Abstract
Background. Effective recovery from functional impairments caused by traumatic brain injury (TBI) requires appropriate rehabilitation therapy. Multiple pathways are involved in secondary injury and recovery suggesting a role for multimodal approaches. Objective. Here, we examined the efficacy of the anti-inflammatory agent minocycline and botulinum toxin (botox)–induced limb constraint with structured physical therapy, delivered alone or in combination, after a severe TBI produced by a controlled cortical impact in rats. Methods. Minocycline was administered at 25 mg/kg daily for 2 weeks beginning 1 day after TBI or sham surgery. For constraint/physical therapy, botox-type A was injected into the nonaffected forearm muscle 1 day after injury and 2 weeks of physical therapy commenced at 5 days after injury. Functional evaluations were conducted 8 weeks after injury. Results. Minocycline, either as a monotherapy or as combination treatment with botox/physical therapy significantly reduced impairments of spatial learning and memory in the water maze test, whereas botox/physical therapy reduced forelimb motor asymmetry and improved manual dexterity in the cylinder and vermicelli handling tests, A synergistic effect between the 2 treatments was observed when rats performed tasks requiring dexterity. Inflammation was attenuated in the peri-contusion cortex and hippocampus in all TBI groups receiving mono or combination therapies, though there was no significant difference in lesion size among groups. Conclusion. These data provide a rationale for incorporating anti-inflammatory treatment during rehabilitation therapy.
Keywords
Introduction
Traumatic brain injury (TBI) is the leading cause of disability in the young in the Western world with an estimated incidence of approximately 1.4 million per year in the United States. 1 Currently, 5.3 million Americans (2% of the population) live with a persisting disabling condition attributable to TBI. 2 Moreover, the prevalence of TBI among military personnel in Iraq and Afghanistan is 19.5%, or more than 320 000 according to the RAND report. 3
Following the initial mechanical damage resulting from the direct impact, TBI-induced brain damage is exacerbated by secondary injury that is orchestrated primarily by the inflammatory response.4-6 Whereas early interventions are important to reduce secondary injury and maximize functional outcome, chronic interventions are instrumental for promoting recovery of persistent debilitating symptoms resulting from TBI. Minocycline, a derivative of tetracycline that is capable of crossing the blood–brain barrier, has not only anti-inflammatory effects7-9 but has also been previously shown to be neuroprotective in many neurological conditions, including human ischemic stroke, 10 animal models of cerebral ischemia, Huntington’s disease, and TBI.11-15
Constraint-induced movement therapy (CIMT), in which the unimpaired limb is restrained, forcing use of the affected limb, has been shown in clinical use to improve upper limb motor function at various stages of stroke recovery.16,17 CIMT has also been reported to improve upper limb motor function following chronic TBI in adults 18 and children. 19 Apart from the benefit in improving motor recovery, CIMT also increased hippocampal gray matter volume bilaterally in stroke patients, 20 suggesting that CIMT might enhance neuroplasticity at functionally remote regions that are involved in cognition. In this study, we sought to determine the treatment effect of multimodal therapies in a rat model of TBI with controlled cortical impact (CCI), by combining treatment with the anti-inflammatory agent minocycline with physical rehabilitation, including unaffected limb restraint using botox-A and ethologically based exercise.
Materials and Methods
Animals, Housing, and General Considerations
All experimental procedures were approved by the San Francisco Veterans Affairs Medical Center Animal Care and Use Committee. Male Sprague–Dawley rats (250-300 g; 8-10 weeks of age, Simonsen Laboratories, Gilroy, CA) were housed 2 per cage on a reverse 12-hour dark/light cycle with access to food and water ad libitum. All procedures were conducted by personnel blinded to experimental conditions.
Induction of Traumatic Brain Injury
Controlled cortical impact was conducted as described 21 with modifications in impact location at anteroposterior 0 and lateral 3.5 mm, from bregma (left hemisphere) using a 5-mm diameter impact tip, and impact parameters, including velocity of 1.5 m/s, depth of 5 mm, and dwell time of 120 ms. The overall mortality rate was less than 5%.
Administration of Minocycline
Following CCI, rats were assigned randomly to treatment groups. To compare effects of the dosing regimen on microglial activation, minocycline (25 mg/kg in 0.1 M phosphate buffer; Sigma-Aldrich, St Louis, MO) or vehicle (0.1 M phosphate buffer) was administered via intraperitoneal injection initially at 24 hours postinjury, and subsequently every 24 hours until the rats were sacrificed at 7 days, 12 days, and 16 days. Animals that underwent behavioral evaluations received a daily minocycline injection for 2 weeks either by itself or in combination with botox/physical therapy, beginning at 24 hours post TBI.
Botox-Induced Limb Constraint and Physical Therapy
To enforce use of the TBI-impaired arm, subgroups of rats received injections of botulinum toxin type A (botox A; Allergan, Inc, Irvine, CA) 1 day following injury in 4 left forelimb muscles (bicep, tricep, brachioradialis, and flexor carpi ulnaris) with 2 doses of 1.25 U botox A in 0.05 mL saline. Daily 1-hour physical therapy began 5 days postinjury and continued for 2 weeks, consisting of daily rope climbing, net traversal, and wheel running for 10 minutes of each activity, and 30 minutes of forced-walking in the rodent balls. Animals that failed to spontaneously engage in the exercise were gently encouraged by the trainer to participate in the activities. Representative sessions during the first and last day of training were shown in supplemental videos 1 and 2, respectively available at http://nnr.sagepub.com/content/by/supplemental-data. The effect of botox on the grip strength of the injected paw was determined via a digital force meter (Chatilon DFE series, Largo, FL) with a T-bar attachment. The peak tensile strength (pound per force) from the paw was measured 3 times and averaged during each time point of assessment.
Immunohistochemistry
Double immunostaining was performed as previously described 22 with 40-µm thick floating sections using a mouse monoclonal anti-rat CD11b (1:200, Serotec, Oxford, UK), rabbit polyclonal anti-IBA-1 (1:250, Wako Chemicals, Inc, Irvine, CA), or rabbit polyclonal anti-glial fibrillary acidic protein (GFAP; 1:1000, Chemicon, Billerica, MA), followed by Alexa Fluor 594 donkey anti-mouse IgG or Alexa Fluor 488 goat anti-rabbit IgG (1:400 dilutions). Fluorescent signals were visualized by confocal microscopy (Zeiss LSM 510).
Assessment of Microglial and Astroglial Activation and Lesion Volume
Microglial activation was evaluated in the ipsilesional and contralesional cortex and dentate gyrus in 4 coronal sections spaced 480 µm apart, centering on the lesion epicenter as indicated in Figure 1A. CD11b- and IBA-1-immunoreactive cells displaying an activated microglial phenotype were counted as previously described. 21 Fluorescent images were captured with a confocal laser scanning microscope and analyzed with National Institutes of Health Image J program among 4 randomly selected and evenly distributed 450 µm2 areas in each brain section. The number of activated microglia was normalized by the total number of cells within the 450 µm2 frame. GFAP expression level in each photographed area was calculated by multiplying the net area of GFAP staining by the intensity of GFAP staining with Image J. TBI lesion cavity size was determined as previously described with modifications 23 using NeuN stained sections.
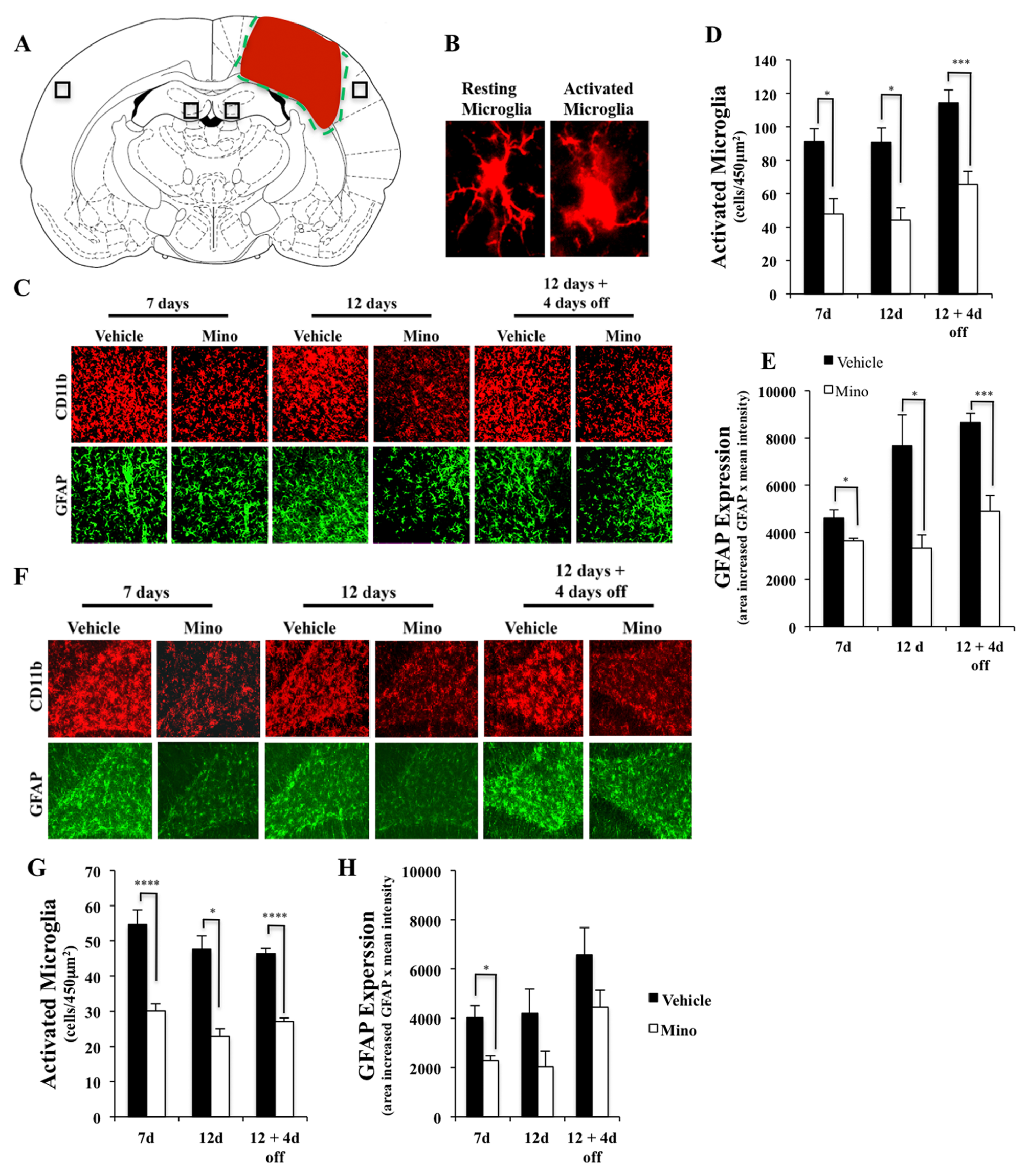
Minocycline reduces microglial activation in the cortex and hippocampus after traumatic brain injury (TBI). One day after TBI surgery, rats were treated with daily minocycline (25 mg/kg) or vehicle for the time periods as indicated. (A) A coronal rat brain section diagram showing the relationship of the lesion cavity (red) to regions photographed (black squares). (B) Representative morphology of resting microglia, with 2 or more thin processes (left), and activated microglia with less than 2 processes and amoeboid cell soma (right). Representative immunofluorescence images (C: CD11b, red; GFAP, green) and quantification of microglial activation (D) and astrocytes (E) in the perilesion cortex. Minocycline reduced TBI-induced microglial activation and astrogliosis in all 3 treatment paradigms. (F-H) Representative images of microglia and astrocytes in the ipsilateral dentate gyrus and corresponding quantification. Minocycline suppressed TBI-induced microglial activation in the hippocampus at designated time points after TBI and reduced astrogliosis only at 7 days treatment, with a trend toward reduction at later time points that did not reach significance; n = 4 to 5 in each group. Scale bars, 20 µm.
Neurobehavioral Assessment
At 8 weeks after TBI or sham surgery, neurobehavioral tests were conducted following the order presented below, in 8 groups of rats: sham–vehicle–no BPT (n = 6), sham–vehicle–BPT (n = 6), sham–MC–no BPT (n = 6), sham–MC–BPT (n = 6), TBI–vehicle–no BPT (n = 8), TBI–vehicle–BPT (n = 8), TBI–MC–no BPT (n = 8), and TBI–MC–BPT (n = 8), where MC indicates minocycline and BPT indicates botox/physical therapy. Following behavioral testing, each cohort was examined for the lesion size and brain inflammation at 24 weeks postinjury.
Forelimb use asymmetry test
Rats were placed individually in a Plexiglas cylinder 20 cm in diameter and 30 cm in height. The number of contacts with the cylinder wall during rearing was determined from 10-minute videotaped sessions. Limb preference was calculated as a percentage of the independent use of either limb during wall exploration. 24
Vermicelli handling test
Rats received 5 pieces of dry vermicelli, each 7 cm in length in their home cages.21,25 Eating and manipulation of the vermicelli was videotaped for analysis. Normal handling behavior involves holding the vermicelli fragment asymmetrically with 2 paws, one as a guide paw and the other as a grasping paw, with the paws apart until the fragment is approximately half in length. Atypical behaviors in handling the pasta were classified into 2 categories. Type I atypical behaviors either occurred or did not, and included (a) symmetrical paw placement when eating the first half of the pasta, (b) in a hunched body posture, and (c) head tilt or face lowered toward the cage floor while eating the pasta. Type II atypical behaviors could occur on multiple occasions, and were scored according to the frequency of the occurrence and included (a) mouth pulling, (b) single paw use, (c) switching the grasp and guide paws, (d) breaking the pasta into small pieces to eat, and (e) dropping or flipping the pasta during eating.21,25
Morris water maze test
The ability of rats to locate a submerged platform (10 cm in diameter, 2.5 cm below the surface of water) was assessed in a circular pool (180 cm in diameter, 60 cm in depth) with a video tracking system (Ethovision, Noldus Information Technology, Sterling, VA). 22 Rats were trained first to locate a visible platform (cued by a flag hanging above the platform, day 1), and then to locate a hidden platform (days 2-5) from different counterbalanced starting positions; in a subsequent trial, the hidden platform was removed (probe trial, day 6, trial 31) to assess short-term memory recall/retention. An annulus was defined by a circular region with a diameter of 20 cm, concentric with the previous platform position during hidden platform training. Time spent in the target quadrant as well as in the annulus, and the numbers of annulus crossings were analyzed for probe trial data. Thigmotaxic behavior, defined as swimming within 20 cm of the perimeter of the pool, was determined as well.
Statistical Analyses
Data were expressed as mean ± standard error of the mean. Water maze and grip strength data were analyzed as repeated measures using mixed model regression in SAS Version 9 (SAS Institute, Cary, NC) Proc MIXED. Type I atypical behaviors during pasta handling were analyzed using a Mann–Whitney nonparametric test. Other data were analyzed using one-way or multivariate analysis of variance with StatView or SAS Version 9. Post hoc tests were conducted when appropriate using Bonferroni corrections. Differences between groups were considered significant when P <. 05.
Results
Minocycline Treatment Effectively Suppresses Glial Responses to TBI
To determine whether minocycline reduces TBI-induced brain inflammation, injured rats were treated with vehicle or minocycline (25 mg/kg) for 7 days, 12 days, or 12 days followed by 4 days without treatment (to test for a rebound effect) beginning at 24 hours post-TBI. In the perilesion cortex and dentate gyrus, minocycline significantly decreased activated microglia when compared with vehicle-treated groups at all time points, as shown in Figure 1C, D, F, and G. Minocycline also reduced the number of GFAP immunoreactive cells in these areas (Figure 1C, E, F, and H). Based on these results a 2-week daily minocycline treatment regimen was chosen for subsequent experiments.
Chronic Minocycline Treatment Improves Cognitive Function in the TBI Rats
Following minocycline, botox/physical therapy (BPT) and combination treatments, rats underwent neurobehavioral tests beginning 8 weeks after TBI. During water maze acquisition, both sham and TBI animals learned to locate a visible (F1, 46 = 121.7, P < .0001; Figure 2A) or a hidden platform (F3, 138 = 52.6, P < .0001; Figure 2B), as evidenced by progressively shortened pathlengths. However, TBI significantly decreased water maze acquisition performance, resulting in longer pathlengths to find the visible (F1, 46 = 42.3, P < .0001) and hidden platform (F1, 46 = 35.2, P < .0001) relative to sham-operated rats.
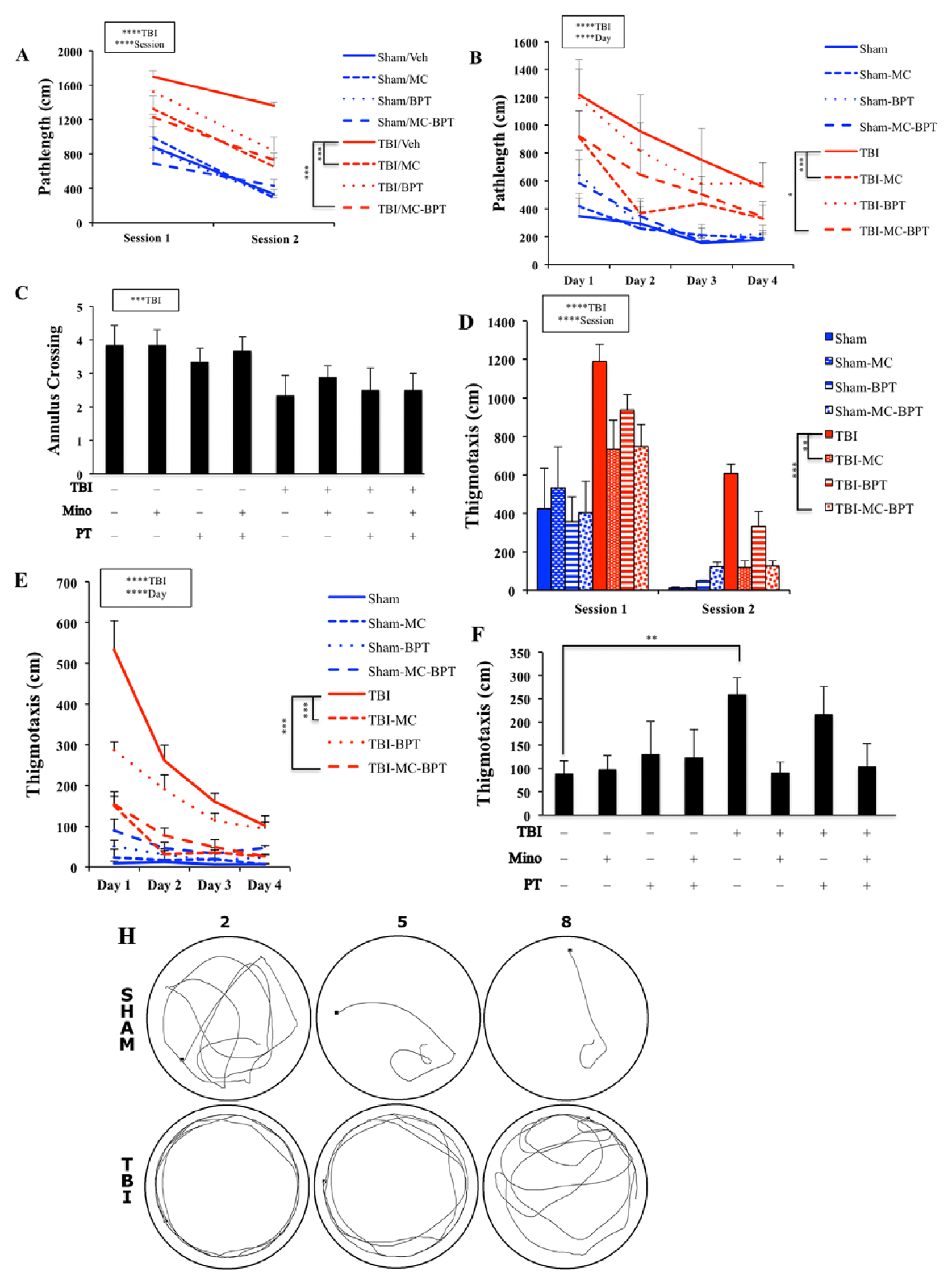
Minocycline treatment improves traumatic brain injury (TBI)–induced learning and memory impairment in the water maze. The main effects of training and TBI were indicated with statistical significance. Injured animals that received minocycline or the combined treatment of minocycline and botox/physical therapy had shortened pathlength to platform during visible (A) and (B) platform training. In the probe trial, injured animals made significantly fewer crosses in the annulus search compared to sham rats. However, there was no significant treatment effect found among the TBI groups (C). Minocycline treatment alone or combined treatment with minocycline and botox/physical therapy also reduced thigmotaxic behavior in visible (D) and hidden (E) platform training. During the probe trial, injured rats continued to demonstrate significantly increased thigmotaxic behavior relative to sham-operated controls (F). Injured animals receiving minocycline had a tendency toward less thigmotaxis than those that did not receive any treatment (P = .06 following Bonferroni corrections). (G) Representative swimming traces from a sham and a TBI rat during trials 2, 5, and 8 of the hidden platform search are shown as indicated. In contrast to the sham rat, the TBI rat tended to swim along the wall of the pool, especially during the initial trials.
Sham and TBI animals responded differently to treatments in the visible and hidden platform trials of the Morris water maze. In the visible platform search, minocycline treatment significantly shortened the pathlength in TBI (F1, 27 = 12.09, P < .01) groups but not in sham-operated animals (Figure 2A). In hidden platform training, minocycline treatment again significantly reduced pathlength in TBI groups (F1, 27 = 18.4, P < .0005). Injured animals receiving the combination of minocycline and BPT also performed significantly better than untreated TBI animals (post hoc: P < .05; Figure 2B). During the probe trial, injured animals showed reduced numbers of annulus crossings compared with sham (F1, 46 = 8.9, P < .005; Figure 2C), suggesting that TBI had an adverse effect on memory recall/retention. However, neither minocycline nor BPT altered the number of annulus crossings.
Injured animals also exhibited thigmotaxic behavior, searching near the perimeter of the pool during visible platform (F1, 46 = 47.9, P < .0001; Figure 2D) and hidden platform trials (F1, 46 = 35.2, P < .0001; Figure 2E) compared with sham controls. Their decreased performance during platform searching may be because of increased use of this unproductive search strategy. Minocycline reduced thigmotaxic behavior in TBI rats during visible platform (F1, 27 = 13.99, P < .0001), hidden platform (F1, 27 = 16.97, P < .0005), and probe trials (F1, 27 = 4.1, P < .05; Figure 2D-F). Similar to the pathlength effects, BPT alone did not result in significant decreases in thigmotaxis, and did not significantly increase or decrease the effects of minocycline.
Botox/Physical Therapy Improves the Use of the TBI-Affected Forelimb
Eight weeks after injury there was persistent preference for the unaffected paw during forelimb asymmetry (cylinder) testing (TBI effect: F1,46 = 47.4, P < .0001). Interestingly, BPT, but not minocycline alone, significantly reduced forelimb asymmetry by increasing the relative use of the affected forelimb and decreasing the use of the unaffected one (F1, 27 = 7.46, P < .01). TBI rats receiving minocycline with BPT had significantly reduced forelimb asymmetry (post hoc: P < .05), similar to that seen with BPT alone (Figure 3A). The severity and duration of the botox-induced weakness was determined by a T-bar force meter over 20 weeks. Sham rats that received botox demonstrated persistent weakness in the left paw compared with those injected with saline (F1, 10 = 189.1, P < .0001; Figure 3B). However, botox/physical therapy in sham animals, either alone or in combination with minocycline, did not appear to significantly influence paw preference (Figure 3A).
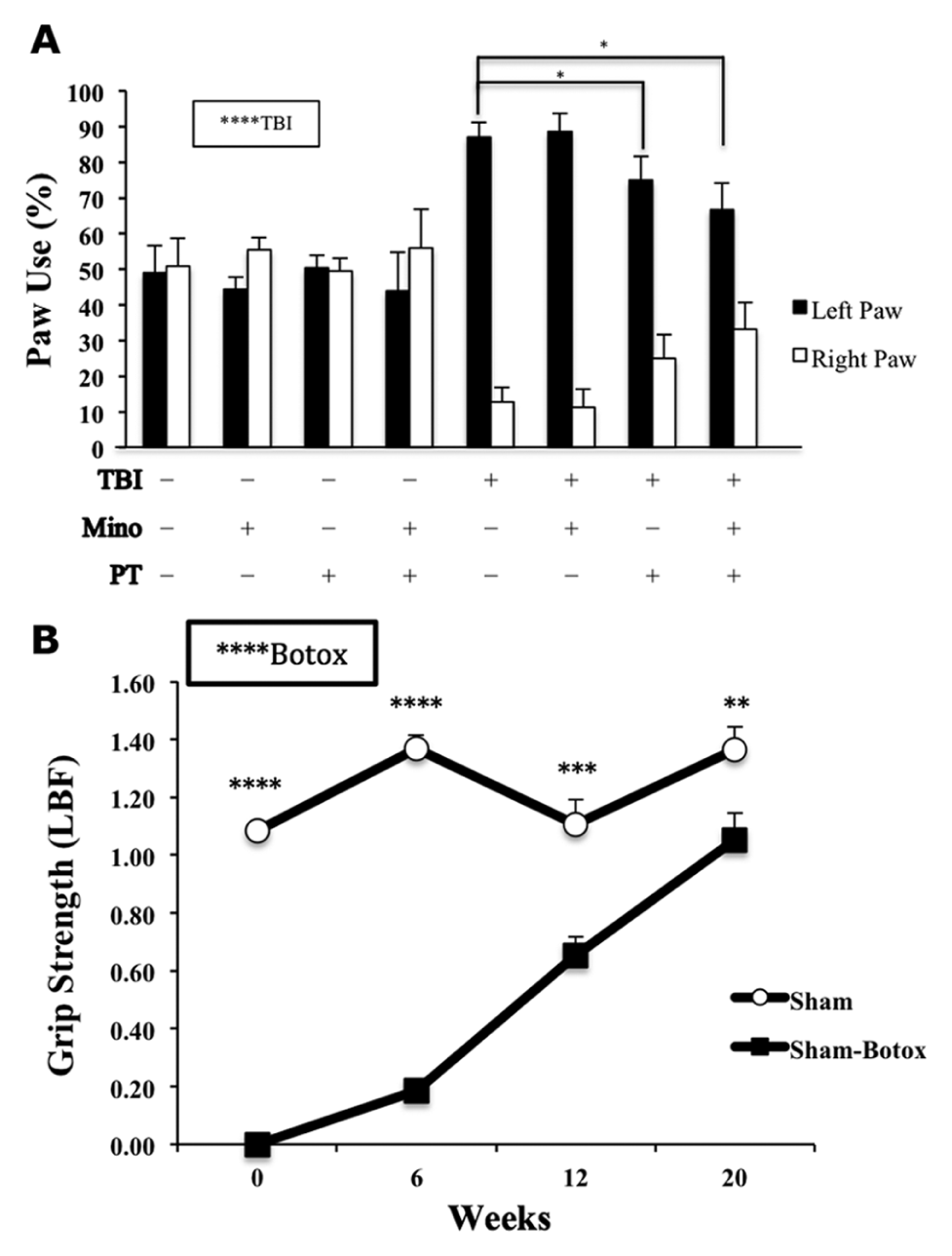
Botox/physical therapy reduces forelimb asymmetry in injured animals. (A) Sham animals showed no preference for either limb during wall exploration. Injured animals displayed a significant preference for the unaffected limb and those receiving either botox/physical therapy or the combined treatment showed a significant increase in the use of the affected paw with consequent decrease in unaffected paw usage. (B) Botox had a persistent effect on grip strength with 59% and 77% recovery at 12 and 20 weeks, respectively.
Botox/Physical Therapy Reduces the Atypical Handling of Vermicelli in TBI Rats
Injured animals displayed significantly greater numbers of type I atypical behaviors over 5 trials of pasta handling than sham animals (F1,46 = 147.7, P < .0001). BPT had an overall effect in reducing type I atypical behaviors (F1, 27 = 12.8, P < .005; Figure 4A), whereas neither minocycline nor BPT alone significantly reduced type I atypical behaviors in injured animals. Interestingly, the combination of minocycline and BPT was effective (post hoc: P < .005).
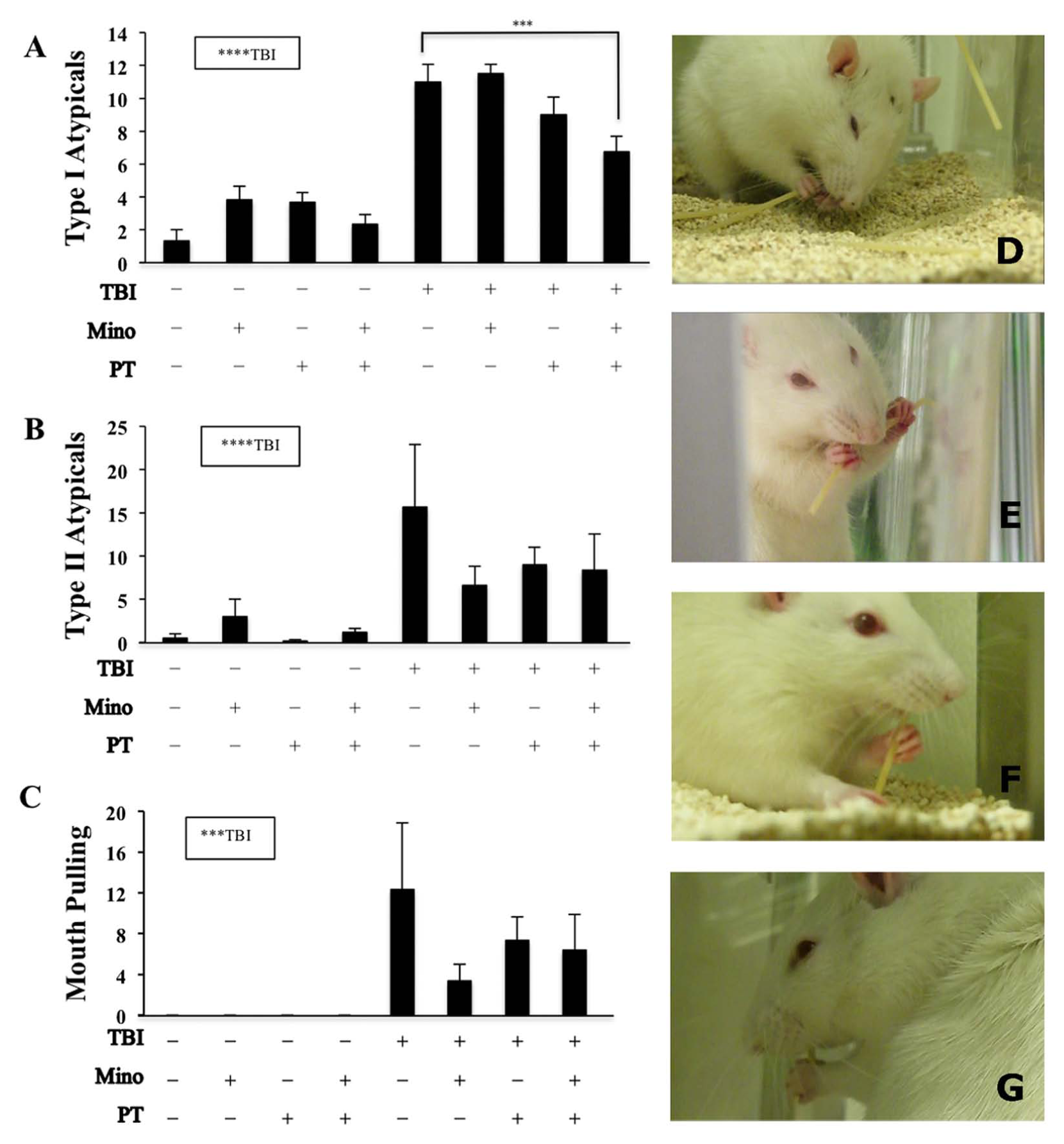
Traumatic brain injury increases atypical behaviors in the vermicelli handling test: Traumatic brain injury (TBI) significantly increased type I (A) and type II (B) atypical behaviors, in particular mouth-pulling behavior (C). Only animals receiving the combination of minocycline and botox/physical therapy showed significantly reduced type I atypical behaviors relative to untreated injured animals. Examples of common atypical adjustments include: face was lowered toward the ground during eating (E), broke pasta while eating (F), flipped pasta with hands to eat other end (G), and used mouth to pull pasta up and through grasp (H).
Traumatic brain injury also increased the total occurrence of type II atypical behaviors (F1,46 = 14.2, P < .001; Figure 4B), among which the mouth pulling was only observed among injured rats (F1,46 = 11.96, P < .005; Figure 4C). Neither minocycline nor constraint/physical therapy significantly reduced the total occurrence of the type II atypical behaviors, including mouth pulling.
Neither Minocycline nor Botox/Physical Therapy Alters TBI Lesion Size
To determine whether the observed benefits in functional outcome were attributable to a neuroprotective effect of minocycline or BPT, we determined the lesion cavity size in NeuN-stained brain sections. We found that the lesion sizes were not significantly different among all TBI treatment groups as shown in Figure 5, suggesting that the delayed treatment of minocycline or constraint physical therapy did not change the overall extent of injury, in spite of the beneficial effect on functional outcomes.
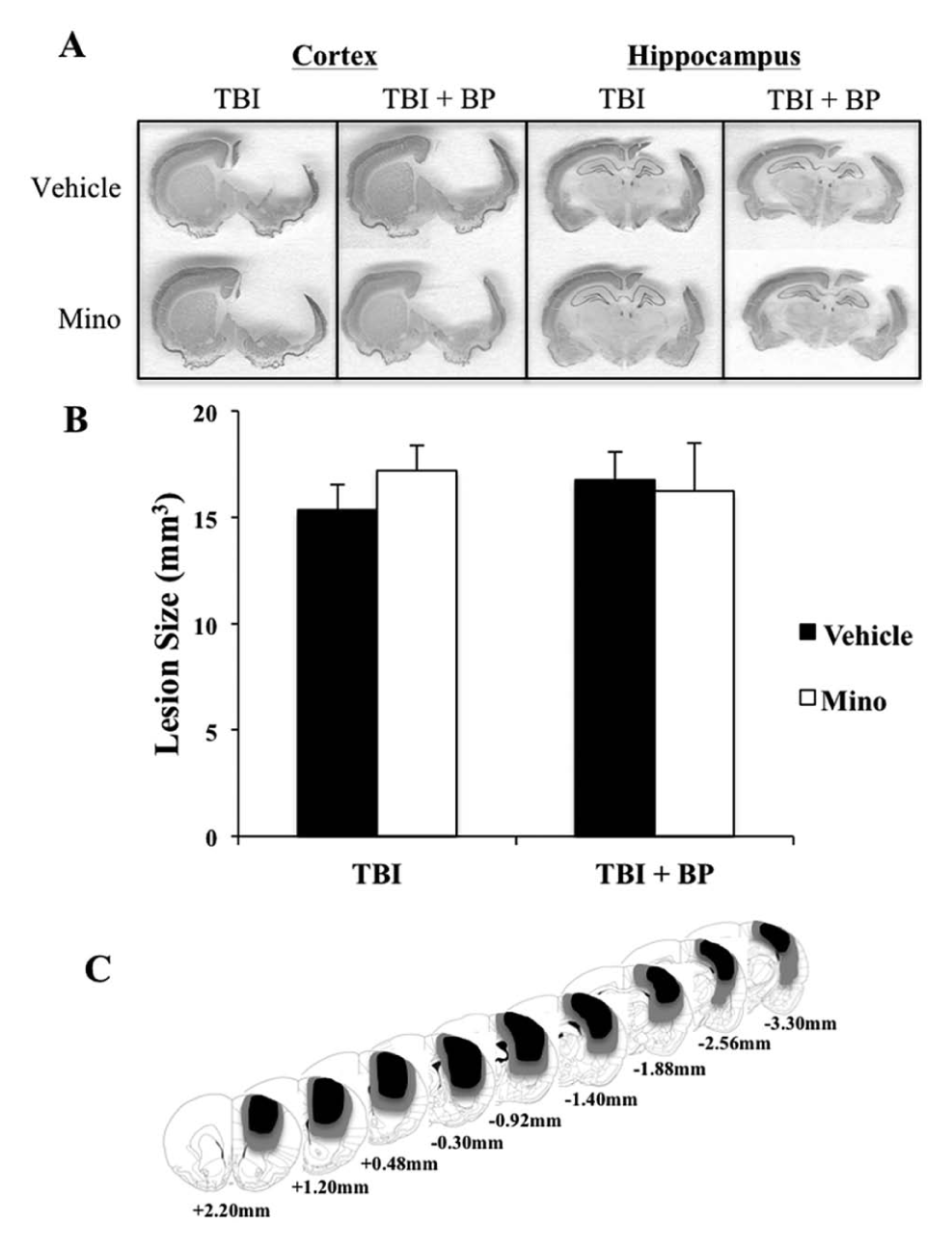
Traumatic brain jury (TBI) lesion size is not affected by either minocycline or botox/physical therapy. (A) Representative coronal sections showing NeuN immunostaining with various treatments. (B) Neither minocycline nor botox/physical therapy altered lesion size. (C) Coronal reconstructions showing the extent of contusion injury in rats following TBI. Smallest and largest damaged areas appear in black and gray, respectively. Numbers indicate the section position in millimeters from the bregma.
Long-Term Beneficial Effect of Minocycline or Constraint Physical Therapy in Reducing TBI-Induced Brain Inflammation
To determine whether the improved functional outcome is related to a long-term effect of minocycline and BPT on glial activation, the number of activated microglia and astrocytes were quantified. Interestingly, 24 weeks after TBI, minocycline alone, BPT alone persistently reduced microglial activation in the perilesion cortex (minocycline effect, P < .0001; BPT effect, P < .0005; Figure 6A and B), and dentate gyrus (minocycline effect, P < .0005; BPT effect, P < .005; Figure 6 D and E). The combination of the treatments had a similarly significant effect; however, synergy could not be determined, as the effect of each of the treatments separately brought activation near to baseline. Significant reduction in GFAP expression occurred only in groups treated with either minocycline or the combined treatment of minocycline and BPT (perilesion cortex, P < .05; hippocampus, P < .05; Figure 6A, C, D, and F); however, there appeared to be a trend toward reduction with BPT alone.
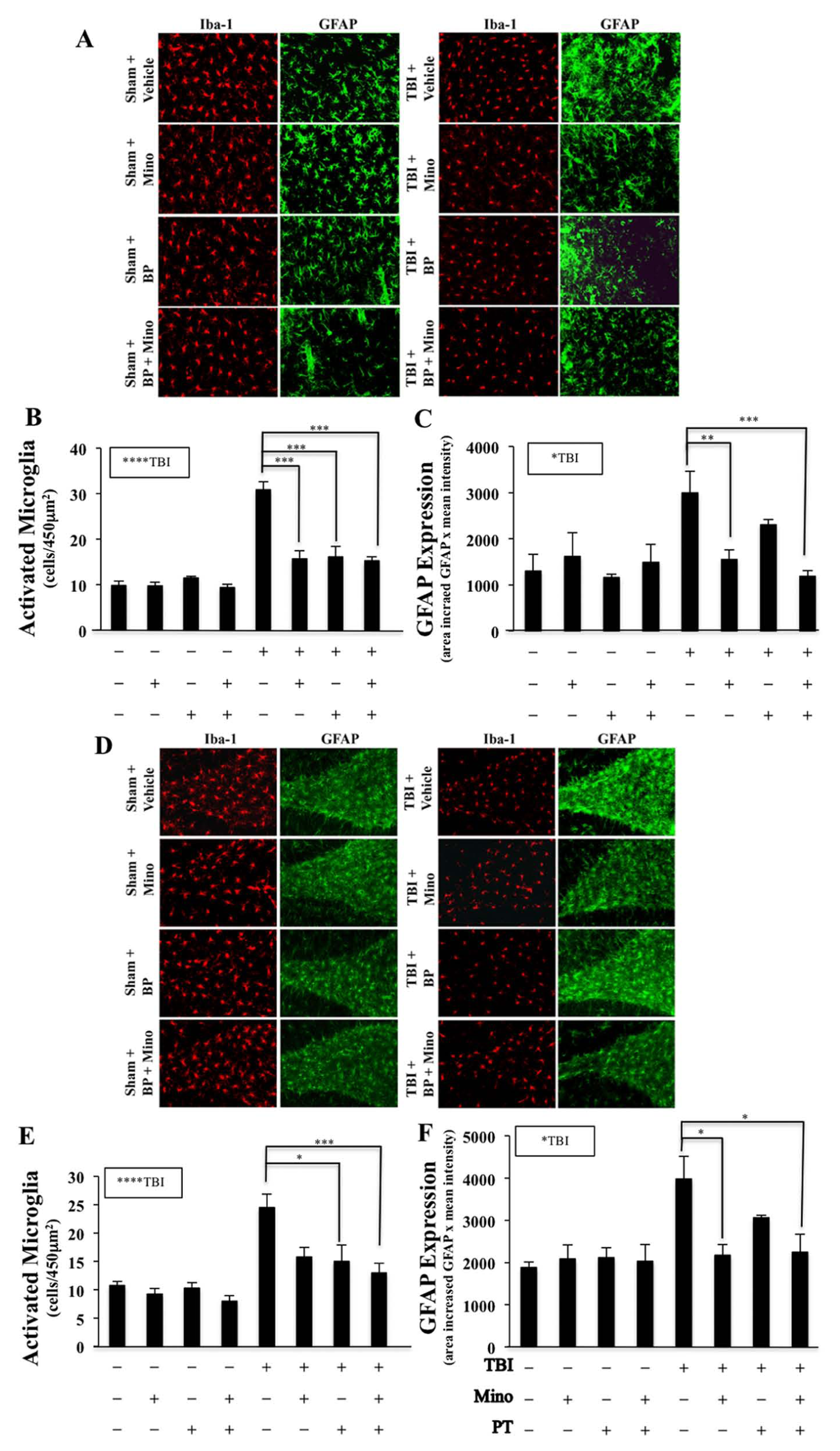
Persistent reduction of microglia in the perilesion cortex and hippocampus in injured animals following treatment with minocycline, botox/physical therapy, or the combination. Brain inflammation was evaluated 24 weeks after traumatic brain injury (TBI). Representative immunofluorescence images (A) and quantitation of microglia (B; IBA-1, red) and astrocytes (C; GFAP, green) in the perilesion cortex. TBI significantly increased microglial and astrocytic activation. Minocycline, botox/physical therapy, and the combination reduced TBI-induced microglial activation (B). Reduction of GFAP expression was observed in injured animals with minocycline and the combined treatment (C). Scale bars, 20 µm.
Discussion
Because of the growing recognition that therapies targeting more than one pathway may have additive or synergistic effects, we determined whether combining the anti-inflammatory agent minocycline and botox-induced constraint therapy with physical exercise promotes functional recovery in an experimental model of TBI. We found that chronic treatment with minocycline not only attenuated TBI-elicited neuroinflammation but also reduced TBI-induced memory impairment. While constraint physical therapy was most effective in reducing forelimb asymmetry, the combined treatment of minocycline and constraint physical therapy synergized to improve forelimb manual dexterity. The observed functional recovery following a single or combined therapy was independent of the extent of brain injury.
Constraint-induced movement therapy, in which the unaffected upper limb is restrained, forcing use of the affected upper limb, resulted in sustained improvements in motor function among stroke patients in one randomized controlled trial (EXCITE), when comparing progressive skills training at high intensity and constraint with no intervention. 26 However, subsequent clinical trials have yielded mixed results.16,27-31 A modified CIMT combined with botox A was shown to reduce spasticity and improve motor function in chronic stroke patients. 32
A recent meta-analysis of CIMT in experimental stroke studies revealed no benefit in motor function or infarct sizes, although 2 of 15 studies showed improved cognitive function after CIMT 33 ; this is at odds with some human stroke studies and our current TBI data showing motor but not cognitive improvements. The reasons for this are unknown, but the onset time, duration, intensity and type of training, time of commencement and duration of constraint, and timing and methods of functional assessment all may be playing a role in the final outcome. Unlike the temporary effect of restraining jackets used in most studies, the restraining effect of botox lasts for weeks and resolves variably. Another pitfall of disabling one forelimb in the quadruped animal is its potential adverse effect on the rat’s ability to locomote and support its stance during feeding and exploration of the environment. Nonetheless, following 2 weeks of physical therapy using the botox constraint method, all rats displayed dramatically improved skills in their daily physical activities, independent of the injury status (supplemental videos available at http://nnr.sagepub.com/content/by/supplemental-data). Most important, the effect of botox did not affect limb preference in the forelimb asymmetry test. Future studies in which these parameters are varied should help to inform clinical applications.
TBI results in microglial and astrocytic activation34,35 and subsequent release of reactive oxygen species, nitric oxide, cytokines, metalloproteinases, and other cytotoxic factors.36-38 This inflammatory response requires hours to days to reach a maximum after TBI, providing a window of opportunity for therapeutic intervention. Conversely, some aspects of the innate immune response can facilitate later tissue repair and functional recovery.34,39 Therefore, the timing of anti-inflammatory treatment may crucially affect the outcome. In a prior study, 21 we found that perilesion microglial and astrocytic activation peaked over 5 to 7 days and diminished, but persisted for at least 14 days. In the current study, with larger TBI lesions, these parameters were elevated for 16 days or more. Minocycline consistently reduced these activations throughout this period, and this effect persisted on discontinuation after 12 days, suggesting the drug had interrupted, rather than delayed or suppressed, some factors driving the inflammatory response. The 24-hour treatment onset used represents a reasonable clinical timeframe and it seems likely that blunting of this early inflammatory spike could have protective effects. However, it remains unclear how this affects the long-term state of the glia, and since some degree of activation may be necessary for the production of a neuroprotective state, and persistent chronic inflammation may be detrimental, it remains to be determined whether shorter or longer minocycline exposure would be beneficial.
In accordance with previous studies, our results suggest that delayed administration of minocycline, either as a monotherapy or combination treatment with CIMT physical therapy, significantly reduced spatial memory impairment and use of an inefficient thigmotaxic search strategy during the water maze acquisition test. Thigmotaxic behavior may be a manifestation of increased anxiety-like behavior or disinhibition induced by TBI. Conversely, our results suggest that minocycline is not effective in reducing motor asymmetry or manual dexterity, in spite of the reduction of inflammation in the perilesion cortex. The TBI model used here produces extensive sensorimotor cortex primary damage, with relative sparing of hippocampal structure, but with substantial microglial and astroglial reaction. Trends in memory behavior responses correlate roughly with the degree of decrease of the astroglial response in the hippocampus (compare Figures 1A and D with 6D and F), suggesting that this component of the inflammatory response, or an upstream activator, may contribute to hippocampal dysfunction. The lack of effect of minocycline in gross motor recovery suggests that the anatomic substrates involved are relatively indifferent to microglial inhibition but responsive to physical training; such substrates might include distant ipsilateral cortex, contralateral cortex and striatum, brainstem, and spinal cord.40,41 Both minocycline sensitive and insensitive functions appear to be necessary for optimal recovery of behavior requiring fine dexterity. Further investigation is needed to isolate the effects of minocycline and botox/physical therapy on motor function and how they affect the regions involved biochemically and structurally.
Footnotes
Acknowledgements
We thank Drs Linda Noble, Gary Abrams, Anthony Chen, and Charles McCulloch for their helpful comments.
Authors’ Note
Tina I. Lam and Deborah Bingham contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a Veterans Affairs Research Enhancement Award (RAS), and Veterans Affairs merit awards (JL, SM).].
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
