Abstract
Background. Recent rehabilitation efforts after stroke often focus on increasing walking speed because it is associated with quality of life. For individuals poststroke, propulsive force generated from the paretic limb has been shown to be correlated to walking speed. However, little is known about the relative contribution of the paretic versus the nonparetic propulsive forces to changes in walking speed. Objective. The primary purpose of this study was to determine the contribution of propulsive force generated from each limb to changes in walking speed during speed modulation within a session and as a result of a 12-week training program. Methods. Gait analysis was performed as participants (N = 38) with chronic poststroke hemiparesis walked at their self-selected and faster walking speeds on a treadmill before and after a 12-week gait retraining program. Results. Prior to training, stroke survivors increased nonparetic propulsive forces as the primary mechanism to change walking speed during speed modulation within a session. Following gait training, the paretic limb played a larger role during speed modulation within a session. In addition, the increases in paretic propulsive forces observed following gait training contributed to the increases in the self-selected walking speeds seen following training. Conclusions. Gait retraining in the chronic phase of stroke recovery facilitates paretic limb neuromotor recovery and reduces the reliance on the nonparetic limb’s generation of propulsive force to increase walking speed. These findings support gait rehabilitation efforts directed toward improving the paretic limb’s ability to generate propulsive force.
Introduction
Stroke is a leading cause of long-term disability. 1 Because improving walking ability has been found to be the best way to reduce dependency after stroke,2-4 restoring gait function has become a critical goal of poststroke rehabilitation. 5 A common measure of gait function is walking speed, which has been shown to be correlated with community walking ability and quality of life. 6 Unfortunately, more than 60% of individuals who achieve independent ambulation still walk at speeds that are insufficient to function effectively in the community (ie, <0.8 m/s).5,7 Consequently, rehabilitation efforts focus on increasing walking speed. 8
During gait, the propulsive forces from both legs propel the body forward and are, therefore, critical to walking speed. 9 For individuals poststroke, it has been shown that the propulsive force generated from the paretic limb is predictive of walking speed 10 and increases when walking speed is increased within a session. 11 Moreover, increases in paretic propulsive force are associated with increases in walking speed observed following rehabilitation. 12 Thus, paretic propulsion has been frequently emphasized in recent studies of hemiparetic walking.12-15 However, because muscle weakness, 16 low propulsive forces, 10 and motor control deficits have been identified in the paretic limb, increases in paretic propulsive force may have relatively insignificant influences on increases in walking speed compared with the nonparetic limb. That is, the nonparetic limb may have greater capacity to increase total forward propulsive force than the paretic limb and could thus dominate increases in walking speed. Indeed, Bowden and colleagues showed that more severely affected stroke participants would use the nonparetic propulsion as a compensation. 10 On the other hand, it is also possible that the paretic limb’s ability to generate propulsive force may be the primary limiting factor to increased walking speed 17 and would therefore have a greater influence on increases in walking speed compared with the nonparetic limb. Although the relationship between increases in paretic propulsive force and walking speed has been documented in previous studies, the relative contribution of paretic versus nonparetic propulsive force to increasing walking speed from a self-selected pace to a faster pace (ie, during poststroke speed modulation) has not been identified and warrants investigation.
Additionally, analysis of the relative contribution of the changes in paretic versus nonparetic propulsive force to the increases in walking speeds resulting from gait training (ie, pre-post an intervention) can provide additional insight into the mechanisms underlying faster poststroke walking speeds. To our knowledge, previous intervention studies that have investigated the relationship between increases in propulsive force and increases in walking speed have primarily been restricted to the study of propulsion symmetry.18-22 However, a potential mechanism to increase walking speed is to increase propulsive force from both the paretic and the nonparetic limbs. Indeed, a study reported increased propulsive impulse in both limbs following an 8-week body-weight-supported treadmill training in individuals with chronic stroke. 22 Interestingly, propulsion symmetry did not change following intervention in their study. Because propulsion symmetry does not capture the changes in propulsion in each limb, propulsion symmetry alone cannot be used to elucidate the contribution from the paretic versus nonparetic limbs in improving walking speed. Examining the changes in propulsive force generated from each limb is necessary for determining the specific mechanisms underlying improved walking speed following gait training.
Despite its clinical and functional importance, the relationship between changes in nonparetic propulsive force and changes in walking speed, both within a session (ie, during speed modulation) or across sessions (ie, pre-post an intervention), has not been investigated. A better understanding of how changes in nonparetic and paretic propulsive forces influence changes in poststroke walking speed would inform future rehabilitation efforts. Thus, the purpose of this study was to determine the relative contribution of the paretic versus the nonparetic propulsive force to walking speed after stroke. Specifically, we studied the relative contribution from propulsive forces in each limb to (a) baseline walking speed, (b) changes in walking speed as a result of speed modulation within a session, and (c) changes in walking speed as a result of 12 weeks of gait training.
Methods
Participants
A total of 45 participants (age = 58.3 ± 11.8 [SD] years; time since stroke = 4.5 ± 6.5 years; 17 females; 16 right hemiparetic; self-selected walking speed = 0.7 ± 0.3 m/s) with poststroke hemiparesis participated in this study. Participant inclusion criteria were a single cortical or subcortical stroke, a poststroke duration of at least 6 months, the ability to ambulate without the assistance of another individual, sufficient cognitive function to follow instruction and communicate with the investigators, the ability to walk for 6 minutes without orthotic support, sufficient passive dorsiflexion range of motion to position the ankle in a neutral position with the knee extended, and sufficient passive hip extension to extend the hip 10°. Individuals were excluded from participating if they had a history of multiple strokes, cerebellar stroke, lower extremity joint replacement, bone or joint problems that limited their ability to walk, a resting heart rate outside of the range of 40 to 100 beats per minute, a resting blood pressure outside of the range of 90/60 to 170/90 mm Hg, neglect and hemianopia, unexplained dizziness during the past 6 months, or chest pain or shortness of breath without exertion. This study was approved by the institutional review board of the University of Delaware, and all participants provided written informed consent to participate in this study.
Gait Evaluation
Participants completed evaluations at baseline (pre) and after 12 weeks of locomotor retraining (post). Kinetic and kinematic data were collected via an 8-camera motion analysis system (Motion Analysis Corp, Santa Rosa, CA) as participants walked at their self-selected (SS) and faster (FS) speeds on a split-belt treadmill (Bertec Corp, Columbus, OH) instrumented with 2 independent 6 degree of freedom force plates capturing at 1080 Hz. Previous work has described in detail the gait analysis setup.12,23,24 For safety, participants were connected to an overhead harness system and were allowed to hold onto handrails if they normally utilized an assistive device or if they felt unsafe walking on a treadmill. Participants were only allowed to use handrails located at the side of the treadmill, which mimicked walking with an assistive device. No bodyweight was supported by the harness, and verbal instructions on using the handrails as minimal as possible were provided. Self-selected walking speed was defined as the participant’s comfortable overground walking speed during a 6-meter walk test and faster walking speed was the fastest speed that participants could maintain for at least 4 minutes of continuous walking on the treadmill. A familiarization period of 1 minute was provided before data collection. Because investigation from our laboratory has identified much stronger correlations between walking speed and peak anterior ground reaction force (AGRF) compared with AGRF impulse (force-time integral), peak AGRF was used in the present study. Peak AGRF was measured as the maximum AGRF normalized to body weight. Kinematic and kinetic data were filtered using a bidirectional Butterworth low-pass filter at 6 and 30 Hz, respectively. Two trials were recorded in each session. Data from the first trial were used in this study. Data from the second trial were used only when the first trial is not useable. Peak propulsive forces were averaged across strides with 30-second trial duration for each trial.
Training
Participants were assigned to 1 of 3 training groups that were equivalent in structure: (a) walking training at a self-selected speed, (b) walking training at the fastest speed that subjects could maintain for at least 4 minutes, and (c) walking training at the fastest speed that subjects could maintain for at least 4 minutes with the addition of functional electrical stimulation applied to the paretic limb dorsiflexors and plantarflexors. Regardless of group, participants completed 3 sessions a week for a total of 12 weeks. Each session consisted of six 6-minute bouts of walking. Participants walked on a treadmill for bouts 1 to 5 and walked overground for bout 6. For the third group, functional electrical stimulation was delivered to ankle dorsiflexor muscles during the first, third, and fifth minutes for bouts 1 to 5. Rest breaks of up to 5 minutes were allowed between walking bouts. More details on the intervention may be found in previous work from our laboratory.12,23
Statistical Analysis
Data from 4 trials were analyzed: pretraining self-selected (Pre-SS) and faster (Pre-FS) walking speed and posttraining self-selected (Post-SS) and faster (Post-FS) walking speed (see Figure 1). Outcome measures included 3 variables: paretic propulsive force, nonparetic propulsive force, and walking speed. For each trial, Pearson correlation coefficients were calculated to determine the correlations between the propulsive forces in each limb versus walking speed. A 3-way speed (SS vs FS) × time (pretraining vs posttraining) × limb (paretic vs nonparetic limb) analysis of variance (ANOVA) and a 2-way speed × time ANOVA were used to analyze propulsive force and walking speed, respectively. Post hoc pairwise comparisons were performed using Fisher’s least significant difference if interactions were significant.
Illustration of trials and changes among trials studied.
Next, the differences in outcome measures between conditions were calculated (see Figure 1). Specifically, “Speed modulationpre” was calculated as the change from Pre-SS to Pre-FS, “Speed modulationpost” was calculated as the change from Post-SS to Post-FS, “Change in SS” was calculated as the change from Pre-SS to Post-SS, and “Change in FS” was calculated as the change from Pre-FS to Post-FS. Because this research was part of a larger study of 3 interventions that varied in intensity, we combined groups with an anticipation of widespread range of responses, as previously done. 25 Correlations between changes in propulsive forces from each limb versus changes in walking speed were analyzed using the Pearson correlation coefficient. In addition, linear regression analysis was performed to identify the independent contribution of paretic and nonparetic propulsive force to the changes in walking speed observed. The significance level was set at an α of .05. All statistics were run using SPSS (version 20.0, SPSS, Inc).
Results
Of the 45 participants, 4 walked with the same self-selected and faster walking speeds at pretraining; therefore, data for these participants were excluded. Of the remaining 41 participants, 3 had unusable ground reaction force data due to technical issues during pretraining. Thus, the results of 38 participants (age = 58.3 ± 12.2 years; time since stroke = 4.1 ± 6.2 years; 14 females; 14 right hemiparetic, self-selected walking speed = 0.68 ± 0.3 m/s) were analyzed.
At pretraining, both the paretic and nonparetic AGRF explained more than 60% of the variance in self-selected and faster walking speeds (Figure 2A-D). At posttraining, the paretic AGRF explained more than 69% of the variance (Figure 2E and G) and the nonparetic AGRF explained more than 58% of the variance (Figure 2F and H) in both self-selected and faster walking speeds. Based on the results of the 3-way ANOVA, significant differences in propulsive force were observed for the FS versus SS (F = 15.7, P < .01), the post versus pre (F = 11.8, P < .01), and the nonparetic versus the paretic limb (F = 63.5, P < .01). Based on the 2-way ANOVA, significant differences in walking speed were observed for the FS versus SS (F = 9.8, P < .01) and the post versus pre (F = 6.6, P = .01). No significant interactions were detected for either analysis.
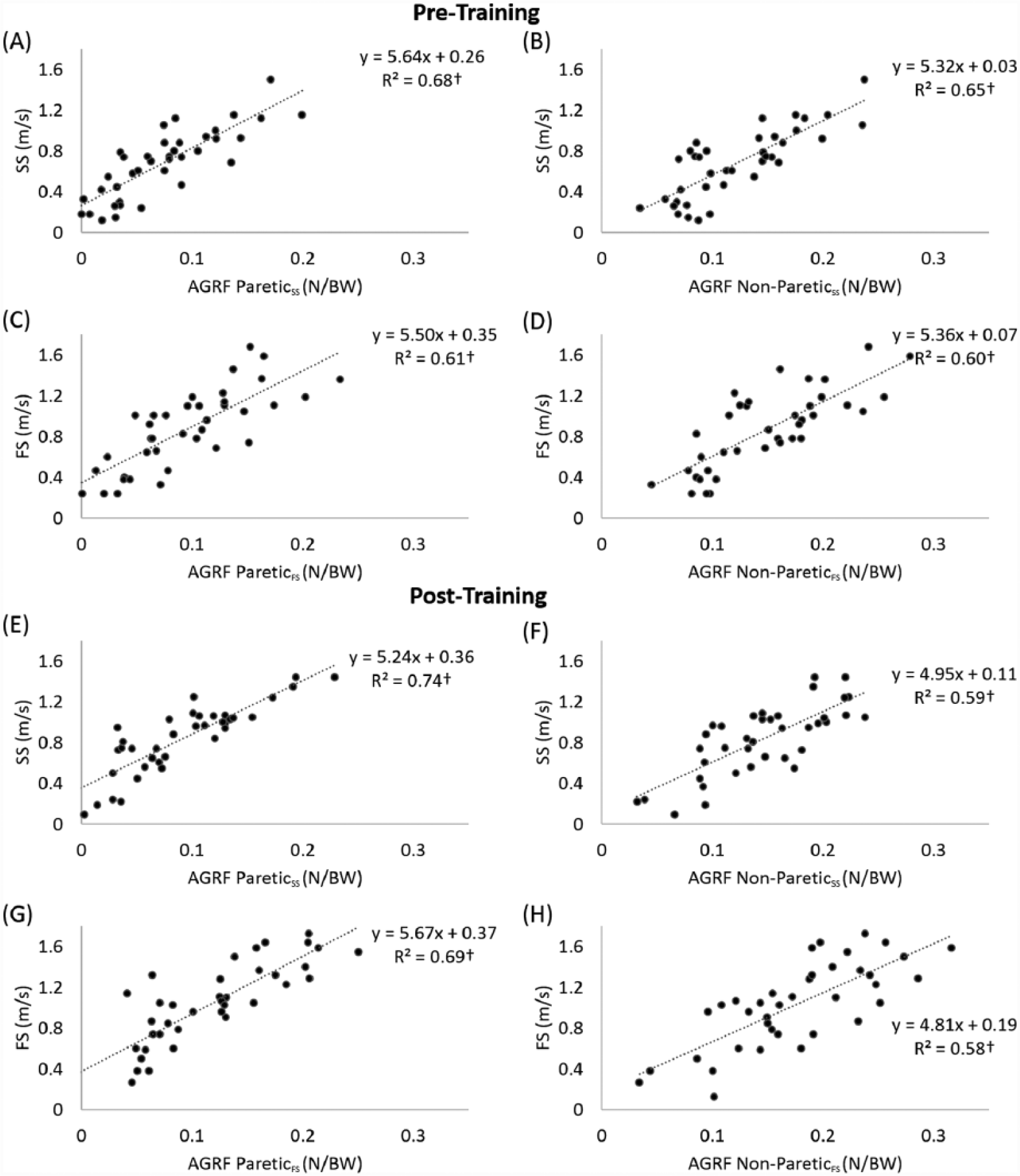
Relationships between anterior ground reaction force (AGRF) and walking speed (N = 38): (A-D) Pretraining; (E-H) Posttraining.
Speed Modulation Within a Session
Speed Modulation (Pretraining)
At baseline, participants’ faster speeds were 28% faster than their self-selected speeds (from 0.68 to 0.87 m/s; Figure 3B). The average corresponding changes in propulsive force from the paretic and the nonparetic limbs were 28% (from 0.074 to 0.095 N/BW) and 22% (from 0.122 to 0.149 N/BW), respectively (Figure 3A). Changes in paretic AGRF explained 26% of the variance, and changes in nonparetic AGRF explained 49% of the variance, of changes in walking speed (Figure 4A and B). Only changes in nonparetic propulsive force independently contributed to changes in walking speed (nonparetic β = 0.6, P < .01; paretic β = 0.18, n.s.). The overall model fit was R2 = 0.48.
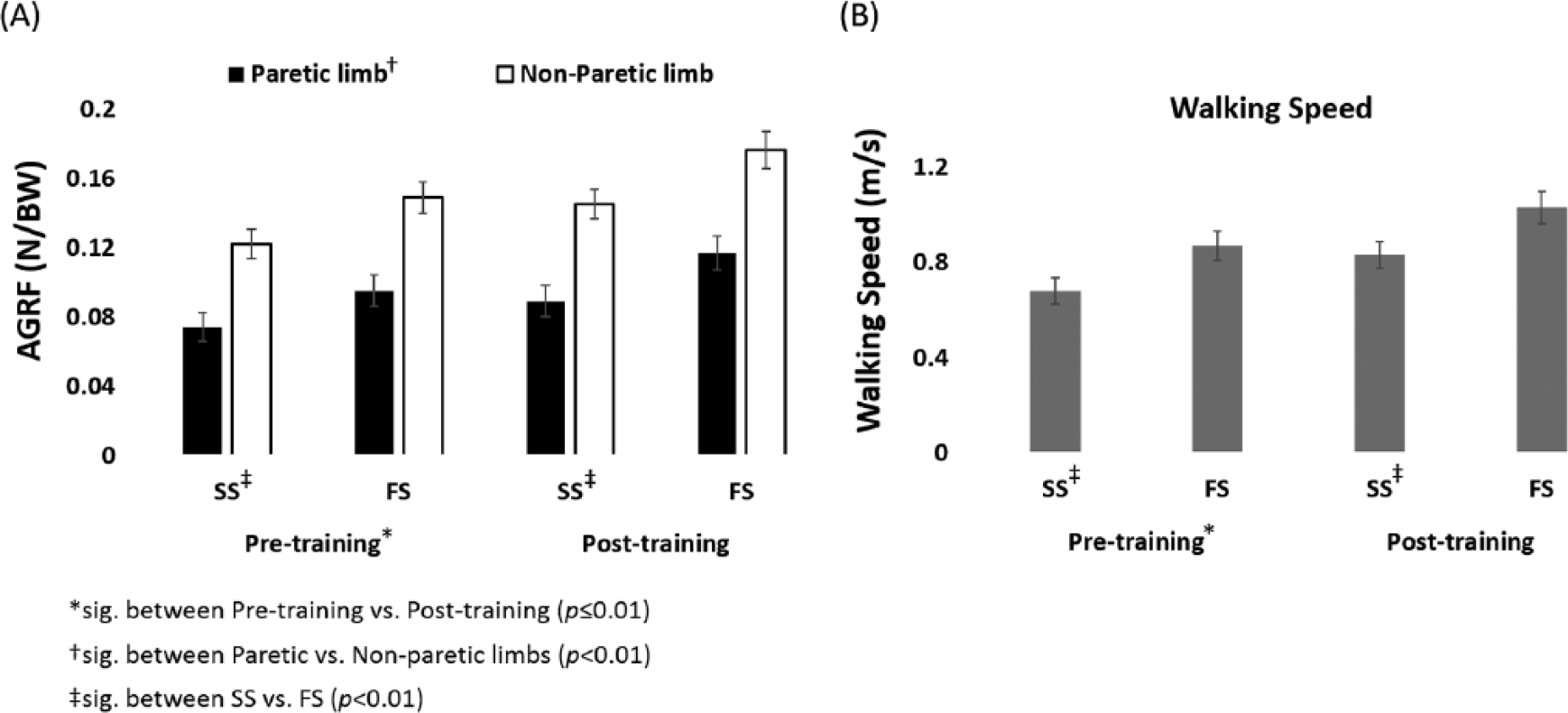
Mean and standard error of propulsive force and walking speeds at self-selected (SS) and faster (FS) walking speeds for pre- and posttraining.
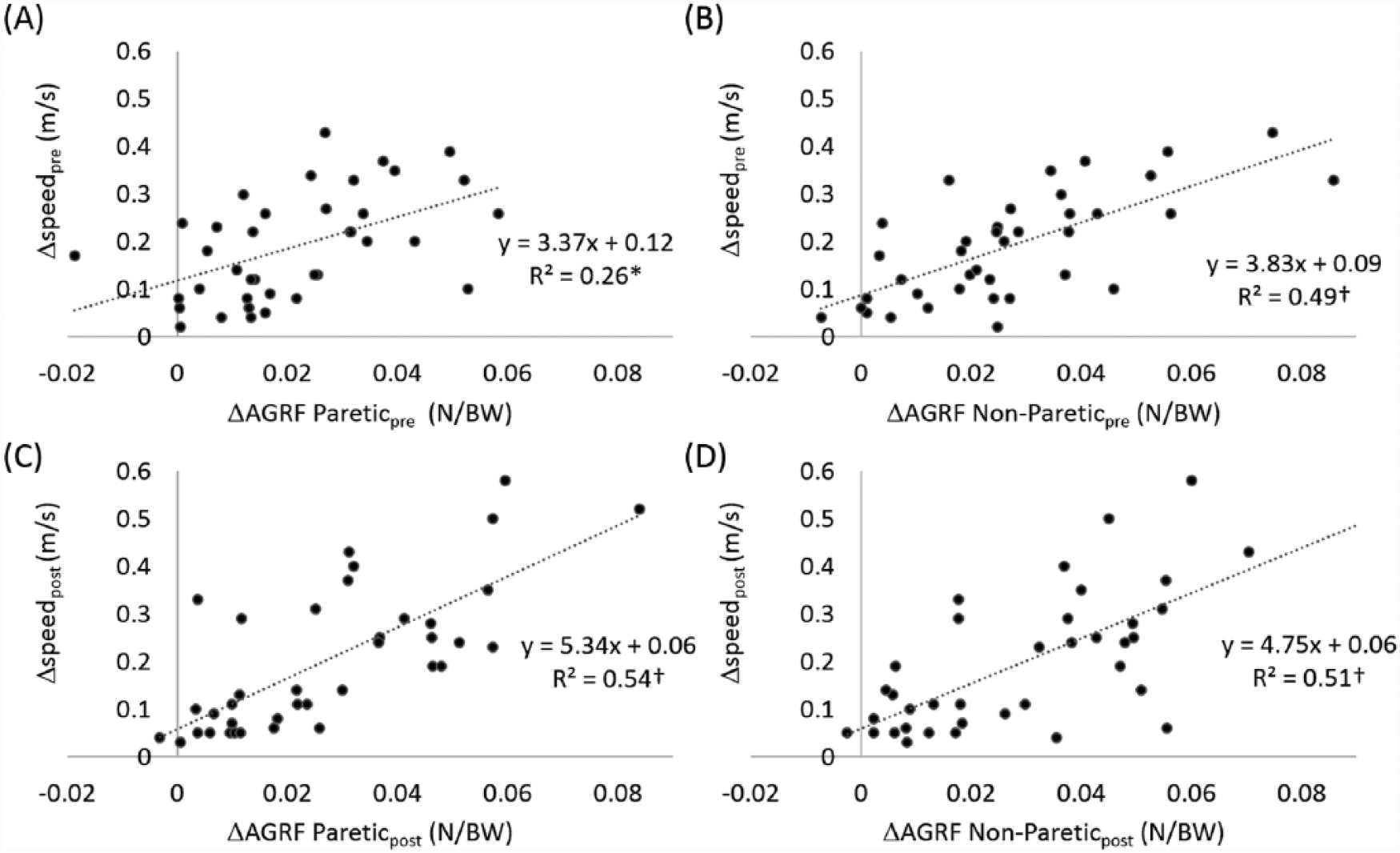
Relationships between changes in anterior ground reaction force (ΔAGRF) and changes in walking speed during speed modulation within a session prior to (Δspeedpre) and following (Δspeedpost) the 12-week training (N = 38).
Speed Modulation (Posttraining)
After training, participants’ faster speeds was 24% faster than their self-selected speeds (from 0.83 to 1.03 m/s; Figure 3B). The average corresponding changes in propulsive force from the paretic and the nonparetic limbs were 31% (from 0.089 to 0.117 N/BW) and 21% (from 0.145 to 0.176 N/BW), respectively (Figure 3A). Changes in paretic AGRF explained 54% of the variance and changes in nonparetic AGRF explained 51% of the variance of changes in walking speed (Figure 4C and D). Both changes in paretic and nonparetic propulsive forces were significant independent predictors of changes in walking speed, with changes in paretic propulsion being the stronger predictor (paretic β = 0.46, P < .01; nonparetic β = 0.39, P < .05). The overall model fit was R2 = 0.59.
Changes as a Result of Training
Change in SS
Average self-selected walking speeds increased 22% from pretraining (0.68 m/s) to posttraining (0.83 m/s; Figure 3B). The average corresponding increases in propulsive force from the paretic and the nonparetic limbs were 20% (from 0.074 to 0.089 N/BW) and 19% (from 0.122 to 0.145 N/BW), respectively. Changes in paretic and nonparetic AGRF explained 54% and 43% of the variance of changes in self-selected walking speed, respectively (Figure 5A and B). Both changes in paretic and nonparetic propulsive forces were significant independent predictors of changes in self-selected walking speed as a result of the training, with changes in paretic propulsion being the stronger predictor (paretic β = 0.53, P < .01; nonparetic β = 0.34, P < .05). The overall model fit was R2 = 0.59.
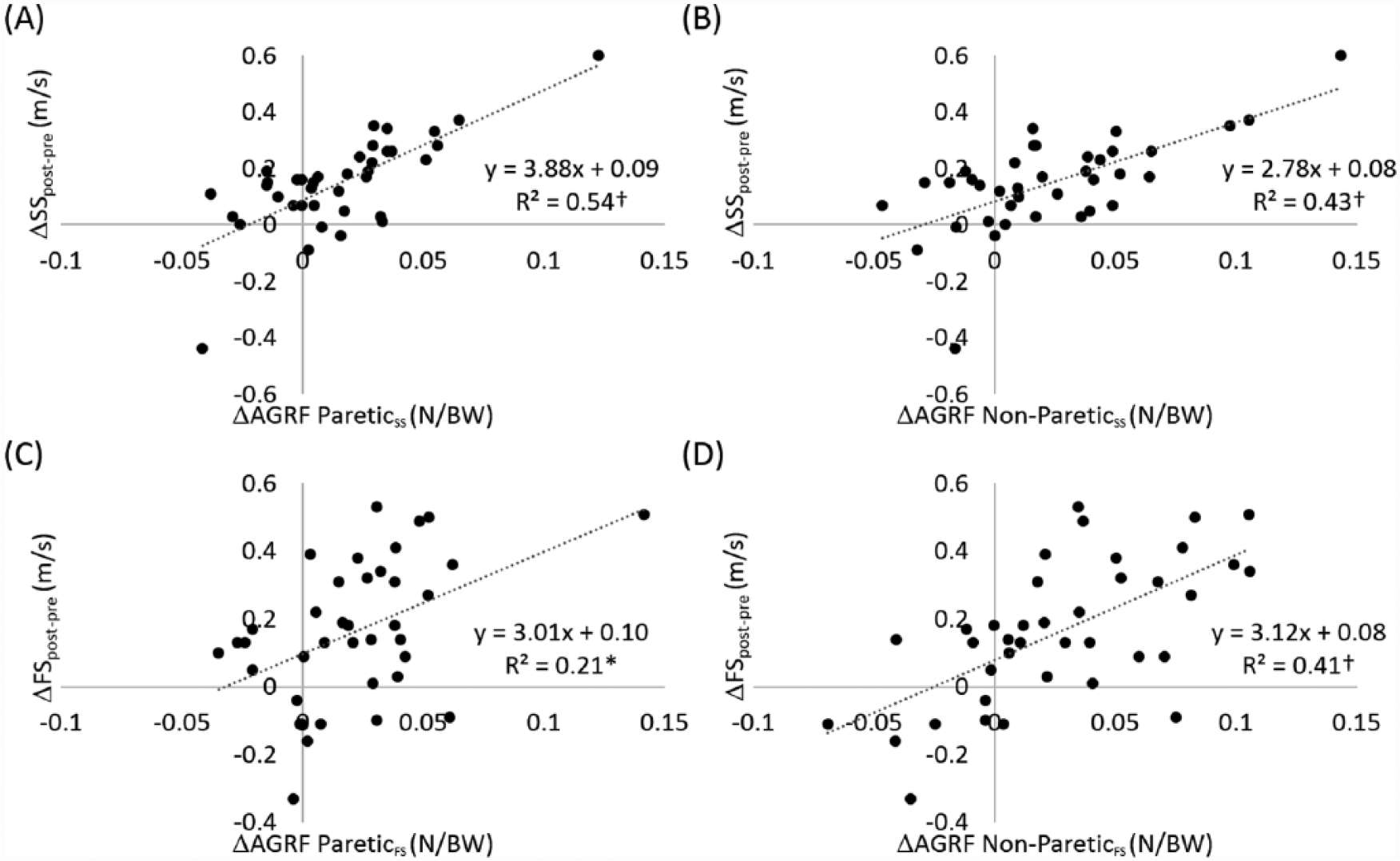
Relationships between changes in anterior ground reaction force (ΔAGRF) and changes in self-selected (ΔSS) and faster (ΔFS) walking speed as a result of the 12-week training.
Change in FS
Average faster walking speeds increased 18% from pretraining (0.87 m/s) to posttraining (1.03 m/s; Figure 3B). The average corresponding increases in propulsive force from the paretic and the nonparetic limbs were 23% (from 0.095 to 0.117 N/BW) and 18% (from 0.149 to 0.176 N/BW) at faster walking speeds, respectively. Changes in paretic and nonparetic AGRF explained 21% and 41% of the variance of changes in faster walking speed, respectively (Figure 5C and D). Only changes in nonparetic propulsive force independently contributed to changes in faster walking speed as a result of the training (nonparetic β = 0.57, P < .01; paretic β = 0.13, n.s.). The overall model fit was R2 = 0.39.
Discussion
In this study, we examined how the propulsive forces generated from the paretic and nonparetic limbs of persons poststroke contributed to speed modulation within a session and the increases in walking speed observed after 12 weeks of gait training. The major finding of this investigation was the more prominent role that paretic propulsive force (vs nonparetic propulsive force) played following gait training in determining faster poststroke walking speeds. Indeed, following 12 weeks of gait training, participants’ paretic propulsive force contributed more to speed modulation within a session and to the increases in self-selected walking speed observed following training. However, despite changes in each paretic and nonparetic propulsive force being related to the increases in faster walking speed observed following gait training, only changes in nonparetic propulsive force independently contributed to the increases in faster walking speeds observed following training. These findings extend previous work that has investigated propulsive force generation after stroke 10 by demonstrating that the compensatory reliance on the nonparetic limb to increase forward propulsion during speed modulation can be reduced following gait training; however, there is an apparent need to study changes in the propulsive force of each limb at both self-selected and faster speeds as changes in paretic and nonparetic limb propulsive forces contribute differently to increases in each speed.
Relationships Between Propulsive Force and Walking Speed
In agreement with the study by Bowden and colleagues, 10 we demonstrated that stroke survivors who walked with greater paretic propulsive force tended to walk faster (Figure 2). Contrary to the findings of Bowden et al, 10 the present study also demonstrated a correlation between nonparetic propulsive force and walking speed (Figure 2). This conflicting finding could originate from methodological differences between the studies. The present study measured peak AGRF, whereas Bowden and colleagues used AGRF impulse. However, data from our laboratory showed that peak AGRF has a higher correlation with walking speed compared to AGRF impulse. Because the measurement of impulse is related to the propulsive duration, which decreases when walking speed increases, AGRF impulse may not be sensitive to changes in walking speed. In contrast, peak AGRF increases with increased walking speed. Thus, we believe that peak AGRF is more appropriate for studying the relationship between propulsion and walking speed. The present results showed that although the propulsive force is lower in the paretic limb compared with the nonparetic limb, propulsive forces from each limb contribute to walking speed.
Speed Modulation (Pretraining)
Previous work investigating changes in joint power during speed modulation offers a biomechanical explanation for our finding that changes in nonparetic propulsion were the primary mechanism for modulating walking speed prior to training. Specifically, Jonkers et al demonstrated that slower walkers “engaged excessive paretic ankle plantarflexor power generation at self-selected walking speeds” and failed to increase paretic ankle plantarflexor power generation at their maximal walking speed. 26 That is, the most affected individuals poststroke utilize their full paretic propulsive force capacity to walk at their self-selected speed and do not have the capacity to increase walking speed via increasing paretic propulsive force and, thus, must compensate with the nonparetic limb to walk at a faster velocity.
Speed Modulation (Posttraining)
Interestingly, at posttraining the change in paretic propulsive force had a greater influence on changes in walking speed compared with the change in nonparetic propulsive force (β = 0.46 vs 0.39). This finding suggests that following gait training, individuals poststroke modulate their walking speeds via a different mechanism than pretraining. It is likely that the gait training increased our participants’ capacity to generate propulsive force by the paretic limb, ultimately reducing their compensatory reliance on the non-paretic limb for propulsion.
It is worth noting that participants increased their paretic and nonparetic propulsive forces during speed modulation similarly at pretraining (29% and 22%, respectively) and posttraining (31% and 21%, respectively). The reduced reliance on the nonparetic limb to increase walking speed was thus not solely due to the generation of more paretic propulsive force. Rather, this was likely due to a better utilization of paretic propulsive force after training. For example, the contribution of a limb’s propulsive force to walking speed could be influenced by braking force. Sousa and colleagues studied poststroke interlimb coordination during the stance phase of gait and found a negative correlation between paretic propulsion impulse and nonparetic limb braking impulse. 27 That is, a greater braking force from the nonparetic limb may counteract the propulsive force generated from the paretic limb and consequently reduce the translation of increased propulsive force to increased walking speed. Thus, our finding of an increased contribution from paretic propulsive force to increases in walking speed could also be due to improved interlimb coordination of propulsive and braking forces after training. Further investigation of the effect of gait training on braking forces can provide additional insight into the biomechanical mechanisms underlying the recovery of walking speed after stroke.
Changes in Self-Selected Walking Speed Following Training
Our results showed that propulsive force from each limb can be improved via gait training and that improvements in both paretic and nonparetic propulsive forces contribute to improvements in self-selected walking speed. A previous study measured the change in propulsion symmetry following a 12-week locomotor intervention for individuals poststroke and found that improvements in self-selected walking speeds were correlated with the improvements in propulsion symmetry (R2 = 0.22). 21 However, for those who improved walking speed more than 0.16 m/s, improvements in propulsion symmetry failed to account for the improvement in self-selected walking speed. Similarly, in a study of the effects of body-weight-supported treadmill training on kinetic symmetry in persons with chronic stroke, increases in walking speed, paretic propulsion, and nonparetic propulsion were observed following intervention. 22 However, propulsion symmetry did not change after training in their study and it was suggested that the increase in speed after training was likely due to strengthening existing compensatory strategies rather than via kinetic symmetry. Although propulsion symmetry can be indicative of stroke severity 10 and muscle coordination, 28 measurements of symmetry do not quantitatively report the output from each limb. 29 Our results also showed that after training the magnitude of propulsive force generated by the nonparetic limb was still greater than the paretic limb. However, increases in paretic propulsive force have stronger effects compared with nonparetic propulsive force on increases in self-selected walking speed (β = 0.53 vs 0.34) regardless of persisting propulsion asymmetry following training. Thus, measuring propulsive forces from each limb can provide critical information to understand the mechanisms underlying increases in poststroke walking speed. Our finding supports recent studies that emphasize improving paretic propulsive force for rehabilitation.12,30
Changes in Faster Walking Speed Following Training
Interestingly, although significant changes in the paretic propulsive force generated at faster walking speeds were observed from pretraining to posttraining, the changes in paretic propulsive force only explained 21% of the variance in the improvements in faster walking speed (vs 41% for the nonparetic; Figure 5C). Previous evidence suggests that walking at a fast speed provides a better assessment of neuromotor impairments for individuals poststroke.29,31 Because the propulsive forces generated by the nonparetic limb remained larger than the forces generated by the paretic limb following training (Figure 3), it is possible that when walking at maximum speed, individuals poststroke rely more on the nonparetic propulsive force compared with the paretic propulsive force. This is consistent with the findings of Jonkers et al discussed previously. These results support previous work that has suggested that assessment at an individual’s self-selected and faster speed can provide different information important for clinical decision making. 29
Study Limitations
A potential limitation of this study is that comparisons between different training groups were not reported. Because the present study was only concerned with the relationship between changes in propulsive force in each limb and changes in walking speed, the results from all groups were combined to show these general relationships. Subsequent reports will test treatment efficacy by investigating group-specific effects. Another potential limitation of this study is that our participants were allowed to hold onto the handrails if needed. The use of handrails could influence gait patterns and force distribution. However, this also replicated walking with an assistive device. Verbal instructions on using the handrail as minimal as possible were provided during data collection. Our results may not be generalizable to individuals who are unable to walk for 6 minutes without orthotic support. Although the ability to do so was an inclusion criterion for this study, it is important to note that many of our participants regularly used orthotics at home and in the community, but were able to safely walk for 6 minutes without one.
Conclusions
This is the first study to reveal that in persons with chronic hemiparesis gait training is able to reduce the compensatory reliance on the nonparetic limb’s ability to generate propulsive force to increase walking speed. Interestingly, the nonparetic limb continued to be the major determinant of participants’ ability to increase faster walking speed—but not self-selected walking speed—following 12 weeks of gait rehabilitation. These findings provide novel evidence that supports recent gait rehabilitation efforts directed toward improving the paretic limb’s ability to generate propulsive force.
Footnotes
Acknowledgements
We thank Ryan Pohlig for statistical consulting. We also thank Christopher Cutsail and Kevin Lenoir for data collection and processing.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: NIH R44HD062065, R01HD038582, RO1NR010786, and U54GM104941.
