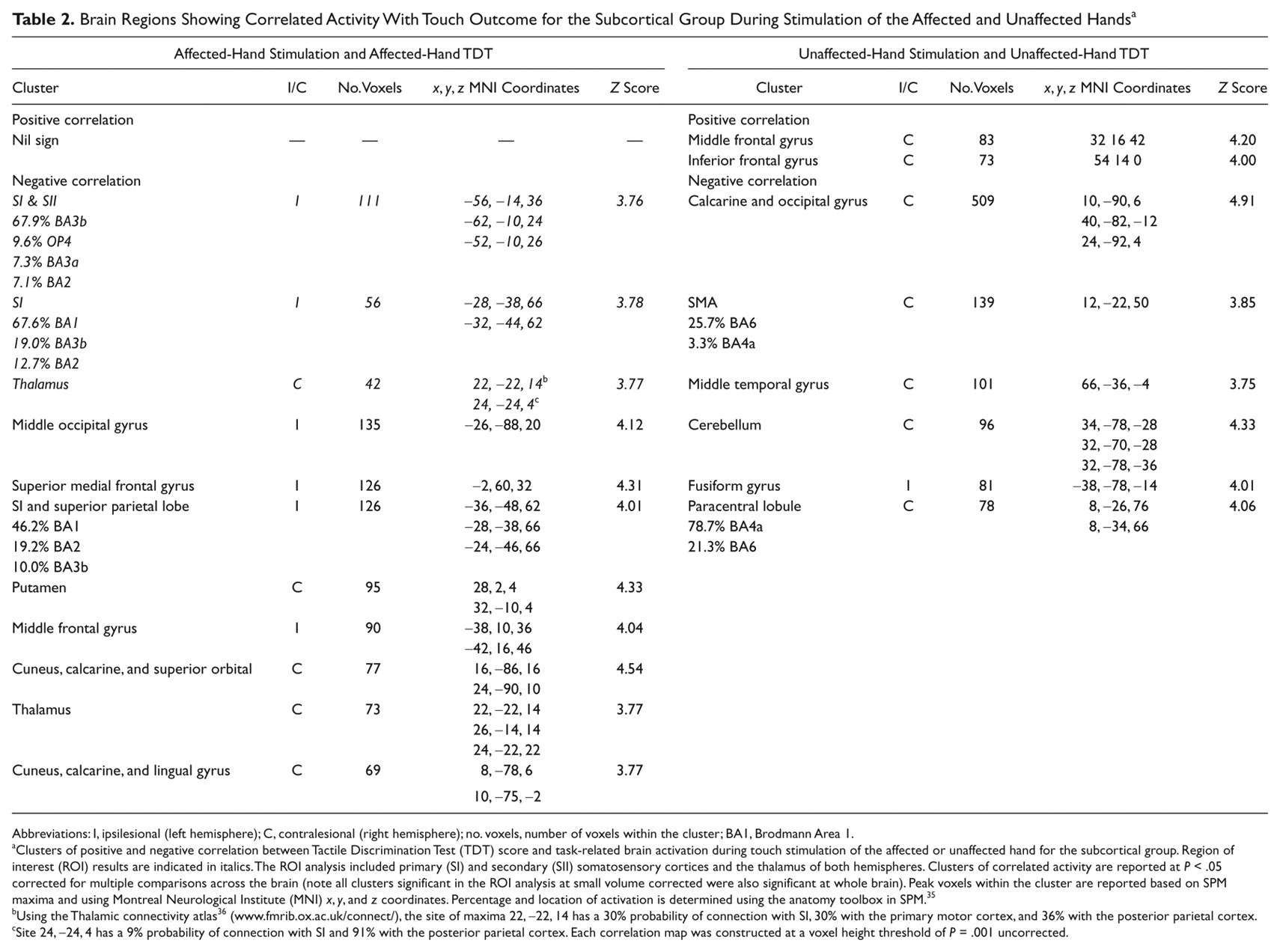
Abbreviations: I, ipsilesional (left hemisphere); C, contralesional (right hemisphere); no. voxels, number of voxels within the cluster; BA1, Brodmann Area 1.
Clusters of positive and negative correlation between Tactile Discrimination Test (TDT) score and task-related brain activation during touch stimulation of the affected or unaffected hand for the subcortical group. Region of interest (ROI) results are indicated in italics. The ROI analysis included primary (SI) and secondary (SII) somatosensory cortices and the thalamus of both hemispheres. Clusters of correlated activity are reported at P < .05 corrected for multiple comparisons across the brain (note all clusters significant in the ROI analysis at small volume corrected were also significant at whole brain). Peak voxels within the cluster are reported based on SPM maxima and using Montreal Neurological Institute (MNI) x, y, and z coordinates. Percentage and location of activation is determined using the anatomy toolbox in SPM. 35
Using the Thalamic connectivity atlas 36 (www.fmrib.ox.ac.uk/connect/), the site of maxima 22, −22, 14 has a 30% probability of connection with SI, 30% with the primary motor cortex, and 36% with the posterior parietal cortex.
Site 24, −24, 4 has a 9% probability of connection with SI and 91% with the posterior parietal cortex. Each correlation map was constructed at a voxel height threshold of P = .001 uncorrected.
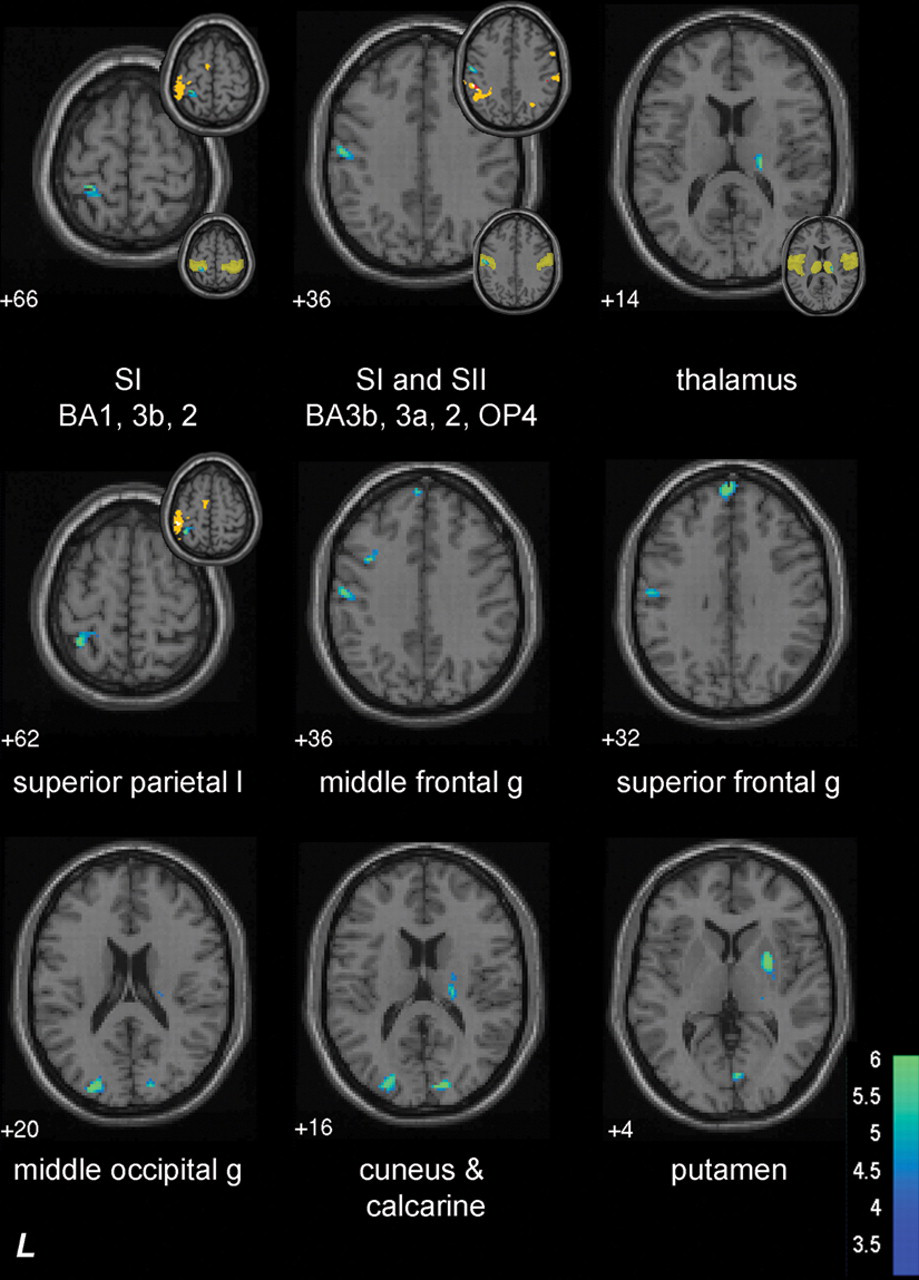
Subcortical group: neural correlates of affected-hand touch discrimination. Axial slices show sites of brain activation correlated with affected-hand touch discrimination during stimulation of the affected hand. Negatively correlated activity is shown using cold colors, which represent t values. There were no significant sites of positive correlation. The small insets to the top right of the brain slice show location of correlated activity relative to activated regions primarily for healthy controls (yellow = activation in healthy controls; red = overlap of activation in healthy controls and subcortical group; there was little or no overlap of activation in SI and SII between healthy controls and the subcortical group in the slices displayed). The small insets (bottom right) show correlated activity overlaid on region of interest mask (transparent yellow) used in analysis. Z values, for example +66, represent axial slice position in Montreal Neurological Institute (MNI) space. There were no sites of positive or negative correlation during stimulation of the affected hand for the cortical group. Abbreviations: BA, Brodmann Area; g, gyrus; SI, primary somatosensory cortex; SII, secondary somatosensory cortex
Inspection of the scatterplots indicates that patients with better touch discrimination were below the zero line; that is, they had a lower parameter estimate for the sensory task relative to the no-stimulation rest condition in ipsilesional SI, ipsilesional SII, and the contralesional thalamus (ie, a relative deactivation; Figure 3). There were no significant positive correlations (Table 2). The signal change observed in age-matched healthy controls (Figure 3) covered a similar range to the stroke patients but was not correlated with touch discrimination.
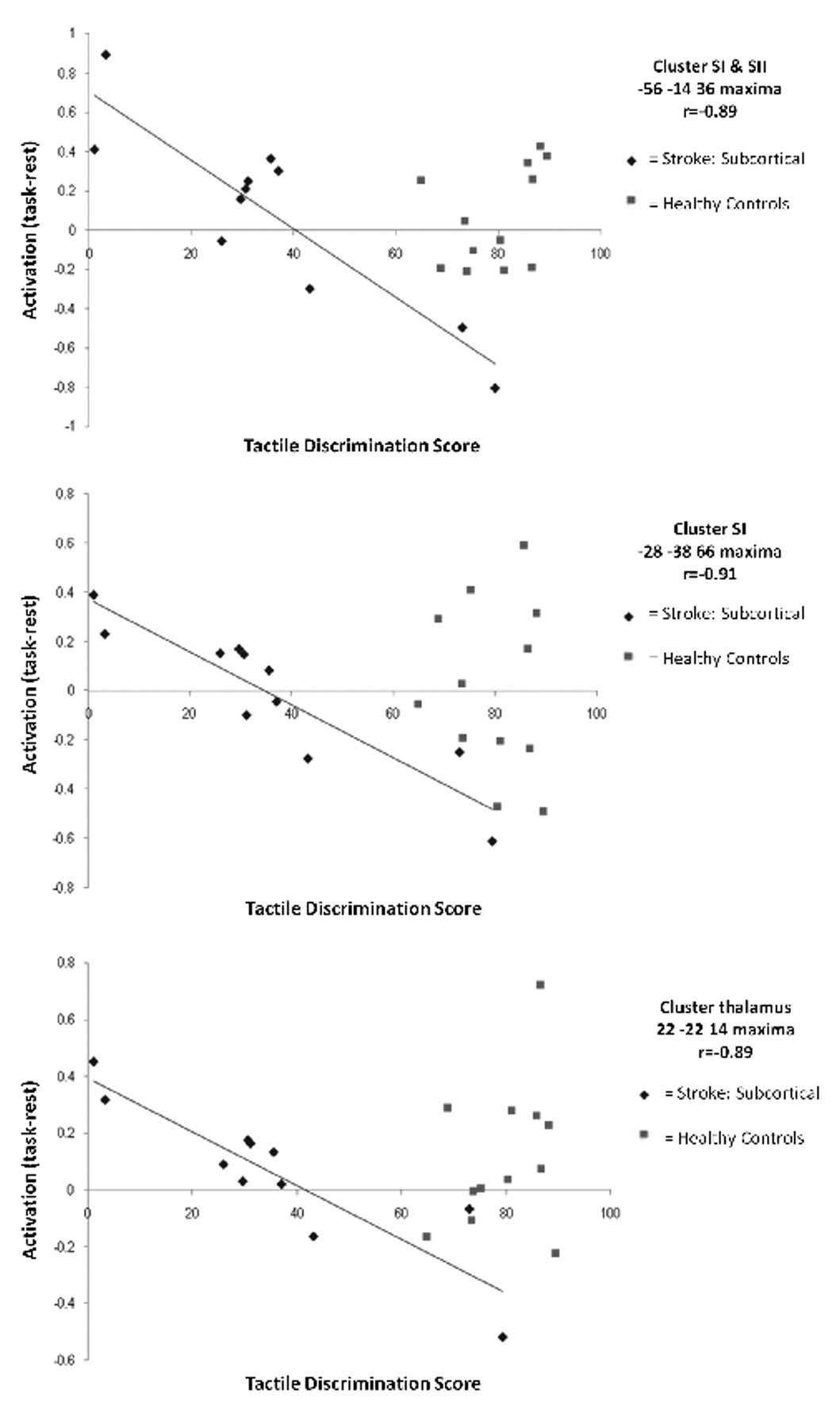
Subcortical group: plots of correlated activity for the affected hand. Scatterplots of relationship between tactile discrimination score (TDT) for the affected hand and task-related activation (task minus rest parameter estimate) for each individual at somatosensory sites that showed significant correlation for the subcortical group (diamonds). A higher tactile discrimination score represents less impairment. Data points for healthy controls (squares) are included to facilitate interpretation. Abbreviations: SI, primary somatosensory cortex; SII, secondary somatosensory cortex
Brain activation during unaffected-hand stimulation and unaffected-hand TDT score were positively correlated in contralesional middle frontal and inferior frontal gyri for the subcortical subgroup (Table 2; Figure 4). A negative correlation was evident in the contralesional occipital gyrus, supplementary motor area, middle temporal gyrus, paracentral lobule, and cerebellum.
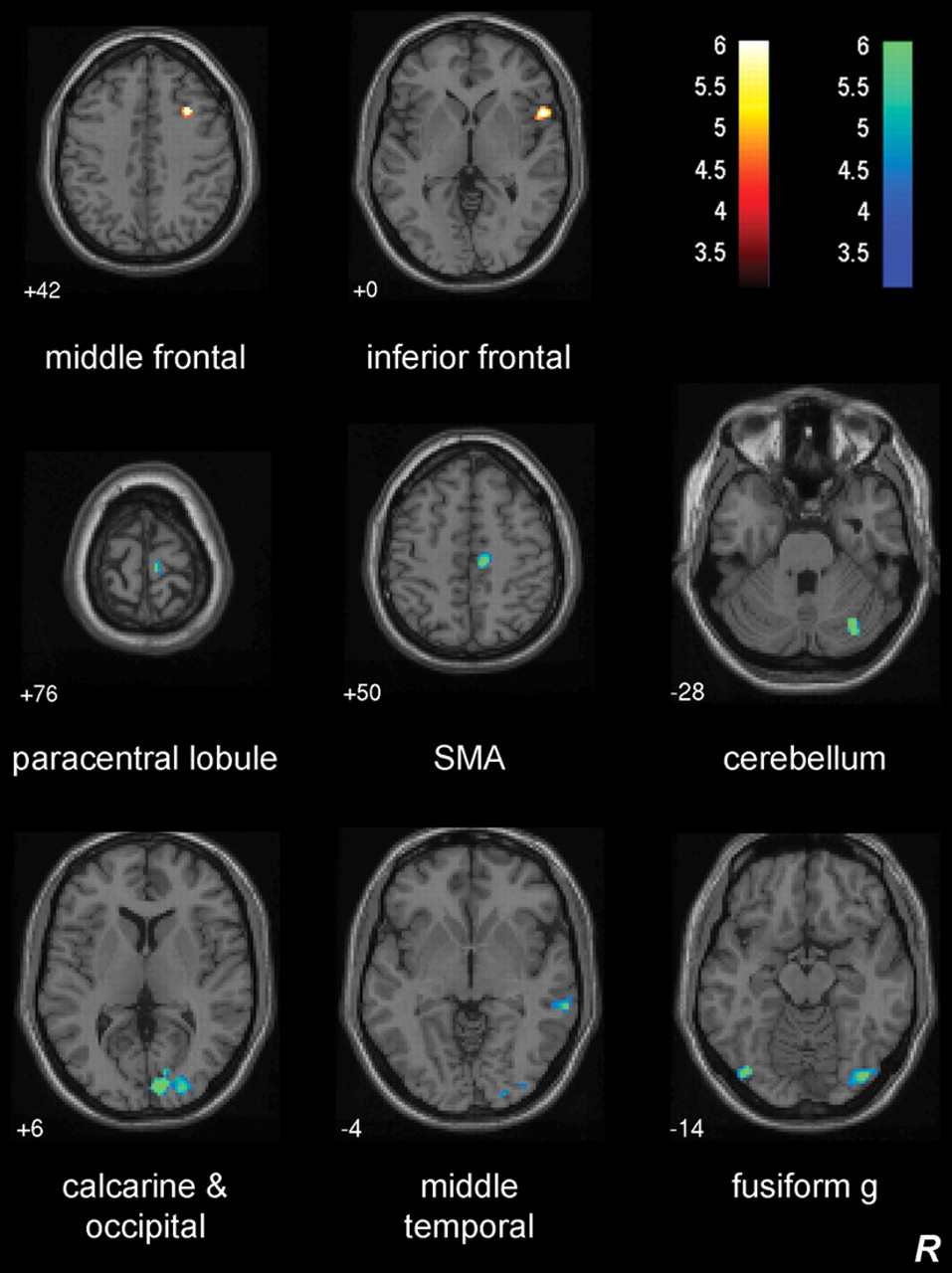
Subcortical group: neural correlates of unaffected-hand touch discrimination. Axial slices showing sites of brain activation correlated with unaffected hand touch discrimination during stimulation of the unaffected hand. Positively correlated activity is shown by t values on a hot color scale and negatively correlated activity using cold colors. Z values represent axial slice position in Montreal Neurological Institute (MNI) space. There were no sites of positive or negative correlation during stimulation of the unaffected hand for the cortical group.
For the cortical group, we did not observe any positive or negative correlations within the somatosensory ROI or at the whole brain level during affected-hand stimulation, although a negative correlation trend was noted in the contralesional thalamus (2, −12, 10; Z = 3.67; P < .05 uncorrected cluster) in our ROI analysis. There were no significant positive or negative correlations during unaffected-hand stimulation.
Investigation of between-group differences of correlated regions for the affected hand of cortical and subcortical groups did not reveal a significant between-group difference in the predefined sensory regions of interest, that is, SI, SII, or the thalamus. However, our whole-brain analysis revealed a significant difference in contralesional superior medial frontal gyrus (2, −60, 34; Z = 4.37; 140 voxels), a region identified as negatively correlated in the subcortical single-group analysis (Table 2). For the unaffected hand,* there was a significant between-group difference in contralesional cerebellum (34, −80, −14; Z = 3.99; 297 voxels); contralesional middle temporal gyrus (64, −34, −4; Z = 4.04; 148 voxels); and middle cingulate cortex/supplementary motor area (SMA) (14, −20, 50; Z = 4.07; 125 voxels).
In the total group, only 1 region showed correlated activity during affected-hand stimulation—namely, the contralesional thalamus (MNI coordinate 6, −10, 8; 68 voxels; Z = 4.22; Figure 5A). The site was identified as the anterior medial thalamus and had a 91% probability of connection with the prefrontal cortex 36 (www.fmrib.ox.ac.uk/connect/). There were no sites of positive correlation. The relationship was not systematically affected by lesion site: individuals with subcortical or cortical lesions were spread throughout the range (Figure 5B). There were no positive or negative correlated sites with unaffected-hand stimulation.
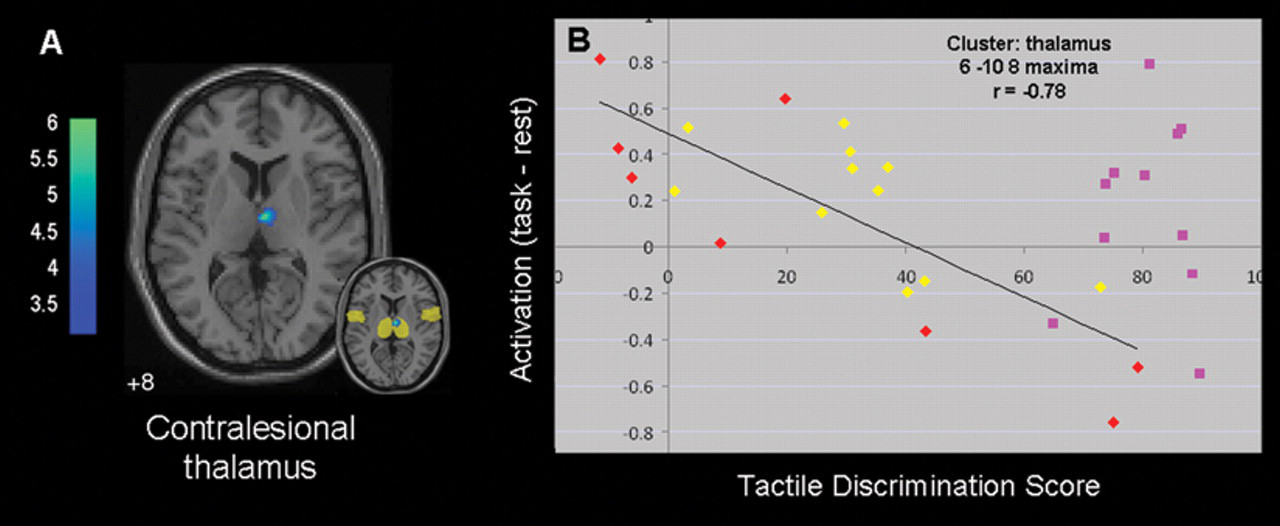
Combined group: neural correlates of affected hand touch discrimination. A. Axial slice depicting brain activation correlated with tactile discrimination during stimulation of the affected hand in the total group (n = 19). Negatively correlated activity is shown using cold colors, which represent t values. There were no significant sites of positive correlation. Inset shows correlated activity overlaid on region-of-interest mask (transparent yellow). B. Plot of relationship between touch discrimination score and task-related activation (task minus rest parameter estimate) for each individual in the contralesional thalamus. Yellow diamonds indicate values for individuals with subcortical lesions, and red diamonds indicate values for those with cortical lesions. A higher tactile discrimination score represents less impairment. Data points for healthy controls (pink squares) are included to facilitate interpretation.
In healthy controls, there were no sites of positive or negative correlated activity in the somatosensory ROI or whole brain for either right- or left-hand stimulation.
Discussion
Our major finding is that touch discrimination impairment after stroke is correlated with brain activity. In the subgroup with lesions in subcortical somatosensory regions, touch outcome was negatively correlated with brain activity in ipsilesional SI (adjacent to the SI hand area activated in our healthy controls), ipsilesional SII, the contralesional thalamus, and distributed attention-based frontal and occipital regions during tactile stimulation of the affected hand. Thus, patients with better touch outcome showed a relative deactivation in these regions. In patients with cortical lesions, we observed little or no correlated activity, likely because of variation in lesion location. Notably, we failed to find a significant difference between subcortical and cortical subgroups in our a priori regions of interest, in particular in the hand representation of SI, in SII, or in the thalamus bilaterally. However, a significant difference between the 2 subgroups at a whole-brain corrected threshold was found only in the contralesional superior medial frontal gyrus. This region is associated with conscious information processing 37 and is reported to be inhibited when the brain needs to divert resources to perceptually demanding tasks. 38 In addition, baseline brain activity in this region is correlated with conscious perception of low-intensity stimuli in healthy participants. 39 Our whole brain finding is hypothesis generating and accords with the notion that poststroke recovery may use different modes and pathways in cortical and subcortical stroke patients. Finally, in our separate analysis of all patients in a single group, we found negatively correlated activity in a single region—namely, the contralesional thalamus. Involvement of the thalamus in the total group is consistent with evidence from animal studies, 40,41 which highlights the fact that gating of sensory inputs, rather than cortical representation alone, is important in recovery. For the therapist, this could suggest the use of intervention approaches that focus on training the ability to attentively discriminate sensory information rather than stimulation-only approaches or bombardment.
Previous studies of somatosensory impairment have charted SI and SII recovery in patients with thalamic lesions and relatively mild impairment or good recovery. 12-15 Rather than simply monitoring the presence or absence of activation, we investigated correlations between brain activation and sensory performance in patients with a wide range of impairment severity. Our findings relate to patients with subcortical and cortical lesions and with good recovery through to severe impairment in the subacute phase of recovery, thus, extending previous findings. They are unlikely to be a result of artifact or nonspecific vascular effects because the correlated activity was differentially observed in somatosensory and distributed regions and involved both hemispheres. All patients and healthy controls were right-hand dominant to minimize variation associated with hand dominance.
Negative Correlation in Somatosensory Regions Following Subcortical Lesions
We observed a negative correlation involving a relative deactivation in SI and SII in those with better touch discrimination following subcortical lesions, commonly involving the thalamus. Post hoc analysis indicated comparable findings in those with only thalamic lesions. This relative deactivation was confirmed through inspection of individual-subject BOLD signal changes in significant clusters (Figure 3). Although this finding differs from positive correlations reported in motor recovery, 42 it is consistent with correlates of somatosensory processing in healthy participants. 17 For subcortical patients with better touch discrimination, one possible mechanism to address the reduced capacity of the infarcted thalamus to inhibit competing input and “gate” task-relevant stimulation 43 would be to establish this modulation locally, in ipsilesional SI. We speculate that the relative deactivation observed in ipsilesional SI may reflect a functionally effective inhibition, 17 based on evidence in healthy participants that unilateral touch of fingers is associated with phasic deactivation of bilateral SI, in addition to the well-known activation of contralateral SI. 44 In stroke survivors with good touch discrimination, deactivation could help suppress the “noisy” signals from the damaged somatosensory system. What is interesting is that a relative deactivation at this site was also observed in some healthy controls, supporting its role in processing of somatosensory information. Observation of a relatively increased parameter estimate during the task relative to baseline/rest in patients with greater impairment is consistent with prior evidence of selective hypoperfusion in ipsilesional SI at rest following thalamic lesions. 14 Thus, brain activity at rest may also be important.
Neuronal inhibition has been associated with negative fMRI response, 18 consistent with our interpretation of increased local inhibition. The relative deactivation with better touch discrimination was adjacent to the SI hand area activated by our healthy controls. Oxygenation decreases at locations surrounding SI activation (2-4 mm away) are consistent with a negative BOLD fMRI signal and with interpretation of inhibitory synaptic activity and sensory “surround inhibition.” 45 Our SI finding in patients with subcortical lesions and intact SI is consistent with these neurophysiological and behavioral studies that support a role for SI in neuromodulation in addition to function-specific processing.
Correlated activity in the contralesional thalamus with subcortical lesions has a high connection probability with SI and the posterior parietal cortex 36 (Table 2). Relative deactivation of the contralesional thalamus with good touch outcome would be associated with decreased suppression of ipsilesional SI and decreased activity in contralesional SI during stimulation of the affected hand. 13 This would help redress the imbalance in cortical activity between hemispheres that commonly occurs after stroke and is an important determinant of impairment and recovery. 1,3,46 The observed correlation in the contralesional thalamus could be influenced by interthalamic communication with the ipsilesional thalamus 47 (affected in 8 patients) or via corticothalamic connections that have an important role in both rapid and more slowly emerging forms of thalamic plasticity. 40,41,48
Involvement of Distributed Frontal and Occipital Regions With Subcortical Lesions
Distributed neural networks and top-down modulation may facilitate somatosensory information processing after stroke, particularly following subcortical lesions. Perceptual awareness occurs from an interaction between specialized sensory cortices and a higher order frontoparietal attention network. 38,39 Our observed changes in frontoparietal regions, involving superior and middle frontal gyri and the superior parietal lobe, are consistent with their role in attention-based top-down modulation of SI and SII. 38,39 These regions have corticothalamic connections 36 and are part of the prefrontal-thalamic inhibitory system that is involved with optimizing the detection of novel or hard-to-sense stimuli. 49 The superior frontal gyrus, the region also significantly different between groups, is inhibited when the brain needs to divert resources to carry out a perceptually demanding task. 38 A relative deactivation in the superior parietal lobe (away from SI hand area) may produce increased focusing in the SI hand area through generalized suppression of background activity. 50 Areas activated preferentially in attentive perception of touch include the superior frontal cortex 51 and middle frontal gyrus, 52 consistent with our findings.
Baseline brain activity in the medial thalamus and the frontoparietal network is also important in perception 39 and may affect information processing following sensory impairment. Healthy participants show a positive relationship between conscious perception of low-intensity somatosensory stimuli and immediately preceding levels of baseline activity in the medial thalamus and frontoparietal cortex—regions thought to relate to vigilance and “external monitoring.” 39 The relative increase in resting signal compared with the task signal, observed in similar regions with better touch outcome, is consistent with the interpretation that baseline brain activity may assist in conscious perception of somatosensory information.
Patients with better touch discrimination might also access functionally effective preexisting, but latent, connections between visual and somatosensory regions during this simple tactile task. Correlated activity in the ipsilesional middle occipital gyrus, contralesional cuneus, calcarine fissure, and lingual gyrus is consistent with activity in visual regions during tactile perception 53 and with effective connectivity of parietal and occipital cortical regions during haptic shape perception. 54 In fact, visual cortical processing may be necessary for normal tactile perception. 55
Lack of Correlated Activity Following Cortical Lesions
There was no evidence of correlated activity following cortical lesions, even with ROI analysis. Cortical lesions involved SI and/or the inferior parietal lobe, supramarginal gyrus, insula, parietal operculum, and superior parietal lobule. Variation in lesion location would reduce the likelihood of finding a correlation, especially in ipsilesional somatosensory regions. However, a trend was observed in the contralesional thalamus. The scatter plot for the total group indicates that the correlation holds for those with cortical infarcts consistent with the observation that they represent the extremes along the regression line. Involvement of the contralesional medial thalamus is consistent with activation of contralesional and medial sites with motor impairment following cortical lesions 5 and has been associated with conscious perception. 39 Systematic investigation of correlated activity in larger samples of patients with homogeneous cortical lesions is indicated.
Role of the Contralesional Thalamus in the Total Group
Despite heterogeneous infarct location, activity in the contralesional thalamus in the total sample correlated with touch severity. Although the thalamus is postulated to play a role in poststroke somatosensory recovery, 13 to our knowledge, this is the first direct demonstration of its involvement in humans poststroke using functional imaging. Corticothalamic pathways play a role in modifying the size, strength, and selectivity of thalamocortical receptive fields and/or establishing cortico-cortical communication via the thalamus. 48 The correlated maxima in the medial thalamus had a 91% probability of connection with the prefrontal cortex 36 ; a region that has a role in intermodality inhibition of irrelevant inputs, regulation of task-relevant touch stimulation, and influences SI of both hemispheres via the thalamus. 43 If the prefrontal-thalamic system was active, we would expect a net inhibitory effect on the thalamus, reflected as a deactivation, as observed with good touch outcome. In contrast, those with severe impairment did not show this, suggesting a reduced opportunity to regulate task-relevant stimuli to the contralesional and ipsilesional SI. Thus, our findings highlight the role of the contralesional thalamus via its prefrontal connections and influence on bilateral SI and its potential to be accessed irrespective of lesion location.
Correlated Activity With Stimulation of the Unaffected Hand
To our knowledge, evidence of correlated activity with sensory stimulation of the unaffected hand has not been previously reported, despite evidence of somatosensory impairment in the hand ipsilateral to the lesion in addition to the more typical contralateral loss. 8 Processing of tactile information requires integration of information from both sides of the body. Better touch sensation in the unaffected hand after subcortical lesions was associated with increased activity in contralesional middle frontal and inferior frontal gyri, regions involved in attentive processing of sensory information. 39 A negative correlation with the contralesional (rather than ipsilesional) cerebellum may be influenced by descending influences from the impaired hemisphere. These findings highlight the need to evaluate the role of the unaffected hand and interactions between hands in sensory recovery. Use of bilateral approaches in therapy require systematic investigation.
Conclusion
We report patterns of correlated brain activity associated with touch discrimination in subcortical and cortical somatosensory stroke. For subcortical lesions, somatosensory and distributed region activity was negatively correlated. Ipsilesional SI and SII showed a relative deactivation in those with better touch discrimination. We speculate that this is consistent with functionally effective local inhibition in SI, as reported in healthy controls. 17,44 Distributed regions involving attention and visual circuits were also correlated, suggesting the possible role of top-down modulation and corticothalamic connections in sensory processing after stroke. We did not observe significant associations for the cortical lesion group alone. Despite this, we did not find significant between-group differences in our a priori regions of interest: SI, SII, or the thalamus in either hemisphere. We did however find a between-group difference in correlated activity in the superior frontal gyrus, a region known to be involved in attentive processing of sensory information. Correlated deactivation in the contralesional thalamus in the total group with heterogeneous lesions and partial recovery is consistent with evidence from animal studies 40,41 that thalamic plasticity and “gating” of sensory information, rather than primary representation alone, is important in recovery. Together with this literature, our finding of correlated activity in somatosensory and frontal regions suggests the importance of modulation of conscious sensation poststroke via local inhibition in the ipsilesional SI and frontal attention regions and via thalamocortical networks bilaterally. Further prospective experiments to tease apart the role of these regions in attentional modulation of sensory perception after stroke are warranted. A review of the theoretical and empirical foundations of sensory rehabilitation support approaches directed at attentive discrimination of sensory stimuli rather than a bombardment approach, 9 consistent with our findings and an attentional modulation hypothesis. Thus, our findings have significance in relation to mechanisms of sensory processing following poststroke impairment as well as direct therapeutic relevance.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: The authors received financial support for the research and authorship of this article from the National Health and Medical Research Council (NHMRC) of Australia [project grant number 307902; and Career Development Award number 307905 to LMC]; an Australian Research Council Future Fellowship [number FT0992299 to LMC]; the Austin Hospital Medical Research Foundation; the National Stroke Research Institute of Australia and by the Victorian Government’s Operational Infrastructure Support Program. The funding sources had no role in conduct of the study or writing of the report.
*
The commonly used term unaffected hand has been used to refer to the hand ipsilateral to the lesion. It should be noted, however, that this hand may also show impairment in addition to the contralateral hand that is traditionally known to be impaired.
