Abstract
Introduction
Multiple sclerosis (MS) is the most common disabling neurological disease afflicting young adults. MS results in impaired balance early in the disease course, even in minimally impaired people.1-4 Balance problems result in falls and reduced mobility, negatively affecting quality of life.5-8 Impaired balance with MS is multifactorial, caused by variable and extensive damage to the central nervous system (CNS). 9 Although studies have shown that exercise training, strength training, and so on can be used to improve balance, no systematic study has been performed to understand if postural control can be learned. 10
Demyelination in the CNS caused by MS may interfere with the ability to learn motor patterns because of the involvement of neural structures responsible for motor learning, including, but not limited to, the sensorimotor cortex, basal ganglia, cerebellum, and spinal somatosensory pathways.11-13 Studies in MS have investigated the capacity for motor learning in people with MS using upper-limb tasks11-15 and gait rehabilitation.16,17 Motor learning has been shown to be preserved during the early phase of MS, 14 but the capacity of motor learning has been shown to be dependent on the severity of MS.12,15 However, the potential for motor learning in postural control tasks in people with MS has not yet been investigated and is the focus of the current study.
Recent findings demonstrate that postural control can be learned with training.18-21 Our laboratory has shown that healthy adults can “implicitly” learn to improve postural responses by reducing their center of mass (CoM) displacements with respect to continuous oscillations of the support surface.18-20 Implicit motor learning refers to the “self-regulated learning” mechanism in which the learned variable is not specified explicitly. In fact, most postural control, such as learning how to ride a bike or how to stand on a boat, is learned implicitly. Understanding the capacity for implicit postural motor learning in people with MS is crucial for mobility rehabilitation.5-7,22 We operationalized the term postural motor learning to describe retention of practice-related improvements in postural stability after 24 hours of practice at a given task.
A critical neurophysiological deficit underlying impaired balance in people with MS is slowed spinal somatosensory conduction, which leads to delayed postural response latencies23-25 and increased postural sway. 26 Automatic postural responses are triggered in response to external disruptions to equilibrium, such as slips or trips or during continuous perturbations such as while standing in a subway car or on a boat. When continuous external perturbations become predictable, however, healthy people learn to anticipate them as indicated by a gradually increasing phase lead of body CoM movement prior to perturbations. 24
In this study, we investigated whether people with MS show postural motor learning deficits during a fixed, continuous pattern of anterior and posterior surface oscillations. Specifically, we investigated whether people with MS have the capacity to (1) change from a feedback control strategy to a predictive, feed-forward control strategy during 1 day of training and (2) retain the learned behavior after 24 hours. We hypothesized that people with MS would demonstrate impaired postural motor performance and postural motor learning and that their motor performance and learning impairments would be related to the extent of their somatosensory impairment, measured as the increase of standing postural sway in the eyes-closed versus eyes-open condition. 27
Methods
Participants
A total of 24 individuals with MS (21 female [F], 3 male [M]; 48.4 ± 10.9 years old) and 14 age-matched non-neurologically impaired adults (11 F, 3 M; 46.9 ± 13.4 years old) volunteered to participate in the study. The institutional review board of the Oregon Health and Science University approved the methods used in this study. All participants signed an informed consent prior to participation. Participants were free of orthopedic, psychological, or other neurological disorders that could affect their ability to perform the task, were able to maintain balance independently by standing on their toes for 3 s, and had the capability to walk 500 m without a walking aid. Patients were mildly affected, based on Expanded Disability Status Score (EDSS) and MiniBESTest scores. EDSS scores for the participants with MS ranged from 2 to 4.
Experimental Procedures
Participants visited the laboratory on two consecutive days for the acquisition (day I) and retention (day II) paradigms with approximately 24 hours in between. Participants stood with each foot on a hydraulically driven, servocontrolled platform.18-20 The platform oscillated at a fixed frequency of 0.5 Hz sinusoidally, with one defined sequence composed of horizontal translations, in forward and backward directions of variable magnitude (Figure 2). Participants were asked to maintain balance while keeping the arms crossed across their chest and looking straight ahead at a picture kept approximately 2 m from them. To ensure safety, participants wore an industrial safety harness tethered to a sliding hook on an overhead rail with enough slack to avoid any support unless a participant began to fall. Trials were 48 s long with 3 s of quiet period at the beginning and a 2 s quiet period at the end. The same sequence was repeated at each trial. For each participant, the maximum distance moved by the platform was scaled to a personal maximum (largest amplitude without taking a step) as determined by a 20 s, constant-amplitude practice trial (range = 7-15 cm). 18 To decrease the likelihood of a step or fall, the first movement of the platform was always in the backward direction.
The testing paradigm consisted of 5 blocks of 5 trials with rest periods between blocks. To separate temporary performance effects from more permanent changes in behavior that would reflect learning, participants returned for a retention test approximately 24 hours following practice.
Body Kinematic Recording
Body CoM was calculated from kinematics recorded via a Motion Analysis System (Motion Analysis Corporation, Santa Rosa, CA) with 8 cameras that captured 3-dimensional spatial coordinate information about body segment displacements and platform movement. Reflective markers were placed bilaterally on the following anatomical landmarks: fifth metatarsophalangeal, lateral malleolus, lateral femoral condyle, greater trochanter, anterior superior iliac spine, iliac crest, styloid process, olecranon, acromium process, lateral mandibular joint, cervical seventh, lumbar fifth level, and the xyphoid process. A marker was also placed on the platform. Data were sampled at 60 Hz and low-pass filtered using a second-order, dual-pass Butterworth filter with a cutoff frequency of 5 Hz. The position of the CoM of each body segment in the anteroposterior (AP) direction was calculated using the kinematic data and anthropometric data provided by Winter. 28 Whole-body CoM position (in space) in the AP direction was derived from the weighted sum of the individual CoM locations using a custom-designed MATLAB program (Mathworks, Natick, MA).
Postural Sway Measure
An instrumented test of postural sway was administered during 1 trial of a 30 s quiet stance with eyes closed and with eyes open while standing on a firm surface with feet together (Figure 1). A single, body-worn inertial sensor (APDM Inc) that included 2D accelerometers was used to provide objective measures of postural sway (Figure 1). 29 The reliability, sensitivity, and validity of the single body-worn sensor has been established in people with Parkinson’s disease. 29 The sensor was positioned with a Velcro belt on the lower trunk, near the body CoM, and data were streamed to a laptop and automatically analyzed with MobilityLab by APDM. 30 The Romberg Quotient was calculated as the area of sway with eyes closed divided by the area of sway with eyes open. 27 The increase of sway with eyes closed, compared with eyes open, has been shown to be significantly larger in patients with poor use of somatosensory information for postural control.31-33
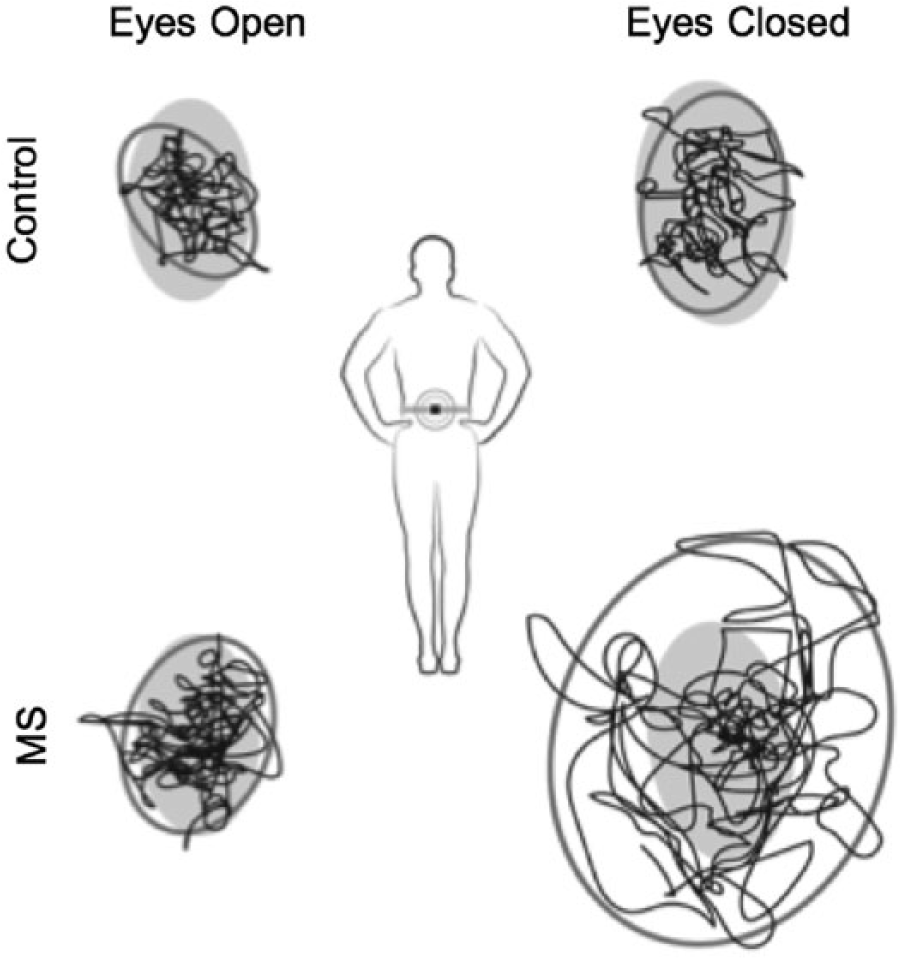
Instrumented test of postural sway during quiet stance on firm surface with eyes open (left) and eyes closed (right): stabilogram shows postural sway from one representative control (top panel) and multiple sclerosis participant (bottom panel). The Romberg quotient, an indicator of somatosensory deficits, is computed as the ratio of the sway area for the eyes closed and eyes open condition. Area in gray represents the normative values from healthy control participants.
Data Analyses
Postural Control During Training
Temporal measure of postural training
Mean relative phase of body CoM displacement relative to platform displacement was derived to examine the temporal control of the CoM with respect to the platform motion with training. 18 Greater negative value for the mean relative phase reflected the CoM motion “lagging” behind the platform perturbations, consistent with feedback control, with postural response latencies contributing to the lag (Figure 2, top panel). The ability of the system to move toward the zero mean relative phase would reflect the ability of the CoM to “lead,” and thus predict, the platform perturbations, consistent with feed-forward postural control (Figure 2, bottom panel). Therefore, a shift from a lag to lead of the CoM versus platform motion was considered to be a shift from a feedback to a feed-forward strategy, indicating the ability of the system to adapt/plan responses to postural perturbations.
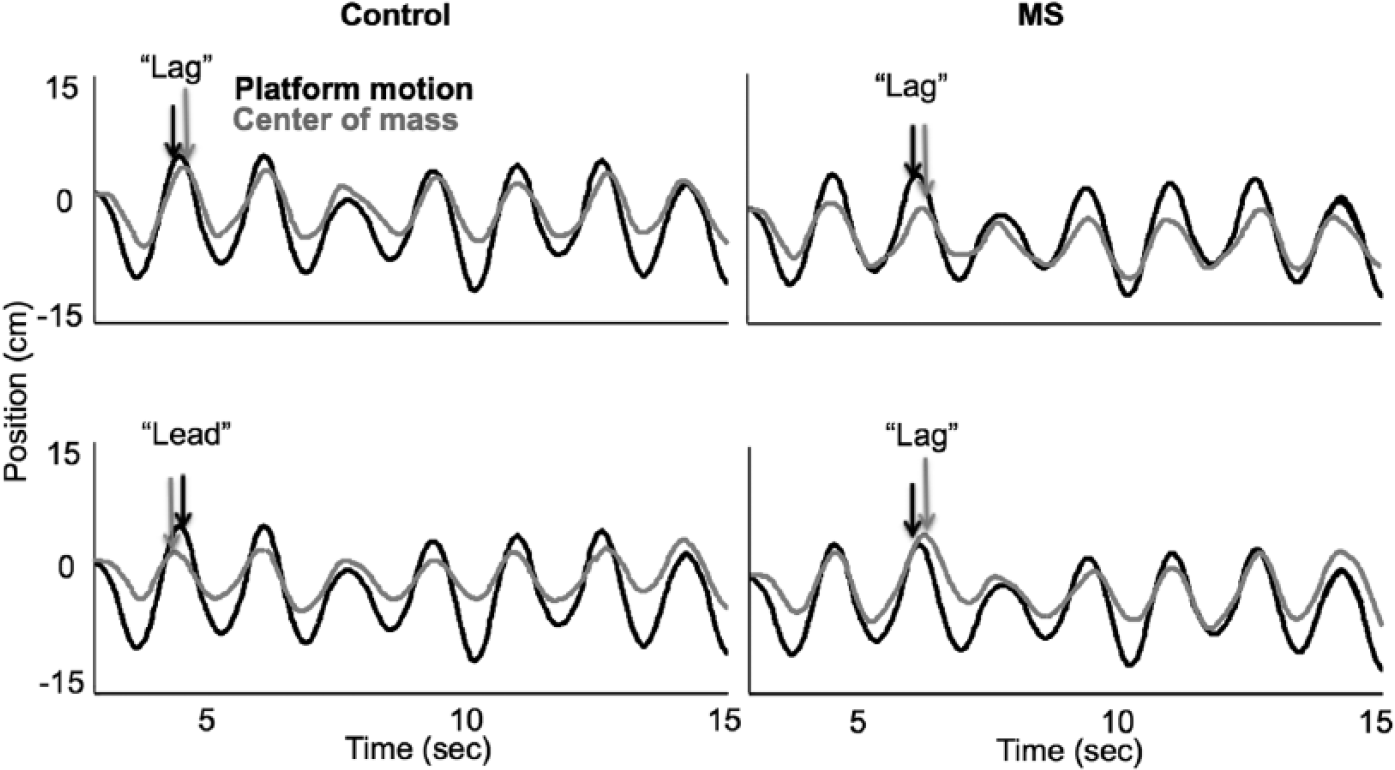
Illustration of the mean relative phase between the platform motion (black) and the center of mass (gray) for a control (left) and multiple sclerosis (MS; right) patient. The top row shows the mean relative phase in the early training block, and the bottom row shows the late training block. With training, the phase “lag” relationship between the platform motion and center of mass changes to phase “lead” for the control participant but not so for the MS patient. The illustration shows the lag and lead relationship for one of the peaks. The average value for mean relative phase across different peaks and valleys were used for the analysis.
Spatial measure of postural training
Mean gain of the CoM (CoM peak displacement/Platform peak displacement) was selected as the spatial measure of CoM control. The ratio of maximum CoM displacement to maximum platform displacement was calculated for each peak and valley event during platform motion, and these values were averaged for each segment within a trial to determine mean gain. For example, a CoM gain of 1.0 would correspond to equal displacements of the platform and CoM in space and would occur if participants were following platform motion. Reduction in CoM gain from 1.0 was considered improved balance control as participants stabilized their CoM in space. 34
We investigated (1) motor performance and (2) motor “learning.” Operationally, we defined motor performance as the comparison of postural control between the control and the MS groups on day I. Statistically, motor performance was tested between MS and control groups (1) using a repeated-measures ANOVA (Bonferroni corrections) with 5 training blocks as within-subjects factors and with group (MS and control) as the between-subjects factor (2 groups x 5 blocks) and (2) by comparing the difference between the final and the baseline (first) training block between two groups.
We investigated postural motor learning with univariate ANOVA, with group (MS and control) as between-subjects factor and the difference between the retention block on day II and the first training block on day I as within-subjects factor.
To determine the relationship of postural motor performance and motor learning with the somatosensory deficits, we performed correlation analysis between the Romberg quotient and the mean relative phase and gain for baseline (first training block, day I), acquisition, and retention. Data were normally distributed; therefore, we used the Pearson test for correlation analysis. An acceptable significance level for all tests was .05.
Postural Sway in Quiet Stance
To estimate the extent of somatosensory deficits, we compared postural sway area while standing on a firm surface with eyes closed versus eyes open with feet together. The Romberg quotient for total postural sway area, which is defined as the ratio of total sway with eyes closed divided by sway with eyes open, served as a surrogate for somatosensory deficits. 27
Results
Postural Motor Performance
The control group performed better than the MS group for the mean relative phase measure—that is, the control group had lower mean relative phase than the MS group (main effect of group: F1,36 = 8.9, P < .01; Figure 3, Table 1) irrespective of the testing block (2 groups × 5 blocks, P = .98). Thus, the body CoM lagged behind the platform motion more for the MS than the control group. However, the extent of improvements in temporal control of CoM as measured by the comparison of final training block versus baseline was similar between the MS and control groups (acquisition, P = .77).
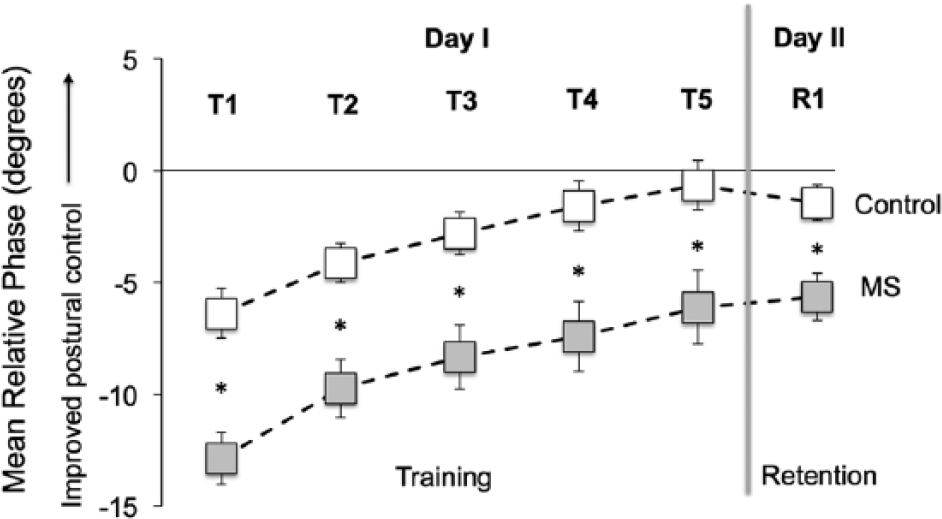
Group changes in the relative phase between the CoM and platform motion during training on day 1 (T1 − T5); retention (R1) testing on day II. To test the retention of the learned pattern, participants revisited the lab the next day after a 24-hour gap (vertical thick gray line). Relative mean phase greater than zero represents the lead relationship between the CoM and platform motion. Error bars represent the standard error of the mean, with some error bars hidden by square symbols. The control group had significantly better postural motor performance across all training blocks, but no group differences were observed in postural motor learning; *P < .05.
Mean Relative Phase and Gain Between the CoM and Platform Motion for the First and Final Training and Retention Blocks and for Acquisition and Retention. a
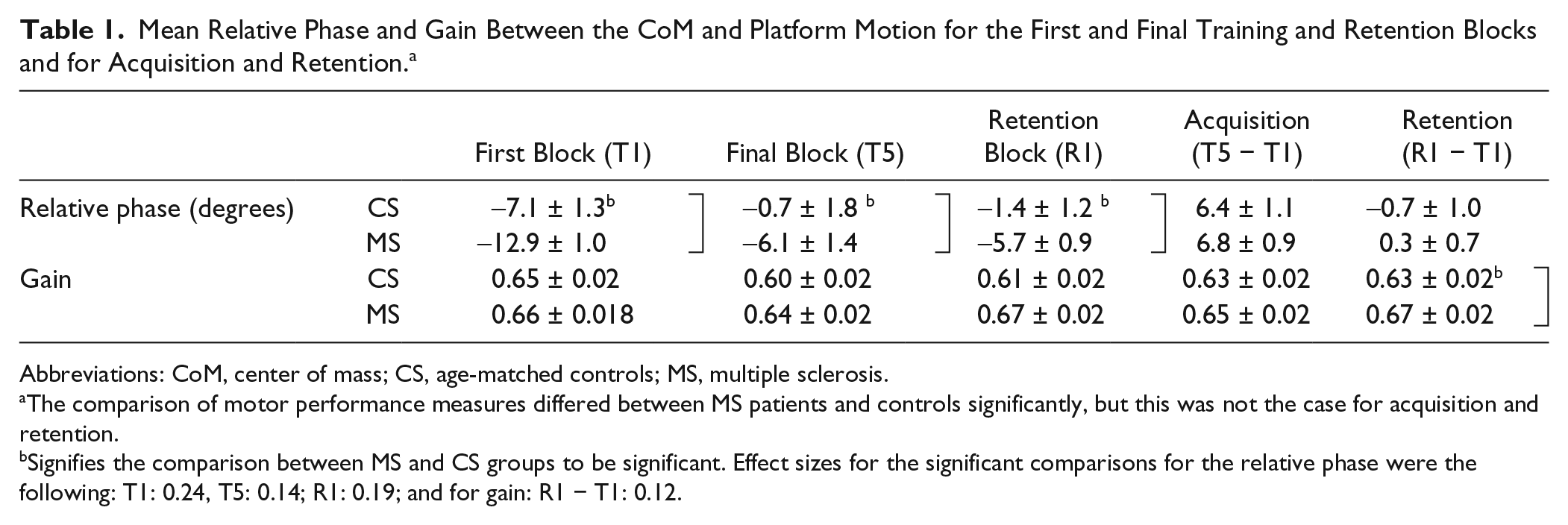
Abbreviations: CoM, center of mass; CS, age-matched controls; MS, multiple sclerosis.
The comparison of motor performance measures differed between MS patients and controls significantly, but this was not the case for acquisition and retention.
Signifies the comparison between MS and CS groups to be significant. Effect sizes for the significant comparisons for the relative phase were the following: T1: 0.24, T5: 0.14; R1: 0.19; and for gain: R1 − T1: 0.12.
In contrast to the mean relative phase measure, performance on the spatial measure of CoM control—that is, CoM gain—was similar between MS and control groups (main effect of group, P = .3, Figure 4, Table 1) irrespective of the testing block (2 groups × 5 blocks, P = .1). Thus, the magnitude of displacement of the body CoM with respect to the platform motion was similar for MS and control groups for the corresponding training blocks on day I. Similar to the mean relative phase measure, improvements in CoM gain (i.e., reduction in gain) with training were similar between the MS and control groups (acquisition, P = .13).
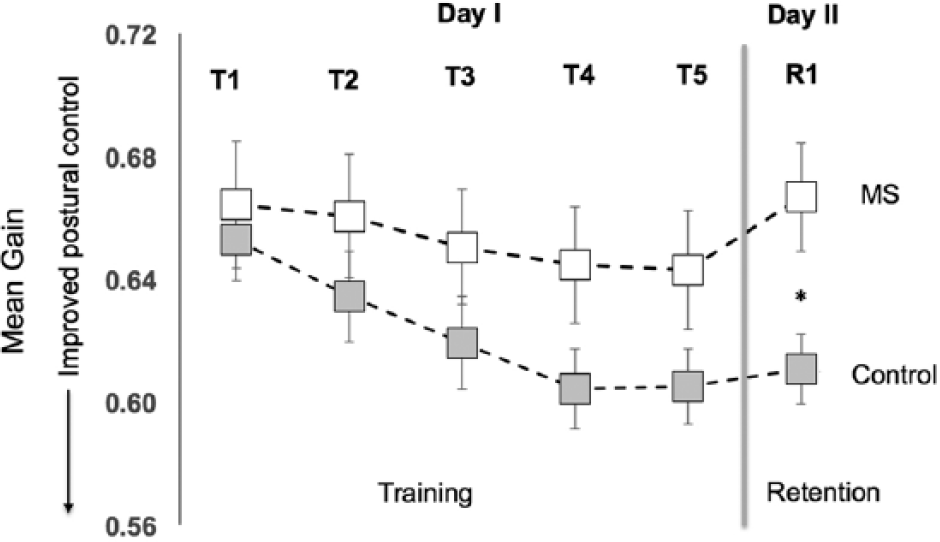
Group changes in the mean gain between the CoM and platform motion during training on day 1 (T1 − T5); retention (R1) testing on day II. To test the retention of the learned pattern, participants revisited the lab the next day after a 24-hour gap (vertical thick gray line). Lower values of the mean gain represent improved postural control. Error bars represent the standard error of the mean. No group differences were found across all training blocks, but the control group had significantly better retention on day II; *P < .05.
Postural Motor Learning
Participants retained the learned motor behavior as tested on the similar perturbation pattern after 24 hours for the relative phase measure (retention: F1,36 = 128.38, P < .01; Figure 3, Table 1), irrespective of the group (P = .22). However, retention of the CoM gain was different between groups (retention: F1,36 = 6.77, P < .05; Figure 4, Table 1). Controls were able to retain the gains in the CoM control, but this was not the case for the MS group (P < .05).
Relationship of Postural Motor Performance/Learning With Balance Deficits and Disease Severity
The MiniBESTest scores for the MS group (mean ± SD = 21 ± 4.8) were significantly lower than that for the control group (mean ± SD = 26 ± 1.8; F1,36 = 8.9; P < .01). The MiniBESTest of the MS group was significantly correlated at the baseline performance with the relative phase measure (r = 0.44; P < .05) but not so for the gain (r = 0.001; P = .99). MiniBESTest was not related to phase or gain for the extents of improvements on day I—that is, acquisition or retention on day II. Disease severity based on EDSS was not significantly related to the baseline performance, acquisition, and retention for either phase or gain measures.
Relationship Between Postural Motor Performance/Learning and Postural Sway
Statistical analysis revealed that the postural sway was greater for the MS group compared with the control group for both eyes open (F1,32 = 9.03; P < .01) and eyes closed (F1,32 = 8.06; P < .01) conditions. However, the Romberg quotient did not differ between groups (P = .84). Figure 1 shows an example of postural sway in an MS patient who did have a larger Romberg quotient than the representative control.
Relative Phase
The Romberg quotient was significantly correlated with postural motor performance in participants with MS at baseline—that is, first training block on day I (r = −0.47; P < .05). However, the Romberg quotient was not related to the extents of improvements on day I (acquisition, r = 0.03; P = .89) or retention on day II (r = −0.14; P = .56). Figure 5 illustrates representative MS participants who improved postural control with practice, although MS patient I (who had a low Romberg quotient) but not MS participant II (who had a high Romberg quotient) showed postural performance similar to the representative control. Unlike, the relative phase measure, there was no correlation between gain and Romberg quotient for motor performance at baseline—that is, first training block on day I (r = 0.35; P = .14). There was also no correlation between the Romberg quotient and extent of gain improvements on day I (acquisition: r = 0.33; P = .16) or retention on day II (retention: r = 0.32; P = .16).
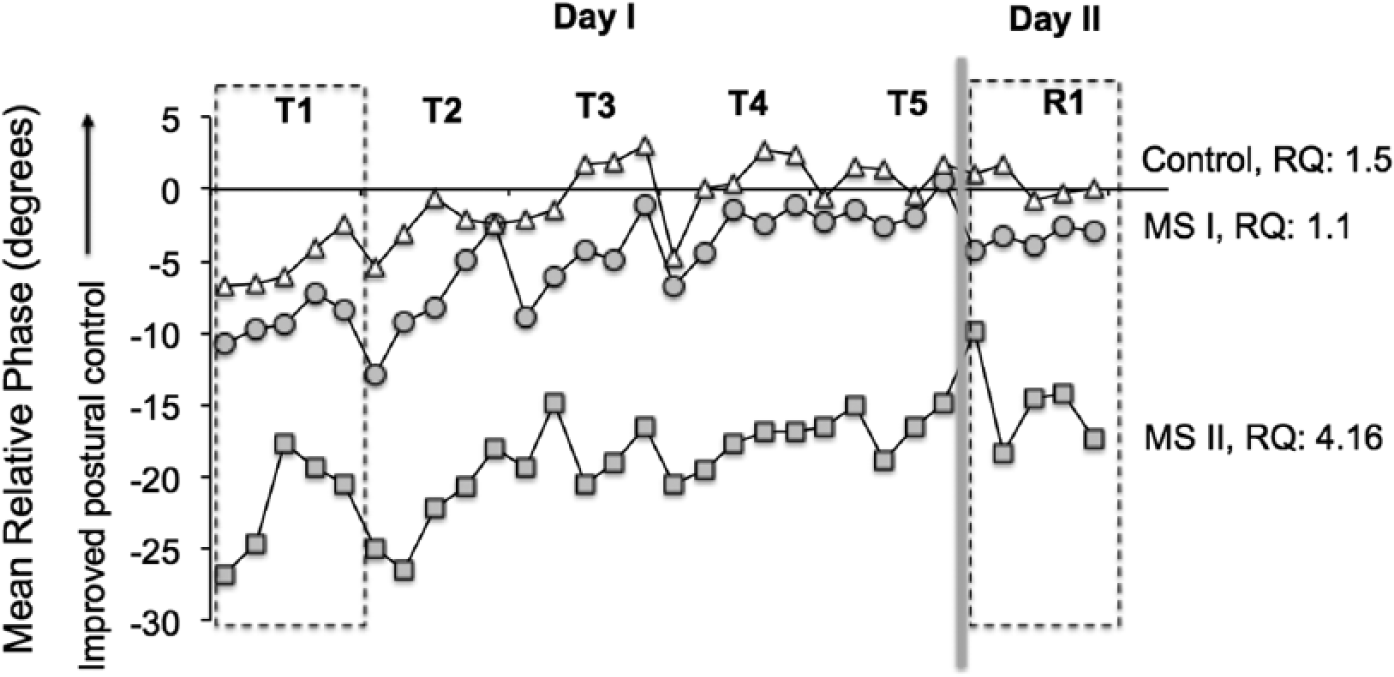
Mean relative phase between the CoM and platform motion for individual trials during training on day 1 (T1 − T5) and retention (R1) testing on day II. Data are presented for one of the controls and two MS patients. Although MS patients I and II differ in the motor performance at baseline depending on the Romberg quotient (RQ), both participants improve postural responses with training and are able to retain the learned responses. Training consisted of 5 blocks of surface perturbations, with each block comprising 5 repetitions. A rest break was provided after every training block. To test the retention of the learned pattern, participants revisited the lab the next day after a 24-hour gap. The retention phase comprised testing with the same perturbation pattern as that for training.
Discussion
To our knowledge, this is the first study to demonstrate that mildly affected MS patients have the ability to improve aspects of postural responses with training. People with MS exhibited preserved capacity, comparable with that of controls, to improve both the temporal and spatial aspects of postural control with practice. However, MS patients exhibited deficits in retaining these improvements only in the spatial domain when tested the next day.
Standing balance requires sensory integration of somatosensory, visual, and vestibular inputs, with somatosensory input being the primary contributor. 35 Although MS can affect any of the three sensory systems, eliminating the use of vision leads to markedly increased postural sway in patients with MS, which is consistent with proprioceptive and/or vestibular-based balance deficits.36-38 Balance deficits in people with MS have been shown to be associated with reduced proprioception and vibration loss.32,39,40 These somatosensory deficits have been associated with damage to the spinal cord32,39,40 and reduced microstructural integrity of the cortical proprioceptive pathway. 41 In the current study, we observed that people with MS were able to learn to improve/predict temporal control of postural responses (Figures 3 and 4). These results provide further evidence for preserved postural learning in people with MS despite deficits in postural performance.13,14,16,42,43
We speculate that the capacity for preserved postural motor learning in temporal control of CoM could be a result of an increased reliance on a feed-forward postural control system. Predictive, feed-forward control is developed with practice, whereby the system registers certain parameters about perturbation characteristics and/or body orientation (eg, constant frequency of platform motion). This feed-forward control then serves to improve stability by generating the postural response earlier by minimizing the reliance on reactive responses.44,45
Based on earlier studies, we know that participants do not learn the exact amplitude of perturbation sequences. 18 However, a CoM shift from phase lag toward lead suggests that frequency of the platform motion is utilized to control postural responses. We speculate that for MS participants, the delay in the timing of the CoM displacement with respect to the platform motion led to greater displacements in CoM. As mentioned earlier, given that the frequency of the platform motion was held constant, improvements in the temporal domain might reflect improvements in anticipatory control of postural responses. Thus, participants with MS learn to improve and retain predictive control of balance but do not retain the reduction in excessive CoM displacement, reflecting continued postural instability.
Preserved ability of patients with MS to predict upcoming surface perturbations, is corroborated by the findings from Cameron et al, 25 in which people with MS demonstrated the ability to scale their postural responses when exposed to predictable magnitudes of discrete surface perturbations. Although predictive scaling was intact, MS patients in Cameron et al 46 study showed impaired reactive scaling to unpredictable velocities of surface perturbations and had long latencies of postural responses that were related to their slowed spinal somatosensory conduction. In fact, the greater the deficit in the somatosensory conduction, the larger was the use of a predictive strategy to scale postural responses.
Postural responses to continuous perturbations require more continuous updating of motor commands than responses to discrete perturbations, so they are even more dependent on somatosensory inputs. 23 It is likely that compensation for MS-related proprioceptive deficit involves increased dependence on the visual or vestibular inputs to provide the error signal to the cerebellum in order to update future motor commands.2,36-38,41,47 Irrespective of the sensory systems used, people with MS demonstrated the ability to increase use of feed-forward strategies by predicting postural perturbations and, more important, retained the learned behavior.24,48,49 Thus, the fact that people with MS are able to use a predictive strategy to control postural responses suggests increased reliance on the corticocerebellar networks for the postural motor learning task. Alternatively, this preserved predictive strategy could be a manifestation of functional reorganization of motor networks, compensating for the deficits incurred in the primary structures responsible for reactive postural strategies.42,43,50
Preserved capacity of postural motor learning in people with MS could also be attributed to the implicit nature of the postural learning tasks. Motor learning studies in people with MS have shown greater improvements with implicit, compared with explicit, motor learning tasks in people with MS.11,13 This greater difficulty with explicit learning has been attributed to cognitive deficits related to cortical atrophy in people with MS.11,13
Rehabilitation Implications
The findings from this study suggest that people with MS have the ability to learn to anticipate external perturbations and thereby improve their postural responses. The ability to improve postural control with practice illustrates that postural responses can be adapted and learned similar to a skilled motor learning task. The ability to acquire and retain the changes in postural control was observed despite balance impairments and postural motor performance deficits observed at baseline. Thus, in mildly impaired patients with MS, exposure to external surface perturbations can be used to improve postural control.
It should, however, be noted that these findings pertain to continuous surface perturbations such as those encountered while standing on a boat or subway. Generalization of these findings to discrete perturbations, such as a slip or trip, requires further investigation of motor learning paradigms with discrete perturbations. Nonetheless, findings of this study suggest that mildly affected MS patients can be trained to anticipate postural perturbations by exposing them to external perturbations.
Limitations
The findings of this study should be cautiously interpreted because of the small sample size. Also, long-term training paradigms should be tested for better clinical applicability of the findings. In addition, the participants included in the study were minimally to mildly affected, which again limits the generalizability of the findings to more severely affected individuals.
Conclusions
This study provides evidence for preserved postural motor learning in people with MS, irrespective of the extent of somatosensory deficits and disease severity. More important, we demonstrate, for the first time, that people with MS have the ability to retain this learned postural control. This retained postural motor learning can be harnessed through physical therapy interventions to bring about desirable mobility changes and, more important, prevent nondesirable changes related to disuse.
Footnotes
Acknowledgements
We thank the volunteers for participating in this study. We are also grateful to Heather Schlueter and Jessica Nyugen for assistance in participant recruitment and data collection.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: This potential conflict of interest has been reviewed and managed by OHSU and the Integrity Program Oversight Council. OHSU and Dr Horak have a significant financial interest in APDM, a company that has a commercial interest in this research and technology. This potential conflict of interest has been reviewed and managed by OHSU and the Integrity Program Oversight Council
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was principally supported by grants from the National Multiple Sclerosis Society: FG 2058-A-1 (GG); RG 5273A1/T (BWF); and MB0011 (FBH). Additional support was provided by the Medical Research Foundation of Oregon (GG and BWF) and the NL Tartar Research Fund (BWF).
