Abstract
Background. People with postpolio syndrome (PPS) commonly experience severe fatigue that persists over time and negatively affects functioning and health-related quality of life (HRQoL). Objectives. To study the efficacy of exercise therapy (ET) and cognitive behavioral therapy (CBT) on reducing fatigue and improving activities and HRQoL in patients with PPS. Methods. We conducted a multicenter, single-blinded, randomized controlled trial. Over 4 months, severely fatigued patients with PPS received ET, CBT, or usual care (UC). The primary end point (fatigue) was assessed using the subscale fatigue severity of the Checklist Individual Strength (CIS20-F). Secondary end points included activities and HRQoL, which were assessed with the Sickness Impact Profile and the 36-Item Short-Form, respectively. End points were measured at baseline and at 4, 7, and 10 months. Results. A total of 68 patients were randomized. No differences were observed between the intervention groups and UC group for fatigue (mean differences in CIS20-F score = 1.47, 95%CI = −2.84 to 5.79, for ET versus UC; and 1.87, 95%CI = −2.24 to 5.98, for CBT versus UC), activities, or HRQoL. Conclusions. Our results demonstrate that neither ET nor CBT were superior to UC in reducing fatigue in severely fatigued PPS patients. Further research should investigate explanations for the lack of efficacy of these 2 currently advised approaches in clinical practice, which may provide clues to improving treatment aimed at reducing fatigue in PPS.
Keywords
Introduction
People with postpolio syndrome (PPS) commonly experience severe fatigue that persists over time1-3 and negatively affects functioning and health-related quality of life (HRQoL).4,5 This fatigue is typically described as tiredness or lack of energy that increases with physical activity and decreases with rest. 6 The etiology of fatigue in PPS is poorly understood, and it is presumed that fatigue is multifactorial. Biological, physical, psychological, and cognitive-behavioral factors are known to be associated with fatigue in PPS.1,7 Because of this multidimensional character of the fatigue experienced, a variety of interventions aimed at alleviating it have been studied. However, because of insufficient good-quality data and lack of randomized studies, no definite conclusions can be made on their effectiveness. 8
Exercise therapy (ET) and cognitive behavioral therapy (CBT) are interventions that have been found to successfully reduce fatigue in other disorders by targeting 2 different fatigue-related factors. The rationale for ET is based on the assumption that it can interrupt the vicious cycle of fatigue, leading to physical inactivity and, consequently, reduction of physical capacity. Reduced physical capacity results in an increased effort to perform daily activities,9,10 which in turn leads to more fatigue. ET aims to improve physical capacity. The rationale for CBT is based on the assumption that cognitive and behavioral factors play a significant role in perpetuating or worsening fatigue.11,12 By challenging the meaning of these dysfunctional cognitions and eliciting more realistic cognitions, emotions and behaviors might be changed, and consequently, fatigue might be reduced.
Evidence on the effectiveness of ET and CBT in PPS is limited. A systematic review on ET for neuromuscular diseases included 10 studies on patients with PPS, 5 of which demonstrated positive effects on muscular strength and aerobic capacity. 13 A more recent study showed improvement in fatigue and HRQoL in PPS; however, no comparison with a control group was made, and long-term effectiveness was not evaluated. 14 Evidence for CBT in PPS is currently limited to an uncontrolled study of cognitive behavioral strategies incorporated in a multidisciplinary rehabilitation program. 15 Although a reduction in fatigue was found, it is unclear whether this can be ascribed to the cognitive behavioral components of the intervention.
This study aimed at investigating the efficacy of ET and CBT in patients with PPS. We hypothesized that both approaches would lead to a sustained reduction of fatigue and improvement in activities and HRQoL compared with usual care (UC). The primary research questions for this study in patients with PPS were the following: (1) Does ET lead to a sustained reduction of fatigue and an improvement in activities and HRQoL compared with UC? and (2) Does CBT lead to a sustained reduction of fatigue and an improvement in activities and HRQoL compared with UC?
Methods
We performed a stratified multicenter, single-blinded randomized controlled trial (RCT) with equal allocation across treatment arms and follow-up over 6 months. Patients were randomized to 1 of 3 treatment arms: ET, CBT, or UC. Randomization was stratified by center. The randomization scheme was computer generated, and random blocks of sequences were created with variable block sizes of 3 and 6. An independent investigator performed the randomization. The investigator responsible for the inclusion and the 2 experimenters who performed the outcome assessments were blinded to the group allocation.
Standard Protocol Approvals, Registrations, and Patient Consents
Our study protocol 16 was approved by the Medical Ethics Committee of the Academic Medical Centre in Amsterdam, and all participating centers granted approval to participate. The RCT was registered at the Netherlands National Trial Register (NTR1371). Informed consent was obtained from all participants.
Participants
Patients were recruited from 7 hospitals and rehabilitation centers in the Netherlands. Medical files were screened for potentially eligible patients. Patients willing to give signed consent were evaluated by a physician to check the inclusion and exclusion criteria. Inclusion criteria were the following: diagnosis of PPS according to the criteria of the March of Dimes, which includes a gradual or sudden onset of progressive and persistent muscle weakness or abnormal muscle fatigability after a period of stable neurological function 17 ; severe perceived fatigue (subscale fatigue severity of the Checklist Individual Strength (CIS20-F) ≥ 35) 18 ; age between 18 and 75 years; life expectancy longer than 1 year, that is, absence of life-threatening comorbidity; walking ability at least indoors with or without a walking aid; and ability to cycle on a ergometer against a load of at least 25 W. Each patient included in the trial completed a Physical Activity Readiness Questionnaire 19 and was approved safe for exercise by the screening physician. Exclusion criteria are described elsewhere. 16
Interventions
Usual care: In all allocation groups, patients received UC at the discretion of their treating physician, which could include the use of assistive devices and/or orthoses, physical therapy, and medication. Patients were not restricted in their activities. Cointerventions were monitored throughout the study.
Exercise therapy: ET was designed specifically to enhance the patient’s physical capacity. The intervention lasted 4 months and consisted of (1) a home-based aerobic training program 3 times weekly and (2) a supervised group-training program once a week. Physiotherapists who were trained in the protocol supervised the therapy. The home-based interval training program included aerobic exercise on a cycle ergometer. Patients were supplied with a cycle ergometer and logbook with training instructions at their home. During training, the heart rate was continuously monitored. Training intensity was gradually increased from 60% to 70% heart rate reserve, 20 and the training duration was gradually increased from 28 to 38 minutes per session. The feasibility of the training schemes was checked weekly by one of the therapists by reading the heart rate monitors and checking the logbooks. The supervised group-training program consisted of individually tailored muscle strengthening and functional exercises in 1-hour group sessions. Only muscle groups with a MRC score 21 ≥3 were selected for the strengthening exercises. Functional exercises aimed to improve the interplay of cognitive, perceptual, and motor functions. 22 Compliance with ET was assessed by recording the number of home-based training sessions and group-training attendance, as monitored by the physiotherapists. All adverse events (such as severe muscle fatigue, joint pain, or other events considered to be related to ET) were recorded. Adverse events were followed until they abated or until a stable situation had been reached.
Cognitive behavioral therapy: CBT was directed at perpetuating factors of fatigue in slowly progressive neuromuscular disorders. 11 These involve dysfunctional cognitions with respect to the disease itself, pain, or fatigue 7 ; dysfunctional attention to pain and fatigue symptoms; deregulation of sleep7,23; deregulation of physical, social, and/or mental activities7,23; and low social support and negative social interactions. 24 For each factor, a standardized module was available as part of the intervention. To determine which modules were appropriate, each perpetuating factor was assessed with specific questionnaires, so as to customize the CBT to the individual patient. 16 The number of CBT sessions was dependent on the number of modules used and the time necessary to reach the goals of the conducted module. Sessions lasted 1 hour and were planned within a 4-month period. Certified cognitive behavioral therapists, who were highly trained in the protocol, treated the patients. Attendance of CBT was monitored by recording the number of treatment sessions as given by the therapists.
A more detailed description of both interventions is available elsewhere. 16
Outcomes
Outcomes were assessed at study entry (pretreatment), at 4 months (posttreatment), at 3 months’ follow-up (short term), and at 6 months’ follow-up (long term). The primary end point was fatigue, assessed with the 8-item subscale CIS20-F. 18 Secondary end points included self-perceived activity limitations (Sickness Impact Profile [SIP-68]; domains of mobility control, social behavior, mobility range 25 ) and HRQoL (Short-Form 36 [SF-36]; Physical Component Summary, Mental Component Summary 26 ).
Exploratory end points included pain (Visual Analogue Scale), total mood disturbance (Profile of Mood States), sleep disturbances (Nottingham Health Profile-sleep), illness cognitions (Illness Cognitions Questionnaire), coping (Coping Inventory for Stressful Situations, CISS-21), and general self-efficacy (Dutch version of the Self-Efficacy Scale, ALCOS-16).
In addition, we assessed cardiorespiratory fitness (submaximal heart rate during exercise [HRsubmax]), muscle strength (maximal isokinetic voluntary torque of quadriceps muscles [MVT]), functional capacity (Timed-Up-and-Go test and 2-Minute Walk test), and actual daily physical activity level (monitored on 7 consecutive days [StepWatch Activity Monitor; Orthocare Innovations, Oklahoma City, OK]). Descriptions and references of all outcomes are available elsewhere. 16
Statistical Analyses
The sample size for this RCT was based on the comparison treatment (ET or CBT) versus UC and 4 repeated measurements, using an estimated correlation coefficient of the repeated measurements of 0.79 (based on unpublished data from a reproducibility study in 37 PPS patients); a clinically relevant decline of 8 points on the CIS20-F (for both treatments) 27 ; and an estimated standard deviation in each treatment group of 9.3. The total sample size needed to detect an 8-point difference in CIS20-F at a 5% level of significance (2 tailed) with a power of 90% was 24 in each group. 28 We expected a maximum drop-out rate of 10%, based on a previous trial in this patient group. 29 Therefore, a total sample size of 81 patients was planned.
To check for selective loss to follow-up, differences between participants who completed the trial and those who were lost to follow-up were examined (2-tailed independent t tests, Mann-Whitney U tests, χ2 tests).
We assessed the primary, secondary, and exploratory end points with linear mixed models, with group and pretreatment score of the outcome as covariates (primary analyses). The models incorporated random intercepts for participants. To allow estimates at the individual time points, we added time and time-by-group interaction to the model (secondary analyses). Analyses were performed blinded for group allocation and based on the intention-to-treat (ITT) sample. No imputation of missing data was performed, under the assumption that data were missing at random. Additionally, per protocol analyses were performed including only patients who attended more than 47 of 63 possible treatment sessions for ET (75%) or completed the CBT intervention according to the protocol. All analyses were done with SPSS Statistics 21. An α level of .05 was used for all tests of significance.
Results
Participant Flow and Recruitment
The phases of the trial are depicted in Figure 1. In total, 490 patients were invited to participate. Of these 490 patients, 129 did not respond, 200 declined to participate, and 93 were excluded. The major reasons for exclusion were not being severely fatigued and having comorbidities. From June 2009 to September 2012, 68 patients were enrolled. One patient allocated to ET withdrew consent after the pretreatment assessment; hence, the ITT sample consisted of 67 patients (22 in UC, 22 in ET, and 23 in CBT). Sociodemographic and polio characteristics of these 67 patients are shown in Table 1. In all, 51 patients were included in the per-protocol analyses (22 in UC, 14 in ET, and 15 in CBT). There were no significant differences between participants who were not lost to follow-up (n = 60) and those who were (n = 7) on pretreatment CIS20-F-scores, treatment allocation, or any of the pretreatment sociodemographic or polio characteristics (P > .08).
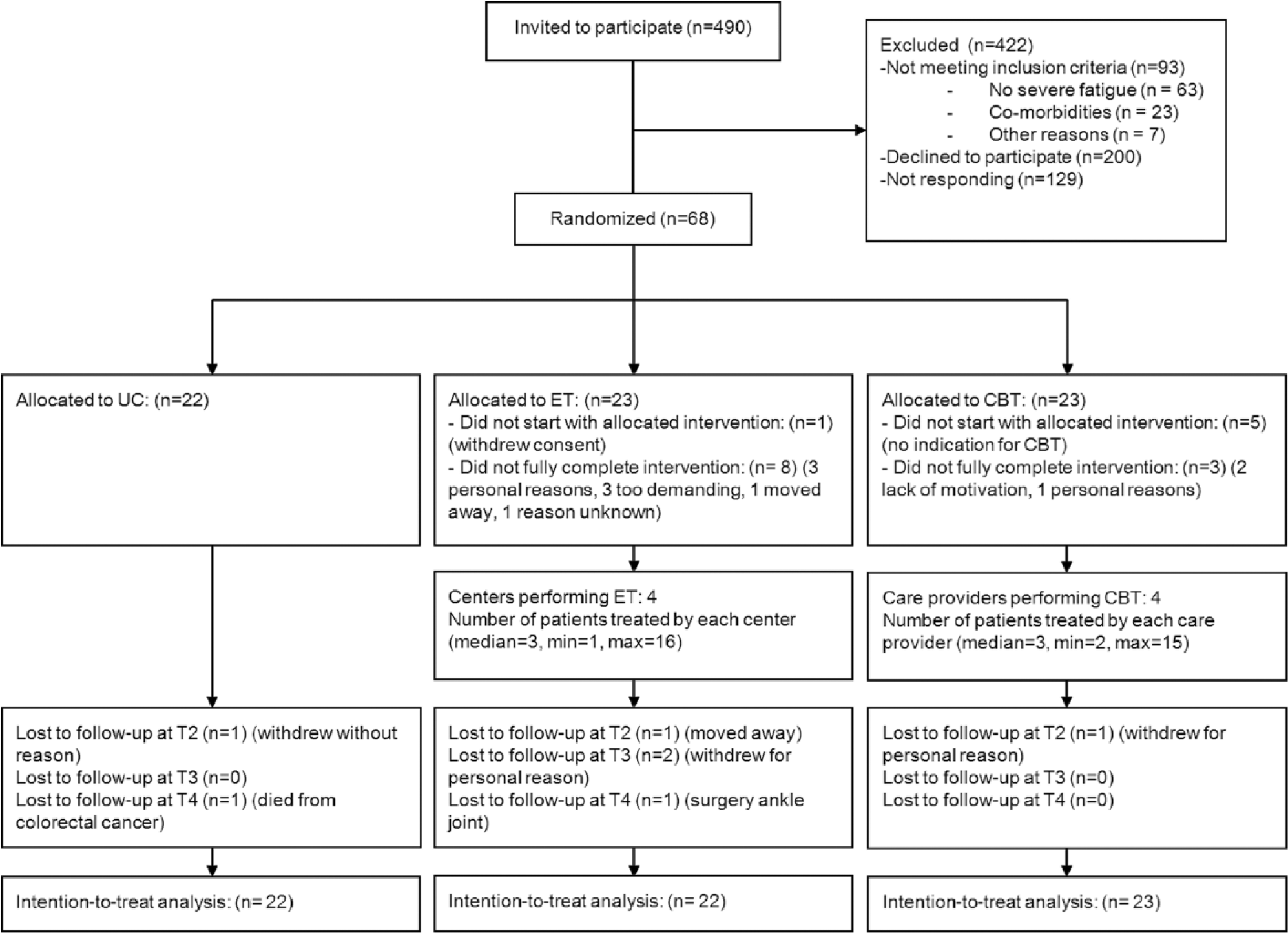
Flow diagram.
Sociodemographic and Polio Characteristics for the Intention-to-Treat Population.
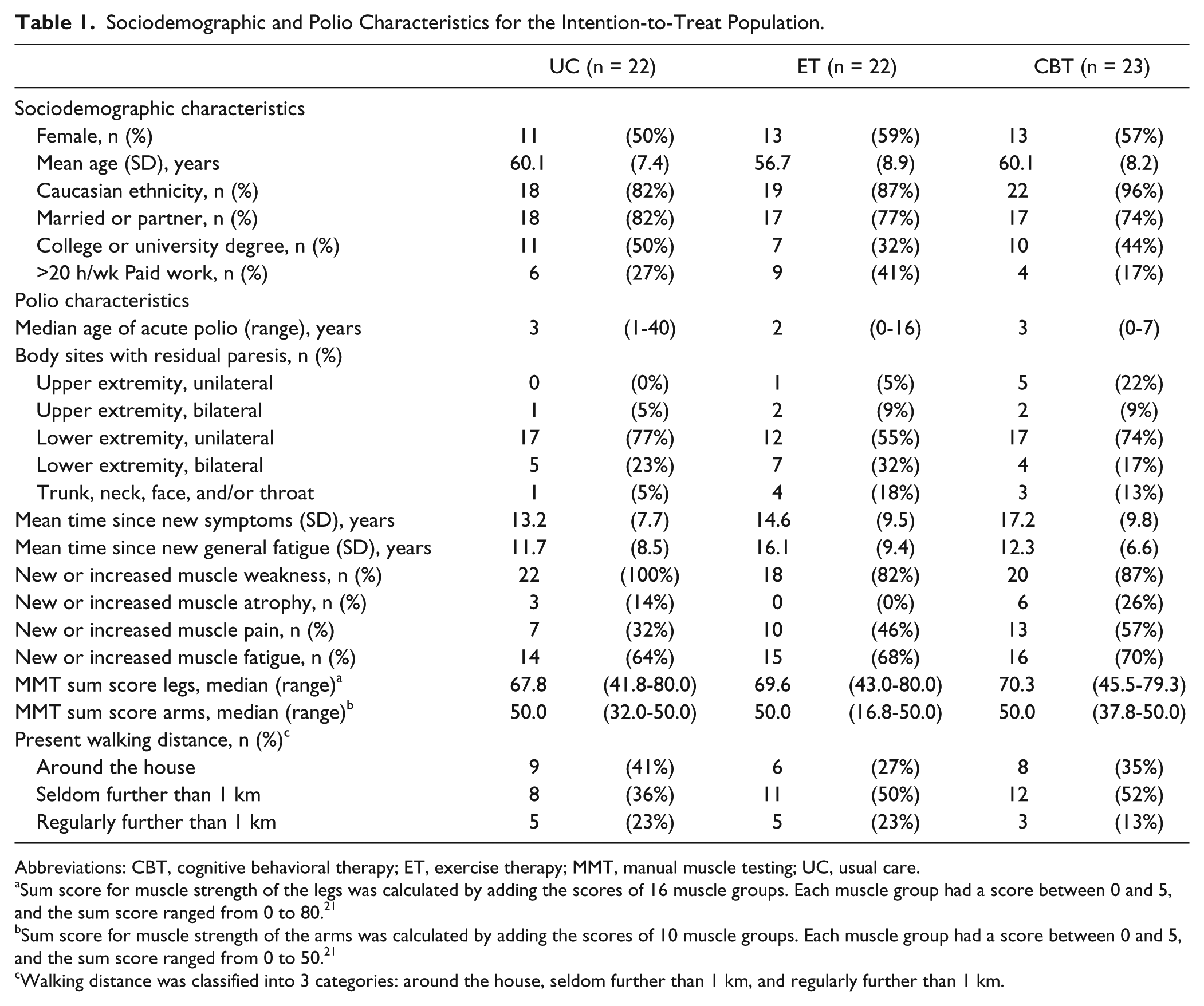
Abbreviations: CBT, cognitive behavioral therapy; ET, exercise therapy; MMT, manual muscle testing; UC, usual care.
Sum score for muscle strength of the legs was calculated by adding the scores of 16 muscle groups. Each muscle group had a score between 0 and 5, and the sum score ranged from 0 to 80. 21
Sum score for muscle strength of the arms was calculated by adding the scores of 10 muscle groups. Each muscle group had a score between 0 and 5, and the sum score ranged from 0 to 50. 21
Walking distance was classified into 3 categories: around the house, seldom further than 1 km, and regularly further than 1 km.
Implementation of Interventions
The median number of overall treatment sessions for the ITT sample was 57 (range = 8-63) for ET and 7 (range = 0-12) for CBT.
Cointerventions
The number of patients who were prescribed new cointerventions during the whole study period was 10 (50%) for the UC-group, 6 (38%) for the ET-group, and 10 (48%) for the CBT-group. The newly prescribed co-interventions are summarized in Table 2.
Newly Prescribed Cointerventions During the 10-Month Study Period. a
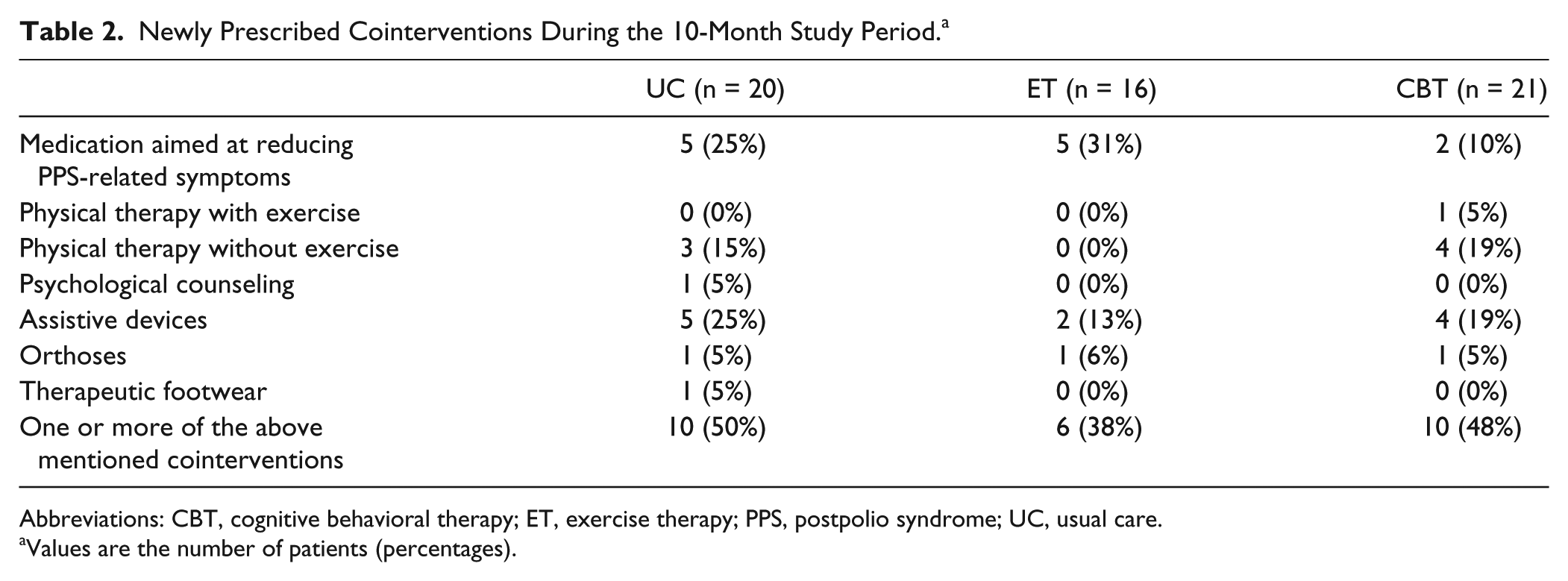
Abbreviations: CBT, cognitive behavioral therapy; ET, exercise therapy; PPS, postpolio syndrome; UC, usual care.
Values are the number of patients (percentages).
Outcomes
The results of the linear mixed modeling for the primary and secondary end points are shown in Table 3. Primary analyses showed no differences in fatigue (CIS20-F), self-perceived activity limitations (SIP-68), and HRQoL (SF-36) for ET or CBT compared with UC. Secondary analyses also showed no differences in primary and secondary end points between the interventions and UC on the individual time points, except for a significantly higher long-term CIS20-F score for ET compared with UC (5.37, 95% CI = 0.19 to 10.56). The efficacies of the interventions compared with UC on the exploratory end points are summarized in Supplemental Table 1. For ET, analyses showed no differences in cardiorespiratory fitness and muscle strength when compared with UC (overall mean difference in HRsubmax = 2.72 bpm, 95% CI = −2.41 to 7.85; and MVT strongest leg = 5.31 N m, 95% CI = −6.13 to 16.74, respectively). Also, for CBT, no differences were found for any of the exploratory end points compared with UC, except for a lower score on the emotional coping domain of the CISS-21 at long-term follow-up for CBT when compared with UC (−2.77, 95% CI = −5.16 to −0.38). Per-protocol analyses showed similar results as ITT analyses (mean differences in CIS20-F score = 2.29, 95% CI = −2.36 to 6.94 for ET vs UC; and 1.03, 95% CI = −3.65 to 5.71 for CBT vs UC).
Effects of ET and CBT Compared With UC for Primary and Secondary End Points.
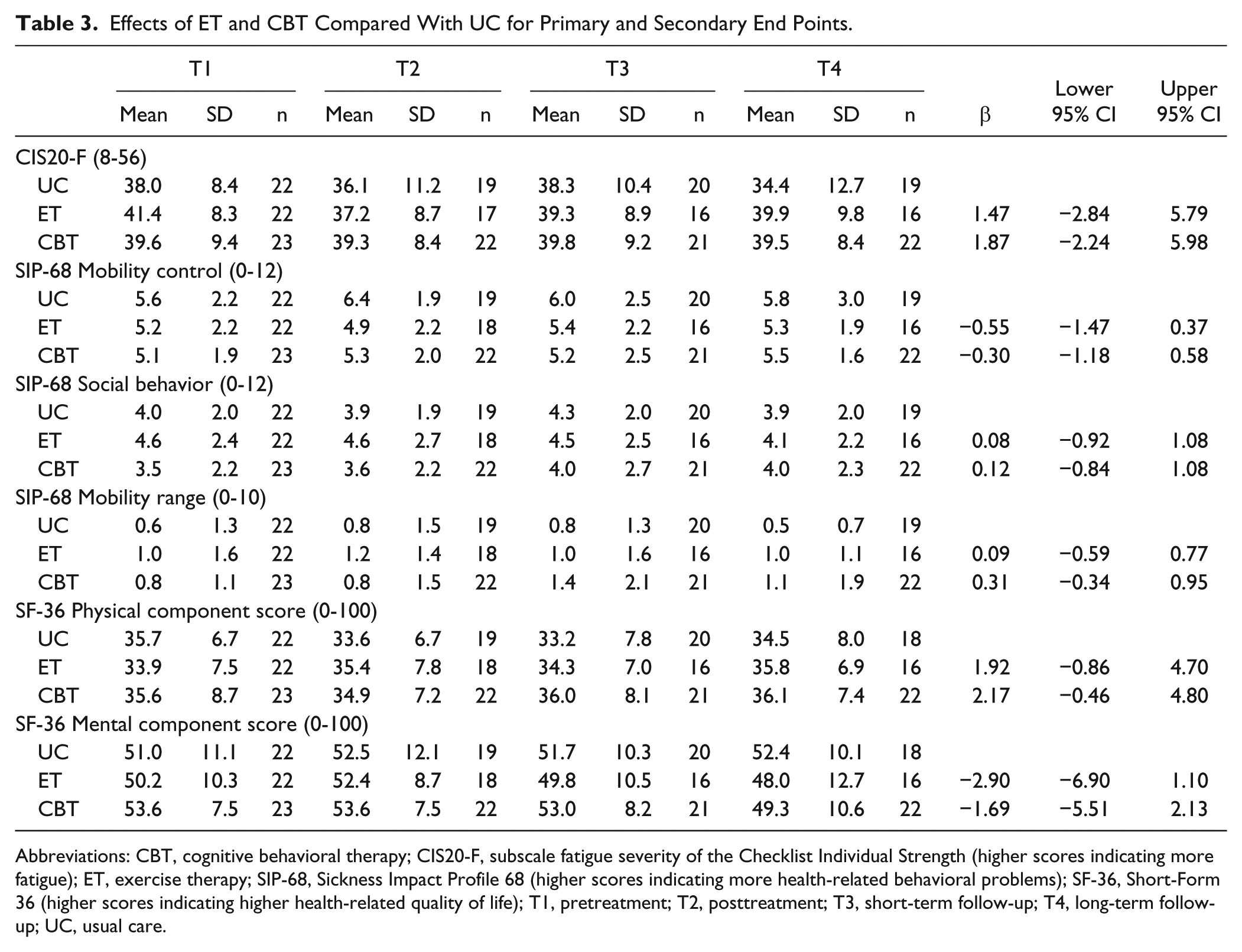
Abbreviations: CBT, cognitive behavioral therapy; CIS20-F, subscale fatigue severity of the Checklist Individual Strength (higher scores indicating more fatigue); ET, exercise therapy; SIP-68, Sickness Impact Profile 68 (higher scores indicating more health-related behavioral problems); SF-36, Short-Form 36 (higher scores indicating higher health-related quality of life); T1, pretreatment; T2, posttreatment; T3, short-term follow-up; T4, long-term follow-up; UC, usual care.
Adverse Events
Three adverse events were reported in the ET group (joint pain at the knee and elbow and trochanteric bursitis). This led to discontinuation of the intervention in 1 patient. All 3 adverse events were resolved.
Discussion
This RCT did not demonstrate a beneficial effect of ET or CBT on fatigue, activities, or HRQoL compared with UC in patients with PPS. These results are consistent with the absence of positive effects on any of the exploratory end points investigated in this study.
Although the required sample size was not reached, we are confident that this is not the reason for the negative study, considering the magnitude of the estimated effects of ET and CBT. Had the pattern of change noted in the included patients continued, the extra precision in estimates from another 14 patients would not have generated a statistically significant result for either intervention. Moreover, when a larger sample size would have led to significant differences, these differences would not be considered clinically relevant because the lower limits of the 95% CIs are, for both interventions, well below the clinically relevant decline of 8 points on the CIS20-F. 27 Furthermore, the estimated effects are well below the effects found in an identical trial (with a sample size of 77 patients) investigating these interventions in patients with facioscapulohumeral dystrophy (mean differences in CIS20-F score = −9.1, 95% CI = −12.4 to −5.8 for ET vs UC; and −13.3, 95% CI = −16.5 to −10.2 for CBT vs UC). 30
Additional newly prescribed cointerventions varied between the groups; however, it seems unlikely that this could explain the absence of positive effects of the interventions. Slightly more new cointerventions were prescribed in the UC group during the study period compared with the intervention groups, but these were almost never specifically targeted at alleviating fatigue. Moreover, we did not find a reduction in fatigue scores within the UC group.
Although, in this study, a number of patients had insufficient exposure to the interventions for reasons related and unrelated to the treatments, the per-protocol analyses and ITT analyses gave similar estimated effects for both interventions, instead of effects leading more in the direction of favoring ET or CBT. Therefore, it is unlikely that underexposure did influence the results of this study.
Regarding ET, only 3 adverse events were reported, and these were all resolved, indicating that this intervention was safe. The absence of effects on fatigue, activities, and HRQoL in the ET group compared with UC is in line with the results of 3 previous studies in PPS that reported no effects of ET on fatigue and activities31-33; also, they are consistent with the absence of any physiological changes in cardiorespiratory fitness and muscle strength. To our knowledge, only 1 study 14 showed positive effects of a hospital-based aerobic training program on cardiorespiratory fitness (VO2peak), fatigue, and HRQoL. One possible explanation for the dissimilarity in results may be related to the fact that severe fatigue was not a prerequisite for inclusion in that study. It may be hypothesized that increasing cardiorespiratory fitness and muscle strength in severely fatigued PPS patients is not possible because they already have adapted muscle function optimally as a result of performing daily life activities with their reduced muscle mass. This hypothesis is supported by the findings of a recent study showing that in a sample of fatigued PPS patients, there was no deconditioning of the knee extensor muscles as compared with healthy individuals. 34
The lack of efficacy of CBT in our study is in contrast with the positive results found in studies evaluating its effectiveness in other patient populations, such as those with postcancer fatigue, chronic fatigue syndrome, multiple sclerosis, and facioscapulohumeral dystrophy.27,30,35,36 People affected with PPS have spent a lifetime managing the challenges of living with functional limitations and have experienced long durations of fatigue. This may have led to a relatively high degree of acceptance of fatigue symptoms. This assumption is supported by the fact that, although all patients included in the study did indeed experience severe fatigue, a number of patients did not prioritize fatigue as a treatment goal, and therefore, the CBT intervention was not initiated (n = 5). Furthermore, it may be hypothesized that patients with PPS have developed different patterns of dysfunctional cognitions and behavior related to fatigue compared with patient populations with a symptom onset later on in their lives. These differences might, at least in part, explain the lack of efficacy of CBT in PPS.
Strengths and Limitations
This is the first study evaluating the effect of CBT on reducing fatigue in PPS and the effect of ET in a randomized controlled design with patients receiving UC as the control group. A principal strength of this study is that we used standardized treatment protocols, of which a detailed description is available. 16 Furthermore, we evaluated patients over a long-term period and utilized a broad arsenal of outcome measures from different domains of the International Classification of Functioning, Disability, and Health, including patient-reported outcomes.
Generalizability of our study results to the PPS population at large may be considered a limitation because only a small proportion of the total number of patients assessed for eligibility in this study (almost 500) was included. Nevertheless, patients were recruited from different centers throughout the Netherlands, and the sociodemographic and disease characteristics in our sample are comparable to that in previous cohort studies in polio survivors in the Netherlands 37 and other countries.7,38
Conclusion
Our results demonstrate that neither ET nor CBT were superior to UC in reducing fatigue or improving activities and HRQoL in severely fatigued PPS patients. Considering the negative results of these 2 substantially different interventions, which have been proved to be effective in other patient populations,27,30,35,36,39,40 and the wide range of other interventions that were unsuccessful, 8 it seems that fatigue in severely fatigued PPS patients is quite resistant to therapy. Further research should investigate explanations for the lack of efficacy of these 2 currently advised approaches in clinical practice, 41 which may provide clues to improve treatment aimed at reducing fatigue in PPS.
Footnotes
Acknowledgements
The authors thank the patients; the referring rehabilitation medicine consultants of the participating hospitals and rehabilitation centers (Academic Medical Centre Amsterdam; University Medical Centre Nijmegen; Roessingh Rehabilitation Centre, Enschede; University Medical Centre Utrecht; University Medical Centre Rotterdam (Erasmus MC); Rehabilitation Centre Merem de Trappenberg, Huizen; Rehabilitation Centre Leijpark, Libra Zorggroep, Tilburg); and the therapists of the ET and CBT interventions.
Authors’ Note
FSK was responsible for the conduct of the study, design of the statistical analytic plan, statistical analysis, interpretation of the data, drafting, and revision of the manuscript. ELV was a coinvestigator of the study and contributed to the conduct of the study, interpretation of the data, and revision of the manuscript. AB was a senior investigator and statistician of the study, oversaw all aspects of the conduct of the study, designed the statistical analytic plan, conducted statistical analysis, oversaw analyses, interpreted data, and contributed to the revision of the manuscript. GB was an advisory member of the study group and contributed to the conceptualization of the study, interpretation of the data, and revision of the manuscript. MdV was an advisory member of the study group and contributed to the conceptualization of the study, interpretation of the data, and revision of the manuscript. MAB was a coinvestigator of the study and oversaw all aspects of the conduct of the study, interpreted data, and contributed to the revision of the manuscript. FN was the principal investigator of the study, obtained funding, and contributed to the conceptualization and conduct of the study, interpretation of the data, and revision of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funded by Prinses Beatrix SpierFonds (PBF; The Dutch Public Fund for Neuromuscular Disorders), ZonMw (the Netherlands Organisation for Health Research and Development, ID: ZonMw 89000003), het Revalidatiefonds/Revalidatie Nederland/de Nederlandse Vereniging van Revalidatieartsen (the Netherlands Society of Physical and Rehabilitation Medicine).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
