Abstract
Background. Handwriting is often impaired in Parkinson’s disease (PD). Several studies have shown that writing in PD benefits from the use of cues. However, this was typically studied with writing and drawing sizes that are usually not used in daily life. Objective. This study examines the effect of visual cueing on a prewriting task at small amplitudes (≤1.0 cm) in PD patients and healthy controls to better understand the working action of cueing for writing. Methods. A total of 15 PD patients and 15 healthy, age-matched controls performed a prewriting task at 0.6 cm and 1.0 cm in the presence and absence of visual cues (target lines). Writing amplitude, variability of amplitude, and speed were chosen as dependent variables, measured using a newly developed touch-sensitive tablet. Results. Cueing led to immediate improvements in writing size, variability of writing size, and speed in both groups in the 1.0 cm condition. However, when writing at 0.6 cm with cues, a decrease in writing size was apparent in both groups (P < .001) and the difference in variability of amplitude between cued and uncued writing disappeared. In addition, the writing speed of controls decreased when the cue was present. Conclusions. Visual target lines of 1.0 cm improved the writing of sequential loops in contrast to lines spaced at 0.6 cm. These results illustrate that, unlike for gait, visual cueing for fine-motor tasks requires a differentiated approach, taking into account the possible increases of accuracy constraints imposed by cueing.
Introduction
The basal ganglia play an important role during self-generated and well-learned movements.1-3 Because handwriting is an internally generated and habitual movement, basal ganglia dysfunction inevitably leads to handwriting problems. In the past, handwriting difficulties in Parkinson’s disease (PD) were often referred to as micrographia—that is, a progressive reduction in amplitude during writing. 4 However, writing problems in PD do not only include an amplitude scaling deficit, but also timing deficits, irregularities, and breakdown of movement.5-8 Initially, writing problems seem to respond well to dopaminergic medication. However, improvements resulting from medication are mainly found for movement speed and, to a lesser extent, for writing size.5,7 Writing faster is often associated with a decreased legibility. 9 With disease progression, writing performance may further deteriorate. Therefore, complementary strategies are necessary to maintain or improve writing amplitude in PD.
One possible approach is offering external cueing. Cues provide discrete external information, serving as a reference, target, or trigger for movement generation. 10 Because patients with PD suffer from difficulties with automatic movement control, cueing was proposed to induce a transition from habitual behavior to a goal-directed mode of motor control. 11 In gait, it was shown that cueing has beneficial effects for patients with PD (for reviews, see Rocha et al 12 and Spaulding et al 13 ). So far, only few studies have investigated the effects of cueing on fine-motor skills, usually during more visually controlled tasks than gait (for a review, see Nackaerts et al 14 ). Both bimanual drawing and writing studies showed that PD patients improve their writing or drawing size in the presence of visual cues.15-18 However, these studies included writing and drawing sizes of 1.0 cm and larger, whereas older adults generally write a self-generated text at a size of, on average, 2.4 mm. 19 PD is increasingly prevalent in people who are still working, 20 and despite the intensive use of computers, handwriting is still necessary in many professions. Though visual cueing was shown to be a valuable tool for writing with a larger amplitude and is recommended as a rehabilitation strategy in PD, 21 techniques that improve daily-life writing sizes should be further investigated, as suggested by Bryant et al. 15 Therefore, we wanted to study the effect of visual cues on 2 different sizes during a prewriting task. We hypothesized that the therapeutic window of cueing may be smaller in PD for movements in the small amplitude range 22 because a recent study showed that patients with PD experienced more problems when writing at 0.6 cm compared with writing at 1.0 cm during dual-task writing. 23 Furthermore, we wanted to investigate whether cueing for different amplitudes has a different effect in PD patients and healthy controls. In line with earlier work, we hypothesize that cueing will lead to a reduction in amplitude of loops at large compared with small amplitudes in healthy controls because they tend to overshoot when writing large in the absence of cues. 16
Materials and Methods
Participants
For this cross-sectional study, 30 right-handed participants were tested, including 15 PD patients and 15 healthy, age-matched controls. Patients with PD were recruited by the neurologist (WV) of the Movement Disorders Clinic at the University Hospital Leuven. Inclusion criteria for PD patients were the following: (1) diagnosis of PD according to the United Kingdom PD Society Brain Bank criteria 24 ; (2) Hoehn and Yahr (H&Y) stage I to III in the on phase of the medication cycle 25 ; (3) writing problems, as identified by a score of 1 or more on item 2.7 (handwriting) of the Movement Disorder Society–sponsored revision of the Unified Parkinson’s Disease Rating Scale (MDS-UPDRS) part II; and (4) no cognitive impairment (Mini-Mental State Examination [MMSE] ≥24). The exclusion criteria for both patients and healthy controls were the following: (1) color blindness or other impairments in vision interfering with handwriting and (2) upper-limb problems other than those related to PD. All patients were tested in the on phase of the medication cycle—that is, approximately 1 hour after the last drug intake. For 11 patients, this was in the morning, whereas 4 patients were tested in the afternoon. The study design and protocol were approved by the local Ethics Committee of the KU Leuven and were in accordance with the code of Ethics of the World Medical Association (Declaration of Helsinki, 1967). After complete explanation of the study protocol, written informed consent was obtained from all participants prior to participation in the study.
Experimental Procedure and Tasks
Before performing the prewriting tests, all participants completed a clinical test battery incorporating overall cognitive status (MMSE 26 ), fine motor skills (Purdue Pegboard Test 27 and Manual Ability Measure [MAM-16] 28 ), and emotional behavior (Hospital Anxiety and Depression Scale [HADS] 29 ). In addition, disease-specific characteristics were assessed by means of the (1) MDS-UPDRS part III 30 and (2) H&Y scale. 25 In addition, participants were asked to perform a writing test in which they had to copy a standardized text for 5 minutes on paper (Systematic Screening of Handwriting Difficulties [SOS test]23,31).
Next, participants performed a simple repetitive prewriting task that allowed assessment of pure writing performance. The same 3-loop sequence was chosen as previously described by Broeder et al. 23 This task offers the advantage that it is a fluent writing-like movement, excluding long left-to-right letter sequences, which would require a shift of the hand over the surface, and avoiding the involvement of language and higher cognitive demands. This approach ensured optimal experimental control while still representing a writing-like movement. The prewriting sequence was performed on a custom-made touch-sensitive writing tablet (Figure 1A) and was performed at 2 different sizes—0.6 and 1.0 cm (Figures 1B and 1C, respectively)—in 2 cueing conditions, consisting of visual target zones. The requested writing amplitude was indicated on the tablet by colored target zones (gray, yellow, and blue) with a bandwidth of 2 mm. For both sizes, participants were instructed to start writing within the start circle and write the loops from the bottom of the blue target zone to the top of the yellow target zone and, then, returning to the bottom of the blue zone. The distance between the bottom of the blue and top of the yellow target zone was 0.6 or 1.0 cm. After completion of the third loop, participants were instructed to return to the start circle via the gray target zone to avoid left-to-right sequence progression. Each loop disappeared from the screen at the end of the loop figure. This allowed continuous repetition of the same figure without hand repositioning movements during the whole trial of 27 s. In the without cue condition, the target zones were initially presented to indicate the requested amplitude but disappeared after 2 s. Before each task, participants performed 1 practice trial on paper and 1 on the writing tablet to become accustomed to writing on the tablet and sudden disappearance of the cue. In addition, participants were instructed to produce natural and fluent loops and write at a comfortable speed.
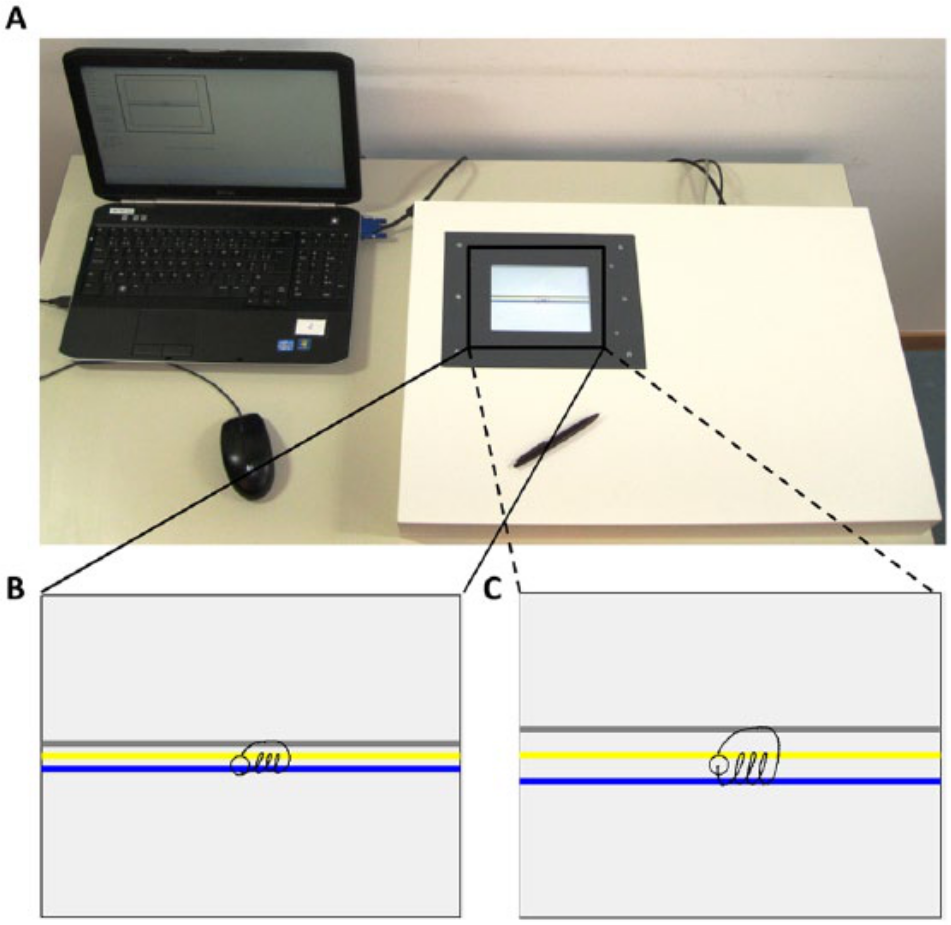
Experimental material and writing tasks: A. The newly developed writing tablet connected with a laptop. B. Writing task at 0.6 cm—between the bottom of the blue (lower) and top of the yellow (middle) target zone. C. Writing task at 1.0 cm—between the bottom of the blue (lower) and top of the yellow (middle) target zone.
Data Processing and Statistical Analysis
All data were filtered at 7 Hz with a fourth-order Butterworth filter 8 and further processed using Matlab R2011b. Amplitude (percentage of target size) and speed (cm/s) were chosen as dependent variables based on previous studies on handwriting.5,17,32 The dependent variables were determined using the same process as previously described in the study by Broeder et al. 23 In short, amplitudes of individual upstrokes and downstrokes were defined by calculating local minima and maxima. To compare the different sizes, the participants’ amplitude was expressed as a percentage of the target amplitude (percentage of target size) to make the data comparable. For each upstroke and downstroke, the time to complete (s) was computed and used to calculate speed (cm/s). In addition to these variables, the within-patient coefficient of variation of writing amplitude was also analyzed (COVampl).
Data processing of the SOS test was performed manually by a blinded researcher. The total SOS score measures the handwriting quality and is calculated by evaluating the following criteria: (1) fluency of letter formation, (2) fluency in connections between letters, (3) regularity of letter height, (4) space between words, and (5) straightness of the sentences. 31 A higher total SOS score indicates worse quality of handwriting. Mean writing size (mm) and writing velocity (number of letters written in 5 minutes) were also determined.
Statistical analysis was performed using Statistica software (version 10). All data were checked for normality and equality of variances, and appropriate parametric or nonparametric analyses were performed. To compare differences in demographic characteristics between PD patients and healthy controls Mann-Whitney U tests were used. Amplitude, writing speed, and COVampl were analyzed using a 2 × 2 × 2 repeated-measures analysis of variance (ANOVA), with group (PD and CT) as a between-subjects factor and cue (with and without cue) and size (0.6 and 1.0 cm) as within-subjects factors. Significant interactions were further investigated using the Tukey Honest Significance test as a post hoc analysis method. Finally, correlations between writing on the tablet and writing on paper were calculated using Spearman rank tests. Significance levels for all tests were set at P < .05.
Results
Participants
Demographics and clinical characteristics of the participants are specified in Table 1. Patients and healthy controls did not differ significantly, except for measures of fine motor skills (MAM-16 and Purdue Pegboard Tests, P < .001) and the depression subscale of the HADS (P = .029). Patients also wrote smaller (P = .056) and slower (P < .001), as compared with controls, on the SOS test.
Participant Characteristics; Median, and Interquartile Range (25%-75%).
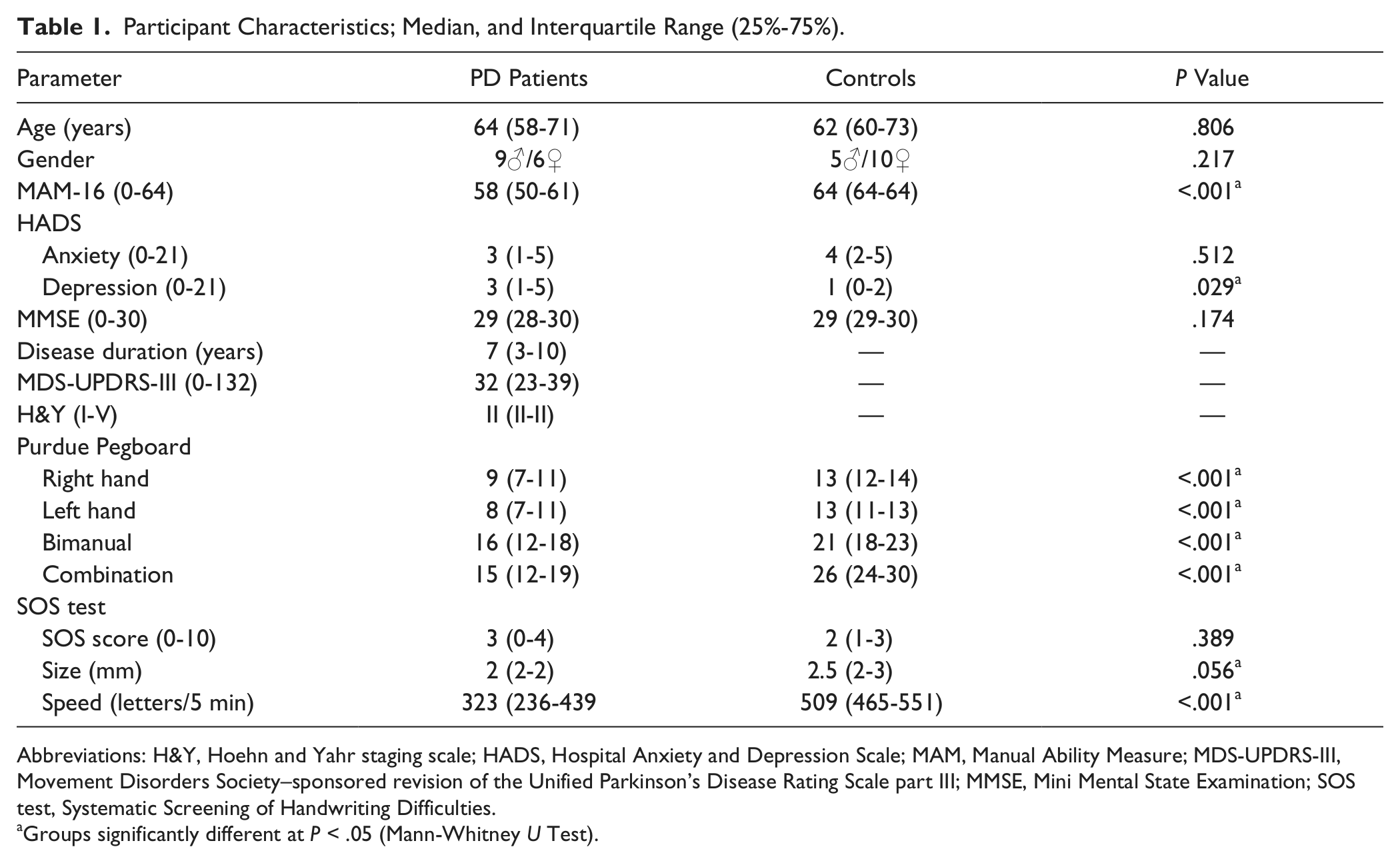
Abbreviations: H&Y, Hoehn and Yahr staging scale; HADS, Hospital Anxiety and Depression Scale; MAM, Manual Ability Measure; MDS-UPDRS-III, Movement Disorders Society–sponsored revision of the Unified Parkinson’s Disease Rating Scale part III; MMSE, Mini Mental State Examination; SOS test, Systematic Screening of Handwriting Difficulties.
Groups significantly different at P < .05 (Mann-Whitney U Test).
Writing Amplitude
A 2 × 2 × 2 ANOVA (Group × Size × Cue) showed a significant interaction between size and cue: F(1, 28) = 65.5, P < .001; Figure 2A. Post hoc analysis revealed that writing in the 0.6-cm condition induced a relatively larger amplitude (with cue, 80% ± 11%; without cue, 86% ± 15%) than in the 1.0-cm condition (with cue, 72% ± 11%; without cue, 65% ± 11%) for both writing with and without cues (both P < .001). In the 0.6-cm condition, both groups wrote with a larger amplitude in the absence of cues compared with the presence of cues (P < .001). In the 1.0-cm condition, the opposite was true: participants wrote larger with cues compared with without (P < .001) irrespective of group. In addition, a significant main effect of group showed that patients with PD wrote significantly smaller overall than healthy controls (F(1, 28) = 9.5; P = .005).
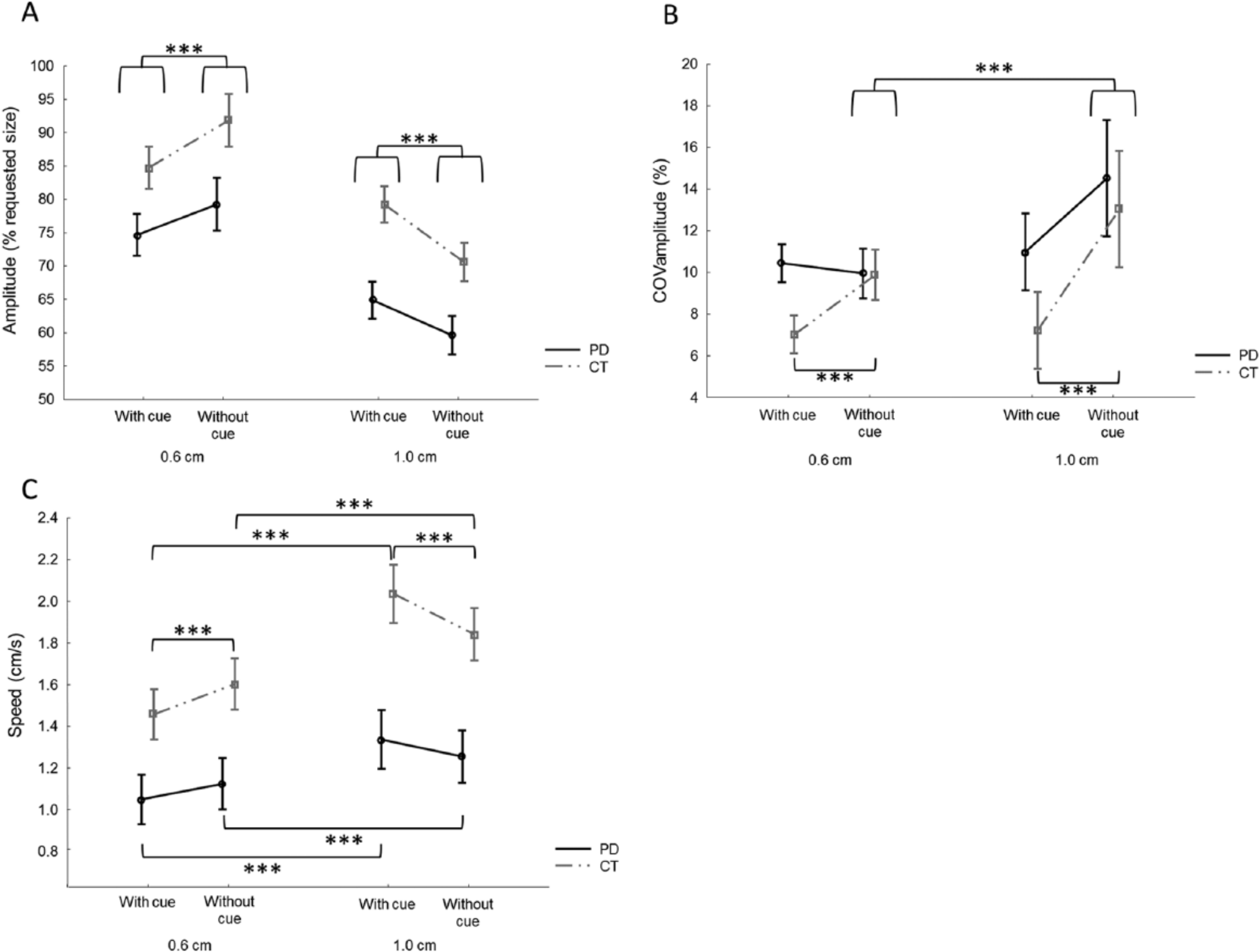
Performance differences between writing with and without cue. A. For amplitude, a significant interaction was found between size and cue (P < .001). Post hoc analysis showed that both patients with Parkinson’s disease (PD) and controls (CT) wrote larger without cues in the 0.6-cm condition, whereas in the l.0-cm condition, this was the opposite. B. For variability of amplitude (COVamplitude), an interaction between group and cue (P = .044) was found, showing that CTs wrote with higher variability during writing without cues in both size conditions. In addition, an interaction between size and cue was found (P = .012), indicating higher variability in the l.0-cm condition compared with the 0.6-cm condition during writing without cue. C. For speed, a significant interaction was found between size, cue, and group (P = .003), showing that both groups wrote faster in the l.0-cm condition compared with the 0.6-cm condition, both during writing with and without cues. CTs also wrote faster without cues in the 0.6-cm condition and slower without cues in the l.0-cm condition.
Variability of Amplitude (COVampl)
For COVampl the 2 × 2 × 2 ANOVA (Group × Size × Cue) revealed 2 significant interactions: between cue and group (F(1, 28) = 4.5; P = .044) and between size and cue (F(1, 28) = 7.2; P = .012). For the first interaction (Cue × Group), the observation was made that PD patients overall had a higher variability; however, this was not significantly different. Post hoc analysis showed that only in healthy controls, there was a significantly higher variability when writing without cues compared to with cues (P < .001; Figure 2B). This was not the case for patients with PD (P = .384). Post hoc analysis of the Size × Cue interaction showed that there was no difference in variability between both size conditions while writing with cues (P = .976; Figure 2B). When writing without cues, there was a higher variability in the 1.0-cm condition compared with the 0.6-cm condition (P = .001). In addition, a significantly higher variability was found when writing without cues compared to with cues (P < .001) in the 1.0-cm condition, whereas there was no difference in variability between cueing conditions in the 0.6-cm condition (P = .572).
Writing Speed
For writing speed, a significant 2 × 2 × 2 interaction was found (F(1, 28) = 10.8; P = .003). Post hoc analysis showed that both groups wrote faster in the 1.0-cm condition compared with the 0.6-cm condition, both while writing with and without cues (all P < .001; Figure 2C). Furthermore, analysis revealed that healthy controls wrote significantly faster without cues in the 0.6-cm condition (P < .001), whereas in the 1.0-cm condition, they wrote faster in the presence of cues (P < .001). No significant differences were found for patients with PD.
Correlation Analysis
The Spearman Rank test showed a high correlation between the total SOS score (quality) and writing of loops at 1.0 cm without cues (R = −0.514; P < .05) and weaker correlation for 0.6 cm without cues (R = −0.306; P = .100). Both analyses showed that a better handwriting quality on paper was correlated with writing larger loops on the tablet. In addition, strong correlations were found between writing speed on the tablet and on paper (0.6 cm with cue: R = 0.584; 0.6 cm without cue: R = 0.650; 1.0 cm with cue: R = 0.613; 1.0 cm without cue: R = 0.675; all P < .001), indicating that writing faster on paper was associated with writing faster on the tablet.
Discussion
In this study, it was investigated for the first time whether there was a different response to cueing during a prewriting task at 2 sizes comparable to the ones used in daily life—that is, 0.6 and 1.0 cm—in patients with PD and healthy controls. As anticipated and consistent with previous literature,15-18 patients displayed micrographia compared with controls in all conditions, as well as during the SOS paper-and-pen test, confirming the validity of the experimental paradigm. The results for writing at 1.0 cm showed immediate improvements in writing size, variability of writing size, and speed in the presence of cues in both groups. In contrast, the cue seemed to hinder writing performance in the 0.6 cm condition: writing was smaller and there was no difference in variability compared with uncued writing in both groups. In addition, the writing speed of healthy controls decreased when the cue was present.
Interfering Effect of Cueing in the Small Writing Condition
During externally triggered movements, the dorsolateral neural network, consisting of parietal and premotor cortices and cerebellum, has been found to be more active compared with when the same movements are internally controlled in healthy young and older adults.1,3,33 This network may bypass the corticobasal ganglia pathway, which is dysfunctional in PD, as was seen for the prewriting task at 1.0 cm. However, our results suggest that visual lines may be perceived differently during writing at the small 0.6 cm size. At 0.6 cm, the target zones seemed to create an additional accuracy constraint, making the task more complex than the self-generated version. This is in line with a study on forearm movements, which demonstrated size-dependent feedback effects in PD. 34 Fast and small movements were performed better when no visual feedback was available. Conversely, slow and large movements improved in the presence of visual feedback. In addition, several studies reported that fast and small movements, both during gait and upper-limb movements, make patients with PD more susceptible to additional difficulties, such as freezing episodes or hastening.16,35-39 Therefore, a potential explanation for the fact that negative effects of cueing are more pronounced at small amplitudes is that these conditions could have led to more disordered motor control, at least in patients with PD.
In gait it was already shown that when task demands increase, the attentional resources may become overloaded, causing gait abnormalities. 40 Gait in PD, and even more so under complex conditions, may no longer be considered as an automated activity, requiring executive functioning and attention.41,42 Although handwriting is very different from gait, it is not implausible that the cues added task complexity during writing at the small size. It has been proposed that handwriting requires executive functioning and attention. 43 As in gait, the capacity to improve writing may, thus, depend on the ability to use attention for compensation, 35 which in turn depends on the individual’s cognitive reserve. This cognitive reserve model suggests that the brain actively recruits preexisting cognitive or other compensatory processes to substitute for ageing or pathology (for a review see Stern 44 ). However, when cognitive reserve wears out, impairments will become noticeable. Applying this knowledge to the current prewriting paradigm, the task may have led to the activation of primarily writing-specific brain regions—that is, superior and middle frontal area, superior parietal area, and cerebellum—in addition to non–writing-specific motor regions.45,46 The additional attention drawn to the writing process by means of visual cueing probably implied less automatic performance, triggering additional activity in the parietal and premotor cortices and cerebellum, drawing on the cognitive reserve. Whereas this led to improved performance in the 1.0-cm condition, the greater accuracy constraints imposed by the visual cues in the small prewriting task added extra stress to these compensatory mechanisms. As a result, we speculate that limitations in neural resources were reached earlier, explaining worse performance in the 0.6-cm condition in the presence of visual cues.
It is also possible that the smaller-sized loops required more visual control regions and more motor areas, in line with greater motor precision demands 47 or increased task complexity.48,49 The greater involvement of these regions may have precluded the efficient allocation of the dorsolateral system, as is normally the case during externally triggered movements, thereby leading to compromised performance during writing of small loops.
Similar Response of Patients and Healthy Controls
Contrary to our hypothesis, the response of both groups to the different cueing conditions was overall similar. Whereas other studies made strict comparisons between writing or drawing with cues compared with spontaneous movements,16,17 in the present study, every trial started with a short display of visual lines, which disappeared in the uncued condition after 2 s. This may have provided an internal reference of the desired amplitude and as such prevented overshooting in controls and, thus, reduced the relative effect of cueing. Similar to previous studies regarding handwriting and aiming movements and consistent with Fitts’ law,50-52 both patients and controls in this study wrote with greater accuracy (percentage of desired amplitude) in the small compared with the large condition, and this was accompanied by slower writing. This speed-accuracy trade-off was observed both in the presence and absence of cueing, supporting the hypothesis that an internal reference of the desired amplitude may have been formed in both groups.
Another explanation for the few between-group effects is that controls and patients were age matched, and age-related changes in handwriting explained the similar responses in controls. 19 It has been reported that both ageing and PD can lead to executive dysfunction and reduced cognitive reserve.44,53-55 Because intact executive functioning is important for handwriting, healthy older controls may also have responded to cueing in a similar way. In support of this hypothesis, it was shown previously that the presence of external cues led to less-efficient writing movements in healthy elderly compared with young adults. 56 More recently, Vitorio et al 57 showed that in gait, PD patients and healthy controls used the same strategy to capture visual information from visual cues. 57
Although PD patients and controls showed comparable behavior during both internally and externally triggered movements in a computerized choice reaction time task, 56 there were great differences at the neural level. 57 Results showed increased functional connectivity between the dorsolateral prefrontal cortex (PFC) and lateral premotor cortex (PMC) in patients compared with controls only in the externally guided condition. These results provided a strong indication for a compensatory role of the PFC-PMC connection, which allowed patients with PD to maintain task performance at a level similar to that of healthy elderly individuals during external cueing. Further research is warranted to determine whether this also applies for a daily-life repetitive activity such as handwriting.
Implications for Neurorehabilitation
The current results have important implications for neurorehabilitation aimed at designing the most optimal learning environment for people with a neurological disorder. Visual cueing was shown to be a very effective method to optimize gait (for review see Rocha et al 12 and Spaulding et al 13 ). We found that visual cueing can be used as a strategy to improve writing of loops at sizes that are larger than the average writing size in PD. However, we also demonstrated that cueing is not an optimal strategy for producing smaller loops, in line with daily-life writing. The current results illustrate that cueing needs to be administered with insight into how it changes the task demands of fine-motor skills. Hence, cueing effects are task dependent in PD. As novel technology is invading the field of neurorehabilitation, the development of digitized pens and tablets holds promise for writing training. 58 Such devices are able to provide cueing or feedback when writing quality starts to deteriorate. Also, intermittent cueing or performance feedback may stimulate the formation of an internal representation of the motor task and, thus, stimulate consolidation of learning. The current results contribute to the development of such technological devices because it was shown that cueing should not be delivered in a one-fits-all-fashion but rather be offered in a differentiated way. Our results suggest further that for letter and loop training of larger sizes, cueing has immediate advantageous effects, which could not be replicated for small sizes. However, long-term training studies should be performed to investigate whether intensive training with visual cues leads to retention and transfer to daily-life performance.
Conclusion
In conclusion, we found that while the effects of cueing on writing of loops at 1.0 cm were positive, this was not the case for writing at the smaller amplitudes of 0.6 cm. We attribute these findings to the additional cognitive load imposed by having to write in a narrow space. These context-dependent effects of cues have an important impact on the rehabilitation of writing skills in PD and illustrate that cueing needs to be administered with care. As a cue may induce a shift from automatic to more controlled processing, requiring more (pre)frontal involvement, ultimately a cue may encourage deautomaticity. Future neuroimaging research should focus on the underlying mechanisms of cueing during writing, both at small and large amplitudes. Our results confirm the use of visual cueing as a rehabilitation strategy when writing large-sized letters. In addition, alternative cues or feedback strategies using a sensory pen 58 may be valuable techniques for writing small-sized letters. Finally, the results of this study illustrate that further knowledge on cueing and feedback strategies in different motor effectors is needed to understand how patients with PD most optimally adapt to their loss of motor control.
Footnotes
Acknowledgements
We thank all participants of this study. We thank Ir. Marc Beirinckx for development of the tablet and for providing technical support.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Research Foundation, Flanders (FWO), Grant Number G.0906.11. EN is a research assistant and EH a postdoctoral researcher at the Research Foundation Flanders (FWO). WV is a senior clinical investigator of the FWO.
