Abstract
Introduction
Motor imagery (MI), defined as making a mental representation of a movement without executing the movement physically, may play a role in the rehabilitation of patients with neurological disorders.1,2 It was, for example, mentioned as a promising intervention for improving upper limb movements in stroke patients.
3
However, in patients with Parkinson disease (PD), studies remain sparse. This technique may be difficult to apply because of reported impairments in MI ability in PD patients.4-6 Although PD patients in the early and middle stages of the disease were able to accurately imagine movements, they showed profound slowness during MI.7,8 In healthy persons, imagined movement speed affects subsequent motor performance.
9
Therefore, to gain the most from MI training, patients must develop correct image characteristics of the duration of movements. This fits well within the mental simulation theory posed by Jeannerod,
10
which states that correct MI should be endowed with the same properties as those of the corresponding covert motor representations. The similarity in duration between imagery and execution, often referred to as
In physical practice, external cueing is known to provide an efficient way to (partly) overcome bradykinesia in PD.18,19 Cueing is defined as the use of external temporal or spatial stimuli to facilitate initiation and continuation of movement. 18 By providing external cues, the basal ganglia, which are the focal point of impairment in PD, 20 can be bypassed and compensatory networks activated, 21 leading to significant gains in performance.18,19,22 Although this strategy is widely accepted as a training tool in gait rehabilitation, it has not been employed to optimize MI performance in patients with PD. Yet the large amount of evidence showing that MI and physical execution have various characteristics in common23-26 suggests that strategies facilitating physical training might also be valid for MI practice. Previously, we successfully combined external cueing and MI in healthy participants. 27 Cues significantly enhanced participants’ MI accuracy, speed, and vividness. In the present study, we investigated whether cues also positively affect MI performance in PD patients. We hypothesized that external cues offering a temporal or spatial reference frame would decrease patients’ bradykinesia during MI and thus increase the MI quality.
Methods
Participants
We included 14 PD patients (9 males; 59.1 ± 9.6 years) and 14 age-matched healthy control subjects (8 males; 61.1 ± 6.6 years). Participants were right handed as measured by the Edinburgh Handedness Inventory Questionnaire. 28 Patients were diagnosed by a neurologist using the Brain Bank criteria 29 and were all in Hoehn and Yahr (H&Y) 30 stages 1 to 3. Tests were done while patients were “on” medication. Their motor status was assessed using the Unified Parkinson’s Disease Rating Scale 31 part III, their cognitive status was assessed using the Mini-Mental State Examination 32 and the Scales for Outcomes in Parkinson’s Disease–Cognition 33 (Table 1), and their eye movements were assessed by means of the International Cooperative Ataxia Rating Scale (items 17-19). 34 Exclusion criteria were Mini-Mental State Examination score <24, severe tremor, neurological comorbidity, unpredictable motor fluctuations, eye movement abnormalities, severe orthopedic problems of the upper limb, and treatment with deep brain stimulation. The study was conducted in accordance with the 1964 Declaration of Helsinki and approved by the ethical committee of the Katholieke Universiteit Leuven. Participants gave written informed consent prior to participation.
Clinical Characteristics of the Parkinson Patients
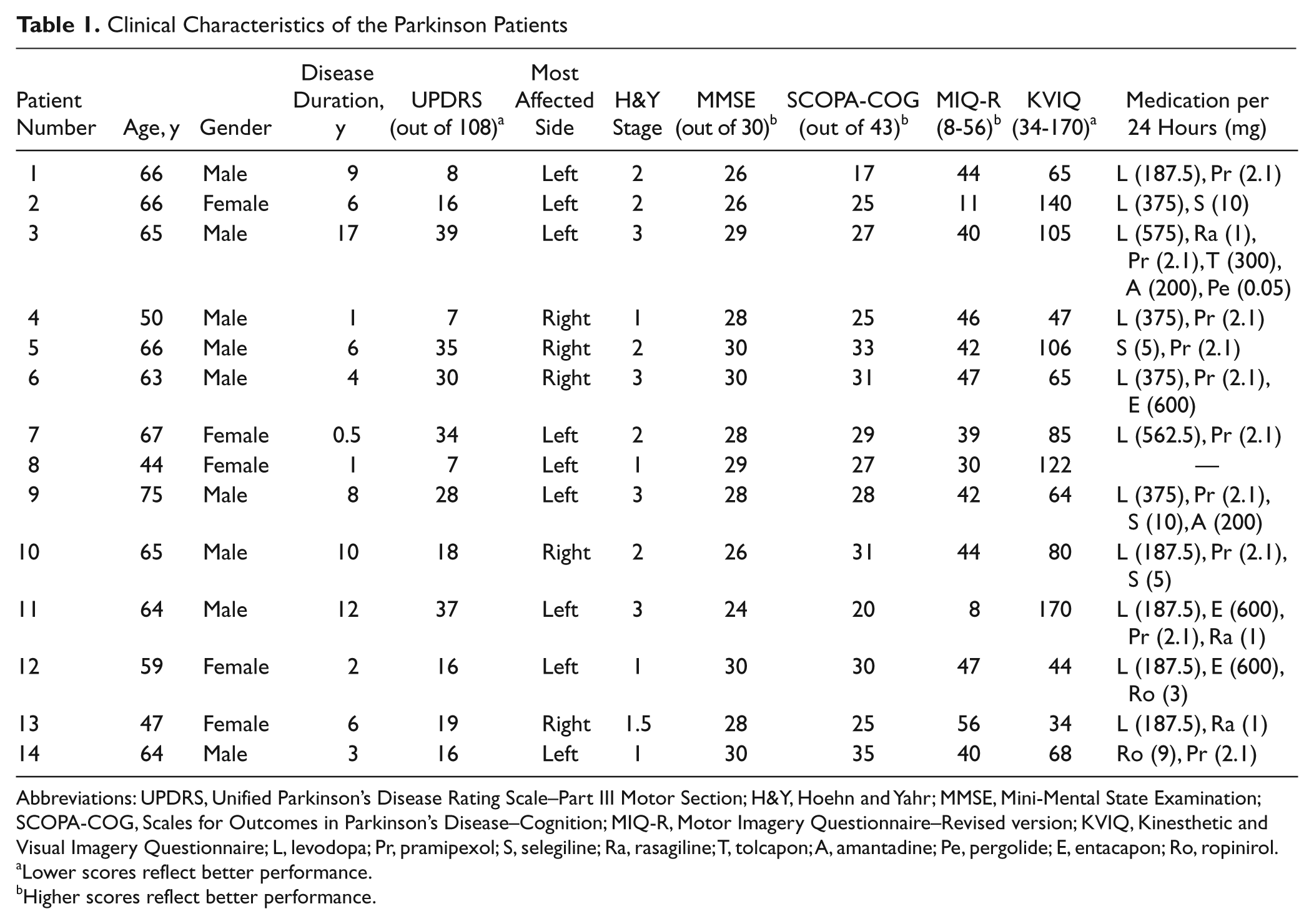
Abbreviations: UPDRS, Unified Parkinson’s Disease Rating Scale–Part III Motor Section; H&Y, Hoehn and Yahr; MMSE, Mini-Mental State Examination; SCOPA-COG, Scales for Outcomes in Parkinson’s Disease–Cognition; MIQ-R, Motor Imagery Questionnaire–Revised version; KVIQ, Kinesthetic and Visual Imagery Questionnaire; L, levodopa; Pr, pramipexol; S, selegiline; Ra, rasagiline; T, tolcapon; A, amantadine; Pe, pergolide; E, entacapon; Ro, ropinirol.
Lower scores reflect better performance.
Higher scores reflect better performance.
Experimental Procedure
The participants’ imagery ability was evaluated in detail in a separate session. An assessment battery was used consisting of the Chaotic Motor Imagery Assessment (CMIA),35,36 Motor Imagery Questionnaire–Revised version (MIQ-R), 37 Kinesthetic and Visual Imagery Questionnaire (KVIQ), 38 and a test using mental chronometry.26,39 The CMIA 35 was previously used in stroke patients to screen for chaotic MI, which can be defined as an inability to perform MI accurately or if having preserved accuracy, demonstrating temporal uncoupling.35,36 The MIQ-R37,40 was shown to be reliable to assess MI ability in healthy persons. 41 The KVIQ was especially developed for assessing MI ability in patients with restricted mobility and was shown to be reliable for healthy persons, stroke patients, and patients with PD.38,42 The final test of the assessment battery was based on a mental chronometry paradigm.26,39 Based on this assessment, we excluded 1 patient because of a lack of imagery ability (Table 1, patient 11), as well as his age-matched control. This patient had a score that deviated more than 2 standard deviations from the mean on both questionnaires and, he was not able to perform the other tests of the assessment battery. A more detailed description of the MI ability assessment can be found in Heremans et al. 8 The remaining 13 patients and controls performed a second session in which they physically executed and visually imagined (first person perspective) 2 upper limb tasks. The goal-directed aiming task (GDAT) consists of cyclical flexion and extension movements of the wrist. It is a fully experimentally controlled task, which was shown to be useful to investigate MI in previous studies.27,43-45 The Box-and-Block Test (BBT) is a functional and clinically applicable task, which requires participants to pick up and transfer blocks. 46 The participants were instructed to use visual MI, since we previously found that both patients and controls performed visual MI more vividly than kinesthetic MI. 8 Patients performed the tasks with their most affected side, and controls did it side-matched to avoid interference of hand dominance. As such, we assured that the number of patients and controls who performed the test with their preferred and nonpreferred hands was equal. The session lasted approximately 2 hours.
Tasks
Goal-directed aiming task
Participants were seated wearing a wrist–hand orthosis. At both sides of the computer screen in front of them, 1 cm2 black squares were projected at intertarget distances of either 12 cm (SMALL intertarget distance) or 20 cm (LARGE intertarget distance). The use of 2 different distances allowed us to monitor whether participants’ eye movements adapted to the task requirements during MI. This was previously shown to be a control parameter for accurate MI.27,43 The wrist position was displayed on the screen as a 2-cm-diameter round cursor. The task was performed for 3 modalities: (
Box and Block Test 46
The participants performed an adapted version of the BBT, consisting of wooden blocks of 2.5 cm2 and a box, divided in 2 equal parts by a 18-cm-high partition. They were instructed to transport 20 blocks as fast as possible from one side of the box to the other. This task was performed under 4 conditions: (
Outcome Measures
During the tasks, we monitored the subjects’ performance by means of 3 assessment methods: (
Electrooculography/electromyography
For both tasks, EMG activity of the forearm was monitored. If muscular activity was increased during MI, the trial was discarded. Participants’ eye movements were recorded by means of EOG. 43 For EOG/EMG recording, a Porti 7 device (TMSi, Enschede, the Netherlands) with a sample frequency of 1024 Hz was used. After careful skin preparation, 5-mm-diameter Ag–AgCl surface electrodes were placed around the right eye and 24-mm-diameter electrodes on the extensor and flexor carpi radialis muscles of the tested arm and on the left pelvic bone as a reference electrode. A chinrest was used to restrict head movements. In line with Heremans et al,27,43 a low-pass filter (cutoff frequency 20 Hz) was used to filter the eye movement data and drift of the signal was corrected by a piecewise second-order polynomial fit. Fixations of gaze were defined as stable gaze positions (standard deviation of point of gaze <1° in the last 100 ms) that were maintained for at least 100 ms. 47 The data points at the end and start of each fixation were taken as start and end points of the eye movements, which could consist of 1 single primary saccade or a combination of a primary saccade and 1 or more corrective saccades. The eye movement time was defined as the time between the end point of the previous fixation and start point of the next fixation. Furthermore, we calculated the eye movement amplitude, corresponding with the distance traveled by the eyes between those 2 points, as well as the number of eye movements per trial.
Mental chronometry
For the BBT, the duration of each executed and imagined trial was measured by means of a chronometer. When using this method, a close match between the duration of execution and imagery is supposed to be an indicator of good MI quality.26,39 For the GDAT, this method could not be used since this task was performed with fixed trial durations.
Visual analogue scale
Vividness scores were measured after each trial during both tasks by means of a 7-point VAS (1 =
Statistical Analyses
Variables showed normal distributions (Shapiro–Wilk test) and equivalent variance (Levene test). Data were analyzed using repeated-measures analyses of variance (ANOVAs) with α set at .05. For the eye movement data of the GDAT, within-subjects factors were modality (EXEC, MI, REST), cueing type (VIS, AUD, NO CUES), and intertarget distance (LARGE, SMALL). For the BBT, only condition (EXEC, MI VIS, MI AUD, MI NO CUES) was taken as the within-subjects factor. For both tasks, group (PD, CONTROLS) was added as the between-subject factor. BBT duration was analyzed by a 3 condition (EXEC, MI VIS, MI NO CUES) × 2 group ANOVA. The MI AUD condition was not included in this analysis, since its duration was metronome paced. Pearson product–moment correlation coefficients were calculated between the duration of physical execution and the different imagery conditions. MI vividness was analyzed by a 2 group × 3 condition (VIS, AUD, NO CUES) ANOVA for the BBT and by a 2 group × 3 cueing type (VIS, AUD, NO CUES) × 2 intertarget distance ANOVA for the GDAT. In case of significant interaction effects, we proceeded with analysis of the simple effects contributing to the interaction effect. 48 Post hoc Tukey honestly significant difference tests were done with α = .05.
Results
Eye Movements
Eye movement time
During the GDAT, 2-way interactions between modality and cueing type,
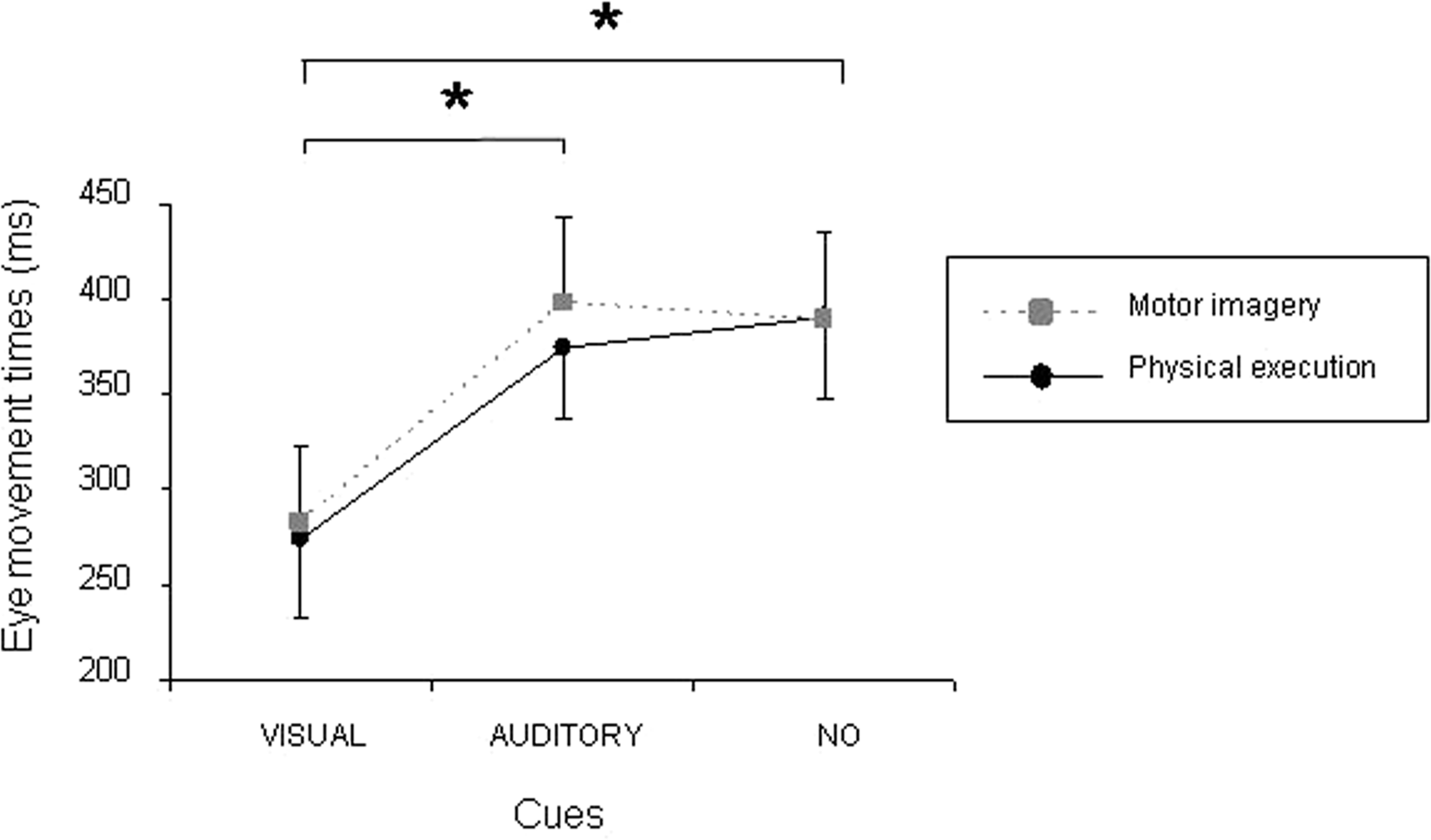
Eye movement times of all participants (mean ± SEM) during the goal-directed aiming task.
The eye movement times of the BBT confirmed those of the GDAT. The main effect for condition,
Eye movement number
For the GDAT, significant effects for cueing type,
Eye movement amplitude
The eye movement amplitudes were analyzed to examine whether they adapted to the task requirements during imagery in a similar way as during execution. For the GDAT, a significant interaction was found between modality and intertarget distance,
Mental Chronometry
For BBT duration, a main effect was found for group,
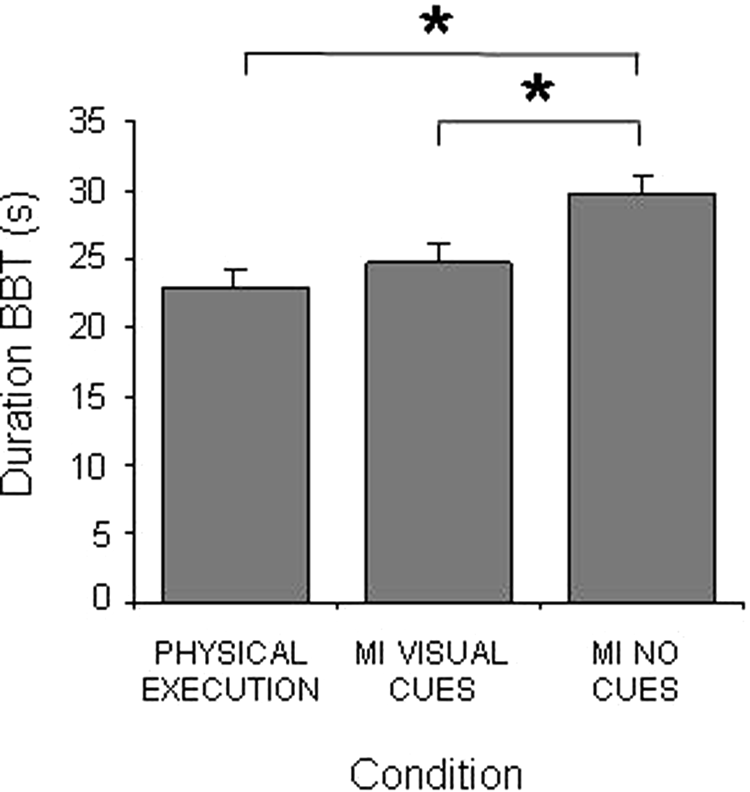
Duration of the Box-and-Block Test (BBT) of all participants (mean ± SEM) in the different experimental conditions.
MI Vividness
Both for the GDAT and the BBT, a main effect for cueing type was found,
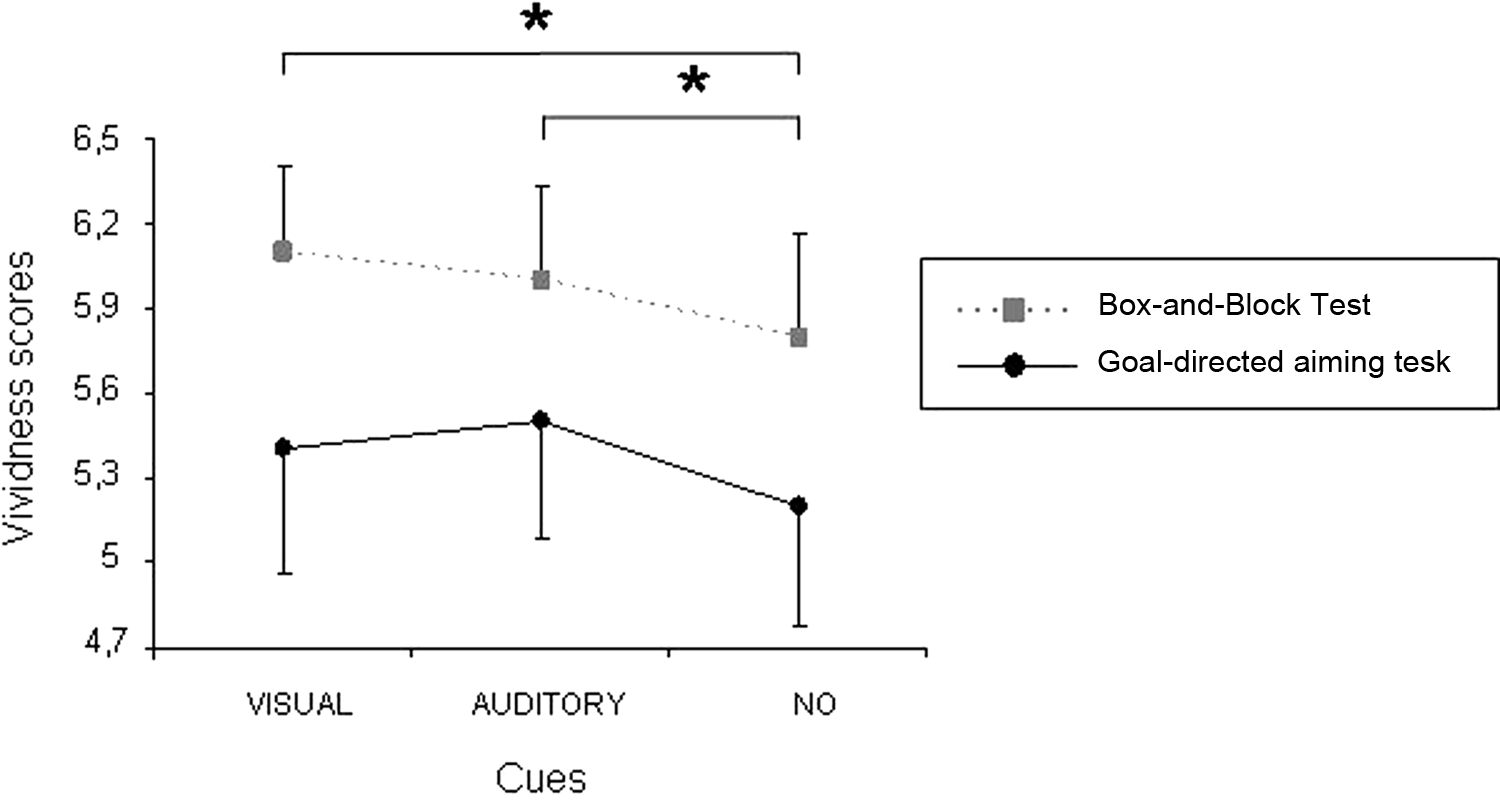
Motor imagery (MI) vividness of all participants (mean ± SEM) in both tasks during the different cueing types.
Discussion
We investigated whether external cues could enhance MI performance in patients with PD and, in particular, whether the cues would diminish patients’ bradykinesia during MI. We found that visual cues significantly increased the temporal isochrony between MI and execution, as well as the speed of both the imagined hand movements and the patients’ eye movements, which are considered as an objective indicator of MI. 43 The participants’ MI vividness was positively affected by visual and auditory cues.
The most widely accepted explanation for the positive effects of cueing in PD is that cues help patients compensate for self-driven, movement generation deficits. 20 External cues can overcome these problems, since cueing activates the lateral system, including the premotor and parietal cortex and the cerebellum to bypass the deficient medial system, including the basal ganglia.19,22 An alternative explanation is that adding visual information compensates for the patients’ deficits in the processing of proprioceptive information.7,49 The provision of visual cues allows patients to adopt a closed control mode, adjusting their movements continuously on the basis of the available visual information. 50
Although the effects of cueing are well known regarding physical practice, the present study shows for the first time that cueing can also be applied to optimize PD patients’ imagined movements. The best results were found when applying visual rather than auditory cues, perhaps because participants were instructed to specifically use visual MI. It is likely that this type of imagery is more sensitive to augmented visual information than to information influencing other senses. Second, auditory cues were mainly shown to be useful for physical execution of rhythmic tasks such as gait, 22 while less is known on their effect on upper limb movements. 51 Possibly, upper limb tasks are more influenced by augmented visual information offering a spatial reference frame. Furthermore, all conditions were initiated by 5 auditory paced movements to indicate the movement rhythm. Although these initial movements were not included in the data analysis, they may have had a sustained effect, diminishing the need for further auditory cueing. Finally, the auditory cues were given at a fixed rhythm, feasible for all patients and perhaps not at their preferred speed. In a study on gait training, Nieuwboer et al 18 reported that the modality (eg, visual, auditory) and parameters (eg, frequency) of the cues need to be adapted to the needs of the individual. Likewise, in MI practice, the way the cues are applied seems crucial. The importance of the cueing parameters was illustrated by Louis et al, 9 who showed that changing MI speed also changes the speed of actual motor performance. Concerning the choice between cueing modalities, some patients prefer to perform MI with the eyes closed. However, if the eyes are kept closed, the imagery quality can no longer be enhanced by visual cueing. Further research may clarify the type of cues and how these cues can be applied during MI practice to get the best effects.
Our findings are important for developing rehabilitation strategies based on MI. Mainly in the early phase of MI training, cues could be used to consciously help patients attend to problematic aspects of the imagined movement to develop a correct internal representation of the trained movement. Additional sensory information may optimize motor learning in PD.50,52 In addition, it has been shown that the effects of cues are retained in patients with PD even when the cues are withdrawn in later stages of training. 53 In healthy persons, 54 augmented visual information during training led to changes in both behavioral data and brain activity when the cues were withdrawn. It is still unclear whether this reflects a positive aftereffect of cueing or the development of cue dependency. If the augmented information becomes an actual part of the central representation of the movement, this may prohibit generalization to daily life situations in which cues are absent. 52 To prevent such cue dependency, one could gradually decrease the provision of cues over the course of training.
MI might be difficult for some patients, especially those with deficits in executive functioning.8,55 We expect that impairments in MI will mainly occur when the attention demands exceed available resources. 56 Cues might be used to provide patients with a framework of the to-be-imagined movement, as such reducing the attentional cost. Our eye movement recording method offers a way to control whether patients really keep their attention to the MI task. In previous studies in young healthy participants, we demonstrated that eye movements act as a mirror of the movements that were imagined.27,43 In this study, the same pattern was found in elderly controls and patients with PD. The participants’ eye movements closely adapted to the to-be-imagined movement, both temporally and spatially, indicating that our participants had well-preserved imagery ability. These task-related eye movements were found for both tasks, showing that they not only occur during a strictly controlled laboratory task such as the GDAT but also during a functionally relevant task such as the BBT. This task can easily be implemented in a clinical setting for the evaluation of MI ability or training of upper limb movements.
A limitation of this study is that it only investigated the effect of cueing within a single imagery session. Future studies should examine whether cueing effects are retained in later sessions, as well as whether cueing enhances the effects of long-term MI training. MI practice provides an opportunity to add additional training without needing a therapist or institutional context and without additional costs or safety risks. Besides, it offers the patients a tool to self-manage their rehabilitation, since they can use it as often as they want. Furthermore, MI can be used as an attentional strategy to optimally prepare in advance for difficult actions such as walking through a narrow door or crossing a busy street. Tamir et al 16 showed that combining MI and real practice may reduce bradykinesia. The present data indicate that shaping the patients’ environment during MI to offer them correct spatial reference frames of the imagined movements might even further enhance these positive effects. Future research should focus on the potential clinical benefits of (cued) MI training in this group, as well as on which patients might benefit most from this technique. The current study was limited to patients in the early and middle stage of the disease, which had no severe cognitive decline and were ‘on’ medication. An additional limitation of the current study is that it only involves tasks that were performed in a laboratory environment. Future research is needed to investigate to what extent our results generalize to tasks that are relevant to patients’ daily life.
Furthermore, the present study was limited to participants with good imagery ability only. Previous studies have shown wide individual differences in imagery ability, both in healthy persons and in neurological patients.1,8,57-59 These differences are not merely due to differences in motivation or concentration but relate to distinct neurological processing characteristics, 60 as revealed by the difference in patterns of cerebral activation during MI between skilled and unskilled imagers. 59 Since a relationship exists between imagery ability and the effectiveness of mental practice, 61 a thorough screening of each individual’s imagery ability is necessary before considering MI practice to augment motor rehabilitation.
Footnotes
Acknowledgements
We thank all participants. The technical assistance of Ir Marc Beirinckx and Ir Paul Meugens is greatly appreciated.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:
Elke Heremans is a research assistant and Wim Vandenberghe a senior clinical investigator at the Research Foundation Flanders (FWO). The authors also acknowledge the support of the Belgian Charcot Foundation to obtain eye movement recording equipment.
