Abstract
Background. Individuals with brain impairment (BI) are less active than the general population and have increased risk of chronic disease. Objective. This controlled trial evaluated the efficacy of a physical activity (PA) intervention for community-dwelling adults with BI. Methods. A total of 43 adults with BI (27 male, 16 female; age 38.1 ± 11.9 years; stage of change 1-3) who walked as their primary means of locomotion were allocated to an intervention (n = 23) or control (n = 20) condition. The intervention comprised 10 face-to-face home visits over 12 weeks, including a tailored combination of stage-matched behavior change activities, exercise prescription, community access facilitation, and relapse prevention strategies. The control group received 10 face-to-face visits over 12 weeks to promote sun safety, healthy sleep, and oral health. Primary outcomes were daily activity counts and minutes of moderate-to-vigorous-intensity PA (MVPA) measured with the ActiGraph GT1M at baseline (0 weeks), postintervention (12 weeks) and follow-up (24 weeks). Between-group differences were evaluated for statistical significance using repeated-measures ANOVA. Results. MVPA for the intervention group increased significantly from baseline to 12 weeks (20.8 ± 3.1 to 31.2 ± 3.1 min/d; P = .01), but differences between baseline and 24 weeks were nonsignificant (20.8 ± 3.1 to 25.3 ± 3.2 min/d; P = .28). MVPA changes for the control group were negligible and nonsignificant. Between-group differences for change in MVPA were significant at 12 weeks (P = .03) but not at 24 weeks (P = .49). Conclusion. The 12-week intervention effectively increased adoption of PA in a sample of community-dwelling adults with BI immediately after the intervention but not at follow-up. Future studies should explore strategies to foster maintenance of PA participation.
Introduction
There is unequivocal evidence that physical activity (PA) is essential for good health. Physical inactivity is the fourth leading cause of death from noncommunicable disease worldwide and is estimated to contribute to more than 3 million preventable deaths each year, including deaths from coronary heart disease, hypertension, colon cancer, and diabetes mellitus.1,2 Based on this evidence, it is recommended that adults should accumulate 30 minutes of moderate-to-vigorous-intensity PA (MVPA) per day.3-5
Brain impairment (BI) is defined as a significant loss or abnormality in the brain structure. 6 A range of health conditions may lead to BI, including the following: cerebral palsy, a disorder of movement or posture resulting from a nonprogressive lesion in the developing brain 7 ; traumatic brain injury, which is an injury resulting from an external force and causing altered brain function 8 ; and stroke, a focal disturbance of cerebral function of vascular origin. 9 These 3 conditions are particularly important because of their high incidence: cerebral palsy affects 1 per 500 live births, 10 traumatic brain injury affects 200 per 100 000 people per year, 11 and stroke affects 94 per 100 000 people per year. 12 Regardless of etiology, BI results in abnormal brain function, including impaired cognitive, social/behavioral, and/or sensorimotor function. Additionally, people with BI participate in significantly less PA than the general population.13-15 Evidence indicates that the benefits accrued by people with BI who increase their PA includes improved cardiorespiratory fitness, muscle strength, and functional independence.16-20 To increase PA participation and improve health, fitness, and functioning in people with BI, it is critical to develop interventions that effectively promote free-living PA participation in this population.
To date, interventions to increase PA in adults with BI have largely been ineffective.21-25 These programs have primarily been conducted in dedicated, fixed facilities, including hospital-based or outpatient-based facilities, and consisted of structured exercise prescription with limited use of tailored evidence-based behavior change strategies.21-25 There is preliminary evidence to indicate that tailored exercise counseling alone, or in combination with supervised exercise, improves PA participation in community-dwelling stroke survivors. 26 However, this approach has not been tested in more general samples of adults with BI. Additionally, evidence indicates that key contextual factors such as lack of transportation and/or finances are significant barriers to attending exercise programs at fixed facilities for individuals with a disability, including BI. 27 PA interventions delivered in an individual’s home or local community help reduce these barriers and increase the prospect of sustained participation, particularly for individuals who have PA preferences beyond structured exercise in a fixed facility. Therefore, studies evaluating the efficacy of lifestyle PA interventions delivered in the home or local community are required.
Community-based rehabilitation is a theory-driven model of service delivery that has been successfully applied to the provision of a range of rehabilitation services for individuals with BI, including physiotherapy, occupational therapy, and community reintegration services. 28 Community-based rehabilitation refers to services delivered in an individual’s home or community (ie, physical surrounds that they typically inhabit or could easily access). 28 An advantage of the community-based rehabilitation model of service delivery is that infrastructure costs are negligible when compared with establishing and maintaining a purpose-built, fixed facility. Community-based rehabilitation aims to enhance the natural support systems within an individual’s home and community and may be a potentially effective service delivery model for promoting PA in people with BI. 29
Lifestyle PA interventions are an effective means of promoting PA 30 and could be delivered using a community-based rehabilitation service delivery model. Lifestyle interventions utilize a range of evidence-based PA promotion strategies (eg, goal setting, self-monitoring, self-reward, and social support) to help individuals increase their physically active behavior.28,30 These strategies increase PA because of their effect on theory-based mediators of PA, including self-efficacy,31-33 social support,34,35 and decisional balance. 35 The effectiveness of lifestyle interventions can be enhanced by using a stage of change algorithm to categorize each participant into 1 of 5 stages of motivational readiness for change, 36 thereby permitting selection of PA promotion strategies that best suit an individual’s motivational readiness for change. This process is known as stage matching an intervention. Delivering a lifestyle PA intervention using a community-based service delivery model allows PA promotion strategies to be discussed, designed, and evaluated in the same setting in which PA will be adopted and maintained. Lifestyle interventions have the potential to be seamlessly integrated into already established community-based rehabilitation services for individuals with BI. To date, studies evaluating the effectiveness of community-based, lifestyle PA interventions for adults with BI have not been conducted.
The purpose of this controlled clinical trial was to evaluate the efficacy of a PA intervention for community-dwelling adults with BI. The intervention adopted a community-based rehabilitation approach to deliver a stage-matched lifestyle PA intervention, and the primary outcome was objectively measured PA. Secondary outcome measures were decisional balance, self-efficacy, and social support.
Methods
Participants
People meeting the following inclusion criteria were eligible to participate: men or women, 18 to 60 years old, with a BI resulting from cerebral palsy, traumatic brain injury, or stroke; living in a community setting within a 150-km radius of the University of Queensland; with walking as their primary means of locomotion (with or without aids); who were deemed medically safe to participate in moderate intensity PA 37 ; who were currently insufficiently active for health; and who had the cognitive ability to participate in the behavior change activities integral to the intervention. Exclusion criteria were the following: lower-limb surgery in the past 6 months or scheduled during the intervention; lower-limb botox in the past 3 months or scheduled during the intervention; serious, regular, or uncontrolled substance abuse; documented violent or aggressive behavior; and suicidal ideation.
Ethical approval for this study was received from The University of Queensland Medical Research Ethics Committee (HMS10/0410) and the Cerebral Palsy League of Queensland Research Ethics Committee (CPL-2011-003). The study was registered through the Australian New Zealand Clinical Trials Registry (ANZCTR) ACTRN: 12610000864022.
Recruitment
Participants were recruited through gatekeeper referral agencies, including community-based service providers for people with BI, and outpatient rehabilitation services in Brisbane from January 2011 to May 2012. Referral agencies were provided with an overview of the intervention and the inclusion and exclusion criteria and asked to identify participants who met the criteria and who would be interested in taking part in a healthy lifestyle program addressing the following health behaviors: PA, sun safety, oral health, and sleep. Potential participants were subsequently contacted by a member of the research team who formally assessed study eligibility and provided details of the study in terms commensurate with their level of understanding. Following an opportunity for questions, people who were eligible and interested in participating provided written, informed consent. Eligibility assessment included the administration of the Marcus and Simkin 38 staging algorithm. People in the first 3 stages of change were, by definition, insufficiently active for health and, therefore, eligible to participate. These stages were the following: stage 1 (precontemplation—not currently physically active and have no intention of increasing their activity in the next 6 months); stage 2 (contemplation—not currently physically active but intend to become physically active in the next 6 months); and stage 3 (preparation—completing some PA however not a volume sufficient to meet the PA guidelines for health). 38
Study Design
A stratified systematic allocation protocol was used to assign participants to either a PA intervention or a control condition. As participants were enrolled in the study, they were first stratified by stage of change, ensuring that both study arms had equal numbers of inactive or insufficiently active participants. Within each stratum, the first participant recruited was allocated to the intervention or control group by flipping a coin. The second participant assigned to the cell was allocated to the alternative condition (eg, if the first participant assigned to the cell was allocated to the control group, the second participant assigned to the cell was automatically allocated to the intervention group). This process was conducted by a researcher external to the study.
Participants allocated to the intervention group were informed that they would be receiving 2 health behavior interventions: the PA intervention first, then an intervention addressing sun safety, oral health, and sleep. Participants allocated to the control intervention were told the reverse.
The participant flow diagram is shown in Figure 1. A total of 54 referrals were received from gatekeeper agencies, and participants with BI were assessed for eligibility. Among them, 11 were excluded because they did not meet the inclusion criteria (n = 5), suffered an unrelated illness or injury (n = 5), or declined to participate (n = 1). The remaining 43 participants were stratified by stage of change (n = 19, stage of change 1 and 2; n = 24, stage of change 3) and allocated into the PA intervention (n = 23) or the control condition (n = 20). One participant from the intervention group and 1 participant from the control group were unable to complete the postintervention measures because of medical issues not associated with their participation in the program.
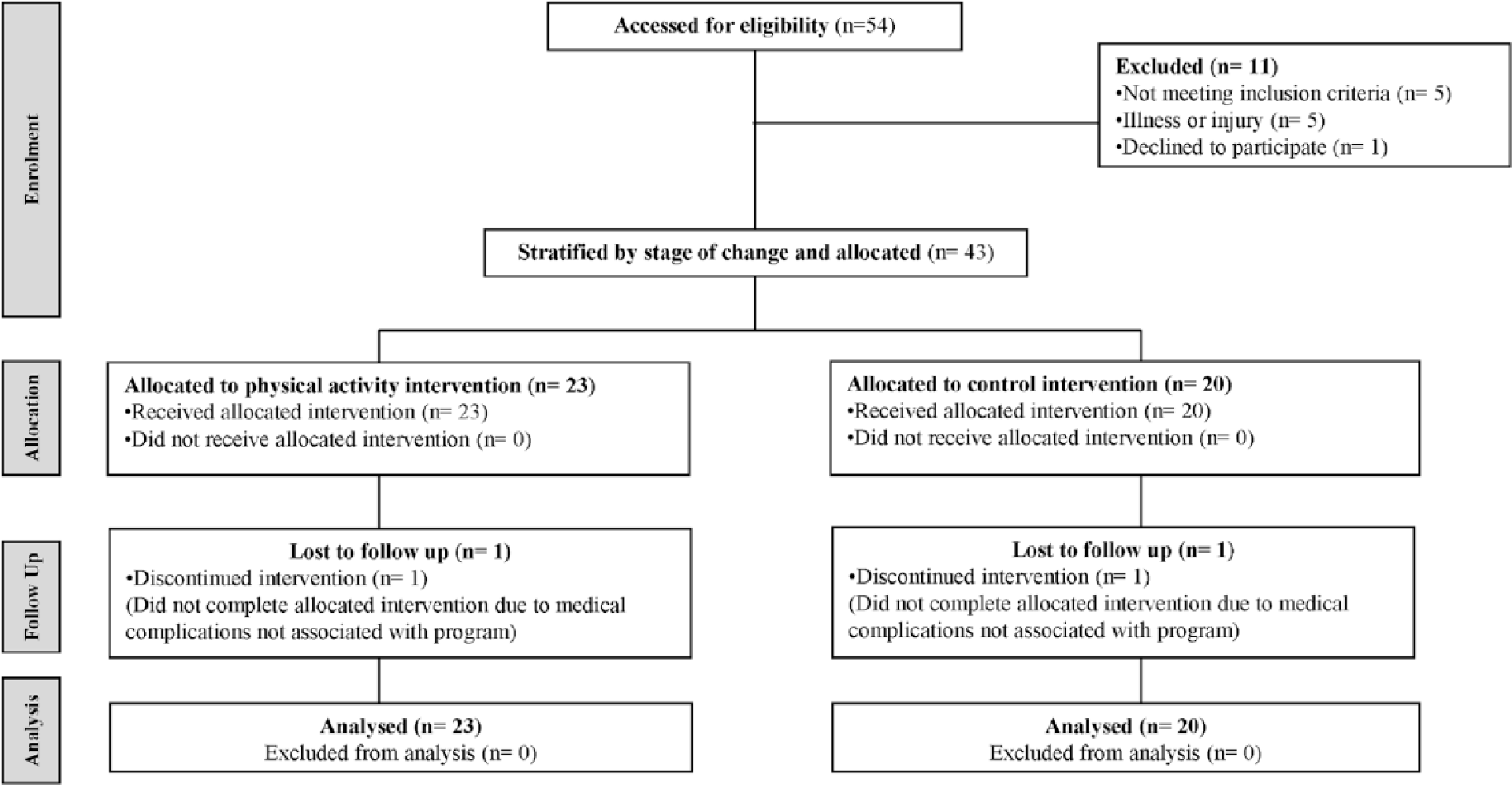
Participant flow diagram for participant inclusion, allocation, follow-up, and analysis.
Physical Activity Intervention
Table 1 provides an overview of the PA intervention. All participants transitioned through steps 1, 2, and 3 during 10 face-to-face sessions over 12 weeks. Each session was 1 hour in duration and the intersession time varied depending on the needs of the participant and the stage of the program, with typically more sessions in the first 6 weeks as community access was arranged, skills were learned, and habits were established. Session frequency diminished in the second 6 weeks, with more emphasis on fostering independence and self-management.
Physical Activity Intervention Outline. a
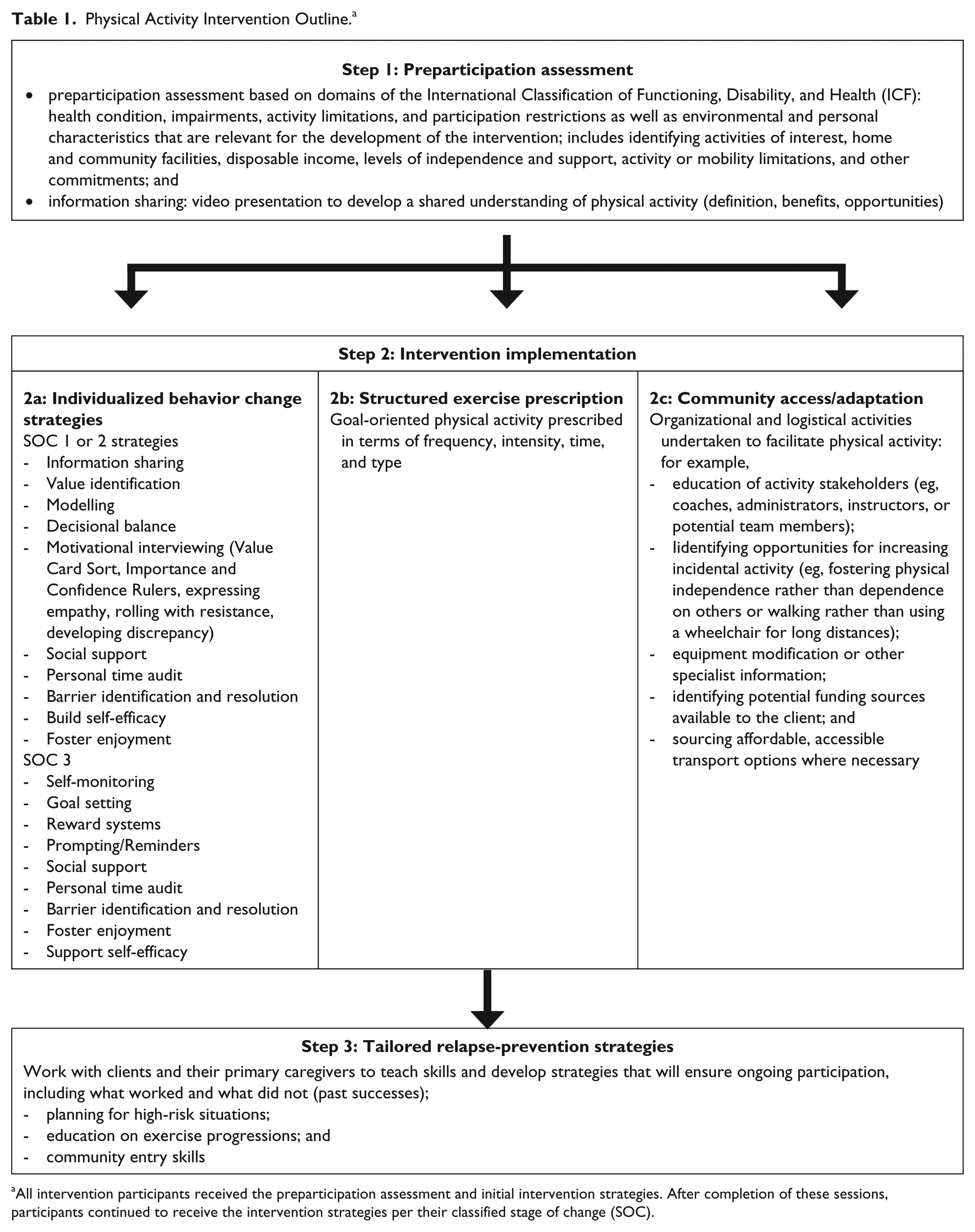
All intervention participants received the preparticipation assessment and initial intervention strategies. After completion of these sessions, participants continued to receive the intervention strategies per their classified stage of change (SOC).
Step 1 was a standardized preparticipation assessment conducted to permit each participant’s PA intervention to be individually tailored. This step had 3 main components, the first being a series of questions and activities based on the domains of the International Classification of Functioning, Disability, and Health (ICF), 6 which aimed to identify key factors that would impede or enhance PA participation for a particular individual. Domains assessed included each participant’s impairments, activity limitations and participation restrictions, and personal and environmental factors likely to affect PA participation. 39 An information-sharing exercise, conducted through a short video discussing the definition and benefits of PA, was also completed in order to establish a shared understanding of the concept of PA. 40 The motivational readiness of each participant, previously evaluated using a stage of change algorithm, was used to permit stage matching of the intervention. 36
Step 2, the intervention implementation, entailed the application of stage-matched behavior change strategies (step 2a), exercise prescription (step 2b), and strategies to promote community access (step 2c). For the purposes of this program, participants who were originally classified as stage of change 1 and 2 were pooled because the information sharing exercise conducted in step 1 explicitly required participants to consider the benefits they may accrue by becoming physically active, so any genuine stage 1 participants who remained in the program would have become stage 2. For participants in stages 1 and 2, emphasis was placed on developing a discrepancy between the participant’s current PA behavior and their broader goals and values, evaluating the benefits and barriers and eliciting change talk for increasing PA. For stage 3 participants, emphasis was placed on strategies that would assist them to increase their activity from insufficient levels to sufficient levels (eg, goal setting, reward systems, and self-monitoring). 36 The specific strategies used for each participant were selected from those presented in Table 1 (step 2a), all of which have been described elsewhere.41-43 The selection of the strategies used was based on the outcomes of step 1 and any subsequent session.
The behavior change strategies implemented as part of step 2a led to a broad array of PAs, which usually included a mixture of structured, graduated exercise prescription (step 2b) as well as sports, outdoor activities, practical activities, or cultural activities facilitated by step 2c. Exercise prescription and recommendations for the type of activity were individually tailored using the information gained during the preparticipation assessment. The structured exercise prescribed in step 2b aimed to assist participants achieve personally relevant goals (eg, to be physically stronger, to be able to walk further, or to “get back into the garden”). Community access facilitation included activities such as liaising with community-based PA providers, equipment modification, and/or the identification of financial assistance programs and transportation. 44
Step 3 entailed the delivery of individually tailored relapse prevention strategies designed to help participants identify potential situations in which their PA routine may be disrupted or stopped and to formulate appropriate strategies to restart or continue with their PA program. 45
During steps 2 and 3, the face-to-face sessions were supplemented by reminder calls, email messages, or electronic notifications to reinforce the session outcomes and provide a prompt for additional action where required (eg, completing an activity log or completing a daily walk). The extent to which these methods were used depended on individual participant preferences, availability and use of the various communication methods, and the individual’s impairment profile (especially impaired memory).
Control Condition
Like the PA intervention, the control condition comprised ten 1-hour face-to-face counseling sessions delivered over 12 weeks. The distribution of face-to-face sessions was similar to the PA intervention, and control participants received an equivalent number of reminder calls and electronic notifications. By controlling for an equivalent amount of contact, this group acted as an attention control, thereby reducing the potential confounding influence of the effect that therapist contact can have on the response of the participants. The control intervention aimed to promote oral health, sun safety, and healthy sleep. Sun safety, oral health, and sleep were chosen because they are health behaviors that can significantly reduce disease risk in the target population and because they can be influenced using behavior change strategies similar to those used for promoting PA. The face-to-face sessions for the control condition followed a sequence similar to the PA intervention outline presented in Table 1, aiming to (1) develop a shared understanding of the target health behaviors, including their importance and the recommended guidelines for good health; (2) discuss the participant’s thoughts and feelings regarding the behavior; (3) discuss the perceived importance of, and their current engagement in, the health behaviors; and (4) use evidence-based behavior change strategies to increase engagement in the target behaviors.
After participants had completed 12 weeks of either the PA intervention or the control condition, there was a 12-week follow-up period (week 13 to week 24) during which there was no contact—either face-to-face or electronic—between the research team and participants. At the conclusion of the follow-up period (24 weeks), participants received an abridged version of the program they had not already received: participants who initially completed the PA intervention received the control intervention, and participants who initially received the control intervention received the PA intervention. Both the PA and control interventions were delivered by the same accredited exercise physiologist (AEP), who had experience in exercise prescription for individuals with BI, behavior change strategies for increasing PA, and the principles of promoting oral health, sun safety, and sleep. Delivery of both interventions by the same person ensured that interpractitioner factors did not influence results.
Primary Outcome Measure: Physical Activity
Both primary and secondary outcome measures were taken at 3 time points: baseline (0 weeks), immediately postintervention (12 weeks), and at follow-up (24 weeks) and were performed in the participant’s home by one assessor who was blinded to the allocation of the participants.
PA was measured using an ActiGraph GT1M accelerometer-based motion sensor (ActiGraph, Pensacola, FL). Using portable indirect calorimetry as a criterion measure, Tweedy and Trost 46 established that the ActiGraph could validly differentiate between MVPA and activity that was light or sedentary in adults with brain injury.
Participants wore the ActiGraph for 7 consecutive days at baseline (0 weeks), postintervention (12 weeks), and follow-up (24 weeks). The ActiGraph was positioned on the midaxilla line at the level of the iliac crest on the side of the body least affected by neurological impairment. Where there was no obvious difference between sides, the ActiGraph was worn on the participant’s dominant side. Participants were provided with a log book with pictures showing how to position the monitor correctly. During these data collection periods, participants received a reminder card, phone calls, or text messages to ensure that they wore the ActiGraph for the full 7-day period. A user-defined time sampling interval (epoch) 47 of 1 minute was used in this study. On completion of the 7-day monitoring period, the accelerometer data were downloaded, and a customized Visual Basic Excel Macro was used to determine daily wear time, daily average counts per minute (cpm), and daily time spent in MVPA. Counts were classified as either MVPA or light/sedentary using the cut-point validated by Tweedy and Trost. 46
For a monitoring day to be considered valid and included for analysis, 600 minutes of wear time was required. 48 Non–wear time was classified as consecutive 0 counts for at least 60 consecutive minutes, allowing for up to 2 consecutive 1-minute epochs with nonzero counts less than or equal to 100 counts. Daily wear time was calculated by subtracting the estimated non–wear time from the total monitoring time. Participants with 4 or more valid monitoring days were included in the analyses.
Secondary Outcome Measures
Self-efficacy was measured using the 5-item scale developed by Marcus et al. 49 A 5-point Likert-type scale was used to rate each item, with 1 indicating not at all confident, and 5 very confident. In this sample, Cronbach’s α was acceptable, at .68. Social support was measured using the 13-item scale developed by Sallis et al. 50 A 5-point Likert-type scale was used to rate the frequency of support received from family and friends in the 3-months prior, with 1 indicating none, and 5 indicating very often. Social support scores were calculated for family and friends separately by computing the sum of the items on the measure. 50 In this sample, Cronbach’s αs for the friends and family subscales were .83 and .86, respectively. Decisional balance was measured using the 16-item scale developed by Marcus et al, 51 which included 6 items representing the avoidance of exercise (cons) and 10 items representing the positive perceptions of exercise (pros). A 5-point Likert-type scale was used to rate how important each statement was, with 1 indicating not at all important, and 5 indicating extremely important. The average of the 10 pro items and the 6 con items was computed. The difference in the averages (ie, average pro score − average con score) was taken as the decisional balance score. In this sample Cronbach’s α was .86 for the positive items and .62 for the negative items.
Statistical Analysis
Descriptive statistics, including means and SDs, were calculated for the primary and secondary outcomes. Differences between the groups at baseline were tested using independent t tests for continuous variables and χ2 tests for categorical variables. For the χ2 analysis, Fisher exact tests were used when cell counts were less than 5. Differences between the intervention and control group changes in PA from baseline to postintervention and baseline to follow-up were evaluated for statistical significance using repeated-measures ANOVA. Because daily MVPA and wear time were positively correlated, accelerometer wear time was included as a covariate. All participants (n = 43) enrolled in the study were included in the final analysis. Assuming a 2-tailed α level of .05, a SD of 25 cpm for change in daily counts per minute, and a SD of 5 minutes for change in daily minutes of MVPA, the sample size of 23 intervention and 20 control participants (n = 43) provided 80% power to detect change scores of 22 cpm and 4.4 minutes of MVPA, respectively. All analyses were conducted on an intention-to-treat basis, with missing follow-up data conservatively imputed using the last observation carried forward method. General linear models were implemented in SAS (version 9.3) using the MIXED procedure, which allows for the specification of the covariance structure for repeated observations. Statistical significance was set at an α level of .05.
Results
Participants
Participant characteristics are presented in Table 2. A total of 43 participants (27 male and 16 female), with a mean age of 38.1 ± 11.9 years, participated in the study (n = 23, intervention; n = 20, control). Etiology included traumatic brain injury (n = 21), stroke (n = 20), and cerebral palsy (n = 2). A total of 40 participants were unemployed, and 3 worked part-time (≤20 h/wk) at the time of the intervention. All participants had completed junior high school, with 42% of participants having completed a trade, vocational certificate, or university degree prior to their injury. All but 2 participants were classified as community ambulators per the Hoffer Functional Ambulation Scale. 52 There were no significant differences between participants in the intervention and control groups with respect to participant characteristics and outcome variables, with the exception of height. Although participants in the control group were significantly taller than participants in the intervention group (P = .034), the 7-cm difference was considered to be of little practical significance. All participants in both the PA and control interventions completed all 10 face-to-face sessions. Although there was some variability in the mean time for each contact session, there was no significant difference between the mean session lengths for the intervention and control groups: the average session times for the intervention and control groups were 55.0 ± 3.8 minutes and 56.9 ± 3.9 minutes, respectively.
Participant Characteristics at Baseline (n = 43). a
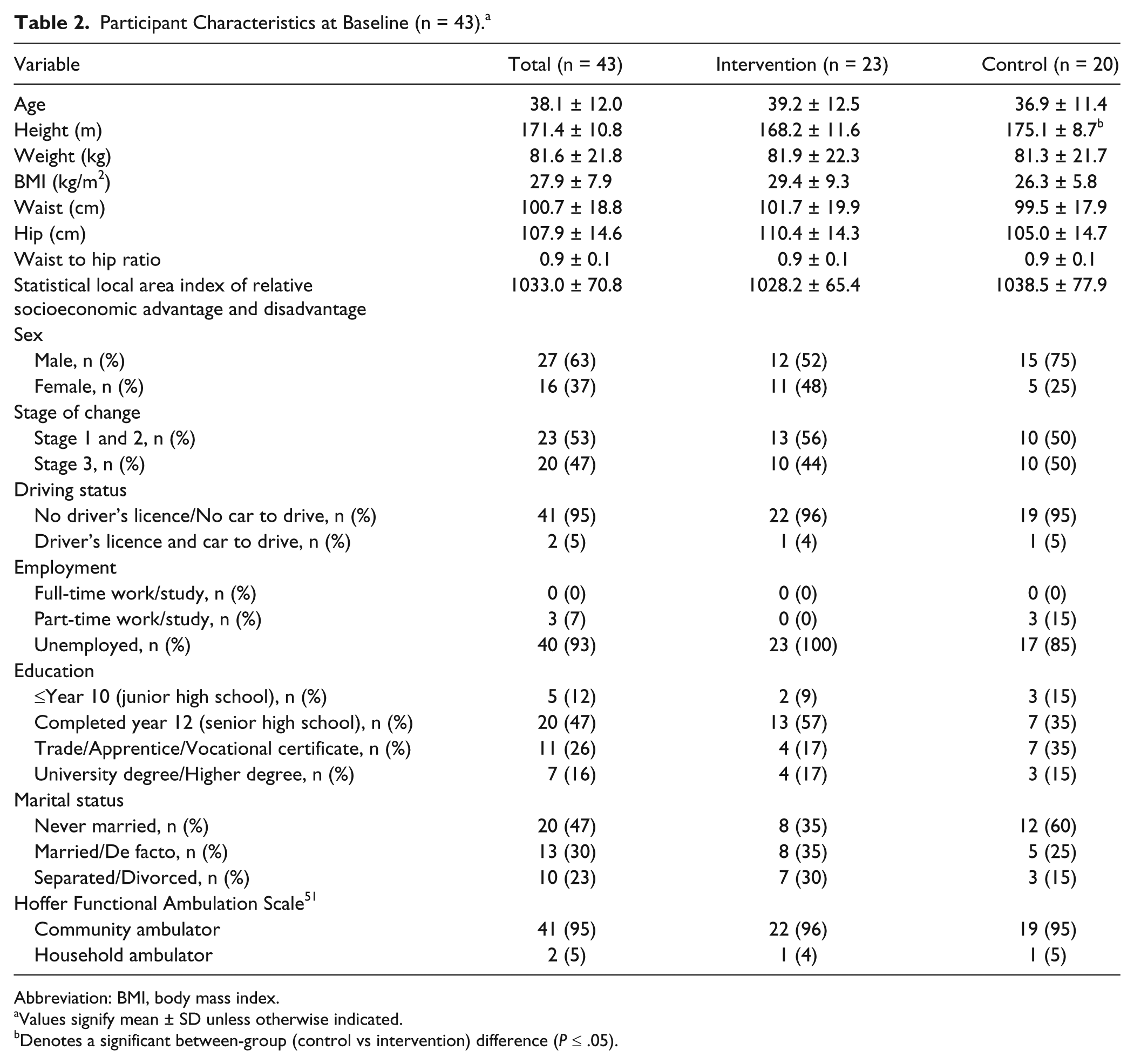
Abbreviation: BMI, body mass index.
Values signify mean ± SD unless otherwise indicated.
Denotes a significant between-group (control vs intervention) difference (P ≤ .05).
Primary Outcome Measure: Physical Activity
Between- and within-group differences in the changes in accelerometer counts per minute and MVPA are presented in Table 3. Mean counts per minute in the intervention group increased significantly from baseline to postintervention (mean change = 60.9 ± 20.7 cpm; P = .004), but changes from baseline to follow-up were nonsignificant (mean change = 34.5 ± 21.0 cpm; P = .11). Changes in counts per minute over time were small and nonsignificant for participants in the control group. The increase in daily counts per minute from baseline to postintervention among intervention participants was significantly greater than that observed among the control participants (net difference = 71.9 ± 30.8 cpm; P = .02). However, the between-group difference for the change from baseline to follow-up was nonsignificant (net difference = 29.3 ± 31.0 cpm; P = .35).
Changes in Outcome Measure (From Baseline to Postintervention and Baseline to 3-Month Follow-up) Scores and Net Differences Between the Intervention and Control Groups for All Outcome Measures.
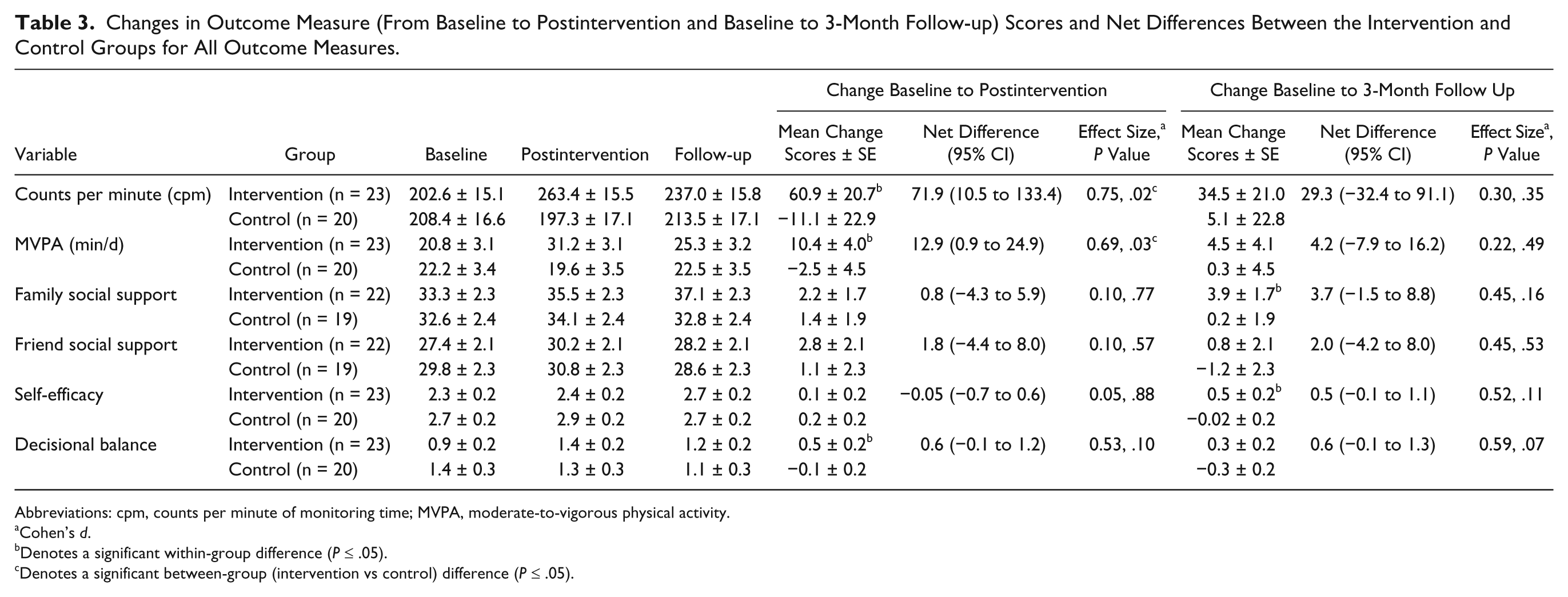
Abbreviations: cpm, counts per minute of monitoring time; MVPA, moderate-to-vigorous physical activity.
Cohen’s d.
Denotes a significant within-group difference (P ≤ .05).
Denotes a significant between-group (intervention vs control) difference (P ≤ .05).
Participants in the intervention group exhibited a significant increase in MVPA from baseline to postintervention (mean change = 10.4 ± 4.0 min/d; P = .01), but changes from baseline to follow-up were nonsignificant (mean change = 4.5 ± 4.1 min/d; P = .28). For participants in the control group, changes in MVPA were small and nonsignificant. After controlling for daily wear time, the increase in MVPA from baseline to postintervention among intervention participants was significantly greater than that observed among control participants (net difference = 12.9 ± 6.0 min/d; P = .03); however, the between-group difference in the change from baseline to follow-up was nonsignificant (net difference = 4.2 ± 6.1 min/d; P = .49).
Secondary Outcome Measures
Changes in the secondary outcomes are also presented in Table 3. Although participants in the intervention group increased their self-efficacy, social support, and decisional balance scores, with the exception of decisional balance, none of the changes was significant postintervention. Within-group changes in family social support and self-efficacy scores for the intervention group were significant at the 24-week follow-up. There were no significant within-group changes for these variables among control participants. Net differences for changes in social support, self-efficacy, and decisional balance were nonsignificant from baseline to postintervention (12 weeks) and from baseline to follow-up (24 weeks).
Discussion
This is the first study to evaluate a community-based, lifestyle PA intervention for adults with BI. The findings indicate that the intervention was effective at increasing daily PA levels in adults with BI. However, there was limited maintenance of the increase 3 months postintervention, indicating that the intervention was successful at promoting PA adoption but was limited in its capacity to promote PA maintenance. These findings are noteworthy, given the particularly low volumes of PA in this population and the significant cognitive, physical, and environmental barriers that individuals with BI face. Although the absolute volume of change may appear modest, evidence indicates that these changes are clinically important. A recent review by Powell et al 53 concluded that even very small increases in physically active behavior confers significant health benefits for people who are completing less than 150 minutes of moderate-intensity PA per week. Furthermore, the review suggests that interventions that prevent people from becoming more physically inactive may also be considered successful. The results of this study indicate that a community-based, lifestyle PA intervention is potentially an efficacious approach for promoting adoption of PA in adults with BI.
Previous studies evaluating interventions to promote PA in individuals with BI have been conducted predominantly in fixed facilities rather than being community based. The shortcoming of fixed-facility PA programs is that penetration is low, being restricted to those who can travel to the facility, and the prospect of maintaining increases after the conclusion of the intervention is reduced because the facilities and staffing support typically cease at study conclusion. A community-based, lifestyle PA program addresses these shortcomings and was a particular strength of this study. Conducting the intervention in the participant’s home also addressed a range of common barriers for PA participation, including, but not limited to, lack of transportation, lack of access to an accessible facility, and the costs associated with participating in a PA program. 27 All participants in the intervention and control group completed all 10 sessions of their allocated program. This high attendance rate is attributed to the intervention being conducted at both a convenient time and convenient location for the participant, removing the requirements for transportation and accessibility. Additionally, the prospects of translating results from this study into practice are enhanced by the fact that a range of other health-related services (eg, physiotherapy, occupational therapy, social work) are currently delivered to people with BI using the community-based rehabilitation model of service delivery, and the intervention evaluated in this study could be seamlessly incorporated into such services.
Relative to baseline, participants in the intervention group exhibited increases in PA at 12 weeks and 24 weeks. However, during the follow-up period, activity levels in the intervention group declined marginally by approximately 10%. Compared with the PA intervention, participants in the control group exhibited a small decrease in PA from baseline (0 weeks) to postintervention (12 weeks) and a small increase in PA from postintervention (12 weeks) to follow-up (24 weeks). The small changes in PA participation in the control group can be attributed to the natural variance in PA participation over time. As a result of these differences, the between-group difference in the change from baseline to follow-up (24 weeks) was not statistically significant. By design, participants did not have contact with the intervention staff during the follow-up period, and it is possible that the withdrawal disrupted the routine that had been established. A more gradual reduction in the frequency and duration of home visits and the provision of relatively cheap remote support strategies (eg, reminders via phone, text, email) may be effective ways of assisting participants to maintain their positive changes in behavior. 54 For example, evidence indicates that follow-up phone calls can improve maintenance of PA following intervention withdrawal.55,56 Accordingly, future research should evaluate strategies to provide support beyond the intervention period.
Although the intervention resulted in significant increases in PA, there were no concomitant changes in the secondary outcome measures, indicating that changes were mediated by variables not measured in the current study. This finding is consistent with the results of previous intervention studies that also reported increases in PA without concomitant changes in the theory-based mediators of PA behavior. 57 A possible explanation for our finding is that, because the constructs targeted and their associated measures were adapted from the nondisabled literature, the changes in PA in our sample were mediated by cognitive, social, and environmental influences specific to people with BI that were not measured (eg, independence in living/function, physical environment or facility access, or expected benefits31-33,58). Further research on the social-ecological influences of PA in adults with BI is, thus, warranted.
The participants in this study included adults with BIs that included cerebral palsy, traumatic brain injury, and stroke. Despite the diversity in the health conditions, from the perspective of promoting PA, the participants were sufficiently similar to be treated as one group. Specifically, the diagnostic groups included in the study are characterized by having impairments to the brain that affect physical, cognitive, or social/behavioral functioning and low levels of PA participation. Additionally, all participants included in this study walked as their primary means of locomotion and lived in a community setting (ie, noninstitutional), ensuring a relatively uniform level of functioning. From the perspective of promoting PA, these shared features mean that many of the strategies used to promote PA are similar, addressing issues such as community access, building social support, and improving self-efficacy for PA. Furthermore, individual tailoring of behavior change strategies according to the physical, cognitive, and behavioral profile of each participant is a central feature of the intervention evaluated in this study, making it appropriate for each of the diagnostic groups included. Previously published PA promotion studies have used even more diverse populations.24,59
This study has several limitations that warrant consideration. First, participants lived in urban settings located within a 150-km radius of a large metropolitan university. Consequently, the findings may not be generalizable to individuals with BI residing in rural and remote areas. Future studies should explore the viability of implementing the intervention in rural areas, including exploring the need for adaptations such as increasing the proportion of the intervention delivered through channels such as internet and phone delivery. Second, because of the difficulties of objectively measuring PA in wheelchair users, the study excluded individuals who used a wheelchair as their primary form of locomotion. The results of this study are also only generalizable to individuals who have the cognitive capacity to engage in intervention activities, such as identifying values of interest, discussing behavior patterns, and setting goals for increased PA participation. Third, although accelerometers are capable of assessing the pattern and intensity of activity, as well as total accumulated activity, they cannot be worn while swimming and are unable to measure the increased energy expenditure associated with bike riding, walking up stairs, or walking while carrying a load. However, this limitation is applied to both the intervention and control participants, and participation in problematic activities was minimal. Fourth, the last observation carried forward method was used to impute missing values. This method is limited because of the potential risk of underestimating the variance in the outcome and inflating the type I error rate. However, this approach is conservative in nature (ie, assumes no change over time), is transparent to the reader, and allows for intention-to-treat analyses to be conducted. 60 Finally, although the method for allocating participants to the intervention and control conditions could not be considered true random assignment, the systematic allocation protocol resulted in 2 groups with no significant differences with respect to participant characteristics or baseline outcome measures, and therefore the impact of any selection bias would be minimal.
These limitations were offset by several strengths. The use of an objective measure of PA, in comparison with a self-report instrument, is advantageous given the cognitive impairments associated with BI. Studies evaluating validity of self-report measures in adults with BI identify difficulties in recalling the duration of activities within the monitoring period and quantifying the time spent being active in assisted activities (eg, the time they spend independently completing housework in comparison to the time that they are provided with in-home assistance). These problems may result in participants either over- or underreporting their time spent active. 61 The veracity of outcomes from this study is enhanced because changes in PA were measured objectively using an instrument specifically validated in this population. 46 Second, the study included an attention-control condition to control for the amount of face-to-face contact with the therapist. Finally, the intervention made use of evidence-based behavior change strategies tailored to the participant’s PA preferences and stage of change.
Conclusions/Implications
In conclusion, a 12-week community-based, lifestyle intervention significantly increased PA in a sample of community-dwelling adults with BI immediately after the intervention but not at follow-up. The results of this study are sufficiently positive to warrant conducting additional efficacy trials, using trained practitioners in a variety of clinical practice and rehabilitation settings. Future studies should explore strategies to foster maintenance of PA participation, including providing support to participants via phone or internet. Additional research for adapting the intervention for effective delivery in rural and remote settings should also be a future research priority.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Sean Tweedy’s position is supported by the Motor Accident Insurance Commission, Australia.
