Abstract
Background and Objective. Favorable prognosis of the upper limb depends on preservation or return of voluntary finger extension (FE) early after stroke. The present study aimed to determine the effects of modified constraint-induced movement therapy (mCIMT) and electromyography-triggered neuromuscular stimulation (EMG-NMS) on upper limb capacity early poststroke. Methods. A total of 159 ischemic stroke patients were included: 58 patients with a favorable prognosis (>10° of FE) were randomly allocated to 3 weeks of mCIMT or usual care only; 101 patients with an unfavorable prognosis were allocated to 3-week EMG-NMS or usual care only. Both interventions started within 14 days poststroke, lasted up until 5 weeks, focused at preservation or return of FE. Results. Upper limb capacity was measured with the Action Research Arm Test (ARAT), assessed weekly within the first 5 weeks poststroke and at postassessments at 8, 12, and 26 weeks. Clinically relevant differences in ARAT in favor of mCIMT were found after 5, 8, and 12 weeks poststroke (respectively, 6, 7, and 7 points; P < .05), but not after 26 weeks. We did not find statistically significant differences between mCIMT and usual care on impairment measures, such as the Fugl-Meyer assessment of the arm (FMA-UE). EMG-NMS did not result in significant differences. Conclusions. Three weeks of early mCIMT is superior to usual care in terms of regaining upper limb capacity in patients with a favorable prognosis; 3 weeks of EMG-NMS in patients with an unfavorable prognosis is not beneficial. Despite meaningful improvements in upper limb capacity, no evidence was found that the time-dependent neurological improvements early poststroke are significantly influenced by either mCIMT or EMG-NMS.
Keywords
Introduction
Several prospective cohort studies among stroke patients have shown that the functional outcome of the upper limb is largely defined within the first 5 weeks poststroke and is mainly driven by (yet poorly understood) mechanisms of spontaneous neurological recovery.1,2 Observational studies showed that the presence of some voluntary finger extension (FE) within 72 hours is a favorable indicator for the return of dexterity poststroke.3,4 This suggests that early control of FE is an important prognostic factor in stratifying patients for upper limb intervention trials early poststroke. 2
For those with a favorable prognosis, indicated by some voluntary FE early poststroke, constraint-induced movement therapy (CIMT) or a modified version (mCIMT) may benefit arm-hand activities and self-reported hand function in daily life. 5 The number of phase II trials on mCIMT within the first days or weeks poststroke is however small and findings are rather inconclusive. For example, Dromerick et al 6 showed in a proof of concept trial that 1 or 2 hours mCIMT per working day for 2 weeks was not superior to an equal dosage of usual care, whereas a high dose of 3 hours mCIMT per working day resulted in less improvement on functional outcome measured with the Action Research Arm Test (ARAT) at 3 months poststroke.
For those with an unfavorable prognosis for functional outcome at 6 months, that is, patients without voluntary FE,1,3,4 no evidence-based therapies have been reported so far. In subacute and chronic stroke, innovative therapies such as electromyography-triggered neuromuscular stimulation (EMG-NMS) of the finger extensors to improve voluntary control have shown promise in terms of increasing active range of motion.7-11 Furthermore, several studies suggest that EMG-NMS may produce changes in cortical activation patterns and excitability in chronic stroke.12,13 For example, Shin et al 13 showed in a small proof of concept trial (n = 14) that a daily 30-minute program for 10 weeks shifted cortical activation patterns as seen in functional magnetic resonance imaging from the ipsilateral sensorimotor cortex to the contralateral sensorimotor cortex in chronic stroke. Despite the growing evidence for enhanced levels of homeostatic neuroplasticity in the first weeks poststroke, 14 early started EMG-NMS trials for patients without FE are lacking in this restricted time window.
The first objective of the present study was to investigate the effects of an early mCIMT program on recovery of upper limb capacity during the first 6 months, starting within 14 days poststroke in patients with some voluntary FE. Our second objective was to investigate the effects of early EMG-NMS on the recovery of voluntary FE and upper limb capacity during the first 6 months, starting within 14 days poststroke in patients with no voluntary control of the finger extensors. We hypothesized that an intensive 3-week mCIMT program would result in a clinically meaningful improvement in ARAT scores compared with usual care alone. For the patients with an unfavorable prognosis we hypothesized that a higher percentage of patients (10% or more increase) would regain some dexterity (ARAT score >9 points on a maximum of 57 points) if they received intensive daily EMG-NMS for 3 weeks, compared with usual care alone.
Materials and Methods
Details of the present study regarding research questions, hypotheses, design, patient selection and stratification, interventions, primary and secondary outcomes, and power and statistical analyses were reported in a design article in 2008. 15
Design
We conducted a stratified, multicenter, observer-blinded randomized controlled trial with 8 repeated measurements within the first 26 weeks poststroke. After stratifying patients within the first 14 days after stroke to either the mCIMT trial (if they had voluntary FE of 10° or more) or the EMG-NMS trial (no voluntary FE), randomization to the experimental treatment or usual care was performed online by a restricted minimization randomization procedure. Randomization was executed by 2 staff members not involved in the treatment protocol (GK and HA).
Two trained observers (RN and CW) who were blinded to the treatment allocation performed all clinical measurements at baseline (before randomization), and at 2, 3, and 4 weeks of follow-up during face-to-face sessions at the location of initial admission and at the location of subsequent rehabilitation. Postintervention assessments were performed at 5, 8, 12, and 26 weeks after stroke. The same assessor performed the serial assessments for an individual subject in both trials. Patients were not blinded to the intervention, however they remained naïve as to the supposed efficacy of the 2 intervention conditions.
Both clinical trials were part of the EXPLICIT-stroke program. EXPLICIT-stroke is an acronym for EXplaining PLastICITy after stroke and was funded for 5 years by the Netherlands Organisation for Health Research and Development (ZonMw: No. 890000001). The study was approved by the Medical Ethics Review Committees of Leiden University Medical Center (No. P08.035) and the Dutch Central Committee on Research Involving Human Subjects (CCMO: No. NL21396.058.08), and was registered in the Netherlands Trial Register (NTR, www.trialregister.nl, TC1424).
Patient Selection
For both trials, we selected patients with (1) first-ever ischemic stroke in one of the cerebral hemispheres; (2) upper limb paresis according to National Institutes of Health Stroke Score (NIHSS) item 5; (3) baseline ARAT score of ≤53 on a maximum of 57 points; (4) ability to communicate and comprehend (Mini Mental State Examination ≥23 points on a maximum of 30 points); (5) ability to sit independently for at least 30 seconds; (6) 18 to 80 years of age; (7) no successful thrombolysis therapy resulting in upper limb motor recovery and attaining 0 points on NIHSS item 5 of the paretic arm; (8) no musculoskeletal impairments of the upper paretic limb; (9) no additional therapies such as botulinum toxin injections or medication intake that may influence upper limb function in the previous 3 months; (10) willing to participate in an intensive rehabilitation treatment program; and (11) written informed consent. At intake, patients were stratified to either the mCIMT trial if able to voluntarily extend the thumb and/or 2 or more fingers of the affected hand (10° or more), or to the EMG-NMS trial if they could not.
Interventions
Patients allocated to the mCIMT group daily received 60 minutes of supervised intensive graded practice focused on improving task-specific use of the paretic arm and hand, including enhancing FE. Therapy was delivered in either 1 session or split into 2 sessions of 30 minutes, depending on the available time and patients’ tolerance. Time between sessions per day was not controlled for. One hour of mCIMT therapy was chosen to not overload patients in the early phase poststroke.16,17 Patients were instructed to wear a padded safety mitt (Sammons Preston #6727; Sammons Preston, Inc, Bolingbrook, IL, USA) for 3 hours per working day, during 3 consecutive weeks and lasting up until 5 weeks poststroke. Details of the mCIMT treatment protocol have been described previously. 17
Patients allocated to the EMG-NMS group received two sessions of 30 minutes stimulation of the finger extensors each working day, for 3 weeks, using the Stiwell-Med4-system (Ottobock Healthcare Products GmbH, Vienna, Austria). Active participation of the patient was required to reach the EMG threshold during the dorsiflexion movement of fingers and wrist in order to trigger the NMS. When triggered, the Stiwell-Med4-system stimulated for 5 seconds, followed by 25 seconds of rest. In case of absent EMG-activity, patients learned to facilitate extensor activity of wrist and fingers by simultaneously abducting their paretic arm (often described as Soques’s phenomenon) or by simultaneously EMG-triggering of extensors with the nonparetic arm. 15 EMG-NMS therapy was augmented by offering patients visual feedback presented in front of the patient on a computer screen. For this purpose, the amount of voluntary EMG-activity of finger extension was visualized in a computer game in which patients had to raise a hot air balloon above a mountain.
Usual upper limb therapy in both strata consisted of exercise therapy based on recommendations from current Dutch guidelines 18 applied face-to-face by a physical therapist or occupational therapist for 30 minutes per working day executed for 3 consecutive weeks. Patients in the control group with an unfavorable prognosis engaged in passive range-of-motion exercises and facilitation of voluntary movements, whereas those with a favorable prognostic group received exercise therapy according to Dutch guidelines. 18
All therapy sessions were performed in each participating center by two to three trained physiotherapists and/or occupational therapists. The content and duration of therapy was recorded by the therapist in patient diaries. After the 3-week intervention phase, all patients received usual care (ie, about 30 minutes arm-hand treatment per working day) during their stay in the rehabilitation center or nursing home for on average 2 months. The exact duration and type of postintervention usual care was not controlled for, and based on individual needs.
Outcomes
The primary outcome variable for both trials was the ARAT score. 19 The ARAT is a performance test that assesses the ability to perform gross movements and the ability to grasp, move, and release objects of various sizes, weights, and shapes. 19 The original test consists of 19 items rated on 4-point ordinal scales. Removing 4 items enabled the construction of a hierarchical 1-dimensional scale. 20 The ARAT has been shown to be valid, reliable, and responsive. 21 It was applied according to the guidelines developed by Yozbatiran et al. 22 The minimal clinically important difference (MCID) was set at 10% of the range of the scale, that is, 6 points. 23
Secondary outcome variables in both trials were the following Upper Extremity motor function section of the Fugl-Meyer Assessment (FMA-UE), Wolf Motor Function Test (WMFT), Motricity Index of the Upper Extremity (MI-UE), Erasmus modification of the Nottingham Sensory Assessment of the Upper Extremity (EmNSA-UE), Nine-Hole Peg Test (NHPT), Frenchay Arm Test (FAT), Motor Activity Log (MAL), and the Hand domain of the Stroke Impact Scale (SIS-Hand, version 3.0).
The FMA-UE is a reliable and valid motor function test24,25 evaluating the ability to make arm movements outside the synergistic pattern. 26 The WMFT is reliable and valid 27 and consists of 2 strength items, 6 timed tasks, and 9 integrative functional tasks. Assessment involves performance time with a maximum of 120 seconds and a 6-point functional ability scale (FAS). 27 The MI reliably and validly assesses upper limb strength by testing 6 functions rated from 0 to 100 points.28,29 The EmNSA is a 3-point ordinal scale measuring sharp-blunt discrimination, 2-point discrimination, and proprioception. Except for the 2-point discrimination, intra- and interrater reliability are good to excellent (κ = 0.58-1.00). 30 The NHPT is a reliable and valid test assessing manual dexterity31,32 by measuring the speed with which a patient grasps and inserts (and removes) 9 pegs into a grid of vertical holes. The test was discontinued after 50 seconds if the patient was still unable to insert any pegs. 32 Reliability and validity have been assessed and norms are available.31,32 The reliable and valid FAT measures dexterity using 5 functional tasks, scoring on a binominal scale (fail/pass). 32 We used a translated and adapted version of the MAL 33 comprising the 14 original activities. It was used to independently rate how well (6-point quality of movement [QOM] scale) and how much (6-point amount of use [AOU] scale) the paretic arm was used spontaneously to accomplish the activities of daily living outside the laboratory. Reliability and validity of the MAL have been proved in a number of studies. 33 The SIS-Hand was used to evaluate patient-perceived outcome for the paretic upper limb. Each item was scored on a 5-point rating scale from “not difficult at all” to “cannot do at all.” The SIS has shown excellent psychometric properties in terms of concurrent and construct validity, test-retest reliability and responsiveness.34,35
Sample size estimation
Power analysis showed that a sample size of 60 patients (including 10% dropout) was estimated to be sufficient for the mCIMT trial, and 120 (including 10% dropouts) for the EMG-NMS trial. Details are described elsewhere. 15
Statistics
Successful blinding of the assessors for treatment allocation was tested by comparing assessors’ guesses with actual treatment assignment in both trials using a Cohen’s κ statistic. In both trials, we tested for differences in baseline values with Fisher’s exact test or the χ2-test for nominal outcomes, the Mann-Whitney U test for ordinal outcomes, and Student’s t-test for independent groups for other outcomes, assuming equal variances for interval or ratio scales. The assumption of normality was tested with Z-scores for skewness and kurtosis, and by visual inspection. To analyze time series of continuous outcomes, the generalized estimating equations model with a compound symmetry as a covariance structure was used to evaluate differences in overall effects in the experimental and control groups within each trial over the time course of 5, 8, 12, and 26 weeks poststroke. This model included time, group, and the interaction between group and time in the regression model. We added the baseline value of the dependent variable to the regression model. The proportional difference in the number of stroke patients showing some return of FE and dexterity in the EMG-NMS trial was assessed using a dichotomous generalized estimating equations model. Intention-to-treat analyses were performed on all patients, including those with incomplete sets of data. Missing values were not imputed. We calculated β-values and standard errors for the group × time interaction effects and, subsequently, used the Wald test to obtain P values. All hypotheses were tested 2-tailed, with an α = .05. All analyses were carried out in SPSS, version 20.
Results
Between October 2008 and November 2013, approximately 4300 patients were assessed for eligibility, and 159 were selected (Figure 1). No adverse effects were reported and none of the dropouts were related to type of therapy in either trial. Forty-one of the 464 (8.8%) measurements in the mCIMT trial and 63 of the 808 (7.8%) measurements in the EMG-NMS trial were missing. Comparing observers’ guesses about treatment allocation and actual allocation showed 89 matches for the 159 patients, which was not statistically significant (P = .132).
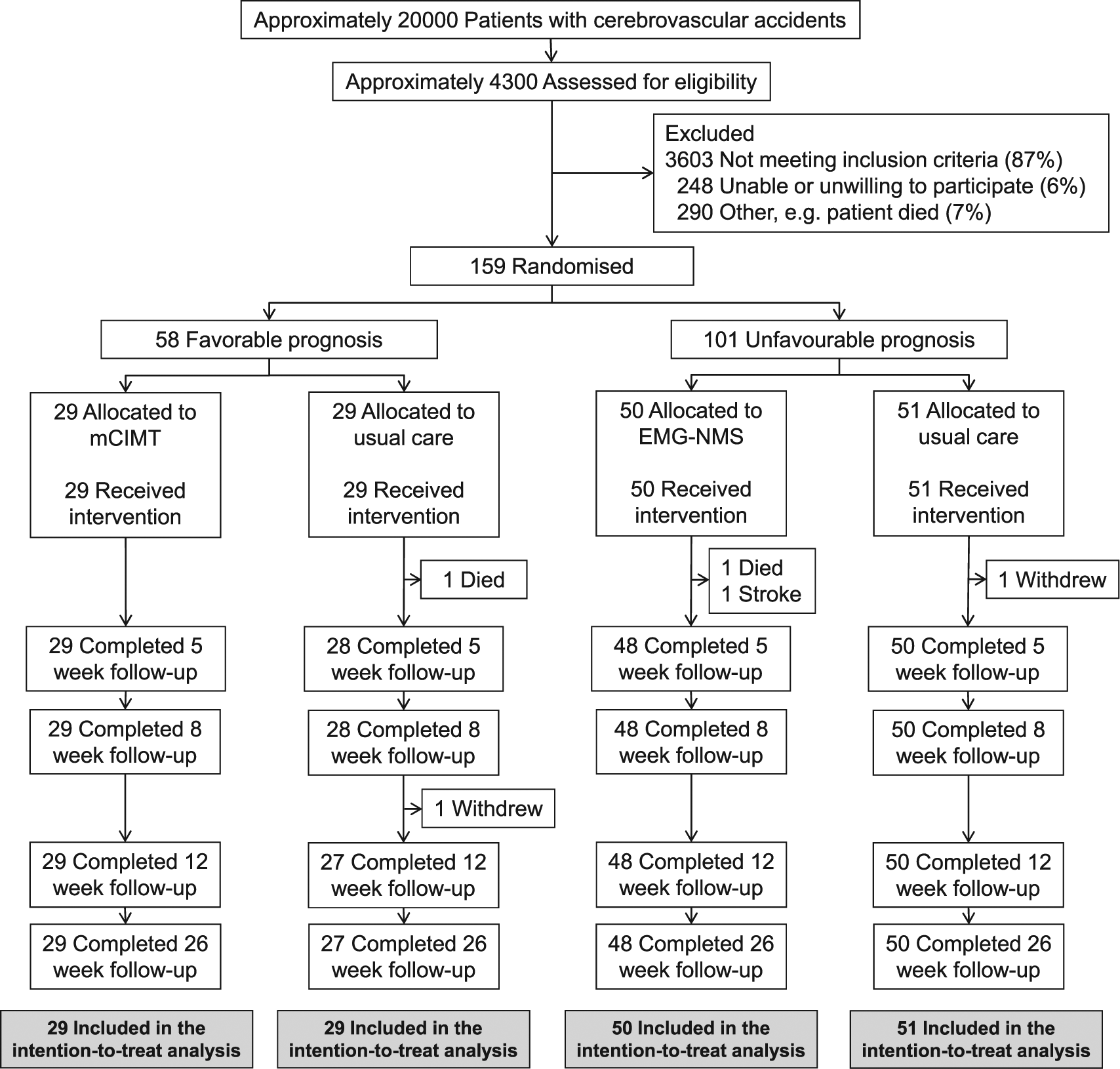
Inclusion flow diagram. The total amount of patients with cerebrovascular accidents was estimated using the number of admitted patients in each participating center. mCIMT: modified constrained-induced movement therapy; EMG-NMS, electromyography-triggered neuromuscular stimulation.
Table 1 shows patients’ baseline characteristics of each trial. Mean start of both trials was at 8 days (SD = 4; interquartile range = 5-10) poststroke. No significant differences were found with respect to demographic, primary, or secondary outcomes at baseline. Table 2 shows the absolute values at 5, 8, 12, and 26 weeks, and the β(SE) and P values for the time × group interaction effects from 2 to 5, 8, 12, and 26 weeks, corrected for baseline. A mean significant time × group interaction effect was found in favor of mCIMT compared with usual care after 5 (P = .011), 8 (P = .002), and 12 (P = .023) weeks poststroke, with respectively 6, 7, and 7 points difference on the primary outcome measure ARAT (Figure 2). In addition, a significant time × group interaction effect was found for SIS-Hand after 8 weeks poststroke in favor of mCIMT in comparison to usual care (P = .038).
Baseline Characteristics of First-Ever Ischemic Stroke Patients Allocated to mCIMT, EMG-NMS, or Usual Care.
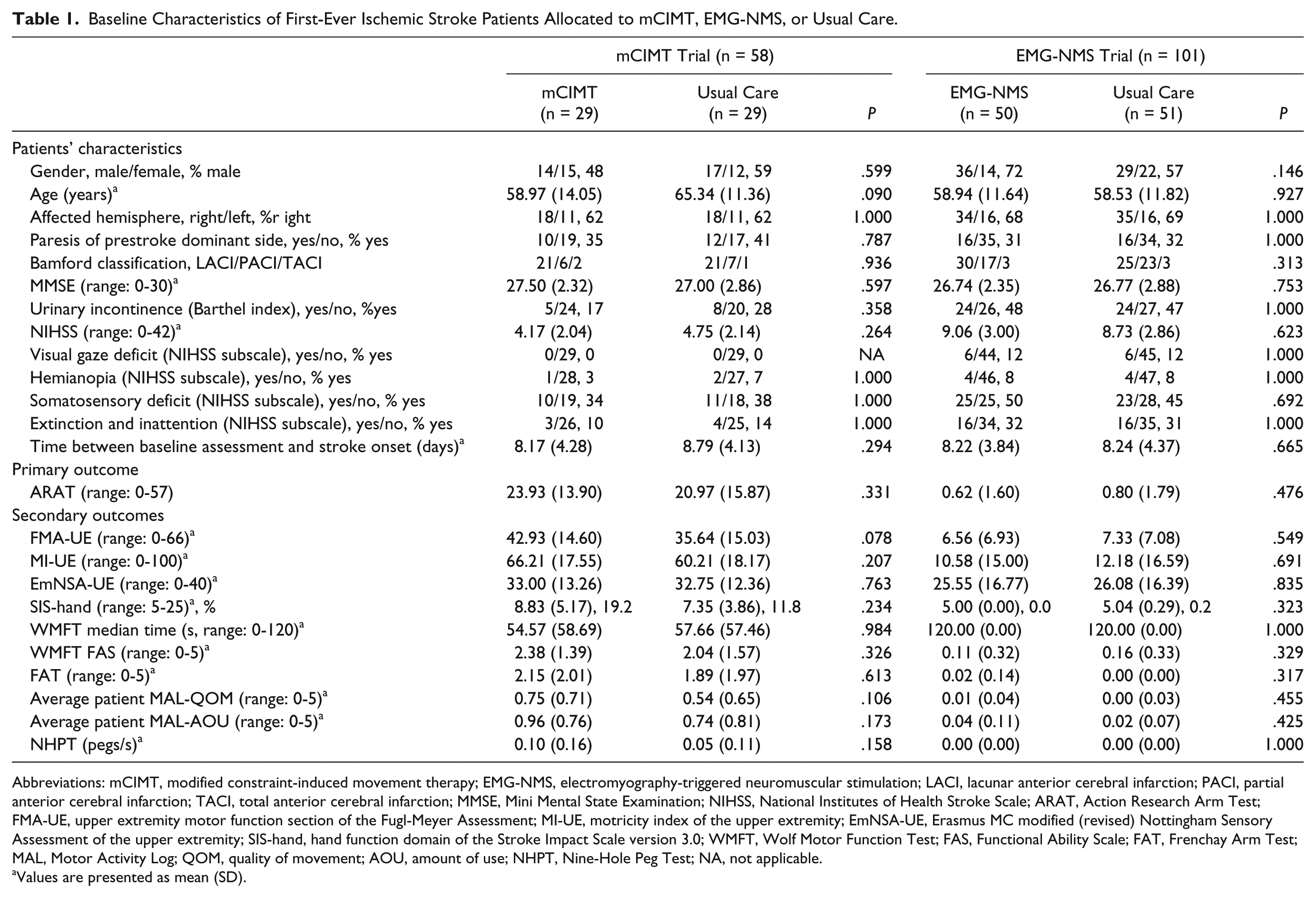
Abbreviations: mCIMT, modified constraint-induced movement therapy; EMG-NMS, electromyography-triggered neuromuscular stimulation; LACI, lacunar anterior cerebral infarction; PACI, partial anterior cerebral infarction; TACI, total anterior cerebral infarction; MMSE, Mini Mental State Examination; NIHSS, National Institutes of Health Stroke Scale; ARAT, Action Research Arm Test; FMA-UE, upper extremity motor function section of the Fugl-Meyer Assessment; MI-UE, motricity index of the upper extremity; EmNSA-UE, Erasmus MC modified (revised) Nottingham Sensory Assessment of the upper extremity; SIS-hand, hand function domain of the Stroke Impact Scale version 3.0; WMFT, Wolf Motor Function Test; FAS, Functional Ability Scale; FAT, Frenchay Arm Test; MAL, Motor Activity Log; QOM, quality of movement; AOU, amount of use; NHPT, Nine-Hole Peg Test; NA, not applicable.
Values are presented as mean (SD).
Effects of Early mCIMT and EMG-NMS on the Primary and Secondary Outcome Measures. a
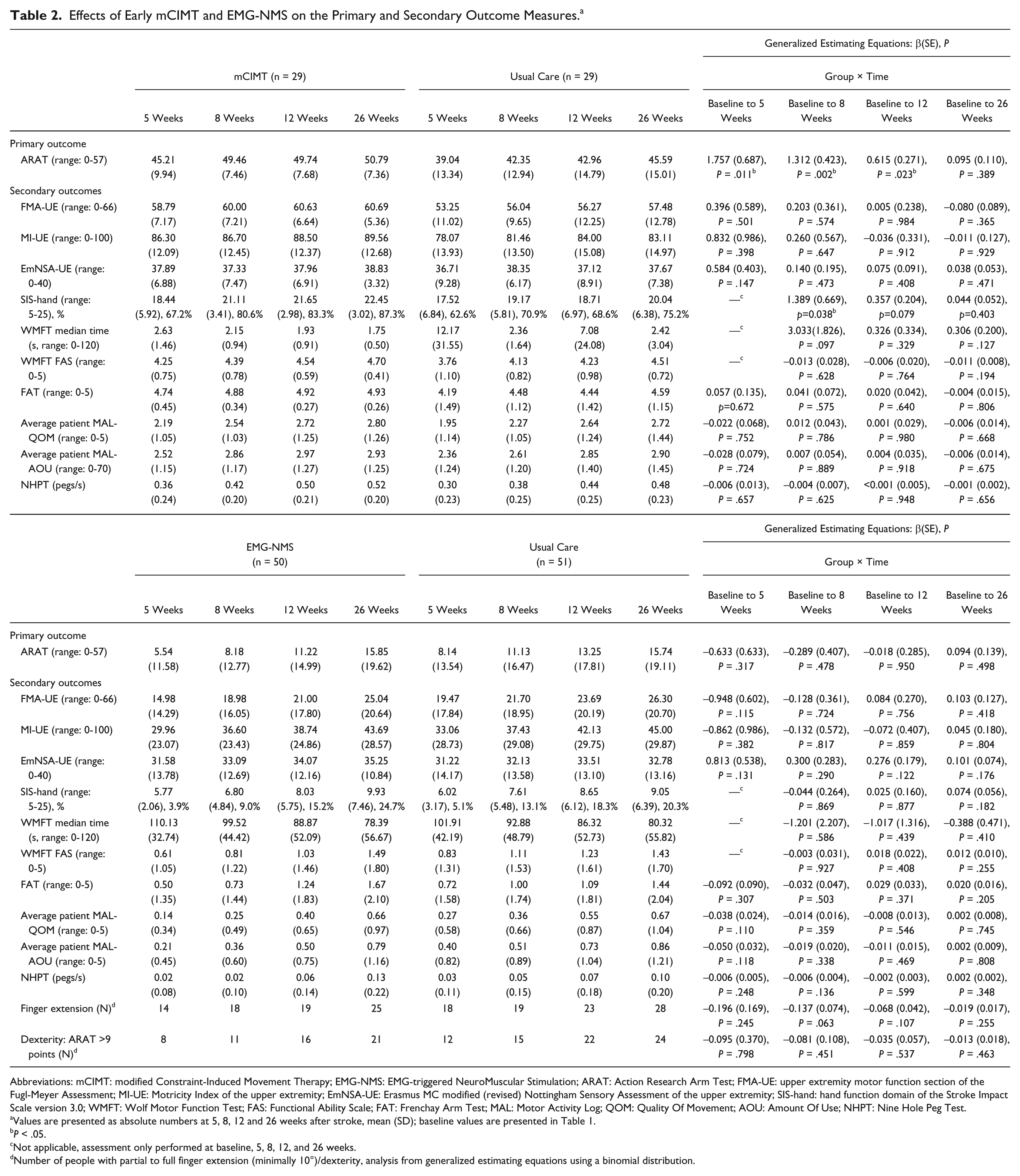
Abbreviations: mCIMT: modified Constraint-Induced Movement Therapy; EMG-NMS: EMG-triggered NeuroMuscular Stimulation; ARAT: Action Research Arm Test; FMA-UE: upper extremity motor function section of the Fugl-Meyer Assessment; MI-UE: Motricity Index of the upper extremity; EmNSA-UE: Erasmus MC modified (revised) Nottingham Sensory Assessment of the upper extremity; SIS-hand: hand function domain of the Stroke Impact Scale version 3.0; WMFT: Wolf Motor Function Test; FAS: Functional Ability Scale; FAT: Frenchay Arm Test; MAL: Motor Activity Log; QOM: Quality Of Movement; AOU: Amount Of Use; NHPT: Nine Hole Peg Test.
Values are presented as absolute numbers at 5, 8, 12 and 26 weeks after stroke, mean (SD); baseline values are presented in Table 1.
P < .05.
Not applicable, assessment only performed at baseline, 5, 8, 12, and 26 weeks.
Number of people with partial to full finger extension (minimally 10°)/dexterity, analysis from generalized estimating equations using a binomial distribution.
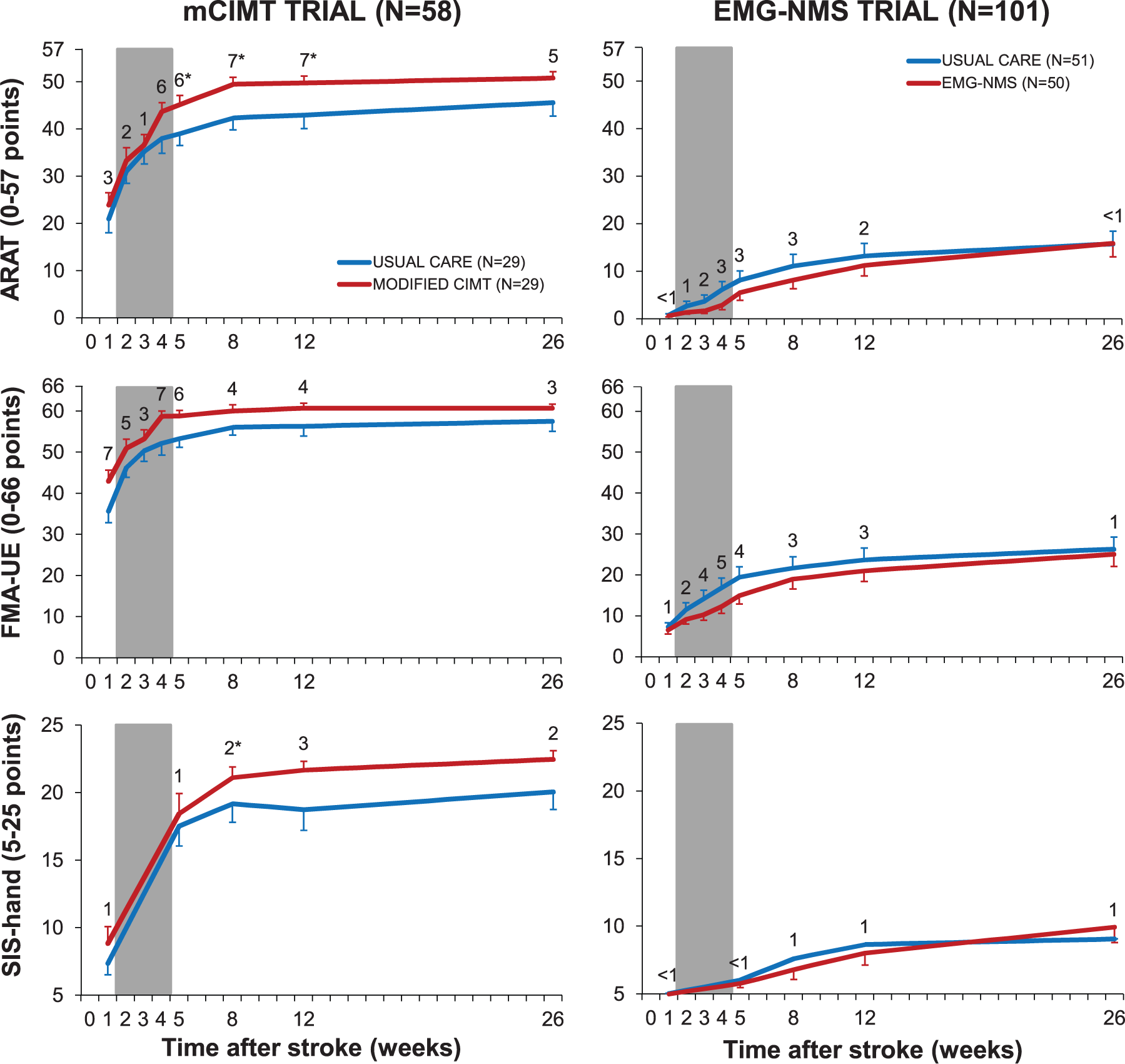
Mean (SEM) recovery patterns for the ARAT, FMA-UE, and SIS-Hand. The modified constrained induced movement therapy (mCIMT) trial and the electromyography-triggered neuromuscular stimulation (EMG-NMS) trial are represented in respectively the left 3 and right 3 panels. The numbers within each panel shows the difference between the intervention (red line) and usual care group (blue line) at all measurements for, from top to bottom, the action research arm test (ARAT), upper extremity motor function section of the Fugl-Meyer assessment (FMA-UE) and the hand domain of the Stroke Impact Scale 3.0 (SIS-hand). The 3-week intervention phase is represented by the gray area. *P < .05.
We found no significant time × group interaction effects on the ARAT in favor of EMG-NMS compared with usual care. As shown in Figure 3 and Table 2, in the EMG-NMS trial, the proportion of patients showing some return of FE and dexterity (ie, ARAT >9 points) did not differ significantly between groups. No significant time × group interaction effects were found for motor impairment such as FMA-UE, MI-UE, and for other secondary outcomes favoring or disfavoring mCIMT or EMG-NMS therapy.
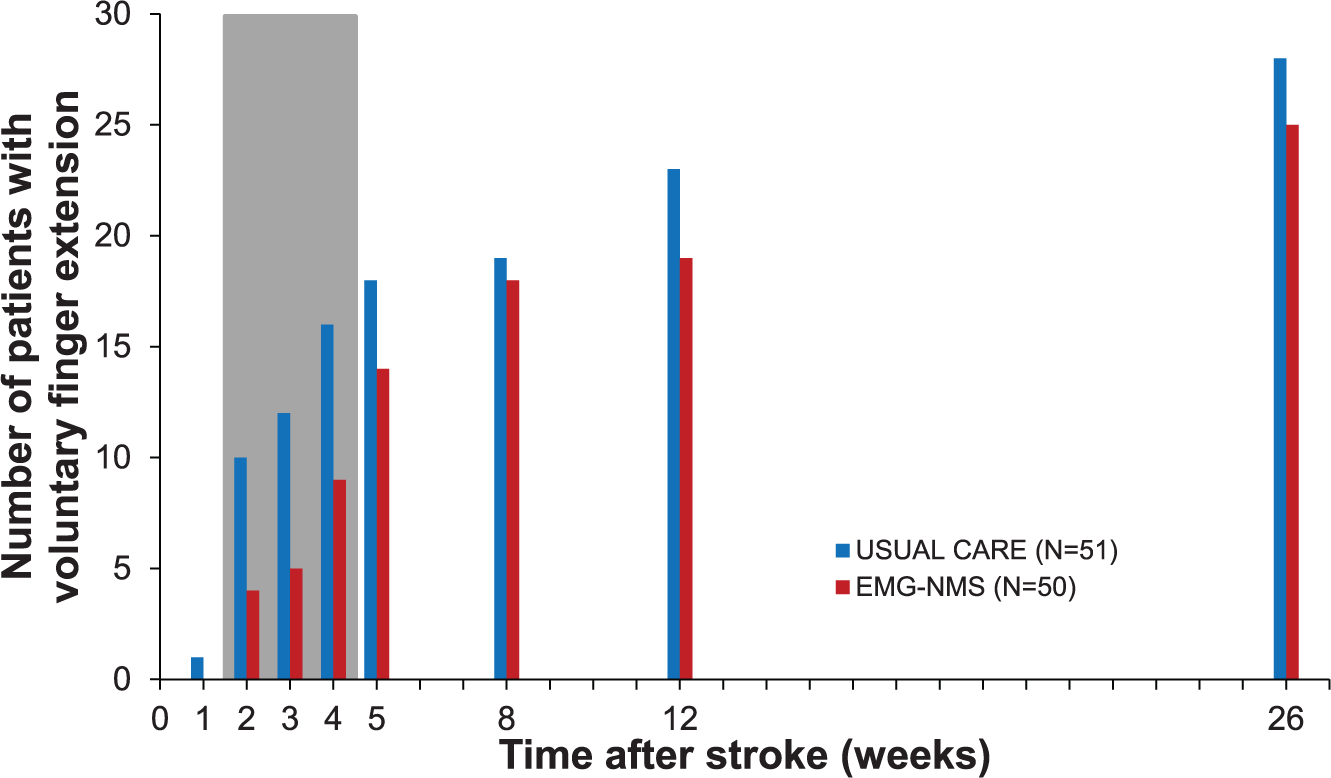
The number of patients within the unfavorable prognosis group who showed return of dexterity. The red and blue bars represent respectively the electromyography-triggered neuromuscular stimulation (EMG-NMS) group and the usual care z group, whereas the 3-week intervention phase is represented by the gray area.
Discussion
The present study showed that an early start of a 3-week mCIMT program in ischemic stroke patients with some voluntary control of FE at stroke onset (minimally 10° of FE) was more effective in improving upper limb capacity as assessed by the ARAT in comparison with solely usual care. However, these clinically meaningful effects did not sustain up to 26 weeks after stroke, which was mainly due to delayed functional improvement of the usual care group with a favorable prognosis. mCIMT also benefitted the self-reported outcome of hand function according to the SIS. Unlike these favorable effects, no superiority of mCIMT was found in terms of motor impairment, including the motor part of the FMA-UE. Finally, no added value of a 3-week EMG-NMS program in patients without initial FE was found with regard to regaining upper limb capacity compared with usual care.
To our knowledge, this is the first trial in which patients were stratified on the basis of the powerful prognostic factor “voluntary FE” to type of therapy within the first 2 weeks poststroke. Our finding of significant and meaningful changes in upper limb capacity for mCIMT in comparison with the usual care without any significant therapy-induced improvements in neurological impairments, such as synergism, is in line with a recent randomized controlled trial 36 as well as longitudinal kinematic studies showing that the observed meaningful improvements in upper limb capacity are predominantly driven by learning adaptive motor strategies. 37 The lack of significant differences on the WMFT, measuring upper extremity motor ability, may be associated with the presence of impairment-related items in this test such as extending the elbow against resistence. True neurological recovery, in terms of regaining degrees of freedom in motor control as expressed by motor synergism and smoothness in reaching tasks, seems to be determined in the same time window of spontaneous neurological recovery restricted to the first 8 weeks37,38 to 10 weeks poststroke. Our findings are supported by a recent proof-of-concept study of Kitago and et al 39 who found no significant changes in kinematic measures of movement coordination after mCIMT despite clinically meaningful improvements of upper limb capacity measured with the ARAT in the chronic phase. In addition, a treatment effect of constraint-induced therapy on the ARAT was found in the chronic phase after stroke, but this effect was not found for the FMA-UE.23,40 This suggests that the effects of mCIMT are mainly the result of learning to use preserved end-effectors in a different, more optimal way to accomplish meaningful tasks (ie, adaptation strategies) and not of actual improvement of neurological impairments.37,41,42 In addition, the follow-up measures beyond 5 weeks showed that the significant effects on the ARAT were restricted to the first 3 months poststroke. A key question that remained unaddressed in the present study is whether a higher dose of mCIMT 6 or continuation of therapy for more than 3 weeks poststroke would have resulted in effects that sustain beyond 3 months poststroke, assuming that higher doses of task-specific training may result in better outcomes.2,41,43,44
Strengths and Limitations
First, patient selection limits the generalizability to other stroke populations. Recruitment of patients with the same stroke type and no limiting comorbidities early poststroke is difficult as is illustrated by the proportion of patients eventually included 3.7%. The proportion of included patients was comparable to, for example, the 2.9% in the VECTORS-trial. 6 Despite this low recruitment rate restricted to patients with a first-ever ischemic hemispheric stroke, this study is the largest and earliest started upper extremity intervention trial conducted thus far. Fewer stroke patients were recruited for the EMG-NMS trial (n = 101) than originally planned (n = 120). 15 However, increasing the number of patients in this trial and, hence, its statistical power for the probability of regaining some upper limb capacity would most likely not have changed our conclusion because the current results do not show any trend toward positive, clinically significant effects of EMG-NMS in comparison with usual care. Omission of the stratification, by allocating all patients to 1 experimental (n = 79) and 1 control group (n = 80), would have resulted in a neutral trial due to lack of homogeneity between subjects. We believe this prognostic stratification is essential when performing rehabilitation trials, and may be a powerful tool to prevent type II errors.2,43 Furthermore, in line with previous experience in rehabilitation trials4,28 we applied a repeated measurements design to create individual time series in order to reduce the unexplained variance due to within-subject measurement error and to enhance the precision of estimating the added value of therapy relative to the underlying logistic pattern of spontaneous neurological recovery. Unfortunately, most rehabilitation trials so far did include patients at arbitrary time points in the first 3 months poststroke, 43 whereas time-dependent spontaneous neurological recovery explains almost 70% of recovery.45,46 Not stratifying patients according to the expected proportional recovery46, as well ass starting recruitment of subjects in trials at nonfixed moments poststroke, such as moment of admission in a rehabilitation center, will affect the variation in spontaneously driven gains between subjects. 44 Patients recruited during the first days poststroke will show on average more change than those recruited after a number of weeks. We are of the opinion that differences in starting times poststroke and lack of stratification increases the within-group variance for measured improvement in trials, and that this will be at the expense of identifying therapy-induced effects between-groups in randomized clinical trials. An important next step is to figure out how differences in timing of recruiting patients for trials in the subacute phase poststroke will affect random as well as systematic error in parallel group designs by using simulation models.
Second, both the mCIMT and the EMG-NMS group received more therapy in comparison to the usual care groups, that is, the amount of therapy was not dose-matched. We therefore cannot answer the question whether the beneficial treatment effect of mCIMT was completely due to the content of the therapy itself or partly by the higher dose of therapy applied. Another limitation may be the choice of EMG-NMS as an intervention for patients with no voluntary control of finger extensors at baseline. Thirty-two patients with an unfavorable prognosis showed return of voluntary FE due to spontaneous neurological recovery within the first 5 weeks. Moreover, these patients showed more improvement in motor function than expected following the shoulder abduction and finger extension model.4,47 Further research should try to identify these “false negatives” in order to improve stratification for intervention. Despite return of some FE, evidence-based therapies for this patient group with an unfavorable prognosis are lacking at this moment. From our data, it cannot be determined if a longer intervention phase, or applying EMG-NMS solely with task specific movements would have changed the current findings. There is some evidence that the sensory-motor integration during EMG-NMS of the paretic arm may activate neuronal networks13,48 and increases the cortical perfusion of the ipsilesional sensorimotor cortex in chronic stroke. 49 Our findings stress the need to explore the meaning of changes in neural networks, as revealed by imaging techniques such as functional magnetic resonance imaging and electroencephalography, regarding the impact of improved motor performance early poststroke. 41 Furthermore, the added value of combining task specific practice with interventions directly focused at the damaged brain should be investigated (eg, noninvasive brain stimulation and neuropharmacological therapies).
Interpretation
This is the first stratified randomized controlled trial of poststroke upper extremity rehabilitation based on functional prognosis using repeated measurements. Our results suggest that 1 hour daily supervised mCIMT for 3 weeks in those with some voluntary FE early poststroke is more effective than usual care alone leading to clinically meaningful effects on arm-hand capacity up to 3 months poststroke. We found no evidence that 1 hour daily EMG-NMS for 3 weeks in those without voluntary FE, influences the likelihood of return of any FE within the first 6 months after stroke. The present study therefore supports the clinical point of view that effects of upper limb training are restricted to patients with some voluntary motor control of finger extensors at baseline.2,43 The literature indeed offers no examples of evidence-based therapies resulting in significant improvements in patients with an initial flaccid hand.1,2,42,43,50 The preservation or return of FE early poststroke is assumed to reflect the integrity of the corticospinal tract. 3 Moreover, some innervation of end-effectors is needed to induce improvements by exercise therapy early poststroke. At the same time, we found no evidence that we were able to influence spontaneous neurological recovery of underlying impairments based on clinical scales, suggesting that functional improvements of the mCIMT group were based on adaptation strategies to use intact end-effectors in a more optimal way.38,42
Footnotes
Acknowledgements
The authors thank the EXPLICIT-stroke physicians, therapist and nurses at the stroke units of the participating university centers and local hospitals (VU University Medical Center Amsterdam; Leiden University Medical Center; University Medical Center Utrecht; Radboud University Medical Center Nijmegen; and Sint Lucas Andreas Hospital Amsterdam; Amstelland Hospital Amstelveen; Rijnland Hospital Leiderdorp; Diaconessen Hospital Leiden; Sint Maartens Hospital Nijmegen; Meander Medical Center Amersfoort; Diakonessen Hospital Utrecht and Zeist) and in the associated rehabilitation centers and nursing homes (Reade Rehabilitation Center, Slotervaart Nursing Home, Sint Jacob Nursing Home, and De Driehoven Nursing Home in Amsterdam; Zonnehuis Nursing Home in Amstelveen; Rijnlands Rehabilitation Center in Leiden; Groot Klimmendaal Rehabilitation Center in Arnhem; Tolbrug Rehabilitation Center in ‘s-Hertogenbosch; Trappenberg Rehabilitation Center in Huizen; De Hoogstraat Rehabilitation Center and Albert van Koningsbruggen Nursing Home in Utrecht; Birkhoven Nursing Home in Amersfoort; Nassau Odijckhof Nursing Home in Driebergen-Rijsenburg; Warande Nursing Home in Zeist; Quarijn Nursing Home and Military Rehabilitation Center Aardenburg in Doorn) for providing the intervention and monitoring rehabilitation, and the patients who participated in the study.
EXPLICIT-Stroke Consortium
In addition to the authors of the present study, the consortium consists of Frans C. van der Helm, PhD, and Asbjørn Klomp, MSc, from the Delft University of Technology; Joost van Kordelaar, PhD, from the VU University Medical Center Amsterdam; Hanneke J. van der Krogt, MD, from the Leiden University Medical Center; Floor E. Buma, MSc, from the Utrecht University; and Chantal D. Bakker, MD, from the Radboud University Medical Center Nijmegen.
Authors’ Note
The funding organizations had no role in the design or conduct of the study, the collection, management, analysis, or interpretation of the data, or in the preparation, review, and approval of the manuscript.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: GK, EW, AK, AV, JG, JA, AG, and CM report grants from The Netherlands Organisation for Health Research and Development (ZonMw), during the conduct of the study. AG reports personal fees from Ipsen Farmaceutics, outside the submitted work. CW, RN, EV have nothing to disclose.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research leading to these results received funding from the Netherlands Organisation for Health Research and Development (ZonMw Grant No. 89000001) and was supported by the European Research Council (ERC) under the European Union’s Seventh Framework Programme (FP/2007-2013)/ERC Grant Agreement no. 291339-4D-EEG and grants awarded to EW and CM from the Dutch Brain Foundation (de Hersenstichting).
